Advances in Molecular Imaging of Locally Delivered Targeted Therapeutics for Central Nervous System Tumors
Abstract
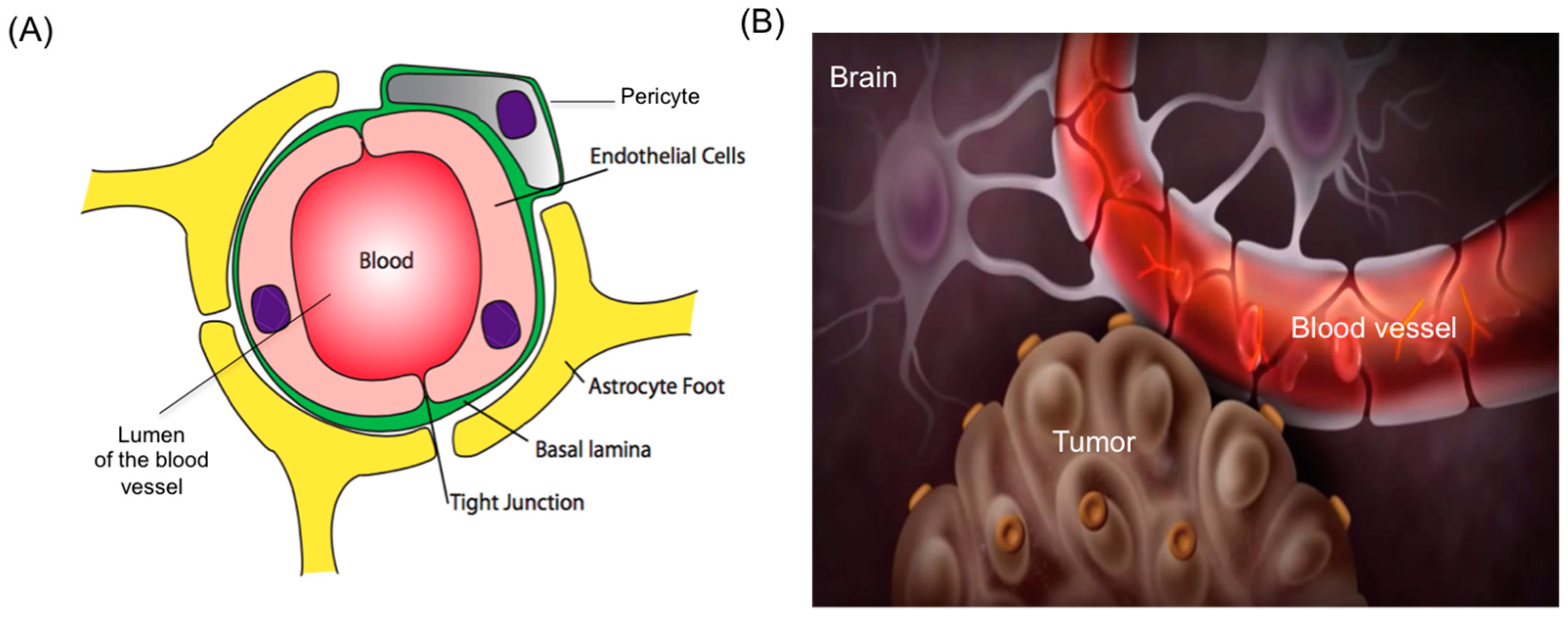
:1. Introduction
2. Strategies for CNS Delivery
2.1. Focused Ultrasound (FUS)
2.2. Osmotic Agents
2.3. Receptor-Mediated Agents
2.4. Convection Enhanced Delivery (CED)
2.5. Advances in Imaging and Theranostics
2.5.1. Antibody Carriers
2.5.2. Nanocarriers
2.5.3. Labeled Small Molecules
3. Challenges for Targeted Therapeutic Agents of Brain Tumors
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Malzkorn, B.; Reifenberger, G. Practical implications of integrated glioma classification according to the world health organization classification of tumors of the central nervous system. Curr. Opin. Oncol. 2016, 28, 494–501. [Google Scholar] [CrossRef] [PubMed]
- Castellano, A.; Falini, A. Progress in neuro-imaging of brain tumors. Curr. Opin. Oncol. 2016, 28, 484–493. [Google Scholar] [CrossRef] [PubMed]
- Cha, S. Update on brain tumor imaging: From anatomy to physiology. Am. J. Neuroradiol. 2006, 27, 475–487. [Google Scholar] [CrossRef] [PubMed]
- Appelboom, G.; Detappe, A.; LoPresti, M.; Kunjachan, S.; Mitrasinovic, S.; Goldman, S.; Chang, S.D.; Tillement, O. Stereotactic modulation of blood-brain barrier permeability to enhance drug delivery. Neuro Oncol. 2016, 18, 1601–1609. [Google Scholar] [CrossRef] [PubMed]
- Morales, J.F.; Montoto, S.S.; Fagiolino, P.; Ruiz, M.E. Current state and future perspectives in qsar models to predict blood brain barrier penetration in central nervous system drug R&D. Mini Rev. Med. Chem. 2017, 17, 247–257. [Google Scholar] [PubMed]
- De Wit, N.M.; Kooij, G.; de Vries, H.E. In vitro and ex vivo model systems to measure abc transporter activity at the blood-brain barrier. Curr. Pharm. Des. 2016, 22, 5768–5773. [Google Scholar] [CrossRef] [PubMed]
- Mahringer, A.; Fricker, G. Abc transporters at the blood-brain barrier. Expert Opin. Drug Metab. Toxicol. 2016, 12, 499–508. [Google Scholar] [CrossRef] [PubMed]
- Etame, A.B.; Diaz, R.J.; Smith, C.A.; Mainprize, T.G.; Hynynen, K.; Rutka, J.T. Focused ultrasound disruption of the blood-brain barrier: A new frontier for therapeutic delivery in molecular neurooncology. Neurosurg. Focus 2012, 32, E3. [Google Scholar] [CrossRef] [PubMed]
- Lammers, T.; Koczera, P.; Fokong, S.; Gremse, F.; Ehling, J.; Vogt, M.; Pich, A.; Storm, G.; Zandvoort, M.V.; Kiessling, F. Theranostic uspio-loaded microbubbles for mediating and monitoring blood-brain barrier permeation. Adv. Funct. Mater. 2015, 25, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Fokong, S.; Theek, B.; Wu, Z.; Koczera, P.; Appold, L.; Jorge, S.; Resch-Genger, U.; van Zandvoort, M.; Storm, G.; Kiessling, F.; et al. Image-guided, targeted and triggered drug delivery to tumors using polymer-based microbubbles. J. Control Release 2012, 163, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Zhang, Y.; Vykhodtseva, N.; Jolesz, F.A.; McDannold, N.J. The kinetics of blood brain barrier permeability and targeted doxorubicin delivery into brain induced by focused ultrasound. J. Control Release 2012, 162, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Treat, L.H.; McDannold, N.; Vykhodtseva, N.; Zhang, Y.; Tam, K.; Hynynen, K. Targeted delivery of doxorubicin to the rat brain at therapeutic levels using MRI-guided focused ultrasound. Int. J. Cancer 2007, 121, 901–907. [Google Scholar] [CrossRef]
- Shang, X.; Wang, P.; Liu, Y.; Zhang, Z.; Xue, Y. Mechanism of low-frequency ultrasound in opening blood-tumor barrier by tight junction. J. Mol. Neurosci. 2011, 43, 364–369. [Google Scholar] [CrossRef] [PubMed]
- Sheikov, N.; McDannold, N.; Sharma, S.; Hynynen, K. Effect of focused ultrasound applied with an ultrasound contrast agent on the tight junctional integrity of the brain microvascular endothelium. Ultrasound Med. Biol. 2008, 34, 1093–1104. [Google Scholar] [CrossRef] [PubMed]
- Baseri, B.; Choi, J.J.; Tung, Y.S.; Konofagou, E.E. Multi-modality safety assessment of blood-brain barrier opening using focused ultrasound and definity microbubbles: A short-term study. Ultrasound Med. Biol. 2010, 36, 1445–1459. [Google Scholar] [CrossRef] [PubMed]
- Hynyen, K.; McDannold, N.; Vykhodtseva, N.; Jolesz, F.A. Ultrasound for drug and gene delivery to the brain. Adv. Drug Deliv. Rev. 2001, 60, 1209–1217. [Google Scholar] [CrossRef] [PubMed]
- Hynyen, K.; McDannold, N.; Vykhodtseva, N.; Raymond, S.; Weissleder, R.; Jolesz, F.A.; Sheikov, N. Focal disruption of the blood–brain barrier due to 260-khz ultrasound bursts: A method for molecular imaging and targeted drug delivery. J. Neurosurg. 2006, 105, 445–454. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.Y.; Lin, G.L.; Horng, S.C.; Chang, T.K.; Wu, S.Y.; Wong, T.T.; Wang, H.E. Pulsed high-intesnity focused ultrasound enhances the relative permeability of the blood-tumor barrier in a glioma-bearing rat model. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2011, 58, 964–970. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.Y.; Teng, M.C.; Lu, M.; Liang, H.F.; Lee, Y.R.; Yen, C.C.; Liang, M.L.; Wong, T.T. Treating glioblastoma multiforme with selective high-dose liposomal doxorubicin chemotherapy induced by repeated focused ultrasound. Int. J. Nanomed. 2012, 7, 965–974. [Google Scholar] [CrossRef] [PubMed]
- Fan, Z.; Kumon, R.E.; Deng, C.X. Mechanisms of microbubble-facilitated sonoporation for drug and gene delivery. Ther. Deliv. 2014, 5, 467–486. [Google Scholar] [CrossRef] [PubMed]
- Delalande, A.; Kotopoulis, S.; Postema, M.; Midoux, P.; Pichon, C. Sonoporation: Mechanistic insights and ongoing challenges for gene transfer. Gene 2013, 525, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Geers, B.; Lentacker, I.; Alonso, A.; Sanders, N.N.; Demeester, J.; Meairs, S.; de Smedt, S.C. Elucidating the mechanisms behind sonoporation with adeno-associated virus-loaded microbubbles. Mol. Pharm. 2011, 8, 2244–2251. [Google Scholar] [CrossRef] [PubMed]
- Mehier-Humbert, S.; Bettinger, T.; Yan, F.; Guy, R.H. Ultrasound-mediated gene delivery: Kinetics of plasmid internalization and gene expression. J. Control Release 2005, 104, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Meijering, B.D.; Juffermans, L.J.; van Wamel, A.; Henning, R.H.; Zuhorn, I.S.; Emmer, M.; Versteilen, A.M.; Paulus, W.J.; van Gilst, W.H.; Kooiman, K.; et al. Ultrasound and microbubble-targeted delivery of macromolecules is regulated by induction of endocytosis and pore formation. Circ. Res. 2009, 104, 679–687. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.L.; Hua, M.Y.; Chen, P.Y.; Chu, P.C.; Yang, H.W.; Yen, T.C.; Wei, K.C. Blood-brain barrier disruption with focused ultrasound enhances delivery of chemotherapeutic drugs for glioblastoma treatment. Radiology 2010, 255, 415–425. [Google Scholar] [CrossRef] [PubMed]
- Mei, J.; Cheng, Y.; Song, Y.; Yang, Y.; Wang, F.; Liu, Y.; Wang, Z. Experimental study on targeted methotrexate delivery to the rabbit brain via magnetic resonance imaging-guided focused ultrasound. J. Ultrasound Med. 2009, 28, 871–880. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, M.; McDannold, N.; Jolesz, F.A.; Hynynen, K. Targeted delivery of antibodies through the blood-brain barrier by MRI-guided focused ultrasound. Biochem. Biophys. Res. Commun. 2006, 340, 1085–1090. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.Y.; Liu, H.L.; Hsu, P.H.; Chiang, C.S.; Tsai, C.H.; Chi, H.S.; Chen, S.Y.; Chen, Y.Y. A multitheragnostic nanobubble system to induce blood-brain barrier disruption with magnetically guided focused ultrasound. Adv. Mater. 2015, 27, 655–661. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, R.; Oda, Y.; Utoguchi, N.; Maruyama, K. Progress in the development of ultrasound-mediated gene delivery systems utilizing nano- and microbubbles. J. Control Release 2011, 149, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Chen, K.J.; Wu, T.H.; Wang, H.; Lin, W.Y.; Ohashi, M.; Chiou, P.Y.; Tseng, H.R. Photothermal effects of supramolecularly assembled gold nanoparticles for the targeted treatment of cancer cells. Angew. Chem. 2010, 49, 3777–3781. [Google Scholar] [CrossRef]
- Yang, F.; Li, Y.; Chen, Z.; Zhang, Y.; Wu, J.; Gu, N. Superparamagnetic iron oxide nanoparticle-embedded encapsulated microbubbles as dual contrast agents of magnetic resonance and ultrasound imaging. Biomaterials 2009, 30, 3882–3890. [Google Scholar] [CrossRef] [PubMed]
- Yin, T.; Wang, P.; Li, J.; Zheng, R.; Zheng, B.; Cheng, D.; Li, R.; Lai, J.; Shuai, X. Ultrasound-sensitive sirna-loaded nanobubbles formed by hetero-assembly of polymeric micelles and liposomes and their therapeutic effect in gliomas. Biomaterials 2013, 34, 4532–4543. [Google Scholar] [CrossRef] [PubMed]
- Neuwelt, E.A.; Barnett, P.A.; Bigner, D.D.; Frenkel, E.P. Effects of adrenal cortical steroids and osmotic blood-brain barrier opening on methotrexate delivery to gliomas in the rodent: The factor of the blood-brain barrier. Proc. Natl. Acad. Sci. USA 1982, 79, 4220–4223. [Google Scholar] [CrossRef]
- Bellavance, M.A.; Blanchette, M.; Fortin, D. Recent advances in blood-brain barrier disruption as a CNS delivery strategy. AAPS J. 2008, 10, 166–177. [Google Scholar] [CrossRef] [PubMed]
- Blanchette, M.; Pellerin, M.; Tremblay, L.; Lepage, M.; Fortin, D. Real-time monitoring of gadolinium diethylenetriamine penta-acetic acid during osmotic blood-brain barrier disruption using magnetic resonance imaging in normal wistar rats. Neurosurgery 2009, 65, 344–350. [Google Scholar] [CrossRef] [PubMed]
- Boockvar, J.A.; Tsiouris, A.J.; Hofstetter, C.P.; Kovanlikaya, I.; Fralin, S.; Kesavabhotla, K.; Seedial, S.M.; Pannullo, S.C.; Schwartz, T.H.; Stieg, P.; et al. Safety and maximum tolerated dose of superselective intraarterial cerebral infusion of bevacizumab after osmotic blood-brain barrier disruption for recurrent malignant glioma: Clinical article. J. Neurosurg. 2011, 114, 624–632. [Google Scholar] [CrossRef] [PubMed]
- Burkhardt, J.K.; Riina, H.; Shin, B.J.; Christos, P.; Kesavabhotla, K.; Hofstetter, C.P.; Tsiouris, A.J.; Boockvar, J.A. Intra-arterial delivery of bevacizumab after blood-brain barrier disruption for the treatment of recurrent glioblastoma: Progression-free survival and overall survival. World Neurosurg. 2012, 77, 130–134. [Google Scholar] [CrossRef] [PubMed]
- Erdlenbruch, B.; Alipour, M.; Fricker, G.; Miller, D.S.; Kugler, W.; Eibl, H.; Lakomek, M. Alkylglycerol opening of the blood-brain barrier to small and large fluorescence markers in normal and c6 glioma-bearing rats and isolated rat brain capillaries. Br. J. Pharmacol. 2003, 140, 1201–1210. [Google Scholar] [CrossRef] [PubMed]
- Erdlenbruch, B.; Kugler, W.; Schinkhof, C.; Neurath, H.; Eibl, H.; Lakomek, M. Blood-brain barrier opening with alkylglycerols: Biodistribution of 1-O-pentylglycerol after intravenous and intracarotid administration in rats. J. Drug Target 2005, 13, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Fortin, D.; Desjardins, A.; Benko, A.; Niyonsega, T.; Boudrias, M. Enhanced chemotherapy delivery by intraarterial infusion and blood-brain barrier disruption in malignant brain tumors: The sherbrooke experience. Cancer 2005, 103, 2606–2615. [Google Scholar] [CrossRef] [PubMed]
- Fortin, D.; Gendron, C.; Boudrias, M.; Garant, M.P. Enhanced chemotherapy delivery by intraarterial infusion and blood-brain barrier disruption in the treatment of cerebral metastasis. Cancer 2007, 109, 751–760. [Google Scholar] [CrossRef] [PubMed]
- Rapoport, S.I.; Robinson, P.J. Tight-junctional modification as the basis of osmotic opening of the blood-brain barrier. Ann. N. Y. Acad. Sci. 1986, 481, 250–267. [Google Scholar] [CrossRef] [PubMed]
- Shin, B.J.; Burkhardt, J.K.; Riina, H.A.; Boockvar, J.A. Superselective intra-arterial cerebral infusion of novel agents after blood-brain disruption for the treatment of recurrent glioblastoma multiforme: A technical case series. Neurosurg. Clin. N. Am. 2012, 23, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Williams, P.C.; Henner, W.D.; Roman-Goldstein, S.; Dahlborg, S.A.; Brummett, R.E.; Tableman, M.; Dana, B.W.; Neuwelt, E.A. Toxicity and efficacy of carboplatin and etoposide in conjunction with disruption of the blood-brain tumor barrier in the treatment of intracranial neoplasms. Neurosurgery 1995, 37, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Hall, W.A.; Doolittle, N.D.; Daman, M.; Bruns, P.K.; Muldoon, L.; Fortin, D.; Neuwelt, E.A. Osmotic blood-brain barrier disruption chemotherapy for diffuse pontine gliomas. J. Neurooncol. 2006, 77, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.C.; Egleton, R.D.; Davis, T.P. Mannitol opening of the blood-brain barrier: Regional variation in the permeability of sucrose, but not 86rb+ or albumin. Brain Res. 2004, 1014, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Kemper, E.M.; Boogerd, W.; Thuis, I.; Beijnen, J.H.; van Tellingen, O. Modulation of the blood-brain barrier in oncology: Therapeutic opportunities for the treatment of brain tumours? Cancer Treat. Rev. 2004, 30, 415–423. [Google Scholar] [CrossRef] [PubMed]
- Chacko, A.M.; Li, C.; Pryma, D.A.; Brem, S.; Coukos, G.; Muzykantov, V. Targeted delivery of antibody-based therapeutic and imaging agents to CNS tumors: Crossing the blood-brain barrier divide. Expert Opin. Drug Deliv. 2013, 10, 907–926. [Google Scholar] [CrossRef] [PubMed]
- Doctrow, S.R.; Abelleira, S.M.; Curry, L.A.; Heller-Harrison, R.; Kozarich, J.W.; Malfroy, B.; McCarroll, L.A.; Morgan, K.G.; Morrow, A.R.; Musso, G.F.; et al. The bradykinin analog RMP-7 increases intracellular free calcium levels in rat brain microvascular endothelial cells. J. Pharmacol. Exp. Ther. 1994, 271, 229–237. [Google Scholar] [PubMed]
- Sanovich, E.; Bartus, R.T.; Friden, P.M.; Dean, R.I.; Le, H.Q.; Brightman, M.W. Pathway across blood-brain barrier opened by the bradykinin agonist, RMP-7. Brain Res. 1995, 705, 125–135. [Google Scholar] [CrossRef]
- Emerich, D.F.; Snodgrass, P.; Dean, R.; Agostino, M.; Hasler, B.; Pink, M.; Xiong, H.; Kim, B.S.; Bartus, R.T. Enhanced delivery of carboplatin into brain tumours with intravenous cereport (RMP-7): Dramatic differences and insight gained from dosing parameters. Br. J. Cancer 1999, 80, 964–970. [Google Scholar] [CrossRef] [PubMed]
- Prados, M.D.; Schold, S.J.S.; Fine, H.A.; Jaeckle, K.; Hocherg, F.; Mechtler, L.; Fetell, M.R.; Phuphanich, S.; Feun, L.; Janus, T.J.; et al. A randomized, double-blind, placebo-controlled, phase 2 study of RMP-7 in combination with carboplatin administered intravenously for the treatment of recurrent malignant glioma. Neuro Oncol. 2003, 5, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Warren, K.; Jakacki, R.; Widemann, B.; Aikin, A.; Libucha, M.; Packer, R.; Vezina, G.; Reaman, G.; Shaw, D.; Krailo, M.; et al. Phase II trial of intravenous lobradimil and carboplatin in childhood brain tumors: A report from the children’s oncology group. Cancer Chemother. Pharmacol. 2006, 58, 343–347. [Google Scholar] [CrossRef] [PubMed]
- Kitazono, T.; Faraci, F.M.; Taguchi, H.; Heistad, D.D. Role of potassium channels in cerebral blood vessels. Stroke 1995, 26, 1713–1723. [Google Scholar] [CrossRef] [PubMed]
- Berg, T.; Koteng, O. Signaling pathways in bradykinin- and nitric oxide-induced hypotension in the normotensive rat: Role of k-channels. Br. J. Pharmacol. 1997, 121, 1113–1120. [Google Scholar] [CrossRef] [PubMed]
- Sobey, C.G.; Heistad, D.D.; Faraci, F.M. Mechanisms of bradykinin-induced cerebral vasodilatation in rats. Stroke 1997, 28, 2290–2295. [Google Scholar] [CrossRef] [PubMed]
- Ningaraj, N.S.; Rao, M.; Hashizume, K.; Asotra, K.; Black, L.K. Regulation of blood-brain tumor barrier permeability by calcium-activated potassium channels. J. Pharmacol. Exp. Ther. 2002, 301, 838–851. [Google Scholar] [CrossRef] [PubMed]
- Lonser, R.R.; Sarntinoranont, M.; Morrison, P.F.; Oldfield, E.H. Convection-enhanced delivery to the central nervous system. J. Neurosurg. 2015, 122, 697–706. [Google Scholar] [CrossRef] [PubMed]
- Laske, D.W.; Youle, R.J.; Oldfield, E.H. Tumor regression with regional distribution of the targeted toxin Tf-CRM107 in patients with malignant brain tumors. Nat. Med. 1997, 3, 1362–1368. [Google Scholar] [CrossRef] [PubMed]
- Bobo, R.H.; Laske, D.W.; Akbasak, A.; Morrisont, P.F.; Dedrickt, R.L.; Oldfield, E.H. Convection-enhanced delivery of macromolecules in the brain. Proc. Natl. Acad. Sci. USA 1994, 91, 2076–2080. [Google Scholar] [CrossRef]
- Sandberg, D.I.; Edgar, M.A.; Souweidane, M.M. Convection-enhanced delivery into the rat brainstem. J. Neurosurg. 2002, 96, 885–891. [Google Scholar] [CrossRef]
- Souweidane, M.M. Convection-enhanced delivery for diffuse intrinsic pontine glioma. J. Neurosurg. Pediatr. 2014, 13, 273–275. [Google Scholar] [CrossRef] [PubMed]
- Raghavan, R.; Brady, M.; Rodriguez-Ponce, M.I.; Hartlep, A.; Pedain, C.; Sampson, J.H. Convection-enhanced delivery of therapeutics for brain disease, and its optimization. Neurosurg. Focus 2006, 20, E12. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Nelson, B.D.; Ai, Y.; Stiles, D.K.; Gash, D.M.; Hardy, P.A.; Zhang, Z. Continuous intraputamenal convection-enhanced delivery in adult rhesus macaques. J. Neurosurg. 2015, 123, 1569–1577. [Google Scholar] [CrossRef] [PubMed]
- Sillay, K.; Hinchman, A.; Akture, E.; Salamat, S.; Miranpuri, G.; Williams, J.; Berndt, D. Convection enhanced delivery to the brain: Preparing for gene therapy and protein delivery to the brain for functional and restorative neurosurgery by understanding low-flow neurocatheter infusions using the Alaris® system infusion pump. Annu. Neurosci. 2013, 20, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Allard, E.; Passirani, C.; Benoit, P.J. Convection-enhanced delivery of nanocarriers for the treatment of brain tumors. Biomaterials 2009, 30, 2302–2318. [Google Scholar] [CrossRef] [PubMed]
- Morrison, P.F.; Laske, D.W.; Bobo, R.H.; Oldfield, E.H.; Dedrickt, R.L. High-flow microinfusions: Tissue penetration and pharmacodynamics. Am. J. Physiol. 1994, 266, 292–305. [Google Scholar]
- Occhiogrosso, G.; Edgar, M.A.; Souweidane, M.M. Prolonged convection-enhanced delivery into the rat brainstem. Neurosurgery 2003, 52, 388–394. [Google Scholar] [CrossRef] [PubMed]
- Tanner, P.G.; Holtmannspötter, M.; Grosshadern, K.; Tonn, J.C.; Goldbrunner, R. Effects of drug efflux on convectionenhanced paclitaxel delivery to malignant gliomas: Technical note. Neurosurgery 2007, 61, 880–882. [Google Scholar] [CrossRef] [PubMed]
- Buonerba, C.; Di Lorenzo, G.; Marinelli, A.; Federico, P.; Palmieri, G.; Imbimbo, M.; Conti, P.; Peluso, G.; de Placido, S.; Sampson, J.H. A comprehensive outlook on intracerebral therapy of malignant gliomas. Crit. Rev. Oncol. Hematol. 2011, 80, 54–68. [Google Scholar] [CrossRef] [PubMed]
- Varenika, V.; Dickinson, P.; Bringas, J.; le Couteur, R.; Higgins, R.; Park, J.; Fiandaca, M.; Berger, M.; Sampson, J.; Bankiewicz, K. Detection of infusate leakage in the brain using real-time imaging of convection-enhanced delivery. J. Neurosurg. 2008, 109, 874–880. [Google Scholar] [CrossRef] [PubMed]
- Han, S.J.; Bankiewicz, K.; Butowski, N.A.; Larson, P.S.; Aghi, M.K. Interventional MRI-guided catheter placement and real time drug delivery to the central nervous system. Expert Rev. Neurother. 2016, 16, 635–639. [Google Scholar] [CrossRef] [PubMed]
- Tykocki, T.; Miekisiak, G. Application of convection-enhanced drug delivery in the treatment of malignant gliomas. World Neurosurg. 2016, 90, 172–178. [Google Scholar] [CrossRef] [PubMed]
- Thomale, U.W.; Tyler, B.; Renard, V.M.; Dorfman, B.; Guarnieri, M.; Haberl, H.E.; Jallo, G.I. Local chemotherapy in the rat brainstem with multiple catheters: A feasibility study. Childs Nerv. Syst. 2009, 25, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Hardy, P.A.; Keeley, D.; Schorn, G.; Forman, E.; Ai, Y.; Venugopalan, R.; Zhang, Z.; Bradley, L.H. Convection enhanced delivery of different molecular weight tracers of gadolinium-tagged polylysine. J. Neurosci. Methods 2013, 219, 169–175. [Google Scholar] [CrossRef] [PubMed]
- Lidar, Z.; Mardor, Y.; Jonas, T.; Pfeffer, R.; Faibel, M.; Nass, D.; Hadani, M.; Ram, Z. Convection-enhanced delivery of paclitaxel for the treatment of recurrent malignant glioma: A phase I/II clinical study. J. Neurosurg. 2004, 100, 472–479. [Google Scholar] [CrossRef] [PubMed]
- Bogdahn, U.; Hau, P.; Stockhammer, G.; Venkataramana, N.K.; Mahapatra, A.K.; Suri, A.; Balasubramaniam, A.; Nair, S.; Oliushine, V.; Parfenov, V.; et al. Targeted therapy for high-grade glioma with the TGF-β2 inhibitor trabedersen: Results of a randomized and controlled phase IIb study. Neuro Oncol. 2011, 13, 132–142. [Google Scholar] [CrossRef] [PubMed]
- Kunwar, S.; Chang, S.; Westphal, M.; Vogelbaum, M.; Sampson, J.; Barnett, G.; Shaffrey, M.; Ram, Z.; Piepmeier, J.; Prados, M.; et al. Phase III randomized trial of ced of IL13-PE38QQR vs. gliadel wafers for recurrent glioblastoma. Neuro Oncol. 2010, 12, 871–881. [Google Scholar] [CrossRef] [PubMed]
- Mehta, A.I.; Choi, B.D.; Raghavan, R.; Brady, M.; Friedman, A.H.; Bigner, D.D.; Pastan, I.; Sampson, J.H. Imaging of convection enhanced delivery of toxins in humans. Toxins 2011, 3, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Sonabend, A.M.; Stuart, R.M.; Yun, J.; Yanagihara, T.; Mohajed, H.; Dashnaw, S.; Bruce, S.S.; Brown, T.; Romanov, A.; Sebastian, M.; et al. Prolonged intracerebral convection-enhanced delivery of topotecan with a subcutaneously implantable infusion pump. Neuro Oncol. 2011, 13, 886–893. [Google Scholar] [CrossRef] [PubMed]
- Muro, K.; Das, S.; Raizer, J.J. Convection-enhanced and local delivery of targeted cytotoxins in the treatment of malignant gliomas. Technol. Cancer Res. Treat. 2006, 5, 201–213. [Google Scholar] [CrossRef] [PubMed]
- Endo, T.; Fujii, Y.; Sugiyama, S.I.; Zhang, R.; Ogita, S.; Funamoto, K.; Saito, R.; Tominaga, T. Properties of convective delivery in spinal cord gray matter: Laboratory investigation and computational simulations. J. Neurosurg. Spine 2015, 30, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Luther, N.; Zhou, Z.; Zanzonico, P.; Cheung, N.K.; Humm, J.; Edgar, M.A.; Souweidane, M.M. The potential of theragnostic 124I-8H9 convection-enhanced delivery in diffuse intrinsic pontine glioma. Neuro Oncol. 2014, 16, 800–806. [Google Scholar] [CrossRef] [PubMed]
- Ghalamfarsa, G.; Hojjat-Farsangi, M.; Mohammadnia-Afrouzi, M.; Anvari, E.; Farhadi, S.; Yousefi, M.; Jadidi-Niaragh, F. Application of nanomedicine for crossing the blood-brain barrier: Theranostic opportunities in multiple sclerosis. J. Immunotoxicol. 2016, 13, 603–619. [Google Scholar] [CrossRef] [PubMed]
- Yoo, S.S.; Guo, L.; Sun, X.; Shaw, A.R.; Yuan, Z.; Lobenberg, R.; Roa, W.H. Fabrication and in vitro characterization of gadolinium-based nanoclusters for simultaneous drug delivery and radiation enhancement. Nanotechnology 2016, 27, 385104. [Google Scholar] [CrossRef] [PubMed]
- Fan, C.H.; Chang, E.L.; Ting, C.Y.; Lin, Y.C.; Liao, E.C.; Huang, C.Y.; Chang, Y.C.; Chan, H.L.; Wei, K.C.; Yeh, C.K. Folate-conjugated gene-carrying microbubbles with focused ultrasound for concurrent blood-brain barrier opening and local gene delivery. Biomaterials 2016, 106, 46–57. [Google Scholar] [CrossRef] [PubMed]
- Fan, C.H.; Cheng, Y.H.; Ting, C.Y.; Ho, Y.J.; Hsu, P.H.; Liu, H.L.; Yeh, C.K. Ultrasound/magnetic targeting with spio-dox-microbubble complex for image-guided drug delivery in brain tumors. Theranostics 2016, 6, 1542–1556. [Google Scholar] [CrossRef] [PubMed]
- Enochs, W.S.; Petherick, P.; Bogdanova, A.; Mohr, U.; Weissleder, R. Paramagnetic metal scavenging by melanin: MR imaging. Radiology 1997, 204, 417–423. [Google Scholar] [CrossRef] [PubMed]
- Michelot, J.M.; Moreau, M.F.; Labarre, P.G.; Madelmont, J.C.; Veyre, A.J.; Papon, J.M.; Parry, D.F.; Bonafous, J.F.; Boire, J.Y.; Desplanches, G.G.; et al. Synthesis and evaluation of new iodine-125 radiopharmaceuticals as potential tracers for malignant melanoma. J. Nucl. Med. 1991, 32, 1573–1580. [Google Scholar] [PubMed]
- Feng, H.; Xia, X.; Li, C.; Song, Y.; Qin, C.; Zhang, Y.; Lan, X. Tyr as a multifunctional reporter gene regulated by the tet-on system for multimodality imaging: An in vitro study. Sci. Rep. 2015, 5, 15502. [Google Scholar] [CrossRef] [PubMed]
- Zonios, G.; Dimou, A.; Bassukas, I.; Galaris, D.; Tsolakidis, A.; Kaxiras, E. Melanin absorption spectroscopy: New method for noninvasive skin investigation and melanoma detection. J. Biomed. Opt. 2008, 13, 014017. [Google Scholar] [CrossRef] [PubMed]
- Ait-Belkacem, R.; Berenguer, C.; Villard, C.; Ouafik, L.; Figarella-Branger, D.; Beck, A.; Chinot, O.; Lafitte, D. Monitoring therapeutic monoclonal antibodies in brain tumor. mAbs 2014, 6, 1385–1393. [Google Scholar] [CrossRef] [PubMed]
- Jansen, M.; Veldhuijzen van Zanten, S.E.; van Vuurden, D.G.; Huisman, M.; Vugts, D.J.; Hoekstra, O.S.; van Dongen, G.A.; Kaspers, G.J. Molecular drug imaging: 89Zr-bevacizumab PET in children with diffuse intrinsic pontine glioma. J. Nucl. Med. 2016. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.E.; Liu, T.; Wang, Y.; Cao, Y.; Du, J.; Luo, X.; Deshmukh, V.; Kim, C.H.; Lawson, B.R.; Tremblay, M.S.; et al. An immunosuppressive antibody-drug conjugate. J. Am. Chem. Soc. 2015, 137, 3229–3232. [Google Scholar] [CrossRef] [PubMed]
- Chatenoud, L.; Baudrihaye, M.F.; Chkoff, N.; Kreis, H.; Goldstein, G.; Bach, J.F. Restriction of the human in vivo immune response against the mouse monoclonal antibody OKT3. J. Immunol. 1986, 137, 830–838. [Google Scholar] [PubMed]
- Chatenoud, L.; Ferran, C.; Legendre, C.; Kurrle, R.; Kreis, H.; Bach, J.F. Immunological follow-up of renal allograft recipients treated with the BMA 031 (anti-TCR) monoclonal antibody. Transplant. Proc. 1990, 22, 1787–1788. [Google Scholar] [PubMed]
- Takahashi, H.; Okazaki, H.; Terasaki, P.I.; Iwaki, Y.; Kinukawa, T.; Taguchi, Y.; Chia, D.; Hardiwidjaja, S.; Miura, K.; Ishizaki, M.; et al. Reversal of transplant rejection by monoclonal antiblast antibody. Lancet 1983, 2, 1155–1158. [Google Scholar] [CrossRef]
- Bharadwaj, V.N.; Lifshitz, J.; Adelson, P.D.; Kodibagkar, V.D.; Stabenfeldt, S.E. Temporal assessment of nanoparticle accumulation after experimental brain injury: Effect of particle size. Sci. Rep. 2016, 6, 29988. [Google Scholar] [CrossRef] [PubMed]
- D’Hollander, A.; Mathieu, E.; Jans, H.; van de Velde, G.; Stakenborg, T.; van Dorpe, P.; Himmelreich, U.; Lagae, L. Development of nanostars as a biocompatible tumor contrast agent: Toward in vivo sers imaging. Int. J. Nanomed. 2016, 11, 3703–3714. [Google Scholar] [CrossRef] [PubMed]
- Saucier-Sawyer, J.K.; Seo, Y.E.; Gaudin, A.; Quijano, E.; Song, E.; Sawyer, A.J.; Deng, Y.; Huttner, A.; Saltzman, W.M. Distribution of polymer nanoparticles by convection-enhanced delivery to brain tumors. J. Control Release 2016, 232, 103–112. [Google Scholar] [CrossRef] [PubMed]
- Gagliardi, M. Novel biodegradable nanocarriers for enhanced drug delivery. Therap. Deliv. 2016, 7, 809–826. [Google Scholar] [CrossRef] [PubMed]
- Marciello, M.; Pellico, J.; Fernandez-Barahona, I.; Herranz, F.; Ruiz-Cabello, J.; Filice, M. Recent advances in the preparation and application of multifunctional iron oxide and liposome-based nanosystems for multimodal diagnosis and therapy. Interface Focus 2016, 6, 20160055. [Google Scholar] [CrossRef] [PubMed]
- Wen, M.M.; El-Salamouni, N.S.; El-Refaie, W.M.; Hazzah, H.A.; Ali, M.M.; Tosi, G.; Farid, R.M.; Blanco-Prieto, M.J.; Billa, N.; Hanafy, A.S. Nanotechnology-based drug delivery systems for Alzheimer’s disease management: Technical, industrial, and clinical challenges. J. Control Release 2016, 245, 95–107. [Google Scholar] [CrossRef] [PubMed]
- Kotov, N.A.; Winter, J.O.; Clements, I.P.; Jan, E.; Timko, B.P.; Campidelli, S.; Pathak, S.; Mazzatenta, A.; Lieber, C.M.; Prato, M.; et al. Nanomaterials for neural interfaces. Adv. Mater. 2009, 21, 3970–4004. [Google Scholar] [CrossRef]
- Gandomi, N.; Varshochian, R.; Atyabi, F.; Ghahremani, M.H.; Sharifzadeh, M.; Amini, M.; Dinarvand, R. Solid lipid nanoparticles surface modified with anti-contactin2 or anti-neurofascin for brain targeted delivery of medicines. Pharm. Dev. Technol. 2016, 11, 1–28. [Google Scholar] [CrossRef] [PubMed]
- Lindqvist, A.; Friden, M.; Hammarlund-Udenaes, M. Pharmacokinetic considerations of nanodelivery to the brain: Using modeling and simulations to predict the outcome of liposomal formulations. Eur. J. Pharm. Sci. 2016, 92, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Muntimadugu, E.; Dhommati, R.; Jain, A.; Challa, V.G.; Shaheen, M.; Khan, W. Intranasal delivery of nanoparticle encapsulated tarenflurbil: A potential brain targeting strategy for Alzheimer’s disease. Eur. J. Pharm. Sci. 2016, 92, 224–234. [Google Scholar] [CrossRef] [PubMed]
- Bakhtiary, Z.; Saei, A.A.; Hajipour, M.J.; Raoufi, M.; Vermesh, O.; Mahmoudi, M. Targeted superparamagnetic iron oxide nanoparticles for early detection of cancer: Possibilities and challenges. Nanomed. Nanotechnol. Biol. Med. 2016, 12, 287–307. [Google Scholar] [CrossRef] [PubMed]
- Rozhkova, E.A. Nanoscale materials for tackling brain cancer: Recent progress and outlook. Adv. Mater. 2011, 23, 136–150. [Google Scholar] [CrossRef] [PubMed]
- Gobbo, O.L.; Sjaastad, K.; Radomski, M.W.; Volkov, Y.; Prina-Mello, A. Magnetic nanoparticles in cancer theranostics. Theranostics 2015, 5, 1249–1263. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, L.R.; Gobin, A.M.; Lowery, A.R.; Tam, F.; Drezek, R.A.; Halas, N.J.; West, J.L. Metal nanoshells. Ann. Biomed. Eng. 2006, 34, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Gobin, A.M.; Lee, M.H.; Halas, N.J.; James, W.D.; Drezek, R.A.; West, J.L. Near-infrared resonant nanoshells for combined optical imaging and photothermal cancer therapy. Nano Lett. 2007, 7, 1929–1934. [Google Scholar] [CrossRef] [PubMed]
- Jordan, A.; Scholz, R.; Maier-Hauff, K.; van Landeghem, F.K.; Waldoefner, N.; Teichgraeber, U.; Pinkernelle, J.; Bruhn, H.; Neumann, F.; Thiesen, B.; et al. The effect of thermotherapy using magnetic nanoparticles on rat malignant glioma. J. Neuro Oncol. 2006, 78, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Thiesen, B.; Jordan, A. Clinical applications of magnetic nanoparticles for hyperthermia. Int. J. Hypertherm. 2008, 24, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Van Landeghem, F.K.; Maier-Hauff, K.; Jordan, A.; Hoffmann, K.T.; Gneveckow, U.; Scholz, R.; Thiesen, B.; Bruck, W.; von Deimling, A. Post-mortem studies in glioblastoma patients treated with thermotherapy using magnetic nanoparticles. Biomaterials 2009, 30, 52–57. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, K.M. Biomedical nanomagnetics: A spin through possibilities in imaging, diagnostics, and therapy. IEEE Trans. Magn. 2010, 46, 2523–2558. [Google Scholar] [CrossRef] [PubMed]
- Maier-Hauff, K.; Rothe, R.; Scholz, R.; Gneveckow, U.; Wust, P.; Thiesen, B.; Feussner, A.; von Deimling, A.; Waldoefner, N.; Felix, R.; et al. Intracranial thermotherapy using magnetic nanoparticles combined with external beam radiotherapy: Results of a feasibility study on patients with glioblastoma multiforme. J. Neuro Oncol. 2007, 81, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Chakravarty, R.; Hong, H.; Cai, W. Positron emission tomography image-guided drug delivery: Current status and future perspectives. Mol. Pharm. 2014, 11, 3777–3797. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Li, F.; Niu, G.; Chen, X. PET imaging of inflammation biomarkers. Theranostics 2013, 3, 448–466. [Google Scholar] [CrossRef] [PubMed]
- Carlucci, G.; Carney, B.; Brand, C.; Kossatz, S.; Irwin, C.P.; Carlin, S.D.; Keliher, E.J.; Weber, W.; Reiner, T. Dual-modality optical/PET imaging of PARP1 in glioblastoma. Mol. Imaging Biol. 2015, 17, 848–855. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Li, Y.; Lozada, J.; Wong, M.Q.; Greene, J.; Lin, K.S.; Yapp, D.; Perrin, D.M. Kit-like 18F-labeling of RGD-19F-arytrifluroborate in high yield and at extraordinarily high specific activity with preliminary in vivo tumor imaging. Nucl. Med. Biol. 2013, 40, 841–849. [Google Scholar] [CrossRef] [PubMed]
- Tsien, R.Y. Imagining imaging’s future. Nat. Rev. Mol. Cell Biol. 2003, 4, 16–21. [Google Scholar]
- Perrin, D.M. 18F-organotrifluoroborates as radioprosthetic groups for PET imaging: From design principles to preclinical applications. Acc. Chem. Res. 2016, 49, 1333–1343. [Google Scholar] [CrossRef] [PubMed]
- Ting, R.; Adam, M.J.; Ruth, T.J.; Perrin, D.M. Arylfluoroborates and alkylfluorosilicates as potential PET imaging agents: High-yielding aqueous biomolecular 18F-labeling. J. Am. Chem. Soc. 2005, 127, 13094–13095. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, E.A.; Wang, Y.; Crisp, J.L.; Vera, D.R.; Tsien, R.Y.; Ting, R. New dioxaborolane chemistry enables 18F-positron-emitting, fluorescent 18F-multimodality biomolecule generation from the solid phase. Bioconj. Chem. 2016, 27, 1390–1399. [Google Scholar] [CrossRef] [PubMed]
- Ting, R.; Aguilera, T.A.; Crisp, J.L.; Hall, D.J.; Eckelman, W.C.; Vera, D.R.; Tsien, R.Y. Fast 18F labeling of a near-infrared fluorophore enables positron emission tomography and optical imaging of sentinel lymph nodes. Bioconj. Chem. 2010, 21, 1811–1819. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Chen, H.; Chen, K.; Shao, Y.; Kiesewetter, D.O.; Niu, G.; Chen, X. Boramino acid as a marker for amino acid transporters. Sci. Adv. 2015, 1, 1500694. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Pourghiasian, M.; Radtke, M.A.; Lau, J.; Pan, J.; Dias, G.M.; Yapp, D.; Lin, K.S.; Benard, F.; Perrin, D.M. An organotrifluoroborate for broadly applicable one-step 18F-labeling. Angew. Chem. 2014, 53, 11876–11880. [Google Scholar] [CrossRef] [PubMed]
- Prabhakaran, J.; Solingapuram Sai, K.K.; Zanderigo, F.; Rubin-Falcone, H.; Jorgensen, M.J.; Kaplan, J.R.; Tooke, K.I.; Mintz, A.; Mann, J.J.; Kumar, J.S. In vivo evaluation of 18F Fecimbi-36, an agonist 5-HT2a/2c receptor PET radioligand in nonhuman primate. Bioorg. Med. Chem. Lett. 2017, 27, 21–23. [Google Scholar] [CrossRef] [PubMed]
- Ettrup, A.; da Cunha-Bang, S.; McMahon, B.; Lehel, S.; Dyssegaard, A.; Skibsted, A.W.; Jorgensen, L.M.; Hansen, M.; Baandrup, A.O.; Bache, S.; et al. Serotonin 2A receptor agonist binding in the human brain with 11C cimbi-36. J. Cerebr. Blood Flow Metab. 2014, 34, 1188–1196. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Cohen, A.L.; Colman, H. Targeted therapeutics in patients with high-grade gliomas: Past, present, and future. Curr. Treat. Opt. Oncol. 2016, 17, 1–11. [Google Scholar]
- Rizzo, D.; Ruggiero, A.; Martini, M.; Rizzo, V.; Maurizi, P.; Riccardi, R. Molecular biology in pediatric high-grade glioma: Impact on prognosis and treatment. BioMed Res. Int. 2015, 2015, 215135. [Google Scholar] [CrossRef] [PubMed]
- Hargrave, D.; Bartels, U.; Bouffet, E. Diffuse brainstem glioma in children: Critical review of clinical trials. Lancet Oncol. 2006, 7, 241–248. [Google Scholar] [CrossRef]
- Louis, D.N.; Perry, A.; Reifenberger, G.; von Deimling, A.; Figarella-Branger, D.; Cavenee, W.K.; Ohgaki, H.; Wiestler, O.D.; Kleihues, P.; Ellison, D.W. The 2016 world health organization classification of tumors of the central nervous system: A summary. Acta Neuropathol. 2016, 131, 803–820. [Google Scholar] [CrossRef] [PubMed]
- Friedman, H.S.; Prados, M.D.; Wen, P.Y.; Mikkelsen, T.; Schiff, D.; Abrey, L.E.; Yung, W.K.A.; Paleologos, N.; Nicholas, M.K.; Jensen, R.; et al. Bevacizumab alone and in combination with irinotecan in recurrent glioblastoma. J. Clin. Oncol. 2009, 27, 4733–4740. [Google Scholar] [CrossRef] [PubMed]
- Cohen, M.H.; Shen, Y.L.; Keegan, P.; Pazdur, R. FDA drug approval summary: Bevacizumab (avastin) as treatment of recurrent glioblastoma multiforme. Oncologist 2009, 14, 1131–1138. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, M.R.; Dignam, J.J.; Armstrong, T.S.; Wefel, J.S.; Blumenthal, D.T.; Vogelbaum, M.A.; Colman, H.; Chakravarti, A.; Pugh, S.; Won, M.; et al. A randomized trial of bevacizumab for newly diagnosed glioblastoma. N. Engl. J. Med. 2014, 370, 699–708. [Google Scholar] [CrossRef] [PubMed]
- Kreisl, T.N.; Kim, L.; Moore, K.; Duic, P.; Royce, C.; Stroud, I.; Garren, N.; Mackey, M.; Butman, J.A.; Camphausen, K.; et al. Phase II trial of single-agent bevacizumab followed by bevacizumab plus irinotecan at tumor progression in recurrent glioblastoma. J. Clin. Oncol. 2009, 27, 740–745. [Google Scholar] [CrossRef] [PubMed]
- Nagengast, W.B.; Hooge, M.N.L.D.; van Straten, E.M.E.; Kruijff, S.; Brouwers, A.H.; den Dunnen, W.F.A.; de Jong, J.R.; Hollema, H.; Dierckx, R.A.; Mulder, N.H.; et al. VEGF-Spect with 111In-bevacizumab in stage III/IV melanoma patients. Eur. J. Cancer 2011, 47, 1595–1602. [Google Scholar] [CrossRef] [PubMed]
- Desar, I.M.E.; Stillebroer, A.B.; Oosterwijk, E.; Leenders, W.P.J.; van Herpen, C.M.L.; van der Graaf, W.T.A.; Boerman, O.C.; Mulders, P.F.A.; Oyen, W.J.G. 111In-bevacizumab imaging of renal cell cancer and evaluation of neoadjuvant treatment with the vascular endothelial growth factor receptor inhibitor sorafenib. J. Nucl. Med. 2010, 51, 1707–1715. [Google Scholar] [CrossRef] [PubMed]
- Scheer, M.G.W.; Stollman, T.H.; Boerman, O.C.; Verrijp, K.; Sweep, F.C.G.J.; Leenders, W.P.J.; Ruers, T.J.M.; Oyen, W.J.G. Imaging liver metastases of colorectal cancer patients with radiolabelled bevacizumab: Lack of correlation with vegf-a expression. Eur. J. Cancer 2008, 44, 1835–1840. [Google Scholar] [CrossRef] [PubMed]
- Gaykema, S.B.M.; Brouwers, A.H.; Hooge, M.N.L.D.; Pleijhuis, R.G.; Timmer-Bosscha, H.; Pot, L.; van Dam, G.M.; van Der Meulen, S.B.; de Jong, J.R.; Bart, J.; et al. 89Zr-bevacizumab PET imaging in primary breast cancer. J. Nucl. Med. 2013, 54, 1014–1018. [Google Scholar] [CrossRef] [PubMed]
- Fleuren, E.D.G.; Versleijen-Jonkers, Y.M.H.; Heskamp, S.; van Herpen, C.M.L.; Oyen, W.J.G.; van der Graaf, W.T.A.; Boerman, O.C. Theranostic applications of antibodies in oncology. Mol. Oncol. 2014, 8, 799–812. [Google Scholar] [CrossRef] [PubMed]
- Parsons, D.W.; Jones, S.; Zhang, X.; Lin, J.C.-H.; Leary, R.J.; Angenendt, P.; Mankoo, P.; Carter, H.; Siu, I.M.; Gallia, G.L.; et al. An integrated genomic analysis of human glioblastoma multiforme. Science 2008, 321, 1807–1812. [Google Scholar] [CrossRef] [PubMed]
- Bender, S.; Tang, Y.; Lindroth, A.M.; Hovestadt, V.; Jones, D.T.W.; Kool, M.; Zapatka, M.; Northcott, P.A.; Sturm, D.; Wang, W.; et al. Reduced H3K27ME3 and DNA hypomethylation are major drivers of gene expression in K27m mutant pediatric high-grade gliomas. Cancer Cell 2013, 24, 660–672. [Google Scholar] [CrossRef] [PubMed]
- Dubuc, A.M.; Remke, M.; Korshunov, A.; Northcott, P.A.; Zhan, S.H.; Mendez-Lago, M.; Kool, M.; Jones, D.T.W.; Unterberger, A.; Morrissy, A.S.; et al. Aberrant patterns of H3K4 and H3K27 histone lysine methylation occur across subgroups in medulloblastoma. Acta Neuropathol. 2013, 125, 373–384. [Google Scholar] [CrossRef] [PubMed]
- Parsons, D.W.; Li, M.; Zhang, X.; Jones, S.; Leary, R.J.; Lin, J.C.-H.; Boca, S.M.; Carter, H.; Samayoa, J.; Bettegowda, C.; et al. The genetic landscape of the childhood cancer medulloblastoma. Science 2011, 331, 435–439. [Google Scholar] [CrossRef] [PubMed]
- Vanan, M.I.; Eisenstat, D.D. DIPG in children—what can we learn from the past? Front. Oncol. 2015, 5, 237. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Diaz, A.K.; Paugh, B.S.; Rankin, S.L.; Ju, B.; Li, Y.; Zhu, X.; Qu, C.; Chen, X.; Zhang, J.; et al. The genomic landscape of diffuse intrinsic pontine glioma and pediatric non-brainstem high-grade glioma. Nat. Genet. 2014, 46, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Broniscer, A.; McEachron, T.A.; Lu, C.; Paugh, B.S.; Becksfort, J.; Qu, C.; Ding, L.; Huether, R.; Parker, M.; et al. Somatic histone H3 alterations in pediatric diffuse intrinsic pontine gliomas and non-brainstem glioblastomas. Nat. Genet. 2012, 44, 251–253. [Google Scholar] [CrossRef] [PubMed]
- Buczkowicz, P.; Hoeman, C.; Rakopoulos, P.; Pajovic, S.; Letourneau, L.; Dzamba, M.; Morrison, A.; Lewis, P.; Bouffet, E.; Bartels, U.; et al. Genomic analysis of diffuse intrinsic pontine gliomas identifies three molecular subgroups and recurrent activating ACVR1 mutations. Nat. Genet. 2014, 46, 451–456. [Google Scholar] [CrossRef] [PubMed]
- Galanis, E.; Jaeckle, K.A.; Maurer, M.J.; Reid, J.M.; Ames, M.M.; Hardwick, J.S.; Reilly, J.F.; Loboda, A.; Nebozhyn, M.; Fantin, V.R.; et al. Phase II trial of vorinostat in recurrent glioblastoma multiforme: A north central cancer treatment group study. J. Clin. Oncol. 2009, 27, 2052–2058. [Google Scholar] [CrossRef] [PubMed]
- Friday, B.B.; Anderson, S.K.; Buckner, J.; Yu, C.; Giannini, C.; Geoffroy, F.; Schwerkoske, J.; Mazurczak, M.; Gross, H.; Pajon, E.; Jaeckle, K.; et al. Phase II trial of vorinostat in combination with bortezomib in recurrent glioblastoma: A north central cancer treatment group study. Neuro Oncol. 2012, 14, 215–221. [Google Scholar] [CrossRef] [PubMed]
- Shi, W.; Palmer, J.D.; Werner-Wasik, M.; Andrews, D.W.; Evans, J.J.; Glass, J.; Kim, L.; Bar-Ad, V.; Judy, K.; Farrell, C.; et al. Phase I trial of panobinostat and fractionated stereotactic re-irradiation therapy for recurrent high grade gliomas. J. Neurooncol. 2016, 127, 535–539. [Google Scholar] [CrossRef] [PubMed]
- Combs, S.E.; Heeger, S.; Haselmann, R.; Edler, L.; Debus, J.; Schulz-Ertner, D. Treatment of primary glioblastoma multiforme with cetuximab, radiotherapy and temozolomide (GERT): Phase I/II trial: Study protocol. BMC Cancer 2006, 18, 133. [Google Scholar] [CrossRef] [PubMed]
- Hasselbalch, B.; Lassen, U.; Hansen, S.; Holmberg, M.; Sorensen, M.; Kosteljanetz, M.; Broholm, H.; Stockhausen, M.-T.; Poulsen, H.S. Cetuximab, bevacizumab, and irinotecan for patients with primary glioblastoma and progression after radiation therapy and temozolomide: A phase II trial. Neuro Oncol. 2010, 12, 508–516. [Google Scholar] [PubMed]
- Rades, D.; Nadrowitz, R.; Buchmann, I.; Hunold, P.; Noack, F.; Schild, S.E.; Meller, B. Radiolabeled cetuximab plus whole-brain irradiation (WBI) for the treatment of brain metastases from non-small cell lung cancer (NSCLC). Strahlenther. Onkol. 2010, 186, 458–462. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, S.A.; Filippi, C.G.; Wong, T.; Ray, A.; Fralin, S.; Tsiouris, A.J.; Praminick, B.; Demopoulos, A.; McCrea, H.J.; Bodhinayake, I.; et al. Superselective intraarterial cerebral infusion of cetuximab after osmotic blood/brain barrier disruption for recurrent malignant glioma: Phase I study. J. Neurooncol. 2016, 128, 405–415. [Google Scholar] [CrossRef] [PubMed]
- Hatanpaa, K.J.; Burma, S.; Zhao, D.; Habib, A.A. Epidermal growth factor receptor in glioma: Signal transduction, neuropathology, imaging, and radioresistance. Neoplasia 2010, 12, 675–684. [Google Scholar] [CrossRef] [PubMed]
- Franceschi, E.; Cavallo, G.; Lonardi, S.; Magrini, E.; Tosoni, A.; Grosso, D.; Scopece, L.; Blatt, V.; Urbini, B.; Pession, A.; et al. Gefitinib in patients with progressive high-grade gliomas: A multicentre phase II study by gruppo italiano cooperativo di neuro-oncologia (GICNO). Br. J. Cancer 2007, 96, 1047–1051. [Google Scholar] [CrossRef] [PubMed]
- Haas-Kogan, D.A.; Prados, M.D.; Tihan, T.; Eberhard, D.A.; Jelluma, N.; Arvold, N.D.; Baumber, R.; Lamborn, K.R.; Kapadia, A.; Malec, M.; et al. Epidermal growth factor receptor, protein kinase B/Akt, and glioma response to erlotinib. J. Natl. Cancer Inst. 2005, 97, 880–887. [Google Scholar] [CrossRef] [PubMed]
- Padfield, E.; Ellis, H.P.; Kurian, K.M. Current therapeutic advances targeting EGFR and EGFRvIII in glioblastoma. Front. Oncol. 2015, 5, 5. [Google Scholar] [CrossRef] [PubMed]
- Reardon, D.A.; Nabors, L.B.; Mason, W.P.; Perry, J.R.; Shapiro, W.; Kavan, P.; Mathieu, D.; Phuphanich, S.; Cseh, A.; Fu, Y.; et al. Phase I/randomized phase II study of afatinib, an irreversible ERBB family blocker, with or without protracted temozolomide in adults with recurrent glioblastoma. Neuro Oncol. 2015, 17, 430–439. [Google Scholar] [CrossRef] [PubMed]
- Ma, D.J.; Galanis, E.; Anderson, S.K.; Schiff, D.; Kaufmann, T.J.; Peller, P.J.; Giannini, C.; Brown, P.D.; Uhm, J.H.; McGraw, S.; et al. A phase II trial of everolimus, temozolomide, and radiotherapy in patients with newly diagnosed glioblastoma: NCCTG N057K. Neuro Oncol. 2015, 17, 1261–1269. [Google Scholar] [CrossRef] [PubMed]
- Phillips, J.J.; Aranda, D.; Ellison, D.W.; Judkins, A.R.; Croul, S.E.; Brat, D.J.; Ligon, K.L.; Horbinski, C.; Venneti, S.; Zadeh, G.; et al. PDGFRA amplification is common in pediatric and adult high-grade astrocytomas and identifies a poor prognostic group in Idh1 mutant glioblastoma. Brain Pathol. 2013, 23, 565–573. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.Q.; Kaley, T.J.; Duda, D.G.; Schiff, D.; Lassman, A.B.; Wong, E.T.; Mikkelsen, T.; Purow, B.W.; Muzikansky, A.; Ancukiewicz, M.; et al. A multicenter, phase II, randomized, noncomparative clinical trial of radiation and temozolomide with or without vandetanib in newly diagnosed glioblastoma patients. Clin. Cancer Res. 2015, 21, 3610–3618. [Google Scholar] [CrossRef] [PubMed]
- Swartz, A.M.; Li, Q.J.; Sampson, J.H. Rindopepimut®: A promising immunotherapeutic for the treatment of glioblastoma multiforme. Immunotherapy 2014, 6, 679–690. [Google Scholar] [CrossRef] [PubMed]
- Schuster, J.; Lai, R.K.; Recht, L.D.; Reardon, D.A.; Paleologos, N.A.; Groves, M.D.; Mrugala, M.M.; Jensen, R.; Baehring, J.M.; Sloan, A.; et al. A phase II, multicenter trial of rindopepimut (Cdx-110) in newly diagnosed glioblastoma: The act III study. Neuro Oncol. 2015, 17, 854–861. [Google Scholar] [CrossRef] [PubMed]
- Preusser, M.; Lim, M.; Hafler, D.A.; Reardon, D.A.; Sampson, J.H. Prospects of immune checkpoint modulators in the treatment of glioblastoma. Nat. Rev. Neurol. 2015, 11, 504–514. [Google Scholar] [CrossRef] [PubMed]
- Neagu, M.R.; Reardon, D.A. An update on the role of immunotherapy and vaccine strategies for primary brain tumors. Curr. Treat. Opt. Oncol. 2015, 11, 54. [Google Scholar] [CrossRef] [PubMed]


| Agent | Mechanisms | Clinical Trials | Theranostics/Local Delivery |
|---|---|---|---|
| Angiogenesis Inhibitors | |||
| VEGF mAbs Bevacizumab | Inhibits vascular endothelial growth factor A (VEGF-A) [135] | No effect on OS in recurrent GBM [135,136,137,138] | 111In-bevacizumab SPECT in melanoma, RCC and CRC [139,140,141]; 89Zr-bevacizumab PET in primary BC [142,143]; Intra-arterial delivery of bevacizumab [37] |
| Epigenetic Therapies | |||
| HDAC inhibitors Panobinostat Vorinostat | Restore histone acetylation in histone mutated gliomas (GBM [144,145], medulloblastoma [146,147] and DIPG [148,149,150,151]) | Single agents in GBM [152] and DIPG [NCT02717455]; Combined agents [153]; Radiosensitizers [154] | - |
| Growth Factor Signaling | |||
| EGFR mAbs Cetuximab ABT-414 | Block EGFR signaling via binding extracellular domain. ABT-414 is an antibody-drug conjugate targeting EGFR/EGFRvIII | Cetuximab + temozolomide + XRT [155,156]; ABT-414: Phase II [NCT02573324] | 123I cetuximab crosses BBB, accumulates in NSCLC brain metastases [157]; Cetuximab SSIACI + mannitol BBBD [158] |
| EGFR TKIs Erlotinib Gefitinib | Block intracellular tyrosine kinase activity of EGFR | Limited single agent effect in Phase II studies; toxicities leading to early termination [159,160,161,162,163] | - |
| PI3K/mTOR inhibitors Everolimus Tacrolimus Sirolimus | Blockade of PI3K/mTOR growth signaling pathways | Everolimus + TMZ + XRT shows PET-visualized antiproliferative effects in GBM [164]; Everolimus in DIPG [NCT02233049] | - |
| PDGF/PDGFR Dasatinib Vandetanib | Targets PDGFR signaling; PDGFRA amplifications common in both adult and pediatric high-grade gliomas [165] | Dasatinib in DIPG [NCT02233049, NCT01644773]; Vandetanib in GBM shows no change in OS [166] | - |
| Immunotherapy/Vaccines | |||
| Vaccines Rindopepimut SL-701 | Vaccines establish immune response to either mutant EGFRvIIII antigen (rindopepimut) [167] or IL-13Ra2, survivin, and Epha2 (SL-701); additional personalized tumor lysate vaccines are under development | Rindopepimut + GM-CSF in newly diagnosed GBM patient prolongs PFS and OS with minimal toxicity [168]; Phase III discontinued [NCT01480479]; SL-701 in Phase I/II for GBM [NCT02078648]; BTIC/Imiquimod in DIPG [NCT01400672] | - |
| Checkpoint Inhibitors Ipilimumab Nivolumab | mAbs which target either CTLA-4 (ipilimumab) or PD-1 (nivolumab) enhancing immune system antitumoral response [169] | Phase III: Nivolumab + ipilimumab in recurrent GBM [NCT02017717]; Nivolumab in new GBM [NCT02617589] | - |
| Cell-based Therapies CAR-T | Chimeric antigen receptor transduced peripheral blood lymphocytes initiate cell-mediated cytotoxicity of target cells (i.e. against EGFRvIII) [170] | Phase I/II: GBM [NCT01454596] | - |
| Other | |||
| 124I-8H9 | MAb 8H9 recognizes B7-H3, extracellular antigen [83] | Phase I: DIPG [NCT01502917] | Agent delivered via CED |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tosi, U.; Marnell, C.S.; Chang, R.; Cho, W.C.; Ting, R.; Maachani, U.B.; Souweidane, M.M. Advances in Molecular Imaging of Locally Delivered Targeted Therapeutics for Central Nervous System Tumors. Int. J. Mol. Sci. 2017, 18, 351. https://doi.org/10.3390/ijms18020351
Tosi U, Marnell CS, Chang R, Cho WC, Ting R, Maachani UB, Souweidane MM. Advances in Molecular Imaging of Locally Delivered Targeted Therapeutics for Central Nervous System Tumors. International Journal of Molecular Sciences. 2017; 18(2):351. https://doi.org/10.3390/ijms18020351
Chicago/Turabian StyleTosi, Umberto, Christopher S. Marnell, Raymond Chang, William C. Cho, Richard Ting, Uday B. Maachani, and Mark M. Souweidane. 2017. "Advances in Molecular Imaging of Locally Delivered Targeted Therapeutics for Central Nervous System Tumors" International Journal of Molecular Sciences 18, no. 2: 351. https://doi.org/10.3390/ijms18020351
APA StyleTosi, U., Marnell, C. S., Chang, R., Cho, W. C., Ting, R., Maachani, U. B., & Souweidane, M. M. (2017). Advances in Molecular Imaging of Locally Delivered Targeted Therapeutics for Central Nervous System Tumors. International Journal of Molecular Sciences, 18(2), 351. https://doi.org/10.3390/ijms18020351







