Dendritic Cells and Their Role in Allergy: Uptake, Proteolytic Processing and Presentation of Allergens
Abstract
:1. Introduction
2. Dendritic Cells Are the Most Important Antigen Presenting Cells in Health and Disease
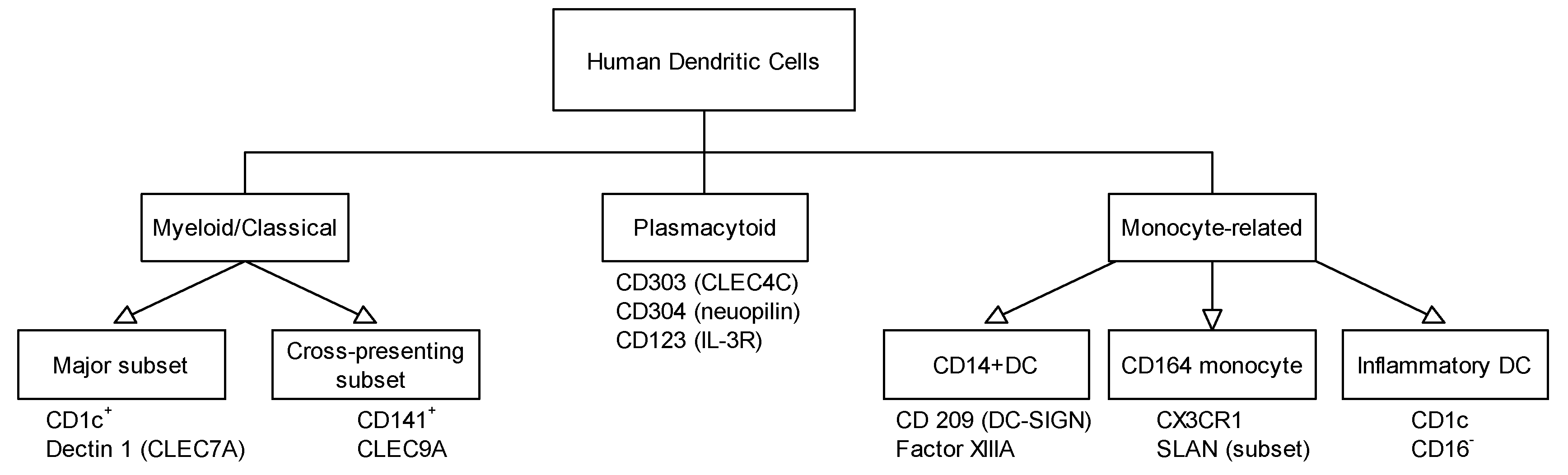
3. Different Dendritic Cell Subsets Identified in Skin, Respiratory, and Gastrointestinal Tract and Their Role in Allergy
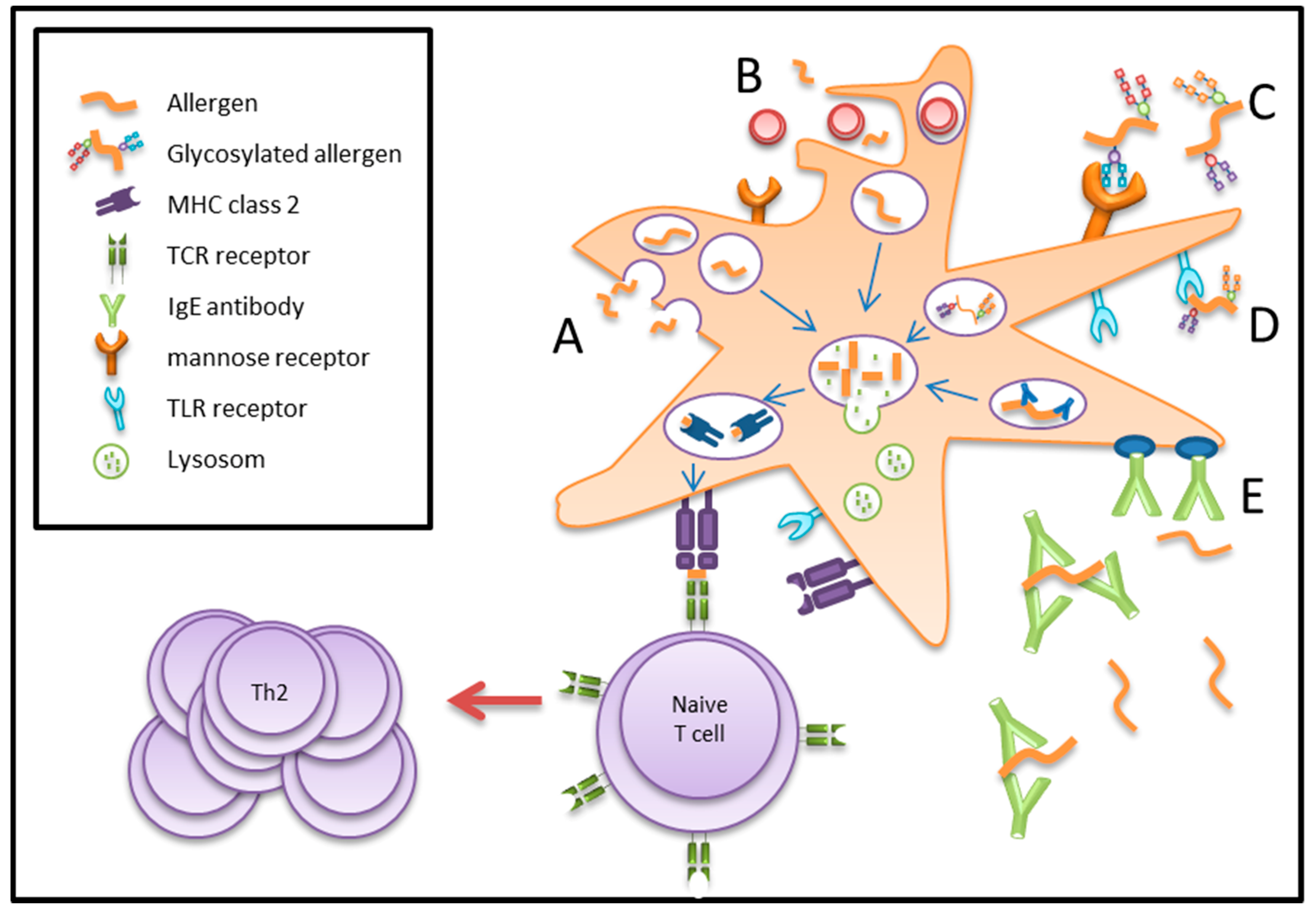
4. Antigen/Allergen Uptake by Dendritic Cells
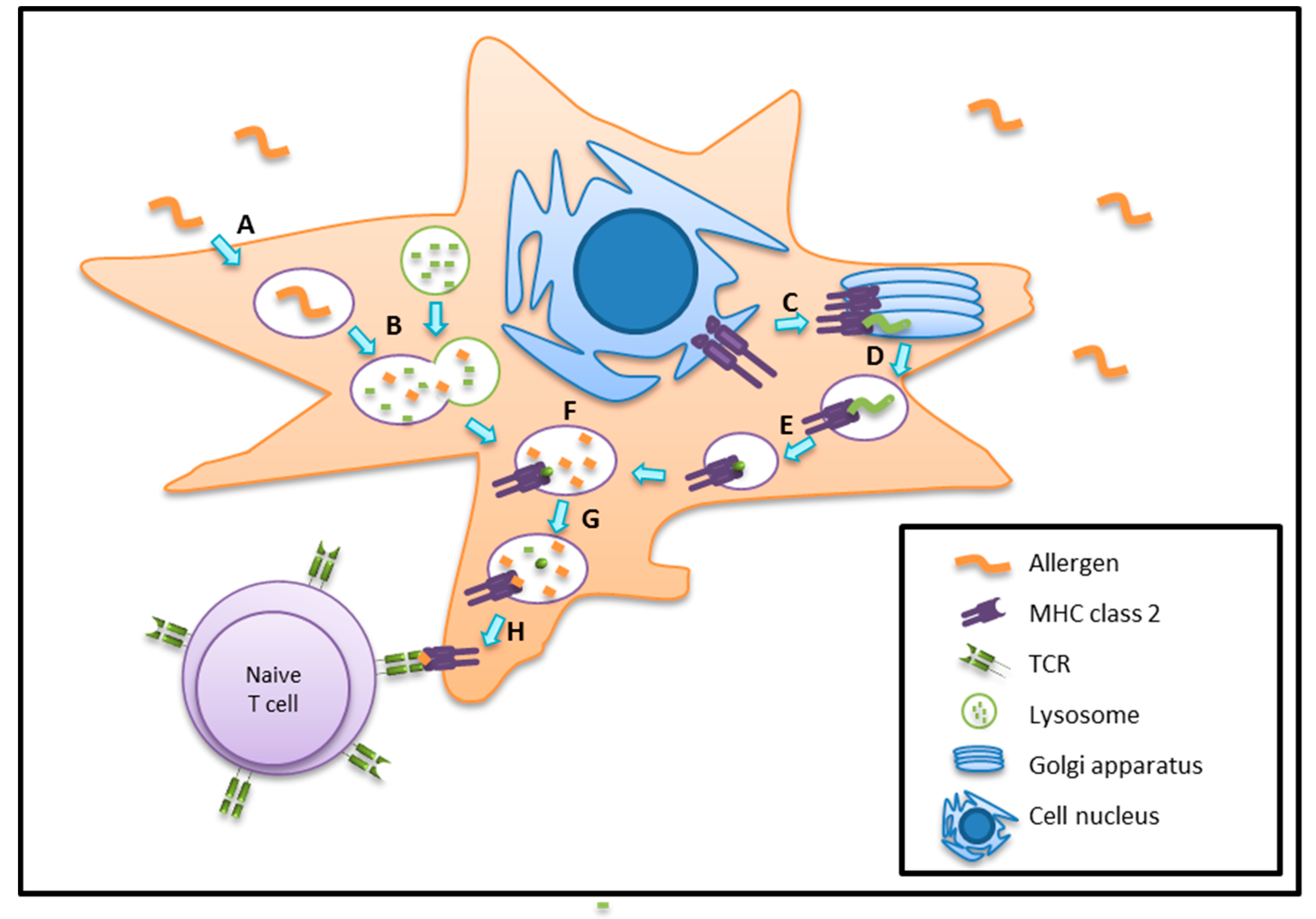
5. Antigen/Allergen Processing and Presentation
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| CCL18 | Chemokine C-C Motif Ligand 18 |
| CD | Cluster of Differentiation |
| cDC | Classic Dendritic Cell |
| CLIP | Class I-Associated Invariant Chain Peptide |
| DC | Dendritic Cell |
| Flk2 | Fetal Liver Kinase-2 |
| Flt3 | Forms Like Tyrosine Kinase-3 |
| GM-CSF | Granulocyte Macrophage-Colony Stimulating Factor |
| IFN | Interferon |
| IL | Interleukin |
| IgE | Immunoglobulin E |
| mDC | Myeloid Dendritic Cells |
| MHC | Major Histacompatibility Complex |
| MIP3-b | Macrophage Inflammatory Protein 3 |
| moDC | Monocyte-Derived Dendritic Cells |
| nsLTP | Non-Specific Lipid Transfer Protein |
| pDC | Plasmocytoid Dendritic Cells |
| Th cells | Helper T Cells |
| TCR | T Cell Receptor |
| TLR | Toll-Like Receptor |
References
- EAACI. Global Atlas of Allergy. European Academy of Allergy and Clinical Immunology: Florence, Italy, 2014. [Google Scholar]
- Radauer, C.; Bublin, M.; Wagner, S.; Mari, A.; Breiteneder, H. Allergens are distributed into few protein families and possess a restricted number of biochemical functions. J. Allergy Clin. Immunol. 2008, 121, 847–852. [Google Scholar] [CrossRef] [PubMed]
- Herz, U.; Renz, H.; Wiedermann, U. Animal models of type I allergy using recombinant allergens. Methods 2004, 32, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Zosky, G.R.; Sly, P.D. Animal models of asthma. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2007, 37, 973–988. [Google Scholar] [CrossRef] [PubMed]
- Bogh, K.L.; van Bilsen, J.; Glogowski, R.; Lopez-Exposito, I.; Bouchaud, G.; Blanchard, C.; Bodinier, M.; Smit, J.; Pieters, R.; Bastiaan-Net, S.; et al. Current challenges facing the assessment of the allergenic capacity of food allergens in animal models. Clin. Transl. Allergy 2016, 6, 21. [Google Scholar] [CrossRef] [PubMed]
- Banchereau, J.; Steinman, R.M. Dendritic cells and the control of immunity. Nature 1998, 392, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Merad, M.; Ginhoux, F.; Collin, M. Origin, homeostasis and function of langerhans cells and other langerin-expressing dendritic cells. Nat. Rev. Immunol. 2008, 8, 935–947. [Google Scholar] [CrossRef] [PubMed]
- Mildner, A.; Jung, S. Development and function of dendritic cell subsets. Immunity 2014, 40, 642–656. [Google Scholar] [CrossRef] [PubMed]
- Steinman, R.M. Decisions about dendritic cells: Past, present, and future. Annu. Rev. Immunol. 2012, 30, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.C.; Brown, B.D.; Shay, T.; Gautier, E.L.; Jojic, V.; Cohain, A.; Pandey, G.; Leboeuf, M.; Elpek, K.G.; Helft, J.; et al. Deciphering the transcriptional network of the dendritic cell lineage. Nat. Immunol. 2012, 13, 888–899. [Google Scholar] [CrossRef] [PubMed]
- Bogunovic, M.; Ginhoux, F.; Helft, J.; Shang, L.; Hashimoto, D.; Greter, M.; Liu, K.; Jakubzick, C.; Ingersoll, M.A.; Leboeuf, M.; et al. Origin of the lamina propria dendritic cell network. Immunity 2009, 31, 513–525. [Google Scholar] [CrossRef] [PubMed]
- Schulz, O.; Jaensson, E.; Persson, E.K.; Liu, X.; Worbs, T.; Agace, W.W.; Pabst, O. Intestinal cd103+, but not cx3cr1+, antigen sampling cells migrate in lymph and serve classical dendritic cell functions. J. Exp. Med. 2009, 206, 3101–3114. [Google Scholar] [CrossRef] [PubMed]
- Randolph, G.J.; Ochando, J.; Partida-Sanchez, S. Migration of dendritic cell subsets and their precursors. Annu. Rev. Immunol. 2008, 26, 293–316. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.J. Ipc: Professional type 1 interferon-producing cells and plasmacytoid dendritic cell precursors. Annu. Rev. Immunol. 2005, 23, 275–306. [Google Scholar] [CrossRef] [PubMed]
- Gordon, S.; Taylor, P.R. Monocyte and macrophage heterogeneity. Nat. Rev. Immunol. 2005, 5, 953–964. [Google Scholar] [CrossRef] [PubMed]
- Leon, B.; Lopez-Bravo, M.; Ardavin, C. Monocyte-derived dendritic cells. Semin. Immunol. 2005, 17, 313–318. [Google Scholar] [CrossRef] [PubMed]
- Leon, B.; Ardavin, C. Monocyte-derived dendritic cells in innate and adaptive immunity. Immunol. Cell Biol. 2008, 86, 320–324. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, G.; Haniffa, M. Human and mouse mononuclear phagocyte networks: A tale of two species? Front. Immunol. 2015, 6, 330. [Google Scholar] [CrossRef] [PubMed]
- Rowden, G.; Lewis, M.G.; Sullivan, A.K. Ia antigen expression on human epidermal langerhans cells. Nature 1977, 268, 247–248. [Google Scholar] [CrossRef] [PubMed]
- Stingl, G.; Wolff-Schreiner, E.C.; Pichler, W.J.; Gschnait, F.; Knapp, W.; Wolff, K. Epidermal langerhans cells bear fc and c3 receptors. Nature 1977, 268, 245–246. [Google Scholar] [CrossRef] [PubMed]
- Aiba, S.; Tagami, H. Dendritic cells play a crucial role in innate immunity to simple chemicals. J. Investig. Dermatol. Symp. Proc. 1999, 4, 158–163. [Google Scholar] [CrossRef] [PubMed]
- Serbina, N.V.; Salazar-Mather, T.P.; Biron, C.A.; Kuziel, W.A.; Pamer, E.G. Tnf/inos-producing dendritic cells mediate innate immune defense against bacterial infection. Immunity 2003, 19, 59–70. [Google Scholar] [CrossRef]
- Wollenberg, A.; Wagner, M.; Gunther, S.; Towarowski, A.; Tuma, E.; Moderer, M.; Rothenfusser, S.; Wetzel, S.; Endres, S.; Hartmann, G. Plasmacytoid dendritic cells: A new cutaneous dendritic cell subset with distinct role in inflammatory skin diseases. J. Investig. Dermatol. 2002, 119, 1096–1102. [Google Scholar] [CrossRef] [PubMed]
- Bieber, T.; Kraft, S.; Geiger, E.; Wollenberg, A.; Koch, S.; Novak, N. Fc [correction of ec] epsilon ri expressing dendritic cells: The missing link in the pathophysiology of atopic dermatitis? J. Dermatol. 2000, 27, 698–699. [Google Scholar] [CrossRef] [PubMed]
- Wollenberg, A.; Mommaas, M.; Oppel, T.; Schottdorf, E.M.; Gunther, S.; Moderer, M. Expression and function of the mannose receptor CD206 on epidermal dendritic cells in inflammatory skin diseases. J. Investig. Dermatol. 2002, 118, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Stary, G.; Bangert, C.; Stingl, G.; Kopp, T. Dendritic cells in atopic dermatitis: Expression of fcepsilonri on two distinct inflammation-associated subsets. Int. Arch. Allergy Immunol. 2005, 138, 278–290. [Google Scholar] [CrossRef] [PubMed]
- Lowes, M.A.; Chamian, F.; Abello, M.V.; Fuentes-Duculan, J.; Lin, S.L.; Nussbaum, R.; Novitskaya, I.; Carbonaro, H.; Cardinale, I.; Kikuchi, T.; et al. Increase in tnf-alpha and inducible nitric oxide synthase-expressing dendritic cells in psoriasis and reduction with efalizumab (anti-cd11a). Proc. Natl. Acad. Sci. USA 2005, 102, 19057–19062. [Google Scholar] [CrossRef] [PubMed]
- Albanesi, C.; Scarponi, C.; Pallotta, S.; Daniele, R.; Bosisio, D.; Madonna, S.; Fortugno, P.; Gonzalvo-Feo, S.; Franssen, J.D.; Parmentier, M.; et al. Chemerin expression marks early psoriatic skin lesions and correlates with plasmacytoid dendritic cell recruitment. J. Exp. Med. 2009, 206, 249–258. [Google Scholar] [CrossRef] [PubMed]
- Gomez de Aguero, M.; Vocanson, M.; Hacini-Rachinel, F.; Taillardet, M.; Sparwasser, T.; Kissenpfennig, A.; Malissen, B.; Kaiserlian, D.; Dubois, B. Langerhans cells protect from allergic contact dermatitis in mice by tolerizing CD8(+) t cells and activating Foxp3(+) regulatory T cells. J. Clin. Investig. 2012, 122, 1700–1711. [Google Scholar] [CrossRef] [PubMed]
- Jahnsen, F.L.; Moloney, E.D.; Hogan, T.; Upham, J.W.; Burke, C.M.; Holt, P.G. Rapid dendritic cell recruitment to the bronchial mucosa of patients with atopic asthma in response to local allergen challenge. Thorax 2001, 56, 823–826. [Google Scholar] [CrossRef] [PubMed]
- Bratke, K.; Lommatzsch, M.; Julius, P.; Kuepper, M.; Kleine, H.D.; Luttmann, W.; Christian Virchow, J. Dendritic cell subsets in human bronchoalveolar lavage fluid after segmental allergen challenge. Thorax 2007, 62, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Sertl, K.; Takemura, T.; Tschachler, E.; Ferrans, V.J.; Kaliner, M.A.; Shevach, E.M. Dendritic cells with antigen-presenting capability reside in airway epithelium, lung parenchyma, and visceral pleura. J. Exp. Med. 1986, 163, 436–451. [Google Scholar] [CrossRef] [PubMed]
- Fokkens, W.J.; Vroom, T.M.; Rijntjes, E.; Mulder, P.G. Cd-1 (T6), HLA-DR-expressing cells, presumably Langerhans cells, in nasal mucosa. Allergy 1989, 44, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Gong, J.L.; McCarthy, K.M.; Telford, J.; Tamatani, T.; Miyasaka, M.; Schneeberger, E.E. Intraepithelial airway dendritic cells: A distinct subset of pulmonary dendritic cells obtained by microdissection. J. Exp. Med. 1992, 175, 797–807. [Google Scholar] [CrossRef] [PubMed]
- McWilliam, A.S.; Nelson, D.J.; Holt, P.G. The biology of airway dendritic cells. Immunol. Cell Biol. 1995, 73, 405–413. [Google Scholar] [CrossRef] [PubMed]
- Condon, T.V.; Sawyer, R.T.; Fenton, M.J.; Riches, D.W. Lung dendritic cells at the innate-adaptive immune interface. J. Leukoc. Biol. 2011, 90, 883–895. [Google Scholar] [CrossRef] [PubMed]
- Wan, H.; Winton, H.L.; Soeller, C.; Tovey, E.R.; Gruenert, D.C.; Thompson, P.J.; Stewart, G.A.; Taylor, G.W.; Garrod, D.R.; Cannell, M.B.; et al. Der p 1 facilitates transepithelial allergen delivery by disruption of tight junctions. J. Clin. Investig. 1999, 104, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Barrett, N.A.; Maekawa, A.; Rahman, O.M.; Austen, K.F.; Kanaoka, Y. Dectin-2 recognition of house dust mite triggers cysteinyl leukotriene generation by dendritic cells. J. Immunol. 2009, 182, 1119–1128. [Google Scholar] [CrossRef] [PubMed]
- Lewkowich, I.P.; Day, S.B.; Ledford, J.R.; Zhou, P.; Dienger, K.; Wills-Karp, M.; Page, K. Protease-activated receptor 2 activation of myeloid dendritic cells regulates allergic airway inflammation. Respir. Res. 2011, 12, 122. [Google Scholar] [CrossRef] [PubMed]
- Rescigno, M.; Urbano, M.; Valzasina, B.; Francolini, M.; Rotta, G.; Bonasio, R.; Granucci, F.; Kraehenbuhl, J.P.; Ricciardi-Castagnoli, P. Dendritic cells express tight junction proteins and penetrate gut epithelial monolayers to sample bacteria. Nat. Immunol. 2001, 2, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Mowat, A.M. Anatomical basis of tolerance and immunity to intestinal antigens. Nat. Rev. Immunol. 2003, 3, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Steinfelder, S.; Andersen, J.F.; Cannons, J.L.; Feng, C.G.; Joshi, M.; Dwyer, D.; Caspar, P.; Schwartzberg, P.L.; Sher, A.; Jankovic, D. The major component in schistosome eggs responsible for conditioning dendritic cells for Th2 polarization is a T2 ribonuclease (omega-1). J. Exp. Med. 2009, 206, 1681–1690. [Google Scholar] [CrossRef] [PubMed]
- Hammad, H.; Plantinga, M.; Deswarte, K.; Pouliot, P.; Willart, M.A.; Kool, M.; Muskens, F.; Lambrecht, B.N. Inflammatory dendritic cells—not basophils—are necessary and sufficient for induction of Th2 immunity to inhaled house dust mite allergen. J. Exp. Med. 2010, 207, 2097–2111. [Google Scholar] [CrossRef] [PubMed]
- Abbas, A.; Lichtman, A.H.; Pillai, S. Cellular and Molecular Immunology, 7th ed.; Elsevier Saunders: Philapelphia, PA, USA, 2012. [Google Scholar]
- Reis, E.S.; Barbuto, J.A.; Isaac, L. Human monocyte-derived dendritic cells are a source of several complement proteins. Inflamm. Res. 2006, 55, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Piqueras, B.; Connolly, J.; Freitas, H.; Palucka, A.K.; Banchereau, J. Upon viral exposure, myeloid and plasmacytoid dendritic cells produce 3 waves of distinct chemokines to recruit immune effectors. Blood 2006, 107, 2613–2618. [Google Scholar] [CrossRef] [PubMed]
- Noirey, N.; Rougier, N.; Andre, C.; Schmitt, D.; Vincent, C. Langerhans-like dendritic cells generated from cord blood progenitors internalize pollen allergens by macropinocytosis, and part of the molecules are processed and can activate autologous naive t lymphocytes. J. Allergy Clin. Immunol. 2000, 105, 1194–1201. [Google Scholar] [CrossRef] [PubMed]
- Bonasio, R.; Scimone, M.L.; Schaerli, P.; Grabie, N.; Lichtman, A.H.; von Andrian, U.H. Clonal deletion of thymocytes by circulating dendritic cells homing to the thymus. Nat. Immunol. 2006, 7, 1092–1100. [Google Scholar] [CrossRef] [PubMed]
- Hadeiba, H.; Lahl, K.; Edalati, A.; Oderup, C.; Habtezion, A.; Pachynski, R.; Nguyen, L.; Ghodsi, A.; Adler, S.; Butcher, E.C. Plasmacytoid dendritic cells transport peripheral antigens to the thymus to promote central tolerance. Immunity 2012, 36, 438–450. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Swiggard, W.J.; Heufler, C.; Peng, M.; Mirza, A.; Steinman, R.M.; Nussenzweig, M.C. The receptor DEC-205 expressed by dendritic cells and thymic epithelial cells is involved in antigen processing. Nature 1995, 375, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Sallusto, F.; Cella, M.; Danieli, C.; Lanzavecchia, A. Dendritic cells use macropinocytosis and the mannose receptor to concentrate macromolecules in the major histocompatibility complex class ii compartment: Downregulation by cytokines and bacterial products. J. Exp. Med. 1995, 182, 389–400. [Google Scholar] [CrossRef] [PubMed]
- Figdor, C.G.; van Kooyk, Y.; Adema, G.J. C-type lectin receptors on dendritic cells and langerhans cells. Nat. Rev. Immunol. 2002, 2, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Sallusto, F.; Lanzavecchia, A. Efficient presentation of soluble antigen by cultured human dendritic cells is maintained by granulocyte/macrophage colony-stimulating factor plus interleukin 4 and downregulated by tumor necrosis factor alpha. J. Exp. Med. 1994, 179, 1109–1118. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Roche, P.A. Macropinocytosis in phagocytes: Regulation of mhc class-II-restricted antigen presentation in dendritic cells. Front. Physiol. 2015, 6, 1. [Google Scholar] [CrossRef] [PubMed]
- Engering, A.J.; Cella, M.; Fluitsma, D.; Brockhaus, M.; Hoefsmit, E.C.; Lanzavecchia, A.; Pieters, J. The mannose receptor functions as a high capacity and broad specificity antigen receptor in human dendritic cells. Eur. J. Immunol. 1997, 27, 2417–2425. [Google Scholar] [CrossRef] [PubMed]
- Mahnke, K.; Guo, M.; Lee, S.; Sepulveda, H.; Swain, S.L.; Nussenzweig, M.; Steinman, R.M. The dendritic cell receptor for endocytosis, DEC-205, can recycle and enhance antigen presentation via major histocompatibility complex class-II -positive lysosomal compartments. J. Cell Biol. 2000, 151, 673–684. [Google Scholar] [CrossRef] [PubMed]
- Holt, P.G.; Stumbles, P.A.; McWilliam, A.S. Functional studies on dendritic cells in the respiratory tract and related mucosal tissues. J. Leukoc. Biol. 1999, 66, 272–275. [Google Scholar] [PubMed]
- Bellinghausen, I.; Brand, U.; Knop, J.; Saloga, J. Comparison of allergen-stimulated dendritic cells from atopic and nonatopic donors dissecting their effect on autologous naive and memory T helper cells of such donors. J. Allergy Clin. Immunol. 2000, 105, 988–996. [Google Scholar] [CrossRef] [PubMed]
- Langenkamp, A.; Messi, M.; Lanzavecchia, A.; Sallusto, F. Kinetics of dendritic cell activation: Impact on priming of Th1, Th2 and nonpolarized T cells. Nat. Immunol. 2000, 1, 311–316. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, H.; Demeure, C.E.; Rubio, M.; Delespesse, G.; Sarfati, M. Human monocyte-derived dendritic cells induce naive T cell differentiation into t helper cell type 2 (Th2) or Th1/Th2 effectors. Role of stimulator/responder ratio. J. Exp. Med. 2000, 192, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Hellman, P.; Eriksson, H. Early activation markers of human peripheral dendritic cells. Hum. Immunol. 2007, 68, 324–333. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, M.; Takeda, K.; Lucas, J.J.; Joetham, A.; Yasutomo, K.; Gelfand, E.W. Low-dose lipopolysaccharide affects lung allergic responses by regulating jagged1 expression on antigen-pulsed dendritic cells. Int. Arch. Allergy Immunol. 2012, 157, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Smole, U.; Balazs, N.; Hoffmann-Sommergruber, K.; Radauer, C.; Hafner, C.; Wallner, M.; Ferreira, F.; Grossinger, R.; de Jong, E.C.; Wagner, S.; et al. Differential T-cell responses and allergen uptake after exposure of dendritic cells to the birch pollen allergens bet v 1.0101, bet v 1.0401 and bet v 1.1001. Immunobiology 2010, 215, 903–909. [Google Scholar] [CrossRef] [PubMed]
- Smole, U.; Wagner, S.; Balazs, N.; Radauer, C.; Bublin, M.; Allmaier, G.; Hoffmann-Sommergruber, K.; Breiteneder, H. Bet v 1 and its homologous food allergen Api g 1 stimulate dendritic cells from birch pollen-allergic individuals to induce different Th-cell polarization. Allergy 2010, 65, 1388–1396. [Google Scholar] [CrossRef] [PubMed]
- Kitzmuller, C.; Nagl, B.; Deifl, S.; Walterskirchen, C.; Jahn-Schmid, B.; Zlabinger, G.J.; Bohle, B. Human blood basophils do not act as antigen-presenting cells for the major birch pollen allergen bet v 1. Allergy 2012, 67, 593–600. [Google Scholar] [CrossRef] [PubMed]
- Gomez, E.; Diaz-Perales, A.; Tordesillas, L.; Dona, I.; Torres, M.J.; Blazquez, A.B.; Gomez, F.; Blanca, M.; Mayorga, C. Effect of pru p 3 on dendritic cell maturation and T-lymphocyte proliferation in peach allergic patients. Ann. Allergy Asthma Immunol. 2012, 109, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Ruiter, B.; Shreffler, W.G. The role of dendritic cells in food allergy. J. Allergy Clin. Immunol. 2012, 129, 921–928. [Google Scholar] [CrossRef] [PubMed]
- Hammad, H.; Charbonnier, A.S.; Duez, C.; Jacquet, A.; Stewart, G.A.; Tonnel, A.B.; Pestel, J. Th2 polarization by der p 1-pulsed monocyte-derived dendritic cells is due to the allergic status of the donors. Blood 2001, 98, 1135–1141. [Google Scholar] [CrossRef] [PubMed]
- Ashjaei, K.; Bublin, M.; Smole, U.; Lengger, N.; Hafner, C.; Breiteneder, H.; Wagner, S.; Hoffmann-Sommergruber, K. Differential T-helper cell polarization after allergen-specific stimulation of autologous dendritic cells in polysensitized allergic patients. Int. Arch. Allergy Immunol. 2015, 166, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Sung, S.J.; Taketomi, E.A.; Smith, A.M.; Platts-Mills, T.A.; Fu, S.M. Efficient presentation of house dust mite allergen der p 2 by monocyte-derived dendritic cells and the role of beta 2 integrins. Scand. J. Immunol. 1999, 49, 96–105. [Google Scholar] [CrossRef] [PubMed]
- De Wit, D.; Amraoui, Z.; Vincart, B.; Michel, O.; Michils, A.; Van Overvelt, L.; Willems, F.; Goldman, M. Helper T-cell responses elicited by der p 1-pulsed dendritic cells and recombinant IL-12 in atopic and healthy subjects. J. Allergy Clin. Immunol. 2000, 105, 346–352. [Google Scholar] [CrossRef]
- Smole, U.; Radauer, C.; Lengger, N.; Svoboda, M.; Rigby, N.; Bublin, M.; Gaier, S.; Hoffmann-Sommergruber, K.; Jensen-Jarolim, E.; Mechtcheriakova, D.; et al. The major birch pollen allergen bet v 1 induces different responses in dendritic cells of birch pollen allergic and healthy individuals. PLoS ONE 2015, 10, e0117904. [Google Scholar] [CrossRef] [PubMed]
- Al-Ghouleh, A.; Johal, R.; Sharquie, I.K.; Emara, M.; Harrington, H.; Shakib, F.; Ghaemmaghami, A.M. The glycosylation pattern of common allergens: The recognition and uptake of der p 1 by epithelial and dendritic cells is carbohydrate dependent. PLoS ONE 2012, 7, e33929. [Google Scholar] [CrossRef] [PubMed]
- Roche, P.A.; Furuta, K. The ins and outs of mhc class-II-mediated antigen processing and presentation. Nat. Rev. Immunol. 2015, 15, 203–216. [Google Scholar] [CrossRef] [PubMed]
- Zulehner, N.; Nagl, B.; Briza, P.; Roulias, A.; Ballmer-Weber, B.; Zlabinger, G.J.; Ferreira, F.; Bohle, B. Characterization of the T-cell response to dau c 1, the bet v 1-homolog in carrot. Allergy 2017, 72, 244–251. [Google Scholar] [CrossRef] [PubMed]
- Prigozy, T.I.; Naidenko, O.; Qasba, P.; Elewaut, D.; Brossay, L.; Khurana, A.; Natori, T.; Koezuka, Y.; Kulkarni, A.; Kronenberg, M. Glycolipid antigen processing for presentation by cd1d molecules. Science 2001, 291, 664–667. [Google Scholar] [CrossRef] [PubMed]
- Turley, S.J.; Inaba, K.; Garrett, W.S.; Ebersold, M.; Unternaehrer, J.; Steinman, R.M.; Mellman, I. Transport of peptide-mhc class II complexes in developing dendritic cells. Science 2000, 288, 522–527. [Google Scholar] [CrossRef] [PubMed]
- Thery, C.; Regnault, A.; Garin, J.; Wolfers, J.; Zitvogel, L.; Ricciardi-Castagnoli, P.; Raposo, G.; Amigorena, S. Molecular characterization of dendritic cell-derived exosomes. Selective accumulation of the heat shock protein hsc73. J. Cell Biol. 1999, 147, 599–610. [Google Scholar] [CrossRef] [PubMed]
- Cyster, J.G. Chemokines and cell migration in secondary lymphoid organs. Science 1999, 286, 2098–2102. [Google Scholar] [CrossRef] [PubMed]
- Cella, M.; Engering, A.; Pinet, V.; Pieters, J.; Lanzavecchia, A. Inflammatory stimuli induce accumulation of mhc class II complexes on dendritic cells. Nature 1997, 388, 782–787. [Google Scholar] [CrossRef] [PubMed]
- Pierre, P.; Turley, S.J.; Gatti, E.; Hull, M.; Meltzer, J.; Mirza, A.; Inaba, K.; Steinman, R.M.; Mellman, I. Developmental regulation of mhc class II transport in mouse dendritic cells. Nature 1997, 388, 787–792. [Google Scholar] [PubMed]
- Calabi, F.; Jarvis, J.M.; Martin, L.; Milstein, C. Two classes of cd1 genes. Eur. J. Immunol. 1989, 19, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Adams, E.J. Lipid presentation by human cd1 molecules and the diverse T cell populations that respond to them. Curr. Opin. Immunol. 2014, 26, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Porcelli, S.A. The cd1 family: A third lineage of antigen-presenting molecules. Adv. Immunol. 1995, 59, 1–98. [Google Scholar] [PubMed]
- Van Wijk, F.; Nierkens, S.; Hassing, I.; Feijen, M.; Koppelman, S.J.; de Jong, G.A.; Pieters, R.; Knippels, L.M. The effect of the food matrix on in vivo immune responses to purified peanut allergens. Toxicol. Sci. 2005, 86, 333–341. [Google Scholar] [CrossRef] [PubMed]
- Dearman, R.J.; Alcocer, M.J.; Kimber, I. Influence of plant lipids on immune responses in mice to the major brazil nut allergen ber e 1. Clin. Exp. Allergy 2007, 37, 582–591. [Google Scholar] [CrossRef] [PubMed]
- Abos-Gracia, B.; del Moral, M.G.; Lopez-Relano, J.; Viana-Huete, V.; Castro, L.; Villalba, M.; Martinez-Naves, E. Olea europaea pollen lipids activate invariant natural killer T cells by upregulating cd1d expression on dendritic cells. J. Allergy Clin. Immunol. 2013, 131, 1393–1399. [Google Scholar] [CrossRef] [PubMed]
| DC Subtype | Human | Cellular Function | Mouse |
|---|---|---|---|
| cDC1 | CD141 | Cross-presentation; IL-12 and IFN-λ production; Expression of TLR3; Induce Th1/Th2 responses | CD103/CD8 |
| XCR1 | XCR1 | ||
| CLEC9A | Clec9A | ||
| CADM1 | CADM1 | ||
| cDC2 | CD1c | CD4+ T cell responses; IL-1B, IL-6, and IL-23 production; Expression of all TLRs apart from TLR3 (mouse) and TLR9 (human); Induce Th2/Th17 responses | CD24 |
| CD11b | CD11b | ||
| SIRPα | SIRPα | ||
| pDC | CD123 | Anti-viral responses; IFN-α production; Expression of TLR7 and TLR9 | SiglecH |
| CD303 | Bst2 | ||
| CD304 | Ly6c | ||
| LC | Langerin CD1a+++ CD11clo | Maintain epidermal integrity; Induce Tregs; Induce Th17 responses | Langerin CD24 CD11b F4/80 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Humeniuk, P.; Dubiela, P.; Hoffmann-Sommergruber, K. Dendritic Cells and Their Role in Allergy: Uptake, Proteolytic Processing and Presentation of Allergens. Int. J. Mol. Sci. 2017, 18, 1491. https://doi.org/10.3390/ijms18071491
Humeniuk P, Dubiela P, Hoffmann-Sommergruber K. Dendritic Cells and Their Role in Allergy: Uptake, Proteolytic Processing and Presentation of Allergens. International Journal of Molecular Sciences. 2017; 18(7):1491. https://doi.org/10.3390/ijms18071491
Chicago/Turabian StyleHumeniuk, Piotr, Pawel Dubiela, and Karin Hoffmann-Sommergruber. 2017. "Dendritic Cells and Their Role in Allergy: Uptake, Proteolytic Processing and Presentation of Allergens" International Journal of Molecular Sciences 18, no. 7: 1491. https://doi.org/10.3390/ijms18071491
APA StyleHumeniuk, P., Dubiela, P., & Hoffmann-Sommergruber, K. (2017). Dendritic Cells and Their Role in Allergy: Uptake, Proteolytic Processing and Presentation of Allergens. International Journal of Molecular Sciences, 18(7), 1491. https://doi.org/10.3390/ijms18071491




