Hydrogen Production by a Chlamydomonas reinhardtii Strain with Inducible Expression of Photosystem II
Abstract
:1. Introduction
2. Results
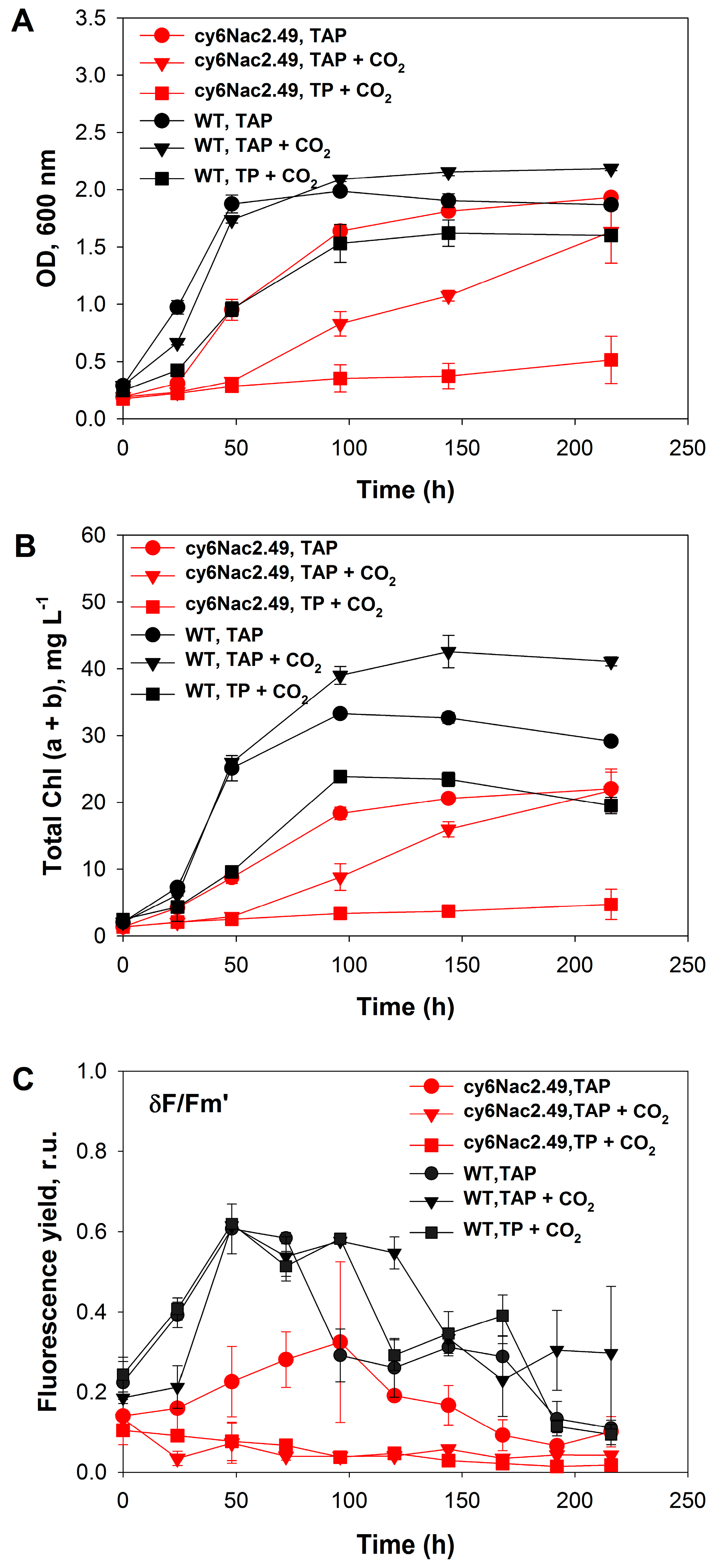
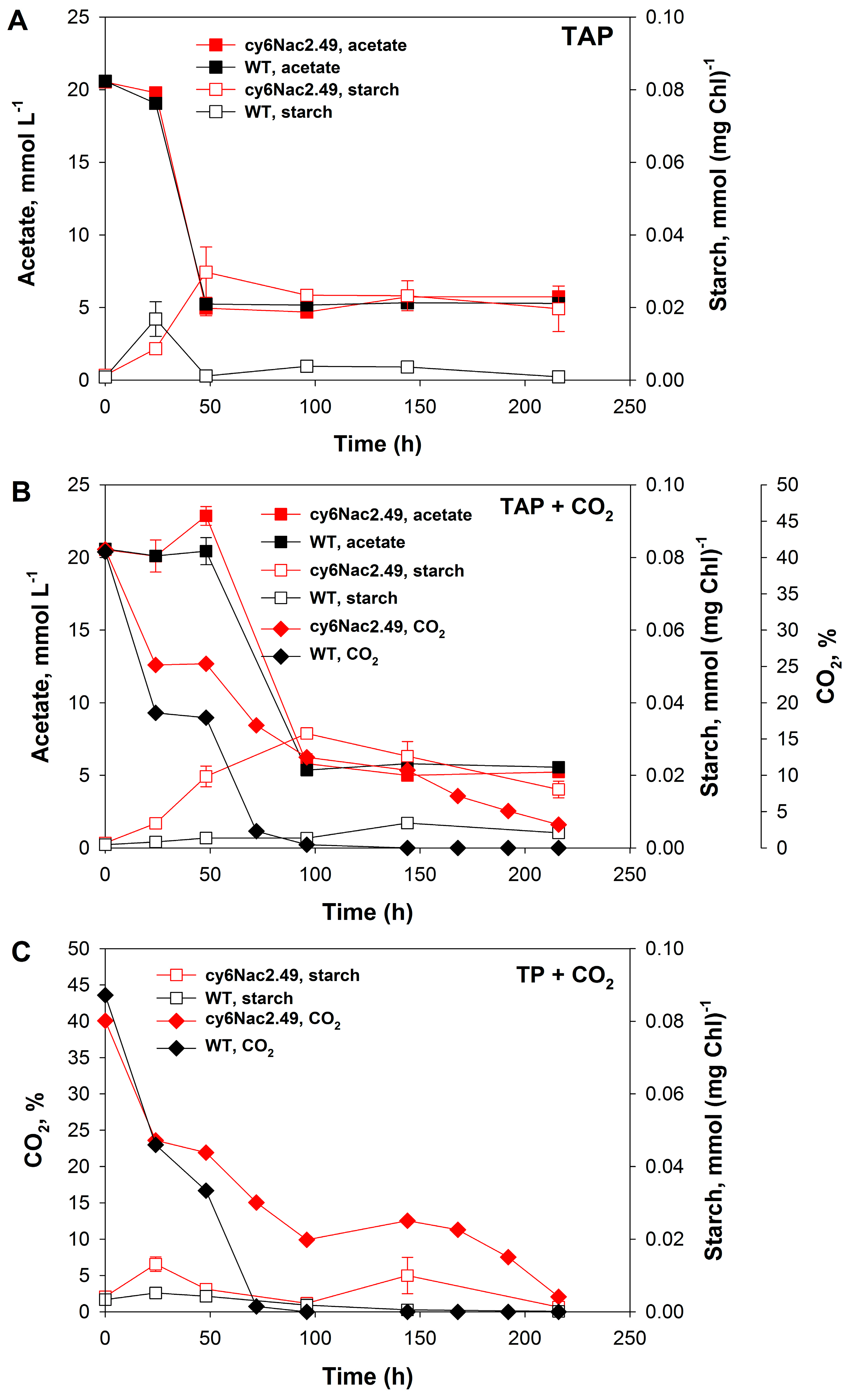
2.1. Cell Growth under Photoheterotrophic, Photomixotrophic and Autotrophic Conditions
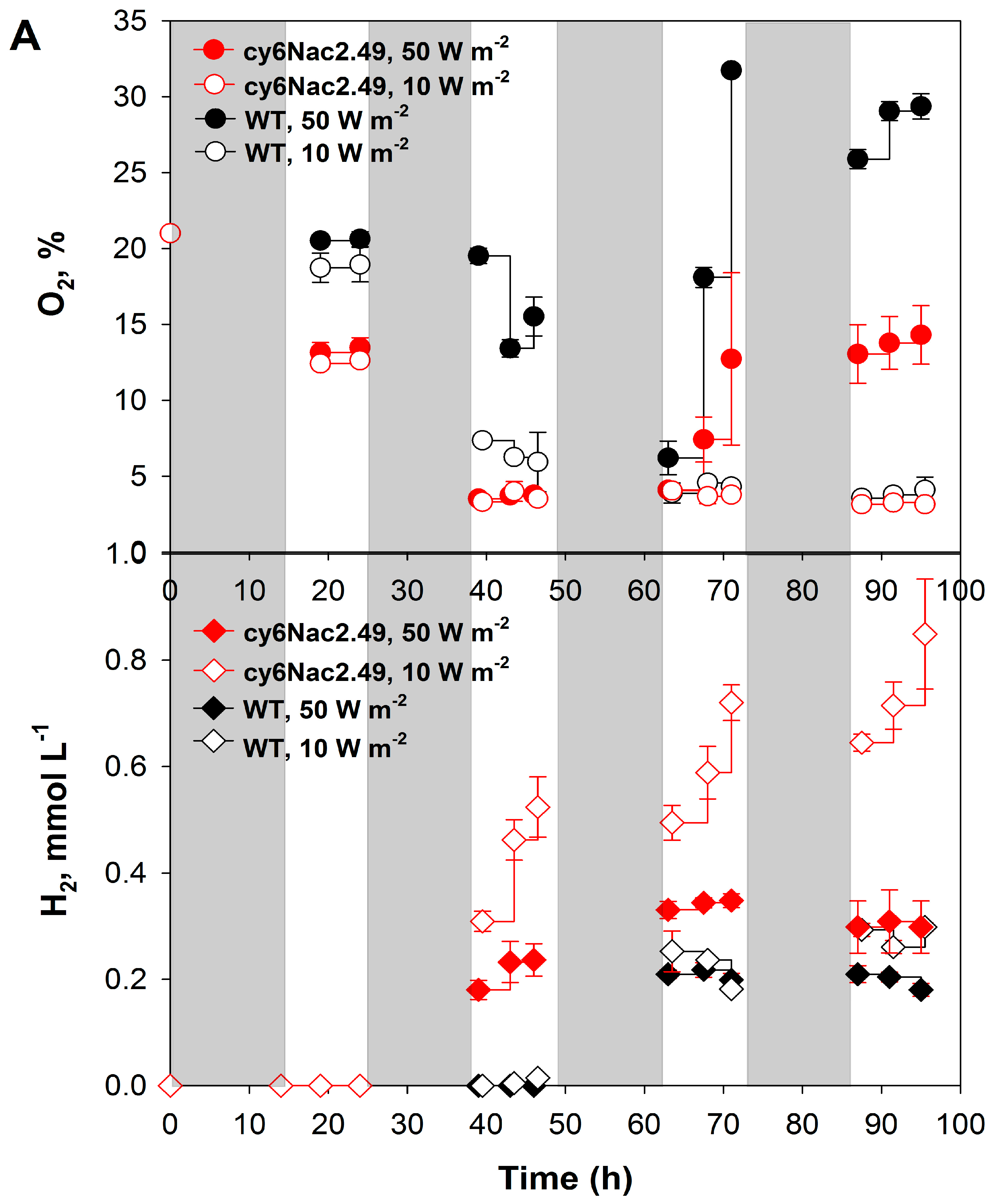
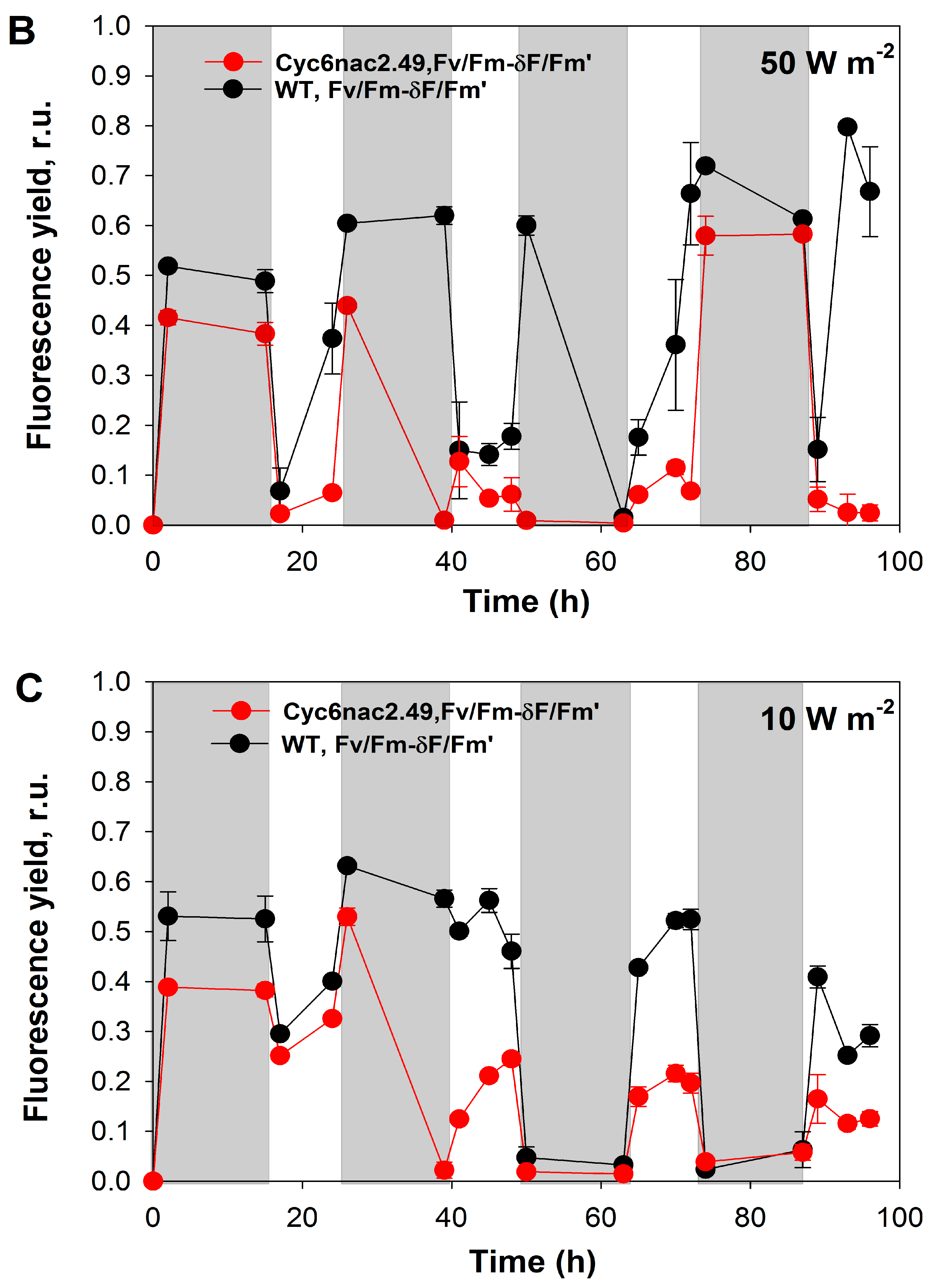
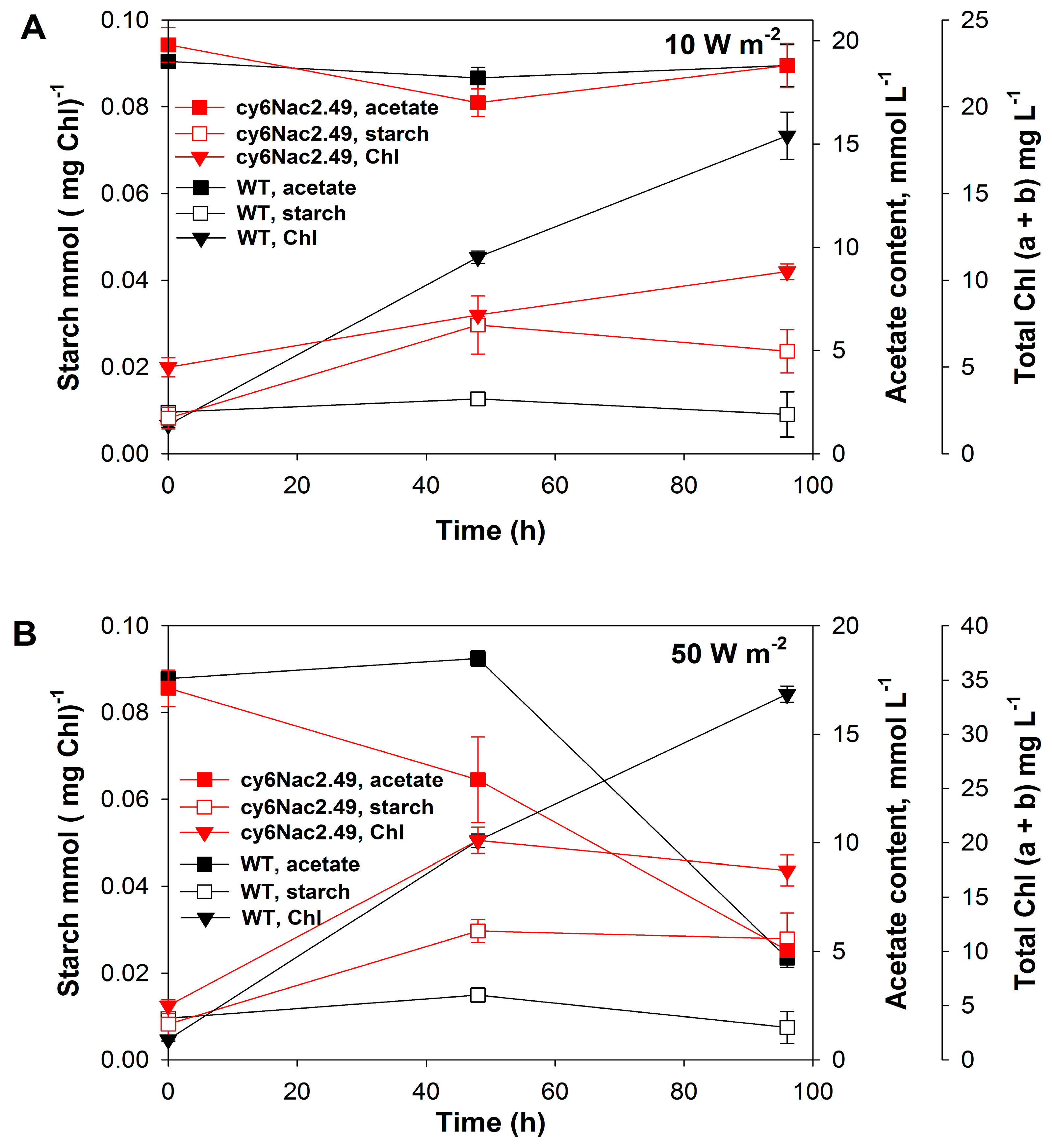
2.2. Effects of Different Light Intensities under a Light/Dark Regime
2.3. Differences in Initial Rates of Oxygen Evolution.
3. Discussion
4. Materials and Methods
4.1. Cell Growth
4.2. H2 Production under a Light/Dark Regime
4.3. Oxygen Evolution and Consumption
4.4. Other Analytical Procedures
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gaffron, H.; Rubin, J. Fermentative and photochemical production of hydrogen in algae. J. Gen. Physiol. 1942, 26, 219–240. [Google Scholar] [CrossRef] [PubMed]
- Forestier, M.; King, P.; Zhang, L.; Posewitz, M.; Schwarzer, S.; Happe, T.; Ghirardi, M.L.; Seibert, M. Expression of two [Fe]-hydrogenases in Chlamydomonas reinhardtii under anaerobic conditions. Eur. J. Biochem. 2003, 270, 2750–2758. [Google Scholar] [CrossRef] [PubMed]
- Happe, T.; Kaminski, A. Differential regulation of the Fe-hydrogenase during anaerobic adaptation in the green alga Chlamydomonas reinhardtii. Eur. J. Biochem. 2002, 269, 1022–1032. [Google Scholar] [CrossRef] [PubMed]
- Melis, A.; Zhang, L.; Forestier, M.; Ghirardi, M.L.; Seibert, M. Sustained photobiological hydrogen gas production upon reversible inactivation of oxygen evolution in the green alga Chlamydomonas reinhardtii. Plant Physiol. 2000, 122, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Batyrova, K.A.; Tsygankov, A.; Kosourov, S. Sustained hydrogen photoproduction by phosphorus deprived Chlamydomonas reinhardtii cultures. Int. J. Hydrog. Energy 2012, 37, 8834–8839. [Google Scholar] [CrossRef]
- Philipps, G.; Happe, T.; Hemschemeier, A. Nitrogen deprivation results in photosynthetic hydrogen production in Chlamydomonas reinhardtii. Planta 2012, 235, 729–745. [Google Scholar] [CrossRef] [PubMed]
- Volgusheva, A.; Kukarskikh, G.; Krendeleva, T.; Rubina, A.; Mamedov, F. Hydrogen photoproduction in green algae Chlamydomonas reinhardtii under magnesium deprivation. RSC Adv. 2015, 5, 5633–5637. [Google Scholar] [CrossRef]
- Posewitz, M.C.; Smolinski, S.L.; Kanakagiri, S.; Melis, A.; Seibert, M.; Ghirardi, M.L. Hydrogen Photoproduction Is Attenuated by Disruption of an Isoamylase Gene in Chlamydomonas reinhardtii. Plant Cell 2004, 16, 2151–2163. [Google Scholar] [PubMed]
- Fouchard, S.; Hemschemeier, A.; Caruana, A.; Pruvost, K.; Legrand, J.; Happe, T.; Peltier, G.; Cournac, L.B. Autotrophic and mixotrophic hydrogen photoproduction in sulfur-deprived Chlamydomonas cells. Appl. Environ. Microbiol. 2005, 71, 6199–6205. [Google Scholar] [CrossRef] [PubMed]
- Melis, A.; Happe, T. Hydrogen production. Green algae as a source of energy. Plant Physiol. 2001, 127, 740–748. [Google Scholar] [CrossRef] [PubMed]
- Surzycki, R.; Cournac, L.; Peltier, G.; Rochaix, J.-D. Potential for hydrogen production with inducible chloroplast gene expression in Chlamydomonas. Proc. Natl. Acad. Sci. USA 2007, 104, 17548–17553. [Google Scholar] [CrossRef] [PubMed]
- Quinn, J.M.; Eriksson, M.; Moseley, J.L.; Merchant, S. Oxygen deficiency responsive gene expression in Chlamydomonas reinhardtii through a copper-sensing signal transduction pathway. Plant Physiol. 2002, 128, 463–471. [Google Scholar] [CrossRef] [PubMed]
- Kosourov, S.; Tsygankov, A.; Seibert, M.; Ghirardi, M.L. Sustained hydrogen photoproduction by Chlamydomonas reinhardtii: Effects of culture parameters. Biotechnol. Bioeng. 2002, 78, 731–740. [Google Scholar] [CrossRef] [PubMed]
- Ghirardi, M.L.; Zhang, L.; Lee, J.W.; Flynn, T.; Seibert, M.; Greenbaum, E. Microalgae: A green source of renewable H2. Trends Biotechnol. 2000, 18, 506–511. [Google Scholar] [CrossRef]
- Grobbelaar, J.U. Microalgal biomass production: Challenges and realities. Photosynth. Res. 2010, 106, 135–144. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.Y.; Liu, C.H.; Lo, Y.C.; Chang, J.S. Perspectives on cultivation strategies and photobioreactor designs for photo-fermentative hydrogen production. Bioresour. Technol. 2011, 102, 8484–8492. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.Y.; Yeh, K.L.; Aisyah, R.; Lee, D.J.; Chang, J.S. Cultivation, photobioreactor design and harvesting of microalgae for biodiesel production:a critical review. Bioresour. Technol. 2011, 102, 71–81. [Google Scholar] [CrossRef] [PubMed]
- Hase, E.; Morimura, Y.; Mihara, S.; Tamiya, H. The role of sulfur in the cell division of Chlorella. Arch. Mikrobiol. 1958, 31, 87–95. [Google Scholar] [CrossRef]
- Tsygankov, A.; Kosourov, S.; Seibert, M.; Ghirardi, M. Hydrogen photoproduction under continuous illumination by sulfur-deprived, synchronous Chlamydomonas reinhardtii cultures. Int. J. Hyd. Energy 2002, 27, 1239–1244. [Google Scholar] [CrossRef]
- Zhang, L.; Happe, T.; Melis, A. Biochemical and morphological characterization of sulfur deprived and H2 producing Chlamydomonas reinhardtii (green alga). Planta 2002, 214, 552–561. [Google Scholar] [CrossRef] [PubMed]
- Matthew, T.; Zhou, W.X.; Rupprecht, J.; Lim, L.; Thomas-Hall, S.R.; Doebbe, A.; Kruse, O.; Hankamer, B.; Marx, U.C.; Smith, S.M.; et al. The metabolome of Chlamydomonas reinhardtii following induction of anaerobic H2 production by sulfur depletion. J. Biol. Chem. 2009, 284, 23415–23425. [Google Scholar] [CrossRef] [PubMed]
- Laurinavichene, T.; Tolstygina, I.; Tsygankov, A. The effect of light intensity on hydrogen production by sulfur-deprived Chlamydomonas reinhardtii. J. Biotechnol. 2004, 114, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Gordillo, F.J.L.; Niell, F.X.; Figueroa, F.L. Non-photosynthetic enhancement of growth by high CO2 level in the nitrophilic seaweed Ulva rigida C. Agardh (Chlorophyta). Planta 2001, 213, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Kasajima, I.; Takahara, K.; Kawai-Yamada, M.; Uchimiya, H. Estimation of the relative sizes of rate constants for chlorophyll de-excitation processes through comparison of inverse fluorescence intensities. Plant Cell. Physiol. 2009, 50, 1600–1616. [Google Scholar] [CrossRef] [PubMed]
- Harris, E.H. The Chlamydomonas Sourcebook: A Comprehensive Guide to Biology and Laboratory Use; Academic Press: San Diego, CA, USA, 1989; pp. 1–780. [Google Scholar]
- Gfeller, R.P.; Gibbs, M. Fermentative metabolism of Chlamydomonas reinhardtii, I: Analysis of fermentative products from starch in dark and light. Plant Physiol. 1984, 75, 212e8. [Google Scholar] [CrossRef]
| Light Intensity (W·m−2) | Rate of Respiration, µmol [O2 (mg·Chl)−1·h−1] | Rate of Photosynthesis, µmol [O2 (mg·Chl)−1·h−1] | Chl (a + b) [mg·(L−1)] | |||
|---|---|---|---|---|---|---|
| WT | Cy6Nac2.49 | WT | Cy6Nac2.49 | WT | Cy6Nac2.49 | |
| 10 W·m−2 | 34.68 ± 0.12 | 28.44 ± 0.53 | 29.89 ± 0.82 | 2.10 ± 0.37 | 6.82 ± 0.52 | 6.68 ± 0.17 |
| 50 W·m−2 | 32.54 ± 0.87 | 28.37 ± 0.37 | 112.29 ± 0.73 | 9.31 ± 0.29 | 6.82 ± 0.52 | 6.68 ± 0.17 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Batyrova, K.; Hallenbeck, P.C. Hydrogen Production by a Chlamydomonas reinhardtii Strain with Inducible Expression of Photosystem II. Int. J. Mol. Sci. 2017, 18, 647. https://doi.org/10.3390/ijms18030647
Batyrova K, Hallenbeck PC. Hydrogen Production by a Chlamydomonas reinhardtii Strain with Inducible Expression of Photosystem II. International Journal of Molecular Sciences. 2017; 18(3):647. https://doi.org/10.3390/ijms18030647
Chicago/Turabian StyleBatyrova, Khorcheska, and Patrick C. Hallenbeck. 2017. "Hydrogen Production by a Chlamydomonas reinhardtii Strain with Inducible Expression of Photosystem II" International Journal of Molecular Sciences 18, no. 3: 647. https://doi.org/10.3390/ijms18030647
APA StyleBatyrova, K., & Hallenbeck, P. C. (2017). Hydrogen Production by a Chlamydomonas reinhardtii Strain with Inducible Expression of Photosystem II. International Journal of Molecular Sciences, 18(3), 647. https://doi.org/10.3390/ijms18030647





