In Situ β-Glucan Fortification of Cereal-Based Matrices by Pediococcus parvulus 2.6: Technological Aspects and Prebiotic Potential
Abstract
1. Introduction
2. Results
2.1. Production of Cereal-Based Fermented Foods
2.2. Formulation of Probiotic Cereal-Based Fermented Substrates
2.3. Prebiotic Potential of the P. parvulus 2.6 β-Glucans in Probiotic Cereal-Based Fermented Products
2.4. Tolerance to Simulated Gastrointestinal Conditions Using Cereal Based Fermented Products as Carrier Matrix
3. Discussion
4. Materials and Methods
4.1. Microbial Strains and Growth Conditions
4.2. Fermented Cereal-Based Foods
4.3. Inhibition Assay of the Barley Matrix
4.4. Viscosity of Cereal-Based Products Fermented by P. parvulus 2.6
4.5. Quantification of the β-Glucan Produced by the P. parvulus 2.6 Strain
4.6. Formulation of Probiotic Cereal-Based Fermented Foods
4.7. Prebiotic Potential of Bacterial β-Glucans in Cereal-Based Fermented Foods
4.8. Tolerance to Gastrointestinal Conditions Using Cereal-Based Fermented Products as Carrier Matrix
4.9. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| EPS | Exopolysaccharides |
| LAB | Lactic acid bacteria |
References
- Kleerebezem, M.; Hols, P.; Bernard, E.; Rolain, T.; Zhou, M.; Siezen, R.J.; Bron, P.A. The extracellular biology of the lactobacilli. FEMS Microbiol. Rev. 2010, 34, 199–230. [Google Scholar] [CrossRef] [PubMed]
- Werning, M.L.; Notararigo, S.; Nácher-Vázquez, M.; Fernández de Palencia, P.; Aznar, R.; López, P. Biosynthesis, purification and biotechnological use of exopolysacchardes produced by lactic acid bacteria. In Food Additives; El-Samragy, Y., Ed.; Intech: Rijeka, Croatia, 2012; pp. 83–114. ISBN 978-953-51-0067-6. [Google Scholar]
- Arena, M.P.; Russo, P.; Fiocco, D.; Capozzi, V.; Spano, G. β-glucans and synbiotic foods. In Probiotics, Prebiotics, and Synbiotics: Bioactive Foods in Health Promotion, 1st ed.; Watson, R.R., Preedy, V.R., Eds.; Academic Press: London, UK, 2016; pp. 432–433. ISBN 978-0-12-802189-7. [Google Scholar]
- Pérez-Ramos, A.; Nácher-Vázquez, M.; Notararigo, S.; López, P.; Mohedano, M.L. Current and future applications of bacterial extracellular polysaccharides. a2—Watson, ronald ross. In Probiotics, Prebiotics, and Synbiotics: Bioactive Foods in Health Promotion, 1st ed.; Watson, R.R., Preedy, V.R., Eds.; Academic Press: London, UK, 2016; pp. 329–344. ISBN 978-0-12-802189-7. [Google Scholar]
- Leroy, F.; de Vuyst, L. Functional lactic acid bacteria starter cultures for the food fermentation industry. Trends Food Sci. Technol. 2004, 15, 67–78. [Google Scholar] [CrossRef]
- Marco, M.L.; Heeney, D.; Binda, S.; Cifelli, C.J.; Cotter, P.D.; Foligné, B.; Gänzle, M.; Kort, R.; Pasin, G.; Pihlanto, A.; et al. Health benefits of fermented foods: Microbiota and beyond. Curr. Opin. Biotechnol. 2017, 44, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Badel, S.; Bernardi, T.; Michaud, P. New perspectives for lactobacilli exopolysaccharides. Biotechnol. Adv. 2011, 29, 54–66. [Google Scholar] [CrossRef] [PubMed]
- Zannini, E.; Waters, D.M.; Coffey, A.; Arendt, E.K. Production, properties, and industrial food application of lactic acid bacteria-derived exopolysaccharides. Appl. Microbiol. Biotechnol. 2016, 100, 1121–1135. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Li, W.; Chen, X.; Feng, M.; Rui, X.; Jiang, M.; Dong, M. Microbiological, physicochemical and rheological properties of fermented soymilk produced with exopolysaccharide (EPS) producing lactic acid bacteria strains. Food Sci. Technol. 2014, 57, 477–485. [Google Scholar] [CrossRef]
- Hickisch, A.; Beer, R.; Vogel, R.F.; Toelstede, S. Influence of lupin-based milk alternative heat treatment and exopolysaccharide-producing lactic acid bacteria on the physical characteristics of lupin-based yogurt alternatives. Food Res. Int. 2016, 84, 180–188. [Google Scholar] [CrossRef]
- Lynch, K.M.; Coffey, A.; Arendt, E.K. Exopolysaccharide producing lactic acid bacteria: Their techno-functional role and potential application in gluten-free bread products. Food Res. Int. 2017. [Google Scholar] [CrossRef]
- Ruas-Madiedo, P.; Gueimonde, M.; Margolles, A.; de los Reyes-Gavilan, C.G.; Salminen, S. Exopolysaccharides produced by probiotic strains modify the adhesion of probiotics and enteropathogens to human intestinal mucus. J. Food Prot. 2006, 69, 2011–2015. [Google Scholar] [CrossRef] [PubMed]
- Nácher-Vázquez, M.; Iturria, I.; Zarour, K.; Mohedano, M.L.; Aznar, R.; Pardo, M.A.; López, P. Dextran production by Lactobacillus sakei MN1 coincides with reduced autoagglutination, biofilm formation and epithelial cell adhesion. Carbohydr. Polym. 2017, 168, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Salazar, N.; Gueimonde, M.; de Los Reyes-Gavilan, C.G.; Ruas-Madiedo, P. Exopolysaccharides produced by lactic acid bacteria and bifidobacteria as fermentable substrates by the intestinal microbiota. Crit. Rev. Food Sci. Nutr. 2016, 56, 1440–1453. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Li, W.; Rui, X.; Chen, X.; Jiang, M.; Dong, M. Characterization of a novel exopolysaccharide with antitumor activity from Lactobacillus plantarum 70810. Int. J. Biol. Macromol. 2014, 63, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Surayot, U.; Wang, J.; Seesuriyachan, P.; Kuntiya, A.; Tabarsa, M.; Lee, Y.; Kim, J.-K.; Park, W.; You, S. Exopolysaccharides from lactic acid bacteria: Structural analysis, molecular weight effect on immunomodulation. Int. J. Biol. Macromol. 2014, 68, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Nácher-Vázquez, M.; Ballesteros, N.; Canales, A.; Rodríguez Saint-Jean, S.; Pérez-Prieto, S.I.; Prieto, A.; Aznar, R.; López, P. Dextrans produced by lactic acid bacteria exhibit antiviral and immunomodulatory activity against salmonid viruses. Carbohydr. Polym. 2015, 124, 292–301. [Google Scholar] [CrossRef] [PubMed]
- Ryan, P.M.; Ross, R.P.; Fitzgerald, G.F.; Caplice, N.M.; Stanton, C. Sugar-coated: Exopolysaccharide producing lactic acid bacteria for food and human health applications. Food Funct. 2015, 6, 679–693. [Google Scholar] [CrossRef] [PubMed]
- Caggianiello, G.; Kleerebezem, M.; Spano, G. Exopolysaccharides produced by lactic acid bacteria: From health-promoting benefits to stress tolerance mechanisms. Appl. Microbiol. Biotechnol. 2016, 100, 3877–3886. [Google Scholar] [CrossRef] [PubMed]
- Fernández, K.; Dueñas, M.T.; Irastorza, A.; Bilbao, A.; del Campo, G. Characterization and DNA plasmid analysis of ropy Pediococcus damnosus spp. strains isolated from Basque country ciders. J. Food Prot. 1995, 59, 35–40. [Google Scholar] [CrossRef]
- Dols-Lafargue, M.; Lee, H.Y.; Le Marrec, C.; Heyraud, A.; Chambat, G.; Lonvaud-Funel, A. Characterization of gtf, a glucosyltransferase gene in the genomes of Pediococcus parvulus and Oenococcus oeni, two bacterial species commonly found in wine. Appl. Environ. Microbiol. 2008, 74, 4079–4090. [Google Scholar] [CrossRef] [PubMed]
- Werning, M.L.; Ibarburu, I.; Dueñas, M.T.; Irastorza, A.; Navas, J.; López, P. Pediococcus parvulus gtf gene encoding the gtf glycosyltransferase and its application for specific PCR detection of β-d-glucan-producing bacteria in foods and beverages. J. Food Prot. 2006, 69, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Werning, M.L.; Corrales, M.A.; Prieto, A.; Fernández de Palencia, P.; Navas, J.; Lopez, P. Heterologous expression of a position 2-substituted (1→3)-β-d-glucan in Lactococcus lactis. Appl. Environ. Microbiol. 2008, 74, 5259–5262. [Google Scholar] [CrossRef] [PubMed]
- Werning, M.L.; Pérez-Ramos, A.; Fernández de Palencia, P.; Mohedano, M.L.; Dueñas, M.T.; Prieto, A.; López, P. A specific immunological method to detect and quantify bacterial 2-substituted (1,3)-β-d-glucan. Carbohydr. Polym. 2014, 113, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Garai-Ibabe, G.; Dueñas, M.T.; Irastorza, A.; Sierra-Filardi, E.; Werning, M.L.; López, P.; Corbi, A.L.; Fernández de Palencia, P. Naturally occurring 2-substituted (1,3)-β-d-glucan producing Lactobacillus suebicus and Pediococcus parvulus strains with potential utility in the production of functional foods. Bioresour. Technol. 2010, 101, 9254–9263. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Ramos, A.; Mohedano, M.L.; Puertas, A.; Lamontanara, A.; Orru, L.; Spano, G.; Capozzi, V.; Dueñas, M.T.; López, P. Draft genome sequence of Pediococcus parvulus 2.6, a probiotic β-glucan producer strain. Genome Announc. 2016, 4, e01381-16. [Google Scholar] [CrossRef] [PubMed]
- Lambo-Fodje, A.M.; Leeman, M.; Wahlund, K.G.; Nyman, M.; Öste, R.; Larsson, H. Molar mass and rheological characterisation of an exopolysaccharide from Pediococcus damnosus 2.6. Carbohydr. Polym. 2007, 68, 577–586. [Google Scholar] [CrossRef]
- Velasco, S.E.; Areizaga, J.; Irastorza, A.; Dueñas, M.T.; Santamaria, A.; Muñoz, M.E. Chemical and rheological properties of the β-glucan produced by Pediococcus parvulus 2.6. J. Agric. Food Chem. 2009, 57, 1827–1834. [Google Scholar] [CrossRef] [PubMed]
- Fernández de Palencia, P.; Werning, M.L.; Sierra-Filardi, E.; Dueñas, M.T.; Irastorza, A.; Corbi, A.L.; López, P. Probiotic properties of the 2-substituted (1,3)-β-d-glucan-producing bacterium Pediococcus parvulus 2.6. Appl. Environ. Microbiol. 2009, 75, 4887–4891. [Google Scholar] [CrossRef] [PubMed]
- Immerstrand, T.; Paul, C.J.; Rosenquist, A.; Deraz, S.; Mårtensson, O.B.; Ljungh, A.; Blucher, A.; Öste, R.; Holst, O.; Karlsson, E.N. Characterization of the properties of Pediococcus parvulus for probiotic or protective culture use. J. Food Prot. 2010, 73, 960–966. [Google Scholar] [CrossRef] [PubMed]
- Notararigo, S.; de Las Casas-Engel, M.; Fernández de Palencia, P.; Corbi, A.L.; López, P. Immunomodulation of human macrophages and myeloid cells by 2-substituted (1-3)-β-d-glucan from P. parvulus 2.6. Carbohydr. Polym. 2014, 112, 109–113. [Google Scholar] [CrossRef] [PubMed]
- Russo, P.; López, P.; Capozzi, V.; Fernández de Palencia, P.; Dueñas, M.T.; Spano, G.; Fiocco, D. β-glucans improve growth, viability and colonization of probiotic microorganisms. Int. J. Mol. Sci. 2012, 13, 6026–6039. [Google Scholar] [CrossRef] [PubMed]
- Mårtensson, O.; Biörklund, M.; Lambo, A.M.; Dueñas-Chasco, M.; Irastorza, A.; Holst, O.; Norin, E.; Welling, G.; Öste, R.; Önning, G. Fermented, ropy, oat-based products reduce cholesterol levels and stimulate the bifidobacteria flora in humans. Nutr. Res. 2005, 25, 429–442. [Google Scholar] [CrossRef]
- Lindström, C.; Holst, O.; Nilsson, L.; Öste, R.; Andersson, K.E. Effects of Pediococcus parvulus 2.6 and its exopolysaccharide on plasma cholesterol levels and inflammatory markers in mice. AMB Express 2012, 2, 66. [Google Scholar] [CrossRef] [PubMed]
- Lindström, C.; Xu, J.; Öste, R.; Holst, O.; Molin, G. Oral administration of live exopolysaccharide-producing Pediococcus parvulus, but not purified exopolysaccharide, suppressed enterobacteriaceae without affecting bacterial diversity in ceca of mice. Appl. Environ. Microbiol. 2013, 79, 5030–5037. [Google Scholar] [CrossRef] [PubMed]
- London, L.E.; Kumar, A.H.; Wall, R.; Casey, P.G.; O’Sullivan, O.; Shanahan, F.; Hill, C.; Cotter, P.D.; Fitzgerald, G.F.; Ross, R.P.; et al. Exopolysaccharide-producing probiotic lactobacilli reduce serum cholesterol and modify enteric microbiota in apoe-deficient mice. J. Nutr. 2014, 144, 1956–1962. [Google Scholar] [CrossRef] [PubMed]
- Mårtensson, O.; Staaf, J.; Dueñas-Chasco, M.; Irastorza, A.; Öste, R.; Holst, O. A fermented, ropy, non-dairy oat product based on the exopolysaccharide-producing strain Pediococcus damnosus. Adv. Food Sci. 2002, 24, 4–11. [Google Scholar]
- Elizaquível, P.; Sánchez, G.; Salvador, A.; Fiszman, S.; Dueñas, M.T.; López, P.; Fernández de Palencia, P.; Aznar, R. Evaluation of yogurt and various beverages as carriers of lactic acid bacteria producing 2-branched (1,3)-β-d-glucan. J. Dairy Sci. 2011, 94, 3271–3278. [Google Scholar] [CrossRef] [PubMed]
- Russo, P.; de Chiara, M.L.V.; Capozzi, V.; Arena, M.P.; Amodio, M.L.; Rascón, A.; Dueñas, M.T.; López, P.; Spano, G. Lactobacillus plantarum strains for multifunctional oat-based foods. LWT-Food Sci. Technol. 2016, 68, 288–294. [Google Scholar] [CrossRef]
- De Vries, M.C.; Vaughan, E.E.; Kleerebezem, M.; de Vos, W.M. Lactobacillus plantarum—Survival, functional and potential probiotic properties in the human intestinal tract. Int. Dairy J. 2006, 16, 1018–1028. [Google Scholar] [CrossRef]
- Bove, P.; Gallone, A.; Russo, P.; Capozzi, V.; Albenzio, M.; Spano, G.; Fiocco, D. Probiotic features of Lactobacillus plantarum mutant strains. Appl. Microbiol. Biotechnol. 2012, 96, 431–441. [Google Scholar] [CrossRef] [PubMed]
- Waters, D.M.; Mauch, A.; Coffey, A.; Arendt, E.K.; Zannini, E. Lactic acid bacteria as a cell factory for the delivery of functional biomolecules and ingredients in cereal-based beverages: A review. Crit. Rev. Food Sci. Nutr. 2015, 55, 503–520. [Google Scholar] [CrossRef] [PubMed]
- Gupta, M.; Bajaj, B.K. Selection criteria for probiotics and potential of cereal based food products as novel probiotic-carriers. Curr. Nutr. Food Sci. 2016, 12, 157–174. [Google Scholar] [CrossRef]
- Mårtensson, O.; Jönsson, C.; Dueñas-Chasco, M.; Irastorza, A.; Öste, R.; Holst, O. Growth and exopolysaccharide formation by Pediococcus damnosus 2.6 in β-glucan suspensions of oat and barley. LWT-Food Sci. Technol. 2005, 38, 151–155. [Google Scholar]
- Wolter, A.; Hager, A.S.; Zannini, E.; Galle, S.; Ganzle, M.G.; Waters, D.M.; Arendt, E.K. Evaluation of exopolysaccharide producing Weissella cibaria MG1 strain for the production of sourdough from various flours. Food Microbiol. 2014, 37, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Leroy, F.; de Vuyst, L. Advances in production and simplified methods for recovery and quantification of exopolysaccharides for applications in food and health. J. Dairy Sci. 2016, 99, 3229–3238. [Google Scholar] [CrossRef] [PubMed]
- Velasco, S.E.; Yebra, M.J.; Monedero, V.; Ibarburu, I.; Dueñas, M.T.; Irastorza, A. Influence of the carbohydrate source on β-glucan production and enzyme activities involved in sugar metabolism in Pediococcus parvulus 2.6. Int. J. Food Microbiol. 2007, 115, 325–334. [Google Scholar] [CrossRef] [PubMed]
- Juvonen, R.; Honkapaa, K.; Maina, N.H.; Shi, Q.; Viljanen, K.; Maaheimo, H.; Virkki, L.; Tenkanen, M.; Lantto, R. The impact of fermentation with exopolysaccharide producing lactic acid bacteria on rheological, chemical and sensory properties of pureed carrots (Daucus carota L.). Int. J. Food Microbiol. 2015, 207, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Coda, R.; Shi, Q.; Tuomainen, P.; Katina, K.; Tenkanen, M. Exopolysaccharides production during the fermentation of soybean and fava bean flours by Leuconostoc mesenteroides DSM 20343. J. Agric. Food Chem. 2017, 65, 2805–2815. [Google Scholar] [CrossRef] [PubMed]
- Gänzle, M.; Schwab, C. Ecology of exopolysaccharide formation by lactic acid bacteria: Sucrose utilization, stress tolerance and biofilm formation. In Bacterial Polysaccharides, Current Innovations and Future Trends; Ullrich, M., Ed.; Caister Academic Press: Norfolk, UK, 2009; pp. 263–278. ISBN 978-1-904455-45-5. [Google Scholar]
- Coulon, J.; Houles, A.; Dimopoulou, M.; Maupeu, J.; Dols-Lafargue, M. Lysozyme resistance of the ropy strain Pediococcus parvulus IOEB 8801 is correlated with β-glucan accumulation around the cell. Int. J. Food Microbiol. 2012, 159, 25–29. [Google Scholar] [CrossRef] [PubMed]
- Alp, G.; Aslim, B. Relationship between the resistance to bile salts and low pH with exopolysaccharide (EPS) production of Bifidobacterium spp. isolated from infants feces and breast milk. Anaerobe 2010, 16, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Fanning, S.; Hall, L.J.; Cronin, M.; Zomer, A.; MacSharry, J.; Goulding, D.; Motherway, M.O.; Shanahan, F.; Nally, K.; Dougan, G.; et al. Bifidobacterial surface-exopolysaccharide facilitates commensal-host interaction through immune modulation and pathogen protection. Proc. Natl. Acad. Sci. USA 2012, 109, 2108–2113. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, S.; Kimoto-Nira, H.; Suganuma, H.; Suzuki, C.; Saito, T.; Yajima, N. Cellular fatty acid composition and exopolysaccharide contribute to bile tolerance in Lactobacillus brevis strains isolated from fermented japanese pickles. Can. J. Microbiol. 2014, 60, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Stack, H.M.; Kearney, N.; Stanton, C.; Fitzgerald, G.F.; Ross, R.P. Association of β-glucan endogenous production with increased stress tolerance of intestinal lactobacilli. Appl. Environ. Microbiol. 2010, 76, 500–507. [Google Scholar] [CrossRef] [PubMed]
- Fanning, S.; Hall, L.J.; van Sinderen, D. Bifidobacterium breve UCC2003 surface exopolysaccharide production is a beneficial trait mediating commensal-host interaction through immune modulation and pathogen protection. Gut Microbes 2012, 3, 420–425. [Google Scholar] [CrossRef] [PubMed]
- Arena, M.P.; Caggianiello, G.; Fiocco, D.; Russo, P.; Torelli, M.; Spano, G.; Capozzi, V. Barley β-glucans-containing food enhances probiotic performances of beneficial bacteria. Int. J. Mol. Sci. 2014, 15, 3025–3039. [Google Scholar] [CrossRef] [PubMed]
- Notararigo, S.; Nácher-Vázquez, M.; Ibarburu, I.; Werning, M.L.; Fernández de Palencia, P.F.; Dueñnas, M.T.; Aznar, R.; López, P.; Prieto, A. Comparative analysis of production and purification of homo- and hetero-polysaccharides produced by lactic acid bacteria. Carbohydr. Polym. 2013, 1, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Bove, P.; Russo, P.; Capozzi, V.; Gallone, A.; Spano, G.; Fiocco, D. Lactobacillus plantarum passage through an oro-gastro-intestinal tract simulator: Carrier matrix effect and transcriptional analysis of genes associated to stress and probiosis. Microbiol. Res. 2013, 168, 351–359. [Google Scholar] [CrossRef] [PubMed]
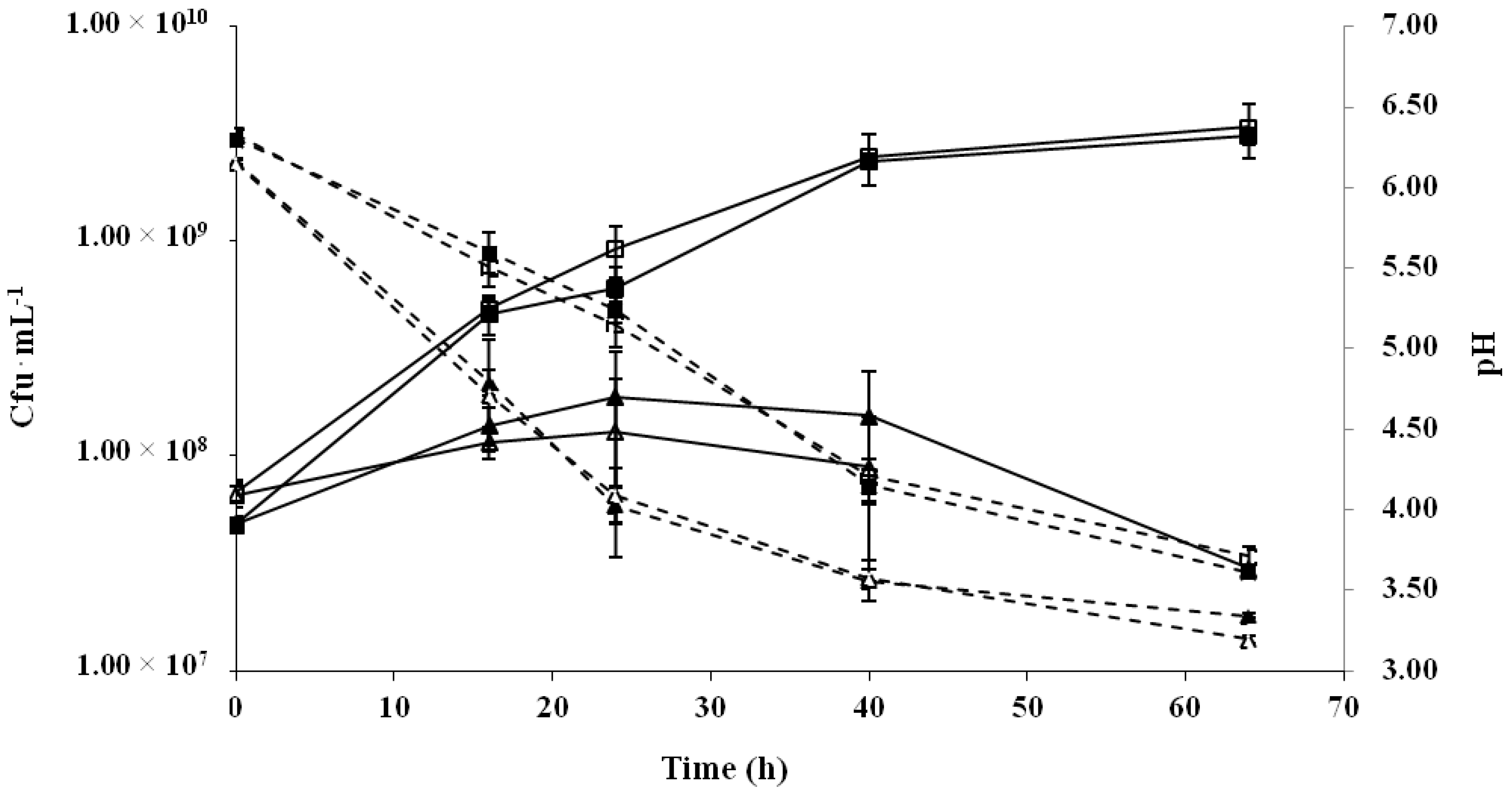
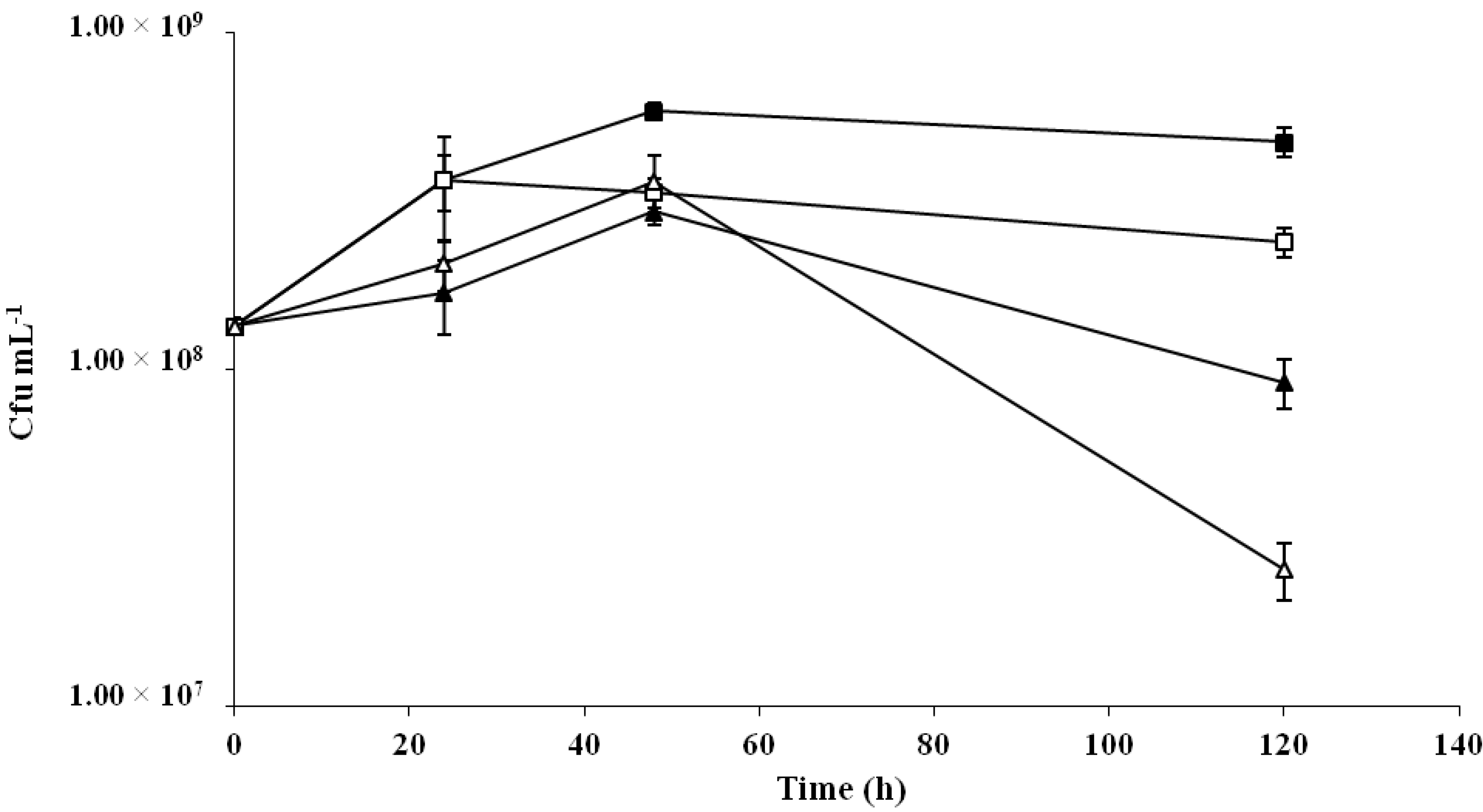
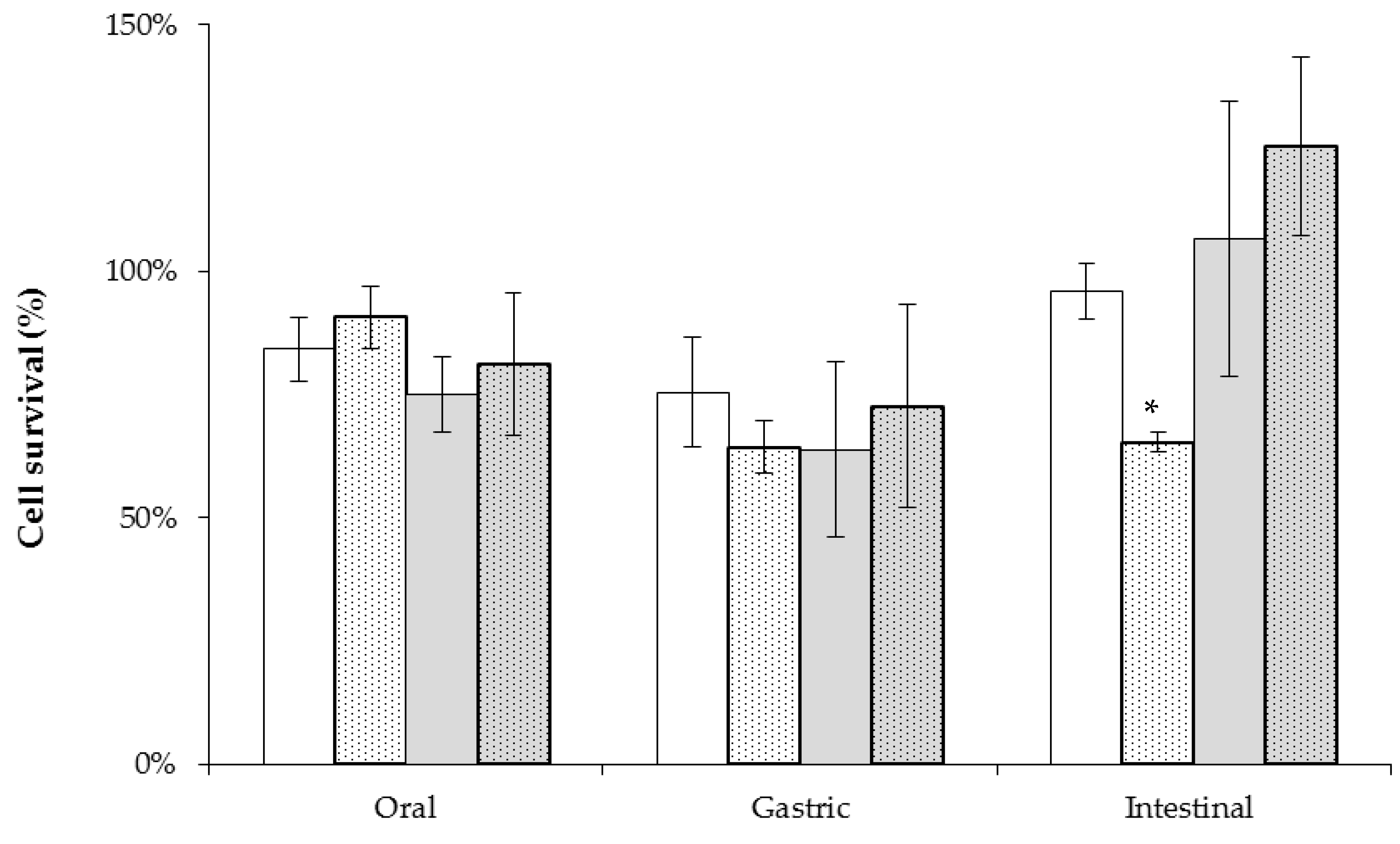
| Time (h) | Oat | Rice | Barley |
|---|---|---|---|
| 16 | 109 ± 17.9 | 34.2 ± 13.5 | 22.8 ± 2.8 |
| 24 | 139.7 ± 40.8 | 58.8 ± 14.0 | 21.3 ± 2.0 |
| 40 | 341.7 ± 44.2 | 129.6 ± 31.0 | 26.7 ± 9.8 |
| 64 | 659.4 ± 45.1 | 164.7 ± 52.3 | 24.9 ± 5.3 |
| Strain | Oat | Rice |
|---|---|---|
| P. parvulus 2.6 | 5407 ± 21 | 4187 ± 108 |
| P. parvulus 2.6NR | 1810 ± 248 | 3523 ± 297 |
| Ratio | 2.99 | 1.19 |
| Time (h) | Oat | Rice |
|---|---|---|
| 0 | 422.7 ± 16 | 81.6 ± 5.7 |
| 120 | 437.5 ± 40.8 | 96.4 ± 27.7 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pérez-Ramos, A.; Mohedano, M.L.; López, P.; Spano, G.; Fiocco, D.; Russo, P.; Capozzi, V. In Situ β-Glucan Fortification of Cereal-Based Matrices by Pediococcus parvulus 2.6: Technological Aspects and Prebiotic Potential. Int. J. Mol. Sci. 2017, 18, 1588. https://doi.org/10.3390/ijms18071588
Pérez-Ramos A, Mohedano ML, López P, Spano G, Fiocco D, Russo P, Capozzi V. In Situ β-Glucan Fortification of Cereal-Based Matrices by Pediococcus parvulus 2.6: Technological Aspects and Prebiotic Potential. International Journal of Molecular Sciences. 2017; 18(7):1588. https://doi.org/10.3390/ijms18071588
Chicago/Turabian StylePérez-Ramos, Adrián, María Luz Mohedano, Paloma López, Giuseppe Spano, Daniela Fiocco, Pasquale Russo, and Vittorio Capozzi. 2017. "In Situ β-Glucan Fortification of Cereal-Based Matrices by Pediococcus parvulus 2.6: Technological Aspects and Prebiotic Potential" International Journal of Molecular Sciences 18, no. 7: 1588. https://doi.org/10.3390/ijms18071588
APA StylePérez-Ramos, A., Mohedano, M. L., López, P., Spano, G., Fiocco, D., Russo, P., & Capozzi, V. (2017). In Situ β-Glucan Fortification of Cereal-Based Matrices by Pediococcus parvulus 2.6: Technological Aspects and Prebiotic Potential. International Journal of Molecular Sciences, 18(7), 1588. https://doi.org/10.3390/ijms18071588








