Oleanolic Acid Alters Multiple Cell Signaling Pathways: Implication in Cancer Prevention and Therapy
Abstract
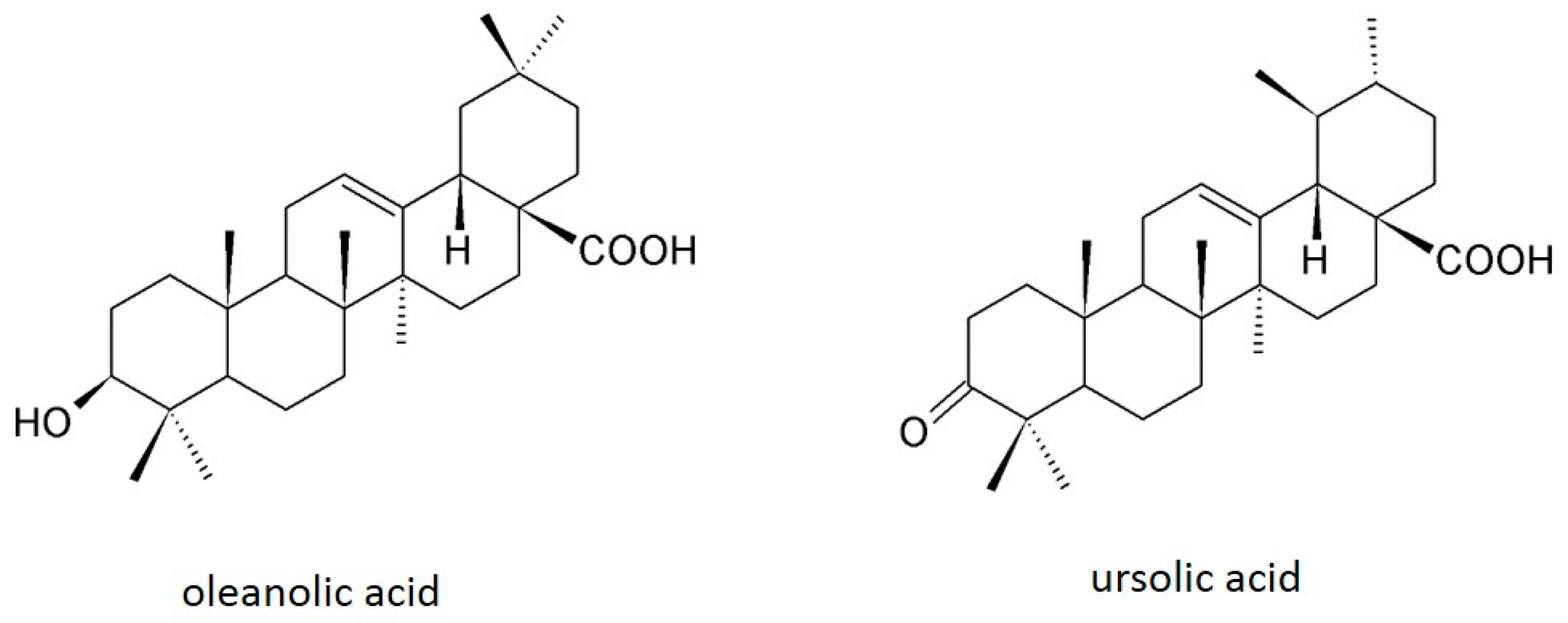
:1. Introduction
2. Chemistry and Biosynthesis
3. Sources and Bioavailability
4. Oleanolic Acid as Anticancer Agent
4.1. Oleanolic Acid Inhibited Tumor Initiation and Development
4.2. Oleanolic Acid Induced Apoptosis
4.3. Oleanolic Acid Mediated Control of TRAIL-Induced Signaling
4.4. Oleanolic Acid Inhibited Angiogenesis, Invasion and Metastasis
4.5. Oleanolic Acid Suppressed Multi-Drug Resistance Proteins
4.6. Oleanolic Acid Exerted Synergistic Activity with Chemotherapeutic Drugs
4.7. Oleanolic Acid Displayed Radiosensitizing Effects
5. Increased Selective Toxicity of Novel Oleanolic Acid Derivatives
6. Perspectives on Using Oleanolic Acid as an Adjuvant of Cancer Treatment
7. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Surh, Y.J. Cancer chemoprevention with dietary phytochemicals. Nat. Rev. Cancer 2003, 3, 768–780. [Google Scholar] [CrossRef] [PubMed]
- Hussain, S.S.; Kumar, A.P.; Ghosh, R. Food-based natural products for cancer management: Is the whole greater than the sum of the parts? Semin. Cancer Biol. 2016, 40–41, 233–246. [Google Scholar] [CrossRef] [PubMed]
- Torre, L.A.; Bray, F.; Siegel, R.L.; Ferlay, J.; Lortet-tieulent, J.; Jemal, A. Global Cancer Statistics, 2012. CA Cancer J. Clin. 2015, 65, 87–108. [Google Scholar] [CrossRef] [PubMed]
- Bandhavkar, S. Cancer stem cells: A metastasizing menace! Cancer Med. 2016, 5, 649–655. [Google Scholar] [CrossRef] [PubMed]
- Neergheen, V.S.; Bahorun, T.; Taylor, E.W.; Jen, L.S.; Aruoma, O.I. Targeting specific cell signaling transduction pathways by dietary and medicinal phytochemicals in cancer chemoprevention. Toxicology 2010, 278, 229–241. [Google Scholar] [CrossRef] [PubMed]
- Bishayee, A. Editorial: Current Advances in Cancer Prevention and Treatment by Natural Products. Curr. Pharm. Biotechnol. 2012, 13, 115–116. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Hubert, J.; Llorach, R.; Scalbert, A. The complex links between dietary phytochemicals and human health deciphered by metabolomics. Mol. Nutr. Food Res. 2009, 53, 1303–1315. [Google Scholar] [CrossRef] [PubMed]
- Bishayee, A.; Sethi, G. Bioactive natural products in cancer prevention and therapy: Progress and promise. Semin. Cancer Biol. 2016, 40–41, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Orlikova, B.; Legrand, N.; Panning, J.; Dicato, M.; Marc, D. Anti-Inflammatory and Anticancer Drugs from Nature. Adv. Nutr. Cancer 2014, 159, 123–143. [Google Scholar]
- Tuorkey, M.J. Cancer Therapy with Phytochemicals: Present and Future Perspectives. Biomed. Environ. Sci. 2015, 28, 808–819. [Google Scholar] [CrossRef]
- Russo, M.; Spagnuolo, C.; Tedesco, I.; Russo, G.L. Phytochemicals in cancer prevention and therapy: Truth or dare? Toxins 2010, 2, 517–551. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Rodriguez, R. Oleanolic Acid and Related Triterpenoids from Olives on Vascular Function: Molecular Mechanisms and Therapeutic Perspectives. Curr. Med. Chem. 2015, 22, 1414–1425. [Google Scholar] [CrossRef] [PubMed]
- Shanmugam, M.K.; Nguyen, A.H.; Kumar, A.P.; Tan, B.K.H.; Sethi, G. Targeted inhibition of tumor proliferation, survival, and metastasis by pentacyclic triterpenoids: Potential role in prevention and therapy of cancer. Cancer Lett. 2012, 320, 158–170. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Men, X.; Lei, P. Review on anti-tumor effect of triterpene acid compounds. J. Cancer Res. Ther. 2014, 10, 14–19. [Google Scholar] [PubMed]
- Parikh, N.R.; Mandal, A.; Bhatia, D.; Siveen, K.S.; Sethi, G.; Bishayee, A. Oleanane triterpenoids in the prevention and therapy of breast cancer: Current evidence and future perspectives. Phytochem. Rev. 2014, 13, 793–810. [Google Scholar] [CrossRef] [PubMed]
- Shanmugam, M.K.; Dai, X.; Kumar, A.P.; Tan, B.K.H.; Sethi, G.; Bishayee, A. Oleanolic acid and its synthetic derivatives for the prevention and therapy of cancer: Preclinical and clinical evidence. Cancer Lett. 2014, 346, 206–216. [Google Scholar] [CrossRef] [PubMed]
- Pollier, J.; Goossens, A. Oleanolic acid. Phytochemistry 2012, 77, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.-P.; Kong, T.; Zhang, L.; Tong, S.; Tian, Z.-Y.; Duan, Y.-H.; Zhang, X.-H. Solubilities of ursolic acid and oleanolic acid in four solvents from (283.2 to 329.7) K. J. Chem. Eng. Data 2011, 56, 2723–2725. [Google Scholar] [CrossRef]
- Liu, L.; Wang, X. Solubility of oleanolic acid in various solvents from (288.3 to 328.3) K. J. Chem. Eng. Data 2007, 52, 2527–2528. [Google Scholar] [CrossRef]
- Liu, J. Pharmacology of oleanolic acid and ursolic acid. J. Ethnopharmacol. 1995, 49, 57–68. [Google Scholar] [CrossRef]
- Yan, S.; Huang, C.; Wu, S.; Yin, M. Oleanolic acid and ursolic acid induce apoptosis in four human liver cancer cell lines. Toxicol. In Vitro 2010, 24, 842–848. [Google Scholar] [CrossRef] [PubMed]
- Senthil, S.; Chandramohan, G.; Pugalendi, K.V. Isomers (oleanolic and ursolic acids) differ in their protective effect against isoproterenol-induced myocardial ischemia in rats. Int. J. Cardiol. 2007, 119, 131–133. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.; Wen, X.; Sun, H. Oleanolic acid derivatives for pharmaceutical use: A patent review. Expert. Opin. Ther. Pat. 2016, 26, 643–655. [Google Scholar] [CrossRef] [PubMed]
- Chin, M.P.; Reisman, S.A.; Bakris, G.L.; O’grady, M.; Linde, P.G.; McCullough, P.A.; Packham, D.; Vaziri, N.D.; Ward, K.W.; Warnock, D.G. Mechanisms contributing to adverse cardiovascular events in patients with type 2 diabetes mellitus and stage 4 chronic kidney disease treated with bardoxolone methyl. Am. J. Nephrol. 2014, 39, 499–508. [Google Scholar] [CrossRef] [PubMed]
- Szakiel, A.; Pączkowski, C.; Pensec, F.; Bertsch, C. Fruit cuticular waxes as a source of biologically active triterpenoids. Phytochem. Rev. 2012, 11, 263–284. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Daimaru, E.; Ohnishi, M.; Kinoshita, M.; Tokuji, Y. Oleanolic acid and ursolic acid in commercial dried fruits. Food Sci. Technol. Res. 2013, 19, 113–116. [Google Scholar] [CrossRef]
- Perez-Camino, M.C.; Cert, A. Quantitative determination of hydroxyl pentacyclic triterpene acids in vegetable oils. J. Agric. Food Chem. 1999, 47, 1558–1562. [Google Scholar] [CrossRef] [PubMed]
- Juan, M.E.; Planas, J.M. 25 Effects of Pentacyclic Triterpenes from Olives on Colon Cancer. Bioact. Foods Extr. Cancer Treat. Prev. 2010, 403–413. [Google Scholar] [CrossRef]
- Kalogeropoulos, N.; Chiou, A.; Ioannou, M.; Karathanos, V.T.; Hassapidou, M.; Andrikopoulos, N.K. Nutritional evaluation and bioactive microconstituents (phytosterols, tocopherols, polyphenols, triterpenic acids) in cooked dry legumes usually consumed in the Mediterranean countries. Food Chem. 2010, 121, 682–690. [Google Scholar] [CrossRef]
- Guo, S.; Duan, J.; Qian, D.; Tang, Y.; Wu, D.; Su, S.; Wang, H.; Zhao, Y. Content variations of triterpenic acid, nucleoside, nucleobase, and sugar in jujube (Ziziphusjujuba) fruit during ripening. Food Chem. 2015, 167, 468–474. [Google Scholar] [CrossRef] [PubMed]
- Chan, T.W.D.; But, P.P.H.; Cheng, S.W.; Kwok, I.M.Y.; Lau, F.W.; Xu, H.X. Differentiation and authentication of Panax ginseng, Panaxquinquefolius, and ginseng products by using HPLC/MS. Anal. Chem. 2000, 72, 1281–1287. [Google Scholar] [CrossRef] [PubMed]
- Banik, R.M.; Pandey, D.K. Optimizing conditions for oleanolic acid extraction from Lantana camara roots using response surface methodology. Ind. Crops Prod. 2008, 27, 241–248. [Google Scholar] [CrossRef]
- Wang, C. Crataeguspinnatifida Bge. (Shanzha, Hawthorn Fruit). In Dietary Chinese Herbs; Springer: Vienna, Austria, 2015; pp. 355–361. [Google Scholar]
- Yim, T.K.; Wu, W.K.; Pak, W.F.; Ko, K.M. Hepatoprotective action of an oleanolic acid-enriched extract of Ligustrumlucidum fruits is mediated through an enhancement on hepatic glutathione regeneration capacity in mice. Phyther. Res. 2001, 15, 589–592. [Google Scholar] [CrossRef] [PubMed]
- Fang, X.; Wang, J.; Yu, X.; Zhang, G.; Zhao, J. Optimization of microwave-assisted extraction followed by RP-HPLC for the simultaneous determination of oleanolic acid and ursolic acid in the fruits of Chaenomelessinensis. J. Sep. Sci. 2010, 33, 1147–1155. [Google Scholar] [PubMed]
- Janicsák, G.; Veres, K.; Kakasy, A.Z.; Máthé, I. Study of the oleanolic and ursolic acid contents of some species of the Lamiaceae. Biochem. Syst. Ecol. 2006, 34, 392–396. [Google Scholar] [CrossRef]
- Caligiani, A.; Malavasi, G.; Palla, G.; Marseglia, A.; Tognolini, M.; Bruni, R. A simple GC–MS method for the screening of betulinic, corosolic, maslinic, oleanolic and ursolic acid contents in commercial botanicals used as food supplement ingredients. Food Chem. 2013, 136, 735–741. [Google Scholar] [CrossRef] [PubMed]
- Razboršek, M.I.; Vončina, D.B.; Doleček, V.; Vončina, E. Determination of oleanolic, betulinic and ursolic acid in Lamiaceae and mass spectral fragmentation of their trimethylsilylated derivatives. Chromatographia 2008, 67, 433–440. [Google Scholar] [CrossRef]
- Yin, M.-C.; Lin, M.-C.; Mong, M.-C.; Lin, C.-Y. Bioavailability, distribution, and antioxidative effects of selected triterpenes in mice. J. Agric. Food Chem. 2012, 60, 7697–7701. [Google Scholar] [CrossRef] [PubMed]
- Kanellos, P.T.; Kaliora, A.C.; Gioxari, A.; Christopoulou, G.O.; Kalogeropoulos, N.; Karathanos, V.T. Absorption and bioavailability of antioxidant phytochemicals and increase of serum oxidation resistance in healthy subjects following supplementation with raisins. Plant Foods Hum. Nutr. 2013, 68, 411–415. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Li, W.; Li, Y.; Xu, H.; Lv, L.; Wang, X.; Chai, Y.; Zhang, G. Simultaneous Determination of Oleanolic and Ursolic Acids in Rat Plasma by HPLC–MS: Application to a Pharmacokinetic Study After Oral Administration of Different Combinations of QingGanSanJie Decoction Extracts. J. Chromatogr. Sci. 2015, 53, 1185–1192. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Q.; Yang, X.; Du, P.; Zhang, H.; Zhang, T. Dual strategies to improve oral bioavailability of oleanolic acid: Enhancing water-solubility, permeability and inhibiting cytochrome P450 isozymes. Eur. J. Pharm. Biopharm. 2016, 99, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Jeong, D.W.; Kim, Y.H.; Kim, H.H.; Ji, H.Y.; Yoo, S.D.; Choi, W.R.; Lee, S.M.; Han, C.; Lee, H.S. Dose-linear pharmacokinetics of oleanolic acid after intravenous and oral administration in rats. Biopharm. Drug Dispos. 2007, 28, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Jäger, S.; Trojan, H.; Kopp, T.; Laszczyk, M.N.; Scheffler, A. Pentacyclictriterpene distribution in various plants-rich sources for a new group of multi-potent plant extracts. Molecules 2009, 14, 2016–2031. [Google Scholar] [CrossRef] [PubMed]
- Li, G.-L.; You, J.-M.; Song, C.-H.; Xia, L.; Zheng, J.; Suo, Y.-R. Development of a new HPLC method with precolumn fluorescent derivatization for rapid, selective and sensitive detection of triterpenic acids in fruits. J. Agric. Food Chem. 2011, 59, 2972–2979. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, T.; Zhou, B.; Gao, W.; Cao, J.; Huang, L. Chemical composition and antioxidant and anti-inflammatory potential of peels and flesh from 10 different pear varieties (Pyrus spp.). Food Chem. 2014, 152, 531–538. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Sun, Y.; Sun, Z.; Wang, X.; You, J.; Suo, Y. Determination of triterpenic acids in fruits by a novel high performance liquid chromatography method with high sensitivity and specificity. Food Chem. 2014, 146, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Romero, C.; Medina, E.; Mateo, M.; Brenes, M. Quantification of bioactive compounds in Picual and Arbequina olive leaves and fruit. J. Sci. Food Agric. 2016. [Google Scholar] [CrossRef] [PubMed]
- Liese, J.; Abhari, B.A.; Fulda, S. Smac mimetic and oleanolic acid synergize to induce cell death in human hepatocellular carcinoma cells. Cancer Lett. 2015, 365, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Song, Q.; Hu, D.; Zhuang, X.; Yu, S.; Teng, D. Oleanolic acid induced autophagic cell death in hepatocellular carcinoma cells via PI3K/Akt/mTOR and ROS-dependent pathway. Korean J. Physiol. Pharmacol. 2016, 20, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Shyu, M.H.; Kao, T.C.; Yen, G.C. Oleanolic acid and ursolic acid induce apoptosis in HuH7 human hepatocellular carcinoma cells through a mitochondrial-dependent pathway and downregulation of XIAP. J. Agric. Food Chem. 2010, 58, 6110–6118. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Bai, H.; Zhang, X.; Liu, J.; Cao, P.; Liao, N.; Zhang, W.; Wang, Z.; Hai, C. Inhibitory effect of oleanolic acid on hepatocellular carcinoma via ERK-p53-mediated cell cycle arrest and mitochondrial-dependent apoptosis. Carcinogenesis 2013, 34, 1323–1330. [Google Scholar] [CrossRef] [PubMed]
- Lucio, K.A.; Rocha Gda, G.; Moncao-Ribeiro, L.C.; Fernandes, J.; Takiya, C.M.; Gattass, C.R. Oleanolic acid initiates apoptosis in non-small cell lung cancer cell lines and reduces metastasis of a B16F10 melanoma model in vivo. PLoS ONE 2011, 6, e28596. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Liu, M.; Li, D. Oleanolic acid suppresses the proliferation of lung carcinoma cells by miR-122/Cyclin G1/MEF2D axis. Mol. Cell. Biochem. 2015, 400, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Yang, C.; Guo, C.; Li, X.; Yang, N.; Zhao, L.; Hang, H.; Liu, S.; Chu, P.; Sun, Z.; et al. SZC015, a synthetic oleanolic acid derivative, induces both apoptosis and autophagy in MCF-7 breast cancer cells. Chem. Biol. Interact. 2016, 244, 94–104. [Google Scholar] [CrossRef] [PubMed]
- Furtado, R.A.; Rodrigues, E.P.; Araujo, F.R.; Oliveira, W.L.; Furtado, M.A.; Castro, M.B.; Cunha, W.R.; Tavares, D.C. Ursolic acid and oleanolic acid suppress preneoplastic lesions induced by 1,2-dimethylhydrazine in rat colon. Toxicol. Pathol. 2008, 36, 576–580. [Google Scholar] [CrossRef] [PubMed]
- Janakiram, N.B.; Indranie, C.; Malisetty, S.V.; Jagan, P.; Steele, V.E.; Rao, C.V. Chemoprevention of colon carcinogenesis by oleanolic acid and its analog in male F344 rats and modulation of COX-2 and apoptosis in human colon HT-29 cancer cells. Pharm. Res. 2008, 25, 2151–2157. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Wei, L.; Shen, A.; Chu, J.; Lin, J.; Peng, J. Oleanolic acid modulates multiple intracellular targets to inhibit colorectal cancer growth. Int. J. Oncol. 2015, 47, 2247–2254. [Google Scholar] [CrossRef] [PubMed]
- Mu, D.W.; Guo, H.Q.; Zhou, G.B.; Li, J.Y.; Su, B. Oleanolic acid suppresses the proliferation of human bladder cancer by Akt/mTOR/S6K and ERK1/2 signaling. Int. J. Clin. Exp. Pathol. 2015, 8, 13864–13870. [Google Scholar] [PubMed]
- Li, X.; Song, Y.; Zhang, P.; Zhu, H.; Chen, L.; Xiao, Y.; Xing, Y. Oleanolic acid inhibits cell survival and proliferation of prostate cancer cells in vitro and in vivo through the PI3K/Akt pathway. Tumour Biol. 2016, 37, 7599–7613. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Liu, H.; Liu, M.; Wu, N.; Zhao, J.; Xiao, L.; Han, L.; Chu, E.; Lin, X. Oleanolic acid potentiates the antitumor activity of 5-fluorouracil in pancreatic cancer cells. Oncol. Rep. 2012, 28, 1339–1345. [Google Scholar] [PubMed]
- Wei, J.; Liu, M.; Liu, H.; Wang, H.; Wang, F.; Zhang, Y.; Han, L.; Lin, X. Oleanolic acid arrests cell cycle and induces apoptosis via ROS-mediated mitochondrial depolarization and lysosomal membrane permeabilization in human pancreatic cancer cells. J. Appl. Toxicol. 2013, 33, 756–765. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.; Xu, Z.; Wang, Y.; Sun, B.; Song, Z.; Yang, B.; Liu, X.; Lin, Y.; Peng, J.; Han, G.; et al. Anticancer effect of SZC017, a novel derivative of oleanolic acid, on human gastric cancer cells. Oncol. Rep. 2016, 35, 1101–1108. [Google Scholar] [CrossRef] [PubMed]
- Li, H.F.; Wang, X.A.; Xiang, S.S.; Hu, Y.P.; Jiang, L.; Shu, Y.J.; Li, M.L.; Wu, X.S.; Zhang, F.; Ye, Y.Y.; et al. Oleanolic acid induces mitochondrial-dependent apoptosis and G0/G1 phase arrest in gallbladder cancer cells. Drug Des. Dev. Ther. 2015, 9, 3017–3030. [Google Scholar] [CrossRef] [PubMed]
- Hua, Y.; Zhang, Z.; Li, J.; Li, Q.; Hu, S.; Li, J.; Sun, M.; Cai, Z. Oleanolic acid derivative Dex-OA has potent anti-tumor and anti-metastatic activity on osteosarcoma cells in vitro and in vivo. Investig. New Drugs 2011, 29, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Li, H.; Chen, D.; Ni, J.; Kang, Y.; Wang, S. Oleanolic acid induces apoptosis in human leukemia cells through caspase activation and poly(ADP-ribose) polymerase cleavage. Acta Biochim. Biophys. Sin. 2007, 39, 803–809. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, Y.; Komohara, Y.; Kudo, R.; Tsurushima, K.; Ohnishi, K.; Ikeda, T.; Takeya, M. Oleanolic acid inhibits macrophage differentiation into the M2 phenotype and glioblastoma cell proliferation by suppressing the activation of STAT3. Oncol. Rep. 2011, 26, 1533–1537. [Google Scholar] [PubMed]
- Guo, G.; Yao, W.; Zhang, Q.; Bo, Y. Oleanolic acid suppresses migration and invasion of malignant glioma cells by inactivating MAPK/ERK signaling pathway. PLoS ONE 2013, 8, e72079. [Google Scholar]
- Tokuda, H.; Ohigashi, H.; Koshimizu, K.; Ito, Y. Inhibitory effects of ursolic and oleanolic acid on skin tumor promotion by 12-O-tetradecanoylphorbol-13-acetate. Cancer Lett. 1986, 33, 279–285. [Google Scholar] [CrossRef]
- Oguro, T.; Liu, J.; Klaassen, C.D.; Yoshida, T. Inhibitory effect of oleanolic acid on 12-O-tetradecanoylphorbol-13-acetate-induced gene expression in mouse skin. Toxicol. Sci. 1998, 45, 88–93. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.Y.; Huang, H.Y.; Wu, Y.L. Anticancer and apoptotic activities of oleanolic acid are mediated through cell cycle arrest and disruption of mitochondrial membrane potential in HepG2 human hepatocellular carcinoma cells. Mol. Med. Rep. 2015, 12, 5012–5018. [Google Scholar] [PubMed]
- Thomas, M.; Deiters, A. MicroRNA miR-122 as a therapeutic target for oligonucleotides and small molecules. Curr. Med. Chem. 2013, 20, 3629–3640. [Google Scholar] [CrossRef] [PubMed]
- Nie, H.; Wang, Y.; Qin, Y.; Gong, X.G. Oleanolic acid induces autophagic death in human gastric cancer cells in vitro and in vivo. Cell Biol. Int. 2016, 40, 770–778. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zheng, L.; Wu, N.; Ma, L.; Zhong, J.; Liu, G.; Lin, X. Oleanolic acid induces metabolic adaptation in cancer cells by activating the AMP-activated protein kinase pathway. J. Agric. Food. Chem. 2014, 62, 5528–5537. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Ma, L.; Chen, X.; Wang, J.; Yu, T.; Gong, Y.; Ma, A.; Zheng, L.; Liang, H. ERK inhibition sensitizes cancer cells to oleanolic acid-induced apoptosis through ERK/Nrf2/ROS pathway. Tumour Biol. 2016, 37, 8181–8187. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, H.; Sugawara, K.; Saito, M.; Saito, S.; Murakami, S.; Miyata, N.; Kawashima, A.; Morimoto, S.; Gao, N.; Zhang, X.; et al. In vitro TGF-β1 antagonistic activity of ursolic and oleanolic acids isolated from Clerodendranthus spicatus. Planta Med. 2003, 69, 673–675. [Google Scholar] [PubMed]
- Akhurst, R.J.; Hata, A. Targeting the TGF β signalling pathway in disease. Nat. Rev. Drug Discov. 2012, 11, 790–811. [Google Scholar] [CrossRef] [PubMed]
- Murakami, S.; Takashima, H.; Sato-Watanabe, M.; Chonan, S.; Yamamoto, K.; Saitoh, M.; Saito, S.; Yoshimura, H.; Sugawara, K.; Yang, J.; et al. Ursolic acid, an antagonist for transforming growth factor (TGF)-β1. FEBS Lett. 2004, 566, 55–59. [Google Scholar] [CrossRef] [PubMed]
- Lagadec, P.; Griessinger, E.; Nawrot, M.P.; Fenouille, N.; Colosetti, P.; Imbert, V.; Mari, M.; Hofman, P.; Czerucka, D.; Rousseau, D.; et al. Pharmacological targeting of NF-κB potentiates the effect of the topoisomerase inhibitor CPT-11 on colon cancer cells. Br. J. Cancer 2008, 98, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Chung, T.W.; Choi, H.; Lee, J.M.; Ha, S.H.; Kwak, C.H.; Abekura, F.; Park, J.Y.; Chang, Y.C.; Ha, K.T.; Cho, S.H.; et al. Oldenlandia diffusa suppresses metastatic potential through inhibiting matrix metalloproteinase-9 and intercellular adhesion molecule-1 expression via p38 and ERK1/2 MAPK pathways and induces apoptosis in human breast cancer MCF-7 cells. J. Ethnopharmacol. 2017, 195, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Willimott, S.; Barker, J.; Jones, L.A.; Opara, E.I. Apoptotic effect of Oldenlandia diffusa on the leukaemic cell line HL60 and human lymphocytes. J. Ethnopharmacol. 2007, 114, 290–299. [Google Scholar] [CrossRef] [PubMed]
- Sadava, D.; Ahn, J.; Zhan, M.; Pang, M.L.; Ding, J.; Kane, S.E. Effects of four Chinese herbal extracts on drug-sensitive and multidrug-resistant small-cell lung carcinoma cells. Cancer Chemother. Pharmacol. 2002, 49, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Wen, L.; Guo, R.; You, L.; Abbasi, A.M.; Li, T.; Fu, X.; Liu, R.H. Major triterpenoids in Chinese hawthorn “Crataegus pinnatifida” and their effects on cell proliferation and apoptosis induction in MDA-MB-231 cancer cells. Food Chem. Toxicol. 2017, 100, 149–160. [Google Scholar] [CrossRef] [PubMed]
- Mishra, T.; Arya, R.K.; Meena, S.; Joshi, P.; Pal, M.; Meena, B.; Upreti, D.K.; Rana, T.S.; Datta, D. Isolation, Characterization and Anticancer Potential of Cytotoxic Triterpenes from Betula utilis Bark. PLoS ONE 2016, 11, e0159430. [Google Scholar] [CrossRef] [PubMed]
- Konopleva, M.; Tsao, T.; Estrov, Z.; Lee, R.M.; Wang, R.Y.; Jackson, C.E.; McQueen, T.; Monaco, G.; Munsell, M. The synthetic triterpenoid 2-cyano-3,12-dioxooleana-1,9-dien-28-oic acid induces caspase-dependent and -independent apoptosis in acute myelogenous leukemia. Cancer Res. 2004, 64, 7927–7935. [Google Scholar] [CrossRef] [PubMed]
- Chakravarti, B.; Maurya, R.; Siddiqui, J.A.; Bid, H.K.; Rajendran, S.M.; Yadav, P.P.; Konwar, R. In vitro anti-breast cancer activity of ethanolic extract of Wrightia tomentosa: Role of pro-apoptotic effects of oleanolic acid and urosolic acid. J. Ethnopharmacol. 2012, 142, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Rui, L.X.; Shu, S.Y.; Jun, W.J.; Mo, C.Z.; Wu, S.Z.; Min, L.S.; Yuan, L.; Yong, P.J.; Cheng, S.Z.; Sheng, W.S.; et al. The dual induction of apoptosis and autophagy by SZC014, a synthetic oleanolic acid derivative, in gastric cancer cells via NF-κB pathway. Tumour Biol. 2016, 37, 5133–5144. [Google Scholar] [CrossRef] [PubMed]
- Zou, W.; Liu, X.; Yue, P.; Zhou, Z.; Sporn, M.B.; Lotan, R.; Khuri, F.R.; Sun, S.Y. c-Jun NH2-terminal kinase-mediated up-regulation of death receptor 5 contributes to induction of apoptosis by the novel synthetic triterpenoid methyl-2-cyano-3,12-dioxooleana-1, 9-dien-28-oate in human lung cancer cells. Cancer Res. 2004, 64, 7570–7578. [Google Scholar] [CrossRef] [PubMed]
- Lange, M.; Abhari, B.A.; Hinrichs, T.M.; Fulda, S.; Liese, J. Identification of a novel oxidative stress induced cell death by Sorafenib and oleanolic acid in human hepatocellular carcinoma cells. Biochem. Pharmacol. 2016, 118, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wu, N.; Ma, L.N.; Zhong, J.T.; Liu, G.; Zheng, L.H.; Lin, X.K. p38 MAPK signaling mediates mitochondrial apoptosis in cancer cells induced by oleanolic acid. Asian Pac. J. Cancer Prev. 2014, 15, 4519–4525. [Google Scholar] [CrossRef] [PubMed]
- Amara, S.; Zheng, M.; Tiriveedhi, V. Oleanolic Acid Inhibits High Salt-Induced Exaggeration of Warburg-like Metabolism in Breast Cancer Cells. Cell Biochem. Biophys. 2016, 74, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wu, N.; Ma, L.; Liu, M.; Liu, G.; Zhang, Y.; Lin, X. Oleanolic acid suppresses aerobic glycolysis in cancer cells by switching pyruvate kinase type M isoforms. PLoS ONE 2014, 9, e91606. [Google Scholar] [CrossRef] [PubMed]
- Walczak, H.; Bouchon, A.; Stahl, H.; Krammer, P.H. Tumor necrosis factor-related apoptosis-inducing ligand retains its apoptosis-inducing capacity on Bcl-2-or Bcl-xL-overexpressing chemotherapy-resistant tumor cells. Cancer Res. 2000, 60, 3051–3057. [Google Scholar] [PubMed]
- Pan, G.; Ni, J.; Wei, Y.F.; Yu, G.L.; Gentz, R.; Dixit, V.M. An antagonist decoy receptor and a death domain-containing receptor for TRAIL. Science 1997, 277, 815–818. [Google Scholar] [CrossRef] [PubMed]
- Lemke, J.V.; Von Karstedt, S.; Zinngrebe, J.; Walczak, H. Getting TRAIL back on track for cancer therapy. Cell Death Differ. 2014, 21, 1350–1364. [Google Scholar] [CrossRef] [PubMed]
- Koornstra, J.J.; Kleibeuker, J.H.; van Geelen, C.M.; Rijcken, F.E.; Hollema, H.; de Vries, E.G.; de Jong, S. Expression of TRAIL (TNF-related apoptosis-inducing ligand) and its receptors in normal colonic mucosa, adenomas, and carcinomas. J. Pathol. 2003, 200, 327–335. [Google Scholar] [CrossRef] [PubMed]
- Twomey, J.D.; Kim, S.R.; Zhao, L.; Bozza, W.P.; Zhang, B. Spatial dynamics of TRAIL death receptors in cancer cells. Drug Resist. Updat. 2015, 19, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Boatright, K.M.; Salvesen, G.S. Mechanisms of caspase activation. Curr. Opin. Cell Biol. 2003, 15, 725–731. [Google Scholar] [CrossRef] [PubMed]
- Salvesen, G.S.; Dixit, V.M. Caspases: Intracellular signaling by proteolysis. Cell 1997, 91, 443–446. [Google Scholar] [CrossRef]
- Kominami, K.; Nakabayashi, J.; Nagai, T.; Tsujimura, Y.; Chiba, K.; Kimura, H.; Miyawaki, A.; Sawasaki, T.; Yokota, H.; Manabe, N.; et al. The molecular mechanism of apoptosis upon caspase-8 activation: Quantitative experimental validation of a mathematical model. Biochim. Biophys. Acta (BBA) Mol. Cell Res. 2012, 1823, 1825–1840. [Google Scholar] [CrossRef] [PubMed]
- Kantari, C.; Walczak, H. Caspase-8 and bid: Caught in the act between death receptors and mitochondria. Biochim. Biophys. Acta (BBA) Mol. Cell Res. 2011, 1813, 558–563. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Wang, X. Cytochrome c promotes caspase-9 activation by inducing nucleotide binding to Apaf-1. J. Biol. Chem. 2000, 275, 31199–31203. [Google Scholar] [CrossRef] [PubMed]
- Jelínek, M.; Balušíková, K.; Schmiedlová, M.; Němcová-Fürstová, V.; Šrámek, J.; Stančíková, J.; Zanardi, I.; Ojima, I.; Kovář, J. The role of individual caspases in cell death induction by taxanes in breast cancer cells. Cancer Cell Int. 2015, 15, 8. [Google Scholar] [CrossRef] [PubMed]
- Yoo, K.H.; Park, J.H.; Cui, E.J.; Kim, K.I.; Kim, J.Y.; Kim, J.; Hong, S.G.; Baek, N.I.; Chung, I.S. 3-O-acetyloleanolic acid induces apoptosis in human colon carcinoma HCT-116 cells. Phytother. Res. 2012, 26, 1541–1546. [Google Scholar] [PubMed]
- Ravanan, P.; Sano, R.; Talwar, P.; Ogasawara, S.; Matsuzawa, S.; Cuddy, M.; Singh, S.K.; Rao, G.S.; Kondaiah, P.; Reed, J.C. Synthetic triterpenoidcyanoenone of methyl boswellate activates intrinsic, extrinsic, and endoplasmic reticulum stress cell death pathways in tumor cell lines. Mol. Cancer Ther. 2011, 10, 1635–1643. [Google Scholar] [CrossRef] [PubMed]
- Zou, W.; Yue, P.; Khuri, F.R.; Sun, S.Y. Coupling of endoplasmic reticulum stress to CDDO-Me-induced up-regulation of death receptor 5 via a CHOP-dependent mechanism involving JNK activation. Cancer Res. 2008, 68, 7484–7492. [Google Scholar] [CrossRef] [PubMed]
- Riccioni, R.; Senese, M.; Diverio, D.; Riti, V.; Mariani, G.; Boe, A.; LoCoco, F.; Foà, R.; Peschle, C.; Sporn, M.; et al. Resistance of acute myeloid leukemic cells to the triterpenoid CDDO-Imidazolide is associated with low caspase-8 and FADD levels. Leuk. Res. 2008, 32, 1244–1258. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Lin, J.; Sun, G.; Wei, L.; Shen, A.; Zhang, M.; Peng, J. Oleanolic acid inhibits colorectal cancer angiogenesis in vivo and in vitro via suppression of STAT3 and Hedgehog pathways. Mol. Med. Rep. 2016, 13, 5276–5282. [Google Scholar] [CrossRef] [PubMed]
- Elsayed, H.E.; Akl, M.R.; Ebrahim, H.Y.; Sallam, A.A.; Haggag, E.G.; Kamal, A.M.; El Sayed, K.A. Discovery, optimization, and pharmacophore modeling of oleanolic acid and analogues as breast cancer cell migration and invasion inhibitors through targeting BRK/Paxillin/Rac1 axis. Chem. Biol. Drug Des. 2015, 85, 231–243. [Google Scholar] [CrossRef] [PubMed]
- Shan, J.Z.; Xuan, Y.Y.; Ruan, S.Q.; Sun, M. Proliferation-inhibiting and apoptosis-inducing effects of ursolic acid and oleanolic acid on multi-drug resistance cancer cells in vitro. Chin. J. Integr. Med. 2011, 17, 607–611. [Google Scholar] [CrossRef] [PubMed]
- Braga, F.; Ayres-Saraiva, D.; Gattass, C.R.; Capella, M.A. Oleanolic acid inhibits the activity of the multidrug resistance protein ABCC1 (MRP1) but not of the ABCB1 (P-glycoprotein): Possible use in cancer chemotherapy. Cancer Lett. 2007, 248, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, J.; Castilho, R.O.; da Costa, M.R.; Wagner-Souza, K.; Coelho Kaplan, M.A.; Gattass, C.R. Pentacyclictriterpenes from Chrysobalanaceae species: Cytotoxicity on multidrug resistant and sensitive leukemia cell lines. Cancer Lett. 2003, 190, 165–169. [Google Scholar] [CrossRef]
- Wang, J.; Yu, M.; Xiao, L.; Xu, S.; Yi, Q.; Jin, W. Radiosensitizing effect of oleanolic acid on tumor cells through the inhibition of GSH synthesis in vitro. Oncol. Rep. 2013, 30, 917–924. [Google Scholar] [PubMed]
- Bednarczyk-Cwynar, B.; Ruszkowski, P.; Bobkiewicz-Kozlowska, T.; Zaprutko, L. Oleanolic Acid A-lactams Inhibit the Growth of HeLa, KB, MCF-7 and Hep-G2 Cancer Cell Lines at Micromolar Concentrations. Anticancer Agents Med. Chem. 2016, 16, 579–592. [Google Scholar] [CrossRef] [PubMed]
- Sommerwerk, S.; Heller, L.; Kuhfs, J.; Csuk, R. Urea derivates of ursolic, oleanolic and maslinic acid induce apoptosis and are selective cytotoxic for several human tumor cell lines. Eur. J. Med. Chem. 2016, 119, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Liu, Y.; Yang, Z.; Niu, R.; Gao, K.; Yang, B.; Liao, X.; Zhang, J. Solid inclusion complexes of oleanolic acid with amino-appended β-cyclodextrins (ACDs): Preparation, characterization, water solubility and anticancer activity. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 69, 68–76. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Liu, Z.; Zhang, X.; Huang, J.; Yu, X.; Li, J.; Xiong, D.; Sun, X.; Zhong, Z. Effect of a controlled-release drug delivery system made of oleanolic acid formulated into multivesicular liposomes on hepatocellular carcinoma in vitro and in vivo. Int. J. Nanomed. 2016, 11, 3111–3129. [Google Scholar]
- Man, D.K.; Casettari, L.; Cespi, M.; Bonacucina, G.; Palmieri, G.F.; Sze, S.C.; Leung, G.P.; Lam, J.K.; Kwok, P.C. Oleanolic Acid Loaded PEGylated PLA and PLGA Nanoparticles with Enhanced Cytotoxic Activity against Cancer Cells. Mol. Pharm. 2015, 12, 2112–2125. [Google Scholar] [CrossRef] [PubMed]
| Fruits | Analyzed Part | Oleanolic Acid Concentration | Method | Reference |
|---|---|---|---|---|
| Apples | Pomace | 16 µg/g·dm | HPLC-DAD | [44] |
| Skin | 28 µg/g·dm | |||
| Pomegranate | Sarocarp | nd | HPLC-DAD | [45] |
| Peel | 26.96 ± 0.93 µg/g dw | |||
| Seed | 1.12 ± 0.09 µg/g dw | |||
| Lemon | Sarocarp | nd | ||
| Peel | 0.62 ± 0.01 µg/g dw | |||
| Mandarin | Sarocarp | nd | ||
| Peel | 1.05 ± 0.04 µg/g dw | |||
| Bilberries | Whole fruit | 1679.2–2029.6 µg/g dw | GC-MS-FID | [25] |
| Pears | Skin | 164.3–3066.6 µg/g fw | HPLC-PAD | [46] |
| Pulp | 34.0–156.0 µg/g fw | |||
| Grapes | Peel | 176.2 µg/g dw | HPLC-FD | [47] |
| Persimmon | Peel | 367.7 µg/g dw | ||
| Flesh | 17.2 µg/g dw | |||
| Jujube | Pulp | 360 ± 10.7 µg/g dw | UHPLC-MS/MS | [30] |
| Olives | Skin | 3094–4356 µg/g fw | HPLC-DAD | [48] |
| Pulp | 27–29 µg/g fw | |||
| Seed | nd |
| Inhibition of tumor initiation and promotion | Cell cycle arrests [52,60,71] |
| Inhibition of PI3K/Akt/mTOR/NF-κB signaling pathway [55] | |
| Inhibition of mitogen-activated protein kinase (ERK/JNK/p38 MAPK) signaling pathways [55,68,73] | |
| Activation of AMPK signaling pathway [74] | |
| Inhibition of TGF-β by binding to its receptors [76,78] | |
| Inhibition of topoisomerase I and IIα proteins [63] | |
| Apoptosis induction | Elevation cytochrome c release [51,55] |
| Decrease anti-apoptotic Bcl-2 proteins [51,55,87,90] | |
| Increase pro-apoptotic Bax proteins [55,87,90] | |
| Up-regulation of caspases [21,51,53,61,62,66,86,91] | |
| Poly (ADP-ribose) polymerase (PARP) cleavage [55,56,57,58,59,60,61,62,63,64,65,66] | |
| Induction of autophagy [50,55,73,87] | |
| Disruption of Warburg metabolism [91,92] | |
| Induction of TRAIL mediated apoptosis [104,105,107] | |
| Inhibition of the multi-drug resistance associated proteins effects [110,111,112] | |
| Metastasis inhibition | Suppression of STAT3 and sonic hedgehog signaling pathways [67,108] |
| Downregulation of proangiogenic vascular endothelial growth factor A and basic fibroblast growth factor [108] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Žiberna, L.; Šamec, D.; Mocan, A.; Nabavi, S.F.; Bishayee, A.; Farooqi, A.A.; Sureda, A.; Nabavi, S.M. Oleanolic Acid Alters Multiple Cell Signaling Pathways: Implication in Cancer Prevention and Therapy. Int. J. Mol. Sci. 2017, 18, 643. https://doi.org/10.3390/ijms18030643
Žiberna L, Šamec D, Mocan A, Nabavi SF, Bishayee A, Farooqi AA, Sureda A, Nabavi SM. Oleanolic Acid Alters Multiple Cell Signaling Pathways: Implication in Cancer Prevention and Therapy. International Journal of Molecular Sciences. 2017; 18(3):643. https://doi.org/10.3390/ijms18030643
Chicago/Turabian StyleŽiberna, Lovro, Dunja Šamec, Andrei Mocan, Seyed Fazel Nabavi, Anupam Bishayee, Ammad Ahmad Farooqi, Antoni Sureda, and Seyed Mohammad Nabavi. 2017. "Oleanolic Acid Alters Multiple Cell Signaling Pathways: Implication in Cancer Prevention and Therapy" International Journal of Molecular Sciences 18, no. 3: 643. https://doi.org/10.3390/ijms18030643
APA StyleŽiberna, L., Šamec, D., Mocan, A., Nabavi, S. F., Bishayee, A., Farooqi, A. A., Sureda, A., & Nabavi, S. M. (2017). Oleanolic Acid Alters Multiple Cell Signaling Pathways: Implication in Cancer Prevention and Therapy. International Journal of Molecular Sciences, 18(3), 643. https://doi.org/10.3390/ijms18030643










