Intraarticularly-Injected Mesenchymal Stem Cells Stimulate Anti-Inflammatory Molecules and Inhibit Pain Related Protein and Chondrolytic Enzymes in a Monoiodoacetate-Induced Rat Arthritis Model
Abstract
1. Introduction
2. Results
2.1. Immunofluorescence Staining of CGRP in the Spinal Dorsal Horn
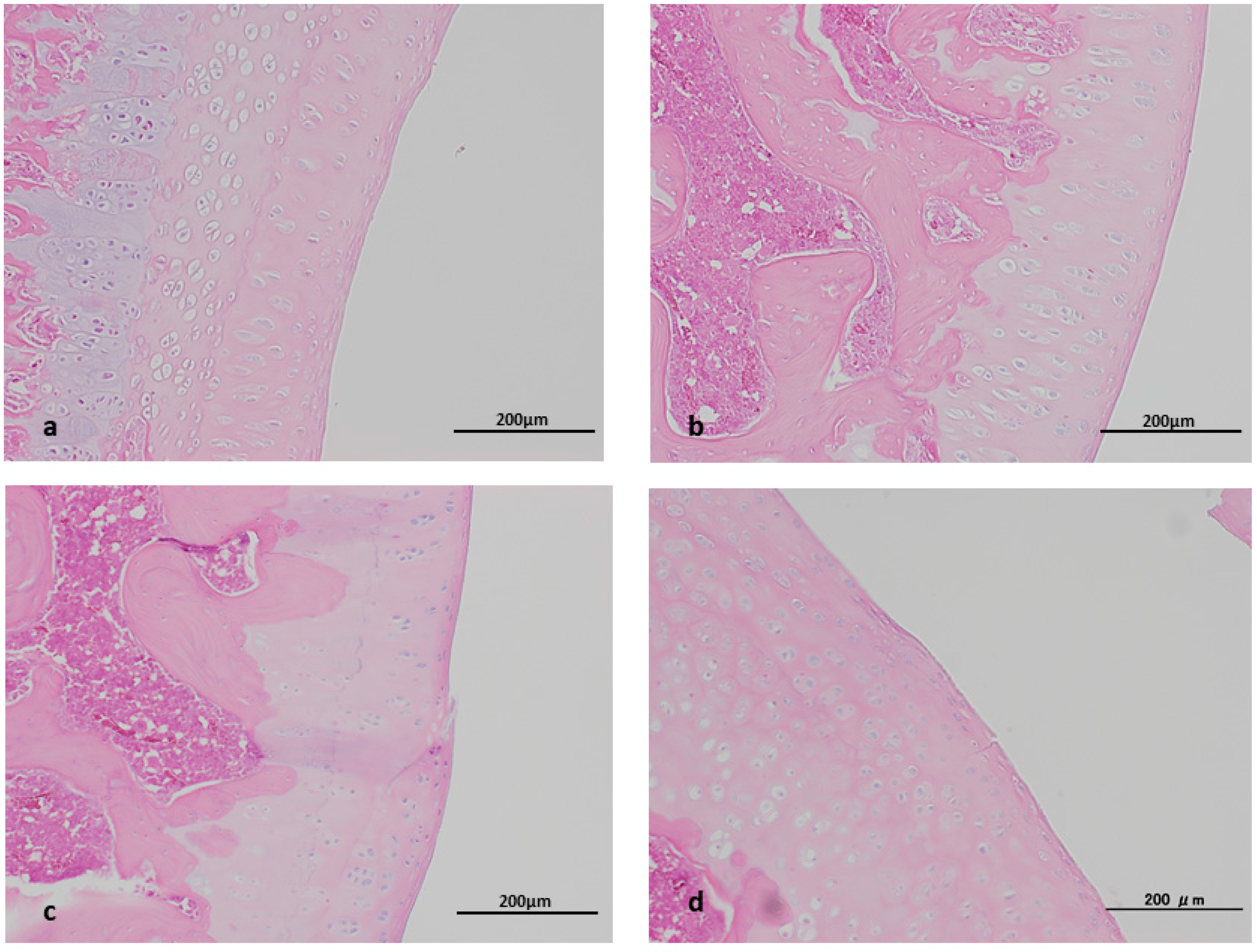
2.2. Histopathological Study
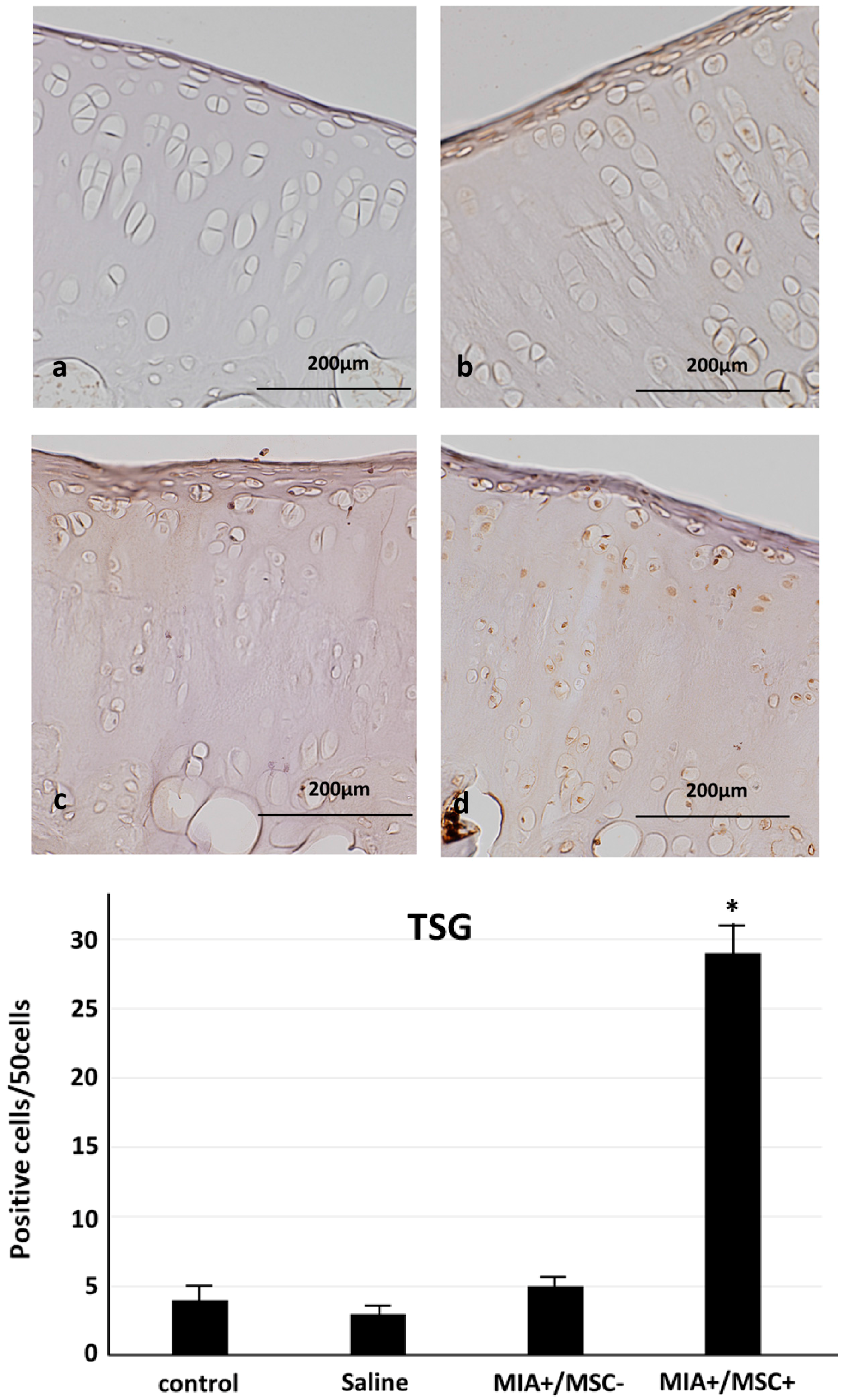
2.3. TSG-6 Expression in Joint Cartilage
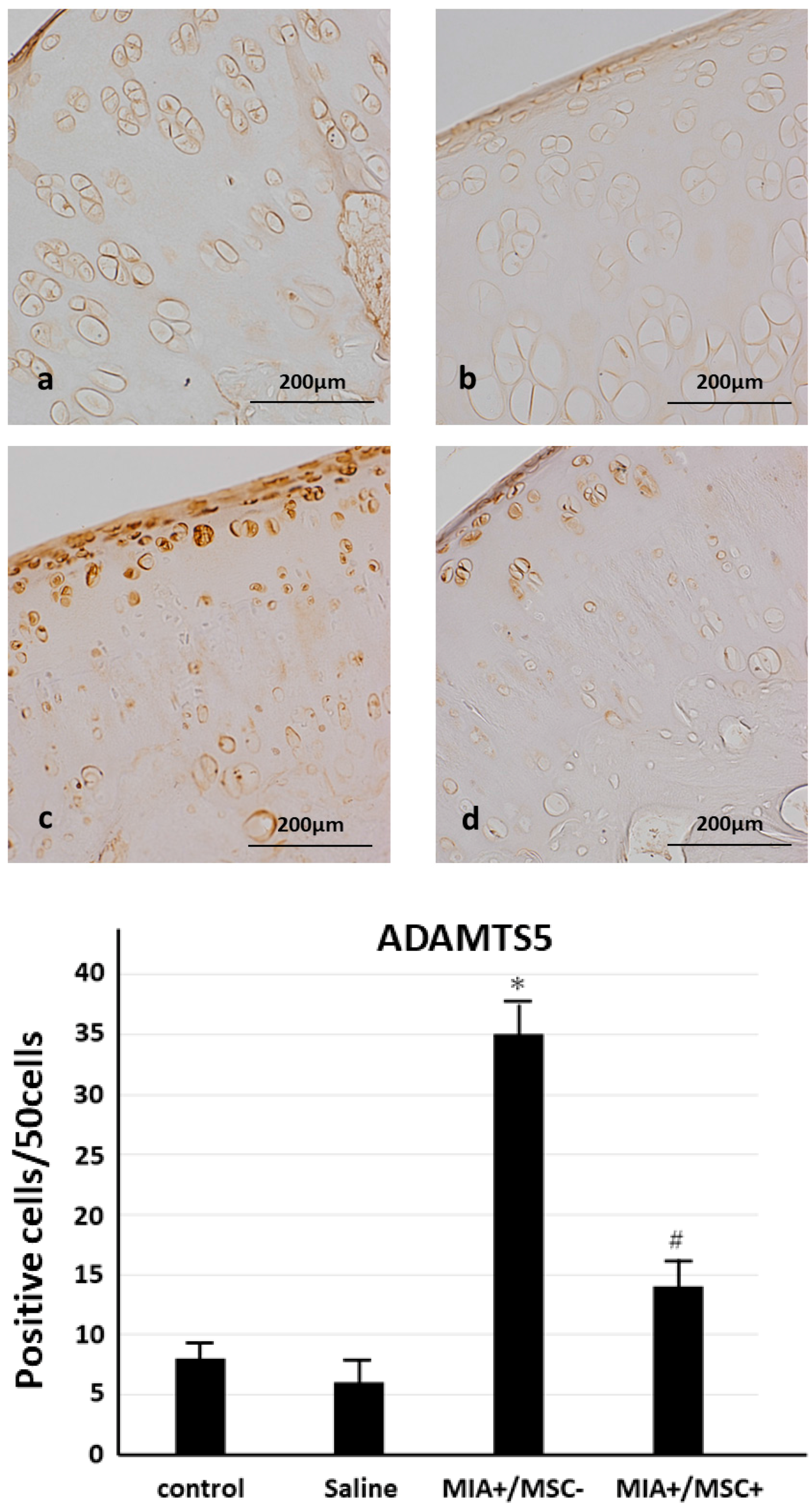
2.4. ADAMTS 5 Expression in Joint Cartilage
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Cell Culture
4.3. Treatment and Tissue Preparation
4.4. Histopathology of the Shoulder Joint
4.5. Immunofluorescence Imaging
4.6. Immunostaining for ADAMTs 5, Stimulated Gene 6 Protein (TSG-6)
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Conflicts of Interest
Abbreviations
| MIA | Monoiodoacetate |
| MSC | Mesenchymal stem cell |
| ADAMTS5 | A disintegrin and metalloproteinase with thrombospondin motifs 5 |
| CGRP | Calcitonin gene related peptide |
| TSG-6 | TNF-α stimulated gene/protein 6 |
| OA | Osteoarthritis |
References
- Yunus, M.B. Fibromyalgia and overlapping disorders: The unifying concept of central sensitivity syndromes. Semin. Arthritis Rheum. 2007, 36, 339–356. [Google Scholar] [CrossRef] [PubMed]
- Woolf, C.J. Central sensitization: Implications for the diagnosis and treatment of pain. Pain 2011, 152, S2–S15. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.W.; Tan, A.; Saab, C.; Waxman, S. Unilateral focal burn injury is followed by long lasting bilateral allodynia and neuronal hyperexcitability in spinal cord dorsal horn. J. Pain 2010, 11, 119–130. [Google Scholar] [CrossRef] [PubMed]
- Neblett, R.; Cohen, H.; Choi, Y.; Hartzell, M.M.; Williams, M.; Mayer, T.G.; Gatchel, R.J. The Central Sensitization Inventory (CSI): Establishing clinically significant values for identifying central sensitivity syndromes in an outpatient chronic pain sample. J. Pain 2013, 14, 438–445. [Google Scholar] [CrossRef] [PubMed]
- Nijs, J.; van Houdenhove, B.; Oostendorp, R.A. Recognition of central sensitization in patients with musculoskeletal pain: Application of pain neurophysiology in manual therapy practice. Man. Ther. 2010, 15, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Raphael, K.G.; Janal, M.N.; Anathan, S.; Cook, D.B.; Staud, R. Temporal summation of heat pain in temporomandibular disorder patients. J. Orofac. Pain 2009, 23, 54–64. [Google Scholar] [PubMed]
- Schur, E.A.; Afari, N.; Furberg, H.; Olarte, M.; Goldberg, J.; Sullivan, P.F.; Buchwald, D. Feeling bad in more ways than one: Comorbidity patterns of medically unexplained and psychiatric conditions. J. Gen. Intern. Med. 2007, 22, 818–821. [Google Scholar] [CrossRef] [PubMed]
- Dieppe, P.A.; Lohmander, L.S. Pathogenesis and management of pain in osteoarthritis. Lancet 2005, 365, 965–973. [Google Scholar] [CrossRef]
- Kim, Y.S.; Choi, Y.J.; Suh, D.S.; Heo, D.B.; Kim, Y.I.; Ryu, J.S.; Koh, Y.G. Mesenchymal stem cell implantation in osteoarthritic knees: Is fibrin glue effective as a scaffold? Am. J. Sports Med. 2015, 43, 176–185. [Google Scholar] [CrossRef] [PubMed]
- Ueda, S.; Shimasaki, M.; Ichiseki, T.; Ueda, Y.; Tsuchiya, M.; Kaneuji, A.; Kawahara, N. Prevention of glucocorticoid-associated osteonecrosis by intravenous administration of mesenchymal stem cells in a rabbit model. BMC Musculoskelet. Disord. 2017, 18, 480. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Yamaoka, K.; Sonomoto, K.; Kaneko, H.; Satake, M.; Yamamoto, Y.; Kondo, M.; Zhao, J.; Miyagawa, I.; Yamagata, K.; et al. Local delivery of mesenchymal stem cells with poly-lactic-co-glycolic acid nano-fiber scaffold suppress arthritis in rats. PLoS ONE 2014, 9, e114621. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Qi, Y.; Du, Y.; Li, W.; Dai, X.; Zhao, T.; Yan, W. Cartilage repair using mesenchymal stem cell (MSC) sheet and MSCs-loaded bilayer PLGA scaffold in a rabbit model. Knee Surg. Sports Traumatol. Arthrosc. 2014, 22, 1424–1433. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.S.; Kwon, O.R.; Choi, Y.J.; Suh, D.S.; Heo, D.B.; Koh, Y.G. Comparative Matched-Pair Analysis of the Injection Versus Implantation of Mesenchymal Stem Cells for Knee Osteoarthritis. Am. J. Sports Med. 2015, 43, 2738–2746. [Google Scholar] [CrossRef] [PubMed]
- Tyndall, A.; Houssiau, F.A. Mesenchymal stem cells in the treatment of autoimmune diseases. Ann. Rheum. Dis. 2010, 69, 1413–1414. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Akiyama, K.; Zhang, H.; Yamaza, T.; Hou, Y.; Zhao, S.; Xu, T.; Le, A.; Shi, S. Mesenchymal stem cell transplantation reverses multiorgan dysfunction in systemic lupus erythematosus mice and humans. Stem Cells 2009, 27, 1421–1432. [Google Scholar] [CrossRef] [PubMed]
- Udo, M.; Muneta, T.; Tsuji, K.; Ozeki, N.; Nakagawa, Y.; Ohara, T.; Saito, R.; Yanagisawa, K.; Koga, H.; Sekiya, I. Monoiodoacetic acid induces arthritis and synovitis in rats in a dose- and time-dependent manner: Proposed model-specific scoring systems. Osteoarthr. Cartil. 2016, 24, 1284–1291. [Google Scholar] [CrossRef] [PubMed]
- Maresca, M.; Micheli, L.; Cinci, L.; Bilia, A.R.; Ghelardini, C.; Di Cesare Mannelli, L. Pain relieving and protective effects of Astragalus hydroalcoholic extract in rat arthritis models. J. Pharm. Pharmacol. 2017, 69, 1858–1870. [Google Scholar] [CrossRef] [PubMed]
- Philpott, H.T.; O’Brien, M.; McDougall, J.J. Attenuation of early phase inflammation by cannabidiol prevents pain and nerve damage in rat osteoarthritis. Pain 2017, 158, 2442–2451. [Google Scholar] [CrossRef] [PubMed]
- Ochiai, N.; Ohtori, S.; Kenmoku, T.; Yamazaki, H.; Ochiai, S.; Saisu, T.; Matsuki, K.; Takahashi, K. Sensory innervation of rat contracture shoulder model. J. Shoulder Elb. Surg. 2013, 22, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, T.; Ochiai, N.; Sasaki, Y.; Kijima, T.; Hashimoto, E.; Sasaki, Y.; Kenmoku, T.; Yamazaki, H.; Miyagi, M.; Ohtori, S.; et al. Efficacy of hyaluronic acid or steroid injections for the treatment of a rat model of rotator cuff injury. J. Orthop. Res. 2015, 33, 1861–1867. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, L.A.; Dutreil, M.; Fattman, C.; Pandey, A.C.; Torres, G.; Go, K.; Phinney, D.G. Interleukin 1 receptor antagonist mediates the anti inflammatory and antifibrotic effect of mesenchymal stem cells during lung injury. Proc. Natl. Acad. Sci. USA 2007, 104, 11002–11007. [Google Scholar] [CrossRef] [PubMed]
- Yagi, H.; Soto-Gutierrez, A.; Navarro-Alvarez, N.; Nahmias, Y.; Goldwasser, Y.; Kitagawa, Y.; Tilles, A.W.; Tompkins, R.G.; Parekkadan, B.; Yarmush, M.L. Reactive bone marrow stromal cells attenuate systemic inflammation via sTNFR1. Mol. Ther. 2010, 18, 1857–1864. [Google Scholar] [CrossRef] [PubMed]
- Wisniewski, H.G.; Vilcek, J. Cytokine-induced gene expression at the crossroads of innate immunity, inflammation and fertility: TSG-6 and PTX3/TSG-14. Cytokine Growth Factor Rev. 2004, 15, 129–146. [Google Scholar] [CrossRef] [PubMed]
- Roddy, G.W.; Oh, J.Y.; Lee, R.H.; Bartosh, T.J.; Ylostalo, J.; Coble, K.; Rosa, R.H., Jr.; Prockop, D.J. Action at a distance: Systemically administered adult stem/progenitor cells (MSCs) reduce inflammatory damage to the cornea without engraftment and primarily by secretion of TNF-α stimulated gene/protein 6. Stem Cells 2011, 29, 1572–1579. [Google Scholar] [CrossRef] [PubMed]
- Nakabayashi, K.; Sakamoto, J.; Kataoka, H.; Kondo, Y.; Hamaue, Y.; Honda, Y.; Nakano, J.; Okita, M. Effect of continuous passive motion initiated after the onset of arthritis on inflammation and secondary hyperalgesia in rats. Physiol. Res. 2016, 65, 683–691. [Google Scholar] [PubMed]
- Hamaue, Y.; Nakano, J.; Sekino, Y.; Chuganji, S.; Sakamoto, J.; Yoshimura, T.; Okita, M.; Origuchi, T. Effects of Vibration Therapy on Immobilization-Induced Hypersensitivity in Rats. Phys. Ther. 2015, 95, 1015–1026. [Google Scholar] [CrossRef] [PubMed]
- Hamaue, Y.; Nakano, J.; Sekino, Y.; Chuganji, S.; Sakamoto, J.; Yoshimura, T.; Origuchi, T.; Okita, M. Immobilization-induced hypersensitivity associated with spinal cord sensitization during cast immobilization and after cast removal in rats. J. Physiol. Sci. 2013, 63, 401–408. [Google Scholar] [CrossRef] [PubMed]
- Glant, T.T.; Kamath, R.V.; Bárdos, T.; Gál, I.; Szántó, S.; Murad, Y.M.; Sandy, J.D.; Mort, J.S.; Roughley, P.J.; Mikecz, K. Cartilage-specific constitutive expression of TSG-6 protein (product of tumor necrosis factor alpha-stimulated gene 6) provides a chondroprotective, but not antiinflammatory, effect in antigen-induced arthritis. Arthritis Rheum. 2002, 46, 2207–2218. [Google Scholar] [CrossRef] [PubMed]
- Sohn, D.H.; Lottman, L.M.; Lum, L.Y.; Kim, S.G.; Pedowitz, R.A.; Coutts, R.D.; Sah, R.L. Effect of gravity on localization of chondrocytes implanted in cartilage defects. Clin. Orthop. Relat. Res. 2002, 394, 254–262. [Google Scholar] [CrossRef]
- Miyaki, S.; Sato, T.; Inoue, A.; Otsuki, S.; Ito, Y.; Yokoyama, S.; Kato, Y.; Takemoto, F.; Nakasa, T.; Yamashita, S.; et al. MicroRNA-140 plays dual roles in both cartilage development and homeostasis. Genes Dev. 2010, 24, 1173–1185. [Google Scholar] [CrossRef] [PubMed]
- Glasson, S.S.; Askew, R.; Sheppard, B.; Carito, B.; Blanchet, T.; Ma, H.L.; Flannery, C.R.; Peluso, D.; Kanki, K.; Yang, Z.; et al. Deletion of active ADAMTS5 prevents cartilage degradation in a murine model of osteoarthritis. Nature 2005, 434, 644–648. [Google Scholar] [CrossRef] [PubMed]
- Stanton, H.; Rogerson, F.M.; East, C.J.; Golub, S.B.; Lawlor, K.E.; Meeker, C.T.; Little, C.B.; Last, K.; Farmer, P.J.; Campbell, I.K.; et al. ADAMTS5 is the major aggrecanase in mouse cartilage in vivo and in vitro. Nature 2005, 434, 648–652. [Google Scholar] [CrossRef] [PubMed]
- Higuchi, Y.; Nishida, Y.; Kozawa, E.; Zhuo, L.; Arai, E.; Hamada, S.; Morita, D.; Ikuta, K.; Kimata, K.; Ushida, T.; et al. Conditional knockdown of hyaluronidase 2 in articular cartilage stimulates osteoarthritic progression in a mice model. Sci. Rep. 2017, 7, 7028. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ichiseki, T.; Shimasaki, M.; Ueda, Y.; Ueda, S.; Tsuchiya, M.; Souma, D.; Kaneuji, A.; Kawahara, N. Intraarticularly-Injected Mesenchymal Stem Cells Stimulate Anti-Inflammatory Molecules and Inhibit Pain Related Protein and Chondrolytic Enzymes in a Monoiodoacetate-Induced Rat Arthritis Model. Int. J. Mol. Sci. 2018, 19, 203. https://doi.org/10.3390/ijms19010203
Ichiseki T, Shimasaki M, Ueda Y, Ueda S, Tsuchiya M, Souma D, Kaneuji A, Kawahara N. Intraarticularly-Injected Mesenchymal Stem Cells Stimulate Anti-Inflammatory Molecules and Inhibit Pain Related Protein and Chondrolytic Enzymes in a Monoiodoacetate-Induced Rat Arthritis Model. International Journal of Molecular Sciences. 2018; 19(1):203. https://doi.org/10.3390/ijms19010203
Chicago/Turabian StyleIchiseki, Toru, Miyako Shimasaki, Yoshimichi Ueda, Shusuke Ueda, Masanobu Tsuchiya, Daisuke Souma, Ayumi Kaneuji, and Norio Kawahara. 2018. "Intraarticularly-Injected Mesenchymal Stem Cells Stimulate Anti-Inflammatory Molecules and Inhibit Pain Related Protein and Chondrolytic Enzymes in a Monoiodoacetate-Induced Rat Arthritis Model" International Journal of Molecular Sciences 19, no. 1: 203. https://doi.org/10.3390/ijms19010203
APA StyleIchiseki, T., Shimasaki, M., Ueda, Y., Ueda, S., Tsuchiya, M., Souma, D., Kaneuji, A., & Kawahara, N. (2018). Intraarticularly-Injected Mesenchymal Stem Cells Stimulate Anti-Inflammatory Molecules and Inhibit Pain Related Protein and Chondrolytic Enzymes in a Monoiodoacetate-Induced Rat Arthritis Model. International Journal of Molecular Sciences, 19(1), 203. https://doi.org/10.3390/ijms19010203