Melatonin and Fertoprotective Adjuvants: Prevention against Premature Ovarian Failure during Chemotherapy
Abstract
:1. Introduction
2. Chemotherapy-Induced Ovarian Disorder
2.1. Chemotherapeutic Drugs
2.2. Mechanisms of Chemotherapy-Induced Ovarian Disorder
3. Melatonin and Other Adjuvant Agents for Protection against Chemotherapy-Induced Follicle Depletion
3.1. Melatonin
3.1.1. Pleiotropic Effects of Melatonin on Cancer Prevention and Immune System
3.1.2. Melatonin as Antioxidant in Ovarian Follicles
3.1.3. New Application of Melatonin as a Fertoprotective Agent in Fertility Preservation
3.2. Other Candidates as a Fertoprotective Agent
3.2.1. Sphingosin-1-phosphate
3.2.2. Imatinib
3.2.3. Tamoxifen
3.2.4. GnRH Analog
3.2.5. Ammonium trichloro (dioxoethylene-O,O′) tellurate (AS101)
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Waxman, J. Chemotherapy and the adult gonad: A review. J. R. Soc. Med. 1983, 76, 144–148. [Google Scholar] [PubMed]
- Loren, A.W.; Mangu, P.B.; Beck, L.N.; Brennan, L.; Magdalinski, A.J.; Partridge, A.H.; Quinn, G.; Wallace, W.H.; Oktay, K.; American Society of Clinical, Oncology. Fertility preservation for patients with cancer: American Society of Clinical Oncology clinical practice guideline update. J. Clin. Oncol. 2013, 31, 2500–2510. [Google Scholar] [CrossRef] [PubMed]
- Peccatori, F.A.; Azim, H.A., Jr.; Orecchia, R.; Hoekstra, H.J.; Pavlidis, N.; Kesic, V.; Pentheroudakis, G.; Group, E.G.W. Cancer, pregnancy and fertility: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2013, 24, vi60–vi70. [Google Scholar] [CrossRef] [PubMed]
- Paluch-Shimon, S.; Pagani, O.; Partridge, A.H.; Bar-Meir, E.; Fallowfield, L.; Fenlon, D.; Friedman, E.; Gelmon, K.; Gentilini, O.; Geraghty, J.; et al. Second international consensus guidelines for breast cancer in young women (BCY2). Breast 2016, 26, 87–99. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, N. Fertility preservation in female cancer patients: An overview. J. Hum. Reprod. Sci. 2015, 8, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Meirow, D.; Wallace, W.H. Preservation of fertility in patients with cancer. N. Engl. J. Med. 2009, 360, 2682. [Google Scholar] [PubMed]
- Sklar, C.A.; Mertens, A.C.; Mitby, P.; Whitton, J.; Stovall, M.; Kasper, C.; Mulder, J.; Green, D.; Nicholson, H.S.; Yasui, Y.; et al. Premature menopause in survivors of childhood cancer: A report from the childhood cancer survivor study. J. Natl. Cancer Inst. 2006, 98, 890–896. [Google Scholar] [CrossRef] [PubMed]
- Bath, L.E.; Wallace, W.H.; Shaw, M.P.; Fitzpatrick, C.; Anderson, R.A. Depletion of ovarian reserve in young women after treatment for cancer in childhood: Detection by anti-Mullerian hormone, inhibin B and ovarian ultrasound. Hum. Reprod. 2003, 18, 2368–2374. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.A.; Themmen, A.P.; Al-Qahtani, A.; Groome, N.P.; Cameron, D.A. The effects of chemotherapy and long-term gonadotrophin suppression on the ovarian reserve in premenopausal women with breast cancer. Hum. Reprod. 2006, 21, 2583–2592. [Google Scholar] [CrossRef] [PubMed]
- Himelstein-Braw, R.; Peters, H.; Faber, M. Morphological study of the ovaries of leukaemic children. Br. J. Cancer 1978, 38, 82–87. [Google Scholar] [CrossRef] [PubMed]
- Meirow, D.; Nugent, D. The effects of radiotherapy and chemotherapy on female reproduction. Hum. Reprod. Update 2001, 7, 535–543. [Google Scholar] [CrossRef] [PubMed]
- Meirow, D.; Biederman, H.; Anderson, R.A.; Wallace, W.H. Toxicity of chemotherapy and radiation on female reproduction. Clin. Obstet. Gynecol. 2010, 53, 727–739. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.A.; Wallace, W.H. Antimullerian hormone, the assessment of the ovarian reserve, and the reproductive outcome of the young patient with cancer. Fertil. Steril. 2013, 99, 1469–1475. [Google Scholar] [CrossRef] [PubMed]
- Familiari, G.; Caggiati, A.; Nottola, S.A.; Ermini, M.; di Benedetto, M.R.; Motta, P.M. Ultrastructure of human ovarian primordial follicles after combination chemotherapy for Hodgkin’s disease. Hum. Reprod. 1993, 8, 2080–2087. [Google Scholar] [CrossRef] [PubMed]
- Ataya, K.; Rao, L.V.; Lawrence, E.; Kimmel, R. Luteinizing hormone-releasing hormone agonist inhibits cyclophosphamide-induced ovarian follicular depletion in rhesus monkeys. Biol. Reprod. 1995, 52, 365–372. [Google Scholar] [CrossRef] [PubMed]
- Meirow, D.; Lewis, H.; Nugent, D.; Epstein, M. Subclinical depletion of primordial follicular reserve in mice treated with cyclophosphamide: Clinical importance and proposed accurate investigative tool. Hum. Reprod. 1999, 14, 1903–1907. [Google Scholar] [CrossRef] [PubMed]
- Maneschi, F.; Benedetti-Panici, P.; Scambia, G.; Salerno, M.G.; D’Agostino, G.; Mancuso, S. Menstrual and hormone patterns in women treated with high-dose cisplatin and bleomycin. Gynecol. Oncol. 1994, 54, 345–348. [Google Scholar] [CrossRef] [PubMed]
- Nozaki, Y.; Furubo, E.; Matsuno, T.; Fukui, R.; Kizawa, K.; Kozaki, T.; Sanzen, T. Collaborative work on evaluation of ovarian toxicity 6. Two- or four-week repeated-dose studies and fertility study of cisplatin in female rats. J. Toxicol. Sci. 2009, 34, 73–81. [Google Scholar] [CrossRef]
- Blommaert, F.A.; Michael, C.; van Dijk-Knijnenburg, H.C.; Schornagel, J.H.; den Engelse, L.; Fichtinger-Schepman, A.M. The formation and persistence of carboplatin-DNA adducts in rats. Cancer Chemother. Pharmacol. 1996, 38, 273–280. [Google Scholar] [CrossRef] [PubMed]
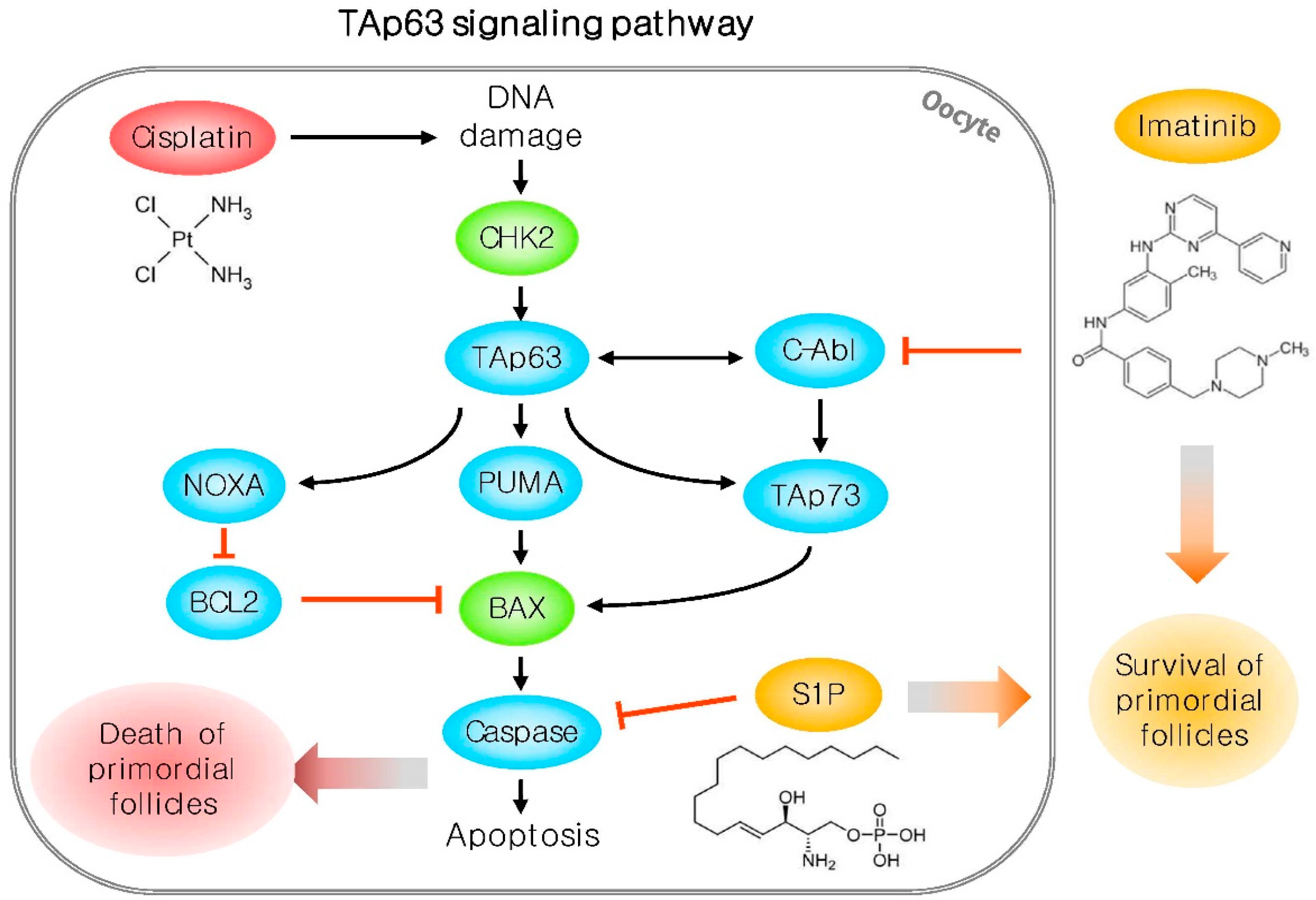
- Gonfloni, S.; di Tella, L.; Caldarola, S.; Cannata, S.M.; Klinger, F.G.; di Bartolomeo, C.; Mattei, M.; Candi, E.; de Felici, M.; Melino, G.; et al. Inhibition of the c-Abl-TAp63 pathway protects mouse oocytes from chemotherapy-induced death. Nat. Med. 2009, 15, 1179–1185. [Google Scholar] [CrossRef] [PubMed]
- Higdon, R.E.; Marchetti, F.; Mailhes, J.B.; Phillips, G.L. The effects of cisplatin on murine metaphase II oocytes. Gynecol. Oncol. 1992, 47, 348–352. [Google Scholar] [CrossRef]
- Mailhes, J.B. Important biological variables that can influence the degree of chemical-induced aneuploidy in mammalian oocyte and zygotes. Mutat. Res. 1995, 339, 155–176. [Google Scholar] [CrossRef]
- Blumenfeld, Z. Chemotherapy and fertility. Best Pract. Res. Clin. Obs. Gynaecol. 2012, 26, 379–390. [Google Scholar] [CrossRef] [PubMed]
- Hortobagyi, G.N.; Buzdar, A.U.; Marcus, C.E.; Smith, T.L. Immediate and long-term toxicity of adjuvant chemotherapy regimens containing doxorubicin in trials at M.D. Anderson Hospital and Tumor Institute. NCI Monogr. 1986, 1, 105–109. [Google Scholar]
- Meirow, D. Reproduction post-chemotherapy in young cancer patients. Mol. Cell. Endocrinol. 2000, 169, 123–131. [Google Scholar] [CrossRef]
- Lee, S.J.; Schover, L.R.; Partridge, A.H.; Patrizio, P.; Wallace, W.H.; Hagerty, K.; Beck, L.N.; Brennan, L.V.; Oktay, K.; American Society of Clinical, Oncology. American Society of Clinical Oncology recommendations on fertility preservation in cancer patients. J. Clin. Oncol. 2006, 24, 2917–2931. [Google Scholar] [CrossRef] [PubMed]
- Bines, J.; Oleske, D.M.; Cobleigh, M.A. Ovarian function in premenopausal women treated with adjuvant chemotherapy for breast cancer. J. Clin. Oncol. 1996, 14, 1718–1729. [Google Scholar] [CrossRef] [PubMed]
- Mol, F.; Mol, B.W.; Ankum, W.M.; van der Veen, F.; Hajenius, P.J. Current evidence on surgery, systemic methotrexate and expectant management in the treatment of tubal ectopic pregnancy: A systematic review and meta-analysis. Hum. Reprod. Update 2008, 14, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Oriol, B.; Barrio, A.; Pacheco, A.; Serna, J.; Zuzuarregui, J.L.; Garcia-Velasco, J.A. Systemic methotrexate to treat ectopic pregnancy does not affect ovarian reserve. Fertil. Steril. 2008, 90, 1579–1582. [Google Scholar] [CrossRef] [PubMed]
- Davis, A.L.; Klitus, M.; Mintzer, D.M. Chemotherapy-induced amenorrhea from adjuvant breast cancer treatment: The effect of the addition of taxanes. Clin. Breast Cancer 2005, 6, 421–424. [Google Scholar] [CrossRef] [PubMed]
- Reh, A.; Oktem, O.; Oktay, K. Impact of breast cancer chemotherapy on ovarian reserve: A prospective observational analysis by menstrual history and ovarian reserve markers. Fertil. Steril. 2008, 90, 1635–1639. [Google Scholar] [CrossRef] [PubMed]
- Han, H.S.; Ro, J.; Lee, K.S.; Nam, B.H.; Seo, J.A.; Lee, D.H.; Lee, H.; Lee, E.S.; Kang, H.S.; Kim, S.W. Analysis of chemotherapy-induced amenorrhea rates by three different anthracycline and taxane containing regimens for early breast cancer. Breast Cancer Res. Treat. 2009, 115, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Abusief, M.E.; Missmer, S.A.; Ginsburg, E.S.; Weeks, J.C.; Partridge, A.H. The effects of paclitaxel, dose density, and trastuzumab on treatment-related amenorrhea in premenopausal women with breast cancer. Cancer 2010, 116, 791–798. [Google Scholar] [CrossRef] [PubMed]
- Ganz, P.A.; Land, S.R.; Geyer, C.E., Jr.; Cecchini, R.S.; Costantino, J.P.; Pajon, E.R.; Fehrenbacher, L.; Atkins, J.N.; Polikoff, J.A.; Vogel, V.G.; et al. Menstrual history and quality-of-life outcomes in women with node-positive breast cancer treated with adjuvant therapy on the NSABP B-30 trial. J. Clin. Oncol. 2011, 29, 1110–1116. [Google Scholar] [CrossRef] [PubMed]
- Petrek, J.A.; Naughton, M.J.; Case, L.D.; Paskett, E.D.; Naftalis, E.Z.; Singletary, S.E.; Sukumvanich, P. Incidence, time course, and determinants of menstrual bleeding after breast cancer treatment: A prospective study. J. Clin. Oncol. 2006, 24, 1045–1051. [Google Scholar] [CrossRef] [PubMed]
- Higgins, M.J.; Liedke, P.E.; Goss, P.E. Extended adjuvant endocrine therapy in hormone dependent breast cancer: The paradigm of the NCIC-CTG MA.17/BIG 1–97 trial. Crit. Rev. Oncol. Hematol. 2013, 86, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Roness, H.; Kalich-Philosoph, L.; Meirow, D. Prevention of chemotherapy-induced ovarian damage: Possible roles for hormonal and non-hormonal attenuating agents. Hum. Reprod. Update 2014, 20, 759–774. [Google Scholar] [CrossRef] [PubMed]
- Bonadonna, G.; Bonfante, V.; Viviani, S.; di Russo, A.; Villani, F.; Valagussa, P. ABVD plus subtotal nodal versus involved-field radiotherapy in early-stage Hodgkin’s disease: Long-term results. J. Clin. Oncol. 2004, 22, 2835–2841. [Google Scholar] [CrossRef] [PubMed]
- Decanter, C.; Morschhauser, F.; Pigny, P.; Lefebvre, C.; Gallo, C.; Dewailly, D. Anti-Mullerian hormone follow-up in young women treated by chemotherapy for lymphoma: Preliminary results. Reprod. Biomed. Online 2010, 20, 280–285. [Google Scholar] [CrossRef] [PubMed]
- Behringer, K.; Mueller, H.; Goergen, H.; Thielen, I.; Eibl, A.D.; Stumpf, V.; Wessels, C.; Wiehlputz, M.; Rosenbrock, J.; Halbsguth, T.; et al. Gonadal function and fertility in survivors after Hodgkin lymphoma treatment within the German Hodgkin Study Group HD13 to HD15 trials. J. Clin. Oncol. 2013, 31, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Kreuser, E.D.; Felsenberg, D.; Behles, C.; Seibt-Jung, H.; Mielcarek, M.; Diehl, V.; Dahmen, E.; Thiel, E. Long-term gonadal dysfunction and its impact on bone mineralization in patients following COPP/ABVD chemotherapy for Hodgkin’s disease. Ann. Oncol. 1992, 3, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Behringer, K.; Breuer, K.; Reineke, T.; May, M.; Nogova, L.; Klimm, B.; Schmitz, T.; Wildt, L.; Diehl, V.; Engert, A.; et al. Secondary amenorrhea after Hodgkin’s lymphoma is influenced by age at treatment, stage of disease, chemotherapy regimen, and the use of oral contraceptives during therapy: A report from the German Hodgkin’s Lymphoma Study Group. J. Clin. Oncol. 2005, 23, 7555–7564. [Google Scholar] [CrossRef] [PubMed]
- Di Giacomo, M.; Barchi, M.; Baudat, F.; Edelmann, W.; Keeney, S.; Jasin, M. Distinct DNA-damage-dependent and -independent responses drive the loss of oocytes in recombination-defective mouse mutants. Proc. Natl. Acad. Sci. USA 2005, 102, 737–742. [Google Scholar] [CrossRef] [PubMed]
- Jurisicova, A.; Lee, H.J.; D’Estaing, S.G.; Tilly, J.; Perez, G.I. Molecular requirements for doxorubicin-mediated death in murine oocytes. Cell Death Differ. 2006, 13, 1466–1474. [Google Scholar] [CrossRef] [PubMed]
- Perez, G.I.; Acton, B.M.; Jurisicova, A.; Perkins, G.A.; White, A.; Brown, J.; Trbovich, A.M.; Kim, M.R.; Fissore, R.; Xu, J.; et al. Genetic variance modifies apoptosis susceptibility in mature oocytes via alterations in DNA repair capacity and mitochondrial ultrastructure. Cell Death Differ. 2007, 14, 524–533. [Google Scholar] [CrossRef] [PubMed]
- Cruz, M.H.; Leal, C.L.; Cruz, J.F.; Tan, D.X.; Reiter, R.J. Essential actions of melatonin in protecting the ovary from oxidative damage. Theriogenology 2014, 82, 925–932. [Google Scholar] [CrossRef] [PubMed]
- Morgan, S.; Anderson, R.A.; Gourley, C.; Wallace, W.H.; Spears, N. How do chemotherapeutic agents damage the ovary? Hum. Reprod. Update 2012, 18, 525–535. [Google Scholar] [CrossRef] [PubMed]
- Carroll, J.; Marangos, P. The DNA damage response in mammalian oocytes. Front. Genet. 2013, 4, 117. [Google Scholar] [CrossRef] [PubMed]
- Oktem, O.; Oktay, K. A novel ovarian xenografting model to characterize the impact of chemotherapy agents on human primordial follicle reserve. Cancer Res. 2007, 67, 10159–10162. [Google Scholar] [CrossRef] [PubMed]
- Picton, H.M.; Harris, S.E.; Muruvi, W.; Chambers, E.L. The in vitro growth and maturation of follicles. Reproduction 2008, 136, 703–715. [Google Scholar] [CrossRef] [PubMed]
- Telfer, E.E.; McLaughlin, M.; Ding, C.; Thong, K.J. A two-step serum-free culture system supports development of human oocytes from primordial follicles in the presence of activin. Hum. Reprod. 2008, 23, 1151–1158. [Google Scholar] [CrossRef] [PubMed]
- Jutras, B.L.; Chenail, A.M.; Rowland, C.L.; Carroll, D.; Miller, M.C.; Bykowski, T.; Stevenson, B. Eubacterial SpoVG homologs constitute a new family of site-specific DNA-binding proteins. PLoS ONE 2013, 8, e66683. [Google Scholar] [CrossRef] [PubMed]
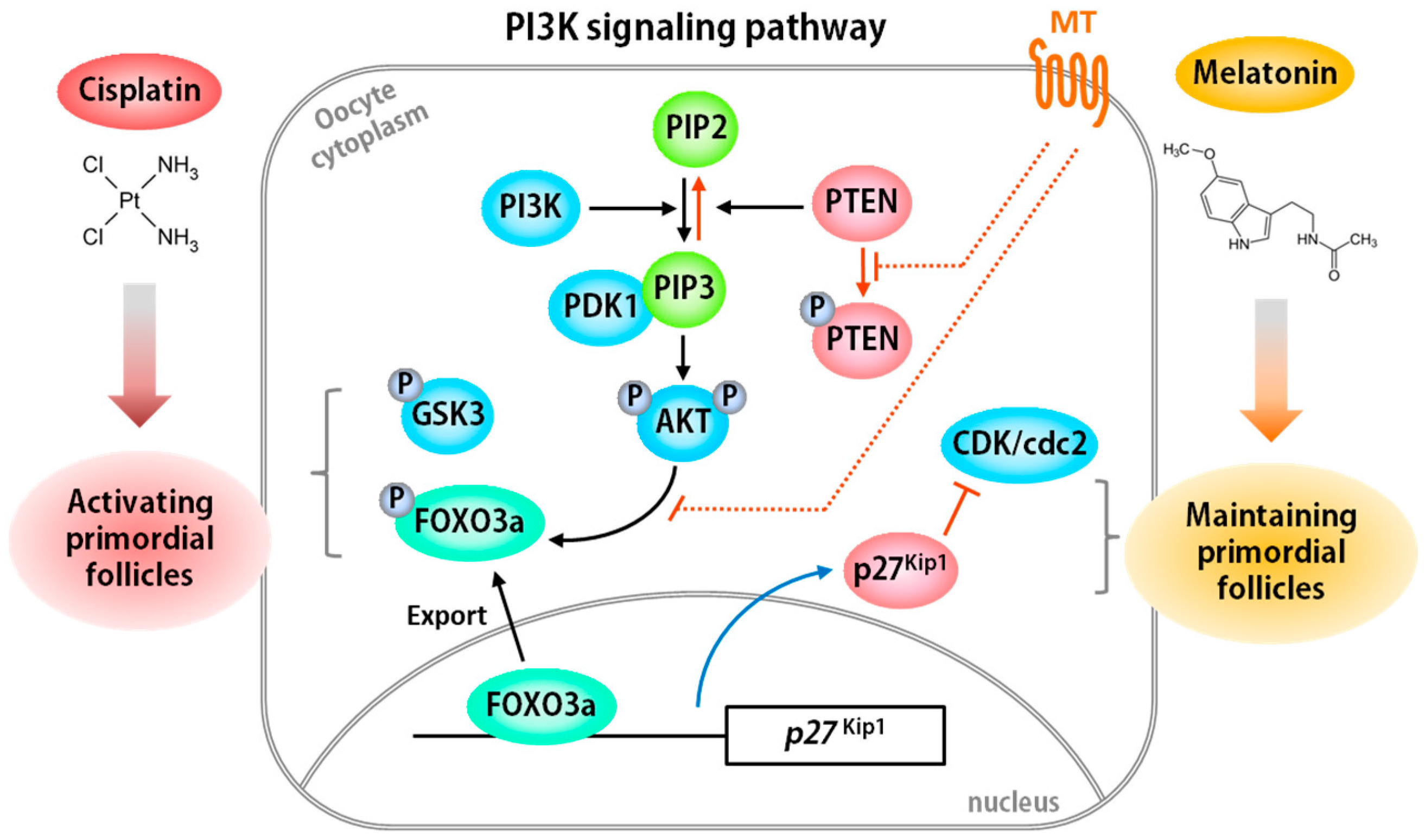
- Jang, H.; Lee, O.H.; Lee, Y.; Yoon, H.; Chang, E.M.; Park, M.; Lee, J.W.; Hong, K.; Kim, J.O.; Kim, N.K.; et al. Melatonin prevents cisplatin-induced primordial follicle loss via suppression of PTEN/AKT/FOXO3a pathway activation in the mouse ovary. J. Pineal Res. 2016, 60, 336–347. [Google Scholar] [CrossRef] [PubMed]
- Chang, E.M.; Lim, E.; Yoon, S.; Jeong, K.; Bae, S.; Lee, D.R.; Yoon, T.K.; Choi, Y.; Lee, W.S. Cisplatin induces overactivation of the dormant primordial follicle through PTEN/AKT/FOXO3a pathway which leads to loss of ovarian reserve in mice. PLoS ONE 2015, 10, e0144245. [Google Scholar] [CrossRef] [PubMed]
- Zwain, I.H.; Amato, P. cAMP-induced apoptosis in granulosa cells is associated with up-regulation of P53 and bax and down-regulation of clusterin. Endocr. Res. 2001, 27, 233–249. [Google Scholar] [CrossRef] [PubMed]
- Harris, S.L.; Levine, A.J. The p53 pathway: Positive and negative feedback loops. Oncogene 2005, 24, 2899–2908. [Google Scholar] [CrossRef] [PubMed]
- Depalo, R.; Nappi, L.; Loverro, G.; Bettocchi, S.; Caruso, M.L.; Valentini, A.M.; Selvaggi, L. Evidence of apoptosis in human primordial and primary follicles. Hum. Reprod. 2003, 18, 2678–2682. [Google Scholar] [CrossRef] [PubMed]
- Perez, G.I.; Knudson, C.M.; Leykin, L.; Korsmeyer, S.J.; Tilly, J.L. Apoptosis-associated signaling pathways are required for chemotherapy-mediated female germ cell destruction. Nat. Med. 1997, 3, 1228–1232. [Google Scholar] [CrossRef] [PubMed]
- Kurita, T.; Cunha, G.R.; Robboy, S.J.; Mills, A.A.; Medina, R.T. Differential expression of p63 isoforms in female reproductive organs. Mech. Dev. 2005, 122, 1043–1055. [Google Scholar] [CrossRef] [PubMed]
- Suh, E.K.; Yang, A.; Kettenbach, A.; Bamberger, C.; Michaelis, A.H.; Zhu, Z.; Elvin, J.A.; Bronson, R.T.; Crum, C.P.; McKeon, F. p63 protects the female germ line during meiotic arrest. Nature 2006, 444, 624–628. [Google Scholar] [CrossRef] [PubMed]
- Livera, G.; Petre-Lazar, B.; Guerquin, M.J.; Trautmann, E.; Coffigny, H.; Habert, R. p63 null mutation protects mouse oocytes from radio-induced apoptosis. Reproduction 2008, 135, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Kerr, J.B.; Hutt, K.J.; Michalak, E.M.; Cook, M.; Vandenberg, C.J.; Liew, S.H.; Bouillet, P.; Mills, A.; Scott, C.L.; Findlay, J.K.; et al. DNA damage-induced primordial follicle oocyte apoptosis and loss of fertility require TAp63-mediated induction of Puma and Noxa. Mol. Cell 2012, 48, 343–352. [Google Scholar] [CrossRef] [PubMed]
- Kharbanda, S.; Yuan, Z.M.; Weichselbaum, R.; Kufe, D. Determination of cell fate by c-Abl activation in the response to DNA damage. Oncogene 1998, 17, 3309–3318. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, K.; Miki, Y. Enabling death by the Abl tyrosine kinase: Mechanisms for nuclear shuttling of c-Abl in response to DNA damage. Cell Cycle 2005, 4, 777–779. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Cordeiro, M.H.; Serna, V.A.; Ebbert, K.; Butler, L.M.; Sinha, S.; Mills, A.A.; Woodruff, T.K.; Kurita, T. Rescue of platinum-damaged oocytes from programmed cell death through inactivation of the p53 family signaling network. Cell Death Differ. 2013, 20, 987–997. [Google Scholar] [CrossRef] [PubMed]
- Bolcun-Filas, E.; Rinaldi, V.D.; White, M.E.; Schimenti, J.C. Reversal of female infertility by Chk2 ablation reveals the oocyte DNA damage checkpoint pathway. Science 2014, 343, 533–536. [Google Scholar] [CrossRef] [PubMed]
- Reddy, P.; Liu, L.; Adhikari, D.; Jagarlamudi, K.; Rajareddy, S.; Shen, Y.; Du, C.; Tang, W.; Hamalainen, T.; Peng, S.L.; et al. Oocyte-specific deletion of PTEN causes premature activation of the primordial follicle pool. Science 2008, 319, 611–613. [Google Scholar] [CrossRef] [PubMed]
- Jagarlamudi, K.; Liu, L.; Adhikari, D.; Reddy, P.; Idahl, A.; Ottander, U.; Lundin, E.; Liu, K. Oocyte-specific deletion of PTEN in mice reveals a stage-specific function of PTEN/PI3K signaling in oocytes in controlling follicular activation. PLoS ONE 2009, 4, e6186. [Google Scholar] [CrossRef] [PubMed]
- Nakahata, S.; Ichikawa, T.; Maneesaay, P.; Saito, Y.; Nagai, K.; Tamura, T.; Manachai, N.; Yamakawa, N.; Hamasaki, M.; Kitabayashi, I.; et al. Loss of NDRG2 expression activates PI3K-AKT signalling via PTEN phosphorylation in ATLL and other cancers. Nat. Commun. 2014, 5, 3393. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.; Yunes, J.A.; Cardoso, B.A.; Martins, L.R.; Jotta, P.Y.; Abecasis, M.; Nowill, A.E.; Leslie, N.R.; Cardoso, A.A.; Barata, J.T. PTEN posttranslational inactivation and hyperactivation of the PI3K/Akt pathway sustain primary T cell leukemia viability. J. Clin. Investig. 2008, 118, 3762–3774. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Kawamura, K.; Cheng, Y.; Liu, S.; Klein, C.; Liu, S.; Duan, E.K.; Hsueh, A.J. Activation of dormant ovarian follicles to generate mature eggs. Proc. Natl. Acad. Sci. USA 2010, 107, 10280–10284. [Google Scholar] [CrossRef] [PubMed]
- Kawamura, K.; Cheng, Y.; Suzuki, N.; Deguchi, M.; Sato, Y.; Takae, S.; Ho, C.H.; Kawamura, N.; Tamura, M.; Hashimoto, S.; et al. Hippo signaling disruption and Akt stimulation of ovarian follicles for infertility treatment. Proc. Natl. Acad. Sci. USA 2013, 110, 17474–17479. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Nagaraju, G.; Liu, Z.; Liu, K. Functional roles of the phosphatidylinositol 3-kinases (PI3Ks) signaling in the mammalian ovary. Mol. Cell. Endocrinol. 2012, 356, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Groszer, M.; Erickson, R.; Scripture-Adams, D.D.; Lesche, R.; Trumpp, A.; Zack, J.A.; Kornblum, H.I.; Liu, X.; Wu, H. Negative regulation of neural stem/progenitor cell proliferation by the PTEN tumor suppressor gene in vivo. Science 2001, 294, 2186–2189. [Google Scholar] [CrossRef] [PubMed]
- Al-Khouri, A.M.; Ma, Y.; Togo, S.H.; Williams, S.; Mustelin, T. Cooperative phosphorylation of the tumor suppressor phosphatase and tensin homologue (PTEN) by casein kinases and glycogen synthase kinase 3β. J. Biol. Chem. 2005, 280, 35195–35202. [Google Scholar] [CrossRef] [PubMed]
- Ning, K.; Miller, L.C.; Laidlaw, H.A.; Burgess, L.A.; Perera, N.M.; Downes, C.P.; Leslie, N.R.; Ashford, M.L. A novel leptin signalling pathway via PTEN inhibition in hypothalamic cell lines and pancreatic β-cells. EMBO J. 2006, 25, 2377–2387. [Google Scholar] [CrossRef] [PubMed]
- Maccario, H.; Perera, N.M.; Davidson, L.; Downes, C.P.; Leslie, N.R. PTEN is destabilized by phosphorylation on Thr366. Biochem. J. 2007, 405, 439–444. [Google Scholar] [CrossRef] [PubMed]
- Manning, B.D.; Tee, A.R.; Logsdon, M.N.; Blenis, J.; Cantley, L.C. Identification of the tuberous sclerosis complex-2 tumor suppressor gene product tuberin as a target of the phosphoinositide 3-kinase/Akt pathway. Mol. Cell 2002, 10, 151–162. [Google Scholar] [CrossRef]
- Shioi, T.; McMullen, J.R.; Kang, P.M.; Douglas, P.S.; Obata, T.; Franke, T.F.; Cantley, L.C.; Izumo, S. Akt/protein kinase B promotes organ growth in transgenic mice. Mol. Cell. Biol. 2002, 22, 2799–2809. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, E.; McGraw, T.E. The Akt kinases: Isoform specificity in metabolism and cancer. Cell Cycle 2009, 8, 2502–2508. [Google Scholar] [CrossRef] [PubMed]
- Roness, H.; Gavish, Z.; Cohen, Y.; Meirow, D. Ovarian follicle burnout: A universal phenomenon? Cell Cycle 2013, 12, 3245–3246. [Google Scholar] [CrossRef] [PubMed]
- Castrillon, D.H.; Miao, L.; Kollipara, R.; Horner, J.W.; DePinho, R.A. Suppression of ovarian follicle activation in mice by the transcription factor Foxo3a. Science 2003, 301, 215–218. [Google Scholar] [CrossRef] [PubMed]
- Dijkers, P.F.; Medema, R.H.; Pals, C.; Banerji, L.; Thomas, N.S.; Lam, E.W.; Burgering, B.M.; Raaijmakers, J.A.; Lammers, J.W.; Koenderman, L.; et al. Forkhead transcription factor FKHR-L1 modulates cytokine-dependent transcriptional regulation of p27(KIP1). Mol. Cell. Biol. 2000, 20, 9138–9148. [Google Scholar] [CrossRef] [PubMed]
- Sherr, C.J.; Roberts, J.M. CDK inhibitors: Positive and negative regulators of G1-phase progression. Genes Dev. 1999, 13, 1501–1512. [Google Scholar] [CrossRef] [PubMed]
- Rajareddy, S.; Reddy, P.; Du, C.; Liu, L.; Jagarlamudi, K.; Tang, W.; Shen, Y.; Berthet, C.; Peng, S.L.; Kaldis, P.; et al. p27kip1 (cyclin-dependent kinase inhibitor 1B) controls ovarian development by suppressing follicle endowment and activation and promoting follicle atresia in mice. Mol. Endocrinol. 2007, 21, 2189–2202. [Google Scholar] [CrossRef] [PubMed]
- Stehle, J.; Reuss, S.; Riemann, R.; Seidel, A.; Vollrath, L. The role of arginine-vasopressin for pineal melatonin synthesis in the rat: Involvement of vasopressinergic receptors. Neurosci. Lett. 1991, 123, 131–134. [Google Scholar] [CrossRef]
- Venegas, C.; Garcia, J.A.; Escames, G.; Ortiz, F.; Lopez, A.; Doerrier, C.; Garcia-Corzo, L.; Lopez, L.C.; Reiter, R.J.; Acuna-Castroviejo, D. Extrapineal melatonin: Analysis of its subcellular distribution and daily fluctuations. J. Pineal Res. 2012, 52, 217–227. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Rosales-Corral, S.A.; Manchester, L.C.; Tan, D.X. Peripheral reproductive organ health and melatonin: Ready for prime time. Int. J. Mol. Sci. 2013, 14, 7231–7272. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Manchester, L.C.; Liu, X.; Rosales-Corral, S.A.; Acuna-Castroviejo, D.; Reiter, R.J. Mitochondria and chloroplasts as the original sites of melatonin synthesis: A hypothesis related to melatonin’s primary function and evolution in eukaryotes. J. Pineal Res. 2013, 54, 127–138. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Tan, D.X.; Tamura, H.; Cruz, M.H.; Fuentes-Broto, L. Clinical relevance of melatonin in ovarian and placental physiology: A review. Gynecol. Endocrinol. 2014, 30, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Tan, D.X.; Korkmaz, A.; Rosales-Corral, S.A. Melatonin and stable circadian rhythms optimize maternal, placental and fetal physiology. Hum. Reprod. Update 2014, 20, 293–307. [Google Scholar] [CrossRef] [PubMed]
- Hardeland, R.; Tan, D.X.; Reiter, R.J. Kynuramines, metabolites of melatonin and other indoles: The resurrection of an almost forgotten class of biogenic amines. J. Pineal Res. 2009, 47, 109–126. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Manchester, L.C.; Terron, M.P.; Flores, L.J.; Reiter, R.J. One molecule, many derivatives: A never-ending interaction of melatonin with reactive oxygen and nitrogen species? J. Pineal Res. 2007, 42, 28–42. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.M.; Zhang, Y. Melatonin: A well-documented antioxidant with conditional pro-oxidant actions. J. Pineal Res. 2014, 57, 131–146. [Google Scholar] [CrossRef] [PubMed]
- Galano, A.; Medina, M.E.; Tan, D.X.; Reiter, R.J. Melatonin and its metabolites as copper chelating agents and their role in inhibiting oxidative stress: A physicochemical analysis. J. Pineal Res. 2015, 58, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J. Oxidative damage in the central nervous system: Protection by melatonin. Prog. Neurobiol. 1998, 56, 359–384. [Google Scholar] [CrossRef]
- Reiter, R.J.; Tan, D.X.; Allegra, M. Melatonin: Reducing molecular pathology and dysfunction due to free radicals and associated reactants. Neuro Endocrinol. Lett. 2002, 23, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Tan, D.X.; Rosales-Corral, S.; Manchester, L.C. The universal nature, unequal distribution and antioxidant functions of melatonin and its derivatives. Mini Rev. Med. Chem. 2013, 13, 373–384. [Google Scholar] [CrossRef] [PubMed]
- Acuna-Castroviejo, D.; Escames, G.; Venegas, C.; Diaz-Casado, M.E.; Lima-Cabello, E.; Lopez, L.C.; Rosales-Corral, S.; Tan, D.X.; Reiter, R.J. Extrapineal melatonin: Sources, regulation, and potential functions. Cell. Mol. Life Sci. 2014, 71, 2997–3025. [Google Scholar] [CrossRef] [PubMed]
- Parlakpinar, H.; Sahna, E.; Ozer, M.K.; Ozugurlu, F.; Vardi, N.; Acet, A. Physiological and pharmacological concentrations of melatonin protect against cisplatin-induced acute renal injury. J. Pineal Res. 2002, 33, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Kilic, U.; Kilic, E.; Tuzcu, Z.; Tuzcu, M.; Ozercan, I.H.; Yilmaz, O.; Sahin, F.; Sahin, K. Melatonin suppresses cisplatin-induced nephrotoxicity via activation of Nrf-2/HO-1 pathway. Nutr. Metab. 2013, 10, 7. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Tan, D.X.; Pappolla, M.A. Melatonin relieves the neural oxidative burden that contributes to dementias. Ann. N. Y Acad. Sci. 2004, 1035, 179–196. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Wu, Q.Q.; Cao, L.F.; Qing, H.Y.; Zhang, C.; Chen, Y.H.; Wang, H.; Liu, R.Y.; Xu, D.X. Melatonin inhibits endoplasmic reticulum stress and epithelial-mesenchymal transition during bleomycin-induced pulmonary fibrosis in mice. PLoS ONE 2014, 9, e97266. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Wang, J.; Li, Y.; Zhu, K.; Xu, Z.; Song, Y.; Song, Y.; Liu, G. Melatonin-related genes expressed in the mouse uterus during early gestation promote embryo implantation. J. Pineal Res. 2015, 58, 300–309. [Google Scholar] [CrossRef] [PubMed]
- Dair, E.L.; Simoes, R.S.; Simoes, M.J.; Romeu, L.R.; Oliveira-Filho, R.M.; Haidar, M.A.; Baracat, E.C.; Soares, J.M. Effects of melatonin on the endometrial morphology and embryo implantation in rats. Fertil. Steril. 2008, 89, 1299–1305. [Google Scholar] [CrossRef] [PubMed]
- Atessahin, A.; Sahna, E.; Turk, G.; Ceribasi, A.O.; Yilmaz, S.; Yuce, A.; Bulmus, O. Chemoprotective effect of melatonin against cisplatin-induced testicular toxicity in rats. J. Pineal Res. 2006, 41, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Galano, A.; Tan, D.X.; Reiter, R.J. Melatonin as a natural ally against oxidative stress: A physicochemical examination. J. Pineal Res. 2011, 51, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Richardson, B.A.; Johnson, L.Y.; Ferguson, B.N.; Dinh, D.T. Pineal melatonin rhythm: Reduction in aging Syrian hamsters. Science 1980, 210, 1372–1373. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Craft, C.M.; Johnson, J.E., Jr.; King, T.S.; Richardson, B.A.; Vaughan, G.M.; Vaughan, M.K. Age-associated reduction in nocturnal pineal melatonin levels in female rats. Endocrinology 1981, 109, 1295–1297. [Google Scholar] [CrossRef] [PubMed]
- Coto-Montes, A.; Boga, J.A.; Tan, D.X.; Reiter, R.J. Melatonin as a potential agent in the treatment of sarcopenia. Int. J. Mol. Sci. 2016, 17, 1771. [Google Scholar] [CrossRef] [PubMed]
- Bubenik, G.A.; Konturek, S.J. Melatonin and aging: Prospects for human treatment. J. Physiol. Pharmacol. 2011, 62, 13–19. [Google Scholar] [PubMed]
- Hill, S.M.; Cheng, C.; Yuan, L.; Mao, L.; Jockers, R.; Dauchy, B.; Blask, D.E. Age-related decline in melatonin and its MT1 receptor are associated with decreased sensitivity to melatonin and enhanced mammary tumor growth. Curr. Aging Sci. 2013, 6, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Caballero, B.; Vega-Naredo, I.; Sierra, V.; Huidobro-Fernandez, C.; Soria-Valles, C.; de Gonzalo-Calvo, D.; Tolivia, D.; Gutierrez-Cuesta, J.; Pallas, M.; Camins, A.; et al. Favorable effects of a prolonged treatment with melatonin on the level of oxidative damage and neurodegeneration in senescence-accelerated mice. J. Pineal Res. 2008, 45, 302–311. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Barcelo, E.J.; Mediavilla, M.D.; Tan, D.X.; Reiter, R.J. Clinical uses of melatonin: Evaluation of human trials. Curr. Med. Chem. 2010, 17, 2070–2095. [Google Scholar] [CrossRef] [PubMed]
- Mockova, K.; Mnichova, M.; Kubatka, P.; Bojkova, B.; Ahlers, I.; Ahlersova, E. Mammary carcinogenesis induced in Wistar:han rats by the combination of ionizing radiation and dimethylbenz(a)anthracene: Prevention with melatonin. Neoplasma 2000, 47, 227–229. [Google Scholar] [PubMed]
- Majidinia, M.; Sadeghpour, A.; Mehrzadi, S.; Reiter, R.J.; Khatami, N.; Yousefi, B. Melatonin: A pleiotropic molecule that modulates DNA damage response and repair pathways. J. Pineal Res. 2017, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.; Adhikari, J.S.; Rizvi, M.A.; Chaudhury, N.K. Radioprotective potential of melatonin against 60Co γ-ray-induced testicular injury in male C57BL/6 mice. J. Biomed. Sci. 2015, 22, 61. [Google Scholar] [CrossRef] [PubMed]
- Cos, S.; Mediavilla, M.D.; Fernandez, R.; Gonzalez-Lamuno, D.; Sanchez-Barcelo, E.J. Does melatonin induce apoptosis in MCF-7 human breast cancer cells in vitro? J. Pineal Res. 2002, 32, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Martin-Renedo, J.; Mauriz, J.L.; Jorquera, F.; Ruiz-Andres, O.; Gonzalez, P.; Gonzalez-Gallego, J. Melatonin induces cell cycle arrest and apoptosis in hepatocarcinoma HepG2 cell line. J. Pineal Res. 2008, 45, 532–540. [Google Scholar] [CrossRef] [PubMed]
- Mediavilla, M.D.; Cos, S.; Sanchez-Barcelo, E.J. Melatonin increases p53 and p21WAF1 expression in MCF-7 human breast cancer cells in vitro. Life Sci. 1999, 65, 415–420. [Google Scholar] [CrossRef]
- Proietti, S.; Cucina, A.; Dobrowolny, G.; D’Anselmi, F.; Dinicola, S.; Masiello, M.G.; Pasqualato, A.; Palombo, A.; Morini, V.; Reiter, R.J.; et al. Melatonin down-regulates MDM2 gene expression and enhances p53 acetylation in MCF-7 cells. J. Pineal Res. 2014, 57, 120–129. [Google Scholar] [CrossRef] [PubMed]
- Santoro, R.; Marani, M.; Blandino, G.; Muti, P.; Strano, S. Melatonin triggers p53Ser phosphorylation and prevents DNA damage accumulation. Oncogene 2012, 31, 2931–2942. [Google Scholar] [CrossRef] [PubMed]
- Santoro, R.; Mori, F.; Marani, M.; Grasso, G.; Cambria, M.A.; Blandino, G.; Muti, P.; Strano, S. Blockage of melatonin receptors impairs p53-mediated prevention of DNA damage accumulation. Carcinogenesis 2013, 34, 1051–1061. [Google Scholar] [CrossRef] [PubMed]
- Reppert, S.M.; Weaver, D.R.; Ebisawa, T.; Mahle, C.D.; Kolakowski, L.F., Jr. Cloning of a melatonin-related receptor from human pituitary. FEBS Lett. 1996, 386, 219–224. [Google Scholar] [CrossRef]
- Overington, J.P.; Al-Lazikani, B.; Hopkins, A.L. How many drug targets are there? Nat. Rev. Drug Discov. 2006, 5, 993–996. [Google Scholar] [CrossRef] [PubMed]
- Kilic, E.; Kilic, U.; Reiter, R.J.; Bassetti, C.L.; Hermann, D.M. Prophylactic use of melatonin protects against focal cerebral ischemia in mice: Role of endothelin converting enzyme-1. J. Pineal Res. 2004, 37, 247–251. [Google Scholar] [CrossRef] [PubMed]
- Cerezo, A.B.; Hornedo-Ortega, R.; Alvarez-Fernandez, M.A.; Troncoso, A.M.; Garcia-Parrilla, M.C. Inhibition of VEGF-Induced VEGFR-2 activation and HUVEC migration by melatonin and other bioactive indolic compounds. Nutrients 2017, 9, 249. [Google Scholar] [CrossRef] [PubMed]
- Lissoni, P.; Rovelli, F.; Malugani, F.; Bucovec, R.; Conti, A.; Maestroni, G.J. Anti-angiogenic activity of melatonin in advanced cancer patients. Neurol. Endocrinol. Lett. 2001, 22, 45–47. [Google Scholar] [PubMed]
- Jardim-Perassi, B.V.; Arbab, A.S.; Ferreira, L.C.; Borin, T.F.; Varma, N.R.; Iskander, A.S.; Shankar, A.; Ali, M.M.; de Campos Zuccari, D.A. Effect of melatonin on tumor growth and angiogenesis in xenograft model of breast cancer. PLoS ONE 2014, 9, e85311. [Google Scholar] [CrossRef] [PubMed]
- Castle-Miller, J.; Bates, D.O.; Tortonese, D.J. Mechanisms regulating angiogenesis underlie seasonal control of pituitary function. Proc. Natl. Acad. Sci. USA 2017, 114, 2514–2523. [Google Scholar] [CrossRef] [PubMed]
- Miller, S.C.; Pandi-Perumal, S.R.; Esquifino, A.I.; Cardinali, D.P.; Maestroni, G.J. The role of melatonin in immuno-enhancement: Potential application in cancer. Int. J. Exp. Pathol. 2006, 87, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Konakchieva, R.; Kyurkchiev, S.; Kehayov, I.; Taushanova, P.; Kanchev, L. Selective effect of methoxyindoles on the lymphocyte proliferation and melatonin binding to activated human lymphoid cells. J. Neuroimmunol. 1995, 63, 125–132. [Google Scholar] [CrossRef]
- Pioli, C.; Caroleo, M.C.; Nistico, G.; Doria, G. Melatonin increases antigen presentation and amplifies specific and non specific signals for T-cell proliferation. Int. J. Immunopharmacol. 1993, 15, 463–468. [Google Scholar] [CrossRef]
- Raghavendra, V.; Singh, V.; Shaji, A.V.; Vohra, H.; Kulkarni, S.K.; Agrewala, J.N. Melatonin provides signal 3 to unprimed CD4+ T cells but failed to stimulate LPS primed B cells. Clin. Exp. Immunol. 2001, 124, 414–422. [Google Scholar] [CrossRef] [PubMed]
- Drazen, D.L.; Bilu, D.; Bilbo, S.D.; Nelson, R.J. Melatonin enhancement of splenocyte proliferation is attenuated by luzindole, a melatonin receptor antagonist. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2001, 280, 1476–1482. [Google Scholar]
- Wajs, E.; Kutoh, E.; Gupta, D. Melatonin affects proopiomelanocortin gene expression in the immune organs of the rat. Eur. J. Endocrinol. 1995, 133, 754–760. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Maurino, S.; Gonzalez-Haba, M.G.; Calvo, J.R.; Rafii-El-Idrissi, M.; Sanchez-Margalet, V.; Goberna, R.; Guerrero, J.M. Melatonin enhances IL-2, IL-6, and IFN-γ production by human circulating CD4+ cells: A possible nuclear receptor-mediated mechanism involving T helper type 1 lymphocytes and monocytes. J. Immunol. 1997, 159, 574–581. [Google Scholar] [PubMed]
- Liu, F.; Ng, T.B.; Fung, M.C. Pineal indoles stimulate the gene expression of immunomodulating cytokines. J. Neural Transm. 2001, 108, 397–405. [Google Scholar] [CrossRef] [PubMed]
- Currier, N.L.; Sun, L.Z.; Miller, S.C. Exogenous melatonin: Quantitative enhancement in vivo of cells mediating non-specific immunity. J. Neuroimmunol. 2000, 104, 101–108. [Google Scholar] [CrossRef]
- Currier, N.L.; Miller, S.C. Echinacea purpurea and melatonin augment natural-killer cells in leukemic mice and prolong life span. J. Altern. Complement. Med. 2001, 7, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Yoo, Y.M.; Jang, S.K.; Kim, G.H.; Park, J.Y.; Joo, S.S. Pharmacological advantages of melatonin in immunosenescence by improving activity of T lymphocytes. J. Biomed. Res. 2016, 30, 314–321. [Google Scholar] [PubMed]
- Agarwal, A.; Gupta, S.; Sharma, R.K. Role of oxidative stress in female reproduction. Reprod. Biol. Endocrinol. 2005, 3, 28. [Google Scholar] [CrossRef] [PubMed]
- Pham-Huy, L.A.; He, H.; Pham-Huy, C. Free radicals, antioxidants in disease and health. Int. J. Biomed. Sci. 2008, 4, 89–96. [Google Scholar] [PubMed]
- Ruder, E.H.; Hartman, T.J.; Blumberg, J.; Goldman, M.B. Oxidative stress and antioxidants: Exposure and impact on female fertility. Hum. Reprod. Update 2008, 14, 345–357. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.; Rajkovic, A. Genetics of early mammalian folliculogenesis. Cell. Mol. Life Sci. 2006, 63, 579–590. [Google Scholar] [CrossRef] [PubMed]
- Lim, E.J.; Choi, Y. Transcription factors in the maintenance and survival of primordial follicles. Clin. Exp. Reprod. Med. 2012, 39, 127–131. [Google Scholar] [CrossRef] [PubMed]
- Tamura, H.; Takasaki, A.; Miwa, I.; Taniguchi, K.; Maekawa, R.; Asada, H.; Taketani, T.; Matsuoka, A.; Yamagata, Y.; Shimamura, K.; et al. Oxidative stress impairs oocyte quality and melatonin protects oocytes from free radical damage and improves fertilization rate. J. Pineal Res. 2008, 44, 280–287. [Google Scholar] [CrossRef] [PubMed]
- Tamura, H.; Nakamura, Y.; Korkmaz, A.; Manchester, L.C.; Tan, D.X.; Sugino, N.; Reiter, R.J. Melatonin and the ovary: Physiological and pathophysiological implications. Fertil. Steril. 2009, 92, 328–343. [Google Scholar] [CrossRef] [PubMed]
- Tamura, H.; Takasaki, A.; Taketani, T.; Tanabe, M.; Kizuka, F.; Lee, L.; Tamura, I.; Maekawa, R.; Aasada, H.; Yamagata, Y.; et al. The role of melatonin as an antioxidant in the follicle. J. Ovarian Res. 2012, 5, 5. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.K.; Park, E.A.; Kim, H.J.; Choi, W.Y.; Cho, J.H.; Lee, W.S.; Cha, K.Y.; Kim, Y.S.; Lee, D.R.; Yoon, T.K. Does supplementation of in vitro culture medium with melatonin improve IVF outcome in PCOS? Reprod. Biomed. Online 2013, 26, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Ma, M.; Chen, X.Y.; Li, B.; Li, X.T. Melatonin protects premature ovarian insufficiency induced by tripterygium glycosides: Role of SIRT1. Am. J. Transl. Res. 2017, 9, 1580–1602. [Google Scholar] [PubMed]
- Chattoraj, A.; Seth, M.; Maitra, S.K. Localization and dynamics of Mel(1a) melatonin receptor in the ovary of carp Catla catla in relation to serum melatonin levels. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2009, 152, 327–333. [Google Scholar] [CrossRef] [PubMed]
- Jablonska, K.; Pula, B.; Zemla, A.; Kobierzycki, C.; Kedzia, W.; Nowak-Markwitz, E.; Spaczynski, M.; Zabel, M.; Podhorska-Okolow, M.; Dziegiel, P. Expression of the MT1 melatonin receptor in ovarian cancer cells. Int. J. Mol. Sci. 2014, 15, 23074–23089. [Google Scholar] [CrossRef] [PubMed]
- Soares, J.M., Jr.; Masana, M.I.; Ersahin, C.; Dubocovich, M.L. Functional melatonin receptors in rat ovaries at various stages of the estrous cycle. J. Pharmacol. Exp. Ther. 2003, 306, 694–702. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Tan, D.X.; Sainz, R.M.; Mayo, J.C.; Lopez-Burillo, S. Melatonin: Reducing the toxicity and increasing the efficacy of drugs. J. Pharm Pharmacol. 2002, 54, 1299–1321. [Google Scholar] [CrossRef] [PubMed]
- Mills, E.; Wu, P.; Seely, D.; Guyatt, G. Melatonin in the treatment of cancer: A systematic review of randomized controlled trials and meta-analysis. J. Pineal Res. 2005, 39, 360–366. [Google Scholar] [CrossRef] [PubMed]
- Casado-Zapico, S.; Rodriguez-Blanco, J.; Garcia-Santos, G.; Martin, V.; Sanchez-Sanchez, A.M.; Antolin, I.; Rodriguez, C. Synergistic antitumor effect of melatonin with several chemotherapeutic drugs on human Ewing sarcoma cancer cells: Potentiation of the extrinsic apoptotic pathway. J. Pineal Res. 2010, 48, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Manchester, L.C.; Reiter, R.J.; Plummer, B.F.; Hardies, L.J.; Weintraub, S.T.; Shepherd, A.M. A novel melatonin metabolite, cyclic 3-hydroxymelatonin: A biomarker of in vivo hydroxyl radical generation. Biochem. Biophys. Res. Commun. 1998, 253, 614–620. [Google Scholar] [CrossRef] [PubMed]
- Pariente, R.; Pariente, J.A.; Rodriguez, A.B.; Espino, J. Melatonin sensitizes human cervical cancer HeLa cells to cisplatin-induced cytotoxicity and apoptosis: Effects on oxidative stress and DNA fragmentation. J. Pineal Res. 2016, 60, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.J.; Do, B.R.; Lee, Y.H.; Park, J.H.; Kim, S.J.; Kim, J.K.; Roh, S.I.; Yoon, Y.D.; Yoon, H.S. Ovarian expression of melatonin Mel(1a) receptor mRNA during mouse development. Mol. Reprod. Dev. 2001, 59, 126–132. [Google Scholar] [CrossRef] [PubMed]
- Niles, L.P.; Wang, J.; Shen, L.; Lobb, D.K.; Younglai, E.V. Melatonin receptor mRNA expression in human granulosa cells. Mol. Cell. Endocrinol. 1999, 156, 107–110. [Google Scholar] [CrossRef]
- Wang, S.J.; Liu, W.J.; Wu, C.J.; Ma, F.H.; Ahmad, S.; Liu, B.R.; Han, L.; Jiang, X.P.; Zhang, S.J.; Yang, L.G. Melatonin suppresses apoptosis and stimulates progesterone production by bovine granulosa cells via its receptors (MT1 and MT2). Theriogenology 2012, 78, 1517–1526. [Google Scholar] [CrossRef] [PubMed]
- Woo, M.M.; Tai, C.J.; Kang, S.K.; Nathwani, P.S.; Pang, S.F.; Leung, P.C. Direct action of melatonin in human granulosa-luteal cells. J. Clin. Endocrinol. Metab. 2001, 86, 4789–4797. [Google Scholar] [CrossRef] [PubMed]
- Kong, P.J.; Byun, J.S.; Lim, S.Y.; Lee, J.J.; Hong, S.J.; Kwon, K.J.; Kim, S.S. Melatonin induces Akt phosphorylation through melatonin receptor- and PI3K-dependent pathways in primary astrocytes. Korean J. Physiol. Pharmacol. 2008, 12, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Kilic, U.; Caglayan, A.B.; Beker, M.C.; Gunal, M.Y.; Caglayan, B.; Yalcin, E.; Kelestemur, T.; Gundogdu, R.Z.; Yulug, B.; Yilmaz, B.; et al. Particular phosphorylation of PI3K/Akt on Thr308 via PDK-1 and PTEN mediates melatonin’s neuroprotective activity after focal cerebral ischemia in mice. Redox Biol. 2017, 12, 657–665. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y.; Tamura, H.; Takayama, H.; Kato, H. Increased endogenous level of melatonin in preovulatory human follicles does not directly influence progesterone production. Fertil. Steril. 2003, 80, 1012–1016. [Google Scholar] [CrossRef]
- Shi, J.M.; Tian, X.Z.; Zhou, G.B.; Wang, L.; Gao, C.; Zhu, S.E.; Zeng, S.M.; Tian, J.H.; Liu, G.S. Melatonin exists in porcine follicular fluid and improves in vitro maturation and parthenogenetic development of porcine oocytes. J. Pineal Res. 2009, 47, 318–323. [Google Scholar] [CrossRef] [PubMed]
- Morita, Y.; Perez, G.I.; Paris, F.; Miranda, S.R.; Ehleiter, D.; Haimovitz-Friedman, A.; Fuks, Z.; Xie, Z.; Reed, J.C.; Schuchman, E.H.; et al. Oocyte apoptosis is suppressed by disruption of the acid sphingomyelinase gene or by sphingosine-1-phosphate therapy. Nat. Med. 2000, 6, 1109–1114. [Google Scholar] [PubMed]
- Hancke, K.; Strauch, O.; Kissel, C.; Gobel, H.; Schafer, W.; Denschlag, D. Sphingosine 1-phosphate protects ovaries from chemotherapy-induced damage in vivo. Fertil. Steril. 2007, 87, 172–177. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Turan, V.; Lierman, S.; Cuvelier, C.; de Sutter, P.; Oktay, K. Sphingosine-1-phosphate prevents chemotherapy-induced human primordial follicle death. Hum. Reprod. 2014, 29, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Kaya, H.; Desdicioglu, R.; Sezik, M.; Ulukaya, E.; Ozkaya, O.; Yilmaztepe, A.; Demirci, M. Does sphingosine-1-phosphate have a protective effect on cyclophosphamide- and irradiation-induced ovarian damage in the rat model? Fertil. Steril. 2008, 89, 732–735. [Google Scholar] [CrossRef] [PubMed]
- Crespo, I.; San-Miguel, B.; Sanchez, D.I.; Gonzalez-Fernandez, B.; Alvarez, M.; Gonzalez-Gallego, J.; Tunon, M.J. Melatonin inhibits the sphingosine kinase 1/sphingosine-1-phosphate signaling pathway in rabbits with fulminant hepatitis of viral origin. J. Pineal Res. 2016, 61, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, D.I.; Gonzalez-Fernandez, B.; San-Miguel, B.; de Urbina, J.O.; Crespo, I.; Gonzalez-Gallego, J.; Tunon, M.J. Melatonin prevents deregulation of the sphingosine kinase/sphingosine 1-phosphate signaling pathway in a mouse model of diethylnitrosamine-induced hepatocellular carcinoma. J. Pineal Res. 2017, 62, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Fernandez, B.; Sanchez, D.I.; Crespo, I.; San-Miguel, B.; Alvarez, M.; Tunon, M.J.; Gonzalez-Gallego, J. Inhibition of the SphK1/S1P signaling pathway by melatonin in mice with liver fibrosis and human hepatic stellate cells. BioFactors 2017, 43, 272–282. [Google Scholar] [CrossRef] [PubMed]
- Kerr, J.B.; Hutt, K.J.; Cook, M.; Speed, T.P.; Strasser, A.; Findlay, J.K.; Scott, C.L. Cisplatin-induced primordial follicle oocyte killing and loss of fertility are not prevented by imatinib. Nat. Med. 2012, 18, 1170–1172. [Google Scholar] [CrossRef] [PubMed]
- Maiani, E.; di Bartolomeo, C.; Klinger, F.G.; Cannata, S.M.; Bernardini, S.; Chateauvieux, S.; Mack, F.; Mattei, M.; de Felici, M.; Diederich, M.; et al. Reply to: Cisplatin-induced primordial follicle oocyte killing and loss of fertility are not prevented by imatinib. Nat. Med. 2012, 18, 1172–1174. [Google Scholar] [CrossRef] [PubMed]
- Ting, A.Y.; Petroff, B.K. Tamoxifen decreases ovarian follicular loss from experimental toxicant DMBA and chemotherapy agents cyclophosphamide and doxorubicin in the rat. J. Assisted Reprod. Genet. 2010, 27, 591–597. [Google Scholar] [CrossRef] [PubMed]
- Lissoni, P.; Barni, S.; Meregalli, S.; Fossati, V.; Cazzaniga, M.; Esposti, D.; Tancini, G. Modulation of cancer endocrine therapy by melatonin: A phase II study of tamoxifen plus melatonin in metastatic breast cancer patients progressing under tamoxifen alone. Br. J. Cancer 1995, 71, 854–856. [Google Scholar] [CrossRef] [PubMed]
- Lissoni, P.; Ardizzoia, A.; Barni, S.; Paolorossi, F.; Tancini, G.; Meregalli, S.; Esposti, D.; Zubelewicz, B.; Braczowski, R. A randomized study of tamoxifen alone versus tamoxifen plus melatonin in estrogen receptor-negative heavily pretreated metastatic breast-cancer patients. Oncol. Rep. 1995, 2, 871–873. [Google Scholar] [CrossRef] [PubMed]
- Lissoni, P.; Paolorossi, F.; Tancini, G.; Ardizzoia, A.; Barni, S.; Brivio, F.; Maestroni, G.J.; Chilelli, M. A phase II study of tamoxifen plus melatonin in metastatic solid tumour patients. Br. J. Cancer 1996, 74, 1466–1468. [Google Scholar] [CrossRef] [PubMed]
- Garcia, J.J.; Reiter, R.J.; Ortiz, G.G.; Oh, C.S.; Tang, L.; Yu, B.P.; Escames, G. Melatonin enhances tamoxifen’s ability to prevent the reduction in microsomal membrane fluidity induced by lipid peroxidation. J. Membr. Biol. 1998, 162, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Dauchy, R.T.; Xiang, S.; Mao, L.; Brimer, S.; Wren, M.A.; Yuan, L.; Anbalagan, M.; Hauch, A.; Frasch, T.; Rowan, B.G.; et al. Circadian and melatonin disruption by exposure to light at night drives intrinsic resistance to tamoxifen therapy in breast cancer. Cancer Res. 2014, 74, 4099–4110. [Google Scholar] [CrossRef] [PubMed]
- Meirow, D.; Assad, G.; Dor, J.; Rabinovici, J. The GnRH antagonist cetrorelix reduces cyclophosphamide-induced ovarian follicular destruction in mice. Hum. Reprod. 2004, 19, 1294–1299. [Google Scholar] [CrossRef] [PubMed]
- Kishk, E.A.; Mohammed Ali, M.H. Effect of a gonadotropin-releasing hormone analogue on cyclophosphamide-induced ovarian toxicity in adult mice. Arch. Gynecol. Obstet. 2013, 287, 1023–1029. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Kang, X.; Deng, Q.; Cai, J.; Wang, Z. Combination of a GnRH agonist with an antagonist prevents flare-up effects and protects primordial ovarian follicles in the rat ovary from cisplatin-induced toxicity: A controlled experimental animal study. Reprod. Biol. Endocrinol. 2013, 11, 16. [Google Scholar] [CrossRef] [PubMed]
- Del Mastro, L.; Boni, L.; Michelotti, A.; Gamucci, T.; Olmeo, N.; Gori, S.; Giordano, M.; Garrone, O.; Pronzato, P.; Bighin, C.; et al. Effect of the gonadotropin-releasing hormone analogue triptorelin on the occurrence of chemotherapy-induced early menopause in premenopausal women with breast cancer: A randomized trial. JAMA 2011, 306, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Badawy, A.; Elnashar, A.; El-Ashry, M.; Shahat, M. Gonadotropin-releasing hormone agonists for prevention of chemotherapy-induced ovarian damage: Prospective randomized study. Fertil. Steril. 2009, 91, 694–697. [Google Scholar] [CrossRef] [PubMed]
- Elgindy, E.A.; El-Haieg, D.O.; Khorshid, O.M.; Ismail, E.I.; Abdelgawad, M.; Sallam, H.N.; Abou-Setta, A.M. Gonadatrophin suppression to prevent chemotherapy-induced ovarian damage: A randomized controlled trial. Obstet. Gynecol. 2013, 121, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Gerber, B.; von Minckwitz, G.; Stehle, H.; Reimer, T.; Felberbaum, R.; Maass, N.; Fischer, D.; Sommer, H.L.; Conrad, B.; Ortmann, O.; et al. Effect of luteinizing hormone-releasing hormone agonist on ovarian function after modern adjuvant breast cancer chemotherapy: The GBG 37 ZORO study. J. Clin. Oncol. 2011, 29, 2334–2341. [Google Scholar] [CrossRef] [PubMed]
- Sverrisdottir, A.; Nystedt, M.; Johansson, H.; Fornander, T. Adjuvant goserelin and ovarian preservation in chemotherapy treated patients with early breast cancer: Results from a randomized trial. Breast Cancer Res. Treat. 2009, 117, 561–567. [Google Scholar] [CrossRef] [PubMed]
- Munster, P.N.; Moore, A.P.; Ismail-Khan, R.; Cox, C.E.; Lacevic, M.; Gross-King, M.; Xu, P.; Carter, W.B.; Minton, S.E. Randomized trial using gonadotropin-releasing hormone agonist triptorelin for the preservation of ovarian function during (neo)adjuvant chemotherapy for breast cancer. J. Clin. Oncol. 2012, 30, 533–538. [Google Scholar] [CrossRef] [PubMed]
- Diaz, E.; Pazo, D.; Esquifino, A.I.; Diaz, B. Effects of ageing and exogenous melatonin on pituitary responsiveness to GnRH in rats. J. Reprod. Fertil. 2000, 119, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Diaz, E.; Castrillon, P.; Esquifino, A.; Diaz, B. Effect of prenatal melatonin on the gonadotropin and prolactin response to the feedback effect of testosterone in male offspring. J. Steroid Biochem. Mol. Biol. 2000, 72, 61–69. [Google Scholar] [CrossRef]
- Montero, R.; Gonsebatt, M.E.; Gerson, R.; Rojas, E.; Herrera, L.A.; Ostrosky-Wegman, P. AS-101: A modulator of in vitro T-cell proliferation. Anti-Cancer Drugs 1993, 4, 351–354. [Google Scholar] [CrossRef] [PubMed]
- Makarovsky, D.; Kalechman, Y.; Sonino, T.; Freidkin, I.; Teitz, S.; Albeck, M.; Weil, M.; Geffen-Aricha, R.; Yadid, G.; Sredni, B. Tellurium compound AS101 induces PC12 differentiation and rescues the neurons from apoptotic death. Ann. N. Y Acad. Sci. 2003, 1010, 659–666. [Google Scholar] [CrossRef] [PubMed]
- Carmely, A.; Meirow, D.; Peretz, A.; Albeck, M.; Bartoov, B.; Sredni, B. Protective effect of the immunomodulator AS101 against cyclophosphamide-induced testicular damage in mice. Hum. Reprod. 2009, 24, 1322–1329. [Google Scholar] [CrossRef] [PubMed]
- Maman, E.; Prokopis, K.; Levron, J.; Carmely, A.; Dor, J.; Meirow, D. Does controlled ovarian stimulation prior to chemotherapy increase primordial follicle loss and diminish ovarian reserve? An animal study. Hum. Reprod. 2009, 24, 206–210. [Google Scholar] [CrossRef] [PubMed]
- Kalich-Philosoph, L.; Roness, H.; Carmely, A.; Fishel-Bartal, M.; Ligumsky, H.; Paglin, S.; Wolf, I.; Kanety, H.; Sredni, B.; Meirow, D. Cyclophosphamide triggers follicle activation and “burnout”; AS101 prevents follicle loss and preserves fertility. Sci. Transl. Med. 2013, 5, 185. [Google Scholar] [CrossRef] [PubMed]
- Hayun, M.; Naor, Y.; Weil, M.; Albeck, M.; Peled, A.; Don, J.; Haran-Ghera, N.; Sredni, B. The immunomodulator AS101 induces growth arrest and apoptosis in multiple myeloma: Association with the Akt/survivin pathway. Biochem. Pharmacol. 2006, 72, 1423–1431. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jang, H.; Hong, K.; Choi, Y. Melatonin and Fertoprotective Adjuvants: Prevention against Premature Ovarian Failure during Chemotherapy. Int. J. Mol. Sci. 2017, 18, 1221. https://doi.org/10.3390/ijms18061221
Jang H, Hong K, Choi Y. Melatonin and Fertoprotective Adjuvants: Prevention against Premature Ovarian Failure during Chemotherapy. International Journal of Molecular Sciences. 2017; 18(6):1221. https://doi.org/10.3390/ijms18061221
Chicago/Turabian StyleJang, Hoon, Kwonho Hong, and Youngsok Choi. 2017. "Melatonin and Fertoprotective Adjuvants: Prevention against Premature Ovarian Failure during Chemotherapy" International Journal of Molecular Sciences 18, no. 6: 1221. https://doi.org/10.3390/ijms18061221
APA StyleJang, H., Hong, K., & Choi, Y. (2017). Melatonin and Fertoprotective Adjuvants: Prevention against Premature Ovarian Failure during Chemotherapy. International Journal of Molecular Sciences, 18(6), 1221. https://doi.org/10.3390/ijms18061221





