Rutin-Enriched Extract from Coriandrum sativum L. Ameliorates Ionizing Radiation-Induced Hematopoietic Injury
Abstract
:1. Introduction
2. Results
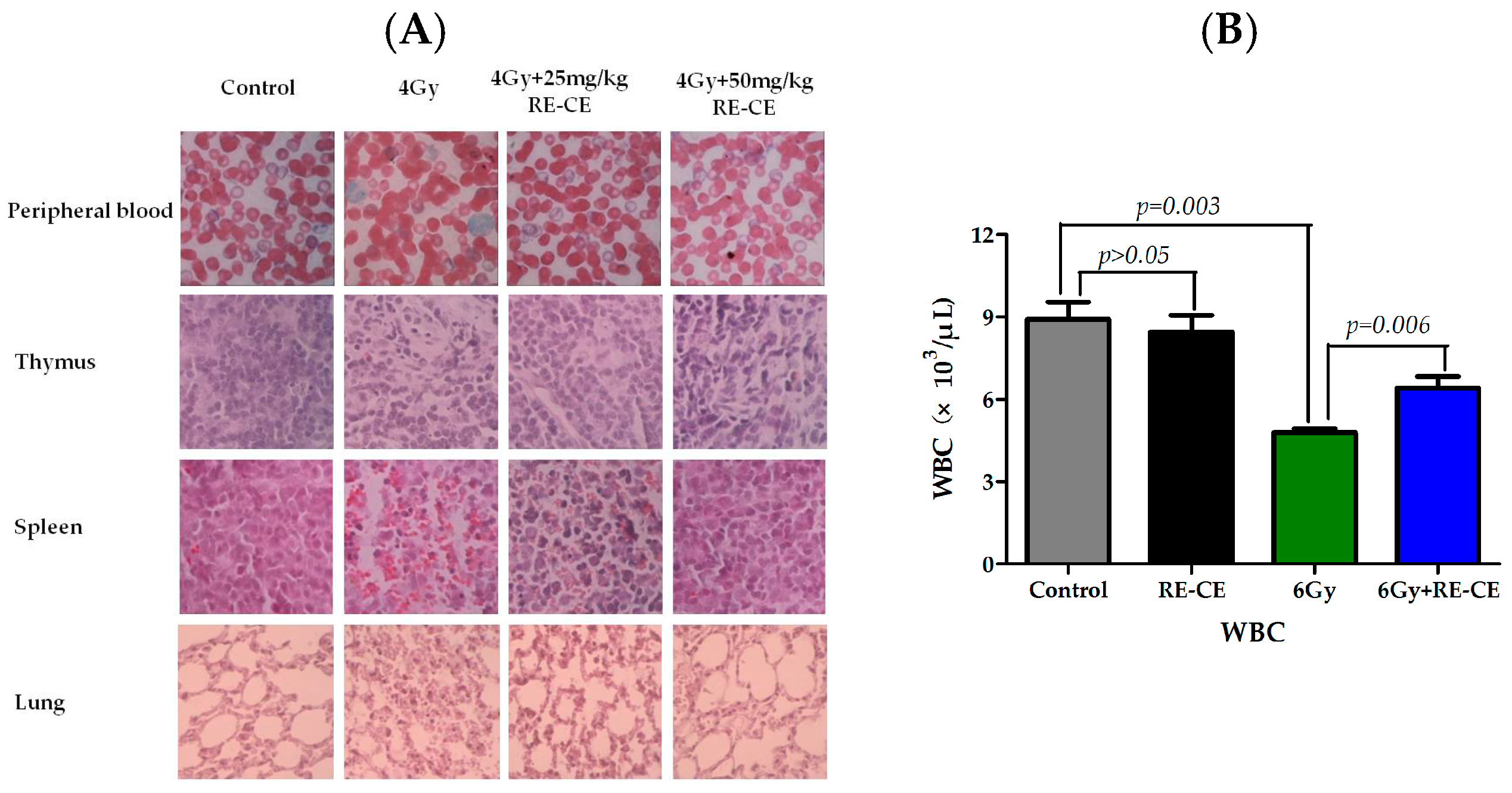
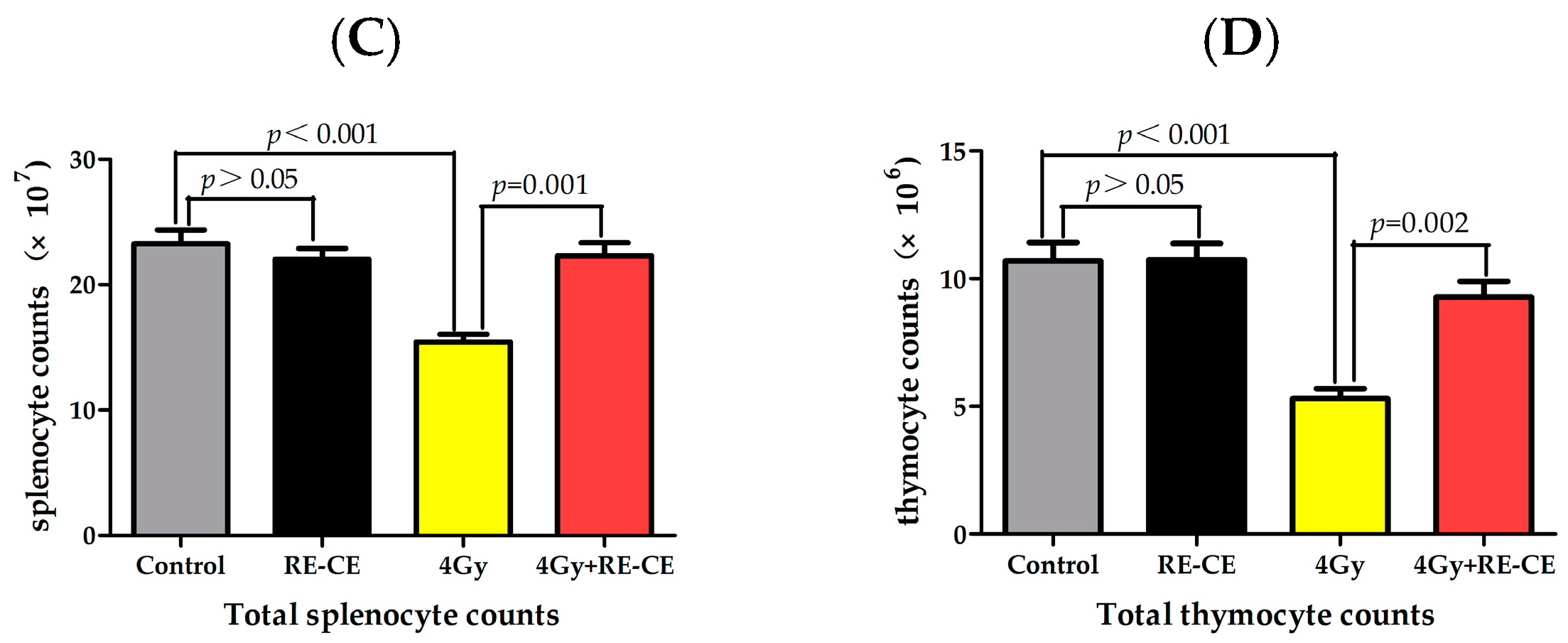
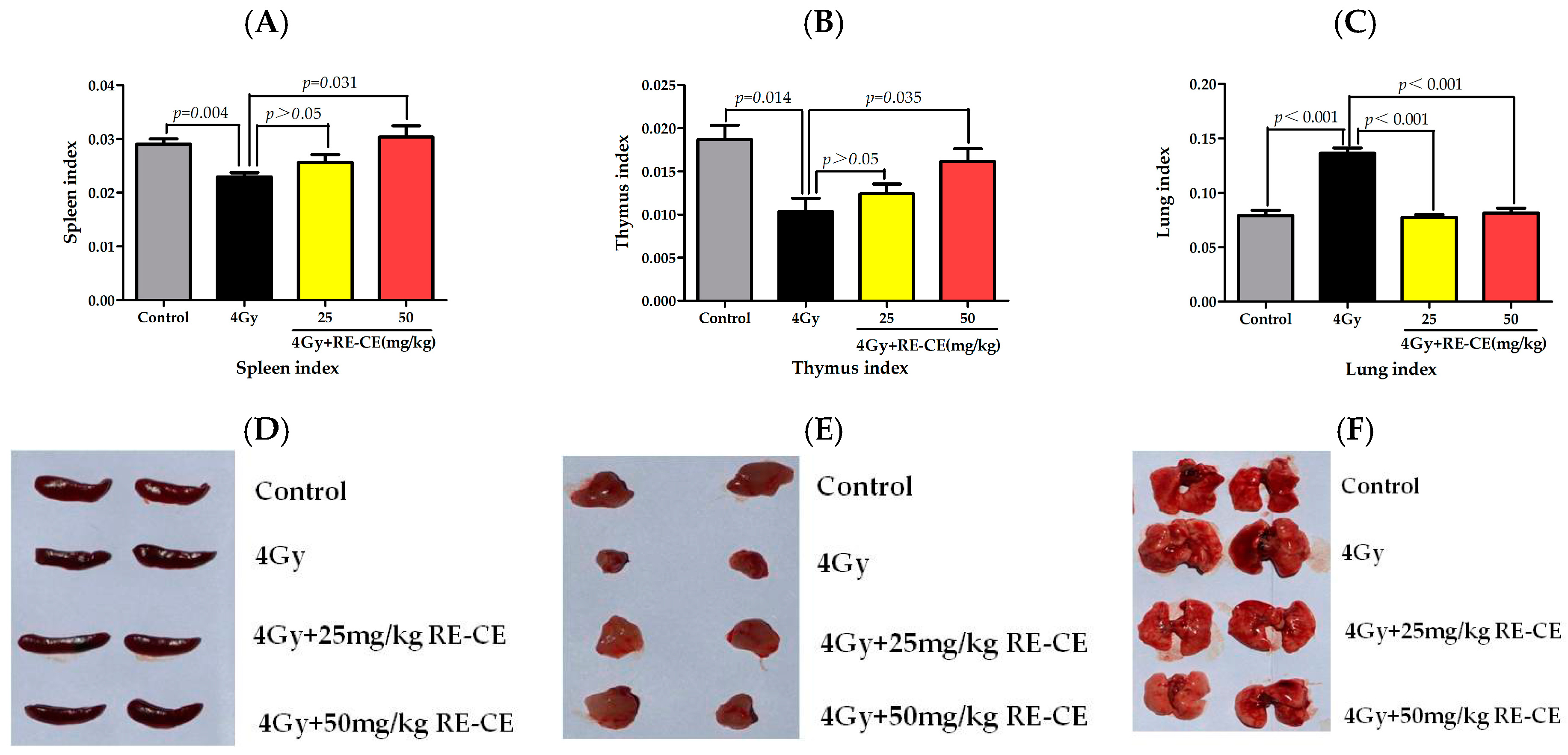
2.1. RE-CE Ameliorates IR-Induced Organ Injury
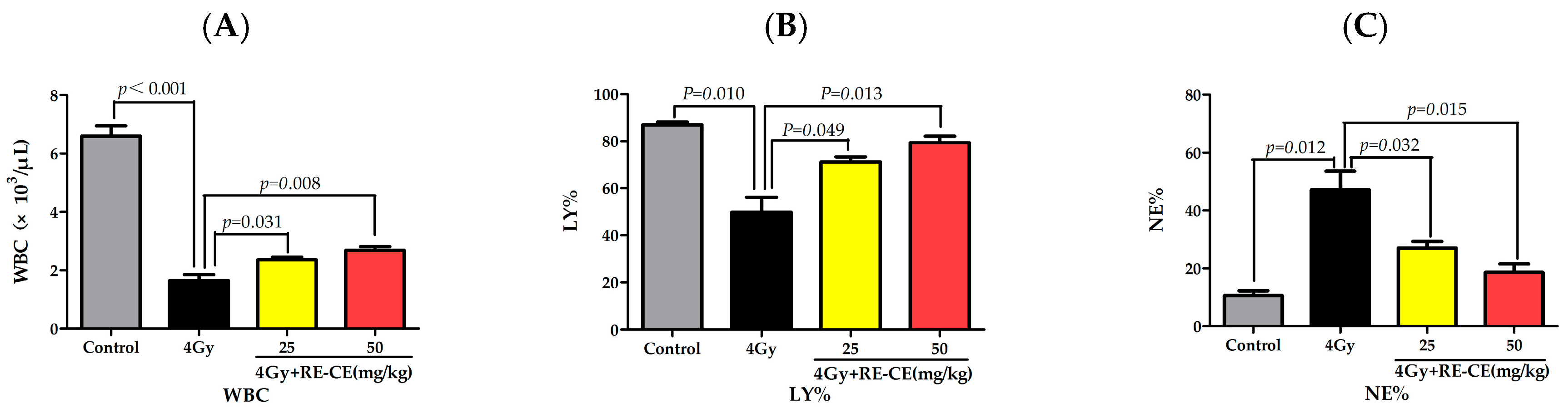
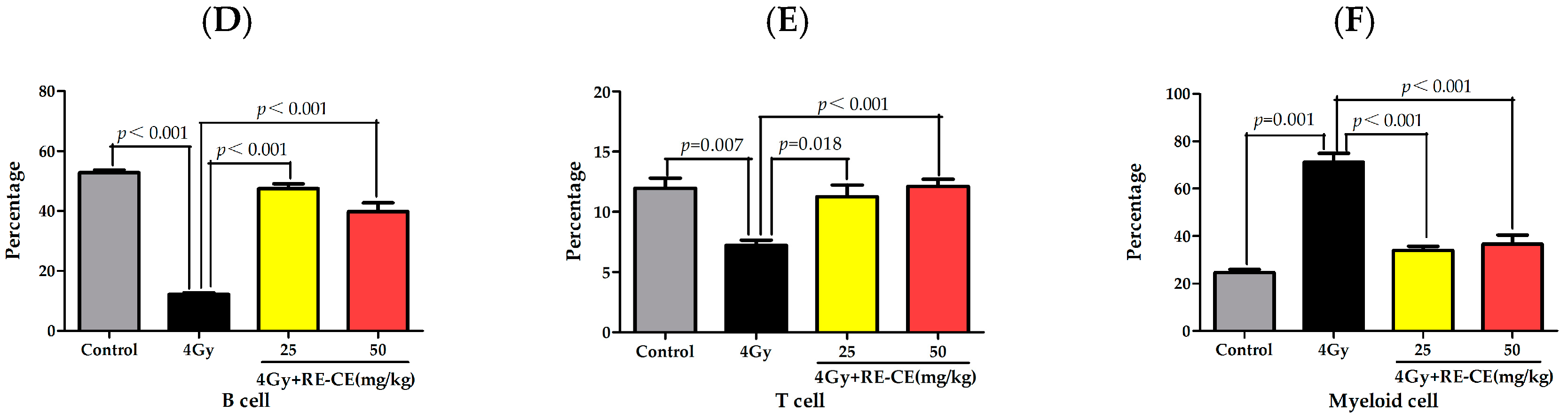
2.2. RE-CE Alleviates IR-Induced Myelosuppression and Promotes Myeloid Skewing Recovery
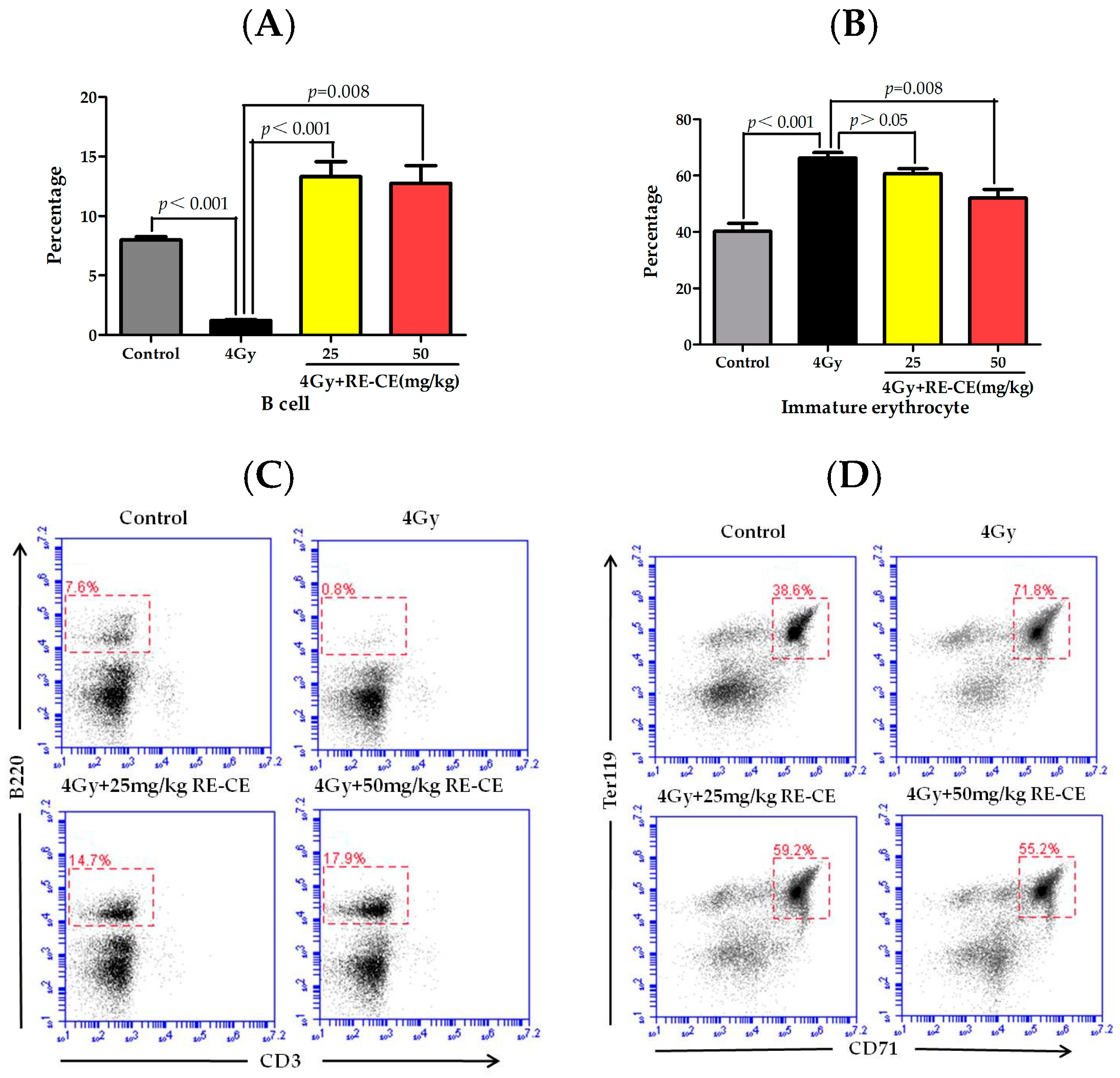
2.3. RE-CE Mitigates IR-Induced Differentiation-Related Dysfunction of B Cells and Erythrocytes in BM
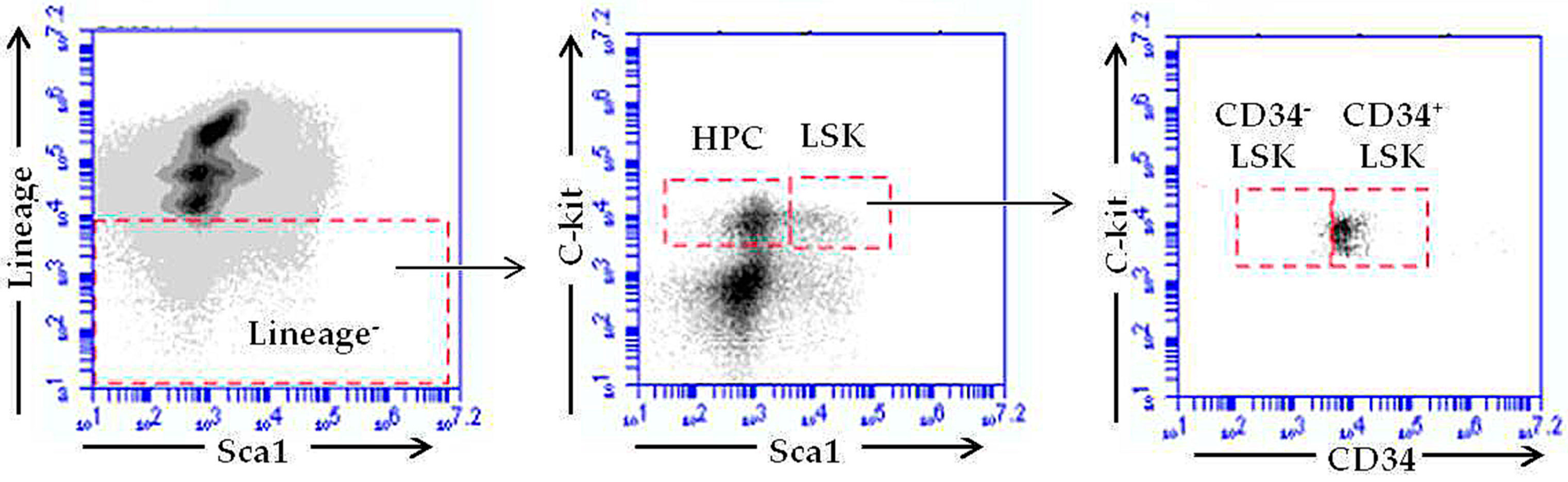
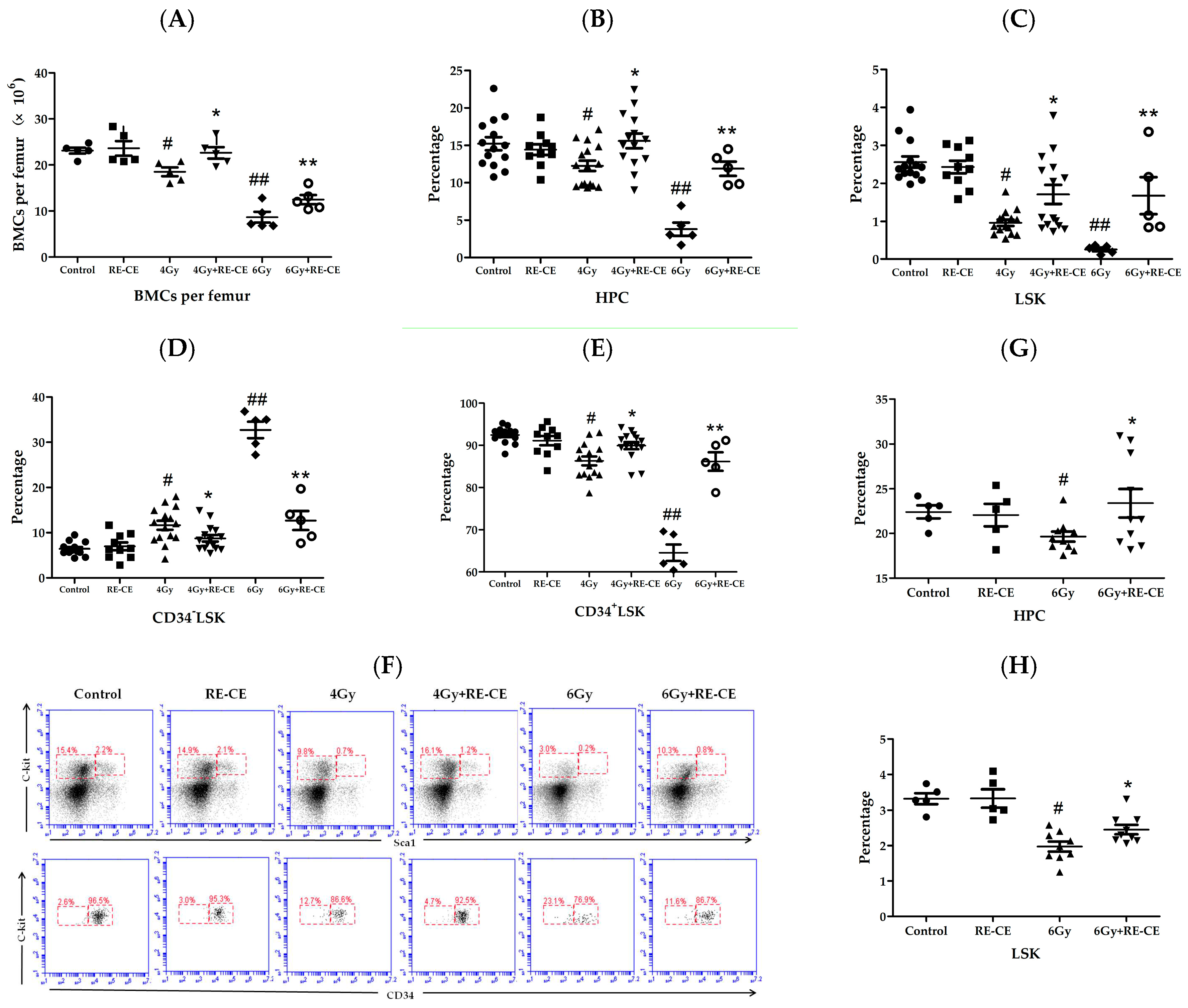
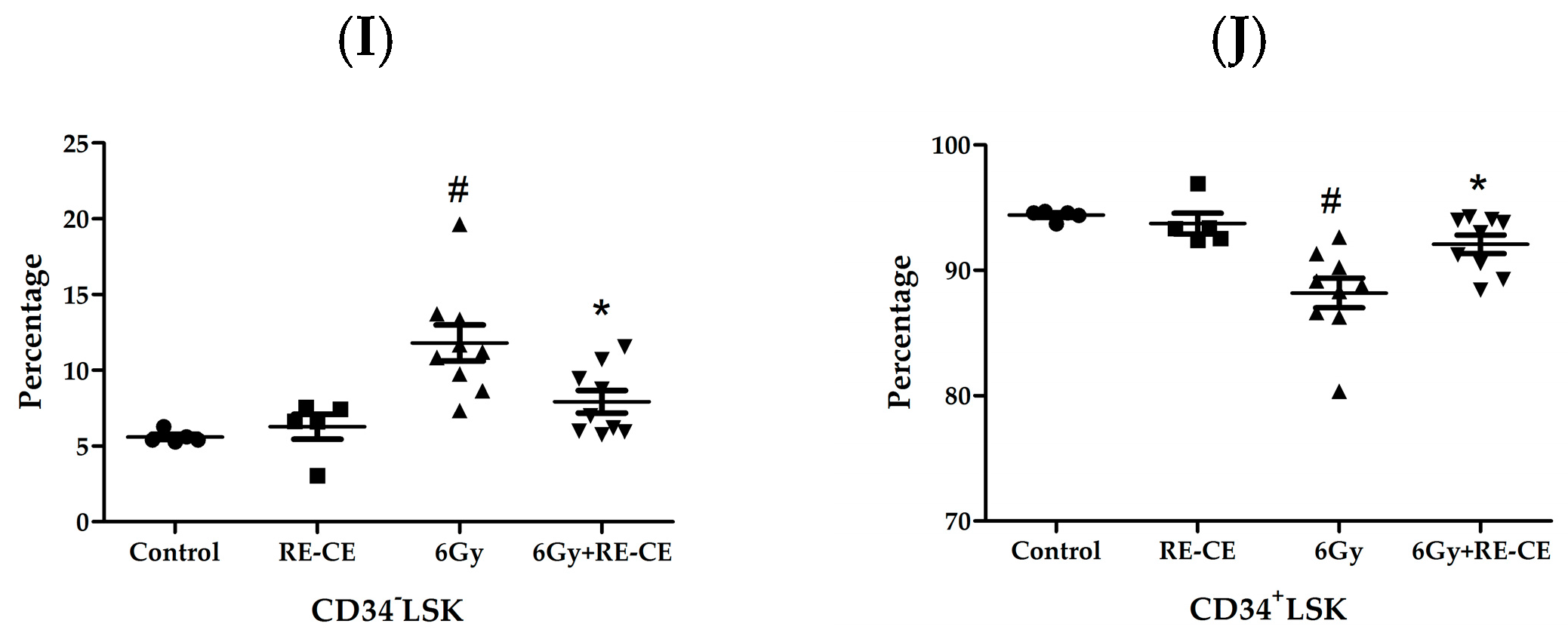
2.4. RE-CE Attenuates IR-Induced Alterations in HSPCs Frequency
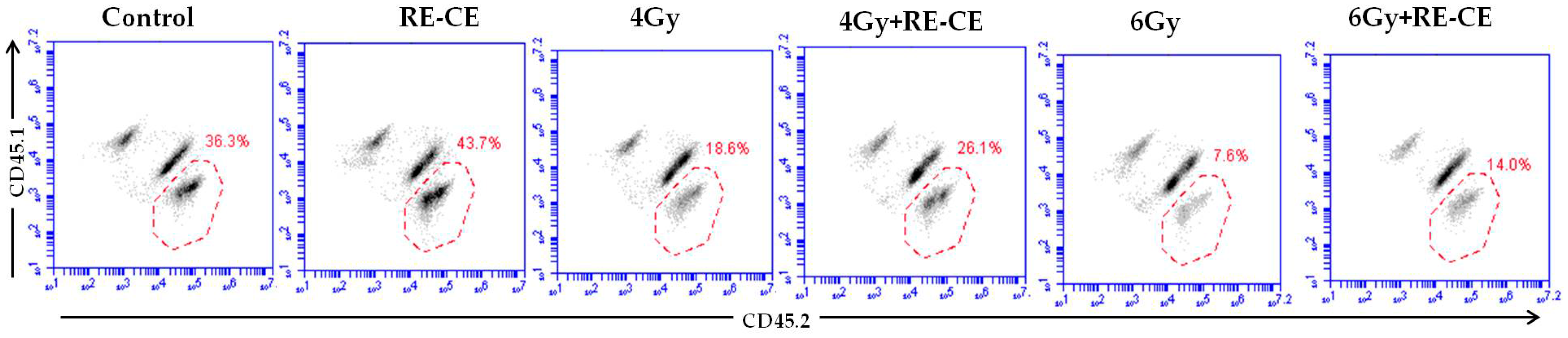
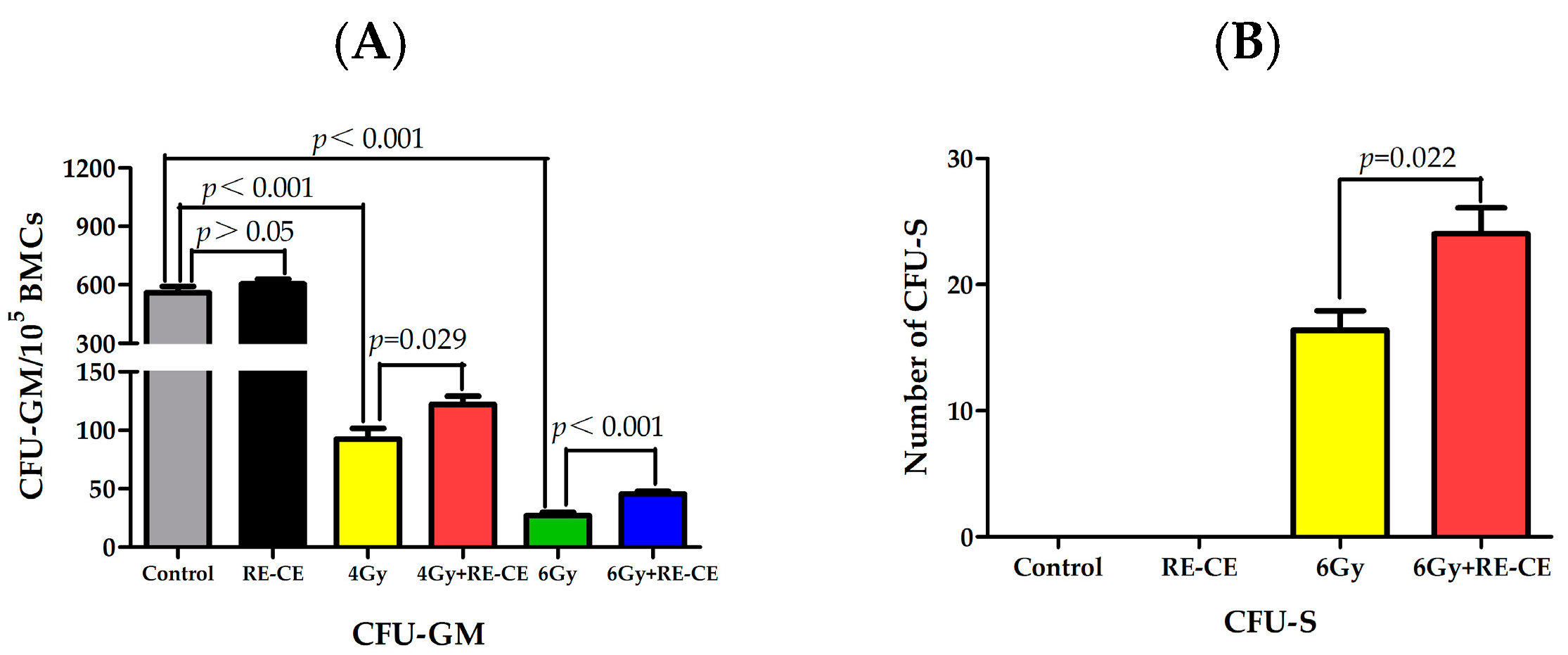
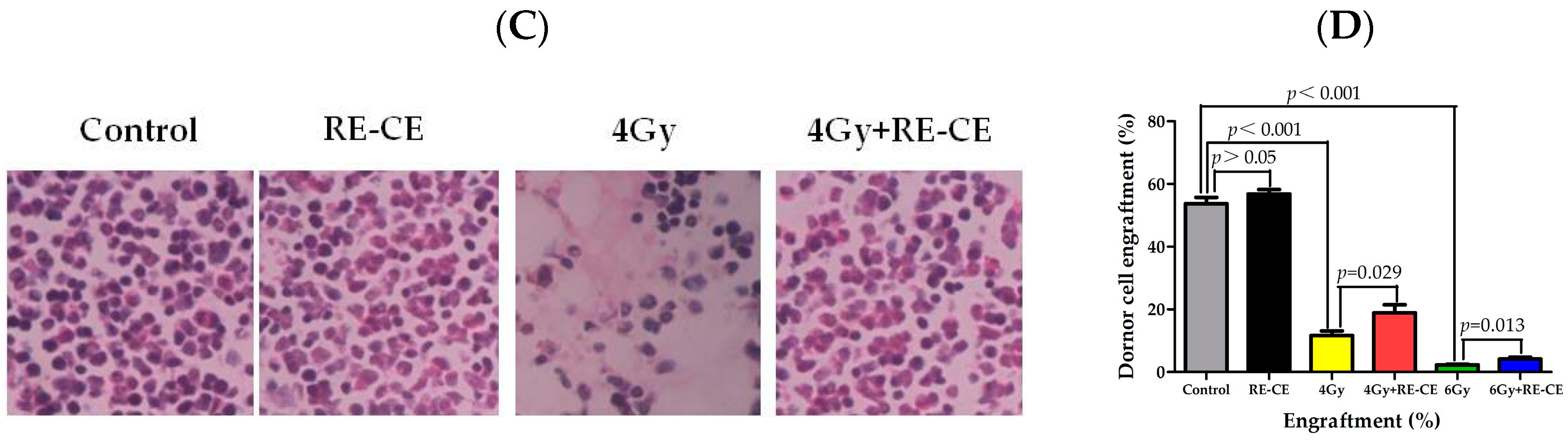
2.5. RE-CE Improves Colony Forming and Engraftment Abilities of HSPCs in Irradiated Mice
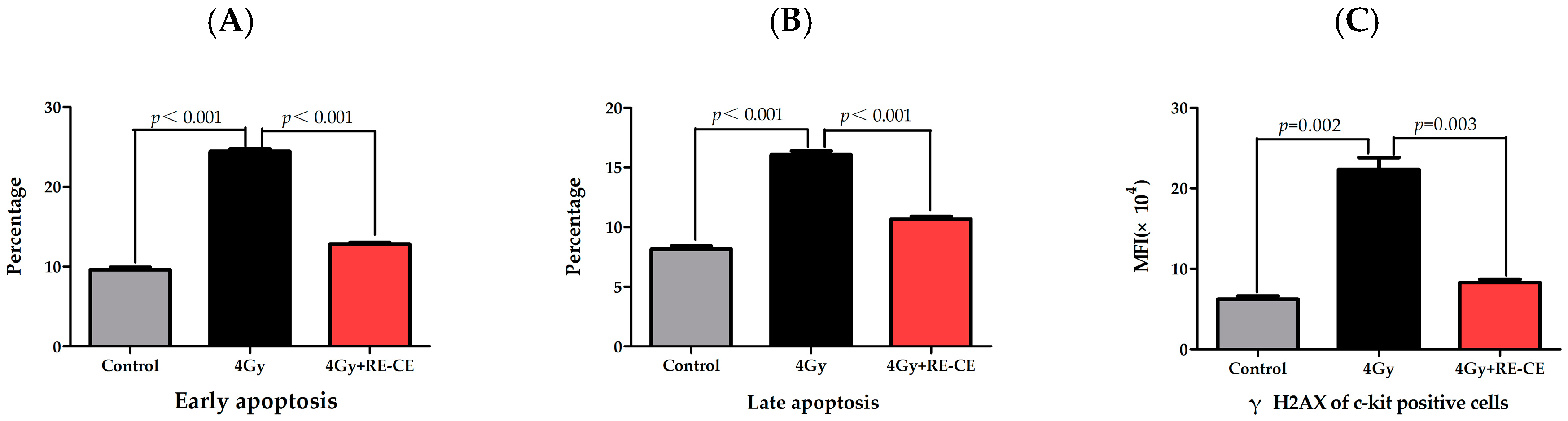
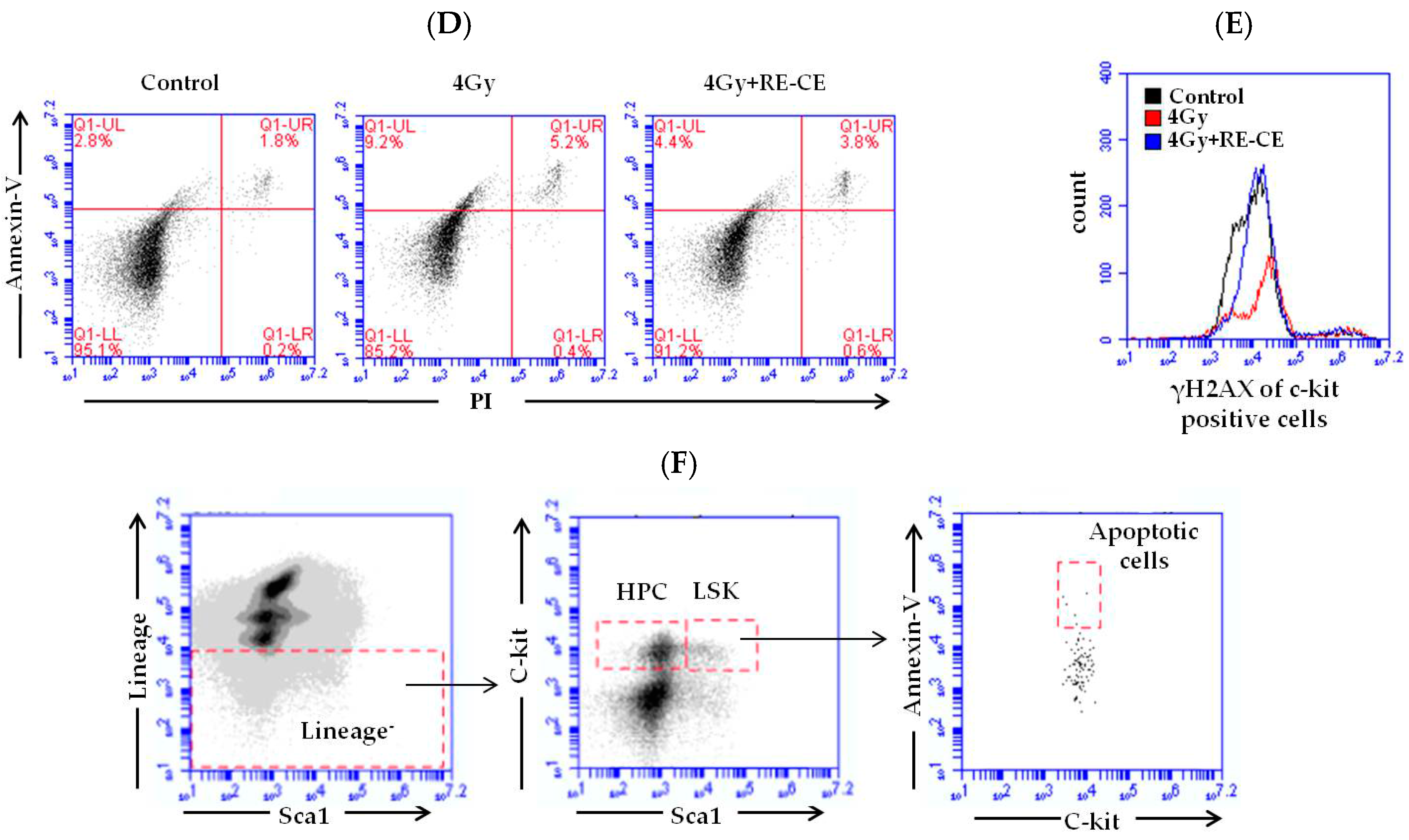
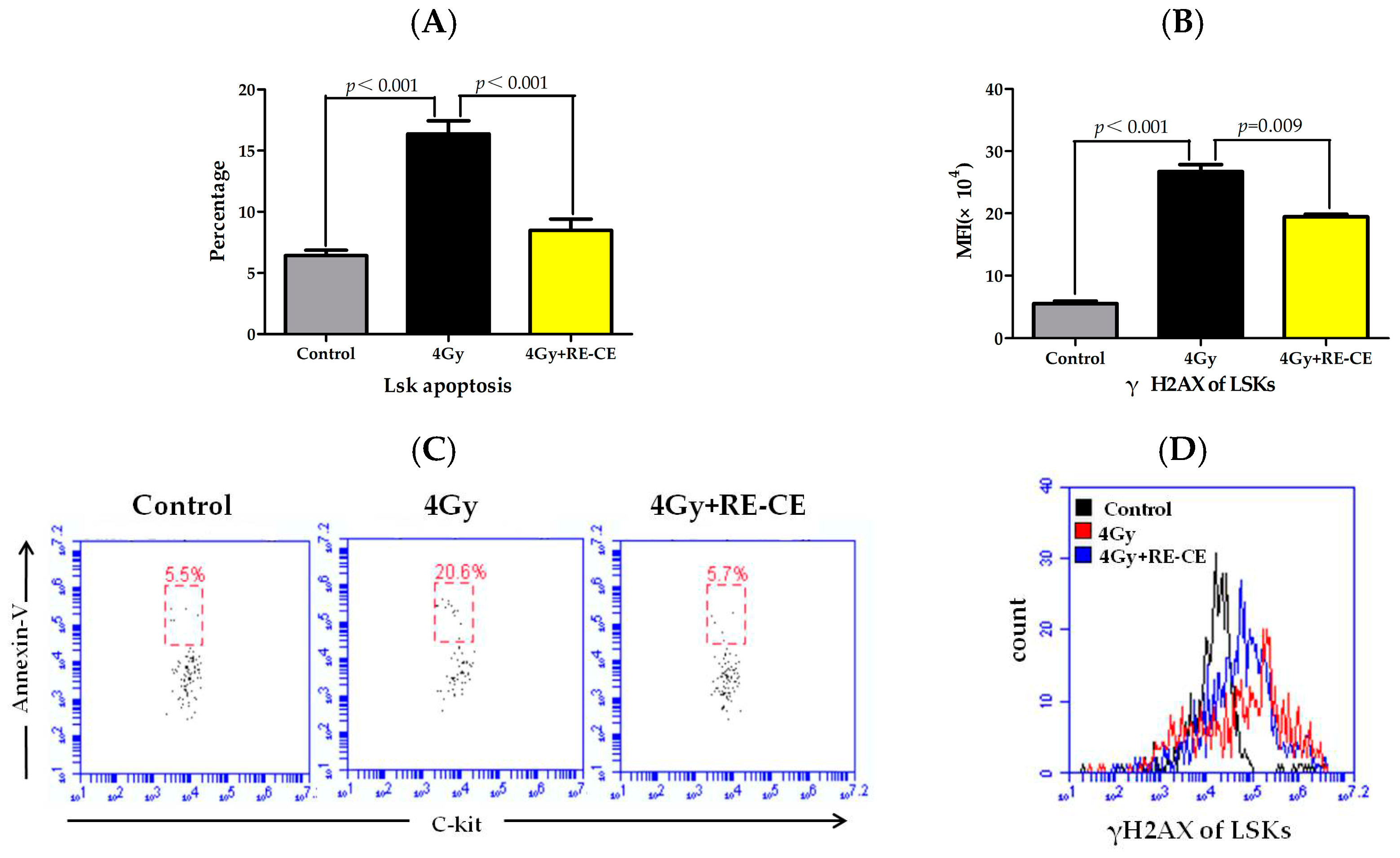
2.6. RE-CE Inhibits IR-Induced Apoptosis and DNA Damage in HSPCs
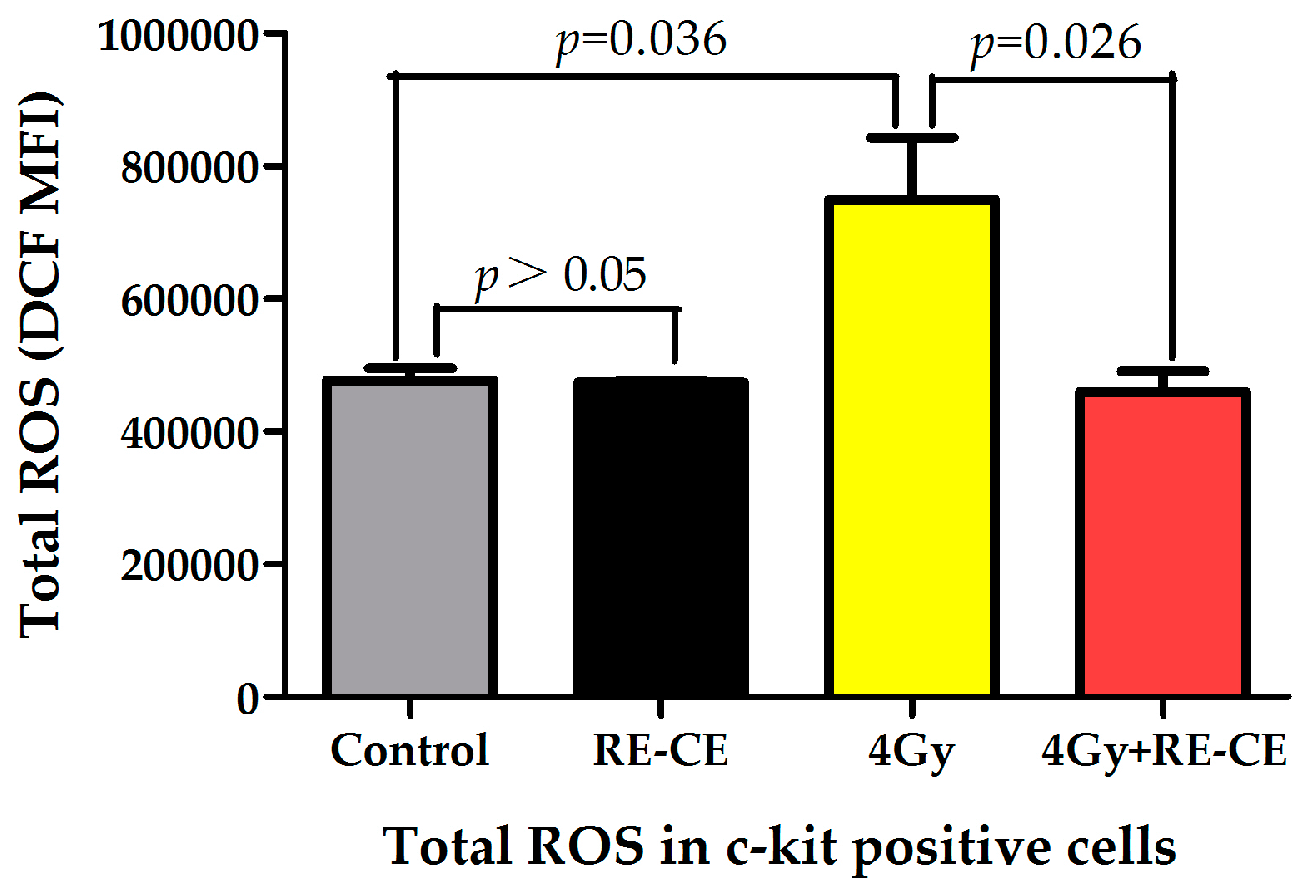
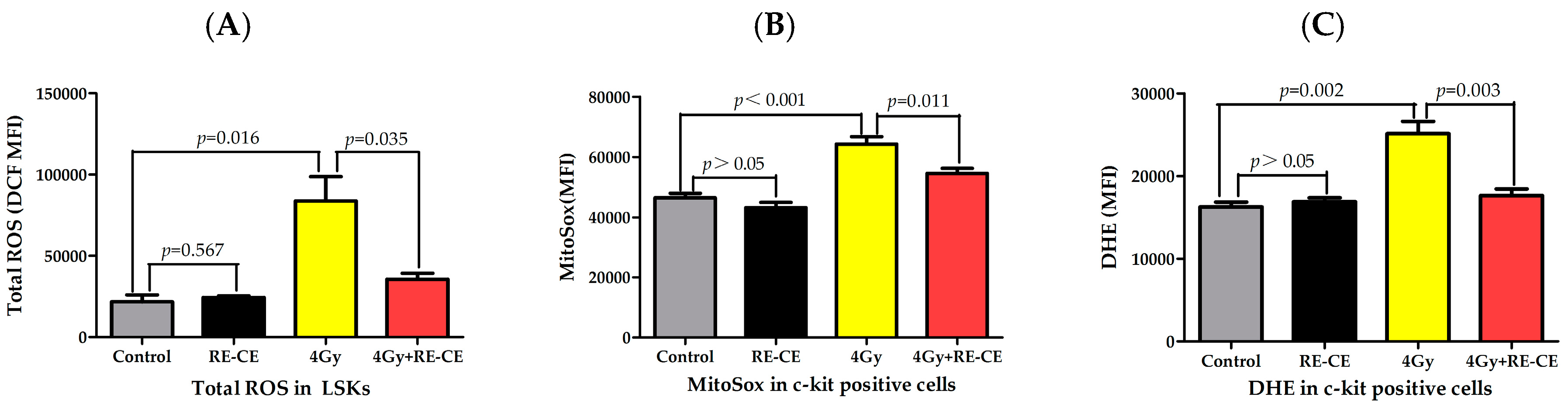
2.7. RE-CE Scavenges IR-Induced ROS in HSPCs
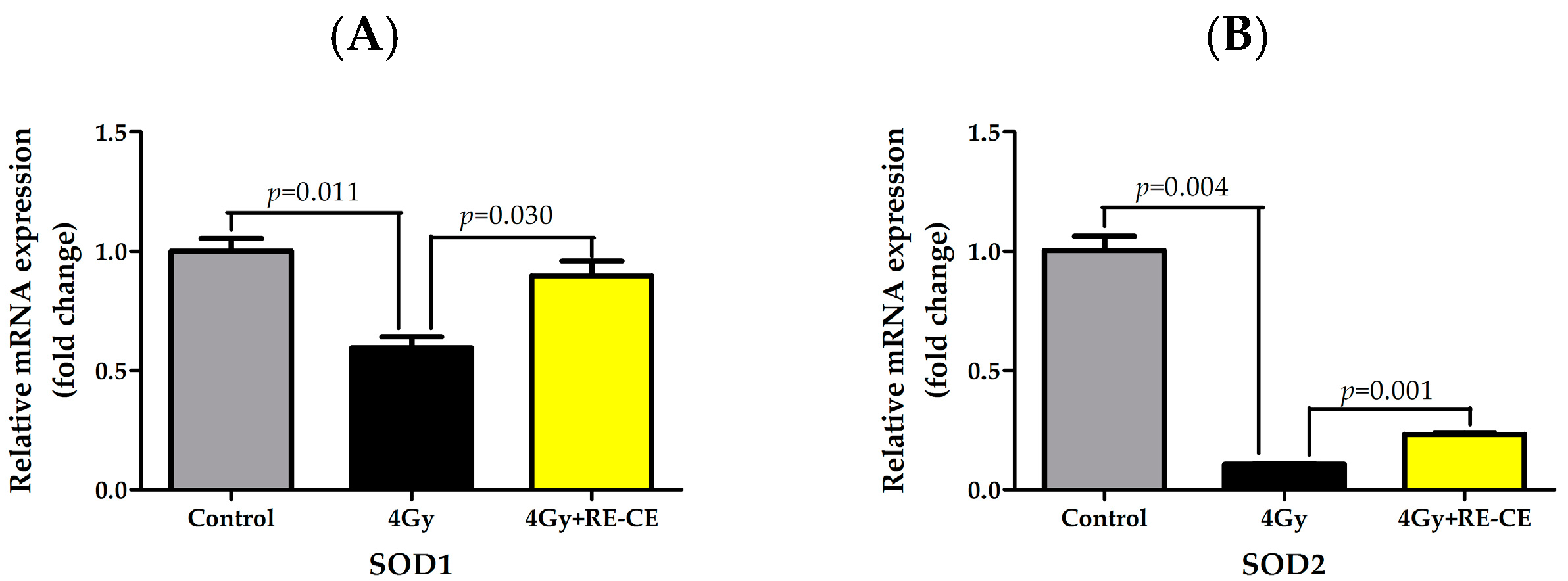
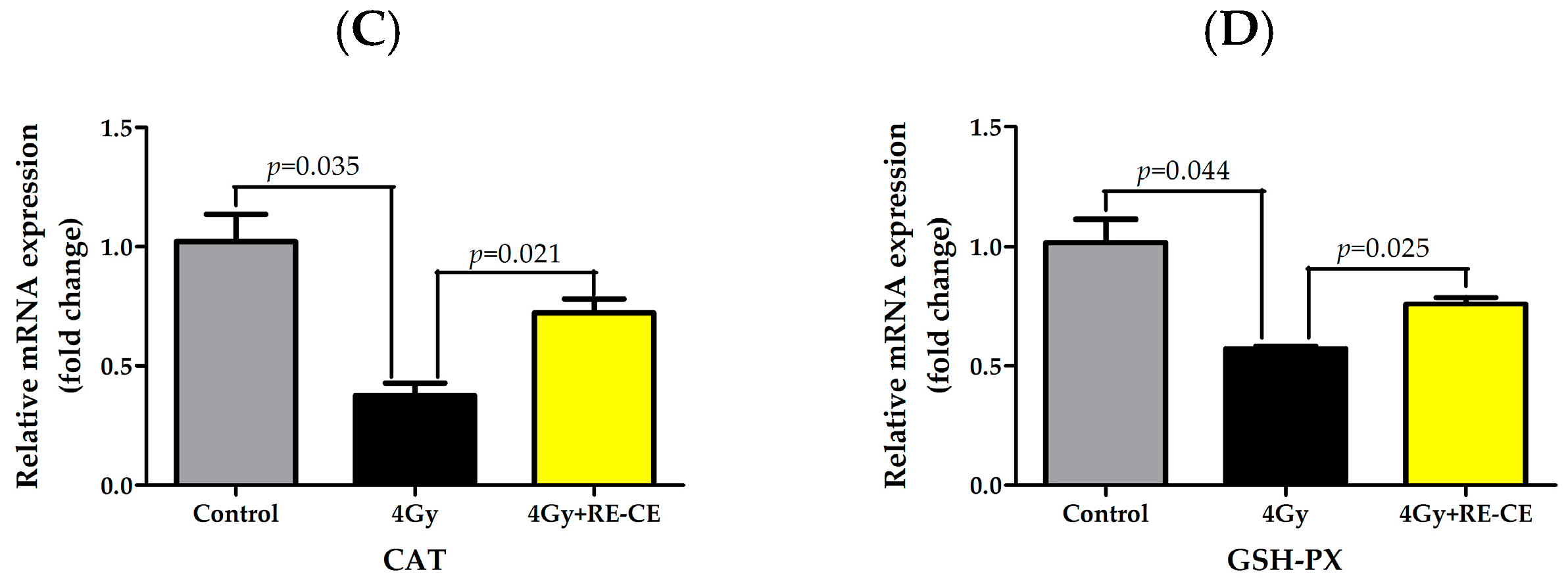
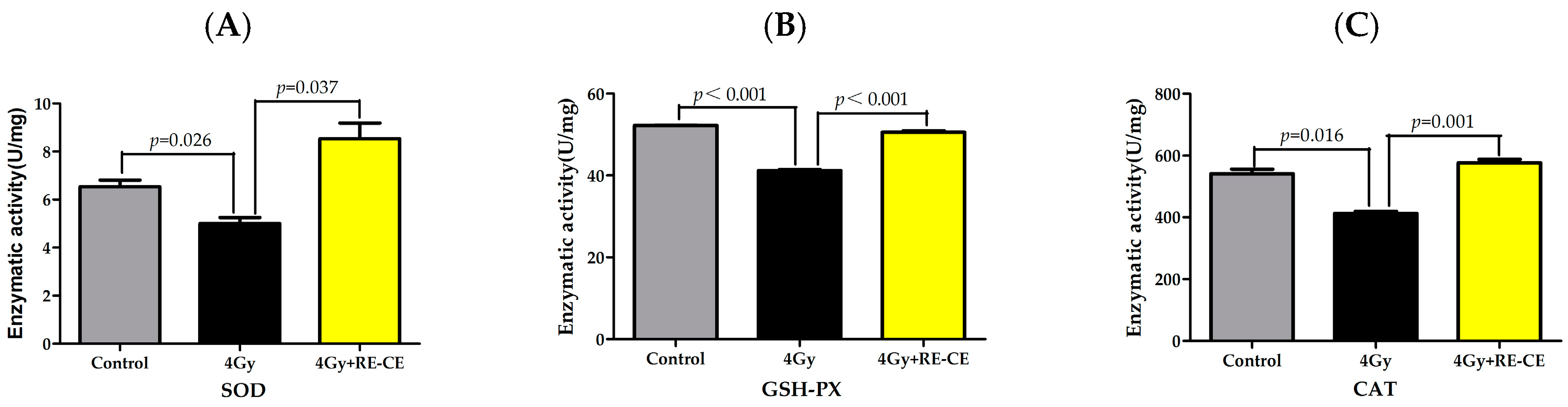
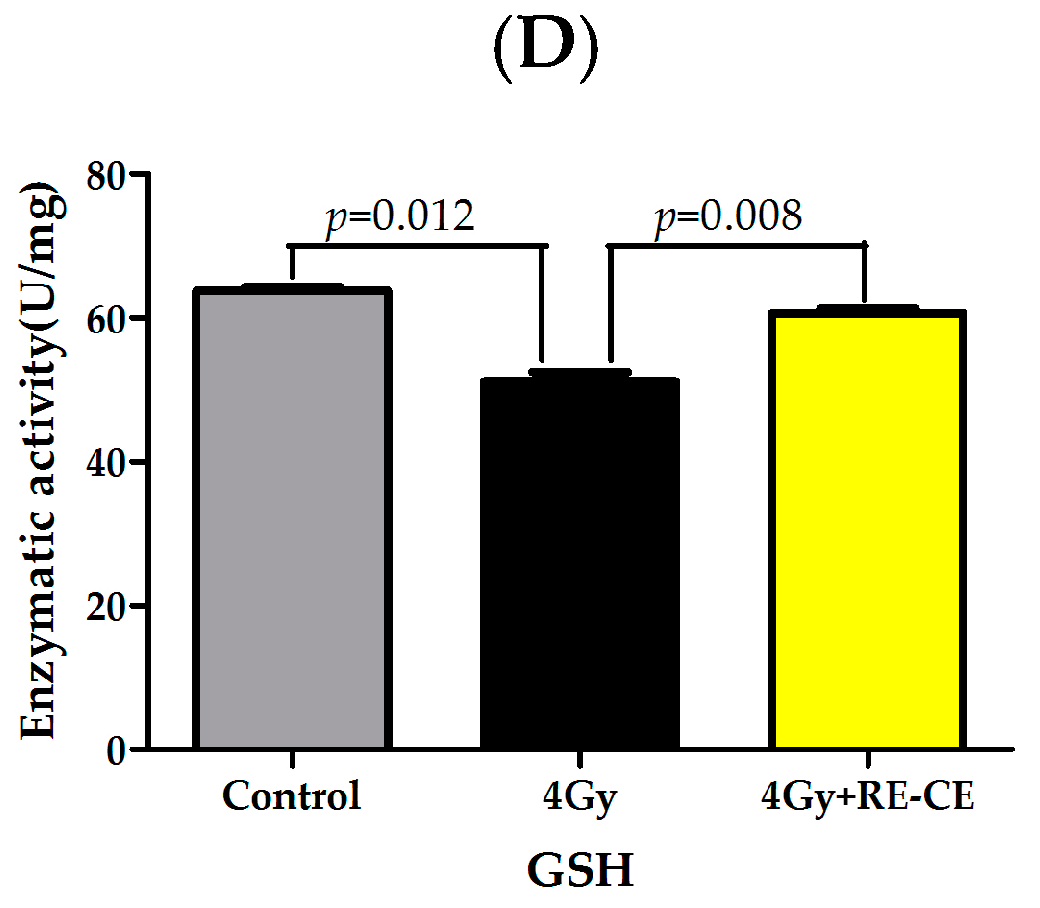
2.8. RE-CE Ameliorates IR-Induced Repression of Antioxidant Enzymatic Activities in c-Kit Positive Cells
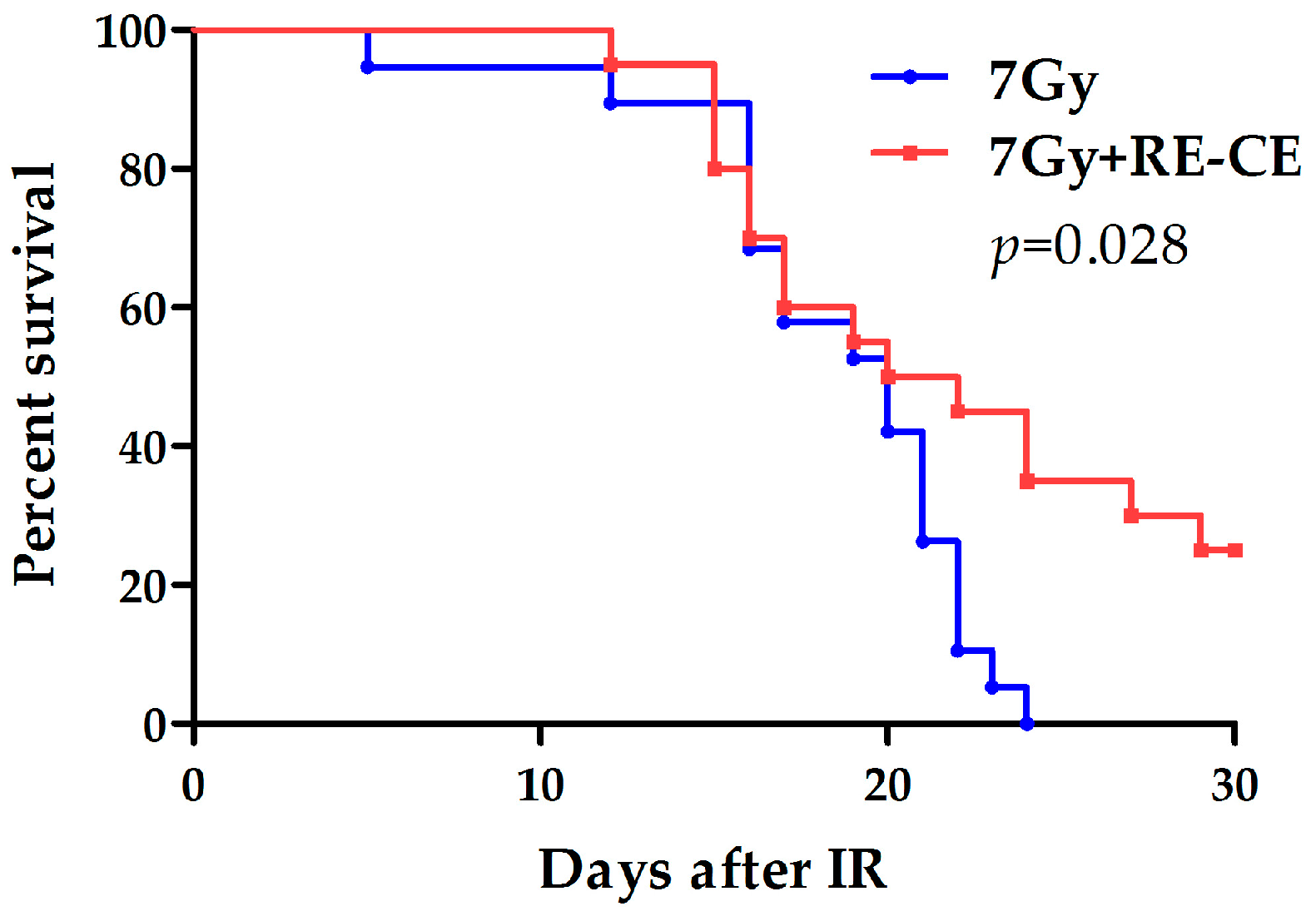
2.9. RE-CE Improves Survival of Lethally Irradiated Mice
2.10. Flavonoids Are Identified as the Major Component of Coriander Extract
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Mice
4.3. TBI and RE-CE Administration
4.4. Weight, Organ Index, Counts of Splenocyte and Thymocyte, and HE Staining
4.5. Peripheral Blood Cell Counts and Wright-Giemsa Staining
4.6. Isolation of BM Cells (BMCs) and Flow Cytometry Analysis
4.7. CFU-GM and CFU-S Assays
4.8. Competitive Bone Marrow Transplantation
4.9. Isolation of c-Kit Positive Cells
4.10. Apoptosis Assay
4.11. Analysis of γH2AX Staining in c-Kit Positive Cells
4.12. Analysis of Intracellular ROS Levels
4.13. Analysis of SOD, GSH-PX, GSH, and CAT Activities
4.14. Quantitative Real-Time PCR
4.15. Preparation and Component Identification of CE
4.16. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| IR | ionizing radiation |
| HSPCs | hematopoietic stem and progenitor cells |
| CE | coriander extract |
| ROS | reactive oxygen species |
| BM | bone marrow |
| TBI | total body irradiation |
| SOD | superoxide dismutase |
| GSH-PX | glutathione peroxidase |
| CAT | catalase |
| WBC | white blood cell |
| LY | lymphocyte |
| NE | neutrophil |
| BMCs | bone marrow cells |
| HPCs | hematopoietic progenitor cells |
| CFU-S | spleen colony-forming units |
| CFU-GM | colony of granulocyte macrophage cells |
Appendix A
References
- Xiao, M.; Whitnall, M.H. Pharmacological countermeasures for the acute radiation syndrome. Curr. Mol. Pharmacol. 2009, 2, 122–133. [Google Scholar] [CrossRef] [PubMed]
- Mauch, P.; Constine, L.; Greenberger, J.; Knospe, W.; Sullivan, J.; Liesveld, J.L.; Deeg, H.J. Hematopoietic stem cell compartment: Acute and late effects of radiation therapy and chemotherapy. Int. J. Radiat. Oncol. Biol. Phys. 1995, 31, 1319–1339. [Google Scholar] [CrossRef]
- Wang, Y.; Schulte, B.A.; LaRue, A.C.; Ogawa, M.; Zhou, D. Total body irradiation selectively induces murine hematopoietic stem cell senescence. Blood 2006, 107, 358–366. [Google Scholar] [CrossRef] [PubMed]
- Shao, L.; Luo, Y.; Zhou, D. Hematopoietic stem cell injury induced by ionizing radiation. Antioxid. Redox Signal. 2014, 20, 1447–1462. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, T.; Hayashi, I.; Shinohara, T.; Morishita, Y.; Nagamura, H.; Kusunoki, Y.; Kyoizumi, S.; Seyama, T.; Nakachi, K. Radiation-induced apoptosis of stem/progenitor cells in human umbilical cord blood is associated with alterations in reactive oxygen and intracellular ph. Mutat. Res. 2004, 556, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Liu, L.; Pazhanisamy, S.K.; Li, H.; Meng, A.; Zhou, D. Total body irradiation causes residual bone marrow injury by induction of persistent oxidative stress in murine hematopoietic stem cells. Free Radic. Biol. Med. 2010, 48, 348–356. [Google Scholar] [CrossRef] [PubMed]
- Shao, L.; Li, H.; Pazhanisamy, S.K.; Meng, A.; Wang, Y.; Zhou, D. Reactive oxygen species and hematopoietic stem cell senescence. Int. J. Hematol. 2011, 94, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.L.; Fang, L.W.; Lu, S.P.; Chou, C.K.; Luh, T.Y.; Lai, M.Z. DNA-damaging reagents induce apoptosis through reactive oxygen species-dependent FAS aggregation. Oncogene 2003, 22, 8168–8177. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, K.; Araki, K.Y.; Naka, K.; Arai, F.; Takubo, K.; Yamazaki, S.; Matsuoka, S.; Miyamoto, T.; Ito, K.; Ohmura, M.; et al. FOXO3A is essential for maintenance of the hematopoietic stem cell pool. Cell Stem Cell 2007, 1, 101–112. [Google Scholar] [CrossRef] [PubMed]
- Ito, K.; Hirao, A.; Arai, F.; Matsuoka, S.; Takubo, K.; Hamaguchi, I.; Nomiyama, K.; Hosokawa, K.; Sakurada, K.; Nakagata, N.; et al. Regulation of oxidative stress by ATM is required for self-renewal of haematopoietic stem cells. Nature 2004, 431, 997–1002. [Google Scholar] [CrossRef] [PubMed]
- Xiao, X.; Luo, H.; Vanek, K.N.; LaRue, A.C.; Schulte, B.A.; Wang, G.Y. Catalase inhibits ionizing radiation-induced apoptosis in hematopoietic stem and progenitor cells. Stem Cells Dev. 2015, 24, 1342–1351. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhai, Z.; Wang, Y.; Zhang, J.; Wu, H.; Wang, Y.; Li, C.; Li, D.; Lu, L.; Wang, X.; et al. Resveratrol ameliorates ionizing irradiation-induced long-term hematopoietic stem cell injury in mice. Free Radic. Biol. Med. 2013, 54, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Citrin, D.; Cotrim, A.P.; Hyodo, F.; Baum, B.J.; Krishna, M.C.; Mitchell, J.B. Radioprotectors and mitigators of radiation-induced normal tissue injury. Oncologist 2010, 15, 360–371. [Google Scholar] [CrossRef] [PubMed]
- Epperly, M.W.; Sikora, C.A.; DeFilippi, S.J.; Gretton, J.A.; Zhan, Q.; Kufe, D.W.; Greenberger, J.S. Manganese superoxide dismutase (SOD2) inhibits radiation-induced apoptosis by stabilization of the mitochondrial membrane. Radiat. Res. 2002, 157, 568–577. [Google Scholar] [CrossRef]
- Singh, V.K.; Romaine, P.L.; Seed, T.M. Medical countermeasures for radiation exposure and related injuries: Characterization of medicines, FDA-approval status and inclusion into the strategic national stockpile. Health Phys. 2015, 108, 607–630. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Lu, L.; Zhang, J.; Wang, X.; Xing, Y.; Wu, H.; Yang, X.; Shi, Z.; Zhao, M.; Fan, S.; et al. Mitigating the effects of xuebijing injection on hematopoietic cell injury induced by total body irradiation with gamma rays by decreasing reactive oxygen species levels. Int. J. Mol. Sci. 2014, 15, 10541–10553. [Google Scholar] [CrossRef] [PubMed]
- Laribi, B.; Kouki, K.; M’Hamdi, M.; Bettaieb, T. Coriander (Coriandrum sativum L.) and its bioactive constituents. Fitoterapia 2015, 103, 9–26. [Google Scholar] [CrossRef] [PubMed]
- Sahib, N.G.; Anwar, F.; Gilani, A.H.; Hamid, A.A.; Saari, N.; Alkharfy, K.M. Coriander (Coriandrum sativum L.): A potential source of high-value components for functional foods and nutraceuticals—A review. Phytother. Res. 2013, 27, 1439–1456. [Google Scholar] [PubMed]
- Gray, A.M.; Flatt, P.R. Insulin-releasing and insulin-like activity of the traditional anti-diabetic plant Coriandrum sativum (Coriander). Br. J. Nutr. 1999, 81, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Cioanca, O.; Hritcu, L.; Mihasan, M.; Hancianu, M. Cognitive-enhancing and antioxidant activities of inhaled coriander volatile oil in amyloid β (1–42) rat model of Alzheimer’s disease. Physiol. Behav. 2013, 120, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Hwang, E.; Lee, D.G.; Park, S.H.; Oh, M.S.; Kim, S.Y. Coriander leaf extract exerts antioxidant activity and protects against UVB-induced photoaging of skin by regulation of procollagen type I and MMP-1 expression. J. Med. Food 2014, 17, 985–995. [Google Scholar] [CrossRef] [PubMed]
- Velaga, M.K.; Yallapragada, P.R.; Williams, D.; Rajanna, S.; Bettaiya, R. Hydroalcoholic seed extract of Coriandrum sativum (Coriander) alleviates lead-induced oxidative stress in different regions of rat brain. Biol. Trace Elem. Res. 2014, 159, 351–363. [Google Scholar] [CrossRef] [PubMed]
- Zielniok, K.; Szkoda, K.; Gajewska, M.; Wilczak, J. Effect of biologically active substances present in water extracts of white mustard and coriander on antioxidant status and lipid peroxidation of mouse C2C12 skeletal muscle cells. J. Anim. Physiol. Anim. Nutr. 2016, 100, 988–1002. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.N.; Guo, Z.W.; Zhang, Y.; Qin, H.; Han, Y. Polysaccharide extracted from rheum tanguticum prevents irradiation-induced immune damage in mice. Asian Pac. J. Cancer Prev. APJCP 2012, 13, 1401–1405. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.K.; Rabbani, Z.N.; Folz, R.J.; Golson, M.L.; Huang, H.; Yu, D.; Samulski, T.S.; Dewhirst, M.W.; Anscher, M.S.; Vujaskovic, Z. Overexpression of extracellular superoxide dismutase protects mice from radiation-induced lung injury. Int. J. Radiat. Oncol. Biol. Phys. 2003, 57, 1056–1066. [Google Scholar] [CrossRef]
- Wang, J.; Sun, Q.; Morita, Y.; Jiang, H.; Gross, A.; Lechel, A.; Hildner, K.; Guachalla, L.M.; Gompf, A.; Hartmann, D.; et al. A differentiation checkpoint limits hematopoietic stem cell self-renewal in response to DNA damage. Cell 2012, 148, 1001–1014. [Google Scholar] [CrossRef] [PubMed]
- Doan, P.L.; Russell, J.L.; Himburg, H.A.; Helms, K.; Harris, J.R.; Lucas, J.; Holshausen, K.C.; Meadows, S.K.; Daher, P.; Jeffords, L.B.; et al. Tie2+ bone marrow endothelial cells regulate hematopoietic stem cell regeneration following radiation injury. Stem Cells 2013, 31, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Lefrancais, E.; Ortiz-Munoz, G.; Caudrillier, A.; Mallavia, B.; Liu, F.; Sayah, D.M.; Thornton, E.E.; Headley, M.B.; David, T.; Coughlin, S.R.; et al. The lung is a site of platelet biogenesis and a reservoir for haematopoietic progenitors. Nature 2017, 544, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Porter, R.L.; Georger, M.A.; Bromberg, O.; McGrath, K.E.; Frisch, B.J.; Becker, M.W.; Calvi, L.M. Prostaglandin E2 increases hematopoietic stem cell survival and accelerates hematopoietic recovery after radiation injury. Stem Cells 2013, 31, 372–383. [Google Scholar] [CrossRef] [PubMed]
- Wilson, A.; Laurenti, E.; Trumpp, A. Balancing dormant and self-renewing hematopoietic stem cells. Curr. Opin. Genet. Dev. 2009, 19, 461–468. [Google Scholar] [CrossRef] [PubMed]
- Tang, E.L.; Rajarajeswaran, J.; Fung, S.; Kanthimathi, M.S. Petroselinum crispum has antioxidant properties, protects against DNA damage and inhibits proliferation and migration of cancer cells. J. Sci. Food Agric. 2015, 95, 2763–2771. [Google Scholar] [CrossRef] [PubMed]
- Lefort, E.C.; Blay, J. Apigenin and its impact on gastrointestinal cancers. Mol. Nutr. Food Res. 2013, 57, 126–144. [Google Scholar] [CrossRef] [PubMed]
- Tian, R.; Yang, W.; Xue, Q.; Gao, L.; Huo, J.; Ren, D.; Chen, X. Rutin ameliorates diabetic neuropathy by lowering plasma glucose and decreasing oxidative stress via NRF2 signaling pathway in rats. Eur. J. Pharmacol. 2016, 771, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Choi, K.S.; Kundu, J.K.; Chun, K.S.; Na, H.K.; Surh, Y.J. Rutin inhibits UVB radiation-induced expression of COX-2 and inos in hairless mouse skin: P38 MAP kinase and JNK as potential targets. Arch. Biochem. Biophys. 2014, 559, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Dutta, S.; Yashavarddhan, M.H.; Srivastava, N.N.; Ranjan, R.; Bajaj, S.; Kalita, B.; Singh, A.; Flora, S.J.; Gupta, M.L. Countering effects of a combination of podophyllotoxin, podophyllotoxin β-d-glucoside and rutin hydrate in minimizing radiation induced chromosomal damage, ROS and apoptosis in human blood lymphocytes. Food Chem. Toxicol. 2016, 91, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Patil, S.L.; Mallaiah, S.H.; Patil, R.K. Antioxidative and radioprotective potential of rutin and quercetin in swiss albino mice exposed to gamma radiation. J. Med. Phys. 2013, 38, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Kalita, B.; Ranjan, R.; Singh, A.; Yashavarddhan, M.H.; Bajaj, S.; Gupta, M.L. A combination of podophyllotoxin and rutin attenuates radiation induced gastrointestinal injury by negatively regulating NF-κB/p53 signaling in lethally irradiated mice. PLoS ONE 2016, 11, e0168525. [Google Scholar] [CrossRef] [PubMed]
- Abd-El-Fattah, A.A.; El-Sawalhi, M.M.; Rashed, E.R.; El-Ghazaly, M.A. Possible role of vitamin E, coenzyme q10 and rutin in protection against cerebral ischemia/reperfusion injury in irradiated rats. Int. J. Radiat. Biol. 2010, 86, 1070–1078. [Google Scholar] [CrossRef] [PubMed]
- Cigremis, Y.; Turel, H.; Adiguzel, K.; Akgoz, M.; Kart, A.; Karaman, M.; Ozen, H. The effects of acute acetaminophen toxicity on hepatic mrna expression of SOD, CAT, GSH-PX, and levels of peroxynitrite, nitric oxide, reduced glutathione, and malondialdehyde in rabbit. Mol. Cell. Biochem. 2009, 327, 277. [Google Scholar] [CrossRef]
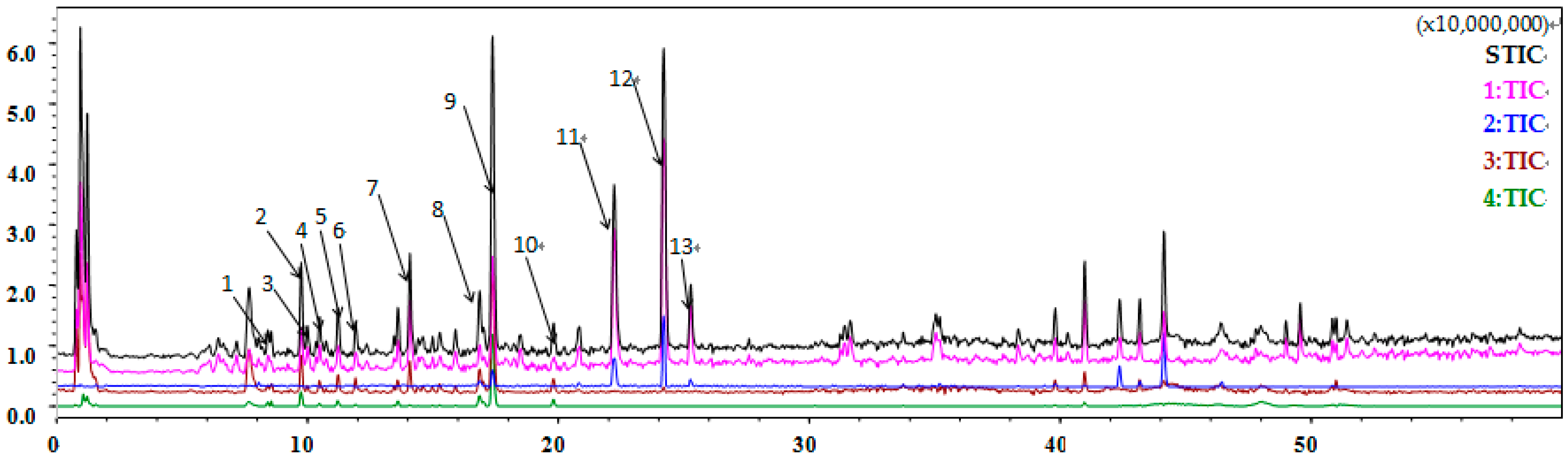
| Peak Number | Retention Time (min) | Molecular Ion Peak m/z | Ion Mode | Molecular Formula | Compound Type | Compound Name |
|---|---|---|---|---|---|---|
| 1 | 8.452 | 377.0836 | [M + Na]+ | C16H18O9 | Phenolic acid | Chlorogenic acid |
| 353.0790 | [M − H]− | |||||
| 2 | 9.735 | 363.0687 | [M + Na]+ | C15H16O9 | Coumarin | Esculin or Daphnetin-8-O-glucoside |
| 339.0614 | [M − H]− | |||||
| 3 | 10.010 | 371.1245 | [M + HCOO]− | C18H18N2O4 | Animo acid | Hydroxybenozyl tryptophan |
| 4 | 10.487 | 365.1189 | [M + Na]+ | C18H22O8 | Phenolic acid | Methoxycinnamic acid glucoside |
| 341.1163 | [M − H]− | |||||
| 5 | 11.220 | 379.0975 | [M + Na]+ | C16H20O9 | Phenolic acid | Ferulic acid glucoside |
| 355.0943 | [M − H]− | |||||
| 6 | 11.917 | 409.1818 | [M + Na]+ | C19H30O8 | Norcarotenoid | Citroside A or B or Icariside B2 |
| 431.1830 | [M + HCOO]− | |||||
| 7 | 14.080 | 433.1079 | [M + Na]+ | C19H22O10 | Coumarin | Coriandrone E hexoside |
| 455.1075 | [M + HCOO]− | |||||
| 8 | 16.848 | 479.0805 | [M + H]+ | C21H18O13 | Flavonoid | Quercetin 3-glucuronide |
| 477.0564 | [M − H]− | |||||
| 9 | 17.398 | 611.1590 | [M + H]+ | C27H30O16 | Flavonoid | Rutin |
| 609.1390 | [M − H]− | |||||
| 10 | 19.800 | 617.1465 | [M + Na]+ | C27H30O15 | Flavonoid | Nicotiflorin |
| 593.1367 | [M − H]− | |||||
| 11 | 22.220 | 255.0614 | [M + Na]+ | C13H12O4 | Coumarin | Dihydrocoriandrin |
| 12 | 24.200 | 293.1311 | [M + H]+ | C16H20O5 | Coumarin | Coriandrone A or B |
| 13 | 25.282 | 253.0453 | [M + Na]+ | C13H10O4 | Coumarin | Coriandrin |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, X.; Xue, X.; Zhao, Y.; Li, Y.; Liu, W.; Zhang, J.; Fan, S. Rutin-Enriched Extract from Coriandrum sativum L. Ameliorates Ionizing Radiation-Induced Hematopoietic Injury. Int. J. Mol. Sci. 2017, 18, 942. https://doi.org/10.3390/ijms18050942
Han X, Xue X, Zhao Y, Li Y, Liu W, Zhang J, Fan S. Rutin-Enriched Extract from Coriandrum sativum L. Ameliorates Ionizing Radiation-Induced Hematopoietic Injury. International Journal of Molecular Sciences. 2017; 18(5):942. https://doi.org/10.3390/ijms18050942
Chicago/Turabian StyleHan, Xiaodan, Xiaolei Xue, Yu Zhao, Yuan Li, Weili Liu, Junling Zhang, and Saijun Fan. 2017. "Rutin-Enriched Extract from Coriandrum sativum L. Ameliorates Ionizing Radiation-Induced Hematopoietic Injury" International Journal of Molecular Sciences 18, no. 5: 942. https://doi.org/10.3390/ijms18050942
APA StyleHan, X., Xue, X., Zhao, Y., Li, Y., Liu, W., Zhang, J., & Fan, S. (2017). Rutin-Enriched Extract from Coriandrum sativum L. Ameliorates Ionizing Radiation-Induced Hematopoietic Injury. International Journal of Molecular Sciences, 18(5), 942. https://doi.org/10.3390/ijms18050942





