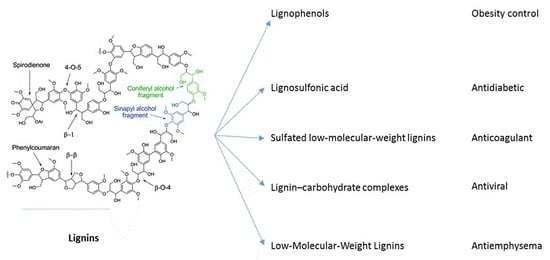
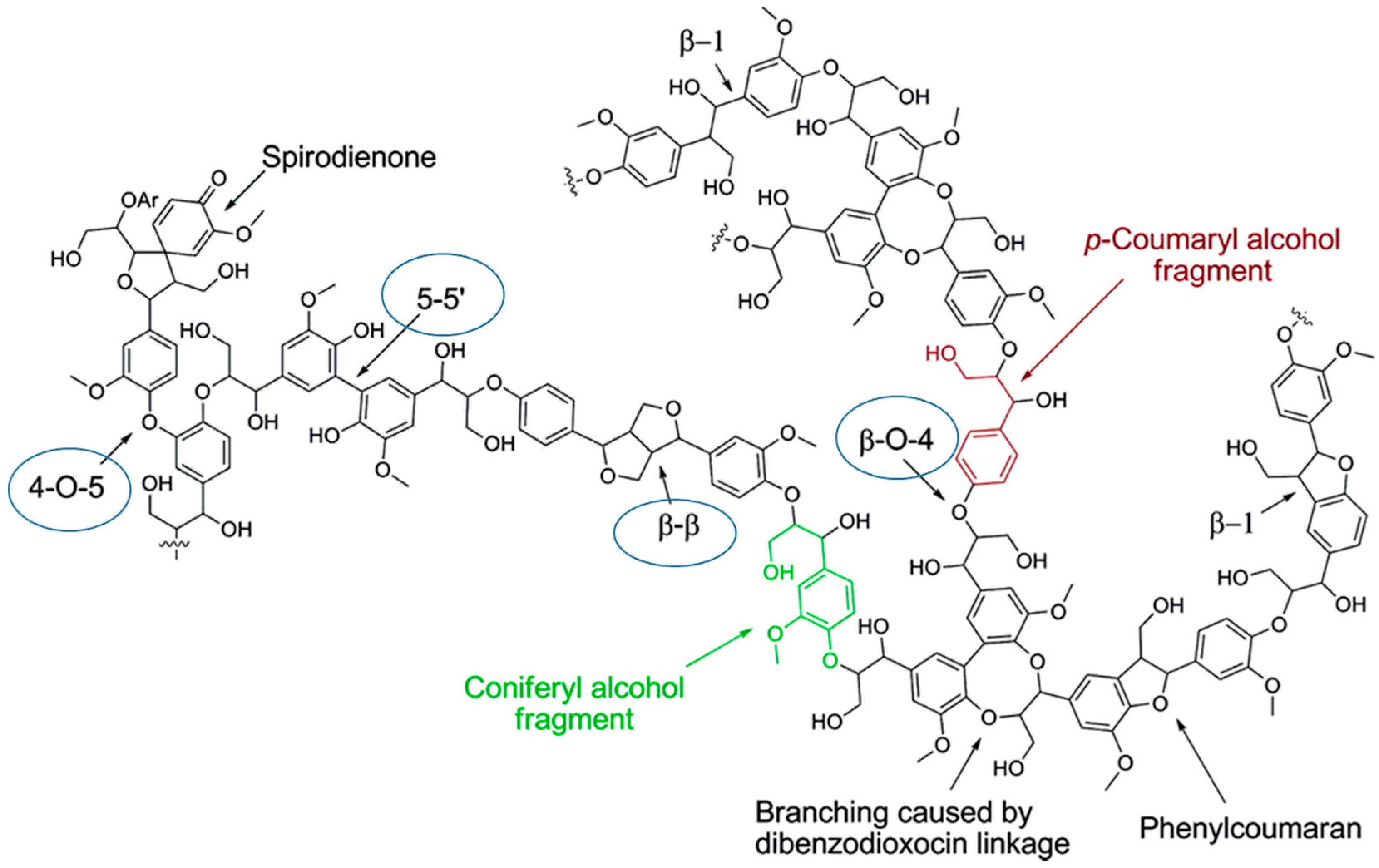
Lignins and Their Derivatives with Beneficial Effects on Human Health
Abstract
:1. Introduction
2. Pharmacological Activities of Lignins and Their Derivatives
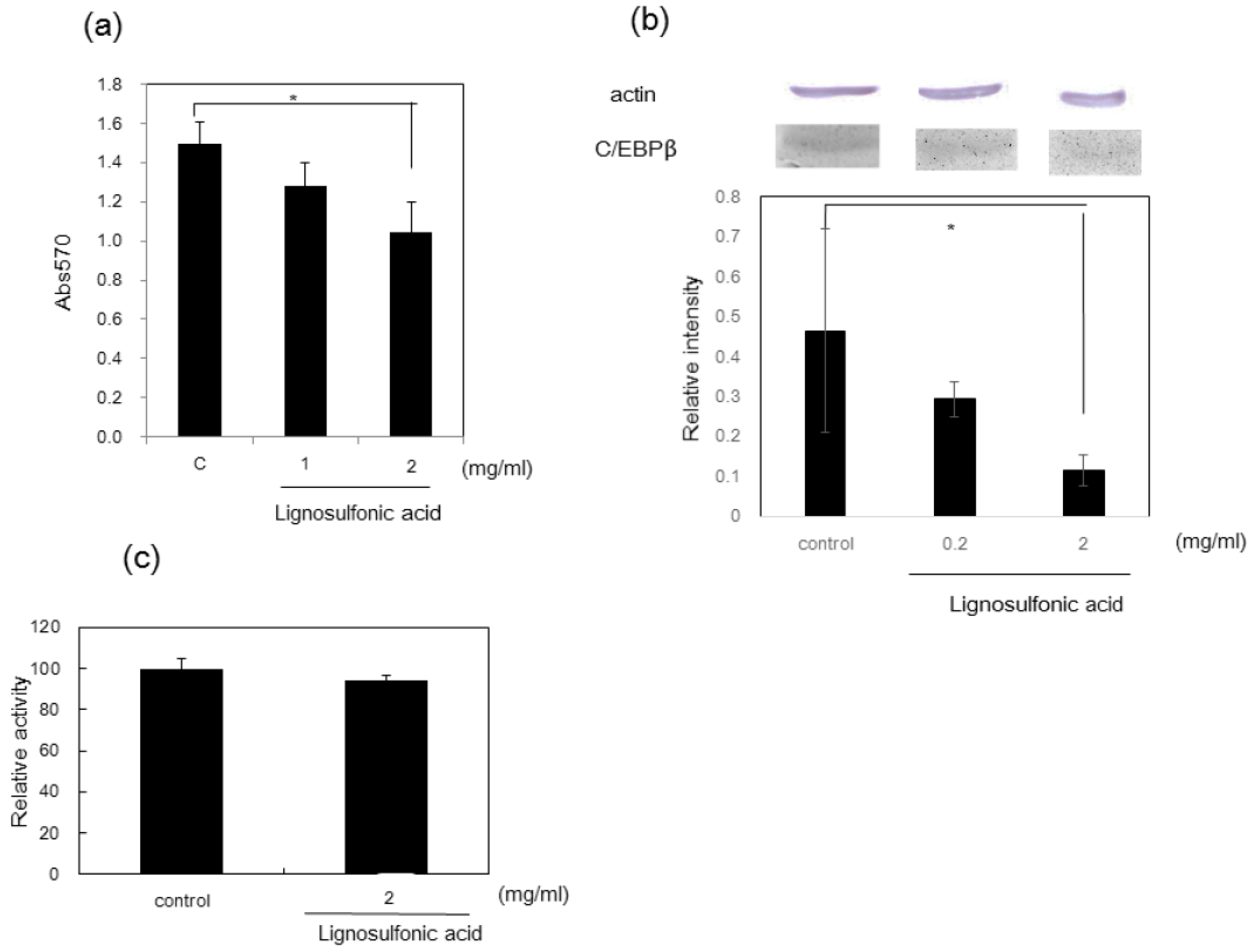
2.1. Lignins and Lignosulfonic Acid for the Treatment of Diabetes
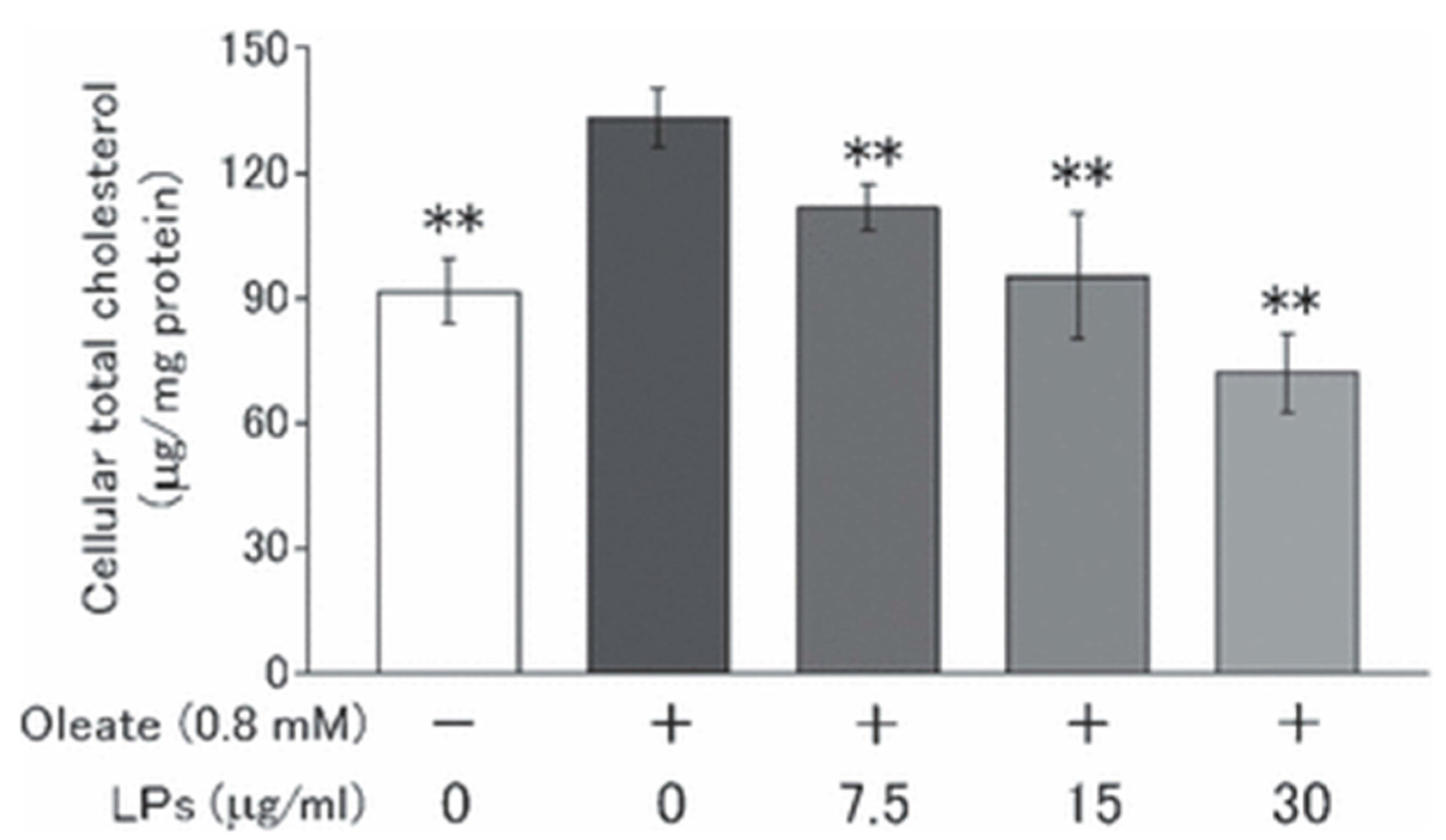
2.2. Lignophenols in Obesity Control
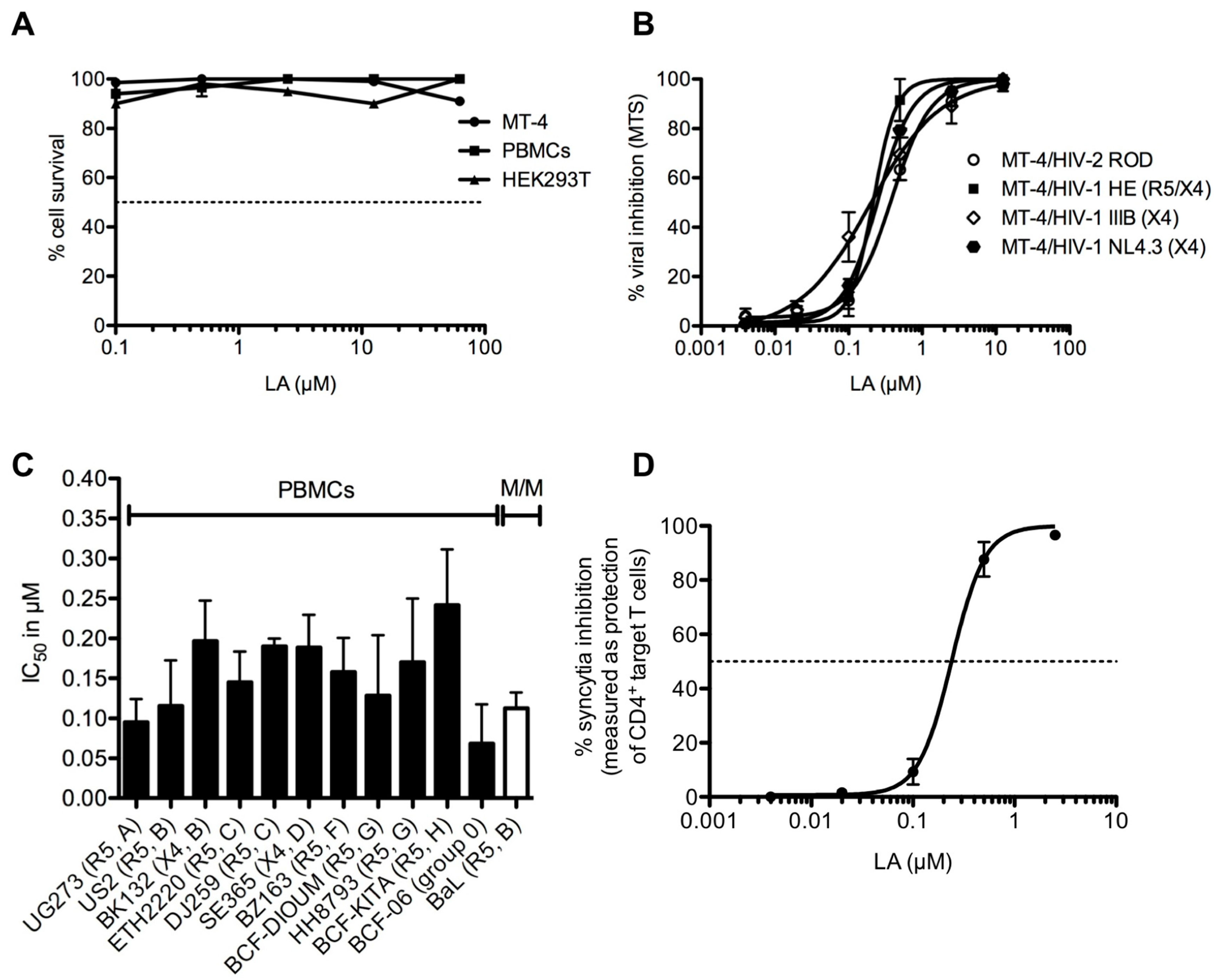
2.3. Lignosulfonic Acid with Antiviral Activity
2.4. Lignin–Carbohydrate Complexes as Antiviral Agents and Immunomodulators
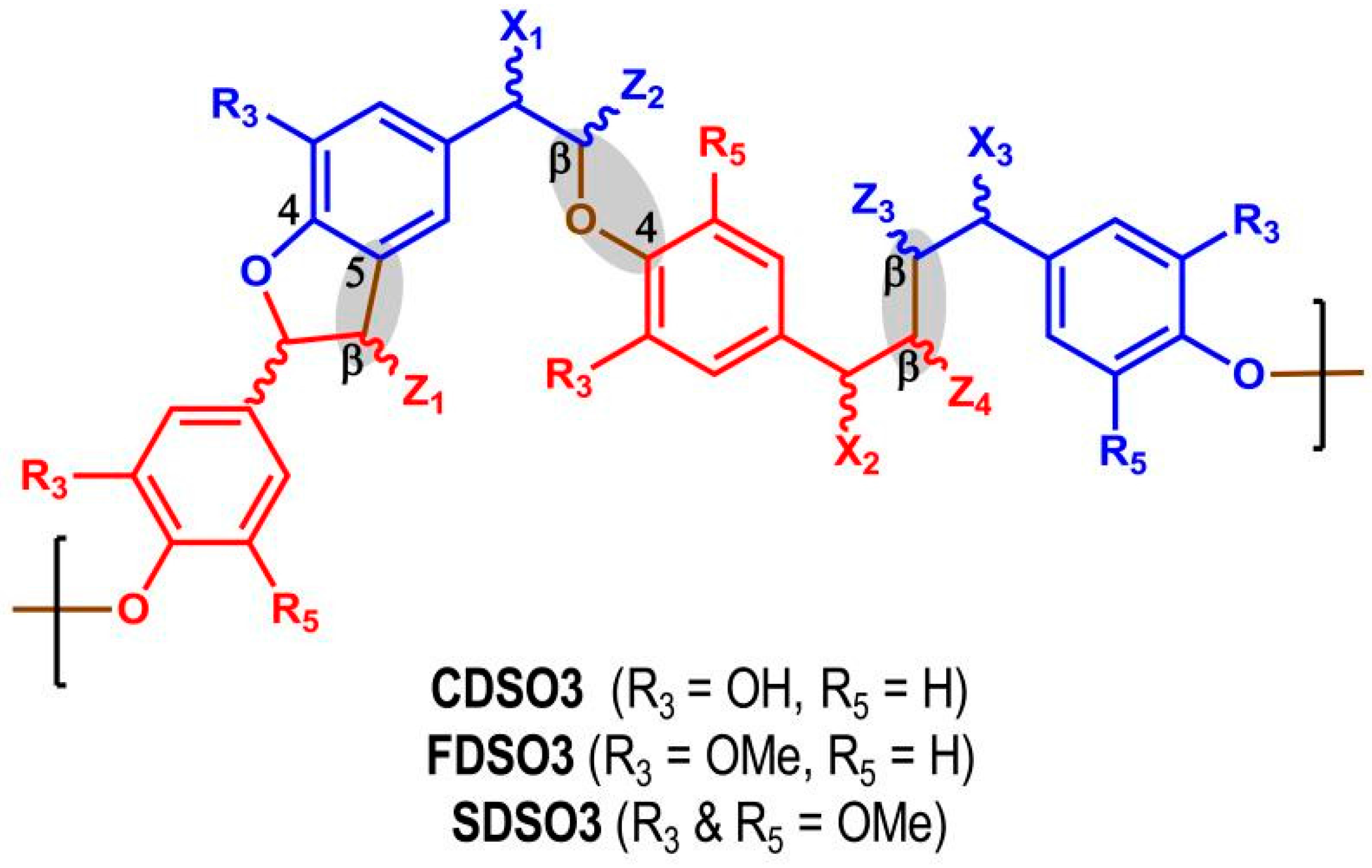
2.5. Low-Molecular-Weight Lignins as Anticoagulant and Anti-Emphysema Agents
2.6. Lignin-Based Nanoparticles for Drug Delivery
2.7. Other Lignins in Drug Delivery
2.8. Lignin Metabolites in Cancer
3. Conclusions
Supplementary Materials
Author Contributions
Conflicts of Interest
References
- Garcia, A.; Erdocia, X.; Gonzalez Alriols, M.; Labidi, J. Physicochemical Properties and Potential Applications of Lignins from Various Sources. In Lignin: Structural Analysis, Applications in Biomaterials and Ecological Significance, 6th ed.; Lu, F., Ed.; NOVA Publisher: New York, NY, USA, 2014; pp. 127–160. [Google Scholar]
- Toledano, A.; Serrano, L.; Balu, A.M.; Luque, R.; Pineda, A.; Labidi, J. Fractionation of organosolv lignin from olive tree clippings and its valorization to simple phenolic compounds. ChemSusChem 2013, 6, 529–536. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Kadla, J.F.; Ehara, K.; Gilkes, N.; Saddler, J.N. Organosolv ethanol lignin from hybrid poplar as a radical scavenger: Relationship between lignin structure, extraction conditions, and antioxidant activity. J. Agric. Food Chem. 2006. [Google Scholar] [CrossRef] [PubMed]
- Azadfar, M.; Gao, A.H.; Bule, M.V.; Chen, S. Structural characterization of lignin: A potential source of antioxidants guaiacol and 4-vinylguaiacol. Int. J. Biol. Macromol. 2015, 75, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Zakzeski, J.; Bruijnincx, P.C.A.; Jongerius, A.L.; Weckhuysen, B.M. The Catalytic Valorization of Lignin for the Production of Renewable Chemicals. Chem. Rev. 2010, 110, 3552–3599. [Google Scholar] [CrossRef] [PubMed]
- Kai, D.; Tan, M.J.; Chee, P.L.; Chua, Y.K.; Yap, Y.L.; Loh, X.J. Towards lignin-based functional materials in a sustainable world. Green Chem. 2016, 18, 1175–1200. [Google Scholar] [CrossRef]
- Ugartondo, V.; Mitjans, M.; Vinardell, M.P. Compartive antioxidant and cytotoxic effects of lignins from different sources. Bioresour. Technol. 2008, 99, 6683–6687. [Google Scholar] [CrossRef] [PubMed]
- Banard, D.L.; Heaton, K.W. Bile acids and vitamin A absorption in man: The effect of two bile acid-binding agents, chloestyramine and lignin. Gut 1973, 14, 316–318. [Google Scholar] [CrossRef]
- Reddy, B.S.; Maeura, Y.; Wayman, M. Effects of dietary corn bran and autohydrolyzed lignin on 3,2′-dimethyl-4-aminobiphenyl-induced intestinal carcinogénesis in male F344 rats. J. Natl. Cancer Inst. 1983, 71, 419–423. [Google Scholar] [PubMed]
- Martinez, V.; Mitjans, M.; Vinardell, M.P. Pharmacological applications of lignins and lignins related compounds: An overview. Curr. Organ. Chem. 2012, 16, 1863–1870. [Google Scholar] [CrossRef]
- Barapatre, A.; Meena, A.S.; Mekala, S.; Das, A.; Jha, H. In vitro evaluation of antioxidant and cytotoxic activities of lignin fractions extracted from Acacia nilotica. Int. J. Biol. Macromol. 2016, 86, 443–453. [Google Scholar] [CrossRef] [PubMed]
- Quesille-Villalobos, A.M.; Torrico, J.S.; Ranilla, L.G. Phenolic compounds, antioxidant capacity, and in vitro α-amylase inhibitory potential of tea infusions (Camellia sinensis) commercialized in Chile. CyTA J. Food 2013, 11, 60–67. [Google Scholar] [CrossRef]
- Kalaivani, T.; Mathew, L. Free radical scavenging activity from leaves of Acacia nilotica (L.) Wild. ex Delile, an Indian medicinal tree. Food Chem. Toxicol. 2010, 48, 298–305. [Google Scholar] [CrossRef] [PubMed]
- Barapatre, A.; Aadil, K.R.; Tiwary, B.N.; Jha, H. In vitro antioxidant and antidiabetic activities of biomodified ligninfrom Acacia nilotica wood. Int. J. Biol. Macromol. 2015, 75, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, Y.; Kadota, Y.; Hasegawa, C.; Kawiminami, S. Lignosulfonic acid-induced inhibition of intestinal glucose absorption. J. Nutr. Sci. Vitaminol. 2015, 61, 449–454. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, Y.; Nakagawa, E.; Kadota, Y.; Kawiminami, S. Lignosulfonic acid promotes hypertrophy in 3T3-L1 cells without increasing lipid content and increases their 2-deoxyglucose uptake. Asian-Austalas. J. Anim. Sci. 2017, 30, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Funaoka, M.; Fukatsu, S. Characteristics of lignin structural conversion in a phase-separative reaction system composed of cresol and sulfuric acid. Holzforschung 1996, 50, 245–252. [Google Scholar] [CrossRef]
- Fujita, S.; Ohmae, E.; Funaoka, M. In Vitro the antioxidant activity of lignophenol from Beech (Fagus crenata Blume) and Hinoki (Cryptomeria japonica D Don). J. Jpn. Assoc. Diet. Fiber Res. 2003, 7, 245–252. [Google Scholar]
- Akao, Y.; Seki, N.; Nakagawa, Y.; Yi, H.; Matsumoto, K.; Ito, Y.; Ito, K.; Funaoka, M.; Maruyama, W.; Naoi, M.; et al. A highly bioactive lignophenol derivative from bamboo lignin exhibits a potent activity to suppress apoptosis induced by oxidative stress in human neuroblastoma SH-SY5Y cells. Bioorgan. Med. Chem. 2004, 12, 4791–4801. [Google Scholar] [CrossRef] [PubMed]
- Norikura, T.; Mukai, Y.; Fujita, S.; Mikame, K.; Funaoka, M.; Sato, S. Lignophenols decrease oleate-induced apolipoprotein-B secretion in HepG2 cells. Basic Clin. Pharmacol. Toxicol. 2010, 107, 813–817. [Google Scholar] [CrossRef] [PubMed]
- Sato, S.; Mukai, Y.; Yamate, J.; Norikura, T.; Morinaga, Y.; Mikame, K.; Funaoka, M.; Fujita, S. Lignin-derived lignophenols attenuate oxidative and inflammatory damage to the kidney in streptozotocin-induced diabetic rats. Free Radic. Res. 2009, 43, 1205–1213. [Google Scholar] [CrossRef] [PubMed]
- Sato, S.; Mukai, Y.; Tokuoka, Y.; Mikame, K.; Funaoka, M.; Fujita, S. Effect of lignin-derived lignophenols on hepatic lipid metabolism in rats fed a high-fat diet. Environ. Toxicol. Pharmacol. 2012, 34, 228–234. [Google Scholar] [CrossRef] [PubMed]
- Fargues, C.; Mathias, A.; Rodrigues, A. Kinetics of Vanillin Production from Kraft Lignin Oxidation. Ind. Eng. Chem. Res. 1996, 35, 28–36. [Google Scholar] [CrossRef]
- Andrei, G.; Lisco, A.; Vanpouille, C.; Introini, A.; Balestra, E.; van den Oord, J.; Cihlar, T.; Perno, C.F.; Snoeck, R.; Margolis, L.; et al. Topical tenofovir, a microbicide effective against HIV, inhibits herpes simplex virus-2 replication. Cell Host Microbe 2011, 10, 379–389. [Google Scholar] [CrossRef] [PubMed]
- Gordts, S.C.; Férir, G.; D’huys, T.; Petrova, M.I.; Lebeer, S.; Snoeck, R.; Andrei, G.; Schols, D. The Low-Cost Compound Lignosulfonic Acid (LA) Exhibits Broad-Spectrum Anti-HIV and Anti-HSV Activity and Has Potential for Microbicidal Applications. PLoS ONE 2015, 10, e0131219. [Google Scholar] [CrossRef] [PubMed]
- Karim, Q.A.; Karim, S.S.A.; Frohlich, J.A.; Grobler, A.C.; Baxter, C.; Mansoor, L.E.; Kharsany, A.B.M.; Sibeko, S.; Mlisana, K.P.; Omar, Z.; et al. Effectiveness and safety of tenofovir gel, an antiretroviral microbicide for the prevention of HIV infection in women. Science 2010, 329, 1168–1174. [Google Scholar] [CrossRef] [PubMed]
- Tao, W.; Richards, C.; Hamer, D. Enhancement of HIV Infection by Cellulose Sulfate. AIDS Res. Hum. Retrovir. 2008, 24, 925–929. [Google Scholar] [CrossRef] [PubMed]
- Qiu, M.; Wang, Q.; Chu, Y.; Yuan, Z.; Song, H.; Chen, Z.; Wu, Z. Lignosulfonic acid exhibits broadly anti-HIV-1 activity-potential as a microbicide candidate for the prevention of HIV-1 sexual transmission. PLoS ONE 2012, 7, e35906. [Google Scholar] [CrossRef] [PubMed]
- Jeffries, T.W. Biodegradation of lignin-carbohydrate complexes. Biodegradation 1990, 1, 163–176. [Google Scholar] [CrossRef]
- Singh, R.; Singh, S.; Trimukhe, K.D.; Pandare, K.V.; M Bastawade, K.B.; Gokhale, D.V.; Varma, A.J. Lignin–carbohydrate complexes from sugarcane bagasse: Preparation, purification, and characterization. Carbohydr. Polym. 2005, 62, 57–66. [Google Scholar] [CrossRef]
- Nanbu, T.; Shimada, J.; Kobayashi, M.; Hirano, K.; Koh, T.; Machino, M.; Ohno, H.; Yamamoto, M.; Sakagami, H. Anti-UV activity of lignin-carbohydrate complex and related compounds. In Vivo 2013, 27, 133–139. [Google Scholar] [PubMed]
- Nanbu, T.; Matsuta, T.; Sakagami, H.; Shimada, J.; Maki, J.; Makino, T. Anti-UV activity of Lentinus edodes mycelia extract (LEM). In Vivo 2011, 25, 733–740. [Google Scholar] [PubMed]
- Sakagami, H.; Sheng, H.; Okudaira, N.; Yasui, T.; Wakabayashi, H.; Jia, J.; Natori, T.; Suguro-Kitajima, M.; Oizumi, H.; Oizumi, T. Prominent Anti-UV Activity and Possible Cosmetic Potential of Lignin-carbohydrate Complex. In Vivo 2016, 30, 331–339. [Google Scholar] [PubMed]
- Zhang, Y.; But, P.P.; Ooi, V.E.; Xu, H.X.; Delaney, G.D.; Lee, S.H.; Lee, S.F. Chemical properties, mode of action, and in vivo anti-herpes activities of a lignin-carbohydrate complex from Prunella vulgaris. Antivir. Res. 2007, 75, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.B.; Yamagishi, C.; Hayashi, K.; Hayashi, T. Antiviral and immunostimulating effects of lignin-carbohydrate-protein complexes from Pimpinella anisum. Biosci. Biotechnol. Biochem. 2011, 75, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Sakagami, H. Biological activities and possible dental application of three major groups of polyphenols. J. Pharmacol. Sci. 2014, 126, 92–106. [Google Scholar] [CrossRef] [PubMed]
- Sakagami, H.; Kawano, M.; Thet, M.M.; Hashimoto, K.; Satoh, K.; Kanamoto, T.; Terakubo, S.; Nakashima, H.; Haishima, Y.; Maeda, Y.; et al. Anti-HIV and immunomodulation activities of cacao lass lignin-carbohydrate complex. In Vivo 2011, 25, 229–236. [Google Scholar] [PubMed]
- Thakkar, J.N.; Tiwari, V.; Desai, U.R. Nonsulfated, Cinnamic Acid-Based Lignins are Potent Antagonists of HSV-1 Entry into Cells. Biomacromolecules 2010, 11, 1412–1416. [Google Scholar] [CrossRef] [PubMed]
- Loomis, T.A.; Beyer, R.E. Heparin-like anticoagulant action of sulfonated lignins from commercial waste sulfite liquour. J. Pharmacol. Exp. Ther. 1953, 109, 21–25. [Google Scholar] [PubMed]
- Monien, B.H.; Henry, B.L.; Raghuraman, A.; Hindle, M.; Desai, U.R. Novel chemo-enzymatic oligomers of cinnamic acids as direct and indirect inhibitors of coagulation proteinases. Bioorgan. Med. Chem. 2006, 14, 7988–7998. [Google Scholar] [CrossRef] [PubMed]
- Henry, B.L.; Monien, B.H.; Bock, P.E.; Desai, U.R. A novel allosteric pathway of thrombin inhibition: Exosite II mediated potent inhibition of thrombin by chemo-enzymatic, sulfated dehydropolymers of 4-hydroxycinnamic acids. J. Biolog. Chem. 2007, 282, 31891–31899. [Google Scholar] [CrossRef] [PubMed]
- Henry, B.L.; Abdel Aziz, M.; Zhou, Q.; Desai, U.R. Sulfated, low-molecular-weight lignins are potent inhibitorsof plasmin, in addition to thrombin and factor Xa: Novel opportunity for controlling complex pathologies. Thromb. Haemost. 2010, 103, 507–515. [Google Scholar] [CrossRef] [PubMed]
- Henry, B.L.; Desai, U.R. Sulfated Low Molecular Weight Lignins, Allosteric Inhibitors of Coagulation Proteinases via the Heparin Binding Site, Significantly Alter the Active Site of Thrombin and Factor Xa Compared to Heparin. Thromb. Res. 2014, 134, 1123–1129. [Google Scholar] [CrossRef] [PubMed]
- Mehta, A.Y.; Mohammed, B.M.; Martin, E.J.; Brophy, D.F.; Gailani, D.; Desai, U.R. Allosterism-based Simultaneous, Dual Anticoagulant and Antiplatelet Action. Allosteric Inhibitor Targeting the Glycoprotein Ibα and Heparin-Binding Site of Thrombin. J. Thromb. Haemost. 2016, 14, 828–838. [Google Scholar] [CrossRef] [PubMed]
- Fischer, B.M.; Pavlisko, E.; Voynow, J.A. Pathogenic triad in COPD: Oxidative stress, proteaseantiprotease imbalance, and inflammation. Int. J. Chronic Obs. Pulm. Dis. 2011, 6, 413–421. [Google Scholar] [CrossRef] [PubMed]
- Saluja, B.; Thakkar, J.N.; Li, H.; Desai, U.R.; Sakagami, M. Novel low molecular weight lignins as potential anti-emphysema agents: In vitro triple inhibitory activity against elastase, oxidation and inflammation. Pulm. Pharmacol. Ther. 2013, 26, 296–304. [Google Scholar] [CrossRef] [PubMed]
- Pereira, F.M.; Gonçalves, A.R.; Ferraz, A.; Silva, F.T.; Oliveira, S.C. Estimation of solubility effect on the herbicide controlled-release kinetics from lignin-based formulations. Appl. Biochem. Biotechnol. 2003, 108, 913–919. [Google Scholar] [CrossRef]
- Fernández-Pérez, M.; Villafranca-Sánchez, M.; Flores-Céspedes, F. Controlled-release formulations of cyromazine-lignin matrix coated with ethylcellulose. J. Environ. Sci. Health B 2007, 42, 863–868. [Google Scholar] [CrossRef] [PubMed]
- Frangville, C.; Rutkevičius, M.; Richter, A.P.; Velev, O.D.; Stoyanov, S.D.; Paunov, V.N. Fabrication of environmentally biodegradable lignin nanoparticles. ChemPhysChem 2012, 13, 4235–4243. [Google Scholar] [CrossRef] [PubMed]
- Lievonen, M.; Valle-Delgado, J.J.; Mattinen, M.L.; Hult, E.L.; Lintinen, K.; Kostiainen, M.A.; Paananen, A.; Szilvay, G.R.; Setala, H.; Osterberg, M. A simple process for lignin nanoparticle preparation. Green Chem. 2016, 18, 1416–1422. [Google Scholar] [CrossRef]
- Richter, A.P.; Bharti, B.; Armstrong, H.B.; Brown, J.S.; Plemmons, D.; Paunov, V.N.; Stoyanov, S.D.; Velev, O.D. Synthesis and characterization of biodegradable lignin nanoparticles with tunable surface properties. Langmuir 2016, 32, 6468–6477. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo, P.; Lintinen, K.; Kiriazis, A.; Hynninen, V.; Liu, Z.; Ramos, T.; Rahikkala, A.; Correia, A.; Kohout, T.; Sarmento, B.; et al. In Vitro Evaluation of biodegradable lignin-based nanoparticles for drug delivery and enhanced antiproliferation effect in cancer cells. Biomaterials 2017, 121, 97–108. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Hernández, J.M.; Escalante, A.; Murillo-Vázquez, R.N.; Delgado, E.; González, F.J.; Toríz, G. Use of Agave tequilana-lignin and zinc oxide nanoparticles for skin photoprotection. J. Photochem. Photobiol. B Biol. 2016, 163, 156–161. [Google Scholar] [CrossRef] [PubMed]
- Klapiszewski, L.; Rzemieniecki, T.; Krawczyk, M.; Malina, D.; Norman, M.; Zdarta, J.; Majchrzak, I.; Dobrowolska, A.; Czaczyk, K.; Jesionowski, T. Kraft lignin/silica-AgNPs as a functional material with antibacterial activity. Colloids Surf. B Biointerfaces 2015, 134, 220–228. [Google Scholar] [CrossRef] [PubMed]
- Nasti, A.; Zaki, N.M.; de Leonardis, P.; Ungphaiboon, S.; Sansongsak, P.; Rimoli, M.G.; Tirelli, N. Chitosan/TPP and chitosan/TPP-hyaluronic acid nanoparticles: Systematic optimisation of the preparative process and preliminary biological evaluation. Pharm. Res. 2009, 26, 1918–1930. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Fernandes, M.M.; Matamá, T.; Loureiro, A.; Gomes, A.C.; Cavaco-Paulo, A. Chitosan-lignosulfonates sono-chemically prepared nanoparticles: Characterisation and potential applications. Colloids Surf. B Biointerfaces 2013, 103, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Faustino, H.; Gil, N.; Baptista, C.; Duarte, A.P. Antioxidant Activity of Lignin Phenolic Compounds Extracted from Kraft and Sulphite Black Liquors. Molecules 2010, 15, 9308–9322. [Google Scholar] [CrossRef] [PubMed]
- Wahba, S.M.; Darwish, A.S.; Shehata, I.H.; Abd Elhalem, S.S. Sugarcane bagasse lignin, and silica gel and magneto-silica as drug vehicles for development of innocuous methotrexate drug against rheumatoid arthritis disease in albino rats. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 48, 599–610. [Google Scholar] [CrossRef] [PubMed]
- Răschip, I.E.; Panainte, A.D.; Pamfil, D.; Profire, L.; Vasile, C. In vitro testing of xanthan/lignin hydrogels as carriers for controlled delivery of bisoprolol fumarare. Rev. Med. Chir. Soc. Med. Nat. Iasi 2015, 119, 1189–1194. [Google Scholar] [PubMed]
- Chowdhury, M.A. The controlled release of bioactive compounds from lignin and lignin-based biopolymer matrices. Int. J. Biol. Macromol. 2014, 65, 136–147. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Hui, J.; Kou, W.; Xin, R.; Jia, F.; Wang, N.; Hu, F.; Zhang, H.; Liu, H. Identification of Inonotus obliquus and Analysis of Antioxidation and Antitumor Activities of Polysaccharides. Curr. Microbiol. 2008, 57, 454–462. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Mu, H.; Zhang, L.; Dong, D.; Zhang, W.; Duan, J. Characterization of two water-soluble lignin metabolites with antiproliferative activities from Inonotus obliquus. Int. J. Biol. Macromol. 2015, 74, 507–514. [Google Scholar] [CrossRef] [PubMed]

| Compound | Effect | Mechanism | Experimental Model | Reference |
|---|---|---|---|---|
| Alkali lignin | Antidiabetic | α-amylase inhibition In vitro decreased glucose diffusion | In vitro glucose movement | [14] |
| Lignosulfonic acid | Antidiabetic | Inhibitor of α-glucosidase Decrease blood glycemia | In vitro inhibitor of α-glucosidase Rat in vivo | [15] |
| Lignophenols | Obesity control | Decrease oleate-induced apo-B secretion | HepG2 in vitro | [20] |
| Lignophenols | Obesity control | Decrease plasma triglyceride levels | Rats fed a high-fat diet | [21] |
| Lignosulfonic acid | Antiviral activity | Inhibition of the replication of herpes simplex virus (HSV) | Infected mice by exposing scarified skin to an HSV-2 G | [24] |
| Lignosulfonic acid (LA) | Antiviral activity | LA mainly binds to the HIV-1 envelope glycoproteins | In vitro cells: T-lymphoma cell lines, HEK293T, HUT-78, Monocyte-derived dendritic cells | [25] |
| Lignin–carbohydrate complexes | Antiviral activity | Inhibiting viral binding and penetration | Vero cells infected with herpes simplex virus | [35] |
| Lignin–carbohydrate complexes | Antiviral activity | Inhibiting viral binding and penetration | HeLa cells infected with herpes simplex virus | [38] |
| Sulfated low-molecular-weight lignins | Anticoagulant | Inhibition of thrombin | Binding to thrombin | [43] |
| Sulfated low-molecular-weight lignins | Anticoagulant | Allosteric inhibition of thrombin | Whole blood thromboelastography, hemostasis analysis and mouse arterial thrombosis models | [44] |
| Sulfated low-molecular-weight lignins | Antiemphysema | Elastase, oxidation and inflammation inhibition | In vitro human alveolar A549 and bronchial Calu-3 epithelial cells | [46] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vinardell, M.P.; Mitjans, M. Lignins and Their Derivatives with Beneficial Effects on Human Health. Int. J. Mol. Sci. 2017, 18, 1219. https://doi.org/10.3390/ijms18061219
Vinardell MP, Mitjans M. Lignins and Their Derivatives with Beneficial Effects on Human Health. International Journal of Molecular Sciences. 2017; 18(6):1219. https://doi.org/10.3390/ijms18061219
Chicago/Turabian StyleVinardell, Maria Pilar, and Montserrat Mitjans. 2017. "Lignins and Their Derivatives with Beneficial Effects on Human Health" International Journal of Molecular Sciences 18, no. 6: 1219. https://doi.org/10.3390/ijms18061219
APA StyleVinardell, M. P., & Mitjans, M. (2017). Lignins and Their Derivatives with Beneficial Effects on Human Health. International Journal of Molecular Sciences, 18(6), 1219. https://doi.org/10.3390/ijms18061219