Adverse Health Effects of Thirdhand Smoke: From Cell to Animal Models
Abstract
:1. Thirdhand Smoke: The Concept and Features
2. Exposure to Thirdhand Smoke (THS) Is Genotoxic
2.1. THS Induces DNA Strand Breaks in Human Cells
2.2. THS Induces Oxidative DNA Damage in Human Cells and Animal Models
2.3. 1-(N-Methyl-N-Nitrosamino)-1-(3-Pyridinyl)-4-Butanal (NNA) Exposure Causes the Formation of DNA Adducts
3. THS Exposure Causes Metabolomic Changes in Reproductive Cells
4. Early Life THS Exposure Affects Body Mass and the Development of Immunity in Mice
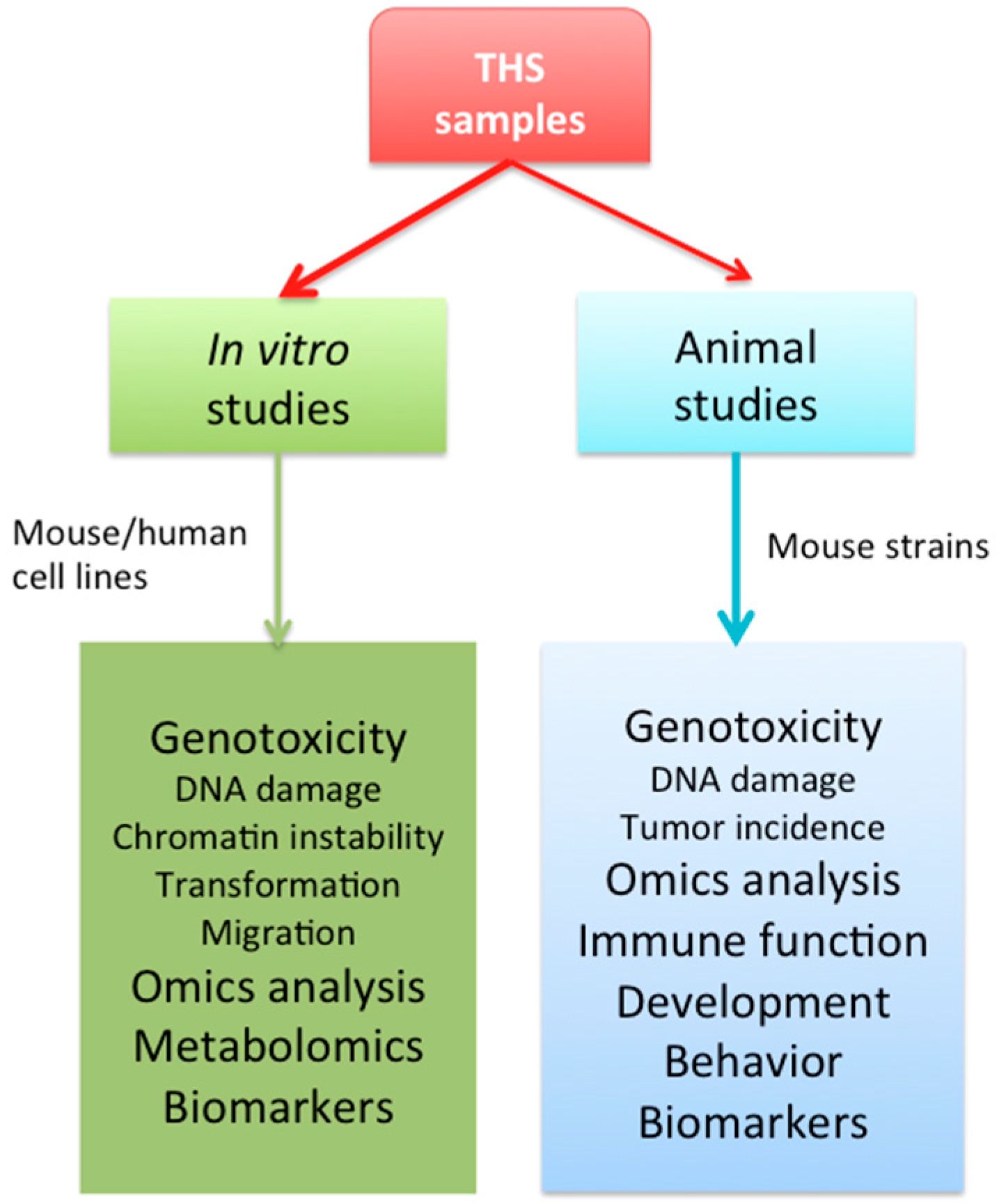
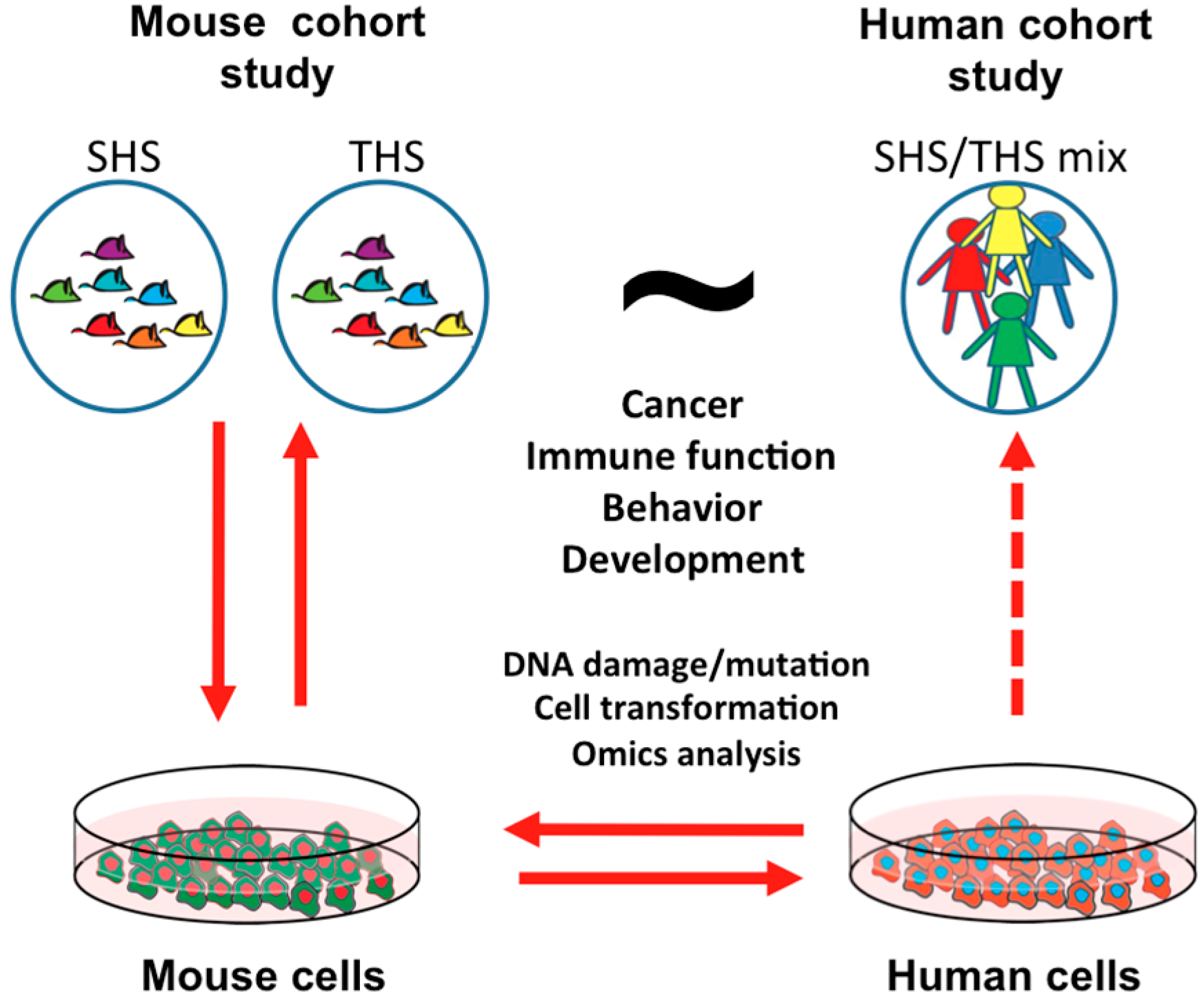
5. Summary and Perspective
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| THS | Thirdhand smoke |
| SHS | Secondhand smoke |
| SSS | Mainstream smoke |
| DSB | Double-strand break |
| dGuo | 2′-Deoxyguanosine |
| dCyt | 2′-Deaxycytidine |
| IARC | International Agency for Research on Cancer |
| NTP | National Toxicology Program |
| HONO | Nitrous acid |
| TSNA | Tobacco-specific nitrosamine |
| NNA | 1-(N-Methyl-N-nitrosamino)-1-(3-pyridinyl)-4-butanal |
| NNK | 4-(Methylnitrosamino)-1-(3-pyridyl)-1-butanone |
| NNN | N′-Nitrosonornicotine |
| LA-qPCR | Long amplicon-qPCR |
| HPRT | Hypoxanthine-guanine phosphoribosyltransferase |
| POLB | DNA polymerase β |
| HPB | 4-Hydroxy-1-(3-pyridyl)-1butanone |
| FACS | Fluorescence-activated cell sorting |
References
- Hang, B.; Chenna, A.; Gundel, L. Smoke: The hidden/emerging risks. Chem. Ind. 2014, 9. [Google Scholar] [CrossRef]
- US-DHHS. Smoking and Health. Report of the Advisory Committee to the Surgeon General of the Public Health Service; United States, Public Health Service, Office of the Surgeon General: Washington, DC, USA, 1964.
- US-DHHS. The Health Consequences of Involuntary Smoking: A Report to the Surgeon General; United States, Public Health Service, Office on Smoking and Health: Washington, DC, USA, 1986.
- Schick, S.F.; Glantz, S. Concentrations of the carcinogen 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone in sidestream cigarette smoke increase after release into indoor air: Results from unpublished tobacco industry research. Cancer Epidemiol. Biomarkers Prev. 2007, 16, 1547–1553. [Google Scholar] [CrossRef] [PubMed]
- Rabin, R.B. A New Cigarette Hazard: “Third-Hand Smoke.”. 2009. The New York Times. Available online: http://www.nytimes.com/2009/01/03/health/research/03smoke.html?_r=1&em=&pagewanted=print (accessed on 3 January 2009).
- Matt, G.E.; Quintana, P.J.; Destaillats, H.; Gundel, L.A.; Sleiman, M.; Singer, B.C.; Jacob, P., III; Benowitz, N.; Winickoff, J.P.; Rehan, V.; et al. Thirdhand tobacco smoke: Emerging evidence and arguments for a multidisciplinary research agenda. Environ. Health Perspect. 2011, 119, 1218–1226. [Google Scholar] [CrossRef] [PubMed]
- Jacob, P.; Benowitz, N.L.; Destaillats, H.; Gundel, L.; Hang, B.; Martins-Green, M.; Matt, G.E.; Quintana, P.J.; Samet, J.M.; Schick, S.F.; et al. Thirdhand Smoke: New Evidence, Challenges, and Future Directions. Chem. Res. Toxicol. 2017, 30, 270–294. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Qiao, S.; Chen, M.; Xia, Y.; Hang, B.; Cheng, S. A investigation of thirdhand smoke pollution in 3 types of places of Nanjing, 2014. Zhonghua Yu Fang Yi Xue Za Zhi 2015, 49, 31–35. [Google Scholar] [PubMed]
- Sleiman, M.; Destaillats, H.; Smith, J.D.; Liu, C.-L.; Ahmed, M.; Wilson, K.R.; Gundel, L.A. Secondary organic aerosol formation from ozone-initiated reactions with nicotine and secondhand tobacco smoke. Atmos. Environ. 2010, 44, 4191–4198. [Google Scholar] [CrossRef]
- Sleiman, M.; Gundel, L.A.; Pankow, J.F.; Jacob, P., III; Singer, B.C.; Destaillats, H. Formation of carcinogens indoors by surface-mediated reactions of nicotine with nitrous acid, leading to potential thirdhand smoke hazards. Proc. Natl. Acad. Sci. USA 2010, 107, 6576–6581. [Google Scholar] [CrossRef] [PubMed]
- Hecht, S.S. Progress and challenges in selected areas of tobacco carcinogenesis. Chem. Res. Toxicol. 2008, 21, 160–171. [Google Scholar] [CrossRef] [PubMed]
- Hang, B. Formation and repair of tobacco carcinogen-derived bulky DNA adducts. J. Nucleic Acids 2010, 2010, 709521. [Google Scholar] [CrossRef] [PubMed]
- Destaillats, H.; Singer, B.C.; Lee, S.K.; Gundel, L.A. Effect of ozone on nicotine desorption from model surfaces: Evidence for heterogeneous chemistry. Environ. Sci. Technol. 2006, 40, 1799–1805. [Google Scholar] [CrossRef] [PubMed]
- Hang, B.; Sarker, A.H.; Havel, C.; Saha, S.; Hazra, T.K.; Schick, S.; Jacob, P., III; Rehan, V.K.; Chenna, A.; Sharan, D.; et al. Thirdhand smoke causes DNA damage in human cells. Mutagenesis. 2013, 28, 381–391. [Google Scholar] [CrossRef] [PubMed]
- Hang, B.; Lavarone, A.; Havel, C.; Jacob, P., III; Villalta, P.; Matter, B.; Sharan, D.; Hang, M.; Sleiman, M.; Destaillats, H.; et al. NNA, a Thirdhand Smoke Constituent, Induces DNA Damage in Vitro and in Human Cells. In Proceedings of the 247th National Meeting of the American Chemical Society (ACS) with Press Release, Dallas, TX, USA, 14–19 March 2014; American Chemical Society: Washington, DC, USA, 2014. [Google Scholar]
- Santos, J.H.; Meyer, J.N.; Mandavilli, B.S.; van Houten, B. Quantitative PCR-based measurement of nuclear and mitochondrial DNA damage and repair in mammalian cells. Methods Mol. Biol. 2006, 314, 183–199. [Google Scholar] [PubMed]
- Dhall, S.; Alamat, R.; Castro, A.; Sarker, A.H.; Mao, J.H.; Chan, A.; Hang, B.; Martins-Green, M. Tobacco toxins deposited on surfaces (third hand smoke) impair wound healing. Clin. Sci. 2016, 130, 1269–1284. [Google Scholar] [CrossRef] [PubMed]
- Lao, Y.; Yu, N.; Kassie, F.; Villalta, P.W.; Hecht, S.S. Formation and accumulation of pyridyloxobutyl DNA adducts in F344 rats chronically treated with 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone and enantiomers of its metabolite, 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol. Chem. Res. Toxicol. 2007, 20, 235–245. [Google Scholar] [CrossRef] [PubMed]
- Peterson, L.A. Formation, repair, and genotoxic properties of bulky DNA adducts formed from tobacco-specific nitrosamines. J. Nucleic Acids 2010, 2010, 284935. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Chen, M.; Yao, M.; Ji, X.; Mao, Z.; Tang, W.; Qiao, S.; Schick, S.F.; Mao, J.H.; Hang, B.; et al. Metabolomics reveals metabolic changes in male reproductive cells exposed to thirdhand smoke. Sci. Rep. 2015, 5, 15512. [Google Scholar] [CrossRef] [PubMed]
- Matt, G.E.; Quintana, P.J.; Zakarian, J.M.; Fortmann, A.L.; Chatfield, D.A.; Hoh, E.; Uribe, A.M.; Hovell, M.F. When smokers move out and non-smokers move in: Residential thirdhand smoke pollution and exposure. Tob. Control. 2011, 20. [Google Scholar] [CrossRef] [PubMed]
- Northrup, T.F.; Khan, A.M.; Jacob, P., III; Benowitz, N.L.; Hoh, E.; Hovell, M.F.; Matt, G.E.; Stotts, A.L. Thirdhand smoke contamination in hospital settings: Assessing exposure risk for vulnerable paediatric patients. Tob. Control. 2015, 25. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, N.; Ozel, M.Z.; Lewis, A.C.; Marce, R.M.; Borrull, F.; Hamilton, J.F. Exposure to nitrosamines in thirdhand tobacco smoke increases cancer risk in non-smokers. Environ. Int. 2014, 71, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Yolton, K.; Khoury, J.; Xu, Y.; Succop, P.; Lanphear, B.; Bernert, J.T.; Lester, B. Low level prenatal exposure to nicotine and infant neurobehavior. Neurotoxicol. Teratol. 2009, 31, 356–363. [Google Scholar] [CrossRef] [PubMed]
- Hang, B.; Snijders, A.M.; Huang, Y.; Schick, S.F.; Wang, P.; Xia, Y.; Havel, C.; Jacob, P., III; Benowitz, N.; Destaillats, H.; et al. Early exposure to thirdhand cigarette smoke affects body mass and the development of immunity in mice. Sci. Rep. 2017, 7, 41915. [Google Scholar] [CrossRef] [PubMed]
- Talbot, P.; Lin, S. The effect of cigarette smoke on fertilization and pre-implantation development: Assessment using animal models, clinical data, and stem cells. Biol. Res. 2011, 44, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Talbot, P.; Lin, S. Mouse and human embryonic stem cells: Can they improve human health by preventing disease? Curr. Top. Med. Chem. 2011, 11, 1638–1652. [Google Scholar] [PubMed]
- Zahedi, A.; On, V.; Lin, S.C.; Bays, B.C.; Omaiye, E.; Bhanu, B.; Talbot, P. Evaluating cell processes, quality, and biomarkers in pluripotent stem cells using video bioinformatics. PLoS ONE 2016, 11, e0148642. [Google Scholar] [CrossRef] [PubMed]
- Bahl, V.; Shim, H.J.; Jacob, P., III; Dias, K.; Schick, S.F.; Talbot, P. Thirdhand smoke: Chemical dynamics, cytotoxicity, and genotoxicity in outdoor and indoor environments. Toxicol. In Vitro 2016, 32, 220–231. [Google Scholar] [CrossRef] [PubMed]
- Figueiro, L.R.; Dantas, D.C.; Linden, R.; Ziulkoski, A.L. Thirdhand tobacco smoke: Procedures to evaluate cytotoxicity in cell cultures. Toxicol. Mech. Methods, 2016, 26, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Riveles, K.; Iv, M.; Arey, J.; Talbot, P. Pyridines in cigarette smoke inhibit hamster oviductal functioning in picomolar doses. Reprod. Toxicol. 2003, 17, 191–202. [Google Scholar] [CrossRef]
- Riveles, K.; Roza, R.; Talbot, P. Phenols, quinolines, indoles, benzene, and 2-cyclopenten-1-ones are oviductal toxicants in cigarette smoke. Toxicol. Sci. 2005, 86, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Bahl, V.; Johnson, K.; Phandthong, R.; Zahedi, A.; Schick, S.F.; Talbot, P. Thirdhand cigarette smoke causes stress-induced mitochondrial hyperfusion and alters the transcriptional profile of stem cells. Toxicol. Sci. 2016, 153, 55–69. [Google Scholar] [CrossRef] [PubMed]
- Martins-Green, M.; Adhami, N.; Frankos, M.; Valdez, M.; Goodwin, B.; Lyubovitsky, J.; Dhall, S.; Garcia, M.; Egiebor, I.; Martinez, B.; et al. Cigarette smoke toxins deposited on surfaces: Implications for human health. PLoS ONE 2014, 9, e86391. [Google Scholar] [CrossRef] [PubMed]
- Adhami, N.; Starck, S. R.; Flores, C.; Martins-Green, M. A health threat to bystanders living in the homes of smokers: How smoke toxins deposited on surfaces can cause insulin resistance. PLoS ONE 2016, 11, e0149510. [Google Scholar]
- Karim, Z.A.; Alshbool, F.Z.; Vemana, H.P.; Adhami, N.; Dhall, S.; Espinosa, E.V.; Martins-Green, M.; Khasawneh, F.T. Third-hand smoke: Impact on hemostasis and thrombogenesis. J. Cardiovasc. Pharmacol. 2015, 66, 177–182. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hang, B.; Wang, P.; Zhao, Y.; Sarker, A.; Chenna, A.; Xia, Y.; Snijders, A.M.; Mao, J.-H. Adverse Health Effects of Thirdhand Smoke: From Cell to Animal Models. Int. J. Mol. Sci. 2017, 18, 932. https://doi.org/10.3390/ijms18050932
Hang B, Wang P, Zhao Y, Sarker A, Chenna A, Xia Y, Snijders AM, Mao J-H. Adverse Health Effects of Thirdhand Smoke: From Cell to Animal Models. International Journal of Molecular Sciences. 2017; 18(5):932. https://doi.org/10.3390/ijms18050932
Chicago/Turabian StyleHang, Bo, Pin Wang, Yue Zhao, Altaf Sarker, Ahmed Chenna, Yankai Xia, Antoine M. Snijders, and Jian-Hua Mao. 2017. "Adverse Health Effects of Thirdhand Smoke: From Cell to Animal Models" International Journal of Molecular Sciences 18, no. 5: 932. https://doi.org/10.3390/ijms18050932
APA StyleHang, B., Wang, P., Zhao, Y., Sarker, A., Chenna, A., Xia, Y., Snijders, A. M., & Mao, J.-H. (2017). Adverse Health Effects of Thirdhand Smoke: From Cell to Animal Models. International Journal of Molecular Sciences, 18(5), 932. https://doi.org/10.3390/ijms18050932







