Muscle Conditional Medium Reduces Intramuscular Adipocyte Differentiation and Lipid Accumulation through Regulating Insulin Signaling
Abstract
1. Introduction
2. Results
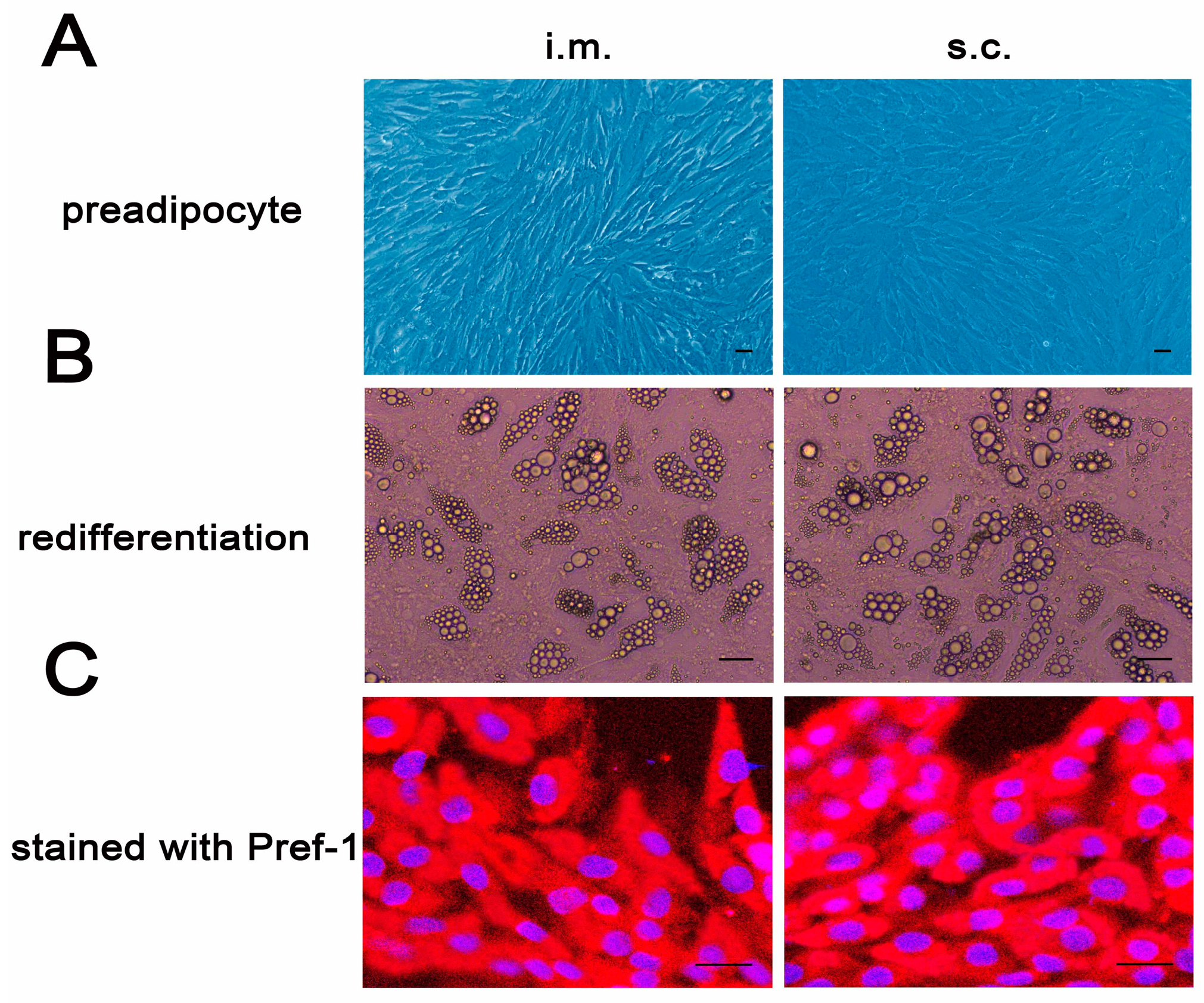
2.1. Cellular Differentiation and Identification of Porcine Intramuscular and Subcutaneous Preadipocytes
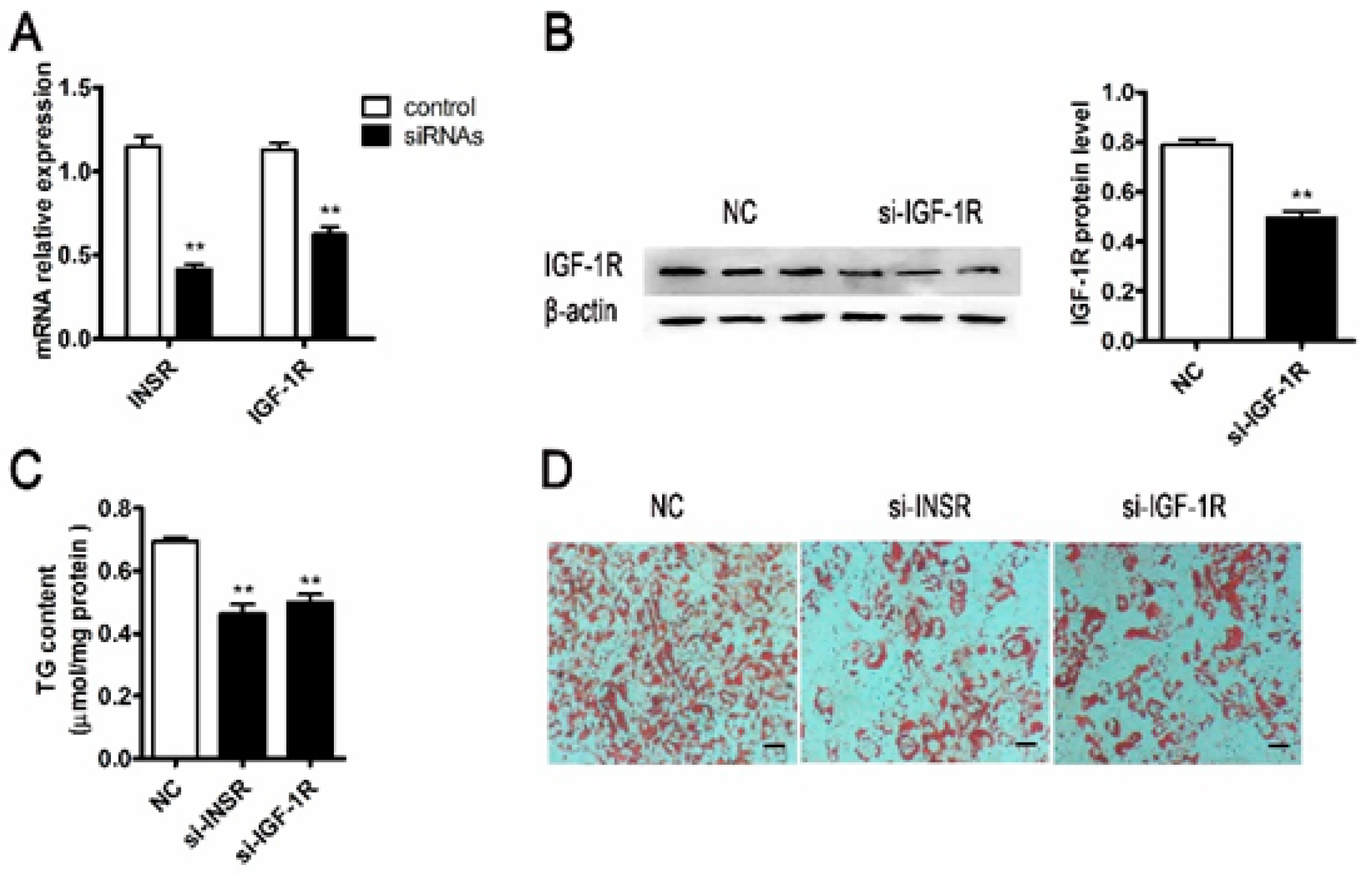
2.2. Inhibition Differentiation of Porcine Adipocyte by Downregulating the Expression of IGF-1R or INSR
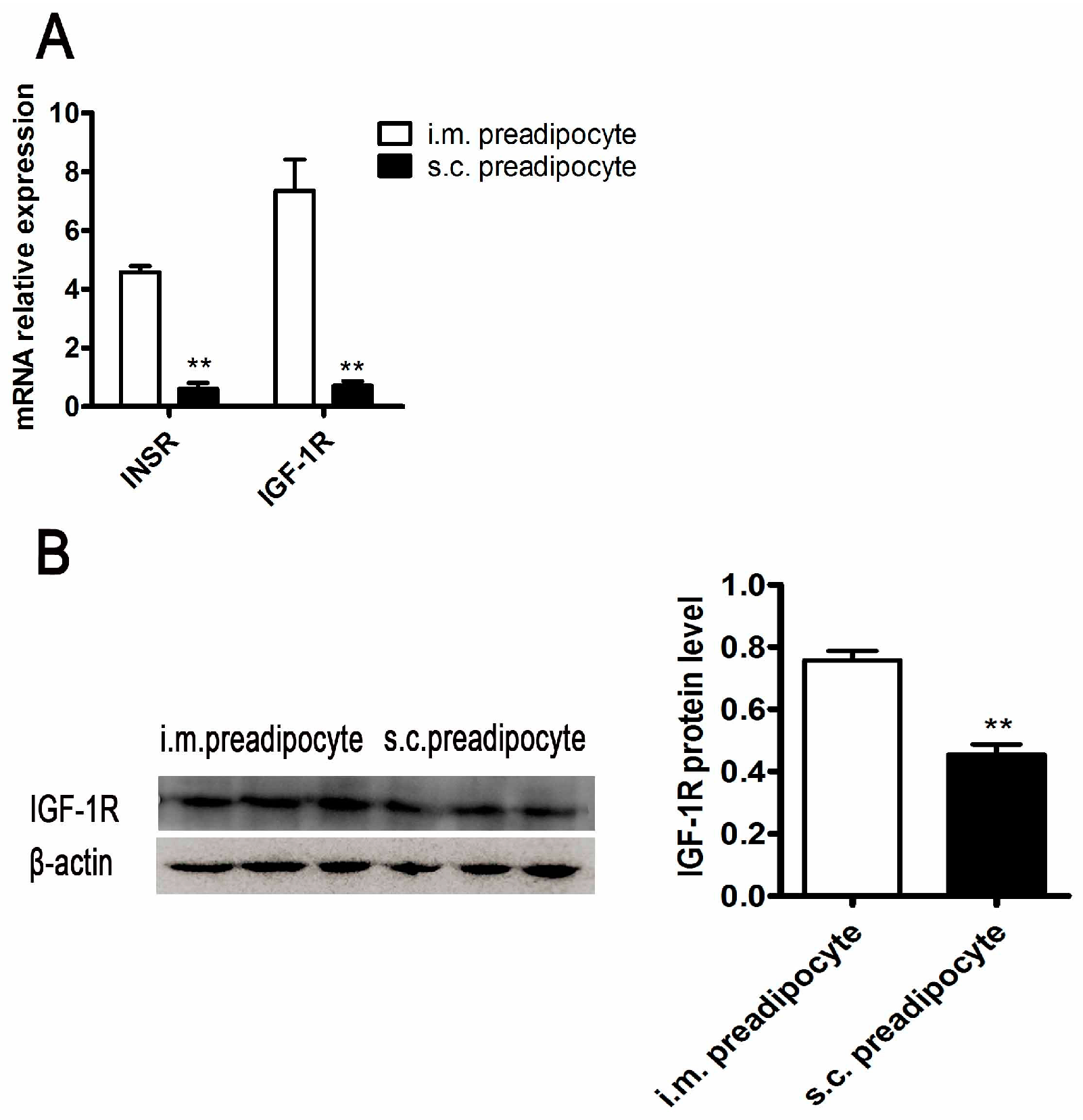
2.3. The Differences of Insulin Signaling between Porcine i.m. Preadipocytes and s.c. Preadipocytes
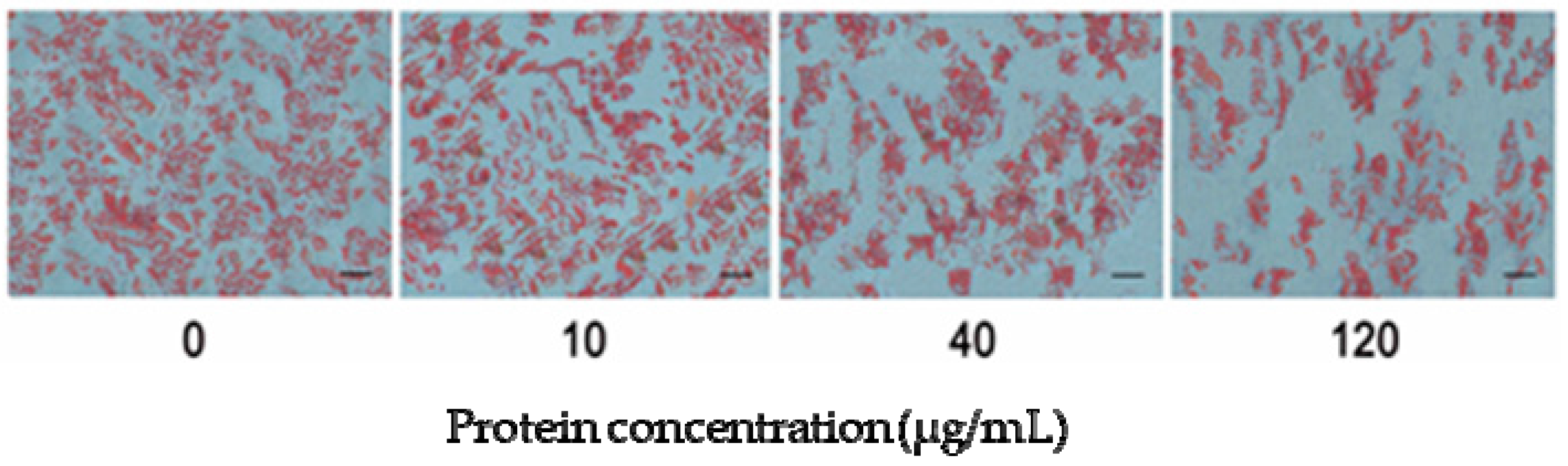
2.4. MCM Identification and Its Effect on Adipocyte Differentiation
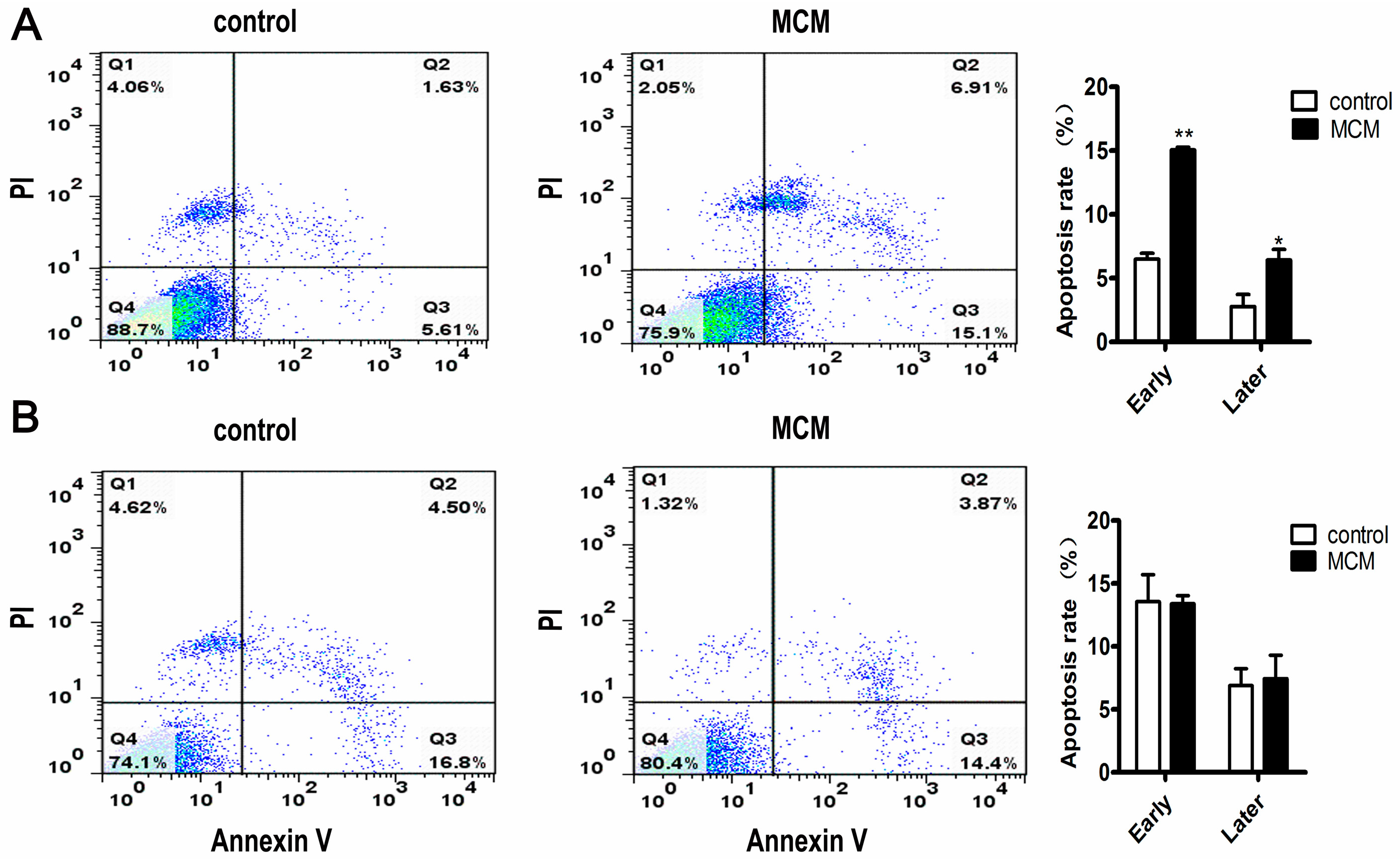
2.5. MCM Induced Apoptosis of Porcine Preadipocytes
2.6. MCM Suppresses Adipocyte Differentiation during the Pre-Differentiation to the Middle Stage
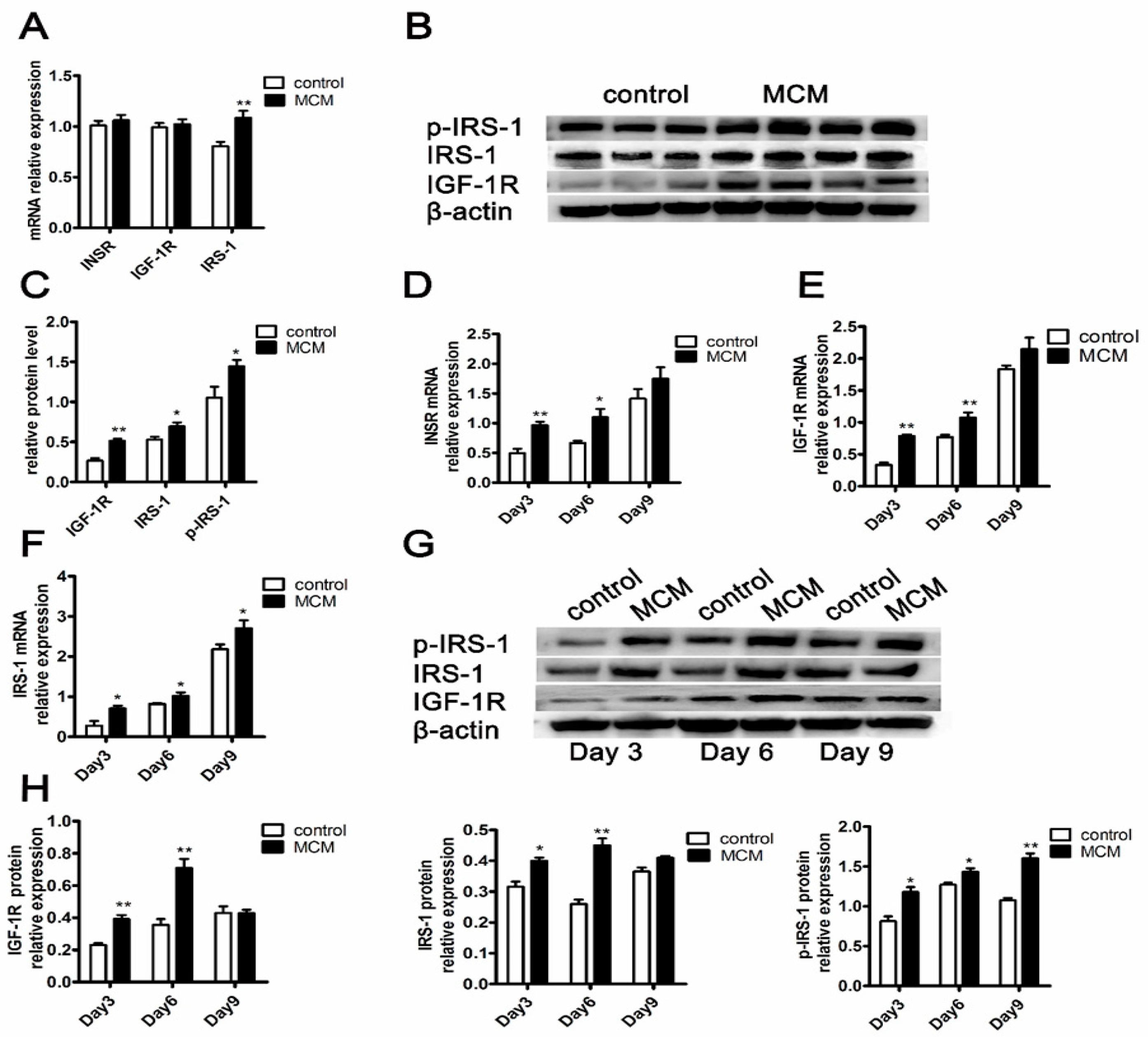
2.7. MCM Increases INSR and IGF-1R Expression and Activates Insulin Signaling
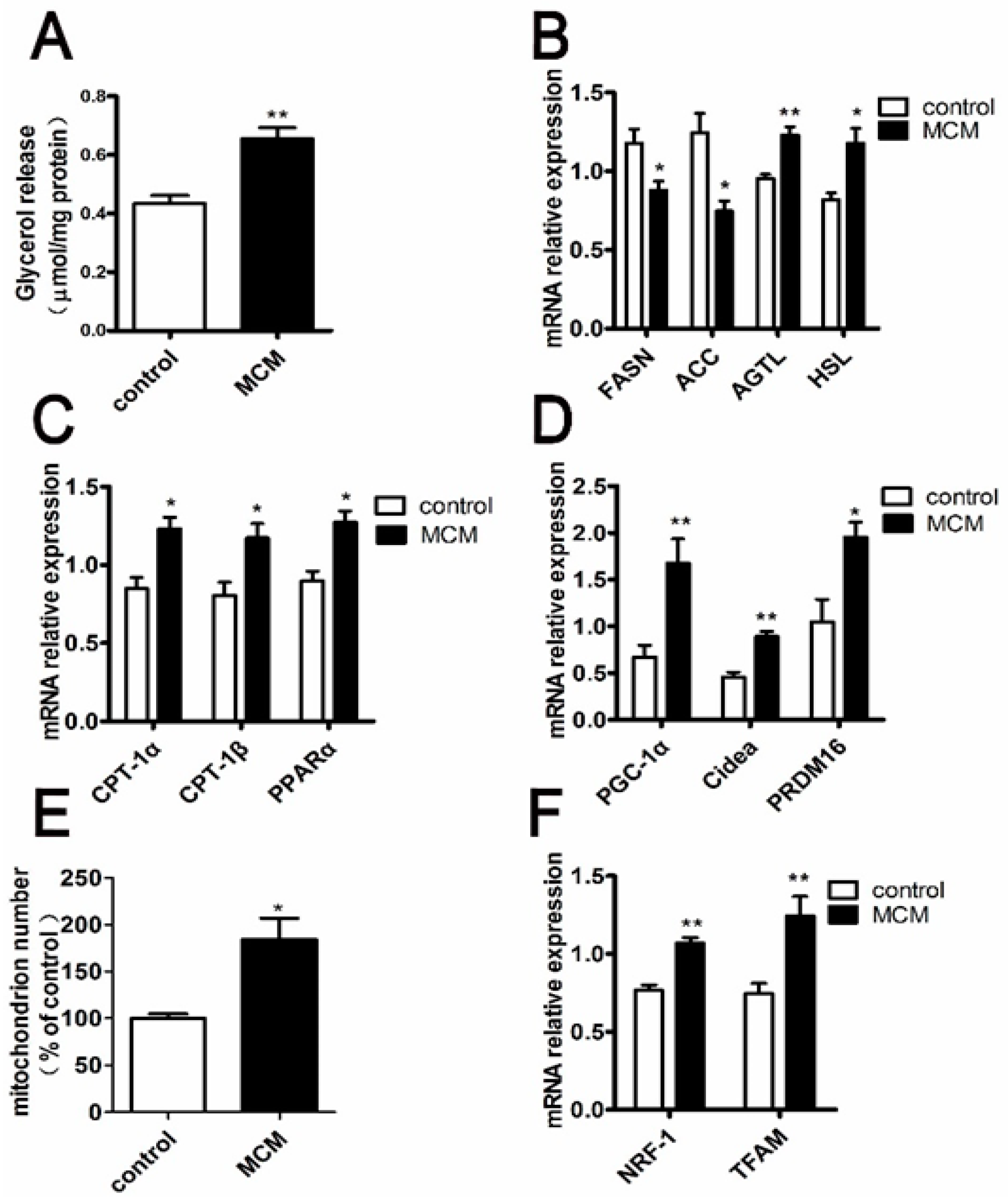
2.8. MCM Increases Adipocyte Lipolysis and Fatty Acid Oxidation
3. Discussion
4. Materials and Methods
4.1. Porcine Preadipocytes Isolation, Culture and Identification
4.2. Oil Red O Staining and Triglyceride Measurements
4.3. mRNA Expression Analysis
4.4. Isolation and Identification of Interstitial Fluid from Skeletal Muscle by a Centrifugation Method
4.5. siRNAs Trasfection
4.6. Western Blotting Analyses
4.7. Glycerol Release
4.8. Analysis of Mitochondrial DNA Content
4.9. Ethics Statement
4.10. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| INSR | Insulin receptor |
| IGF-1R | Insulin-like growth factor 1 receptor |
| MCM | Muscle conditional cultured medium |
| IMF | Intramuscular fat |
| DMEM | Dulbecco’s Modified Eagle’s medium |
| FBS | Fetal bovine serum |
| SDS-PAGE | Sodium dodecyl sulphate-polyacrylamide gel electrophoresis |
| PVDF | Polyvinylidene fluoride membranes |
| mtDNA | Mitochondrial DNA |
| COX2 | Cytochrome c oxidase subunit II |
| RPLP0 | Ribosomal protein lateral stalk subunit P0 |
| PI | Propidium iodine |
| NRF-1 | Nuclear respiratory factor-1 |
| TFAM | Mitochondrial transcription factor A |
| WAT | White adipose tissue |
| BAT | Brown adipose tissue |
References
- Gardan, D.; Gondret, F.; Louveau, I. Lipid metabolism and secretory function of porcine intramuscular adipocytes compared with subcutaneous and perirenal adipocytes. Am. J. Physiol. Endocrinol. Metab. 2006, 291, E372–E380. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.B.; Crouse, J.D. Relative contributions of acetate, lactate and glucose to lipogenesis in bovine, intramuscular and subcutaneous adipose tissue. J. Nutr. 1984, 114, 792–800. [Google Scholar] [PubMed]
- Lee, Y.B.; Kauffman, R.G. Cellularity and lipogenic enzyme activities of porcine intramuscular adipose tissue. J. Anim. Sci. 1974, 38, 538–544. [Google Scholar] [CrossRef] [PubMed]
- Spiegelman, B.M.; Flier, J.S. Adipogenesis and obesity: Rounding out the big picture. Cell 1996, 87, 377–389. [Google Scholar] [CrossRef]
- Suryawan, A.; Swanson, L.V.; Hu, C.Y. Insulin and hydrocortisone, but not triiodothyronine, are required for the differentiation of pig preadipocytes in primary culture. J. Anim. Sci. 1997, 75, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Gagnon, A.; Sorisky, A. The effect of glucose concentration on insulin-induced 3T3-L1 adipose cell differentiation. Obes. Res. 1998, 6, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Klemm, D.J.; Leitner, J.W.; Watson, P.; Nesterova, A.; Reusch, J.E.; Goalstone, M.L.; Draznin, B. Insulin-induced adipocyte differentiation. Activation of CREB rescues adipogenesis from the arrest caused by inhibition of prenylation. J. Biol. Chem. 2001, 276, 28430–28435. [Google Scholar] [CrossRef] [PubMed]
- Reed, B.C.; Lane, M.D. Insulin receptor synthesis and turnover in differentiating 3T3-L1 preadipocytes. Proc. Natl. Acad. Sci. USA 1980, 77, 285–289. [Google Scholar] [CrossRef] [PubMed]
- Klaman, L.D.; Boss, O.; Peroni, O.D.; Kim, J.K.; Martino, J.L.; Zabolotny, J.M.; Moghal, N.; Lubkin, M.; Kim, Y.B.; Sharpe, A.H.; et al. Increased energy expenditure, decreased adiposity, and tissue-specific insulin sensitivity in protein-tyrosine phosphatase 1B-deficient mice. Mol. Cell Biol. 2000, 20, 5479–5489. [Google Scholar] [CrossRef] [PubMed]
- Smith, P.J.; Wise, L.S.; Berkowitz, R.; Wan, C.; Rubin, C.S. Insulin-like growth factor-I is an essential regulator of the differentiation of 3T3-L1 adipocytes. J. Biol. Chem. 1988, 263, 9402–9408. [Google Scholar] [PubMed]
- Miki, H.; Yamauchi, T.; Suzuki, R.; Komeda, K.; Tsuchida, A.; Kubota, N.; Terauchi, Y.; Kamon, J.; Kaburagi, Y.; Matsui, J.; et al. Essential role of insulin receptor substrate 1 (IRS-1) and IRS-2 in adipocyte differentiation. Mol. Cell Biol. 2001, 21, 2521–2532. [Google Scholar] [CrossRef] [PubMed]
- Myers, M.G., Jr.; White, M.F. New frontiers in insulin receptor substrate signaling. Trends Endocrinol. Metab. 1995, 6, 209–215. [Google Scholar] [CrossRef]
- Accili, D.; Taylor, S.I. Targeted inactivation of the insulin receptor gene in mouse 3T3-L1 fibroblasts via homologous recombination. Proc. Natl. Acad. Sci. USA 1991, 88, 4708–4712. [Google Scholar] [CrossRef] [PubMed]
- Hirai, S.; Matsumoto, H.; Hino, N.; Kawachi, H.; Matsui, T.; Yano, H. Myostatin inhibits differentiation of bovine preadipocyte. Domest. Anim. Endocrinol. 2007, 32, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Quinn, L.S. Interleukin-15: A muscle-derived cytokine regulating fat-to-lean body composition. J. Anim. Sci. 2008, 86, E75–E83. [Google Scholar] [CrossRef] [PubMed]
- Huh, J.Y.; Dincer, F.; Mesfum, E.; Mantzoros, C.S. Irisin stimulates muscle growth-related genes and regulates adipocyte differentiation and metabolism in humans. Int. J. Obes. 2014, 38, 1538–1544. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Li, D.; Yin, J.; Ni, J.; Dong, B.; Zhang, J.; Du, M. CLA differently regulates adipogenesis in stromal vascular cells from porcine subcutaneous adipose and skeletal muscle. J. Lipid Res. 2007, 48, 1701–1709. [Google Scholar] [CrossRef] [PubMed]
- Shan, T.; Liu, W.; Kuang, S. Fatty acid binding protein 4 expression marks a population of adipocyte progenitors in white and brown adipose tissues. FASEB J. 2013, 27, 277–287. [Google Scholar] [CrossRef] [PubMed]
- Wood, J.D.; Enser, M.; Fisher, A.V.; Nute, G.R.; Richardson, R.I.; Sheard, P.R. Manipulating meat quality and composition. Proc. Nutr. Soc. 1999, 58, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Zhou, G.; Shu, G.; Wang, L.; Zhu, X.; Gao, P.; Xi, Q.; Zhang, Y.; Yuan, L.; Jiang, Q. Glucose utilization, lipid metabolism and BMP-Smad signaling pathway of porcine intramuscular preadipocytes compared with subcutaneous preadipocytes. Cell Physiol. Biochem. 2013, 31, 981–996. [Google Scholar] [CrossRef] [PubMed]
- Wan, R.; Du, J.; Ren, L.; Meng, Q. Selective adipogenic effects of propionate on bovine intramuscular and subcutaneous preadipocytes. Meat Sci. 2009, 82, 372–378. [Google Scholar] [CrossRef] [PubMed]
- Nobusue, H.; Kano, K. Establishment and characteristics of porcine preadipocyte cell lines derived from mature adipocytes. J. Cell Biochem. 2010, 109, 542–552. [Google Scholar] [CrossRef] [PubMed]
- Ullrich, A.; Schlessinger, J. Signal transduction by receptors with tyrosine kinase activity. Cell 1990, 61, 203–212. [Google Scholar] [CrossRef]
- Entingh-Pearsall, A.; Kahn, C.R. Differential roles of the insulin and insulin-like growth factor-I (IGF-I) receptors in response to insulin and IGF-I. J. Biol. Chem. 2004, 279, 38016–38024. [Google Scholar] [CrossRef] [PubMed]
- Bluher, S.; Kratzsch, J.; Kiess, W. Insulin-like growth factor I, growth hormone and insulin in white adipose tissue. Best. Pract. Res. Clin. Endocrinol. Metab. 2005, 19, 577–587. [Google Scholar] [CrossRef] [PubMed]
- Boucher, J.; Mori, M.A.; Lee, K.Y.; Smyth, G.; Liew, C.W.; Macotela, Y.; Rourk, M.; Bluher, M.; Russell, S.J.; Kahn, C.R. Impaired thermogenesis and adipose tissue development in mice with fat-specific disruption of insulin and IGF-1 signalling. Nat. Commun. 2012, 3, 902. [Google Scholar] [CrossRef] [PubMed]
- Bluher, M.; Kahn, B.B.; Kahn, C.R. Extended longevity in mice lacking the insulin receptor in adipose tissue. Science 2003, 299, 572–574. [Google Scholar] [CrossRef] [PubMed]
- Avram, A.S.; Avram, M.M.; James, W.D. Subcutaneous fat in normal and diseased states: 2. Anatomy and physiology of white and brown adipose tissue. J. Am. Acad. Dermatol. 2005, 53, 671–683. [Google Scholar] [CrossRef] [PubMed]
- Avram, M.M.; Avram, A.S.; James, W.D. Subcutaneous fat in normal and diseased states 3. Adipogenesis: From stem cell to fat cell. J. Am. Acad. Dermatol. 2007, 56, 472–492. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, B.K.; Febbraio, M.A. Muscle as an endocrine organ: Focus on muscle-derived interleukin-6. Physiol. Rev. 2008, 88, 1379–1406. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, B.K.; Steensberg, A.; Fischer, C.; Keller, C.; Keller, P.; Plomgaard, P.; Febbraio, M.; Saltin, B. Searching for the exercise factor: Is IL-6 a candidate? J. Muscle Res. Cell Motil. 2003, 24, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, B.K.; Akerström, T.C.; Nielsen, A.R.; Fischer, C.P. Role of myokines in exercise and metabolism. J. Appl. Physiol. 2007, 103, 1093–1098. [Google Scholar] [CrossRef] [PubMed]
- Halberg, N.; Wernstedt-Asterholm, I.; Scherer, P.E. The adipocyte as an endocrine cell. Endocrinol. Metab. Clin. N. Am. 2008, 37, 753–768. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, B.K.; Febbraio, M.A. Muscles, exercise and obesity: Skeletal muscle as a secretory organ. Nat. Rev. Endocrinol. 2012, 8, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Dietze, D.; Koenen, M.; Röhrig, K.; Horikoshi, H.; Hauner, H.; Eckel, J. Impairment of insulin signaling in human skeletal muscle cells by co-culture with human adipocytes. Diabetes 2002, 51, 2369–2376. [Google Scholar] [CrossRef] [PubMed]
- Chu, W.; Wei, W.; Yu, S.; Han, H.; Shi, X.; Sun, W.; Gao, Y.; Zhang, L.; Chen, J. C2C12 myotubes inhibit the proliferation and differentiation of 3T3-L1 preadipocytes by reducing the expression of glucocorticoid receptor gene. Biochem. Biophys. Res. Commun. 2016, 472, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Loncar, D. Convertible adipose tissue in mice. Cell Tissue Res. 1991, 266, 149–161. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Cohen, P.; Spiegelman, B.M. Adaptive thermogenesis in adipocytes: Is beige the new brown? Genes Dev. 2013, 27, 234–250. [Google Scholar] [CrossRef] [PubMed]
- Cohen, P.; Levy, J.D.; Zhang, Y.; Frontini, A.; Kolodin, D.P.; Svensson, K.J.; Lo, J.C.; Zeng, X.; Ye, L.; Khandekar, M.J.; et al. Ablation of PRDM16 and beige adipose causes metabolic dysfunction and a subcutaneous to visceral fat switch. Cell 2014, 156, 304–316. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Pan, R.; Pfeifer, A. Regulation of brown and beige fat by microRNAs. Pharmacol. Ther. 2017, 170, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, R.; Meng, Y.; Li, S.; Donelan, W.; Zhao, Y.; Qi, L.; Zhang, M.; Wang, X.; Cui, T.; et al. Irisin stimulates browning of white adipocytes through mitogen-activated protein kinase p38 MAP kinase and ERK MAP kinase signaling. Diabetes 2014, 63, 514–525. [Google Scholar] [CrossRef] [PubMed]
- Wiig, H.; Aukland, K.; Tenstad, O. Isolation of interstitial fluid from rat mammary tumors by a centrifugation method. Am. J. Physiol. Heart Circ. Physiol. 2003, 284, H416–H424. [Google Scholar] [CrossRef] [PubMed]
- Reiling, E.; Ling, C.; Uitterlinden, A.G.; Van’t, R.E.; Welschen, L.M.; Ladenvall, C.; Almgren, P.; Lyssenko, V.; Nijpels, G.; van Hove, E.C.; et al. The association of mitochondrial content with prevalent and incident type 2 diabetes. J. Clin. Endocrinol. Metab. 2010, 95, 1909–1915. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Galilea, M.; Pérez-Matute, P.; Prieto-Hontoria, P.L.; Houssier, M.; Burrell, M.A.; Langin, D.; Martínez, J.A.; Moreno-Aliaga, M.J. α-Lipoic acid treatment increases mitochondrial biogenesis and promotes beige adipose features in subcutaneous adipocytes from overweight/obese subjects. Biochim. Biophys. Acta 2015, 1851, 273–281. [Google Scholar] [CrossRef] [PubMed]

| Gene | Primer Sequence (5′–3′) | Product Size (bp) | GeneBank No. |
|---|---|---|---|
| INSR | F: GTGTTGTGATTGGAAGCATTTAT | 108 | XM_021083940.1 |
| R: CATCGCTGGCACTGAGGTA | |||
| FABP4 | F: GCCAAACCCAACCTGATCAT | 188 | NM_001002817.1 |
| R: TCCCACTTCTGCACCTGTAC | |||
| PPARγ | F: TTGCTGTGAAGTTCAACGCA | 167 | NM_214379.1 |
| R: GTGGTTCAACTTGAGCTGCA | |||
| IGF-1R | F: CAACCTCCGGCCTTTTACTTT | 134 | NM_214172.1 |
| R: CAGGAATGTCATCTGCTCCTTCT | |||
| RPLP0 | F: TCCAGGCTTTAGGCATCACC | 95 | NM_001098598.1 |
| R: GGCTCCCACTTTGTCTCCAG | |||
| FASN | F: GCTTGTCCTGGGAAGAGTGTA | 114 | NM_001099930.1 |
| R: AGGAACTCGGACATAGCGG | |||
| ACC | F: CGCTTCATAATTGGTTCTGTG | 147 | NM_001114269.1 |
| R: GCTAGAAATCCCCAAGTCAGA | |||
| ATGL | F: GCGAAAATGTCATCATAACC | 175 | NM_001098605.1 |
| R: ATGGTGCTCTTGAGTTCGT | |||
| HSL | F: GCCCGAGACGAGATTAG | 143 | NM_214315.2 |
| R: ATGAAGGGATTCTTGACG | |||
| CPT-1α | F: AACCTTCTGGCGGACGACG | 213 | NM_001129805.1 |
| R: GCAGGAACGCACGGTCTCA | |||
| CPT-1β | F:ACTGTCTGGGCAAACCAAAC | 176 | NM_001007191.1 |
| R: CTTCTTGATGAGGCCTTTGC | |||
| PPARα | F:CAGCCTCCAGCCCCTCGTC | 382 | NM_001044526.1 |
| R: GCGGTCTCGGCATCTTCTAGG | |||
| PGC-1α | F: CTGTGGATGAAGACGGATTG | 92 | NM_213963.2 |
| R: GTCAGGCATGGAGGAAGGA | |||
| Cidea | F: TTCCGAGTTTCCAACCACAA | 97 | NM_001112696 |
| R: CGATAACCAGGGCATCCAG | |||
| PRDM16 | F: CCACAAGTCCTACACGCAGTTCTC | 121 | XM_021095209.1 |
| R: GTTGAGGGACGAGGTAGTGCTGA | |||
| TFAM | F:GGTCCATCACAGGTAAAGCTGAA | 167 | NM_001130211.1 |
| R:ATAAGATCGTTTCGCCCAACTTC | |||
| NRF-1 | F: CAACAGGAAAGAAACGGAAAC | 157 | XM_021078993.1 |
| R: GAGGGTGAGATACAAAGGACAAT |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, H.; Wei, W.; Chu, W.; Liu, K.; Tian, Y.; Jiang, Z.; Chen, J. Muscle Conditional Medium Reduces Intramuscular Adipocyte Differentiation and Lipid Accumulation through Regulating Insulin Signaling. Int. J. Mol. Sci. 2017, 18, 1799. https://doi.org/10.3390/ijms18081799
Han H, Wei W, Chu W, Liu K, Tian Y, Jiang Z, Chen J. Muscle Conditional Medium Reduces Intramuscular Adipocyte Differentiation and Lipid Accumulation through Regulating Insulin Signaling. International Journal of Molecular Sciences. 2017; 18(8):1799. https://doi.org/10.3390/ijms18081799
Chicago/Turabian StyleHan, Haiyin, Wei Wei, Weiwei Chu, Kaiqing Liu, Ye Tian, Zaohang Jiang, and Jie Chen. 2017. "Muscle Conditional Medium Reduces Intramuscular Adipocyte Differentiation and Lipid Accumulation through Regulating Insulin Signaling" International Journal of Molecular Sciences 18, no. 8: 1799. https://doi.org/10.3390/ijms18081799
APA StyleHan, H., Wei, W., Chu, W., Liu, K., Tian, Y., Jiang, Z., & Chen, J. (2017). Muscle Conditional Medium Reduces Intramuscular Adipocyte Differentiation and Lipid Accumulation through Regulating Insulin Signaling. International Journal of Molecular Sciences, 18(8), 1799. https://doi.org/10.3390/ijms18081799




