Effects of a Sativex-Like Combination of Phytocannabinoids on Disease Progression in R6/2 Mice, an Experimental Model of Huntington’s Disease
Abstract
:1. Introduction
2. Results
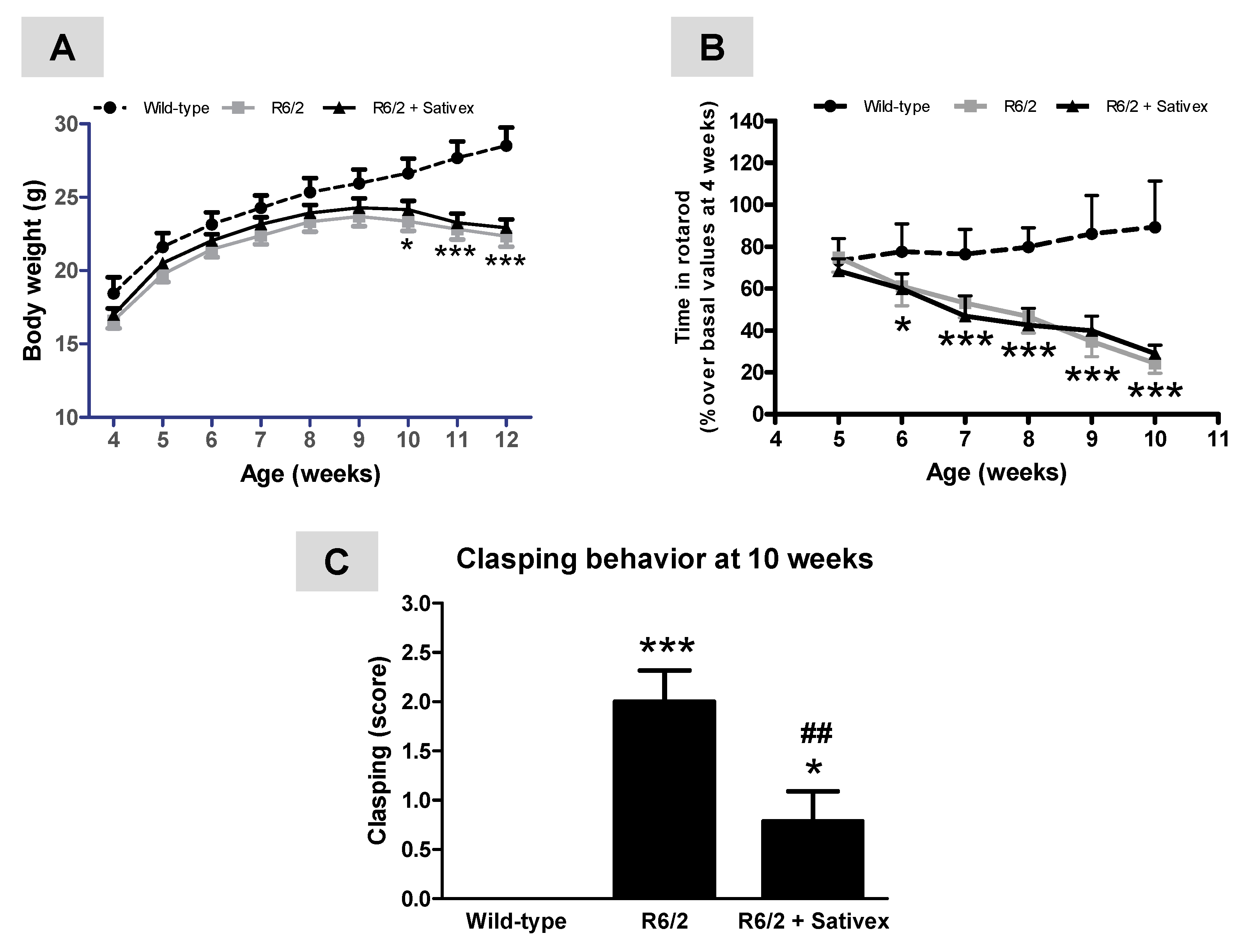
2.1. Behavioral Analysis of R6/2 Mice Treated with the Sativex-Like Combination of Phytocannabinoids
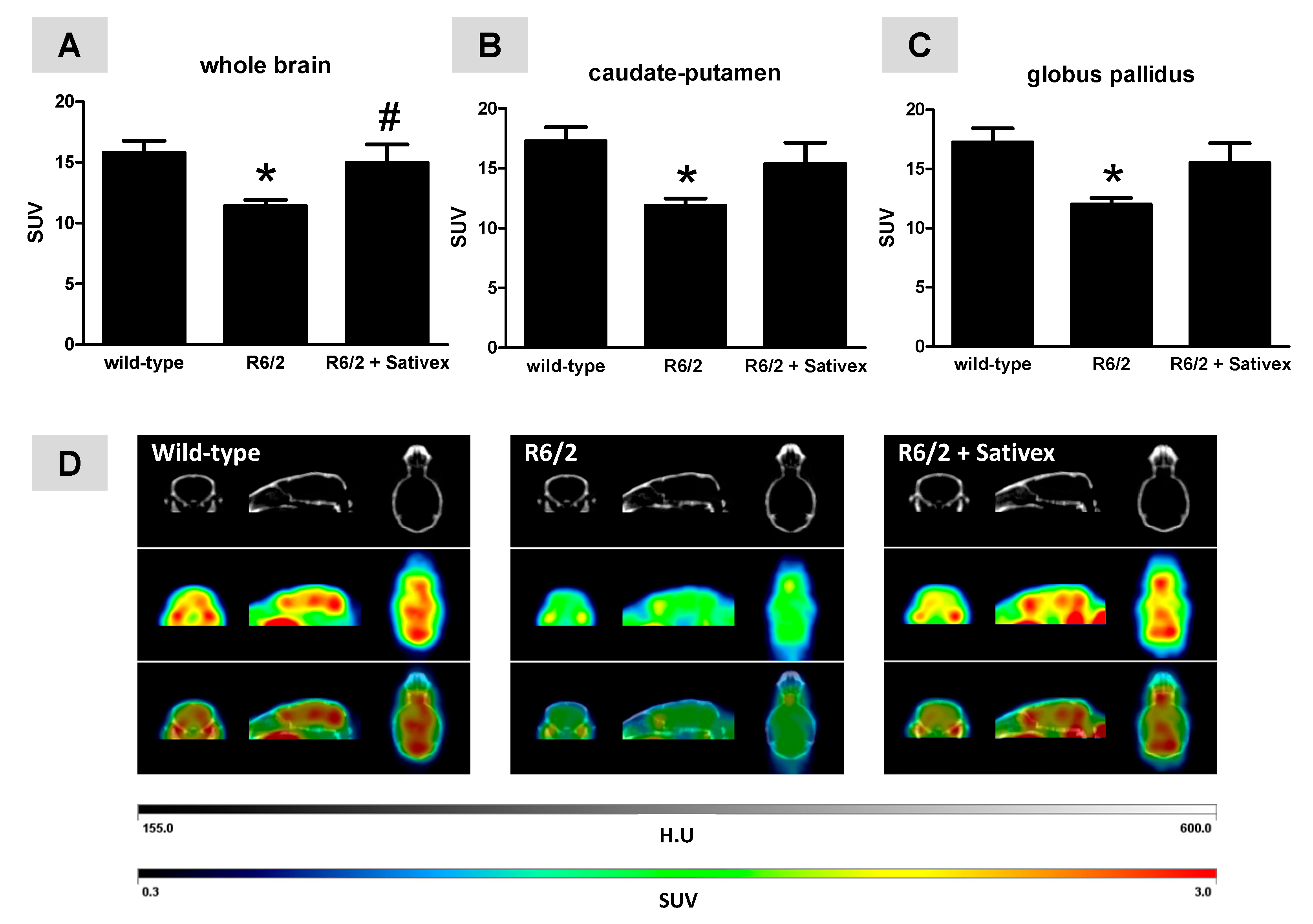
2.2. In Vivo Imaging Analysis of Regional Brain Metabolic Activity in R6/2 Mice Treated with the Sativex-Like Combination of Phytocannabinoids
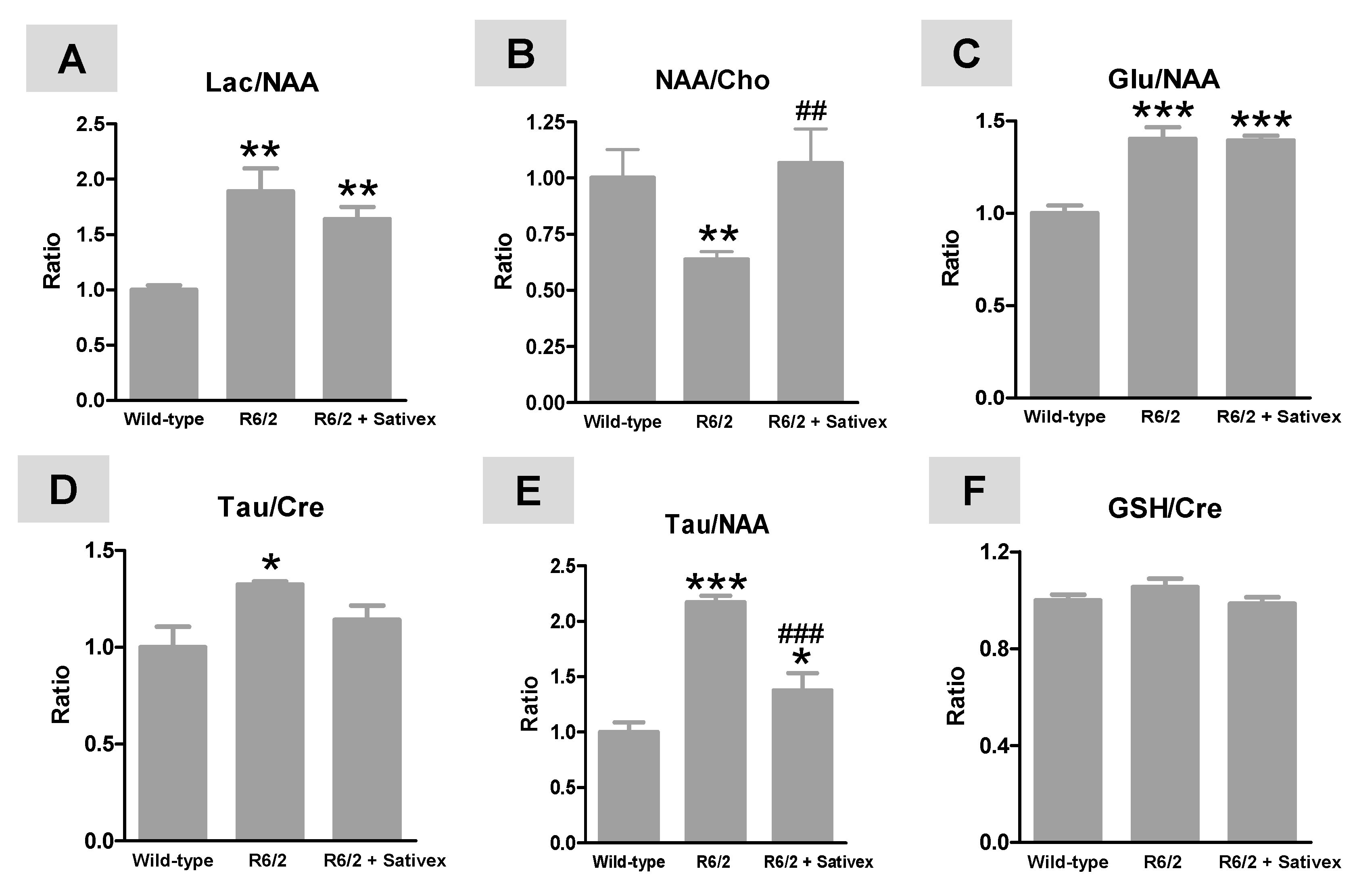
2.3. H+-MRS Analysis of Several Markers of Brain Integrity in R6/2 Mice Treated with the Sativex-Like Combination of Phytocannabinoids
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Treatments and Sampling
4.3. Behavioral Recording
4.3.1. Rotarod Test
4.3.2. Hindlimb Clasping
4.4. In Vivo Analysis of Glucose Metabolism: [18F]FDG PET Imaging
4.5. Proton Magnetic Resonance Spectroscopy (H+-MRS)
4.6. Statistics
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Roze, E.; Bonnet, C.; Betuing, S.; Caboche, J. Huntington’s disease. Adv. Exp. Med. Biol. 2010, 685, 45–63. [Google Scholar] [PubMed]
- Zuccato, C.; Valenza, M.; Cattaneo, E. Molecular mechanisms and potential therapeutical targets in Huntington’s disease. Physiol. Rev. 2010, 90, 905–981. [Google Scholar] [CrossRef] [PubMed]
- Johnson, C.D.; Davidson, B.L. Huntington’s disease: Progress toward effective disease-modifying treatments and a cure. Hum. Mol. Genet. 2010, 19, R98–R102. [Google Scholar] [CrossRef] [PubMed]
- López-Sendón Moreno, J.L.; García Caldentey, J.; Trigo Cubillo, P.; Ruiz Romero, C.; García Ribas, G.; Alonso Arias, M.A.; García de Yébenes, M.J.; Tolón, R.M.; Galve-Roperh, I.; Sagredo, O.; et al. A double-blind, randomized, cross-over, placebo-controlled, pilot trial with Sativex in Huntington’s disease. J. Neurol. 2016, 263, 1390–1400. [Google Scholar] [PubMed]
- Pazos, M.R.; Sagredo, O.; Fernández-Ruiz, J. The endocannabinoid system in Huntington’s disease. Curr. Pharm. Des. 2008, 14, 2317–2325. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Ruiz, J.; García, C.; Sagredo, O.; Gómez-Ruiz, M.; de Lago, E. The endocannabinoid system as a target for the treatment of neuronal damage. Expert Opin. Ther. Targets 2010, 14, 387–404. [Google Scholar] [CrossRef] [PubMed]
- Lastres-Becker, I.; Bizat, N.; Boyer, F.; Hantraye, P.; Fernández-Ruiz, J.; Brouillet, E. Potential involvement of cannabinoid receptors in 3-nitropropionic acid toxicity in vivo. Neuroreport 2004, 15, 2375–2379. [Google Scholar] [CrossRef] [PubMed]
- Sagredo, O.; Ramos, J.A.; Decio, A.; Mechoulam, R.; Fernández-Ruiz, J. Cannabidiol reduced the striatal atrophy caused 3-nitropropionic acid in vivo by mechanisms independent of the activation of cannabinoid, vanilloid TRPV1 and adenosine A2A receptors. Eur. J. Neurosci. 2007, 26, 843–851. [Google Scholar] [CrossRef] [PubMed]
- Valdeolivas, S.; Navarrete, C.; Cantarero, I.; Bellido, M.L.; Muñoz, E.; Sagredo, O. Neuroprotective properties of cannabigerol in Huntington’s disease: Studies in R6/2 mice and 3-nitropropionate-lesioned mice. Neurotherapeutics 2015, 12, 185–199. [Google Scholar] [CrossRef] [PubMed]
- Blázquez, C.; Chiarlone, A.; Sagredo, O.; Aguado, T.; Pazos, M.R.; Resel, E.; Palazuelos, J.; Julien, B.; Salazar, M.; Börner, C.; et al. Loss of striatal type 1 cannabinoid receptors is a key pathogenic factor in Huntington’s disease. Brain 2011, 134, 119–136. [Google Scholar] [CrossRef] [PubMed]
- Palazuelos, J.; Aguado, T.; Pazos, M.R.; Julien, B.; Carrasco, C.; Resel, E.; Palazuelos, J.; Julien, B.; Salazar, M.; Börner, C.; et al. Microglial CB2 cannabinoid receptors are neuroprotective in Huntington’s disease excitotoxicity. Brain 2009, 132, 3152–3164. [Google Scholar] [CrossRef] [PubMed]
- Sagredo, O.; González, S.; Aroyo, I.; Pazos, M.R.; Benito, C.; Lastres-Becker, I.; Romero, J.P.; Tolón, R.M.; Mechoulam, R.; Brouillet, E.; et al. Cannabinoid CB2 receptor agonists protect the striatum against malonate toxicity: Relevance for Huntington’s disease. Glia 2009, 57, 1154–1167. [Google Scholar] [CrossRef] [PubMed]
- Pintor, A.; Tebano, M.T.; Martire, A.; Grieco, R.; Galluzzo, M.; Scattoni, M.L.; Pèzzola, A.; Coccurello, R.; Felici, F.; Cuomo, V.; et al. The cannabinoid receptor agonist WIN 55,212-2 attenuates the effects induced by quinolinic acid in the rat striatum. Neuropharmacology 2006, 51, 1004–1012. [Google Scholar] [CrossRef] [PubMed]
- Dowie, M.J.; Howard, M.L.; Nicholson, L.F.; Faull, R.L.; Hannan, A.J.; Glass, M. Behavioural and molecular consequences of chronic cannabinoid treatment in Huntington’s disease transgenic mice. Neuroscience 2010, 170, 324–336. [Google Scholar] [CrossRef] [PubMed]
- Sastre-Garriga, J.; Vila, C.; Clissold, S.; Montalban, X. THC and CBD oromucosal spray (Sativex®) in the management of spasticity associated with multiple sclerosis. Expert Rev. Neurother. 2011, 11, 627–637. [Google Scholar] [PubMed]
- Zajicek, J.P.; Apostu, V.I. Role of cannabinoids in multiple sclerosis. CNS Drugs 2011, 25, 187–201. [Google Scholar] [CrossRef] [PubMed]
- Pertwee, R.G. Emerging strategies for exploiting cannabinoid receptor agonists as medicines. Br. J. Pharmacol. 2009, 156, 397–411. [Google Scholar] [CrossRef] [PubMed]
- Sagredo, O.; Pazos, M.R.; Satta, V.; Ramos, J.A.; Pertwee, R.G.; Fernández-Ruiz, J. Neuroprotective effects of phytocannabinoid-based medicines in experimental models of Huntington’s disease. J. Neurosci. Res. 2011, 89, 1509–1518. [Google Scholar] [CrossRef] [PubMed]
- Lastres-Becker, I.; Bizat, N.; Boyer, F.; Hantraye, P.; Brouillet, E.; Fernández-Ruiz, J. Effects of cannabinoids in the rat model of Huntington’s disease generated by an intrastriatal injection of malonate. Neuroreport 2003, 14, 813–816. [Google Scholar] [CrossRef] [PubMed]
- Valdeolivas, S.; Satta, V.; Pertwee, R.G.; Fernández-Ruiz, J.; Sagredo, O. Sativex-like combination of phytocannabinoids is neuroprotective in malonate-lesioned rats, an inflammatory model of Huntington’s disease: Role of CB1 and CB2 receptors. ACS Chem. Neurosci. 2012, 3, 400–406. [Google Scholar] [PubMed]
- Jiménez-Xarrié, E.; Davila, M.; Gil-Perotín, S.; Jurado-Rodríguez, A.; Candiota, A.P.; Delgado-Mederos, R.; Lope-Piedrafita, S.; García-Verdugo, J.M.; Arús, C.; Martí-Fàbregas, J. In vivo and ex vivo magnetic resonance spectroscopy of the infarct and the subventricular zone in experimental stroke. J. Cereb. Blood Flow Metab. 2015, 35, 828–834. [Google Scholar] [CrossRef] [PubMed]
- Pazos, M.R.; Mohammed, N.; Lafuente, H.; Santos, M.; Martínez-Pinilla, E.; Moreno, E.; Valdizan, E.; Romero, J.; Pazos, A.; Franco, R.; et al. Mechanisms of cannabidiol neuroprotection in hypoxic-ischemic newborn pigs: Role of 5HT1A and CB2 receptors. Neuropharmacology 2013, 71, 282–291. [Google Scholar] [PubMed]
- Lafuente, H.; Pazos, M.R.; Alvarez, A.; Mohammed, N.; Santos, M.; Arizti, M.; Alvarez, F.J.; Martinez-Orgado, J.A. Effects of cannabidiol and hypothermia on short-term brain damage in new-born piglets after acute hypoxia-ischemia. Front. Neurosci. 2016, 10, 323. [Google Scholar] [CrossRef] [PubMed]
- Tsang, T.M.; Woodman, B.; McLoughlin, G.A.; Griffin, J.L.; Tabrizi, S.J.; Bates, G.P.; Holmes, E. Metabolic characterization of the R6/2 transgenic mouse model of Huntington’s disease by high-resolution MAS 1H NMR spectroscopy. J. Proteome Res. 2006, 5, 483–492. [Google Scholar] [PubMed]
- Verwaest, K.A.; Vu, T.N.; Laukens, K.; Clemens, L.E.; Nguyen, H.P.; van Gasse, B.; Martins, J.C.; van der Linden, A.; Dommisse, R. 1H NMR based metabolomics of CSF and blood serum: A metabolic profile for a transgenic rat model of Huntington disease. Biochim. Biophys. Acta 2011, 1812, 1371–1379. [Google Scholar] [CrossRef] [PubMed]
- Heikkinen, T.; Lehtimäki, K.; Vartiainen, N.; Puoliväli, J.; Hendricks, S.J.; Glaser, J.R.; Bradaia, A.; Wadel, K.; Touller, C.; Kontkanen, O.; et al. Characterization of neurophysiological and behavioral changes, MRI brain volumetry and 1H MRS in zQ175 knock-in mouse model of Huntington’s disease. PLoS ONE 2012, 7, e50717. [Google Scholar] [CrossRef] [PubMed]
- Wegrzynowicz, M.; Bichell, T.J.; Soares, B.D.; Loth, M.K.; McGlothan, J.S.; Mori, S.; Alikhan, F.S.; Hua, K.; Coughlin, J.M.; Holt, H.K.; et al. Novel BAC mouse model of Huntington’s disease with 225 CAG repeats exhibits an early widespread and stable degenerative phenotype. J. Huntingt. Dis. 2015, 4, 17–36. [Google Scholar]
- Tallaksen-Greene, S.J.; Sadagurski, M.; Zeng, L.; Mauch, R.; Perkins, M.; Banduseela, V.C.; Lieberman, A.P.; Miller, R.A.; Paulson, H.L.; Albin, R.L. Differential effects of delayed aging on phenotype and striatal pathology in a murine model of Huntington disease. J. Neurosci. 2014, 34, 15658–15668. [Google Scholar] [PubMed]
- Graham, S.F.; Kumar, P.; Bahado-Singh, R.O.; Robinson, A.; Mann, D.; Green, B.D. Novel metabolite biomarkers of Huntington’s disease as detected by High-Resolution Mass Spectrometry. J. Proteome Res. 2016, 15, 1592–1601. [Google Scholar] [PubMed]
- Satoh, T.; Yoshioka, Y. Contribution of reduced and oxidized glutathione to signals detected by magnetic resonance spectroscopy as indicators of local brain redox state. Neurosci. Res. 2006, 55, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Curtis, A.; Mitchell, I.; Patel, S.; Ives, N.; Rickards, H. A pilot study using nabilone for symptomatic treatment in Huntington’s disease. Mov. Disord. 2009, 24, 2254–2259. [Google Scholar] [CrossRef] [PubMed]
- Curtis, A.; Rickards, H. Nabilone could treat chorea and irritability in Huntington’s disease. J. Neuropsychiatry Clin. Neurosci. 2006, 18, 553–554. [Google Scholar] [CrossRef] [PubMed]
- Müller-Vahl, K.R.; Schneider, U.; Emrich, H.M. Nabilone increases choreatic movements in Huntington’s disease. Mov. Disord. 1999, 14, 1038–1040. [Google Scholar] [CrossRef]
- Consroe, P.; Laguna, J.; Allender, J.; Snider, S.; Stern, L.; Sandyk, R.; Kennedy, K.; Schram, K. Controlled clinical trial of cannabidiol in Huntington’s disease. Pharmacol. Biochem. Behav. 1991, 40, 701–708. [Google Scholar] [CrossRef]
- Petr, G.T.; Schultheis, L.A.; Hussey, K.C.; Sun, Y.; Dubinsky, J.M.; Aoki, C.; Rosenberg, P.A. Decreased expression of GLT-1 in the R6/2 model of Huntington’s disease does not worsen disease progression. Eur. J. Neurosci. 2013, 38, 2477–2490. [Google Scholar] [CrossRef] [PubMed]
- Wade, D. Evaluation of the safety and tolerability profile of Sativex: Is it reassuring enough? Expert Rev. Neurother. 2012, 12, 9–14. [Google Scholar] [PubMed]
- Ferrante, R.J.; Andreassen, O.A.; Jenkins, B.G.; Dedeoglu, A.; Kuemmerle, S.; Kubilus, J.K.; Kaddurah-Daouk, R.; Hersch, S.M.; Beal, M.F. Neuroprotective effects of creatine in a transgenic mouse model of Huntington’s disease. J. Neurosci. 2000, 20, 4389–4397. [Google Scholar] [PubMed]
- Andreassen, O.A.; Dedeoglu, A.; Ferrante, R.J.; Jenkins, B.G.; Ferrante, K.L.; Thomas, M.; Friedlich, A.; Browne, S.E.; Schilling, G.; Borchelt, D.R.; et al. Creatine increase survival and delays motor symptoms in a transgenic animal model of Huntington’s disease. Neurobiol. Dis. 2001, 8, 479–491. [Google Scholar] [CrossRef] [PubMed]
- Tabrizi, S.J.; Blamire, A.M.; Manners, D.N.; Rajagopalan, B.; Styles, P.; Schapira, A.H.; Warner, T.T. Creatine therapy for Huntington’s disease: Clinical and MRS findings in a 1-year pilot study. Neurology 2003, 61, 141–142. [Google Scholar] [CrossRef] [PubMed]
- Deleye, S.; Verhaeghe, J.; wyffels, L.; Dedeurwaerdere, S.; Stroobants, S.; Staelens, S. Towards a reproducible protocol for repetitive and semi-quantitative rat brain imaging with 18F-FDG: Exemplified in a memantine pharmacological challenge. Neuroimage 2014, 96, 276–287. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valdeolivas, S.; Sagredo, O.; Delgado, M.; Pozo, M.A.; Fernández-Ruiz, J. Effects of a Sativex-Like Combination of Phytocannabinoids on Disease Progression in R6/2 Mice, an Experimental Model of Huntington’s Disease. Int. J. Mol. Sci. 2017, 18, 684. https://doi.org/10.3390/ijms18040684
Valdeolivas S, Sagredo O, Delgado M, Pozo MA, Fernández-Ruiz J. Effects of a Sativex-Like Combination of Phytocannabinoids on Disease Progression in R6/2 Mice, an Experimental Model of Huntington’s Disease. International Journal of Molecular Sciences. 2017; 18(4):684. https://doi.org/10.3390/ijms18040684
Chicago/Turabian StyleValdeolivas, Sara, Onintza Sagredo, Mercedes Delgado, Miguel A. Pozo, and Javier Fernández-Ruiz. 2017. "Effects of a Sativex-Like Combination of Phytocannabinoids on Disease Progression in R6/2 Mice, an Experimental Model of Huntington’s Disease" International Journal of Molecular Sciences 18, no. 4: 684. https://doi.org/10.3390/ijms18040684
APA StyleValdeolivas, S., Sagredo, O., Delgado, M., Pozo, M. A., & Fernández-Ruiz, J. (2017). Effects of a Sativex-Like Combination of Phytocannabinoids on Disease Progression in R6/2 Mice, an Experimental Model of Huntington’s Disease. International Journal of Molecular Sciences, 18(4), 684. https://doi.org/10.3390/ijms18040684






