Identification of Reference and Biomarker Proteins in Chlamydomonas reinhardtii Cultured under Different Stress Conditions
Abstract
:1. Introduction
2. Results and Discussion
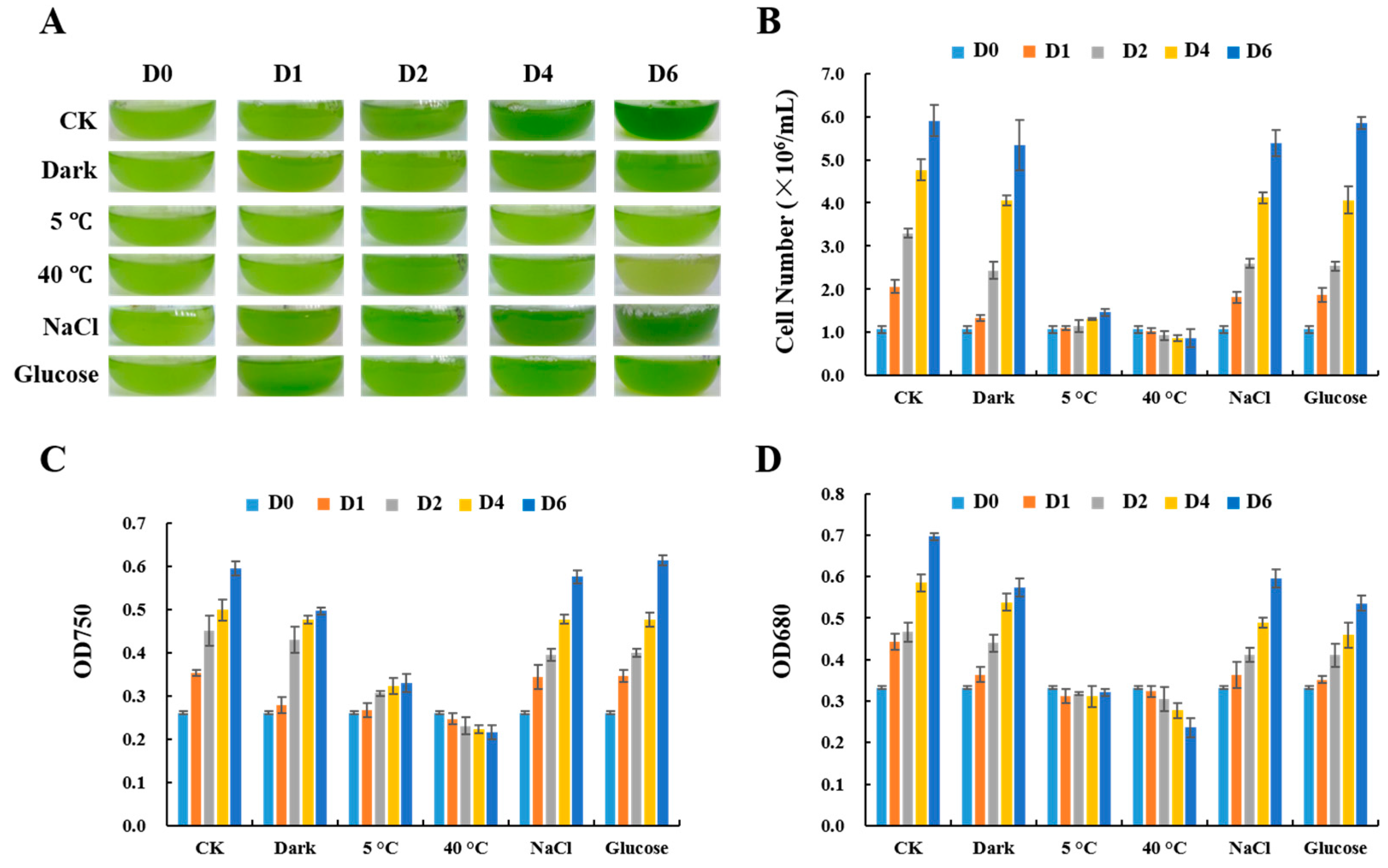
2.1. Growth of C. reinhardtii Cells under Stress
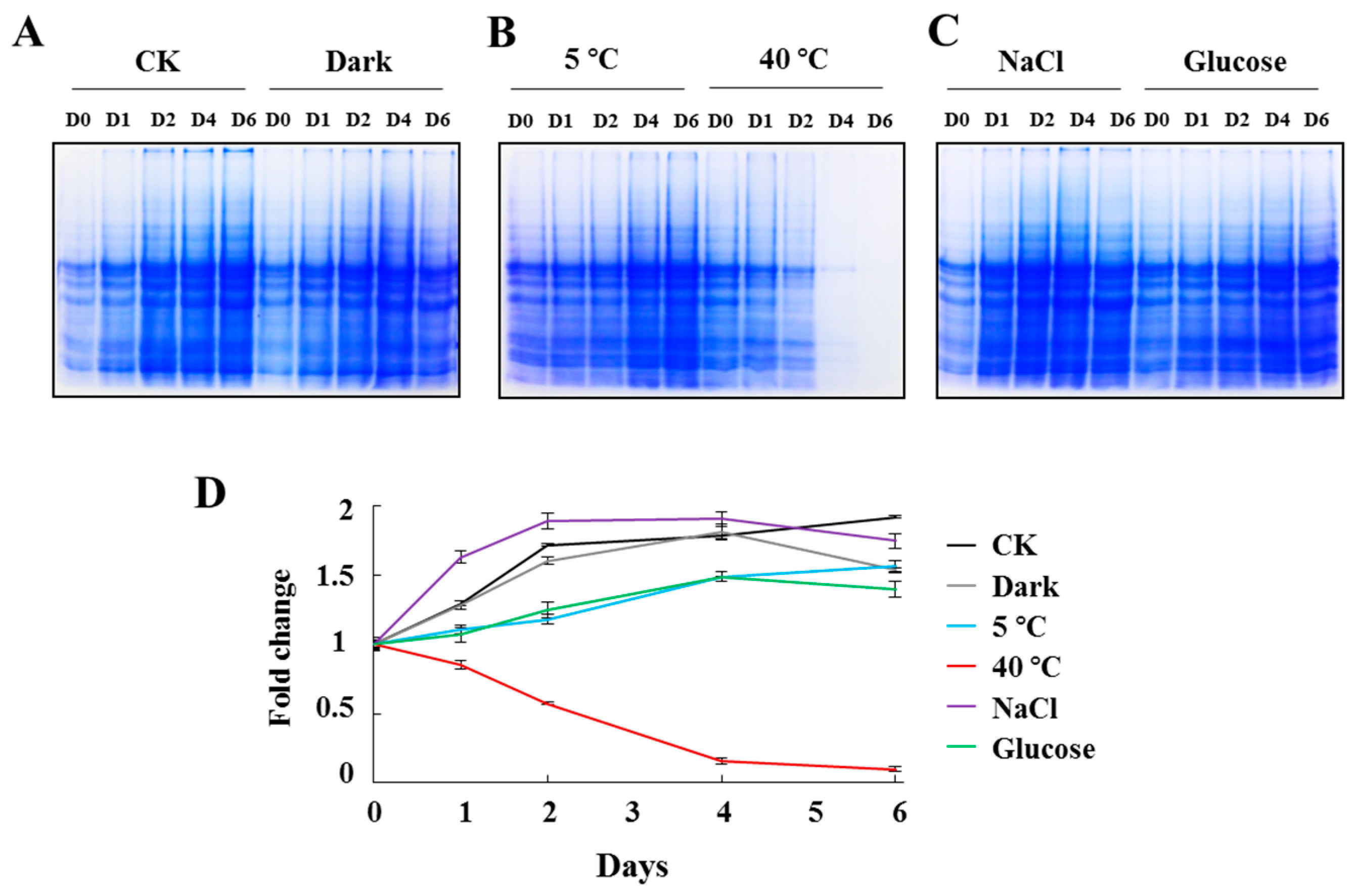
2.2. Total Protein Alteration of C. reinhardtii Cells under Stress
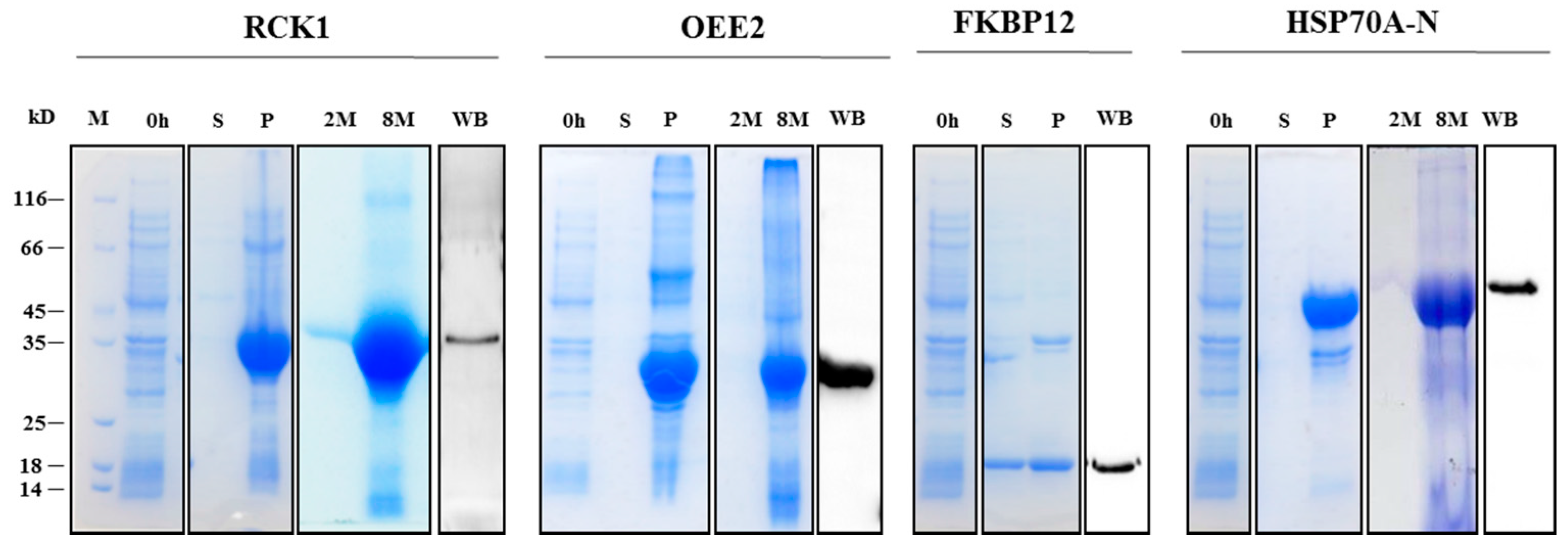
2.3. Recombinant Protein Expression, Antibody Generation, and Specificity Validation
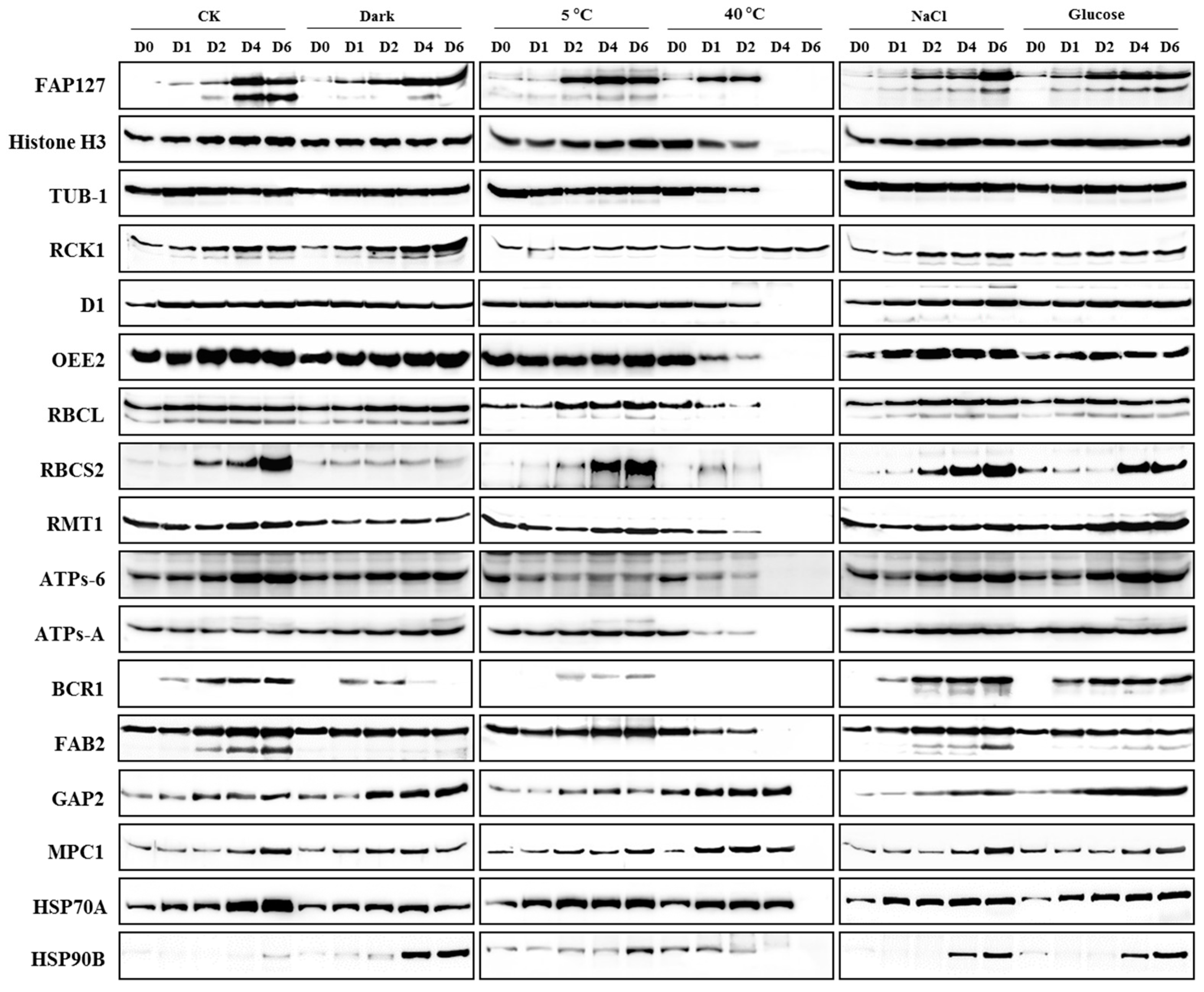
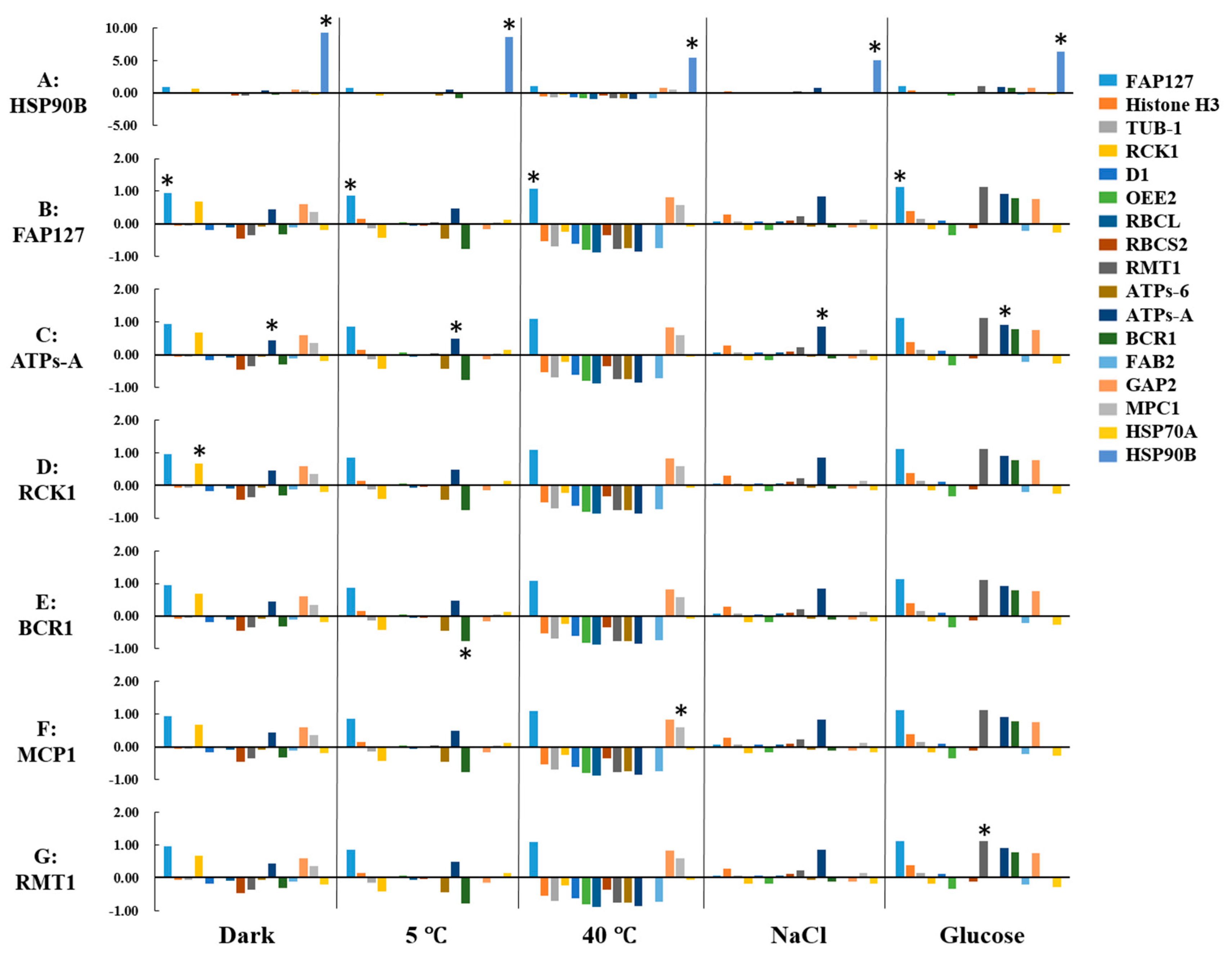
2.4. Expression Profiling of Candidate Proteins
2.5. Identification of Reference Proteins under Abiotic Stresses
2.6. Identification of Biomarkers under Abiotic Stresses
3. Materials and Methods
3.1. C. reinhardtii Strain and Culture
3.2. Abiotic Stress Treatments
3.3. Detection of Cell Density and Cell Number
3.4. Candidate Proteins
3.5. Recombinant Protein Expression and Antibody Generation
3.6. C. reinhardtii Protein Extraction and Quantification
3.7. Western Blot Analyses
3.8. Pearson’s Correlation Coefficient (PCC) Analysis
3.9. Average Relative Fold Change (ARF) Calculation and Biomarker Selection
4. Conclusions
Supplementary Materials
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mata, T.M.; Martins, A.A.; Caetano, N.S. Microalgae for biodiesel production and other applications: A review. Renew. Sustain. Energy Rev. 2010, 14, 217–232. [Google Scholar] [CrossRef]
- Klassen, V.; Blifernez-Klassen, O.; Wobbe, L.; Schluter, A.; Kruse, O.; Mussgnug, J.H. Efficiency and biotechnological aspects of biogas production from microalgal substrates. J. Biotechnol. 2016, 234, 7–26. [Google Scholar] [CrossRef] [PubMed]
- Razzak, S.A.; Ilyas, M.; Ali, S.A.; Hossain, M.M. Effects of CO(2) concentration and pH on mixotrophic growth of nannochloropsis oculata. Appl. Biochem. Biotechnol. 2015, 176, 1290–1302. [Google Scholar] [CrossRef] [PubMed]
- Williams, P.J. Biofuel: Microalgae cut the social and ecological costs. Nature 2007, 450, 478. [Google Scholar] [CrossRef] [PubMed]
- Merchant, S.S.; Prochnik, S.E.; Vallon, O.; Harris, E.H.; Karpowicz, S.J.; Witman, G.B.; Terry, A.; Salamov, A.; Fritz-Laylin, L.K.; Marechal-Drouard, L.; et al. The Chlamydomonas genome reveals the evolution of key animal and plant functions. Science 2007, 318, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhang, R.; Patena, W.; Gang, S.S.; Blum, S.R.; Ivanova, N.; Yue, R.; Robertson, J.M.; Lefebvre, P.A.; Fitz-Gibbon, S.T.; et al. An indexed, mapped mutant library enables reverse genetics studies of biological processes in Chlamydomonas reinhardtii. Plant Cell 2016, 28, 367–387. [Google Scholar] [CrossRef] [PubMed]
- Hema, R.; Senthil-Kumar, M.; Shivakumar, S.; Chandrasekhara Reddy, P.; Udayakumar, M. Chlamydomonas reinhardtii, a model system for functional validation of abiotic stress responsive genes. Planta 2007, 226, 655–670. [Google Scholar] [CrossRef] [PubMed]
- Batyrova, K.; Hallenbeck, P.C. Hydrogen production by a Chlamydomonas reinhardtii strain with inducible expression of photosystem II. Int. J. Mol. Sci. 2017, 18, 647. [Google Scholar] [CrossRef] [PubMed]
- Hemschemeier, A.; Casero, D.; Liu, B.; Benning, C.; Pellegrini, M.; Happe, T.; Merchant, S.S. Copper response regulator1-dependent and -independent responses of the Chlamydomonas reinhardtii transcriptome to dark anoxia. Plant Cell 2013, 25, 3186–3211. [Google Scholar] [CrossRef] [PubMed]
- Chaudhari, V.R.; Vyawahare, A.; Bhattacharjee, S.K.; Rao, B.J. Enhanced excision repair and lack of PSII activity contribute to higher UV survival of Chlamydomonas reinhardtii cells in dark. Plant Physiol. Biochem. 2015, 88, 60–69. [Google Scholar] [CrossRef] [PubMed]
- Valledor, L.; Furuhashi, T.; Hanak, A.M.; Weckwerth, W. Systemic cold stress adaptation of Chlamydomonas reinhardtii. Mol. Cell Proteom. 2013, 12, 2032–2047. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.J.; Jung, W.; Lim, S.; Kim, S.; Han, S.J.; Choi, H.G. Growth and lipid content at low temperature of Arctic alga Chlamydomonas sp. KNM0029C. Bioprocess. Biosyst. Eng. 2016, 39, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, Y.; Nishiyama, Y.; Murata, N. Acclimation of the photosynthetic machinery to high temperature in Chlamydomonas reinhardtii requires synthesis de novo of proteins encoded by the nuclear and chloroplast genomes. Plant Physiol. 2000, 124, 441–449. [Google Scholar] [CrossRef] [PubMed]
- Schroda, M.; Hemme, D.; Muhlhaus, T. The Chlamydomonas heat stress response. Plant J. 2015, 82, 466–480. [Google Scholar] [CrossRef] [PubMed]
- Muhlhaus, T.; Weiss, J.; Hemme, D.; Sommer, F.; Schroda, M. Quantitative shotgun proteomics using a uniform (1)(5)N-labeled standard to monitor proteome dynamics in time course experiments reveals new insights into the heat stress response of Chlamydomonas reinhardtii. Mol. Cell Proteom. 2011, 10, 1–27. [Google Scholar] [CrossRef] [PubMed]
- Hemme, D.; Veyel, D.; Muhlhaus, T.; Sommer, F.; Juppner, J.; Unger, A.K.; Sandmann, M.; Fehrle, I.; Schonfelder, S.; Steup, M.; et al. Systems-wide analysis of acclimation responses to long-term heat stress and recovery in the photosynthetic model organism Chlamydomonas reinhardtii. Plant Cell 2014, 26, 4270–4297. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.H.; Pan, K.Y.; Hung, C.H.; Huang, H.E.; Chen, C.L.; Feng, T.Y.; Huang, L.F. Overexpression of ferredoxin, PETF, enhances tolerance to heat stress in Chlamydomonas reinhardtii. Int. J. Mol. Sci. 2013, 14, 20913–20929. [Google Scholar] [CrossRef] [PubMed]
- Legeret, B.; Schulz-Raffelt, M.; Nguyen, H.M.; Auroy, P.; Beisson, F.; Peltier, G.; Blanc, G.; Li-Beisson, Y. Lipidomic and transcriptomic analyses of Chlamydomonas reinhardtii under heat stress unveil a direct route for the conversion of membrane lipids into storage lipids. Plant Cell Environ. 2016, 39, 834–847. [Google Scholar] [CrossRef] [PubMed]
- Yokthongwattana, C.; Mahong, B.; Roytrakul, S.; Phaonaklop, N.; Narangajavana, J.; Yokthongwattana, K. Proteomic analysis of salinity-stressed Chlamydomonas reinhardtii revealed differential suppression and induction of a large number of important housekeeping proteins. Planta 2012, 235, 649–659. [Google Scholar] [CrossRef] [PubMed]
- Hom, E.F.; Murray, A.W. Niche engineering demonstrates a latent capacity for fungal-algal mutualism. Science 2014, 345, 94–98. [Google Scholar] [CrossRef] [PubMed]
- Bennett, M.E.; Hobbie, J.E. The uptake of glucose by Chlamydomonas sp. J. Phycol. 1972, 8, 392–398. [Google Scholar] [CrossRef]
- Allorent, G.; Tokutsu, R.; Roach, T.; Peers, G.; Cardol, P.; Girard-Bascou, J.; Seigneurin-Berny, D.; Petroutsos, D.; Kuntz, M.; Breyton, C.; et al. A dual strategy to cope with high light in Chlamydomonas reinhardtii. Plant Cell 2013, 25, 545–557. [Google Scholar] [CrossRef] [PubMed]
- Schloss, J.A. A Chlamydomonas gene encodes a G protein beta subunit-like polypeptide. Mol. Gen. Genet. 1990, 221, 443–452. [Google Scholar] [CrossRef] [PubMed]
- Kohinata, T.; Nishino, H.; Fukuzawa, H. Significance of zinc in a regulatory protein, CCM1, which regulates the carbon-concentrating mechanism in Chlamydomonas reinhardtii. Plant Cell Physiol. 2008, 49, 273–283. [Google Scholar] [CrossRef] [PubMed]
- Iwasaki, Y.; Komano, M.; Ishikawa, A.; Sasaki, T.; Asahi, T. Molecular cloning and characterization of cDNA for a rice protein that contains seven repetitive segments of the Trp-Asp forty-amino-acid repeat (WD-40 repeat). Plant Cell Physiol. 1995, 36, 505–510. [Google Scholar] [CrossRef] [PubMed]
- Tokutsu, R.; Minagawa, J. Energy-dissipative supercomplex of photosystem II associated with LHCSR3 in Chlamydomonas reinhardtii. Proc. Natl. Acad. Sci. USA 2013, 110, 10016–10021. [Google Scholar] [CrossRef] [PubMed]
- Heinnickel, M.L.; Alric, J.; Wittkopp, T.; Yang, W.; Catalanotti, C.; Dent, R.; Niyogi, K.K.; Wollman, F.A.; Grossman, A.R. Novel thylakoid membrane GreenCut protein CPLD38 impacts accumulation of the cytochrome b6f complex and associated regulatory processes. J. Biol. Chem. 2013, 288, 7024–7036. [Google Scholar] [CrossRef] [PubMed]
- Meyer, M.T.; Genkov, T.; Skepper, J.N.; Jouhet, J.; Mitchell, M.C.; Spreitzer, R.J.; Griffiths, H. Rubisco small-subunit alpha-helices control pyrenoid formation in Chlamydomonas. Proc. Natl. Acad. Sci. USA 2012, 109, 19474–19479. [Google Scholar] [CrossRef] [PubMed]
- Elyse Ireland, H.; Harding, S.J.; Bonwick, G.A.; Jones, M.; Smith, C.J.; Williams, J.H. Evaluation of heat shock protein 70 as a biomarker of environmental stress in Fucus serratus and Lemna minor. Biomarkers 2004, 9, 139–155. [Google Scholar] [CrossRef] [PubMed]
- Chankova, S.G.; Dimova, E.G.; Mitrovska, Z.; Miteva, D.; Mokerova, D.V.; Yonova, P.A.; Yurina, N.P. Antioxidant and HSP70B responses in Chlamydomonas reinhardtii genotypes with different resistance to oxidative stress. Ecotoxicol. Environ. Saf. 2014, 101, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Girolomoni, L.; Ferrante, P.; Berteotti, S.; Giuliano, G.; Bassi, R.; Ballottari, M. The function of LHCBM4/6/8 antenna proteins in Chlamydomonas reinhardtii. J. Exp. Bot. 2017, 68, 627–641. [Google Scholar] [PubMed]
- Long, J.C.; Sommer, F.; Allen, M.D.; Lu, S.F.; Merchant, S.S. FER1 and FER2 encoding two ferritin complexes in Chlamydomonas reinhardtii chloroplasts are regulated by iron. Genetics 2008, 179, 137–147. [Google Scholar] [CrossRef] [PubMed]
- Lechtreck, K.F.; Gould, T.J.; Witman, G.B. Flagellar central pair assembly in Chlamydomonas reinhardtii. Cilia 2013, 2, 15–33. [Google Scholar] [CrossRef] [PubMed]
- Oda, T.; Kikkawa, M. Novel structural labeling method using cryo-electron tomography and biotin-streptavidin system. J. Struct. Biol. 2013, 183, 305–311. [Google Scholar] [CrossRef] [PubMed]
- Knepper, M.A.; Masilamani, S. Targeted proteomics in the kidney using ensembles of antibodies. Acta Physiol. Scand. 2001, 173, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Lindskog, C.; Asplund, A.; Engkvist, M.; Uhlen, M.; Korsgren, O.; Ponten, F. Antibody-based proteomics for discovery and exploration of proteins expressed in pancreatic islets. Discov. Med. 2010, 9, 565–578. [Google Scholar] [PubMed]
- Montoro-Molina, S.; Quesada, A.; Zafra-Ruiz, P.V.; O’Valle, F.; Vargas, F.; de Gracia Mdel, C.; Osuna, A.; Wangensteen, R. Immunological detection of glutamyl aminopeptidase in urine samples from cisplatin-treated rats. Proteom. Clin. Appl. 2015, 9, 630–635. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Bai, H.; Wang, X.; Li, L.; Cao, Y.; Wei, J.; Liu, Y.; Liu, L.; Gong, X.; Wu, L.; et al. Identification and validation of rice reference proteins for western blotting. J. Exp. Bot. 2011, 62, 4763–4772. [Google Scholar] [CrossRef] [PubMed]
- Bai, H.; Lan, J.P.; Gan, Q.; Wang, X.Y.; Hou, M.M.; Cao, Y.H.; Li, L.Y.; Liu, L.J.; Hao, Y.J.; Yin, C.C.; et al. Identification and expression analysis of components involved in rice Xa21-mediated disease resistance signalling. Plant Biol. 2012, 14, 914–922. [Google Scholar] [CrossRef] [PubMed]
- Hou, M.; Xu, W.; Bai, H.; Liu, Y.; Li, L.; Liu, L.; Liu, B.; Liu, G. Characteristic expression of rice pathogenesis-related proteins in rice leaves during interactions with Xanthomonas oryzae pv. oryzae. Plant Cell Rep. 2012, 31, 895–904. [Google Scholar] [CrossRef] [PubMed]
- Chlamydomonas Resource Center. Available online: https://www.chlamycollection.org/ (accessed on 21 August 2017).
- Lechtreck, K.F.; Brown, J.M.; Sampaio, J.L.; Craft, J.M.; Shevchenko, A.; Evans, J.E.; Witman, G.B. Cycling of the signaling protein phospholipase D through cilia requires the BBSome only for the export phase. J. Cell Biol. 2013, 201, 249–261. [Google Scholar] [CrossRef] [PubMed]
- Perez-Perez, M.E.; Couso, I.; Crespo, J.L. Carotenoid deficiency triggers autophagy in the model green alga Chlamydomonas reinhardtii. Autophagy 2012, 8, 376–388. [Google Scholar] [CrossRef] [PubMed]
- Dorn, K.V.; Willmund, F.; Schwarz, C.; Henselmann, C.; Pohl, T.; Hess, B.; Veyel, D.; Usadel, B.; Friedrich, T.; Nickelsen, J.; et al. Chloroplast DnaJ-like proteins 3 and 4 (CDJ3/4) from Chlamydomonas reinhardtii contain redox-active Fe-S clusters and interact with stromal HSP70B. Biochem. J. 2010, 427, 205–215. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, H.; Clowez, S.; Wollman, F.A.; Vallon, O.; Rappaport, F. Cyclic electron flow is redox-controlled but independent of state transition. Nat. Commun. 2013, 4, 1954–1961. [Google Scholar] [CrossRef] [PubMed]
- Coragliotti, A.T.; Beligni, M.V.; Franklin, S.E.; Mayfield, S.P. Molecular factors affecting the accumulation of recombinant proteins in the Chlamydomonas reinhardtii chloroplast. Mol. Biotechnol. 2011, 48, 60–75. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Huang, J.; Fan, K.W.; Jiang, Y.; Zhong, Y.; Sun, Z.; Chen, F. Production potential of Chlorella zofingienesis as a feedstock for biodiesel. Bioresour. Technol. 2010, 101, 8658–8663. [Google Scholar] [CrossRef] [PubMed]
- Erales, J.; Lignon, S.; Gontero, B. CP12 from Chlamydomonas reinhardtii, a permanent specific “chaperone-like” protein of glyceraldehyde-3-phosphate dehydrogenase. J. Biol. Chem. 2009, 284, 12735–12744. [Google Scholar] [CrossRef] [PubMed]
- PepDesign. Available online: http://www.221.192.241.69:8010/pepdesign/index.jsp (accessed on 21 August 2017).
- Ferrante, P.; Diener, D.R.; Rosenbaum, J.L.; Giuliano, G. Nickel and low CO(2)-controlled motility in Chlamydomonas through complementation of a paralyzed flagella mutant with chemically regulated promoters. BMC Plant Biol. 2011, 11, 22–29. [Google Scholar] [CrossRef] [PubMed]
| CK | Dark | 5 °C | 40 °C | NaCl | Glucose | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| OD750 | TP | OD750 | TP | OD750 | TP | OD750 | TP | OD750 | TP | OD750 | TP | TP with * | |
| OD750 | 1.000 | 0.988 ** | 1.000 | 0.781 | 1.000 | 0.626 | 1.000 | 0.890 * | 1.000 | 0.844 | 1.000 | 0.935 * | |
| FAP127 | 0.913 * | 0.862 | 0.949 * | 0.921 * | 0.634 | 0.905 * | 0.133 | 0.434 | 0.948 * | 0.633 | 0.999 ** | 0.933 * | 3 |
| Histone H3 | 0.957 * | 0.950 * | 0.685 | 0.827 | 0.697 | 0.937 * | 0.722 | 0.916 * | 0.963 ** | 0.949 * | 0.879 * | 0.713 | 4 |
| TUB-1 | 0.953 * | 0.949 * | 0.787 | 0.769 | 0.480 | 0.956 * | 0.819 | 0.982 ** | 0.903 * | 0.986 ** | 0.930 * | 0.765 | 4 |
| RCK1 | 0.945 * | 0.944 * | 0.996 ** | 0.808 | 0.491 | 0.868 | −0.870 | −0.849 | 0.948 * | 0.769 | 0.994 ** | 0.899 * | 2 |
| D1 | 0.977 ** | 0.946 * | −0.541 | −0.336 | 0.208 | 0.650 | 0.812 | 0.974 ** | 0.997 ** | 0.804 | 0.968 ** | 0.865 | 2 |
| OEE2 | 0.974 ** | 0.969 ** | 0.937 * | 0.866 | 0.439 | 0.742 | 0.737 | 0.952 * | 0.944 * | 0.963 ** | 0.984 ** | 0.868 | 3 |
| RBCL | 0.929 * | 0.895 * | 0.862 | 0.919 * | 0.800 | 0.881 * | 0.914 * | 0.921 * | 0.938 * | 0.771 | 0.984 ** | 0.879 * | 5 |
| RBCS2 | 0.883 * | 0.862 | 0.856 | 0.896 * | 0.747 | 0.978 ** | 0.449 | 0.778 | 0.914 * | 0.615 | 0.795 | 0.876 | 2 |
| RMT1 | 0.819 | 0.787 | −0.678 | −0.491 | 0.912 * | 0.799 | 0.901 * | 0.954 * | 0.938 * | 0.637 | 0.975 ** | 0.959 ** | 2 |
| ATPs-6 | 0.939 * | 0.918 * | 0.920 * | 0.958 * | −0.666 | −0.944 * | 0.920 * | 0.996 ** | 0.959 ** | 0.782 | 0.962 ** | 0.994 ** | 5 |
| ATPs-A | 0.384 | 0.279 | 0.986 ** | 0.818 | 0.523 | 0.917 * | 0.964 ** | 0.889 * | 0.932 * | 0.919 * | 0.778 | 0.592 | 3 |
| BCR1 | 0.961 ** | 0.982 ** | −0.125 | −0.002 | 0.745 | 0.836 | - | - | 0.884 * | 0.681 | 0.929 * | 0.790 | 1 |
| FAB2 | 0.915 * | 0.914 * | −0.286 | 0.207 | 0.668 | 0.973 ** | 0.958 * | 0.982 ** | 0.915 * | 0.774 | −0.815 | −0.851 | 3 |
| GAP2 | 0.903 * | 0.957 * | 0.893 * | 0.824 | 0.701 | 0.991 ** | −0.275 | 0.071 | 0.798 | 0.577 | 0.973 ** | 0.963 ** | 3 |
| MPC1 | 0.762 | 0.708 | 0.930 * | 0.944 * | 0.854 | 0.899 * | 0.000 | 0.315 | 0.831 | 0.431 | 0.804 | 0.836 | 2 |
| HSP70A | 0.900 * | 0.848 | 0.951 * | 0.920 * | 0.515 | 0.764 | −0.045 | 0.287 | 0.844 | 0.922 * | 0.985 ** | 0.901 * | 3 |
| HSP90B | 0.531 | 0.457 | 0.827 | 0.668 | 0.860 | 0.838 | 0.149 | 0.361 | 0.778 | 0.427 | 0.770 | 0.804 | 0 |
| Protein with * | 13(11)11 | 8(4)6 | 1(0)10 | 5(5)9 | 13(4)5 | 12(7)7 | |||||||
| No. | Gene Name | Au10_Locus/ID | Category | Subcellular Localization | Annotation | MW (kD) | Antigen Sequence | References |
|---|---|---|---|---|---|---|---|---|
| 1 | FAP127 | Cre17.g737100 | Structure | Motile cilium | Flagellar associated protein | 51 | YSEGLQEDKKIRN | |
| 2 | Histone H3 | Cre06.g268350 | Structure | Nucleus | Histone H3 | 15 | TELLIRKLPFQRLV | [28] |
| 3 | IC2 | Cre12.g506000 | Structure | Flagella | Flagellar outer dynein arm Intermediate chain 2 | 64 | WDLRKMNECVENMPL | [33,42] |
| 4 | TUB-1 | Cre12.g542250 | Structure | Cytoskeleton | Beta tubulin 1 | 50 | QYQDATADEEGEYEDEEQQ | |
| 5 | FKBP12 | Cre13.g586300 | Signaling | Endoplasmic reticulum | Peptidyl-prolyl cis-trans isomerase FKBP-type | 12 | Expressed protein (Full length) | [43] |
| 6 | RCK1 | Cre06.g278222 | Signaling | Whole cell | Receptor of activated protein kinase C1 | 35 | Expressed protein (Full length) | [44] |
| 7 | D1 | NP_958377.1 | Photosynthesis | Chloroplast | Photosystem II protein D1 | 39 | IRETTENESANE | [26] |
| 8 | OEE2 | Cre12.g550850 | Photosynthesis | Chloroplast | Oxygen-evolving enhancer protein 2 of photosystem II | 30 | Expressed protein (Full length) | [45] |
| 9 | RBCL | NP_958405.1 | Photosynthesis | Chloroplast | Ribulose-1,5-bisphosphate carboxylase/oxygenase large subunit (chloroplast) | 53 | MRDDYVEKDRSR | [46] |
| 10 | RBCS2 | Cre02.g120150 | Photosynthesis | Chloroplast | Ribulose-15-bisphosphate carboxylase/oxygenase small subunit 2 | 21 | QRPKSARDWQPA | |
| 11 | RMT1 | Cre16.g661350 | Photosynthesis | Chloroplast | Rubisco large subunit N-methyltransferase | 52 | LRPSMTYSITPDQQ | |
| 12 | ATPs-6 | Cre01.g018800 | Metabolism | Mitochondria | Mitochondrial F1F0 ATP synthase subunit 6 | 35 | NMMAGHSSVKILSG | |
| 13 | ATPs-A | ACS16334 | Metabolism | Mitochondria | ATP synthase CF0 A subunit, partial (chloroplast) | 13 | AGLSKKGLSYFEKY | |
| 14 | ATPs-β | Cre17.g698000 | Metabolism | Mitochondria | ATP synthase CF1 beta subunit | 53 | FAGVGERTREGNDL | [22] |
| 15 | BCR1 | Cre08.g359350 | Metabolism | Chloroplast | Biotin carboxylase (ACCase complex) | 56 | EFVEICTDHGLEFIG | [47] |
| 16 | FAB2 | Cre17.g701700 | Metabolism | Chloroplast | Plastid acyl-ACP desaturase, d-9 stearate desaturase | 45 | DEGRHEIAYCKIMDG | [47] |
| 17 | GAP2 | Cre07.g354200 | Metabolism | Whole cell | Glyceraldehyde 3-phosphate dehydrogenase | 40 | DLVSTDFQGDNRSSIFDAKAGI | [48] |
| 18 | MPC1 | Cre03.g172300 | Metabolism | Integral component of membrane | Mitochondrial phosphate carrier protein | 37 | DVAKKTDSTKYAMP | |
| 19 | HSP70A | Cre08.g372100 | HSP | Whole cell | Heat shock protein 70A | 71 | EQVFSTYSDNQPGV/Expressed protein (N-terminal) | [19] |
| 20 | HSP90B | Cre17.g730950 | HSP | Whole cell | Heat shock protein 90B | 87 | GKDSKLKDLKESFK |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, J.; Huang, T.; Chai, S.; Guo, Y.; Wei, J.; Dou, S.; Li, L.; Liu, G. Identification of Reference and Biomarker Proteins in Chlamydomonas reinhardtii Cultured under Different Stress Conditions. Int. J. Mol. Sci. 2017, 18, 1822. https://doi.org/10.3390/ijms18081822
Shi J, Huang T, Chai S, Guo Y, Wei J, Dou S, Li L, Liu G. Identification of Reference and Biomarker Proteins in Chlamydomonas reinhardtii Cultured under Different Stress Conditions. International Journal of Molecular Sciences. 2017; 18(8):1822. https://doi.org/10.3390/ijms18081822
Chicago/Turabian StyleShi, Jianan, Teng Huang, Shuaijie Chai, Yalu Guo, Jian Wei, Shijuan Dou, Liyun Li, and Guozhen Liu. 2017. "Identification of Reference and Biomarker Proteins in Chlamydomonas reinhardtii Cultured under Different Stress Conditions" International Journal of Molecular Sciences 18, no. 8: 1822. https://doi.org/10.3390/ijms18081822
APA StyleShi, J., Huang, T., Chai, S., Guo, Y., Wei, J., Dou, S., Li, L., & Liu, G. (2017). Identification of Reference and Biomarker Proteins in Chlamydomonas reinhardtii Cultured under Different Stress Conditions. International Journal of Molecular Sciences, 18(8), 1822. https://doi.org/10.3390/ijms18081822




