Antioxidant and Anti-Senescence Effect of Metformin on Mouse Olfactory Ensheathing Cells (mOECs) May Be Associated with Increased Brain-Derived Neurotrophic Factor Levels—An Ex Vivo Study
Abstract
:1. Introduction
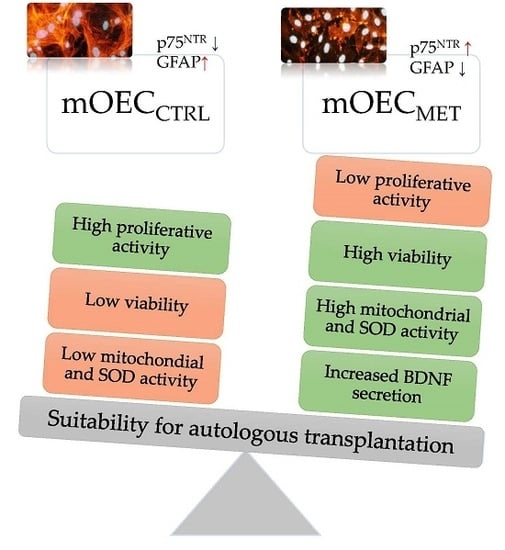
2. Results and Discussion
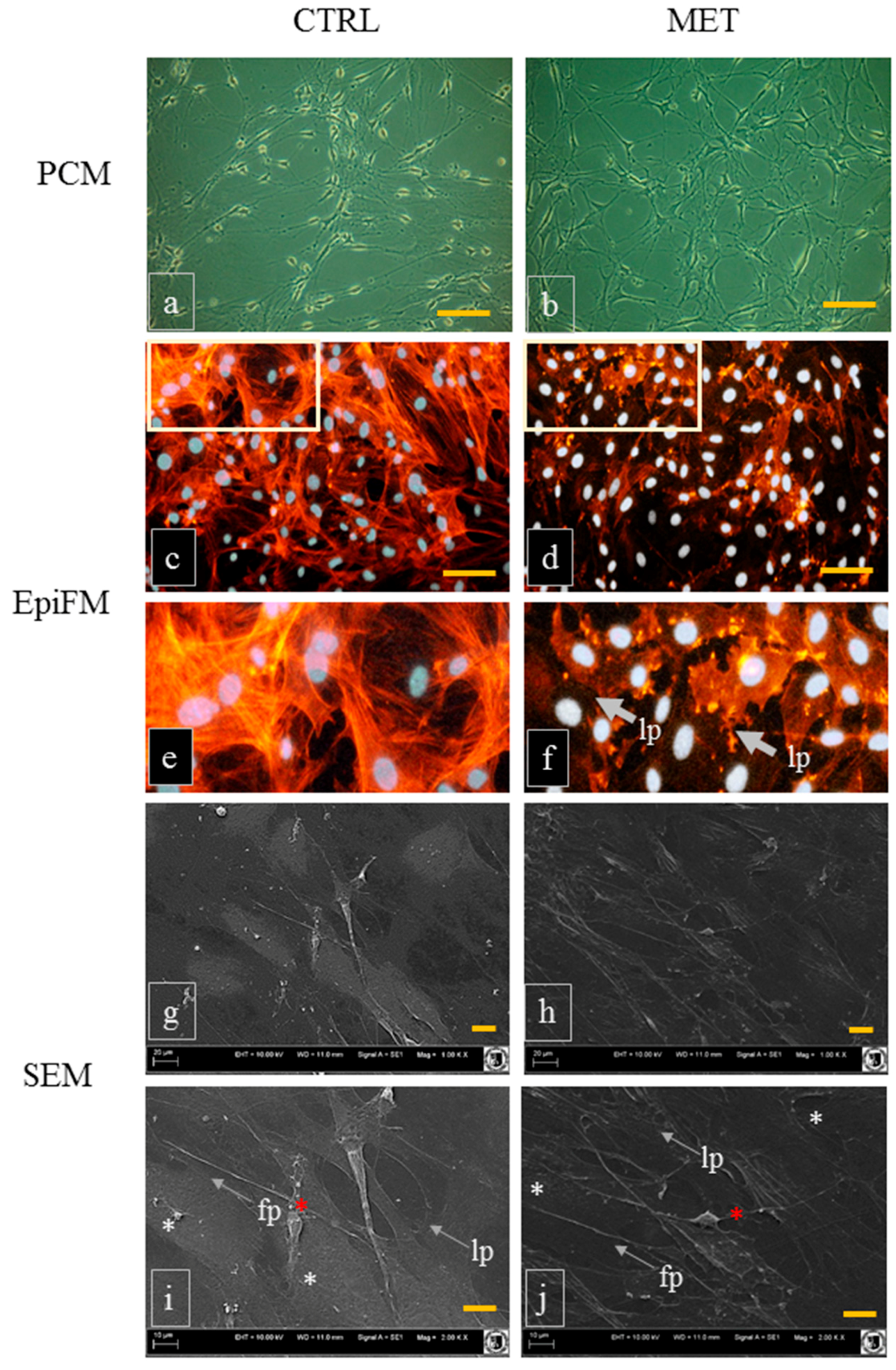
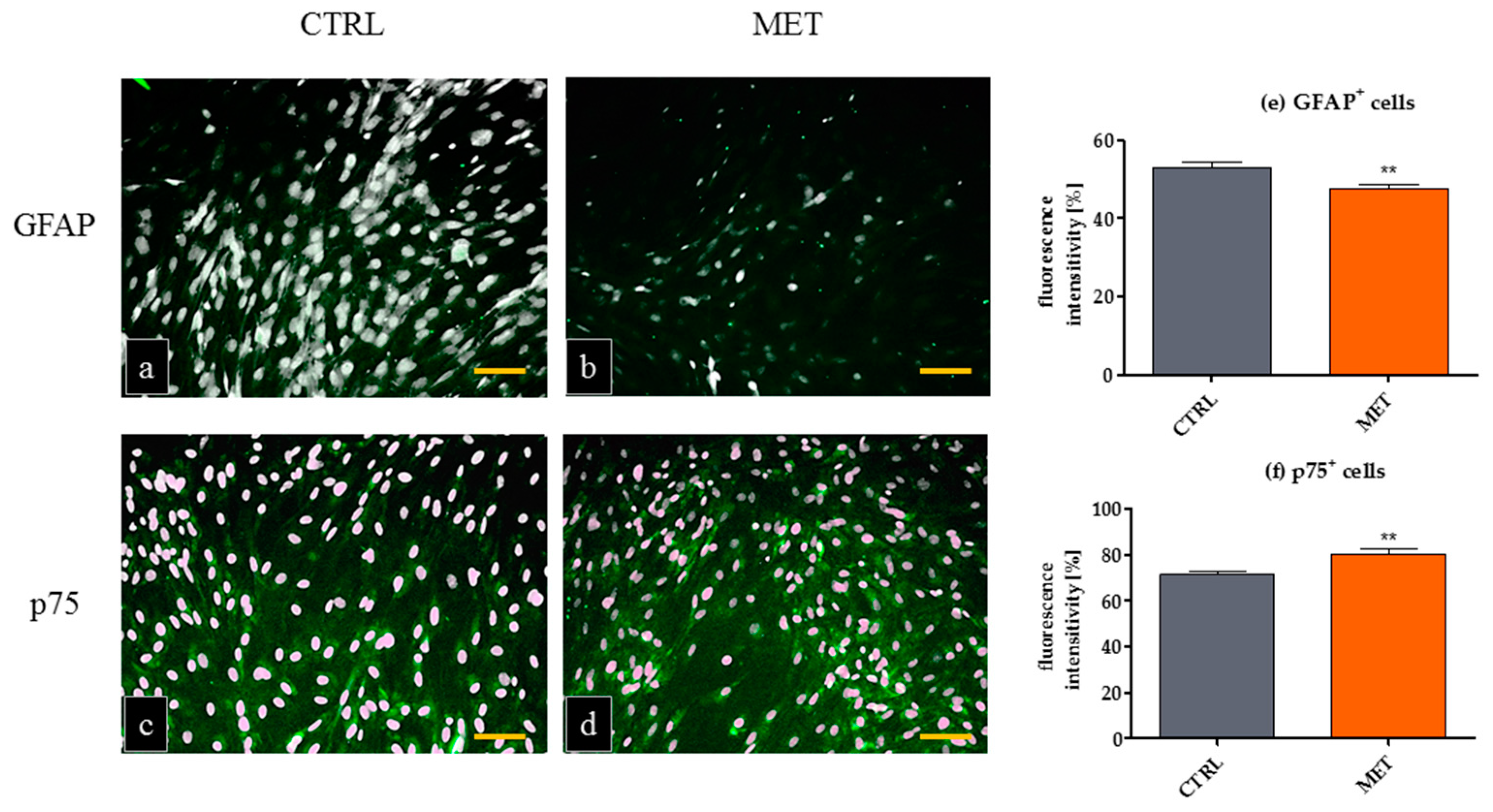
2.1. Cultures of mOECs Derived Ex Vivo from Metformin-Treated and Untreated Animals Show Distinct Morphological and Phenotypical Features
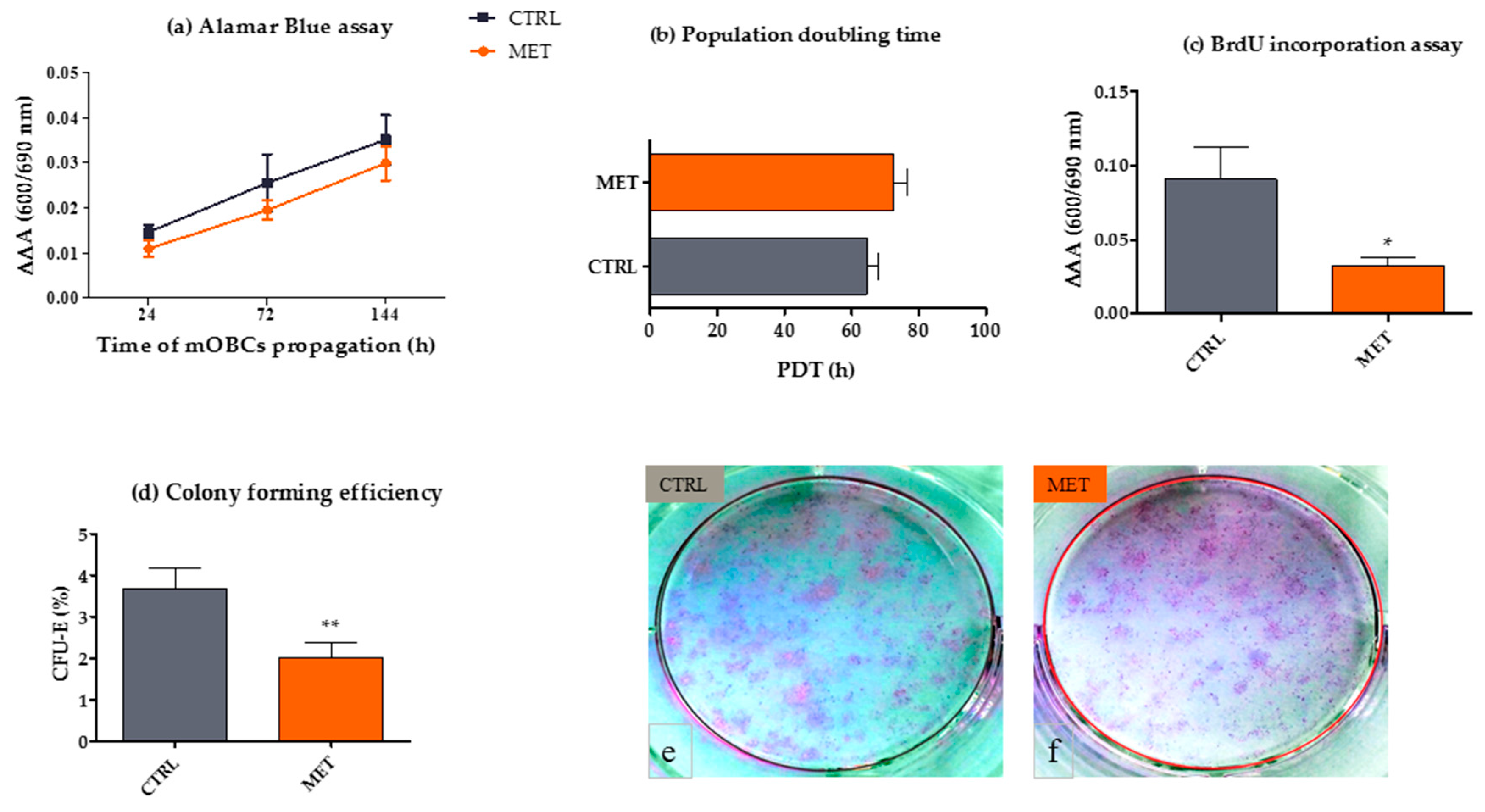
2.2. Metformin Administration in Mice Affects the Proliferative Activity of mOECs Ex Vivo
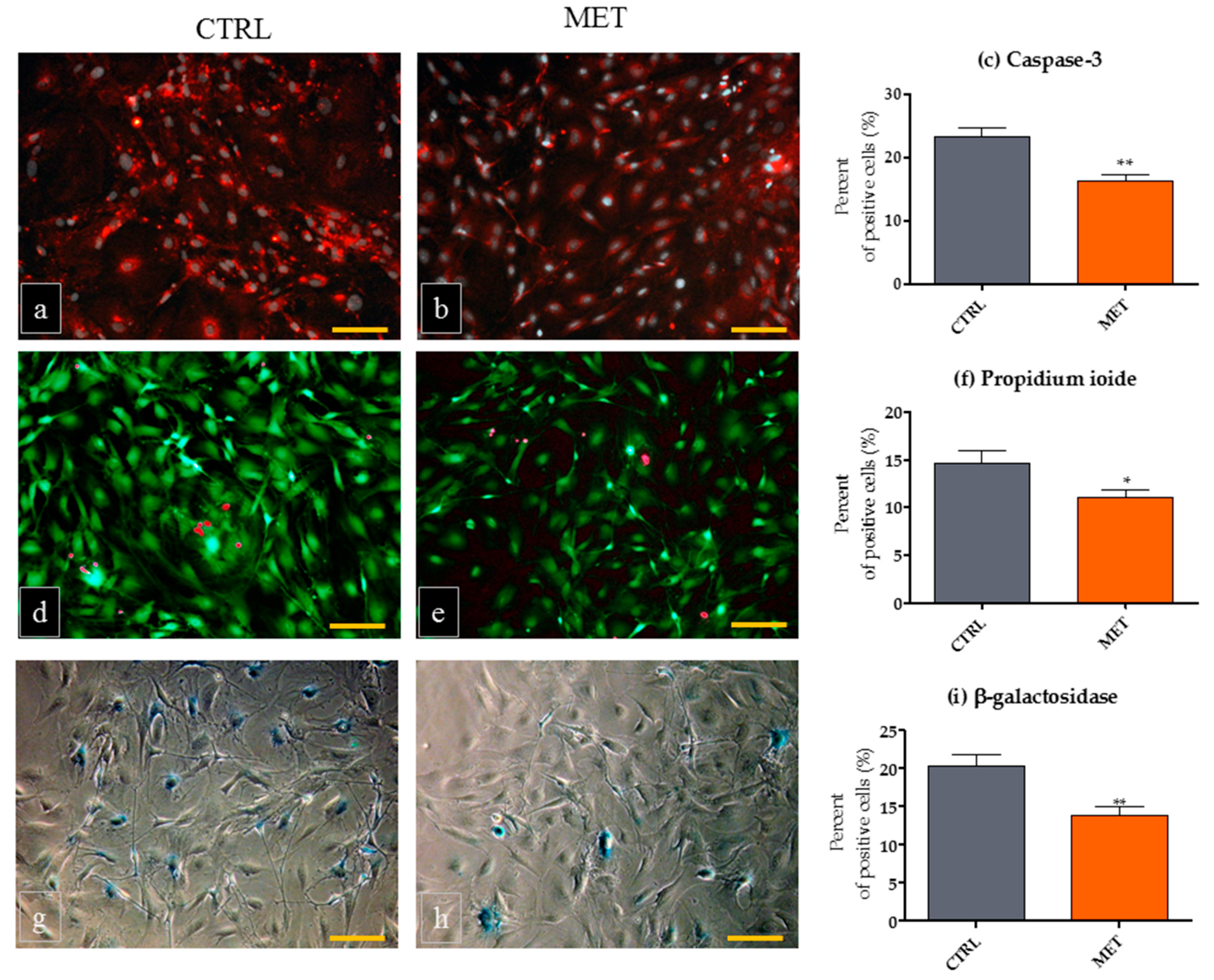
2.3. Metformin Administration May Ameliorate the Viability of mOECs
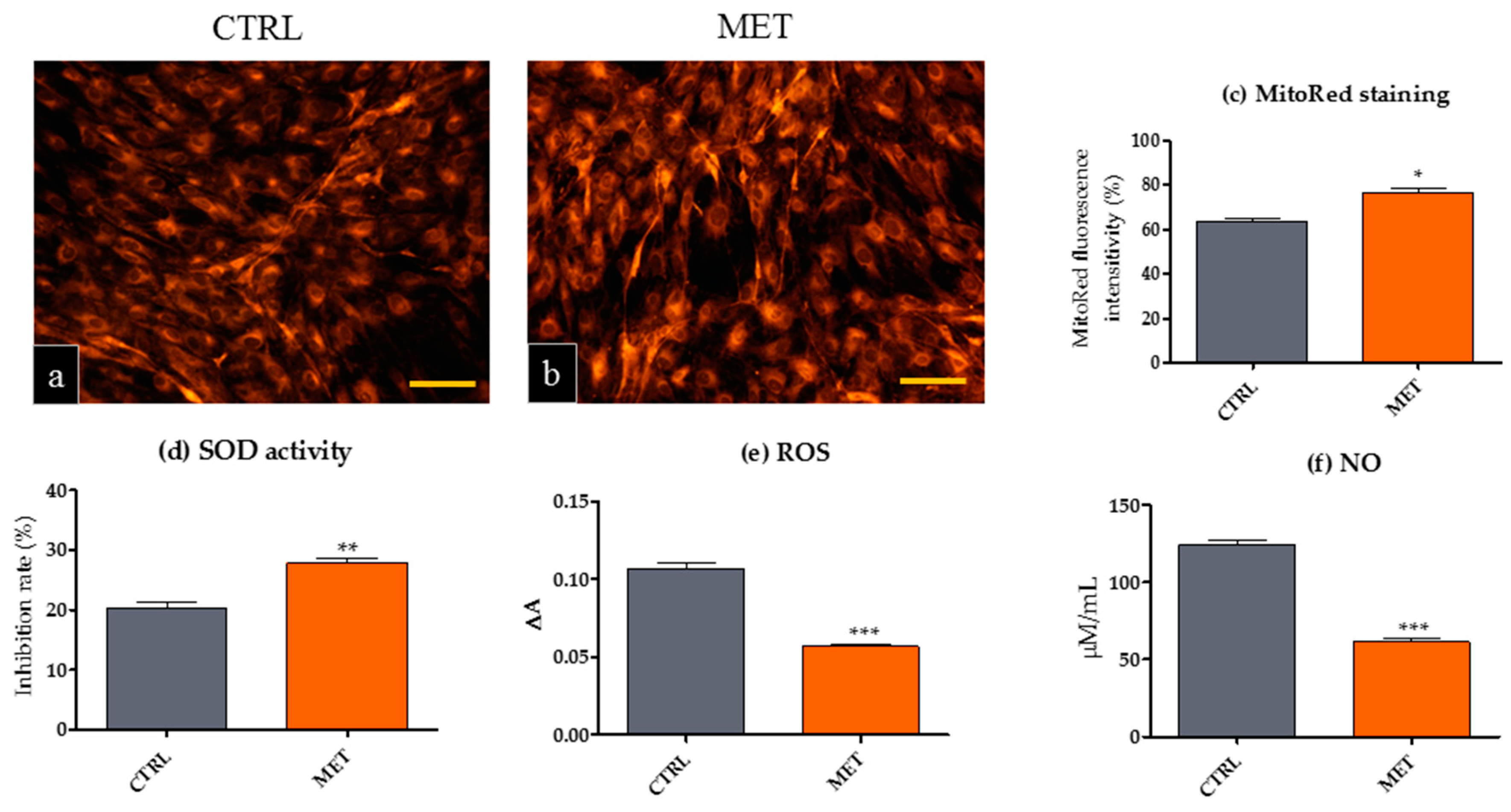
2.4. Metformin Reduces the Expression of Oxidative Stress Markers in mOEC Cultures Derived from Animals Receiving MET
2.5. Metformin Increases Circulating Levels of BDNF and Affects Downstream Genes in the BDNF Pathway
3. Materials and Methods
3.1. Experimental Animals
3.2. Isolation and Culture of Mice Olfactory Ensheathing Glial Cells (mOECs)
3.3. Analysis of mOEC Morphology
3.4. Phenotype of Mice OECs
3.5. Analysis of mOEC Proliferation
3.6. Analysis of mOEC viability
3.7. Visualization of Mitochondria and Determination of Oxidative Stress Factors in mOEC Cultures
3.8. Analysis of mRNA for BDNF and Its Downstream Target Genes
3.9. Determination of BDNF Protein Levels—Secretory Activity of mOECs and Circulating Level of the Protein
3.10. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- He, L.; Wondisford, F.E. Metformin action: Concentrations matter. Cell Metab. 2015, 21, 159–162. [Google Scholar] [CrossRef] [PubMed]
- Hundal, R.S.; Krssak, M.; Dufour, S.; Laurent, D.; Lebon, V.; Chandramouli, V.; Inzucchi, S.E.; Schumann, W.C.; Petersen, K.F.; Landau, B.R.; et al. Mechanism by which metformin reduces glucose production in Type 2 diabetes. Diabetes 2000, 49, 2063–2069. [Google Scholar] [CrossRef] [PubMed]
- Allard, J.S.; Perez, E.J.; Fukui, K.; Carpenter, P.; Ingram, D.K.; de Cabo, R. Prolonged metformin treatment leads to reduced transcription of Nrf2 and neurotrophic factors without cognitive impairment in older C57BL/6J mice. Behav. Brain Res. 2016, 301, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Cho, K.; Chung, J.Y.; Cho, S.K.; Shin, H.-W.; Jang, I.-J.; Park, J.-W.; Yu, K.-S.; Cho, J.-Y. Antihyperglycemic mechanism of metformin occurs via the AMPK/LXRα/POMC pathway. Sci. Rep. 2015, 5, 8145. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Yang, G.; Kim, Y.; Kim, J.; Ha, J. AMPK activators: Mechanisms of action and physiological activities. Exp. Mol. Med. 2016, 48, e224. [Google Scholar] [CrossRef] [PubMed]
- Barzilai, N.; Crandall, J.P.; Kritchevsky, S.B.; Espeland, M.A. Metformin as a tool to target aging. Cell Metab. 2016, 23, 1060–1065. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Gallagher, D.; DeVito, L.M.; Cancino, G.I.; Tsui, D.; He, L.; Keller, G.M.; Frankland, P.W.; Kaplan, D.R.; Miller, F.D. Metformin activates an atypical PKC-CBP pathway to promote neurogenesis and enhance spatial memory formation. Cell Stem Cell 2012, 11, 23–35. [Google Scholar] [CrossRef] [PubMed]
- Potts, M.B.; Lim, D.A. An Old Drug for New Ideas: Metformin promotes adult neurogenesis and spatial memory formation. Cell Stem Cell 2012, 11, 5–6. [Google Scholar] [CrossRef] [PubMed]
- Łabuzek, K.; Liber, S.; Gabryel, B.; Okopień, B. Metformin has adenosine-monophosphate activated protein kinase (AMPK)-independent effects on LPS-stimulated rat primary microglial cultures. Pharmacol. Rep. 2010, 62, 827–848. [Google Scholar] [CrossRef]
- Łabuzek, K.; Suchy, D.; Gabryel, B.; Bielecka, A.; Liber, S.; Okopień, B. Quantification of metformin by the HPLC method in brain regions, cerebrospinal fluid and plasma of rats treated with lipopolysaccharide. Pharmacol. Rep. 2010, 62, 956–965. [Google Scholar] [CrossRef]
- Gupta, A.; Bisht, B.; Dey, C.S. Peripheral insulin-sensitizer drug metformin ameliorates neuronal insulin resistance and Alzheimer’s-like changes. Neuropharmacology 2011, 60, 910–920. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Song, D.; Leng, S.X. Link between type 2 diabetes and Alzheimer’s disease: From epidemiology to mechanism and treatment. Clin. Interv. Aging 2015, 10, 549–560. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Dong, R.R.; Zhong, K.L.; Ghosh, A.; Tang, S.S.; Long, Y.; Hu, M.; Miao, M.X.; Liao, J.M.; Sun, H.B.; et al. Antidiabetic drugs restore abnormal transport of amyloid-β across the blood–brain barrier and memory impairment in db/db mice. Neuropharmacology 2016, 101, 123–136. [Google Scholar] [CrossRef] [PubMed]
- Butterfield, D.A.; Di Domenico, F.; Barone, E. Elevated risk of type 2 diabetes for development of Alzheimer disease: A key role for oxidative stress in brain. Biochim. Biophys. Acta 2014, 1842, 1693–1706. [Google Scholar] [CrossRef] [PubMed]
- Chung, M.-M.; Chen, Y.-L.; Pei, D.; Cheng, Y.-C.; Sun, B.; Nicol, C.J.; Yen, C.-H.; Chen, H.-M.; Liang, Y.-J.; Chiang, M.-C. The neuroprotective role of metformin in advanced glycation end product treated human neural stem cells is AMPK-dependent. Biochim. Biophys. Acta 2015, 1852, 720–731. [Google Scholar] [CrossRef] [PubMed]
- Suski, M.; Olszanecki, R.; Chmura, Ł.; Stachowicz, A.; Madej, J.; Okoń, K.; Adamek, D.; Korbut, R. Influence of metformin on mitochondrial subproteome in the brain of apoE knockout mice. Eur. J. Pharmacol. 2016, 772, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Su, C.; Qiao, C.; Bian, Y.; Ding, J.; Hu, G. Metformin prevents dopaminergic neuron death in MPTP/P-induced mouse model of Parkinson’s disease via autophagy and mitochondrial ROS clearance. Int. J. Neuropsychopharmacol. 2016, 19. [Google Scholar] [CrossRef] [PubMed]
- Davalli, P.; Mitic, T.; Caporali, A.; Lauriola, A.; D’Arca, D. ROS, cell senescence, and novel molecular mechanisms in aging and age-related diseases. Oxid. Med. Cell. Longev. 2016, 2016, e3565127. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Xia, D.; Pan, Z.; Xu, D.; Zhou, Y.; Wu, Y.; Cai, N.; Tang, Q.; Wang, C.; Yan, M.; et al. Metformin protects against apoptosis and senescence in nucleus pulposus cells and ameliorates disc degeneration in vivo. Cell Death Dis. 2016, 7, e2441. [Google Scholar] [CrossRef] [PubMed]
- Golde, T.E.; Miller, V.M. Proteinopathy-induced neuronal senescence: A hypothesis for brain failure in Alzheimer’s and other neurodegenerative diseases. Alzheimers Res. Ther. 2009, 1, 5. [Google Scholar] [CrossRef] [PubMed]
- Chinta, S.J.; Lieu, C.A.; DeMaria, M.; Laberge, R.-M.; Campisi, J.; Andersen, J.K. Environmental stress, ageing and glial cell senescence: A novel mechanistic link to Parkinson’s disease? J. Intern. Med. 2013, 273, 429–436. [Google Scholar] [CrossRef] [PubMed]
- Allen, N.J.; Barres, B.A. Neuroscience: Glia—More than just brain glue. Nature 2009, 457, 675–677. [Google Scholar] [CrossRef] [PubMed]
- Mansour, H.; Chamberlain, C.G.; Weible, M.W.; Hughes, S.; Chu, Y.; Chan-Ling, T. Aging-related changes in astrocytes in the rat retina: Imbalance between cell proliferation and cell death reduces astrocyte availability. Aging Cell 2008, 7, 526–540. [Google Scholar] [CrossRef] [PubMed]
- Higginson, J.R.; Barnett, S.C. The culture of olfactory ensheathing cells (OECs)—A distinct glial cell type. Exp. Neurol. 2011, 229, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Raisman, G.; Barnett, S.C.; Ramón-Cueto, A. Repair of central nervous system lesions by transplantation of olfactory ensheathing cells. Handb. Clin. Neurol. 2012, 109, 541–549. [Google Scholar] [PubMed]
- Wewetzer, K.; Radtke, C.; Kocsis, J.; Baumgärtner, W. Species-specific control of cellular proliferation and the impact of large animal models for the use of olfactory ensheathing cells and Schwann cells in spinal cord repair. Exp. Neurol. 2011, 229, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Féron, F.; Perry, C.; Cochrane, J.; Licina, P.; Nowitzke, A.; Urquhart, S.; Geraghty, T.; Mackay-Sim, A. Autologous olfactory ensheathing cell transplantation in human spinal cord injury. Brain 2005, 128, 2951–2960. [Google Scholar] [CrossRef] [PubMed]
- Tabakow, P.; Jarmundowicz, W.; Czapiga, B.; Fortuna, W.; Miedzybrodzki, R.; Czyz, M.; Huber, J.; Szarek, D.; Okurowski, S.; Szewczyk, P.; et al. Transplantation of autologous olfactory ensheathing cells in complete human spinal cord injury. Cell Transpl. 2013, 22, 1591–1612. [Google Scholar] [CrossRef] [PubMed]
- Moon, J.; Lee, S.-T.; Kong, I.G.; Byun, J.-I.; Sunwoo, J.-S.; Shin, J.-W.; Shim, J.-Y.; Park, J.-H.; Jeon, D.; Jung, K.-H.; et al. Early diagnosis of Alzheimer’s disease from elevated olfactory mucosal miR-206 level. Sci. Rep. 2016, 6, 20364. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Teng, H.-L.; Gao, Y.; Zhang, F.; Ding, Y.-Q.; Huang, Z.-H. Brain-derived neurotrophic factor promotes the migration of olfactory ensheathing cells through TRPC channels. Glia 2016, 64, 2154–2165. [Google Scholar] [CrossRef] [PubMed]
- Boruch, A.V.; Conners, J.J.; Pipitone, M.; Deadwyler, G.; Storer, P.D.; Devries, G.H.; Jones, K.J. Neurotrophic and migratory properties of an olfactory ensheathing cell line. Glia 2001, 33, 225–229. [Google Scholar] [CrossRef]
- Lipson, A.C.; Widenfalk, J.; Lindqvist, E.; Ebendal, T.; Olson, L. Neurotrophic properties of olfactory ensheathing glia. Exp. Neurol. 2003, 180, 167–171. [Google Scholar] [CrossRef]
- Woodhall, E.; West, A.K.; Chuah, M.I. Cultured olfactory ensheathing cells express nerve growth factor, brain-derived neurotrophic factor, glia cell line-derived neurotrophic factor and their receptors. Brain Res. Mol. Brain Res. 2001, 88, 203–213. [Google Scholar] [CrossRef]
- Imfeld, P.; Bodmer, M.; Jick, S.S.; Meier, C.R. Metformin, other antidiabetic drugs, and risk of Alzheimer’s disease: A population-based case-control study. J. Am. Geriatr. Soc. 2012, 60, 916–921. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhou, K.; Wang, R.; Liu, Y.; Kwak, Y.D.; Ma, T.; Thompson, R.C.; Zhao, Y.; Smith, L.; Gasparini, L.; et al. Antidiabetic drug metformin (GlucophageR) increases biogenesis of Alzheimer’s amyloid peptides via up-regulating BACE1 transcription. Proc. Natl. Acad. Sci. USA 2009, 106, 3907–3912. [Google Scholar] [CrossRef] [PubMed]
- Barnett, S.C.; Riddell, J.S. Olfactory ensheathing cells (OECs) and the treatment of CNS injury: Advantages and possible caveats. J. Anat. 2004, 204, 57–67. [Google Scholar] [CrossRef] [PubMed]
- Windus, L.C.E.; Lineburg, K.E.; Scott, S.E.; Claxton, C.; Mackay-Sim, A.; Key, B.; John, J.A.S. Lamellipodia mediate the heterogeneity of central olfactory ensheathing cell interactions. Cell. Mol. Life Sci. 2010, 67, 1735–1750. [Google Scholar] [CrossRef] [PubMed]
- Windus, L.C.E.; Claxton, C.; Allen, C.L.; Key, B.; St John, J.A. Motile membrane protrusions regulate cell–cell adhesion and migration of olfactory ensheathing glia. Glia 2007, 55, 1708–1719. [Google Scholar] [CrossRef] [PubMed]
- Ekberg, J.A.K.; St John, J.A. Crucial Roles for Olfactory Ensheathing Cells and Olfactory Mucosal Cells in the Repair of Damaged Neural Tracts. Anat. Rec. 2014, 297, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Cragnolini, A.B.; Friedman, W.J. The function of p75NTR in glia. Trends Neurosci. 2008, 31, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Vilar, M.; Murillo-Carretero, M.; Mira, H.; Magnusson, K.; Besset, V.; Ibáñez, C.F. Bex1, a novel interactor of the p75 neurotrophin receptor, links neurotrophin signaling to the cell cycle. EMBO J. 2006, 25, 1219–1230. [Google Scholar] [CrossRef] [PubMed]
- Kawaja, M.D.; Boyd, J.G.; Smithson, L.J.; Jahed, A.; Doucette, R. Technical strategies to isolate olfactory ensheathing cells for intraspinal implantation. J. Neurotrauma 2009, 26, 155–177. [Google Scholar] [CrossRef] [PubMed]
- Tetzlaff, W.; Okon, E.B.; Karimi-Abdolrezaee, S.; Hill, C.E.; Sparling, J.S.; Plemel, J.R.; Plunet, W.T.; Tsai, E.C.; Baptiste, D.; Smithson, L.J.; et al. A systematic review of cellular transplantation therapies for spinal cord injury. J. Neurotrauma 2011, 28, 1611–1682. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Bunge, M.B.; Wood, P.M.; Plant, G.W. Mitogenic response of adult rat olfactory ensheathing glia to four growth factors. Glia 2001, 33, 334–342. [Google Scholar] [CrossRef]
- Techangamsuwan, S.; Imbschweiler, I.; Kreutzer, R.; Kreutze, R.M.; Baumgärtner, W.; Wewetzer, K. Similar behaviour and primate-like properties of adult canine Schwann cells and olfactory ensheathing cells in long-term culture. Brain Res. 2008, 1240, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Hannila, S.S.; Filbin, M.T. The role of cyclic AMP signaling in promoting axonal regeneration after spinal cord injury. Exp. Neurol. 2008, 209, 321–332. [Google Scholar] [CrossRef] [PubMed]
- Miller, R.A.; Chu, Q.; Xie, J.; Foretz, M.; Viollet, B.; Birnbaum, M.J. Biguanides suppress hepatic glucagon signalling by decreasing production of cyclic AMP. Nature 2013, 494, 256–260. [Google Scholar] [CrossRef] [PubMed]
- Au, E.; Roskams, A.J. Olfactory ensheathing cells of the lamina propria in vivo and in vitro. Glia 2003, 41, 224–236. [Google Scholar] [CrossRef] [PubMed]
- Walton, R.M.; Wolfe, J.H. In vivo growth and differentiation of canine olfactory bulb-derived neural progenitor cells under variable culture conditions. J. Neurosci. Methods 2008, 169, 158–167. [Google Scholar] [CrossRef] [PubMed]
- Moiseeva, O.; Deschênes-Simard, X.; St-Germain, E.; Igelmann, S.; Huot, G.; Cadar, A.E.; Bourdeau, V.; Pollak, M.N.; Ferbeyre, G. Metformin inhibits the senescence-associated secretory phenotype by interfering with IKK/NF-κB activation. Aging Cell 2013, 12, 489–498. [Google Scholar] [CrossRef] [PubMed]
- Algire, C.; Moiseeva, O.; Deschênes-Simard, X.; Amrein, L.; Petruccelli, L.; Birman, E.; Viollet, B.; Ferbeyre, G.; Pollak, M.N. Metformin reduces endogenous reactive oxygen species and associated DNA Damage. Cancer Prev. Res. 2012, 5, 536–543. [Google Scholar] [CrossRef] [PubMed]
- Marycz, K.; Tomaszewski, K.A.; Kornicka, K.; Henry, B.M.; Wroński, S.; Tarasiuk, J.; Maredziak, M. Metformin decreases reactive oxygen species, enhances osteogenic properties of adipose-derived multipotent mesenchymal stem cells in vitro, and increases bone density in vivo. Oxid. Med. Cell. Longev. 2016, 2016, e9785890. [Google Scholar] [CrossRef] [PubMed]
- Hou, X.; Song, J.; Li, X.-N.; Zhang, L.; Wang, X.; Chen, L.; Shen, Y.H. Metformin reduces intracellular reactive oxygen species levels by upregulating expression of the antioxidant thioredoxin via the AMPK-FOXO3 pathway. Biochem. Biophys. Res. Commun. 2010, 396, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Le Belle, J.E.; Orozco, N.M.; Paucar, A.A.; Saxe, J.P.; Mottahedeh, J.; Pyle, A.D.; Wu, H.; Kornblum, H.I. Proliferative neural stem cells have high endogenous ROS levels that regulate self-renewal and neurogenesis in a PI3K/Akt-dependant manner. Cell Stem Cell 2011, 8, 59–71. [Google Scholar] [CrossRef] [PubMed]
- Patil, S.P.; Jain, P.D.; Ghumatkar, P.J.; Tambe, R.; Sathaye, S. Neuroprotective effect of metformin in MPTP-induced Parkinson’s disease in mice. Neuroscience 2014, 277, 747–754. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, M.; Hains, B.C.; Lankford, K.L.; Waxman, S.G.; Kocsis, J.D. Protection of corticospinal tract neurons after dorsal spinal cord transection and engraftment of olfactory ensheathing cells. Glia 2006, 53, 352–359. [Google Scholar] [CrossRef] [PubMed]
- Skaper, S.D. The neurotrophin family of neurotrophic factors: An overview. Methods Mol. Biol. 2012, 846, 1–12. [Google Scholar] [PubMed]
- Bathina, S.; Das, U.N. Brain-derived neurotrophic factor and its clinical implications. Arch. Med. Sci. 2015, 11, 1164–1178. [Google Scholar] [CrossRef] [PubMed]
- Vaillant, A.R.; Mazzoni, I.; Tudan, C.; Boudreau, M.; Kaplan, D.R.; Miller, F.D. Depolarization and neurotrophins converge on the phosphatidylinositol 3-kinase–akt pathway to synergistically regulate neuronal survival. J. Cell Biol. 1999, 146, 955–966. [Google Scholar] [CrossRef] [PubMed]
- Brunet, A.; Bonni, A.; Zigmond, M.J.; Lin, M.Z.; Juo, P.; Hu, L.S.; Anderson, M.J.; Arden, K.C.; Blenis, J.; Greenberg, M.E. Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell 1999, 96, 857–868. [Google Scholar] [CrossRef]
- Fatt, M.; Hsu, K.; He, L.; Wondisford, F.; Miller, F.D.; Kaplan, D.R.; Wang, J. Metformin acts on two different molecular pathways to enhance adult neural precursor proliferation/self-renewal and differentiation. Stem Cell Rep. 2015, 5, 988–995. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.; Naia, L.; Dominguez, A.; Ribeiro, M.; Rodrigues, J.; Vieira, O.V.; Lessmann, V.; Rego, A.C. Overexpression of BDNF and full-length TrkB receptor ameliorate striatal neural survival in Huntington’s Disease. Neurodegener. Dis. 2015, 15, 207–218. [Google Scholar] [CrossRef] [PubMed]
- Śmieszek, A.; Basińska, K.; Chrząstek, K.; Marycz, K. In vitro and in vivo effects of metformin on osteopontin expression in mice adipose-derived multipotent stromal cells and adipose tissue. J. Diabetes Res. 2015, 2015, e814896. [Google Scholar] [CrossRef] [PubMed]
- Grzesiak, J.; Fryczkowski, R.; Lis, A.; Szarek, D.; Laska, J.; Marycz, K. Characterization of olfactory ensheathing glial cells cultured on polyurethane/polylactide electrospun nonwovens. Int. J. Polym. Sci. 2015, 2015, e908328. [Google Scholar] [CrossRef]
- Śmieszek, A.; Czyrek, A.; Basinska, K.; Trynda, J.; Skaradzińska, A.; Siudzińska, A.; Marędziak, M.; Marycz, K. Effect of metformin on viability, morphology, and ultrastructure of mouse bone marrow-derived multipotent mesenchymal stromal cells and Balb/3T3 embryonic fibroblast cell line. BioMed Res. Int. 2015, 2015, e769402. [Google Scholar] [CrossRef] [PubMed]
- Grzesiak, J.; Marycz, K.; Szarek, D.; Bednarz, P.; Laska, J. Polyurethane/polylactide-based biomaterials combined with rat olfactory bulb-derived glial cells and adipose-derived mesenchymal stromal cells for neural regenerative medicine applications. Mater. Sci. Eng. C 2015, 52, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Marycz, K.; Śmieszek, A.; Grzesiak, J.; Siudzińska, A.; Marędziak, M.; Donesz-Sikorska, A.; Krzak, J. The Osteogenic properties of multipotent mesenchymal stromal cells in cultures on TiO2 sol-gel-derived biomaterial. BioMed Res. Int. 2015, 2015, e651097. [Google Scholar] [CrossRef] [PubMed]
- Marędziak, M.; Marycz, K.; Lewandowski, D.; Siudzińska, A.; Śmieszek, A. Static magnetic field enhances synthesis and secretion of membrane-derived microvesicles (MVs) rich in VEGF and BMP-2 in equine adipose-derived stromal cells (EqASCs)—A new approach in veterinary regenerative medicine. In Vitro Cell. Dev. Biol. Anim. 2015, 51, 230–240. [Google Scholar] [CrossRef] [PubMed]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An open-source platform for biological-image analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef] [PubMed]
- Jensen, E.C. Quantitative analysis of histological staining and fluorescence using ImageJ. Anat. Rec. 2013, 296, 378–381. [Google Scholar] [CrossRef] [PubMed]
- Kornicka, K.; Marycz, K.; Tomaszewski, K.A.; Marędziak, M.; Śmieszek, A. The effect of age on osteogenic and adipogenic differentiation potential of human adipose derived stromal stem cells (hASCs) and the impact of stress factors in the course of the differentiation process. Oxid. Med. Cell. Longev. 2015, 2015, e309169. [Google Scholar] [CrossRef] [PubMed]
- Heuer, G.G.; Skorupa, A.F.; Prasad Alur, R.K.; Jiang, K.; Wolfe, J.H. Accumulation of abnormal amounts of glycosaminoglycans in murine mucopolysaccharidosis type VII neural progenitor cells does not alter the growth rate or efficiency of differentiation into neurons. Mol. Cell. Neurosci. 2001, 17, 167–178. [Google Scholar] [CrossRef] [PubMed]
- Roth, V. 2006 Doubling Time Computing. Available online: http://www.doubling-time.com/compute.php (accessed on 27 November 2016).
- Chomczynski, P.; Sacchi, N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal. Biochem. 1987, 162, 156–159. [Google Scholar] [CrossRef]


| Gene | Abbreviation | Primer | Sequence 5′–3′ | Loci | Aplicon Lenght (bp) | Accesion No. |
|---|---|---|---|---|---|---|
| Brain derived neurotrophic factor | BDNF | F | GCCGCAAACATGTCTATGAGGGTT | 670–693 | 174 | NM_001316310.1 |
| R | TTGGCCTTTGGATACCGGGACTTT | 843–820 | ||||
| Tropomyosin receptor kinase B | TrkB/NTRK2 | F | GCGAACCTGCAGATACCCAAT | 1306–1326 | 148 | XM_006517152.2 |
| R | CCAAATTCCCAACGTCCCA | 1453–1435 | ||||
| B cell leukemia/lymphoma 2 | Bcl-2 | F | ATCGCCCTGTGGATGACTGAG | 1918–1938 | 129 | NM_009741.5 |
| R | CAGCCAGGAGAAATCAAACAGAGG | 2046–2023 | ||||
| Bcl-2-associated death promoter | Bad | F | ACATTCATCAGCAGGGACGG | 199–218 | 115 | NM_001285453.1 |
| R | ATCCCTTCATCCTCCTCGGT | 313–294 | ||||
| Bcl-2-associated X protein | Bax | F | TGCTAGCAAACTGGTGCTCA | 476–495 | 113 | XM_011250780.1 |
| R | CTTGGATCCAGACAAGCAGC | 588–569 | ||||
| RAC-γ serine/threonine-protein kinase | Akt3 | F | ATCCCCTCAACAACTTCTCAGT | 450–471 | 156 | XM_011238805.1 |
| R | CTTCCGTCCACTCTTCTCTTTC | 605–584 | ||||
| Phosphatidylinositol-4,5-bisphosphate 3-kinase | PI3K | F | CTCTCCTGTGCTGGCTACTGT | 2932–2952 | 157 | XM_006536015.2 |
| R | GCTCTCGGTTGATTCCAAACT | 3088–3068 | ||||
| β-actin | ACTB | F | CCTGAGGCTCTTTTCCAGCC | 881–900 | 110 | NM_007393.5 |
| R | TAGAGGTCTTTACGGATGTCAACGT | 990–966 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Śmieszek, A.; Stręk, Z.; Kornicka, K.; Grzesiak, J.; Weiss, C.; Marycz, K. Antioxidant and Anti-Senescence Effect of Metformin on Mouse Olfactory Ensheathing Cells (mOECs) May Be Associated with Increased Brain-Derived Neurotrophic Factor Levels—An Ex Vivo Study. Int. J. Mol. Sci. 2017, 18, 872. https://doi.org/10.3390/ijms18040872
Śmieszek A, Stręk Z, Kornicka K, Grzesiak J, Weiss C, Marycz K. Antioxidant and Anti-Senescence Effect of Metformin on Mouse Olfactory Ensheathing Cells (mOECs) May Be Associated with Increased Brain-Derived Neurotrophic Factor Levels—An Ex Vivo Study. International Journal of Molecular Sciences. 2017; 18(4):872. https://doi.org/10.3390/ijms18040872
Chicago/Turabian StyleŚmieszek, Agnieszka, Zuzanna Stręk, Katarzyna Kornicka, Jakub Grzesiak, Christine Weiss, and Krzysztof Marycz. 2017. "Antioxidant and Anti-Senescence Effect of Metformin on Mouse Olfactory Ensheathing Cells (mOECs) May Be Associated with Increased Brain-Derived Neurotrophic Factor Levels—An Ex Vivo Study" International Journal of Molecular Sciences 18, no. 4: 872. https://doi.org/10.3390/ijms18040872
APA StyleŚmieszek, A., Stręk, Z., Kornicka, K., Grzesiak, J., Weiss, C., & Marycz, K. (2017). Antioxidant and Anti-Senescence Effect of Metformin on Mouse Olfactory Ensheathing Cells (mOECs) May Be Associated with Increased Brain-Derived Neurotrophic Factor Levels—An Ex Vivo Study. International Journal of Molecular Sciences, 18(4), 872. https://doi.org/10.3390/ijms18040872