Melatonin: A Review of Its Potential Functions and Effects on Dental Diseases
Abstract
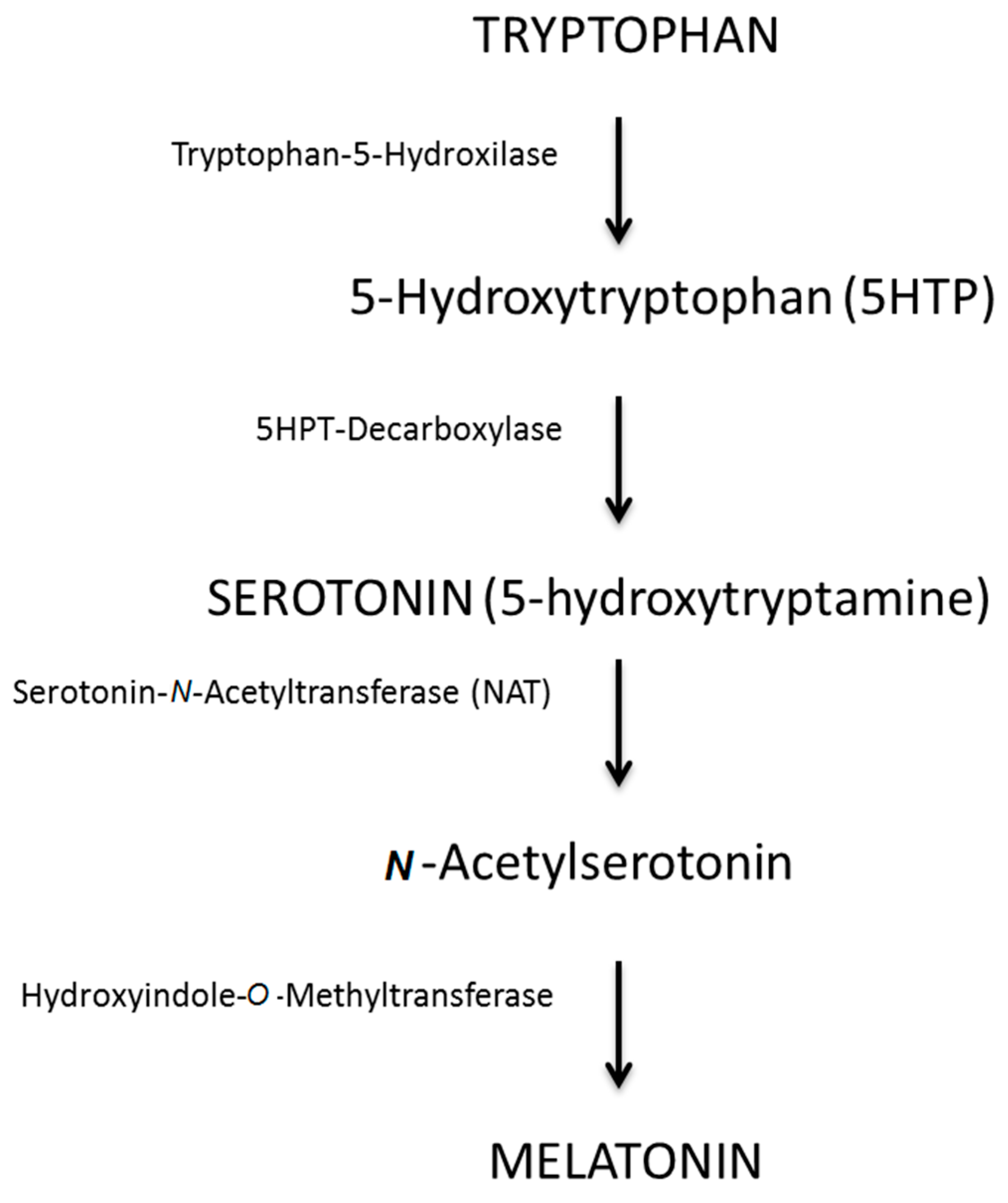
:1. Introduction
2. Material and Methods
- melatonin
- melatonin and oral cavity
- melatonin and implant
- melatonin and periodontal disease
- melatonin and cancer
- melatonin and microorganisms, bacteria or virus
- Main effects of melatonin related to the oral cavity;
- Melatonin and dental implants;
- Melatonin and periodontal disease;
- Other effects of melatonin on the oral cavity.
3. Results
3.1. Main Effects of Melatonin Related to the Oral Cavity
3.2. Melatonin and Dental Implants
3.3. Melatonin and Periodontal Disease
3.4. Other Effects of Melatonin on the Oral Cavity
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hardeland, R.; Pandi-Perumal, S.R.; Cardinali, D.P. Melatonin. Int. J. Biochem. Cell Biol. 2006, 38, 313–316. [Google Scholar] [CrossRef] [PubMed]
- Cutando, A.; Gómez-Moreno, G.; Arana, C.; Muñoz, F.; Lopez-Peña, M.; Stephenson, J.; Reiter, R.J. Melatonin stimulates osteointegration of dental implants. J. Pineal Res. 2008, 45, 174–179. [Google Scholar] [CrossRef] [PubMed]
- Girgert, R.; Hanf, V.; Emons, G.; Gründker, C. Membrane-bound melatonin receptor MT1 down-regulates estrogen responsive genes in breast cancer cells. J. Pineal Res. 2009, 47, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Acuña-Castroviejo, D.; Escames, G.; Macías, M.; Muñoz Hoyos, A.; Molina Carballo, A.; Arauzo, M.; Montes, R. Cell protective role of melatonin in the brain. J. Pineal Res. 1995, 19, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Manchester, L.C.; Reiter, R.J.; Qi, W.B.; Zhang, M.; Weintraub, S.T.; Cabrera, J.; Sainz, R.M.; Mayo, J.C. Identification of highly elevated levels of melatonin in bone marrow: Its origin and significance. Biochim. Biophys. Acta 1999, 1472, 206–214. [Google Scholar] [CrossRef]
- Antón-Tay, F.; Ramírez, G.; Martínez, I.; Benítez-King, G. In vitro stimulation of protein kinase C by melatonin. Neurochem. Res. 1998, 23, 601–606. [Google Scholar] [CrossRef] [PubMed]
- Huerto-Delgadillo, L.; Anton-Tay, F.; Benitz-King, G. Effects of melatonin on microtubule assembly depend on hormone concentration: Role of melatonin as a calmodulin antagonist. J. Pineal Res. 1992, 13, 55–62. [Google Scholar] [CrossRef]
- Macías, M.; Escames, G.; Leon, J.; Coto, A.; Sbihi, Y.; Osuna, A.; Acuña-Castroviejo, D. Calreticulin-melatonin. An unexpected relationship. Eur. J. Biochem. 2003, 270, 832–840. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Manchester, L.C.; Hardeland, R.; López-Burillo, S.; Mayo, J.C.; Sainz, R.M.; Reiter, R.J. Melatonin: A hormone, a tissue factor, an autocoid, a paracoid, and an antioxidant vitamin. J. Pineal Res. 2003, 34, 75–78. [Google Scholar] [CrossRef] [PubMed]
- Axelrod, J. The pineal gland: A neurochemical transducer. Science 1974, 184, 1341–1348. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J. Pineal melatonin: Cell biology of its synthesis and of its physiological interactions. Endocr. Rev. 1991, 12, 151–180. [Google Scholar] [CrossRef] [PubMed]
- Simonneaux, V.; Ribelayga, C. Generation of the melatonin endocrine message in mammals: A review of the complex regulation of melatonin synthesis by norepinephrine, peptides, and other pineal transmitters. Pharmacol. Rev. 2003, 55, 325–395. [Google Scholar] [CrossRef] [PubMed]
- Laakso, M.L.; Porkka-Heiskanen, T.; Alila, A.; Stenberg, D.; Johansson, G. Correlation between salivary and serum melatonin: Dependence on serum melatonin levels. J. Pineal Res. 1990, 9, 39–50. [Google Scholar] [CrossRef] [PubMed]
- Okatani, Y.; Okamoto, K.; Hayashi, K.; Wakatsuki, A.; Sagara, Y. Maternal-fetal transfer of melatonin in human pregnancy near term. J. Pineal Res. 1998, 25, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Redman, J.; Armstrong, S.; Ng, K.T. Free-running activity rhythms in the rat: Entrainment by melatonin. Science 1983, 219, 1089–1091. [Google Scholar] [CrossRef] [PubMed]
- McArthur, A.J.; Hunt, A.E.; Gillette, M.U. Melatonin action and signal transduction in the rat suprachiasmatic circadian clock: Activation of protein kinase C at dusk and dawn. Endocrinology 1997, 138, 627–634. [Google Scholar] [CrossRef] [PubMed]
- Dollins, A.B.; Zhdanova, I.V.; Wurtman, R.J.; Lynch, H.J.; Deng, M.H. Effect of inducing nocturnal serum melatonin concentrations in daytime on sleep, mood, body temperature, and performance. Proc. Natl. Acad. Sci. USA 1994, 91, 1824–1828. [Google Scholar] [CrossRef] [PubMed]
- Esquifino, A.I.; Villanúa, M.A.; Agrasal, C. Effect of neonatal melatonin administration on sexual development in the rat. J. Steroid Biochem. 1987, 27, 1089–1093. [Google Scholar] [CrossRef]
- Kennaway, D.J.; Rowe, S.A. Melatonin binding sites and their role in seasonal reproduction. J. Reprod. Fertil. Suppl. 1995, 49, 423–435. [Google Scholar] [PubMed]
- García-Mauriño, S.; Pozo, D.; Calvo, J.R.; Guerrero, J.M. Correlation between nuclear melatonin receptor expression and enhanced cytokine production in human lymphocytic and monocytic cell lines. J. Pineal Res. 2000, 29, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Guerrero, J.M.; Reiter, R.J. Melatonin-immune system relationships. Curr. Top. Med. Chem. 2002, 2, 167–179. [Google Scholar] [CrossRef] [PubMed]
- Tresguerres, I.F.; Clemente, C.; Donado, M.; Gómez-Pellico, L.; Blanco, L.; Alobera, M.A.; Tresguerres, J.A. Local administration of growth hormone enhances periimplant bone reaction in an osteoporotic rabbit model. Clin. Oral Implants Res. 2002, 13, 631–636. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, N.; Somei, M.; Seki, A.; Reiter, R.J.; Hattori, A. Novel bromomelatonin derivatives as potentially effective drugs to treat bone diseases. J. Pineal Res. 2008, 45, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Dröge, W. Free radicals in the physiological control of cell function. Physiol. Rev. 2002, 82, 47–95. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Melchiorri, D.; Sewerynek, E.; Poeggeler, B.; Barlow-Walden, L.; Chuang, J.; Ortiz, G.G.; Acuña-Castroviejo, D. A review of the evidence supporting melatonin's role as an antioxidant. J. Pineal Res. 1995, 18, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Manchester, L.C.; Reiter, R.J.; Plummer, B.F.; Limson, J.; Weintraub, S.T.; Qi, W. Melatonin directly scavenges hydrogen peroxide: A potentially new metabolic pathway of melatonin biotransformation. Free Radic. Biol. Med. 2000, 29, 1177–1185. [Google Scholar] [CrossRef]
- Garrett, I.R.; Boyce, B.F.; Oreffo, R.O.; Bonewald, L.; Poser, J.; Mundy, G.R. Oxygen-derived free radicals stimulate osteoclastic bone resorption in rodent bone in vitro and in vivo. J. Clin. Investig. 1990, 85, 632–639. [Google Scholar] [CrossRef] [PubMed]
- Koyama, H.; Nakade, O.; Takada, Y.; Kaku, T.; Lau, K.H. Melatonin at pharmacologic doses increases bone mass by suppressing resorption through down-regulation of the RANKL-mediated osteoclast formation and activation. J. Bone Miner. Res. 2002, 17, 1219–1229. [Google Scholar] [CrossRef] [PubMed]
- Roth, J.A.; Kim, B.G.; Lin, W.L.; Cho, M.I. Melatonin promotes osteoblast differentiation and bone formation. J. Biol. Chem. 1999, 274, 22041–22047. [Google Scholar] [CrossRef] [PubMed]
- Satomura, K.; Tobiume, S.; Tokuyama, R.; Yamasaki, Y.; Kudoh, K.; Maeda, E.; Nagayama, M. Melatonin at pharmacological doses enhances human osteoblastic differentiation in vitro and promotes mouse cortical bone formation in vivo. J. Pineal Res. 2007, 42, 31–239. [Google Scholar] [CrossRef] [PubMed]
- Sethi, S.; Radio, N.M.; Kotlarczyk, M.P.; Chen, C.T.; Wei, Y.H.; Jockers, R.; Witt-Enderby, P.A. Determination of the minimal melatonin exposure required to induce osteoblast differentiation from human mesenchymal stem cells and these effects on downstream signaling pathways. J. Pineal Res. 2010, 49, 222–238. [Google Scholar] [CrossRef] [PubMed]
- Nakade, O.; Koyama, H.; Ariji, H.; Yajima, A.; Kaku, T. Melatonin stimulates proliferation and type I collagen synthesis in human bone cells in vitro. J. Pineal Res. 1999, 27, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Acuña-Castroviejo, D.; Reiter, R.J.; Menéndez-Peláez, A.; Pablos, M.I.; Burgos, A. Characterization of high-affinity melatonin binding sites in purified cell nuclei of rat liver. J. Pineal Res. 1994, 16, 100–112. [Google Scholar] [CrossRef] [PubMed]
- López-Martínez, F.; Olivares Ponce, P.N.; Guerra Rodríguez, M.; Martínez Pedraza, R. Melatonin: Bone metabolism in oral cavity. Int. J. Dent. 2012, 2012, 628406. [Google Scholar] [CrossRef] [PubMed]
- Witt-Enderby, P.A.; Radio, N.M.; Doctor, J.S.; Davis, V.L. Therapeutic treatments potentially mediated by melatonin receptors: Potential clinical uses in the prevention of osteoporosis, cancer and as an adjuvant therapy. J. Pineal Res. 2006, 41, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Fernández, M.P.; Calvo-Guirado, J.L.; de-Val, J.E.; Delgado-Ruiz, R.A.; Negri, B.; Pardo-Zamora, G.; Peñarrocha, D.; Barona, C.; Granero, J.M.; Alcaraz-Baños, M. Melatonin promotes angiogenesis during repair of bone defects: A radiological and histomorphometric study in rabbit tibiae. Clin. Oral Investig. 2013, 17, 147–158. [Google Scholar] [CrossRef] [PubMed]
- Pugazhenthi, K.; Kapoor, M.; Clarkson, A.N.; Hall, I.; Appleton, I. Melatonin accelerates the process of wound repair in full-thickness incisional wounds. J. Pineal Res. 2008, 44, 387–396. [Google Scholar] [CrossRef] [PubMed]
- Soybir, G.; Topuzlu, C.; Odabaş, O.; Dolay, K.; Bilir, A.; Köksoy, F. The effects of melatonin on angiogenesis and wound healing. Surg. Today 2003, 33, 896–901. [Google Scholar] [CrossRef] [PubMed]
- Franchi, M.; Fini, M.; Martini, D.; Orsini, E.; Leonardi, L.; Ruggeri, A.; Giavaresi, G.; Ottani, V. Biological fixation of endosseous implants. Micron 2005, 36, 665–671. [Google Scholar] [CrossRef] [PubMed]
- Takechi, M.; Tatehara, S.; Satomura, K.; Fujisawa, K.; Nagayama, M. Effect of FGF-2 and melatonin on implant bone healing: A histomorphometric study. J. Mater. Sci. Mater. Med. 2008, 19, 2949–2952. [Google Scholar] [CrossRef] [PubMed]
- Calvo-Guirado, J.L.; Gómez-Moreno, G.; López-Marí, L.; Guardia, J.; Marínez-González, J.M.; Barone, A.; Tresguerres, I.F.; Paredes, S.D.; Fuentes-Breto, L. Actions of melatonin mixed with collagenized porcine bone versus porcine bone only on osteointegration of dental implants. J. Pineal Res. 2010, 48, 194–203. [Google Scholar] [CrossRef] [PubMed]
- Guardia, J.; Gómez-Moreno, G.; Ferrera, M.J.; Cutando, A. Evaluation of effects of topic melatonin on implant surface at 5 and 8 weeks in Beagle dogs. Clin. Implant. Dent. Relat. Res. 2011, 13, 262–268. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, F.; López-Peña, M.; Miño, N.; Gómez-Moreno, G.; Guardia, J.; Cutando, A. Topical application of melatonin and growth hormone accelerates bone healing around dental implants in dogs. Clin. Implant. Dent. Relat. Res. 2012, 14, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Tresguerres, I.F.; Clemente, C.; Blanco, L.; Khraisat, A.; Tamimi, F.; Tresguerres, J.A. Effects of local melatonin application on implant osseointegration. Clin. Implant. Dent. Relat. Res. 2012, 14, 395–399. [Google Scholar] [CrossRef] [PubMed]
- Salomó-Coll, O.; Maté-Sánchez de Val, J.E.; Ramírez-Fernández, M.P.; Satorres-Nieto, M.; Gargallo-Albiol, J.; Calvo-Guirado, J.L. Osseoinductive elements for promoting osseointegration around immediate implants: A pilot study in the foxhound dog. Clin. Oral Implants Res. 2016, 27, e167–e175. [Google Scholar] [CrossRef] [PubMed]
- Calvo-Guirado, J.L.; Gómez-Moreno, G.; Maté-Sánchez, J.E.; López-Marí, L.; Delgado-Ruiz, R.; Romanos, G.E. New bone formation in bone defects after melatonin and porcine bone grafts: Experimental study in rabbits. Clin. Oral Implants Res. 2015, 26, 399–406. [Google Scholar] [CrossRef] [PubMed]
- Calvo-Guirado, J.L.; Aguilar Salvatierra, A.; Gargallo-Albiol, J.; Delgado-Ruiz, R.A.; Maté Sanchez, J.E.; Satorres-Nieto, M. Zirconia with laser-modified microgrooved surface vs. titanium implants covered with melatonin stimulates bone formation. Experimental study in tibia rabbits. Clin. Oral Implants Res. 2015, 26, 1421–1429. [Google Scholar] [CrossRef] [PubMed]
- Calvo-Guirado, J.L.; López-López, P.J.; Domínguez, M.F.; Gosálvez, M.M.; Prados-Frutos, J.C.; Gehrke, S.A. Histologic evaluation of new bone in post-extraction sockets induced by melatonin and apigenin: An experimental study in American fox hound dogs. Clin. Oral Implants Res. 2016. [Google Scholar] [CrossRef] [PubMed]
- Cutando, A.; Arana, C.; Gómez-Moreno, G.; Escaes, G.; López, A.; Ferrera, M.J.; Reiter, R.J.; Acuña-Castroviejo, D. Local application of melatonin into alveolar sockets of beagle dogs reduces tooth-removal oxidative stress. J. Periodontol. 2007, 78, 576–583. [Google Scholar] [CrossRef] [PubMed]
- Mitri, F.F.; Yoshimoto, M.; Allegrini Júnior, S.; Koo, S.; Carbonari, M.J.; König Júnior, B. Histological findings in titanium implants coated with calcium phosphate ceramics installed in rabbit's tibias. Ann. Anat. 2005, 187, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.C.; Yoshino, F.; Shoji, H.; Takahashi, S.; Todoki, K.; Shimada, S.; Kuse-Barouch, K. Characterization by electron spin resonance spectroscopy of reactive oxygen species generated by titanium dioxide and hydrogen peroxide. J. Dent. Res. 2005, 84, 178–182. [Google Scholar] [CrossRef] [PubMed]
- Taylor, G.C.; Waddington, R.J.; Moseley, R.; Williams, K.R.; Embery, G. Influence of titanium oxide and titanium peroxy gel on the breakdown of hyaluronan by reactive oxygen species. Biomaterials 1996, 17, 1313–1319. [Google Scholar] [CrossRef]
- Cutando, A.; Gómez-Moreno, G.; Arana, C.; Acuña-Castroviejo, D.; Reiter, R.J. Melatonin: Potential functions in the oral cavity. J. Periodontol. 2007, 78, 1094–1102. [Google Scholar] [CrossRef] [PubMed]
- De la Rocha, N.; Rotelli, A.; Aguilar, C.F.; Pelzer, L. Structural basis of the anti-inflammatory activity of melatonin. Arzneimittelforschung 2007, 57, 782–786. [Google Scholar] [PubMed]
- Gustafsson, A.; Asman, B. Increased release of free oxygen radicals from peripheral neutrophils in adult periodontitis after Fc delta-receptor stimulation. J. Clin. Periodontol. 1996, 23, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Battino, M.; Bullon, P.; Wilson, M.; Newman, H. Oxidative injury and inflammatory periodontal diseases: The challenge of anti-oxidants to free radicals and reactive oxygen species. Crit. Rev. Oral Biol. Med. 1999, 10, 458–476. [Google Scholar] [CrossRef] [PubMed]
- Kimura, S.; Yonemura, T.; Kaya, H. Increased oxidative product formation by peripheral blood polymorphonuclear leukocytes in human periodontal diseases. J. Periodontal Res. 1993, 28, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Sies, H. Oxidative stress: Oxidants and antioxidants. Exp. Physiol. 1997, 82, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Najeeb, S.; Khurshid, Z.; Zohaib, S.; Zafar, M.S. Therapeutic potential of melatonin in oral medicine and periodontology. Kachsiung J. Med. Sci. 2016, 32, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Srinath, R.; Acharya, A.B.; Thakur, S.L. Salivary and gingival crevicular fluid melatonin in periodontal health and disease. J. Periodontol. 2010, 81, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Cutando, A.; Galindo, P.; Gómez-Moreno, G.; Arana, C.; Bolaños, J.; Acuña-Castroviejo, D.; Wang, H.L. Relationship between salivary melatonin and severity of periodontal disease. J. Periodontol. 2006, 77, 1533–1538. [Google Scholar] [CrossRef] [PubMed]
- Almughrabi, O.M.; Marzouk, K.M.; Hasanato, R.M.; Shafik, S.S. Melatonin levels in periodontal health and disease. J. Periodontal Res. 2013, 48, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Moreno, G.; Cutando-Soriano, A.; Arana, C.; Galindo, P.; Bolaños, J.; Acuña-Castroviejo, D.; Wang, H.L. Melatonin expression in periodontal disease. J. Periodontal Res. 2007, 42, 536–540. [Google Scholar] [CrossRef] [PubMed]
- Cutando, A.; Gómez-Moreno, G.; Villalba, J.; Ferrera, M.J.; Escames, G.; Acuña-Castroviejo, D. Relationship between salivary melatonin levels and periodontal status in diabetic patients. J. Pineal Res. 2003, 35, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Kara, A.; Akman, S.; Ozkanlar, S.; Tozoglu, U.; Kalkan, Y.; Canakci, C.F.; Tozoglu, S. Immune modulatory and antioxidant effects of melatonin in experimental periodontitis in rats. Free Radic. Biol. Med. 2013, 55, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Ghallab, N.A.; Hamdy, E.; Shaker, O.G. Malondialdehyde, superoxide dismutase and melatonin levels in GCF of aggressive and chronic periodontitis patients. Aust. Dent. J. 2016, 61, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Lodhi, K.; Saimbi, C.S.; Khan, M.A.; Nath, C.; Shukla, R. Evaluation of melatonin levels in saliva in gingivitis and periodontitis cases: A pilot study. Contemp. Clin. Dent. 2016, 7, 519–523. [Google Scholar] [CrossRef] [PubMed]
- Shino, H.; Hasuike, A.; Arai, Y.; Honda, M.; Isokawa, K.; Sato, S. Melatonin enhances vertical bone augmentation in rat calvaria secluded spaces. Med. Oral Patol. Oral Cir. Bucal. 2016, 21, e122–e126. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, S.; Cooke, J.; Fotek, P.; Wang, H.L. Vertical bone augmentation: Where are we now? Implant. Dent. 2006, 15, 219–228. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Huang, F.; He, H.W. Melatonin effects on hard tissues: Bone and tooth. Int. J. Mol. Sci. 2013, 14, 10063–10074. [Google Scholar] [CrossRef] [PubMed]
- Kumasaka, S.; Shimozuma, M.; Kawamoto, T.; Mishima, K.; Tokuyama, R.; Kamiya, J.; Davvdorj, P.; Saito, I.; Satomura, K. Possible involvement of melatonin in tooth development: Expression of melatonin 1a receptor in human and mouse tooth germs. Histochem. Cell Biol. 2010, 133, 577–584. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J. Mechanisms of cancer inhibition by melatonin. J. Pineal Res. 2004, 37, 213–214. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, E.; Kozaki, K.; Tsuda, H.; Suzuki, E.; Pimkhaokham, A.; Yamamoto, G.; Irie, T.; Tachinawa, T.; Amagasa, T.; Inazawa, J.; Imoto, I. Frequent silencing of a putative tumor suppressor gene melatonin receptor 1A (MTNR1A) in oral squamous-cell carcinoma. Cancer Sci. 2008, 99, 1390–1400. [Google Scholar] [CrossRef] [PubMed]
- Lissoni, P. Is there a role for melatonin in supportive care? Support Care Cancer 2002, 10, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Niscola, P.; Tendas, A.; Cupelli, L.; Catalano, G.; Scaramucci, L.; Giovannini, M.; Trinchieri, V.; Sharma, A.; Efficace, F.; Cartoni, C.; et al. The prevention of oral mucositis in patirnts with blood cancers: Current concepts and emerging landscapes. Cardiovasc. Hematol. Agents Med. Chem. 2012, 10, 362–375. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Tan, D.X.; Korkmaz, A.; Erren, T.C.; Piekarski, C.; Tamura, H. Light at night, chronodisruption, melatonin suppression and cancer risk: A review. Crit. Rev. Oncog. 2007, 13, 303–328. [Google Scholar] [CrossRef] [PubMed]
- Chaiyarit, P.; Man, N.; Hiraku, Y.; Pinlaor, S.; Yongvanit, P.; Jintakanon, D.; Murata, M.; Oikawa, S.; Kawanishi, S. Nitrative and oxidative DNA damage in oral lichen planus in relation to human oral carcinogenesis. Cancer Sci. 2005, 96, 553–559. [Google Scholar] [CrossRef] [PubMed]
- Bonilla, E.; Valero, N.; Chacín-Bonilla, L.; Medina-Leendertz, S. Melatonin and viral infections. J. Pineal Res. 2004, 36, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Nunes, O.D.S.; Pereira, R.D.S. Regression of herpes viral infection symptoms using melatonin and SB-73: Comparison with Acyclovir. J. Pineal Res. 2008, 44, 373–378. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, K.B. Oxidative stress during viral infection: A review. Free Radic. Biol. Med. 1996, 21, 641–649. [Google Scholar] [CrossRef]
- Reynolds, F.D.; Dauchy, R.; Blask, D.; Dietz, P.A.; Lynch, D.; Zuckerman, R. The pineal gland hormone melatonin improves survival in a rat model of sepsis/shock induced by zymosan. Surgery 2003, 134, 474–479. [Google Scholar] [CrossRef]
- Nunnari, G.; Nigro, L.; Palermo, F.; leto, D.; Pomerantz, R.J.; Cacopardo, B. Reduction of serum melatonin levels in Hiz-1-infected individuals parallel disease progression: Correlation with serum interleukin-12 leves. Infection 2003, 31, 379–382. [Google Scholar]
- Tekbas, O.F.; Ogur, R.; Korkmaz, A.; Kilic, A.; Reiter, R.J. Melatonin as an antibiotic: New insights into the actions of this ubiquitous molecule. J. Pineal Res. 2008, 44, 222–226. [Google Scholar] [CrossRef] [PubMed]
- Limson, J.; Nyokong, T.; Daya, S. The interaction of melatonin and its precursors with aluminum, cadmium, copper, iron, lead and zinc: An adsorptive volumetric study. J. Pineal Res. 1998, 24, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Cengiz, M.I.; Cengiz, S.; Wang, H.L. Melatonin and oral cavity. Int. J. Dent. 2012, 2012, 491872. [Google Scholar] [CrossRef] [PubMed]
- Simon, Z.; Watson, P.A. Biomimetic dental implants—New ways to enhance osseointegration. J. Can. Dent. Assoc. 2002, 68, 286–288. [Google Scholar] [PubMed]
- Gibbs, F.P.; Vriend, J. The half-life of melatonin elimination from rat plasma. Endocrinology 1981, 109, 1796–1798. [Google Scholar] [CrossRef] [PubMed]
- Salvi, G.E.; Beck, J.D.; Offenbacher, S. PGE2, IL-1β, and TNF-α responses in diabetics as modifiers of periodontal disease expression. Ann. Periodontol. 1998, 3, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Poon, A.M.; Liu, Z.M.; Pang, C.S.; Brown, G.M.; Pang, S.F. Evidence for a direct action of melatonin on the immune system. Biol. Signals 1994, 3, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Calvo, J.R.; Karbownik, M.; Qi, W.; Tan, D.X. Melatonin and its relation to the immune system and inflammation. Ann. N. Y. Acad. Sci. 2000, 917, 376–386. [Google Scholar] [CrossRef] [PubMed]
- Mrnka, L.; Hock, M.; Rybová, M.; Pácha, J. Melatonin inhibits prostaglandin E2 and sodium nitroprusside-induced ion secretion in rat distal colon. Eur. J. Pharmacol. 2008, 581, 164–170. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Tan, D.X.; Sainz, R.M.; Mayo, J.C. Melatonin: Reducing the toxicity and increasing the efficacy of drugs. J. Pharm. Pharmacol. 2002, 54, 1299–1321. [Google Scholar] [CrossRef] [PubMed]
| Study | Animal Model | Animals/Implants | Time | Alone or Combination | Results |
|---|---|---|---|---|---|
| Cutando et al., 2008 [2] | Beagle dog | 12/72 | 2 weeks | Alone | ↑ BIC in melatonin group |
| Takechi et al., 2008 [40] | Wistar rat | 10/20 | 4 weeks | + FGF-2 | ↑ BIC and bone density in combination group |
| Calvo-Guirado et al., 2010 [41] | Beagle dog | 12/48 | 4 weeks | + porcine bone | ↑ BIC, bone density and new bone in combination group |
| Guardia et al., 2015 [42] | Beagle dog | 12/72 | 5 and 8 weeks | Alone | ↑ Bone formation in melatonin group |
| Muñoz et al., 2012 [43] | Beagle dog | 12/48 | 2, 5 and 8 weeks | + Growth hormone | ↑ BIC ando bone density at 2 and 5 weeks in combination group |
| Tresguerres et al., 2012 [44] | Rabbit | 10/40 | 4 weeks | Alone | ↑ trabecular BIC in melatonin group |
| Salomó-Coll et al., 2015 [45] | Foxhound dog | 6/24 | 12 weeks | Alone | ↑ total BIC in melatonin group |
| Calvo-Guirado et al., 2015 [47] | Rabbit | 20/80 | 1 and 10 weeks | Alone | ↑ BIC in titanium and zirconium implants at 1 week, in zirconium at 10 weeks |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Permuy, M.; López-Peña, M.; González-Cantalapiedra, A.; Muñoz, F. Melatonin: A Review of Its Potential Functions and Effects on Dental Diseases. Int. J. Mol. Sci. 2017, 18, 865. https://doi.org/10.3390/ijms18040865
Permuy M, López-Peña M, González-Cantalapiedra A, Muñoz F. Melatonin: A Review of Its Potential Functions and Effects on Dental Diseases. International Journal of Molecular Sciences. 2017; 18(4):865. https://doi.org/10.3390/ijms18040865
Chicago/Turabian StylePermuy, Maria, Mónica López-Peña, Antonio González-Cantalapiedra, and Fernando Muñoz. 2017. "Melatonin: A Review of Its Potential Functions and Effects on Dental Diseases" International Journal of Molecular Sciences 18, no. 4: 865. https://doi.org/10.3390/ijms18040865
APA StylePermuy, M., López-Peña, M., González-Cantalapiedra, A., & Muñoz, F. (2017). Melatonin: A Review of Its Potential Functions and Effects on Dental Diseases. International Journal of Molecular Sciences, 18(4), 865. https://doi.org/10.3390/ijms18040865






