Cryptomphalus aspersa Mollusc Egg Extract Promotes Regenerative Effects in Human Dermal Papilla Stem Cells
Abstract
:1. Introduction
2. Results
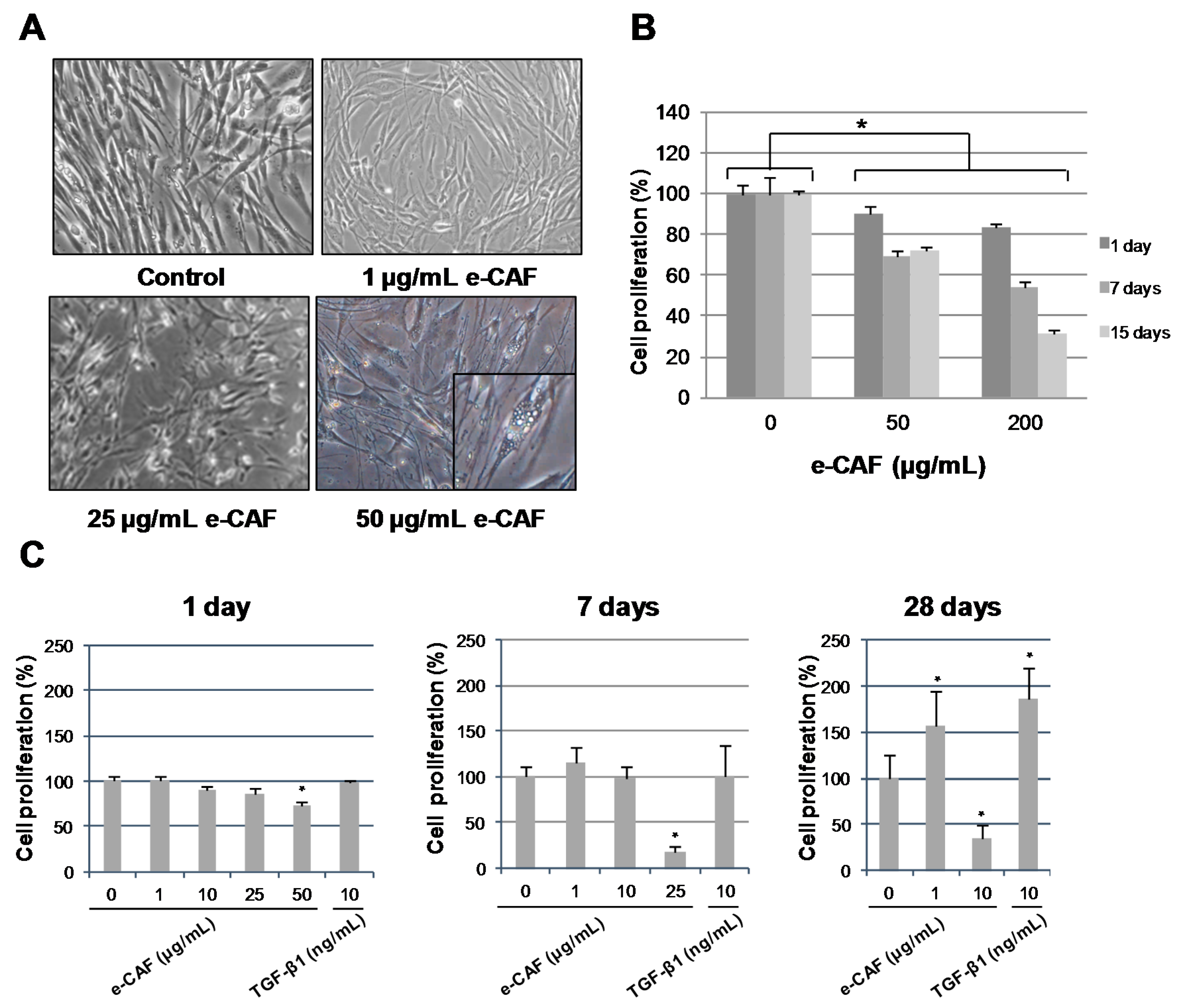
2.1. e-CAF Treatment Affects Cell Morphology and Proliferation of HHDPCs
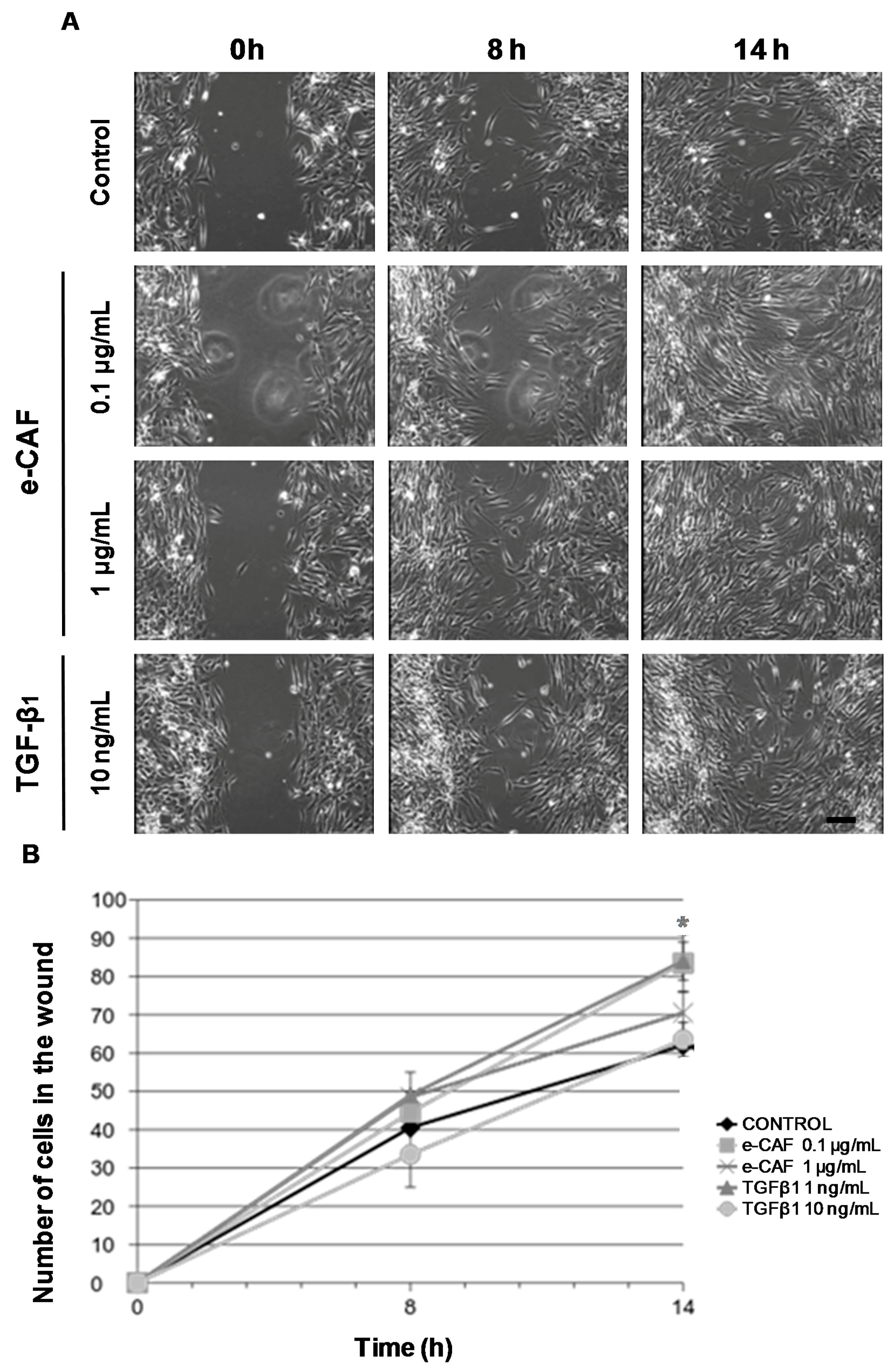
2.2. e-CAF Affects Cell Migration of HHDPCs
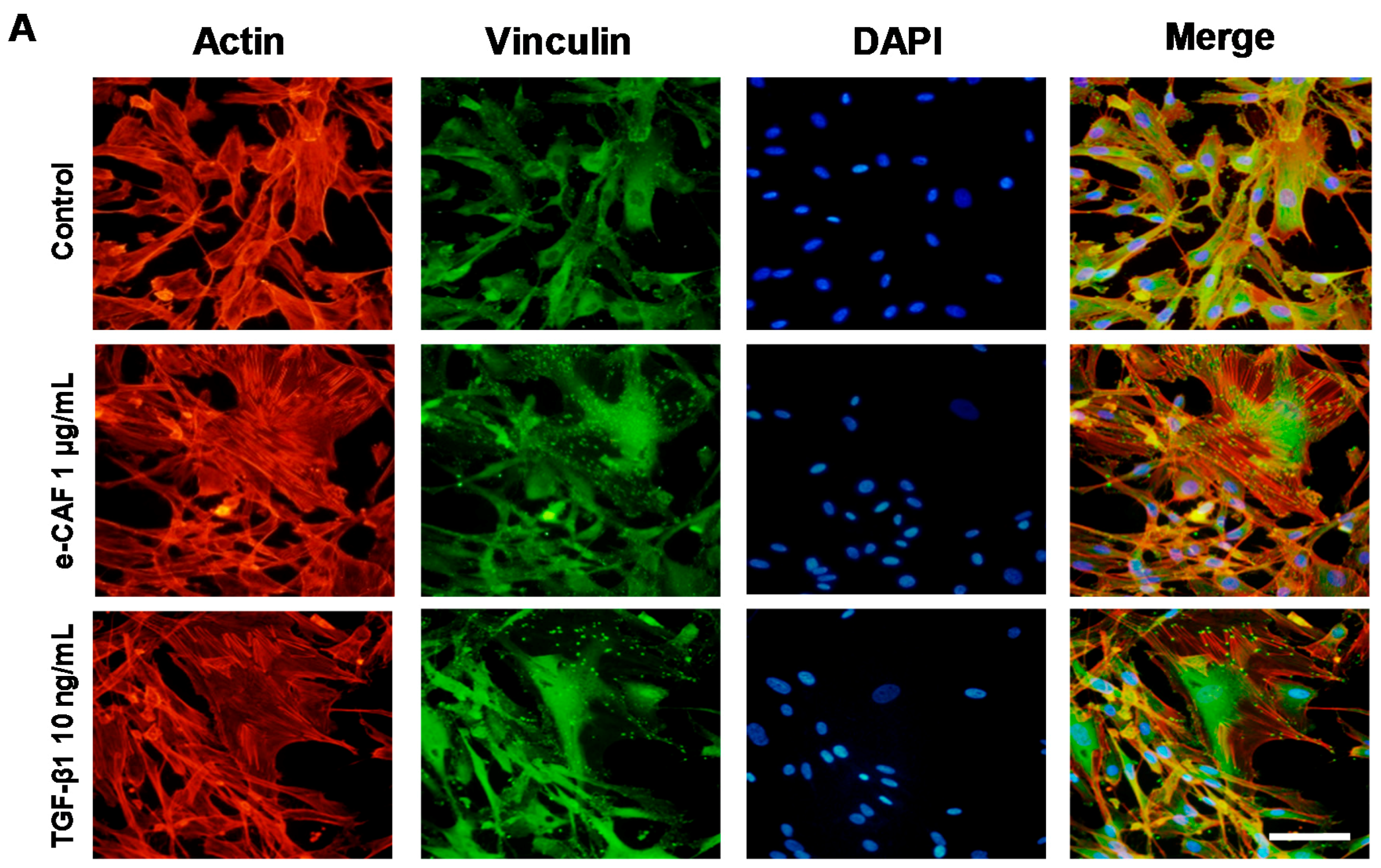
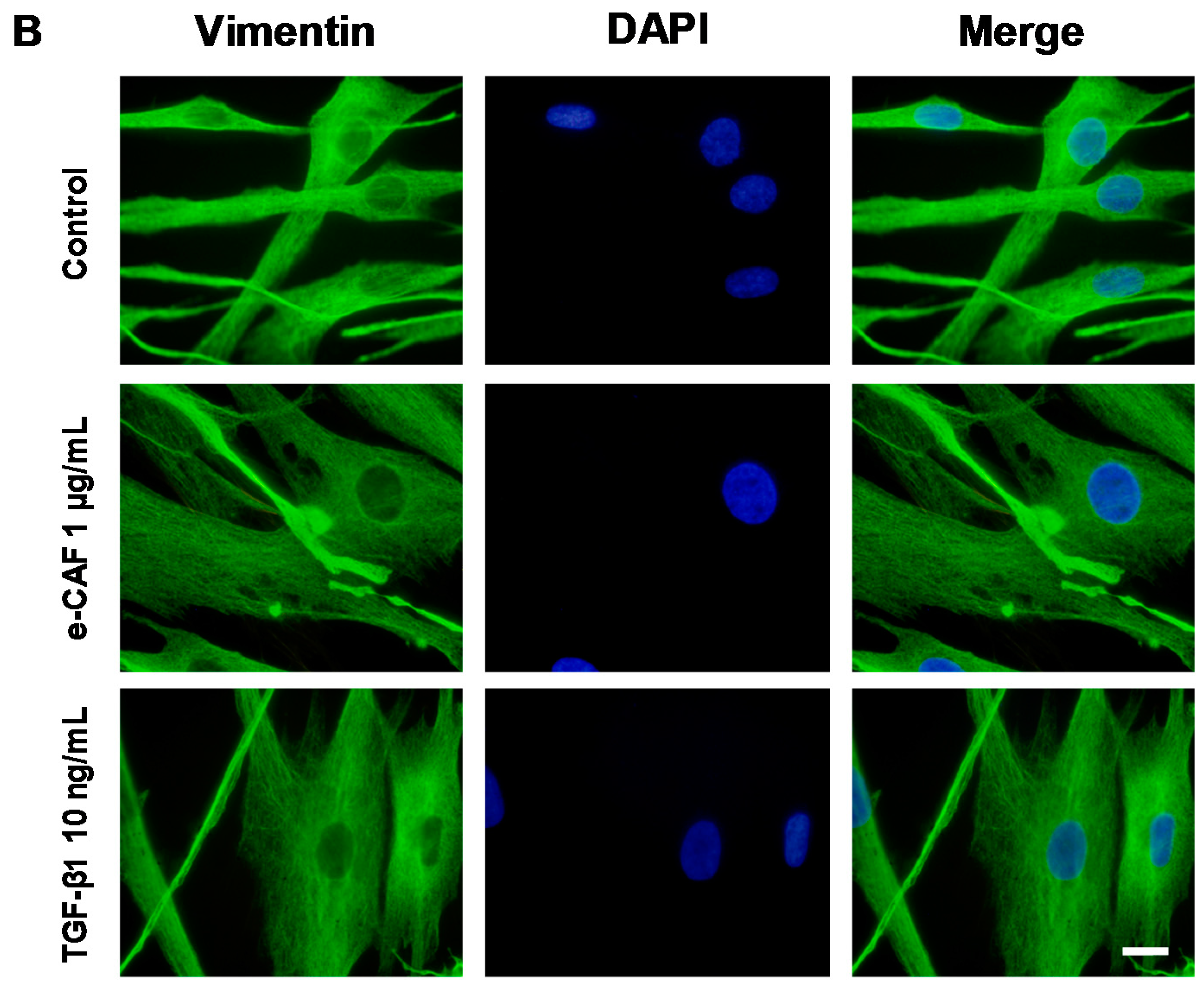
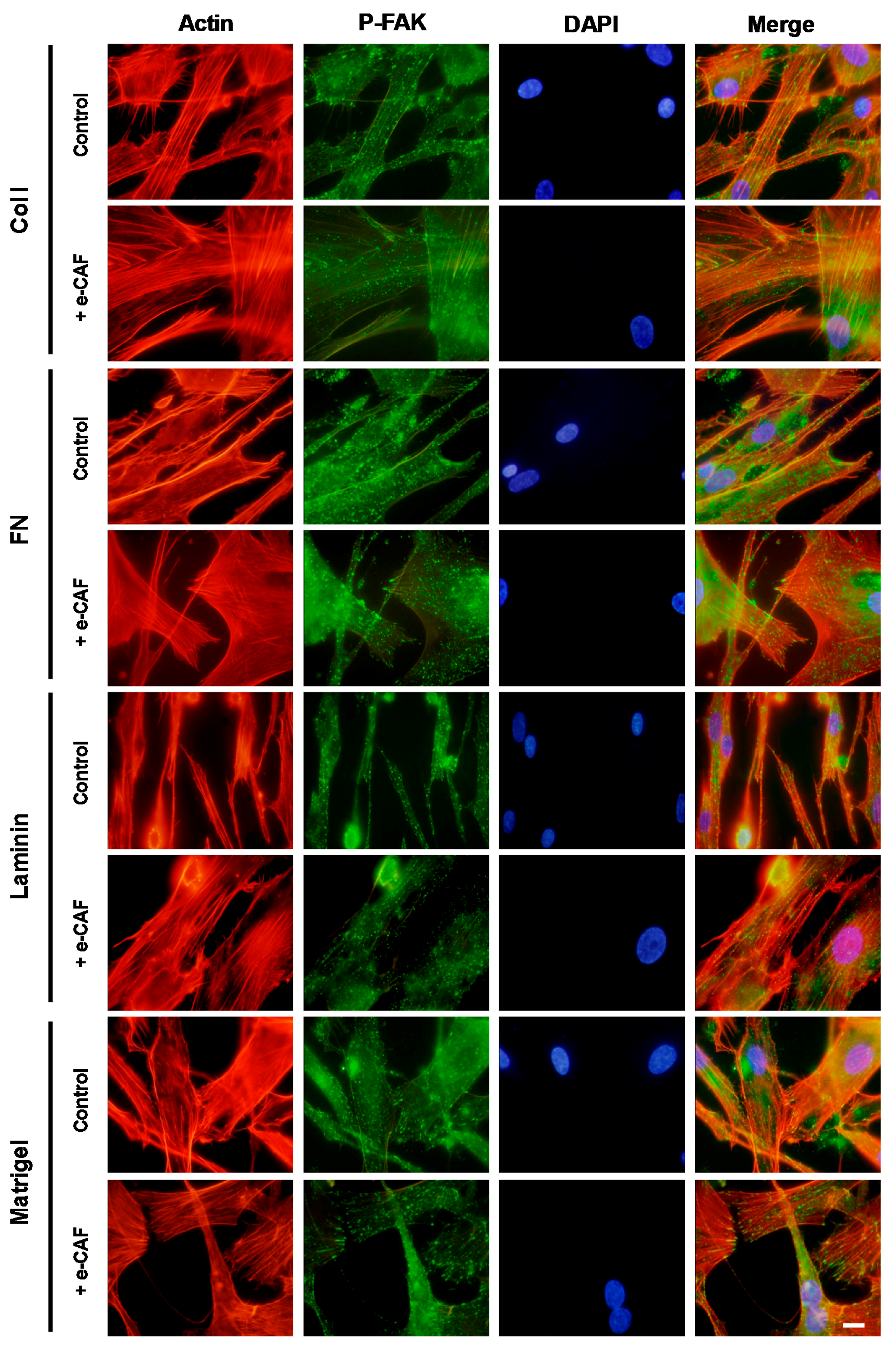
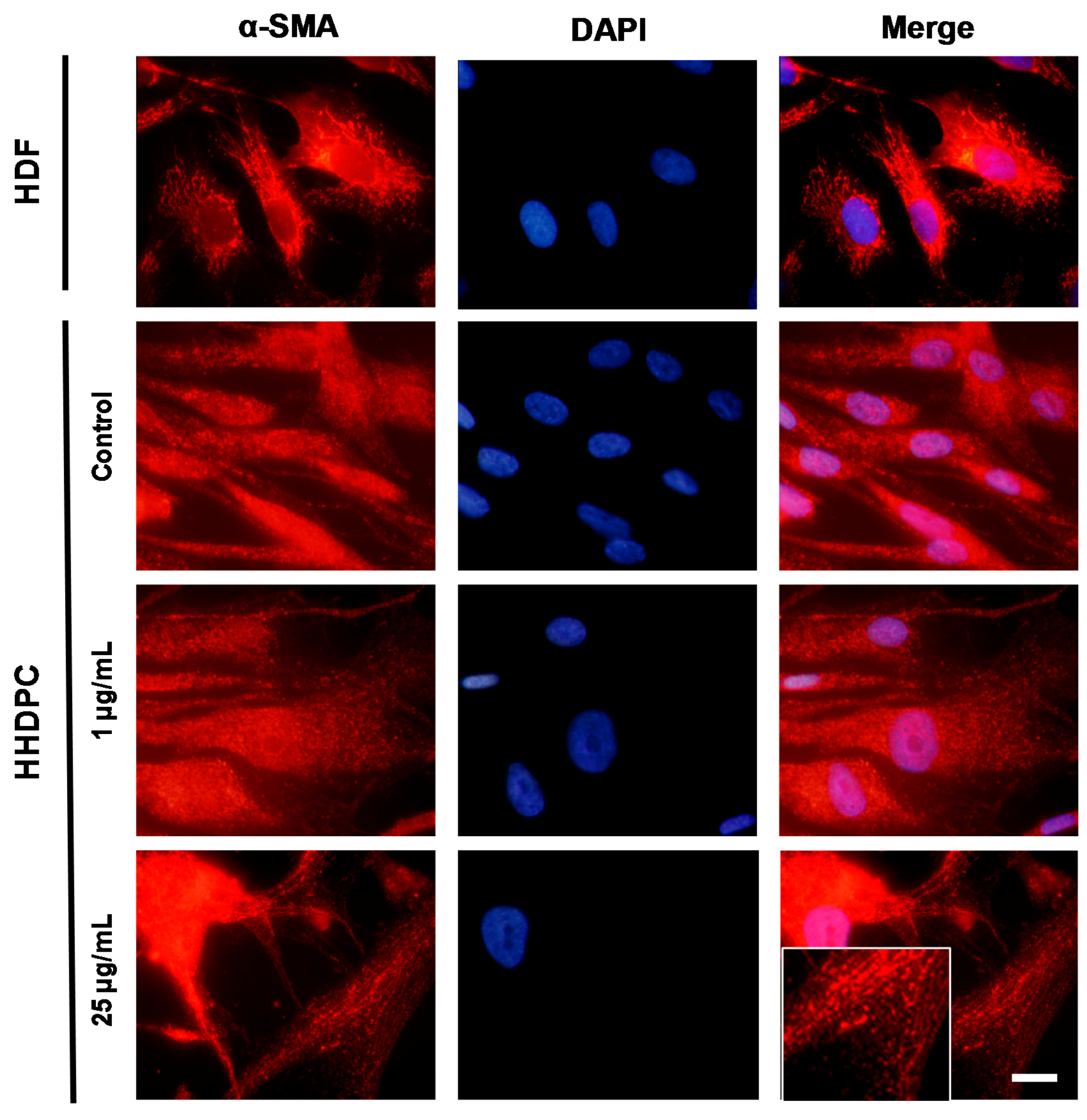
2.3. e-CAF Modulates Cytoskeleton Organization and Cell Adhesion
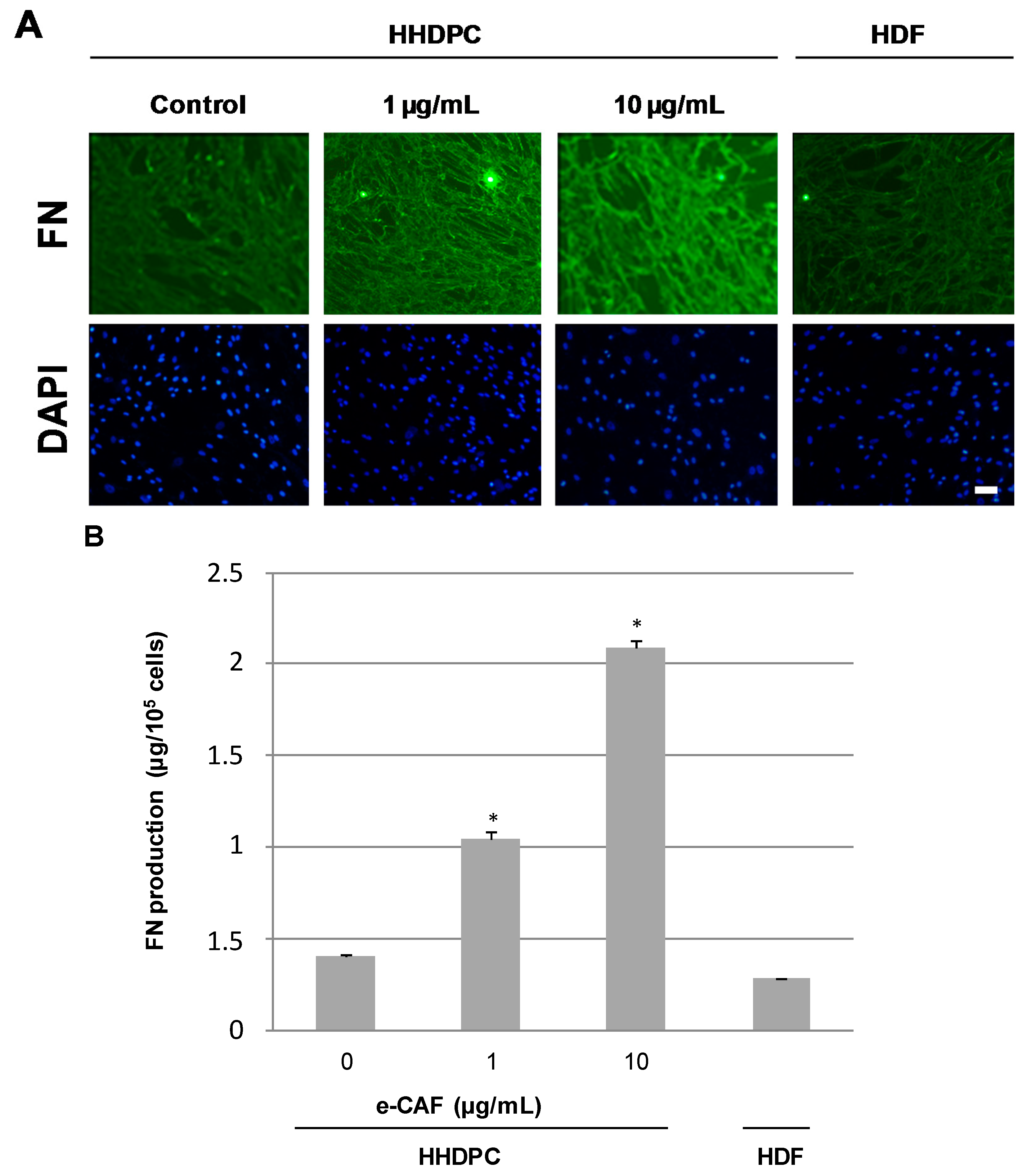
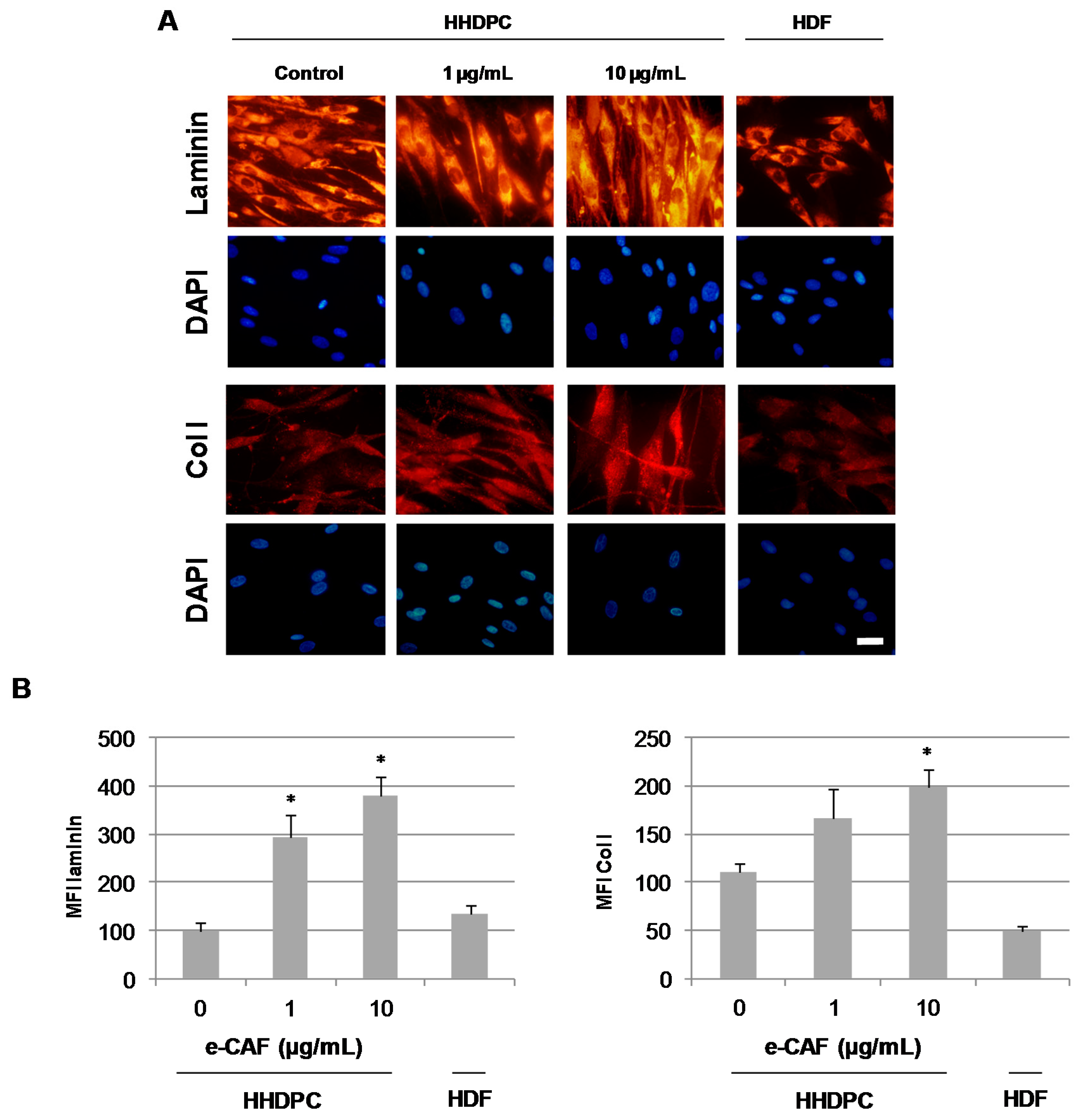
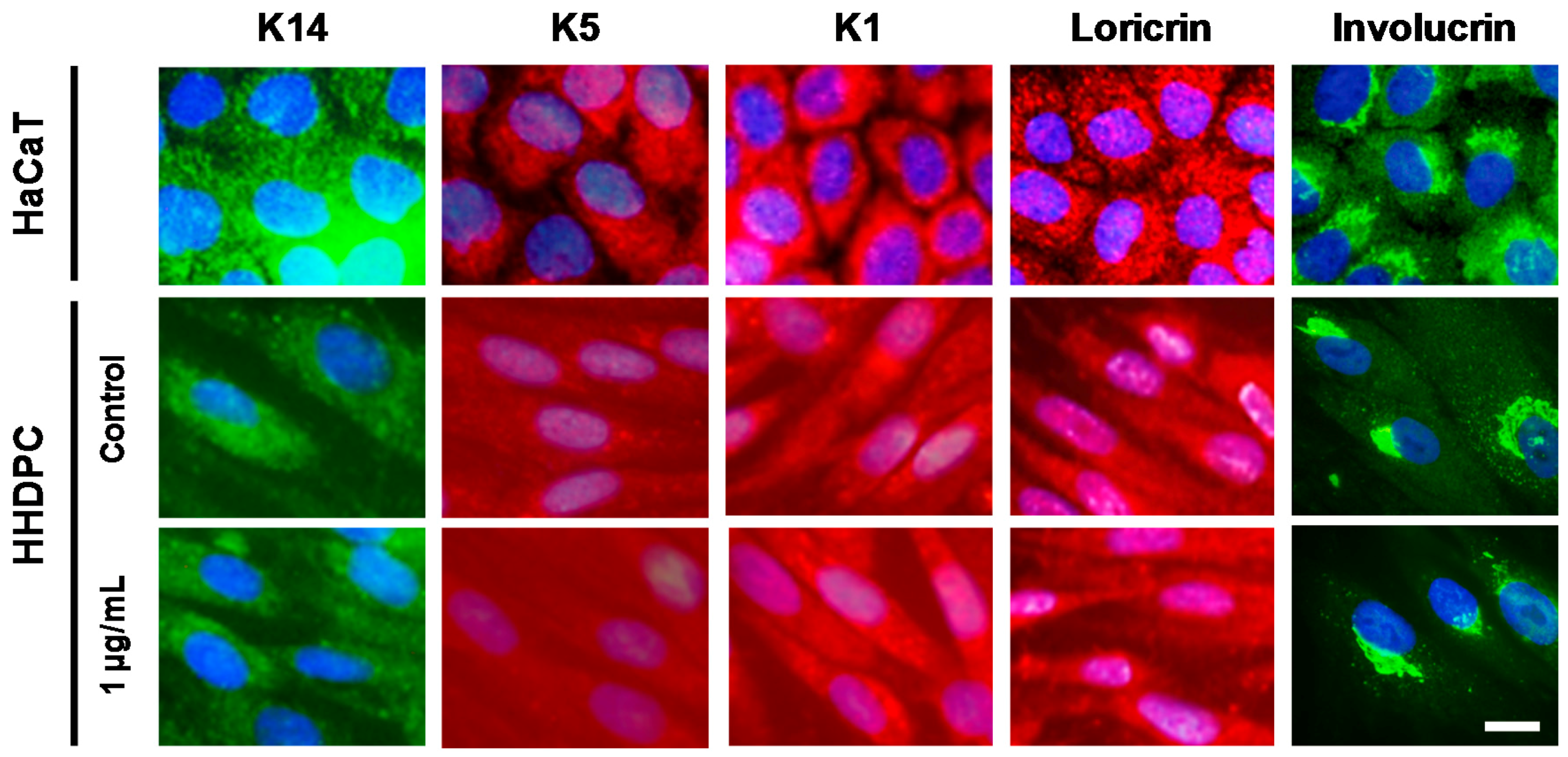
2.4. e-CAF Effects on the HHDPC Differentiation into Skin Lineages
3. Discussion
4. Materials and Methods
4.1. Cell Cultures
4.2. Reagents
4.3. MTT Assay
4.4. Migration Assay
4.5. Immunofluorescent Assays
4.6. ELISA Assay
4.7. Data Processing and Image Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- López-Otín, C.; Blasco, M.A.; Partridge, L.; Serrano, M.; Kroemer, G. The hallmarks of aging. Cell 2013, 153, 1194–1217. [Google Scholar] [CrossRef] [PubMed]
- Kohl, E.; Steinbauer, J.; Landthaler, M.; Szeimies, R.M. Skin ageing. J. Eur. Acad. Dermatol. Venereol. 2011, 25, 873–884. [Google Scholar] [CrossRef] [PubMed]
- Sanches Silveira, J.E.; Myaki Pedroso, D.M. UV light and skin aging. Rev. Environ. Health 2014, 29, 243–254. [Google Scholar] [CrossRef] [PubMed]
- Panich, U.; Sittithumcharee, G.; Rathviboon, N.; Jirawatnotai, S. Ultraviolet radiation-induced skin aging: The role of DNA damage and oxidative stress in epidermal stem cell damage mediated skin aging. Stem Cells Int. 2016, 2016, 7370642. [Google Scholar] [CrossRef] [PubMed]
- Watson, R.E.; Gibbs, N.K.; Griffiths, C.E.; Sherratt, M.J. Damage to skin extracellular matrix induced by UV exposure. Antioxid. Redox. Signal. 2014, 21, 1063–1077. [Google Scholar] [CrossRef] [PubMed]
- Valluru, M.; Staton, C.A.; Reed, M.W.R.; Brown, N.J. Transforming growth factor-β and endoglin signaling orchestrate wound healing. Front. Physiol. 2011, 2, 89. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Abbas, A.K.; Fausto, N.; Aster, J. Tissue renewal, regeneration and repair. In Pathologic Basis of Disease, 8th ed.; Elsevier: Philadelphia, PA, USA, 2010; pp. 79–110. [Google Scholar]
- Thannickal, V.J.; Lee, D.Y.; White, E.S.; Cui, Z.; Larios, J.M.; Chacon, R.; Horowitz, J.C.; Day, R.M.; Thomas, P.E. Myofibroblast differentiation by transforming growth factor-β1 is dependent on cell adhesion and integrin signaling via focal adhesion kinase. J. Biol. Chem. 2003, 278, 12384–12389. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.; Liu, J.-Q.; Yang, C.; Zheng, Z.; Zhou, Q.; Guan, H.; Su, L.-L.; Hu, D.-H. Human amniotic epithelial cells attenuate TGF-β1-induced human dermal fibroblast transformation to myofibroblasts via TGF-β1/Smad3 pathway. Cytotherapy 2016, 8, 1012–1024. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Xu, S.-W.; Kennedy, L.; Pala, D.; Chen, Y.; Eastwood, M.; Carter, D.E.; Black, C.M.; Abraham, D.J.; Leask, A. FAK is required for TGFβ-induced JNK phosphorylation in fibroblasts: Implications for acquisition of a matrix-remodeling phenotype. Mol. Biol. Cell 2007, 18, 2169–2178. [Google Scholar] [CrossRef] [PubMed]
- Rustad, K.C.; Wong, V.W.; Gurtner, G.C. The role of focal adhesion complexes in fibroblast mechanotransduction during scar formation. Differentiation 2013, 86, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Giannone, G.; Sheetz, M.P. Substrate rigidity and force define form through tyrosine phosphatase and kinase pathways. Trends Cell Biol. 2006, 16, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Case, L.B.; Baird, M.A.; Shtengel, G.; Campbell, S.L.; Hess, H.F.; Davidson, M.W.; Waterman, C.M. Molecular mechanism of vinculin activation and nanoscale spatial organization in focal adhesions. Nat. Cell Biol. 2015, 17, 880–892. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.; Nelson, E.S.; Maiers, J.L.; DeMali, K.A. New insights into vinculin function and regulation. Int. Rev. Cell Mol. Biol. 2011, 287, 191–231. [Google Scholar]
- DeMali, K.A.; Sun, X.; Bui, G.A. Force transmission at cell–cell and cell–matrix adhesions. Biochemistry 2014, 53, 7706–7717. [Google Scholar] [CrossRef] [PubMed]
- Fabi, S.; Sundaram, H. The potential of topical and injectable growth factors and cytokines for skin rejuvenation. Facial Plast. Surg. 2014, 30, 157–171. [Google Scholar] [CrossRef] [PubMed]
- Tundis, R.; Loizzo, M.R.; Bonesi, M.; Menichini, F. Potential role of natural compounds against skin aging. Curr. Med. Chem. 2015, 22, 1515–1538. [Google Scholar] [CrossRef] [PubMed]
- Brieva, A.; Guerrero, A.; Pivel, J. Antioxidative properties of a mollusc secretion (SCA): A skin protective product. Methods Find. Exp. Clin. Pharmacol. 1999, 21, 175. [Google Scholar]
- Brieva, A.; Philips, N.; Tejedor, R.; Guerrero, A.; Pivel, J.P.; Alonso-Lebrero, J.L.; Gonzalez, S. Molecular basis for the regenerative properties of a secretion of the mollusk Cryptomphalus aspersa. Pharmacol. Physiol. 2008, 21, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Tribo-Boixareu, M.; Parrado-Romero, C.; Rais, B.; Reyes, E.; Vitale-Villarejo, M.; González, S. Clinical and histological efficacy of a secretion of the mollusk Cryptomphalus aspersa in the treatment of cutaneous photoaging. J. Cosmet. Dermatol. 2009, 22, 247–252. [Google Scholar]
- Ledo, E.; Ledo, A. Treatment of acute radiodermatitis with Cryptomphalus aspersa secretion. Radioproteccion 1999, 23, 34–38. [Google Scholar]
- Tsoutsos, D.; Kakagia, D.; Tamparopoulos, K. The efficacy of helix aspersa muller extract in the healing of partial thickness burns: A novel treatment for open burn management protocols. J. Dermatol. Treat. 2009, 20, 219–222. [Google Scholar] [CrossRef] [PubMed]
- Cruz, M.C.; Sanz-Rodriguez, F.; Zamarron, A.; Reyes, E.; Carrasco, E.; Gonzalez, S.; Juarranz, A. A secretion of the mollusc Cryptomphalus aspersa promotes proliferation, migration and survival of keratinocytes and dermal fibroblasts in vitro. Int. J. Cosmet. Sci. 2012, 34, 183–189. [Google Scholar] [CrossRef]
- Fabi, S.G.; Cohen, J.L.; Peterson, J.D.; Kiripolsky, M.G.; Goldman, M.P. The effects of filtrate of the secretion of the Cryptomphalus aspersa on photoaged skin. J. Drugs Dermatol. 2013, 12, 453–457. [Google Scholar] [PubMed]
- Espada, J.; Matabuena, M.; Salazar, N.; Lucena, S.; Kourani, O.; Carrasco, E.; Calvo, M.; Rodriguez, C.; Reyes, E.; Gonzalez, S.; et al. Cryptomphalus aspersa mollusc eggs extract promotes migration and prevents cutaneous ageing in keratinocytes and dermal fibroblasts in vitro. Int. J. Cosmet. Sci. 2015, 37, 41–55. [Google Scholar] [PubMed]
- De Los Angeles, A.; Ferrari, F.; Xi, R.; Fujiwara, Y.; Benvenisty, N.; Deng, H.; Hochedlinger, K.; Jaenisch, R.; Lee, S.; Leitch, H.G.; et al. Hallmarks of pluripotency. Nature 2015, 525, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Zhou, J.; Zhang, X.; Liu, Y.; Chen, J.; Hu, B.; Song, J.; Zhang, Y. Strategies to optimize adult stem cell therapy for tissue regeneration. Int. J. Mol. Sci. 2016, 17, 982. [Google Scholar] [CrossRef] [PubMed]
- Chamberlain, G.; Fox, J.; Ashton, B.; Middleton, J. Concise review: Mesenchymal stem cells: Their phenotype, differentiation capacity, immunological features, and potential for homing. Stem Cells 2007, 25, 2739–2749. [Google Scholar] [CrossRef] [PubMed]
- Rahmani, W.; Abbasi, S.; Hagner, A.; Raharjo, E.; Kumar, R.; Hotta, A.; Magness, S.; Metzger, D.; Biernaskie, J. Hair follicle dermal stem cells regenerate the dermal sheath, repopulate the dermal papilla, and modulate hair type. Dev. Cell 2014, 31, 543–558. [Google Scholar] [CrossRef] [PubMed]
- Driskell, R.R.; Clavel, C.; Rendl, M.; Watt, F.M. Hair follicle dermal papilla cells at a glance. J. Cell Sci. 2011, 124, 1179–1182. [Google Scholar] [CrossRef] [PubMed]
- Jahoda, C.A.; Whitehouse, C.J.; Reynolds, A.J.; Hole, N. Hair follicle dermal cells differentiate into adipogenic and osteogenic lineages. Exp. Dermatol. 2003, 12, 849–859. [Google Scholar] [CrossRef] [PubMed]
- Vapniarsky, N.; Arzi, B.; Hu, J.C.; Nolta, J.A.; Athanasiou, K.A. Concise review: Human dermis as an autologous source of stem cells for tissue engineering and regenerative medicine. Stem Cells Transl. Med. 2015, 4, 1187–1198. [Google Scholar] [CrossRef] [PubMed]
- Tsai, S.Y.; Clavel, C.; Kim, S.; Ang, Y.S.; Grisanti, L.; Lee, D.F.; Kelley, K.; Rendl, M. Oct4 and klf4 reprogram dermal papilla cells into induced pluripotent stem cells. Stem Cells 2010, 28, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Gharzi, A.; Reynolds, A.J.; Jahoda, C.A.B. Plasticity of hair follicle dermal cells in wound healing and induction. Exp. Dermatol. 2003, 12, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Qi, S.H.; Liu, P.; Xie, J.L.; Shu, B.; Xu, Y.B.; Ke, C.N.; Liu, X.S.; Li, T.Z. Experimental study on repairing of nude mice skin defects with composite skin consisting of xenogeneic dermis and epidermal stem cells and hair follicle dermal papilla cells. Burns 2008, 34, 385–392. [Google Scholar] [CrossRef] [PubMed]
- Higgins, C.A.; Roger, M.; Hill, R.; Ali-Khan, A.S.; Garlick, J.; Christiano, A.M.; Jahoda, C.A.B. Multifaceted role of hair follicle dermal cells in bioengineered skins. Br. J. Dermatol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Berrier, A.L.; Yamada, K.M. Cell-matrix adhesion. J. Cell. Physiol. 2007, 213, 565–573. [Google Scholar] [CrossRef] [PubMed]
- Ciobanasu, C.; Faivre, B.; Le Clainche, C. Integrating actin dynamics, mechanotransduction and integrin activation: The multiple functions of actin binding proteins in focal adhesions. Eur. J. Cell Biol. 2013, 92, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Seo, C.H.; Furukawa, K.; Montagne, K.; Jeong, H.; Ushida, T. The effect of substrate microtopography on focal adhesion maturation and actin organization via the RhoA/ROCK pathway. Biomaterials 2011, 32, 9568–9575. [Google Scholar] [CrossRef] [PubMed]
- Balaban, N.Q.; Schwarz, U.S.; Riveline, D.; Goichberg, P.; Tzur, G.; Sabanay, I.; Mahalu, D.; Safran, S.; Bershadsky, A.; Addadi, L. Force and focal adhesion assembly: A close relationship studied using elastic micropatterned substrates. Nat. Cell Biol. 2001, 3, 466–472. [Google Scholar] [CrossRef] [PubMed]
- Young, T.-H.; Tu, H.-R.; Chan, C.-C.; Huang, Y.-C.; Yen, M.-H.; Cheng, N.-C.; Chiu, H.-C.; Lin, S.-J. The enhancement of dermal papilla cell aggregation by extracellular matrix proteins through effects on cell–substratum adhesivity and cell motility. Biomaterials 2009, 30, 5031–5040. [Google Scholar] [CrossRef] [PubMed]
- Leirós, G.J.; Kusinsky, A.G.; Drago, H.; Bossi, S.; Sturla, F.; Castellanos, M.L.; Stella, I.Y.; Balañá, M.E. Dermal papilla cells improve the wound healing process and generate hair bud-like structures in grafted skin substitutes using hair follicle stem cells. Stem Cells Transl. Med. 2014, 3, 1209–1219. [Google Scholar] [CrossRef] [PubMed]
- Hanks, S.K.; Ryzhova, L.; Shin, N.-Y.; Brabek, J. Focal adhesion kinase signaling activities and their implications in the control of cell survival and motility. Front. Biosci. 2003, 8, d982–d996. [Google Scholar] [PubMed]
- Parsons, J.T.; Martin, K.H.; Slack, J.K.; Taylor, J.M.; Weed, S.A. Focal adhesion kinase: A regulator of focal adhesion dynamics and cell movement. Oncogene 2000, 19, 5606–5613. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Hochwald, S.N. The role of FAK in tumor metabolism and therapy. Pharmacol. Ther. 2014, 142, 154–163. [Google Scholar] [CrossRef] [PubMed]
- Desai, V.D.; Hsia, H.C.; Schwarzbauer, J.E. Reversible modulation of myofibroblast differentiation in adipose-derived mesenchymal stem cells. PLoS ONE 2014, 9, e86865. [Google Scholar] [CrossRef] [PubMed]
- Li, H.-D.; Shu, B.; Xu, Y.-B.; Shi, Y.; Qi, S.-H.; Li, T.-Z.; Liu, X.-S.; Tang, J.M.; Xie, J.-L. Differentiation of rat dermal papilla cells into fibroblast-like cells induced by transforming growth factor β1. J. Cutan. Med. Surg. 2012, 16, 400–406. [Google Scholar]
- Bin, S.; Li, H.D.; Xu, Y.B.; Qi, S.H.; Li, T.Z.; Liu, X.S.; Tang, J.M.; Xie, J.L. BMP-7 attenuates TGF-β1-induced fibroblast-like differentiation of rat dermal papilla cells. Wound Repair Regen. 2013, 21, 275–281. [Google Scholar] [CrossRef] [PubMed]
| Antibody | Source | Manufacturer | Dilution |
|---|---|---|---|
| Anti-α-smooth muscle actin (α-SMA) | Rabbit (polyclonal) | Abcam® | 1:200 |
| Anti-Keratin 1 | Rabbit (polyclonal) | Abcam® | 1:100 |
| Anti-Keratin 5 | Rabbit (polyclonal) | Abcam® | 1:500 |
| Anti-Keratin 10 | Mouse (monoclonal) | DAKO® | 1:50 |
| Anti-Keratin 14 | Mouse (monoclonal) | Abcam® | 1:200 |
| Anti-Collagen I | Rabbit (polyclonal) | Santa Cruz® | 1:100 |
| Anti-Mouse IgG Alexa Fluor 488 | Goat (polyclonal) | Interchim® | 1:250 |
| Anti-Rabbit IgG Alexa Fluor 546 | Goat (monoclonal) | Interchim® | 1:250 |
| Anti-Mouse IgG Alexa Fluor 488 | Goat (polyclonal) | Interchim® | 1:250 |
| Anti-Rabbit IgG Alexa Fluor 546 | Goat (monoclonal) | Interchim® | 1:250 |
| Anti-Involucrin | Mouse (monoclonal) | Sigma-Aldrich® | 1:50 |
| Phalloidin Alexa Fluor 546 | - | Invitrogen® | 1:100 |
| Anti-phospho-FAK (pY397) | Mouse (monoclonal) | BD Transduction Laboratories® | 1:100 |
| Anti-Fibronectin | Rabbit (polyclonal) | Abcam® | 1:100 |
| Anti-Laminin | Rabbit (polyclonal) | Abcam® | 1:100 |
| Anti-Loricrin | Rabbit (polyclonal) | Sigma-Aldrich® | 1:100 |
| Anti-Vimentin | Mouse (monoclonal) | Sigma-Aldrich® | - |
| Anti-Vinculin | Mouse (monoclonal) | Sigma-Aldrich® | 1:50 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alameda, M.T.; Morel, E.; Parrado, C.; González, S.; Juarranz, Á. Cryptomphalus aspersa Mollusc Egg Extract Promotes Regenerative Effects in Human Dermal Papilla Stem Cells. Int. J. Mol. Sci. 2017, 18, 463. https://doi.org/10.3390/ijms18020463
Alameda MT, Morel E, Parrado C, González S, Juarranz Á. Cryptomphalus aspersa Mollusc Egg Extract Promotes Regenerative Effects in Human Dermal Papilla Stem Cells. International Journal of Molecular Sciences. 2017; 18(2):463. https://doi.org/10.3390/ijms18020463
Chicago/Turabian StyleAlameda, María Teresa, Esther Morel, Concepción Parrado, Salvador González, and Ángeles Juarranz. 2017. "Cryptomphalus aspersa Mollusc Egg Extract Promotes Regenerative Effects in Human Dermal Papilla Stem Cells" International Journal of Molecular Sciences 18, no. 2: 463. https://doi.org/10.3390/ijms18020463
APA StyleAlameda, M. T., Morel, E., Parrado, C., González, S., & Juarranz, Á. (2017). Cryptomphalus aspersa Mollusc Egg Extract Promotes Regenerative Effects in Human Dermal Papilla Stem Cells. International Journal of Molecular Sciences, 18(2), 463. https://doi.org/10.3390/ijms18020463








