Changes in the Expression of AQP4 and AQP9 in the Hippocampus Following Eclampsia-Like Seizure
Abstract
:1. Introduction
2. Results and Discussion
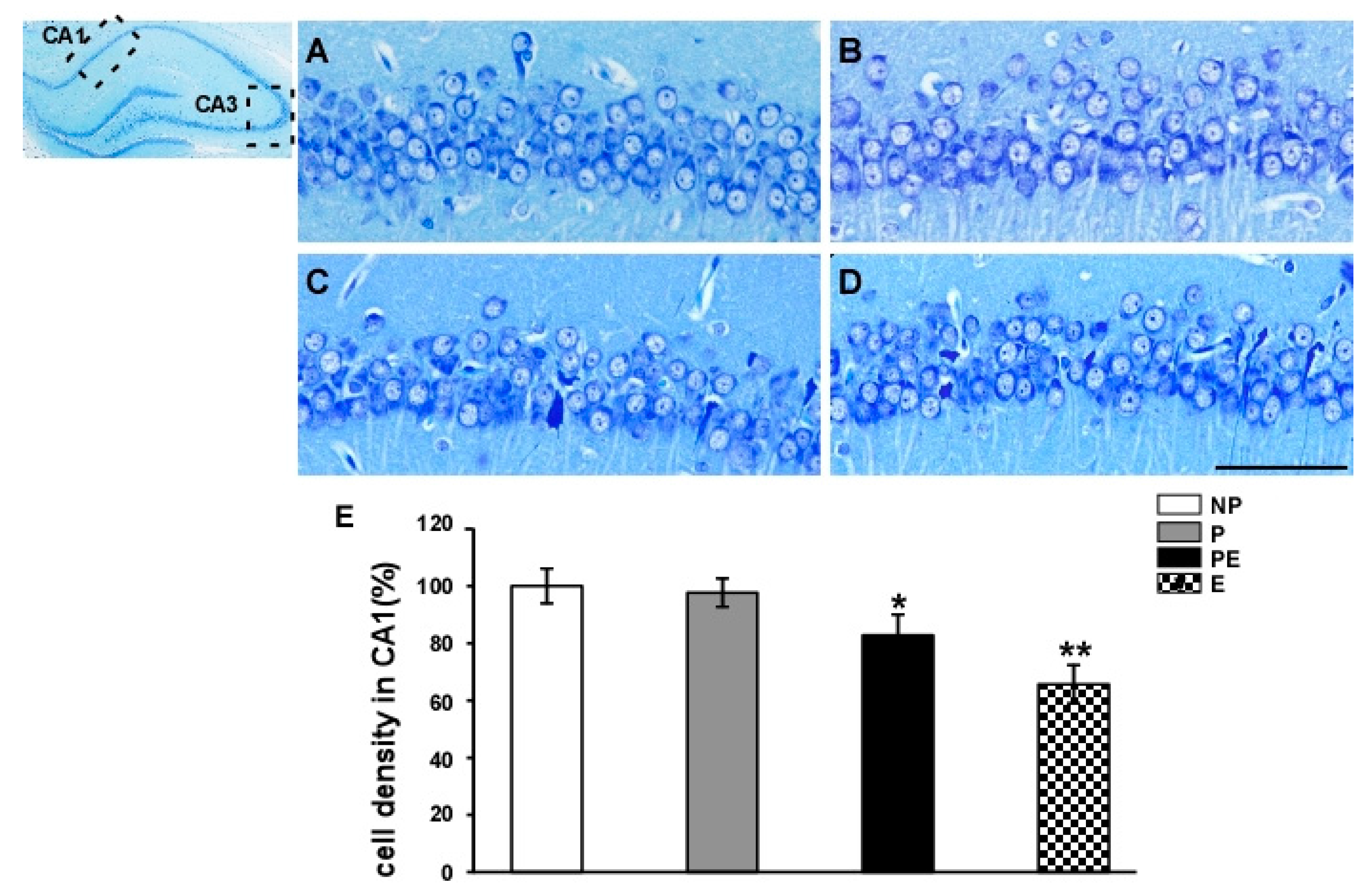
2.1. Neuronal Cell Loss in the Hippocampal CA1 Area before and after Eclampsia-Like Seizures
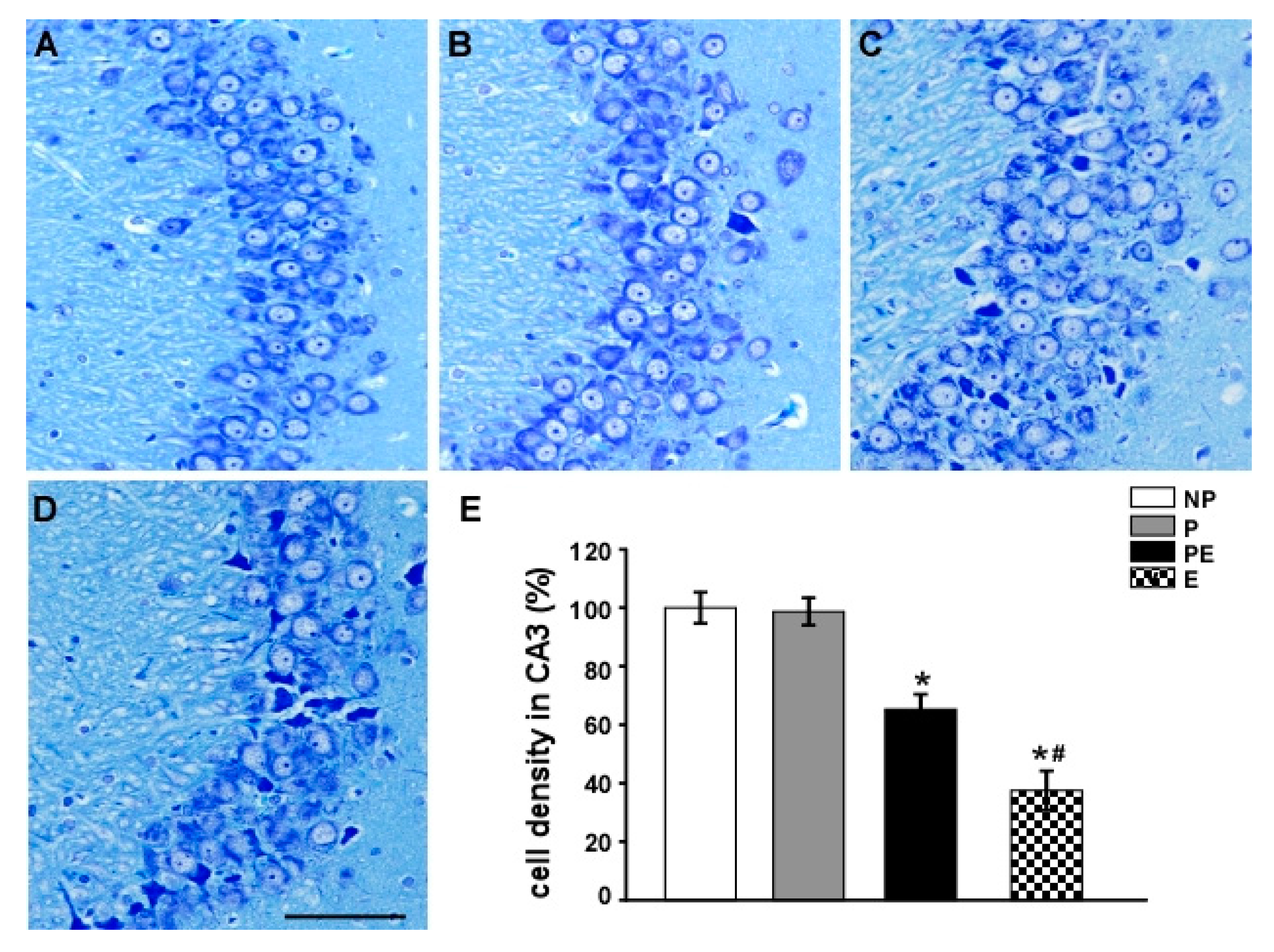
2.2. Neuronal Cell Loss in the Hippocampal CA3 Area before and after Eclampsia-Like Seizures
2.3. Changes in AQP4 and AQP9 mRNA Levels in the Hippocampus after Eclampsia
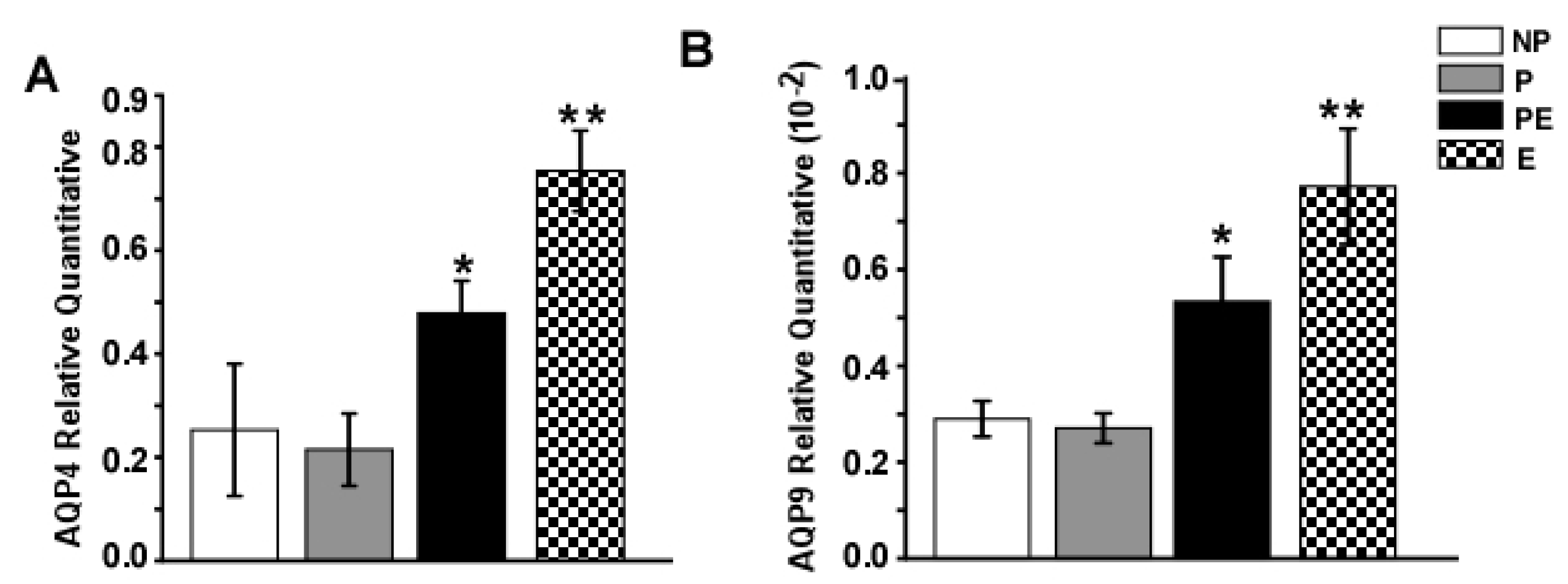
2.4. Changes in Protein Levels of AQP4 and AQP9 in the Hippocampus after Eclampsia
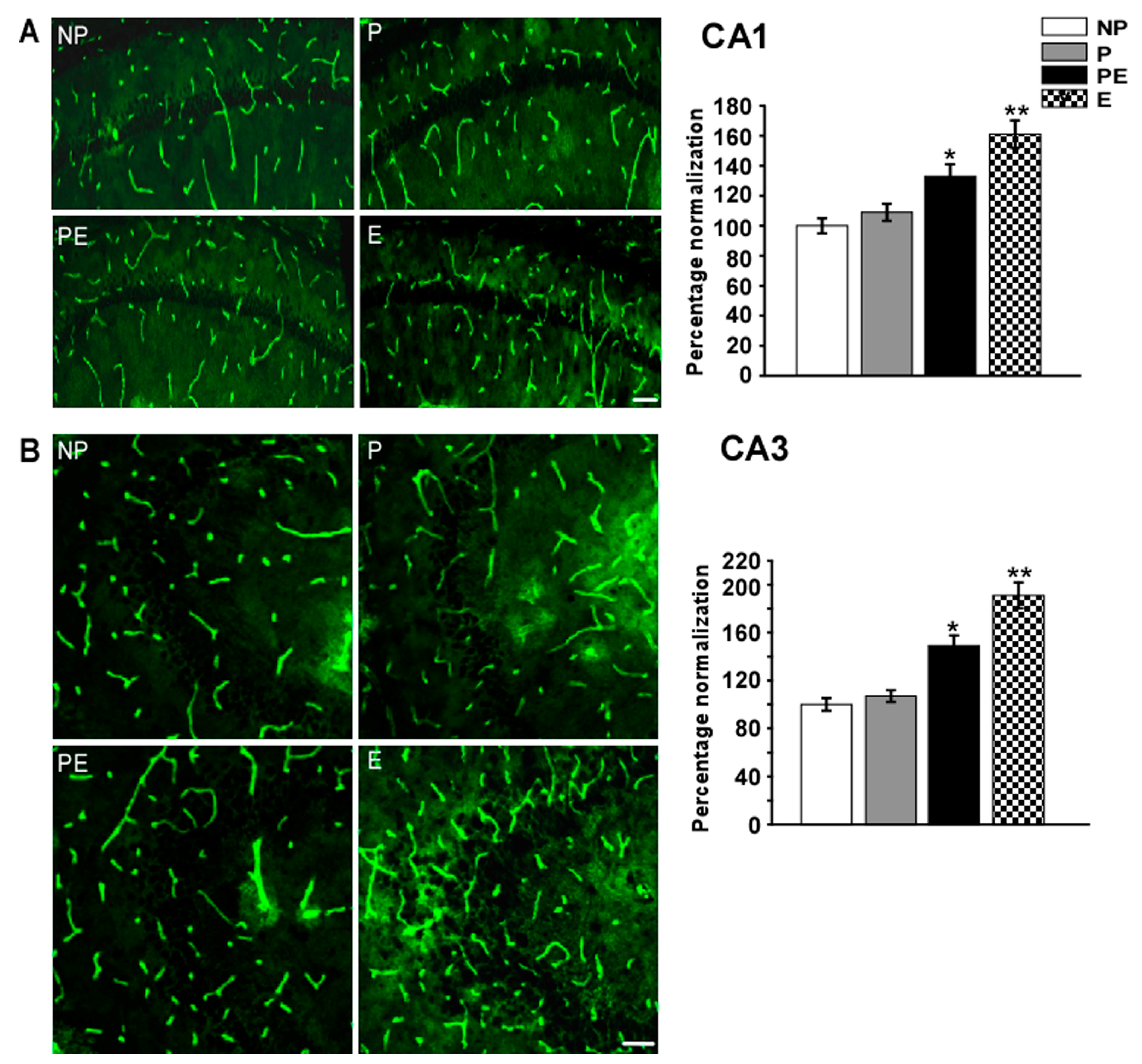
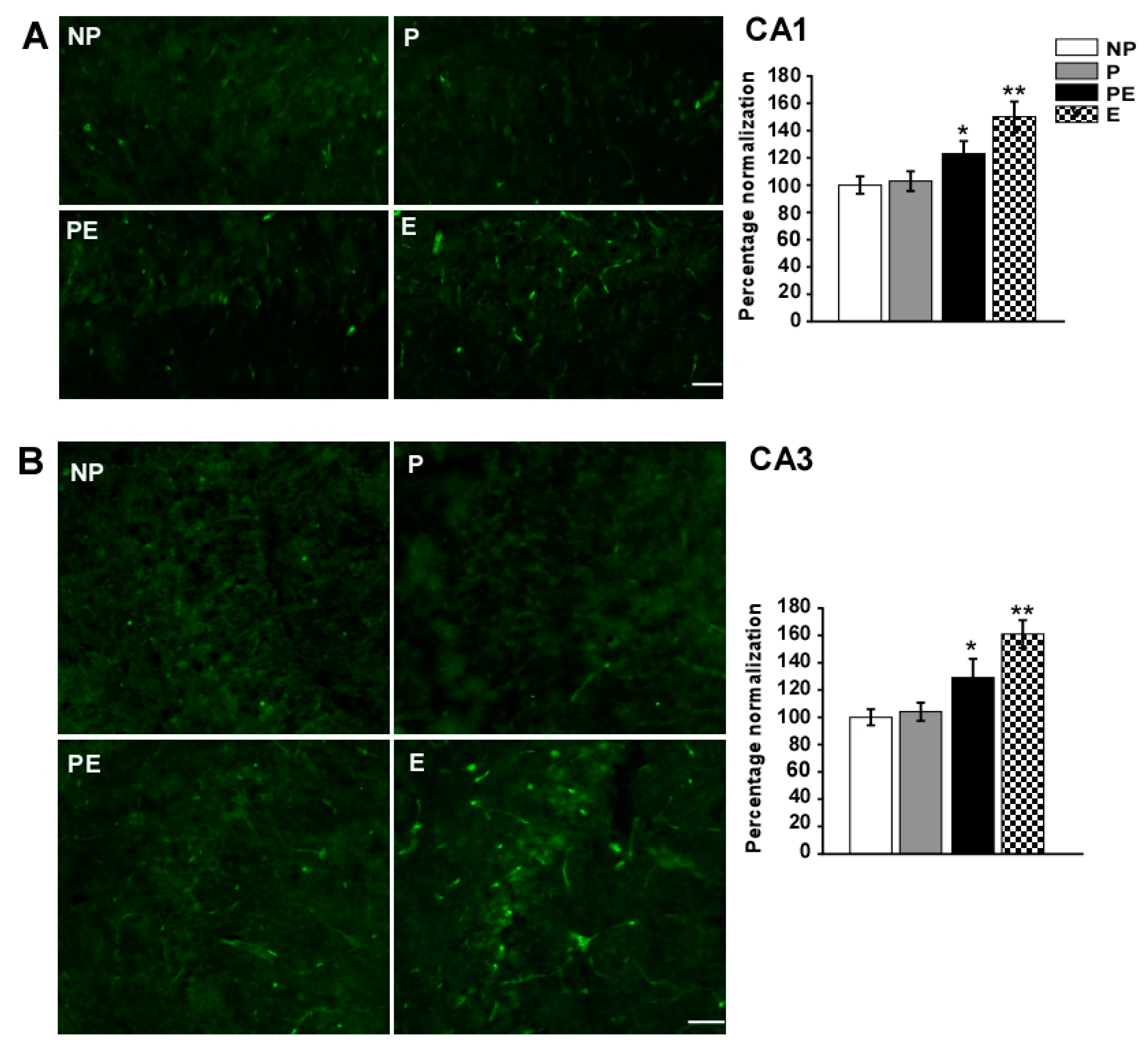
2.5. Changes in Immunoreactivities of AQP4 in the CA1 and CA3 Area after Eclampsia
2.6. Changes in Immunoreactivities of AQP9 in the CA1 and CA3 Area after Eclampsia
2.7. Discussion
3. Materials and Methods
3.1. Animals
3.2. Eclampsia Model
3.3. Nissl and Immunofluorescence Staining
3.4. Quantitative Reverse Transcription-PCR
3.5. Western Blotting
3.6. Statistics
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sibai, B.M. Diagnosis, prevention, and management of eclampsia. Obstet. Gynecol. 2005, 105, 402–410. [Google Scholar] [CrossRef] [PubMed]
- Abalos, E.; Cuesta, C.; Grosso, A.L.; Chou, D.; Say, L. Global and regional estimates of preeclampsia and eclampsia: A systematic review. Eur. J. Obstet. Gynecol. Reprod. Biol. 2013, 170, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Ghulmiyyah, L.; Sibai, B. Maternal mortality from preeclampsia/eclampsia. Semin. Perinatol. 2012, 36, 56–59. [Google Scholar] [CrossRef] [PubMed]
- Zeeman, G.G.; Fleckenstein, J.L.; Twickler, D.M.; Cunningham, F.G. Cerebral infarction in eclampsia. Am. J. Obstet. Gynecol. 2004, 190, 714–720. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Han, X.; Yang, J.; Bao, J.; Di, X.; Zhang, G.; Liu, H. Magnesium sulfate provides neuroprotection in eclampsia-like seizure model by ameliorating neuroinflammation and brain edema. Mol. Neurobiol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Badaut, J.; Lasbennes, F.; Magistretti, P.J.; Regli, L. Aquaporins in brain: Distribution, physiology, and pathophysiology. J. Cereb. Blood Flow Metab. 2002, 22, 367–378. [Google Scholar] [CrossRef] [PubMed]
- Badaut, J.; Regli, L. Distribution and possible roles of aquaporin 9 in the brain. Neuroscience 2004, 129, 971–981. [Google Scholar] [CrossRef] [PubMed]
- Filippidis, A.S.; Carozza, R.B.; Rekate, H.L. Aquaporins in Brain Edema and Neuropathological Conditions. Int. J. Mol. Sci. 2016, 18, 55. [Google Scholar] [CrossRef] [PubMed]
- Laird, M.D.; Sukumari-Ramesh, S.; Swift, A.E.; Meiler, S.E.; Vender, J.R.; Dhandapani, K.M. Curcumin attenuates cerebral edema following traumatic brain injury in mice: A possible role for aquaporin-4? J. Neurochem. 2010, 113, 637–648. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Yang, M.; Qiu, G.P.; Zhuo, F.; Yu, W.H.; Sun, S.Q.; Xiu, Y. Aquaporin 9 in rat brain after severe traumatic brain injury. Arq. Neuro-Psiquiatr. 2012, 70, 214–220. [Google Scholar] [CrossRef]
- Hubbard, J.A.; Szu, J.I.; Yonan, J.M.; Binder, D.K. Regulation of astrocyte glutamate transporter-1 (GLT1) and aquaporin-4 (AQP4) expression in a model of epilepsy. Exp. Neurol. 2016, 283, 85–96. [Google Scholar] [CrossRef] [PubMed]
- Badaut, J.; Hirt, L.; Granziera, C.; Bogousslavsky, J.; Magistretti, P.J.; Regli, L. Astrocyte-specific expression of aquaporin-9 in mouse brain is increased after transient focal cerebral ischemia. J. Cereb. Blood Flow Metab. 2001, 21, 477–482. [Google Scholar] [CrossRef] [PubMed]
- Binder, D.K.; Yao, X.; Zador, Z.; Sick, T.J.; Verkman, A.S.; Manley, G.T. Increased seizure duration and slowed potassium kinetics in mice lacking aquaporin-4 water channels. Glia 2006, 53, 631–636. [Google Scholar] [CrossRef] [PubMed]
- Schwartzkroin, P.A.; Baraban, S.C.; Hochman, D.W. Osmolarity, ionic flux, and changes in brain excitability. Epilepsy Res. 1998, 32, 275–285. [Google Scholar] [CrossRef]
- Dudek, F.E.; Obenaus, A.; Tasker, J.G. Osmolality-induced changes in extracellular volume alter epileptiform bursts independent of chemical synapses in the rat: Importance of non-synaptic mechanisms in hippocampal epileptogenesis. Neurosci. Lett. 1990, 120, 267–270. [Google Scholar] [CrossRef]
- Sendrowski, K.; Sobaniec, W. Hippocampus, hippocampal sclerosis and epilepsy. Pharmacol. Rep. 2013, 65, 555–565. [Google Scholar] [CrossRef]
- Lin, T.Y.; Lu, C.W.; Wang, S.J.; Huang, S.K. Protective effect of hispidulin on kainic acid-induced seizures and neurotoxicity in rats. Eur. J. Pharmacol. 2015, 755, 6–15. [Google Scholar] [CrossRef] [PubMed]
- Pohle, W.; Becker, A.; Grecksch, G.; Juhre, A.; Willenberg, A. Piracetam prevents pentylenetetrazol kindling-induced neuronal loss and learning deficits. Seizure 1997, 6, 467–474. [Google Scholar] [CrossRef]
- Tchekalarova, J.; Atanasova, D.; Nenchovska, Z.; Atanasova, M.; Kortenska, L.; Gesheva, R.; Lazarov, N. Agomelatine protects against neuronal damage without preventing epileptogenesis in the kainate model of temporal lobe epilepsy. Neurobiol. Dis. 2017, 104, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.C.; Hammer, E.S.; Sakkaki, S.; Tremble, S.M.; Holmes, G.L.; Cipolla, M.J. Inhibition of blood-brain barrier efflux transporters promotes seizure in pregnant rats: Role of circulating factors. Brain Behav. Immun. 2017. [Google Scholar] [CrossRef] [PubMed]
- Faas, M.M.; Schuiling, G.A.; Baller, J.F.; Visscher, C.A.; Bakker, W.W. A new animal model for human preeclampsia: Ultra-low-dose endotoxin infusion in pregnant rats. Am. J. Obstet. Gynecol. 1994, 171, 158–164. [Google Scholar] [CrossRef]
- Huang, Q.; Liu, L.; Hu, B.; Di, X.; Brennecke, S.P.; Liu, H. Decreased seizure threshold in an eclampsia-like model induced in pregnant rats with lipopolysaccharide and pentylenetetrazol treatments. PLoS ONE 2014, 9, e89333. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Han, X.; Bao, J.; Liu, Y.; Ye, A.; Thakur, M.; Liu, H. Nicotine increases eclampsia-like seizure threshold and attenuates microglial activity in rat hippocampus through the alpha7 nicotinic acetylcholine receptor. Brain Res. 2016, 1642, 487–496. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.S.; Eid, T.; Mane, S.; Kim, J.H.; Spencer, D.D.; Ottersen, O.P.; de Lanerolle, N.C. Aquaporin-4 is increased in the sclerotic hippocampus in human temporal lobe epilepsy. Acta Neuropathol. 2004, 108, 493–502. [Google Scholar] [CrossRef] [PubMed]
- Eid, T.; Lee, T.S.; Thomas, M.J.; Amiry-Moghaddam, M.; Bjornsen, L.P.; Spencer, D.D.; Agre, P.; Ottersen, O.P.; de Lanerolle, N.C. Loss of perivascular aquaporin 4 may underlie deficient water and K+ homeostasis in the human epileptogenic hippocampus. Proc. Natl. Acad. Sci. USA 2005, 102, 1193–1198. [Google Scholar] [CrossRef] [PubMed]
- Quick, A.M.; Cipolla, M.J. Pregnancy-induced up-regulation of aquaporin-4 protein in brain and its role in eclampsia. FASEB J. 2005, 19, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.E.; Ryu, H.J.; Yeo, S.I.; Seo, C.H.; Lee, B.C.; Choi, I.G.; Kim, D.S.; Kang, T.C. Differential expressions of aquaporin subtypes in astroglia in the hippocampus of chronic epileptic rats. Neuroscience 2009, 163, 781–789. [Google Scholar] [CrossRef] [PubMed]
- Badaut, J.; Ashwal, S.; Obenaus, A. Aquaporins in cerebrovascular disease: A target for treatment of brain edema? Cerebrovasc. Dis. 2011, 31, 521–531. [Google Scholar] [CrossRef] [PubMed]
- Badaut, J.; Petit, J.M.; Brunet, J.F.; Magistretti, P.J.; Charriaut-Marlangue, C.; Regli, L. Distribution of Aquaporin 9 in the adult rat brain: Preferential expression in catecholaminergic neurons and in glial cells. Neuroscience 2004, 128, 27–38. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro Mde, C.; Hirt, L.; Bogousslavsky, J.; Regli, L.; Badaut, J. Time course of aquaporin expression after transient focal cerebral ischemia in mice. J. Neurosci. Res. 2006, 83, 1231–1240. [Google Scholar] [CrossRef] [PubMed]
- Haram, K.; Bjorge, L.; Guttu, K. Pathophysiology and clinical manifestations in pre-eclampsia. Tidsskr. Nor. Laegeforening Tidsskr. Prakt. Med. Ny Raekke 2000, 120, 1426–1431. [Google Scholar]
- Hecht, J.L.; Ordi, J.; Carrilho, C.; Ismail, M.R.; Zsengeller, Z.K.; Karumanchi, S.A.; Rosen, S. The pathology of eclampsia: An autopsy series. Hypertens. Pregnancy 2017, 36, 259–268. [Google Scholar] [CrossRef] [PubMed]
- Pereira de Vasconcelos, A.; Ferrandon, A.; Nehlig, A. Local cerebral blood flow during lithium-pilocarpine seizures in the developing and adult rat: Role of coupling between blood flow and metabolism in the genesis of neuronal damage. J. Cereb. Blood Flow Metab. 2002, 22, 196–205. [Google Scholar] [CrossRef] [PubMed]
- Chesler, M.; Kaila, K. Modulation of pH by neuronal activity. Trends Neurosci. 1992, 15, 396–402. [Google Scholar] [CrossRef]
- Xiong, Z.Q.; Stringer, J.L. Extracellular pH responses in CA1 and the dentate gyrus during electrical stimulation, seizure discharges, and spreading depression. J. Neurophysiol. 2000, 83, 3519–3524. [Google Scholar] [CrossRef] [PubMed]
- Walsh, S.K.; English, F.A.; Johns, E.J.; Kenny, L.C. Plasma-mediated vascular dysfunction in the reduced uterine perfusion pressure model of preeclampsia: A microvascular characterization. Hypertension 2009, 54, 345–351. [Google Scholar] [CrossRef] [PubMed]
- LaMarca, B.B.; Bennett, W.A.; Alexander, B.T.; Cockrell, K.; Granger, J.P. Hypertension produced by reductions in uterine perfusion in the pregnant rat: Role of tumor necrosis factor-alpha. Hypertension 2005, 46, 1022–1025. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, P.; Weaver, L.E.; Doersch, K.M.; Kopriva, S.E.; Chiasson, V.L.; Allen, S.J.; Narayanan, A.M.; Young, K.J.; Jones, K.A.; Kuehl, T.J.; et al. Placental Toll-like receptor 3 and Toll-like receptor 7/8 activation contributes to preeclampsia in humans and mice. PLoS ONE 2012, 7, e41884. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Han, X.; Huang, Q.; Zhu, X.; Yang, J.; Liu, H. Increased neuronal seizure activity correlates with excessive systemic inflammation in a rat model of severe preeclampsia. Hypertens. Res. 2016, 39, 701–708. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Zhang, H.; Varrin-Doyer, M.; Zamvil, S.S.; Verkman, A.S. Proinflammatory role of aquaporin-4 in autoimmune neuroinflammation. FASEB J. 2011, 25, 1556–1566. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Qi, G.L.; Wang, J.; Chen, L.; Deng, Z.; Zhao, Y.S.; Lei, S.S.; Zhu, X.Q. Aquaporin 4 inhibition decreased synthesis of cytokines by acetazolamide in the hippocampus of rats with pentrazol-induced chronic epilepsy. Genet. Mol. Res. 2016, 15. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, X.; Huang, Q.; Liu, L.; Sha, X.; Hu, B.; Liu, H. Changes in the Expression of AQP4 and AQP9 in the Hippocampus Following Eclampsia-Like Seizure. Int. J. Mol. Sci. 2018, 19, 300. https://doi.org/10.3390/ijms19010300
Han X, Huang Q, Liu L, Sha X, Hu B, Liu H. Changes in the Expression of AQP4 and AQP9 in the Hippocampus Following Eclampsia-Like Seizure. International Journal of Molecular Sciences. 2018; 19(1):300. https://doi.org/10.3390/ijms19010300
Chicago/Turabian StyleHan, Xinjia, Qian Huang, Lei Liu, Xiaoyan Sha, Bihui Hu, and Huishu Liu. 2018. "Changes in the Expression of AQP4 and AQP9 in the Hippocampus Following Eclampsia-Like Seizure" International Journal of Molecular Sciences 19, no. 1: 300. https://doi.org/10.3390/ijms19010300
APA StyleHan, X., Huang, Q., Liu, L., Sha, X., Hu, B., & Liu, H. (2018). Changes in the Expression of AQP4 and AQP9 in the Hippocampus Following Eclampsia-Like Seizure. International Journal of Molecular Sciences, 19(1), 300. https://doi.org/10.3390/ijms19010300



