Low Levels of IgG Recognizing the α-1-Antitrypsin Peptide and Its Association with Taiwanese Women with Primary Sjögren’s Syndrome
Abstract
:1. Introduction
2. Results
2.1. Identification and Validation of Differentially Expressed Serum Proteins by In-Solution Digestion and LC-MS/MS
2.2. Novel HNE Modification Identification of Serum Proteins by In-Gel Digestion and LC-MS/MS
2.3. Autoreactivity against A1AT50–63 and A1AT50–63 HNE Peptides
2.4. Determination of HNE-Protein Adducts
2.5. Association of Elevated HNE-Protein Adduct, Lowed A1AT Level or Decreased Autoreactivity against A1AT50–63 and A1AT50–63 HNE Peptides with pSS Patients
3. Discussion
4. Materials and Methods
4.1. Patient Samples
4.2. Depleted-Albumin and IgG Serum Proteins, In-Solution Digestion, and Protein Identification by LC-MS/MS
4.3. Western Blotting
4.4. 1-D SDS-PAGE, In-Gel Digestion, and HNE Identification by LC-MS/MS
4.5. IP
4.6. Detection of Autoreactivity against A1AT50–63 and Their HNE-Modified Peptides
4.7. Detection of Serum HNE-Protein Adducts
4.8. Statistical Analyses
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| pSS | primary Sjögren’s syndrome |
| RA | rheumatoid arthritis |
| SLE | systemic lupus erythematosus |
| HNE | 4-Hydroxy-2-nonenal |
| 1-D SDS-PAGE | one-dimensional sodium dodecyl sulfate polyacrylamide gel electrophoresis |
| nano-LC–MS/MS | nano-liquid chromatography–tandem mass spectrometry |
| HC | healthy control |
| Ig | immunoglobulin |
| ACR | American College of Rheumatology |
| IP | immunoprecipitation |
| A1AG1 | α-1-acid glycoprotein 1 |
| RSD | relative standard deviation |
| SD | standard deviation |
| ROC | receiver operating characteristic |
| AUC | area under the ROC curve |
| CBB | Coomassie brilliant blue |
| RF | rheumatoid factor |
| ANA | antinuclear antibody |
| anti-Ro (SSA) | anti-Sjögren’s-syndrome-related antigen A |
| anti-La (SSB) | anti-Sjögren’s-syndrome-related antigen B |
| CRP | C-Reactive protein |
| ESR | erythrocyte sedimentation rate |
| OR | odds ratio |
| ESSDAI | EULAR Sjögren’s syndrome disease activity index |
| NSAIDs | nonsteroidal anti-inflammatory drugs |
| DMARDs | Disease-modifying anti-rheumatic drugs |
| DMARDs | Disease-modifying anti-rheumatic drugs |
References
- Jonsson, R.; Vogelsang, P.; Volchenkov, R.; Espinosa, A.; Wahren-Herlenius, M.; Appel, S. The complexity of Sjögren’s syndrome : Novel aspects on pathogenesis. Immunol. Lett. 2011, 141, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Yu, K.H.; See, L.C.; Kuo, C.F.; Chou, I.J.; Chou, M.J. Prevalence and incidence in patients with autoimmune rheumatic diseases: A nationwide population-based study in Taiwan. Arthritis Care Res. 2013, 65, 244–250. [Google Scholar] [CrossRef] [PubMed]
- Maslinska, M.; Przygodzka, M.; Kwiatkowska, B.; Sikorska-Siudek, K. Sjögren’s syndrome: Still not fully understood disease. Rheumatol. Int. 2015, 35, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, R.; Brun, J.G. Sjögren’s syndrome. In Els; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2010. [Google Scholar]
- Norheim, K.B.; Jonsson, G.; Harboe, E.; Hanasand, M.; Goransson, L.; Omdal, R. Oxidative stress, as measured by protein oxidation, is increased in primary Sjögren’s syndrome. Free Radic. Res. 2012, 46, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Wakamatsu, T.H.; Dogru, M.; Matsumoto, Y.; Kojima, T.; Kaido, M.; Ibrahim, O.M.; Sato, E.A.; Igarashi, A.; Ichihashi, Y.; Satake, Y.; et al. Evaluation of lipid oxidative stress status in Sjögren’s syndrome patients. Investig. Ophthalmol. Vis. Sci. 2013, 54, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Uchida, K. Protein-bound 4-hydroxy-2-nonenal as a marker of oxidative stress. J. Clin. Biochem. Nutr. 2005, 36, 1–10. [Google Scholar] [CrossRef]
- Zhao, J.; Chen, J.; Zhu, H.; Xiong, Y.L. Mass spectrometric evidence of malonaldehyde and 4-hydroxynonenal adductions to radical-scavenging soy peptides. J. Agric. Food Chem. 2012, 60, 9727–9736. [Google Scholar] [CrossRef] [PubMed]
- Lv, L.; Lin, H.; Li, Z.; Yuan, F.; Gao, Q.; Ma, J. Effect of 4-hydroxy-2-nonenal treatment on the IgE binding capacity and structure of shrimp (Metapenaeus ensis) tropomyosin. Food Chem. 2016, 212, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Uchida, K. 4-hydroxy-2-nonenal: A product and mediator of oxidative stress. Prog. Lipid Res. 2003, 42, 318–343. [Google Scholar] [CrossRef]
- Eggleton, P.; Nissim, A.; Ryan, B.J.; Whiteman, M.; Winyard, P.G. Detection and isolation of human serum autoantibodies that recognize oxidatively modified autoantigens. Free Radic. Biol. Med. 2013, 57, 79–91. [Google Scholar] [CrossRef] [PubMed]
- Alzolibani, A.A.; Al Robaee, A.A.; Al-Shobaili, H.A.; Rasheed, Z. 4-hydroxy-2-nonenal modified histone-H2A: A possible antigenic stimulus for systemic lupus erythematosus autoantibodies. Cell. Immunol. 2013, 284, 154–162. [Google Scholar] [CrossRef] [PubMed]
- Breit, S.N.; Wakefield, D.; Robinson, J.P.; Luckhurst, E.; Clark, P.; Penny, R. The role of α 1-antitrypsin deficiency in the pathogenesis of immune disorders. Clin. Immunol. Immunopathol. 1985, 35, 363–380. [Google Scholar] [CrossRef]
- Macen, J.L.; Upton, C.; Nation, N.; McFadden, G. SERP1, a serine proteinase inhibitor encoded by myxoma virus, is a secreted glycoprotein that interferes with inflammation. Virology 1993, 195, 348–363. [Google Scholar] [CrossRef] [PubMed]
- Lindstrom, F.D.; Skogh, T.; Lundstrom, I.M. α1 antitrypsin deficiency in a patient with systemic vasculitis and primary Sjögren’s syndrome. Ann. Rheum. Dis. 2002, 61, 945–946. [Google Scholar] [CrossRef] [PubMed]
- Malfait, R.; Gorus, F.; Sevens, C. Electrophoresis of serum protein to detect α1-antitrypsin deficiency: Five illustrative cases. Clin. Chem. 1985, 31, 1397–1399. [Google Scholar] [PubMed]
- Barrera, G.; Pizzimenti, S.; Ciamporcero, E.S.; Daga, M.; Ullio, C.; Arcaro, A.; Cetrangolo, G.P.; Ferretti, C.; Dianzani, C.; Lepore, A.; et al. Role of 4-hydroxynonenal-protein adducts in human diseases. Antioxid. Redox Signal. 2015, 22, 1681–1702. [Google Scholar] [CrossRef] [PubMed]
- Fournier, T.; Medjoubi, N.N.; Porquet, D. α1-acid glycoprotein. Biochim. Biophys. Acta 2000, 1482, 157–171. [Google Scholar] [CrossRef]
- Hochepied, T.; Berger, F.G.; Baumann, H.; Libert, C. α1-acid glycoprotein: An acute phase protein with inflammatory and immunomodulating properties. Cytokine Growth Factor Rev. 2003, 14, 25–34. [Google Scholar] [CrossRef]
- Gemelli, C.; Martello, A.; Montanari, M.; Zanocco Marani, T.; Salsi, V.; Zappavigna, V.; Parenti, S.; Vignudelli, T.; Selmi, T.; Ferrari, S.; et al. The orosomucoid 1 protein is involved in the vitamin D-mediated macrophage de-activation process. Exp. Cell Res. 2013, 319, 3201–3213. [Google Scholar] [CrossRef] [PubMed]
- Rantapaa-Dahlqvist, S.; Backman, C.; Sandgren, H.; Ostberg, Y. Echocardiographic findings in patients with primary Sjogren's syndrome. Clin. Rheumatol. 1993, 12, 214–218. [Google Scholar] [CrossRef] [PubMed]
- Saroha, A.; Biswas, S.; Chatterjee, B.P.; Das, H.R. Altered glycosylation and expression of plasma α1-acid glycoprotein and haptoglobin in rheumatoid arthritis. J. Chromatogr. B 2011, 879, 1839–1843. [Google Scholar] [CrossRef] [PubMed]
- Grimstein, C.; Choi, Y.K.; Wasserfall, C.H.; Satoh, M.; Atkinson, M.A.; Brantly, M.L.; Campbell-Thompson, M.; Song, S. α1 antitrypsin protein and gene therapies decrease autoimmunity and delay arthritis development in mouse model. J. Transl. Med. 2011, 9, 21. [Google Scholar] [CrossRef] [PubMed]
- Elshikha, A.S.; Lu, Y.; Chen, M.J.; Akbar, M.; Zeumer, L.; Ritter, A.; Elghamry, H.; Mahdi, M.A.; Morel, L.; Song, S. α1 antitrypsin inhibits dendritic cell activation and attenuates nephritis in a mouse model of lupus. PLoS ONE 2016, 11, e0156583. [Google Scholar] [CrossRef] [PubMed]
- Ciobanu, A.; Ciobanu, I.R.; Ionita, A.; Stoicescu, M.; Stroescu, I.; Caralicea, M.; Curut, O. Some humoral immunological aspects of the rheumatoid arthritis correlated with the ultrastructural changes of the rheumatoid synovial fluid. Romanian J. Morphol. Embryol. 1992, 38, 143–154. [Google Scholar]
- Stefanescu, M.; Szegli, G.; Cremer, L.; Zarma, L.; Mazilu, E.; Naghiu, M.; Niculescu, D.; Gaches, A.; Onu, A. The presence and significance of some anti-enzyme antibodies (anti-plasminogen, anti-trypsin, anti-phospholipase C) in rheumatoid arthritis (RA) and reactive arthritis (RA). Arch. Roum. Pathol. Exp. Microbiol. 1989, 48, 47–53. [Google Scholar] [PubMed]
- Struthers, G.R.; Lewin, I.V.; Stanworth, D.R. IgA-α1 antitrypsin complexes in ankylosing spondylitis. Ann. Rheum. Dis. 1989, 48, 30–34. [Google Scholar] [CrossRef] [PubMed]
- Lacki, J.K.; Schochat, T.; Leszczynski, P.; Mackiewicz, S.H.; Muller, W. IgA-α1-antitrypsin complex in systemic lupus erythematosus: Preliminary report. Lupus 1995, 4, 221–224. [Google Scholar] [CrossRef] [PubMed]
- Lacki, J.K.; Schochat, T.; Porawaska, W.; Mackiewicz, S.H.; Muller, W. Does the serum level of IgA-α1-antitrypsin complex correlate with radiological progression in early rheumatoid arthritis? A 3-year follow-up study. Rheumatol. Int. 1995, 14, 227–230. [Google Scholar] [CrossRef] [PubMed]
- Silosi, I.; Silosi, C.A.; Boldeanu, M.V.; Cojocaru, M.; Biciusca, V.; Avramescu, C.S.; Cojocaru, I.M.; Bogdan, M.; FolcuTi, R.M. The role of autoantibodies in health and disease. Romanian J. Morphol. Embryol. 2016, 57, 633–638. [Google Scholar]
- Binder, C.J.; Papac-Milicevic, N.; Witztum, J.L. Innate sensing of oxidation-specific epitopes in health and disease. Nat. Rev. Immunol. 2016, 16, 485–497. [Google Scholar] [CrossRef] [PubMed]
- Schwartz-Albiez, R.; Monteiro, R.C.; Rodriguez, M.; Binder, C.J.; Shoenfeld, Y. Natural antibodies, intravenous immunoglobulin and their role in autoimmunity, cancer and inflammation. Clin. Exp. Immunol. 2009, 158 (Suppl. 1), 43–50. [Google Scholar] [CrossRef] [PubMed]
- Sutti, S.; Jindal, A.; Locatelli, I.; Vacchiano, M.; Gigliotti, L.; Bozzola, C.; Albano, E. Adaptive immune responses triggered by oxidative stress contribute to hepatic inflammation in nash. Hepatology 2014, 59, 886–897. [Google Scholar] [CrossRef] [PubMed]
- Aletaha, D.; Neogi, T.; Silman, A.J.; Funovits, J.; Felson, D.T.; Bingham, C.O., III; Birnbaum, N.S.; Burmester, G.R.; Bykerk, V.P.; Cohen, M.D.; et al. 2010 rheumatoid arthritis classification criteria: An american college of rheumatology/european league against rheumatism collaborative initiative. Arthritis Rheum. 2010, 62, 2569–2581. [Google Scholar] [CrossRef] [PubMed]
- Arnett, F.C.; Edworthy, S.M.; Bloch, D.A.; McShane, D.J.; Fries, J.F.; Cooper, N.S.; Healey, L.A.; Kaplan, S.R.; Liang, M.H.; Luthra, H.S.; et al. The american rheumatism association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum. 1988, 31, 315–324. [Google Scholar] [CrossRef] [PubMed]
- Vitali, C.; Bombardieri, S.; Jonsson, R.; Moutsopoulos, H.M.; Alexander, E.L.; Carsons, S.E.; Daniels, T.E.; Fox, P.C.; Fox, R.I.; Kassan, S.S.; et al. Classification criteria for Sjögren’s syndrome: A revised version of the European criteria proposed by the American-European consensus group. Ann. Rheum. Dis. 2002, 61, 554–558. [Google Scholar] [CrossRef] [PubMed]
- Hochberg, M.C. Updating the american college of rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum. 1997, 40, 1725. [Google Scholar] [CrossRef] [PubMed]
- Uen, Y.H.; Lin, K.Y.; Sun, D.P.; Liao, C.C.; Hsieh, M.S.; Huang, Y.K.; Chen, Y.W.; Huang, P.H.; Chen, W.J.; Tai, C.C.; et al. Comparative proteomics, network analysis and post-translational modification identification reveal differential profiles of plasma con a-bound glycoprotein biomarkers in gastric cancer. J. Proteom. 2013, 83, 197–213. [Google Scholar] [CrossRef] [PubMed]
- Huo, X.; Wu, B.; Feng, M.; Han, B.; Fang, Y.; Hao, Y.; Meng, L.; Wubie, A.J.; Fan, P.; Hu, H.; et al. Proteomic analysis reveals the molecular underpinnings of mandibular gland development and lipid metabolism in two lines of honeybees (Apis mellifera ligustica). J. Proteome Res. 2016, 15, 3342–3357. [Google Scholar] [CrossRef] [PubMed]
- Liao, C.C.; Chou, P.L.; Cheng, C.W.; Chang, Y.S.; Chi, W.M.; Tsai, K.L.; Chen, W.J.; Kung, T.S.; Tai, C.C.; Lee, K.W.; et al. Comparative analysis of novel autoantibody isotypes against citrullinated-inter-α-trypsin inhibitor heavy chain 3 (ITIH3)542–556 peptide in serum from taiwanese females with rheumatoid arthritis, primary Sjögren’s syndrome and secondary Sjögren’s syndrome in rheumatoid arthritis. J. Proteom. 2016, 141, 1–11. [Google Scholar]
- Weber, D.; Milkovic, L.; Bennett, S.J.; Griffiths, H.R.; Zarkovic, N.; Grune, T. Measurement of HNE-protein adducts in human plasma and serum by ELISA-comparison of two primary antibodies. Redox Biol. 2013, 1, 226–233. [Google Scholar] [CrossRef] [PubMed]
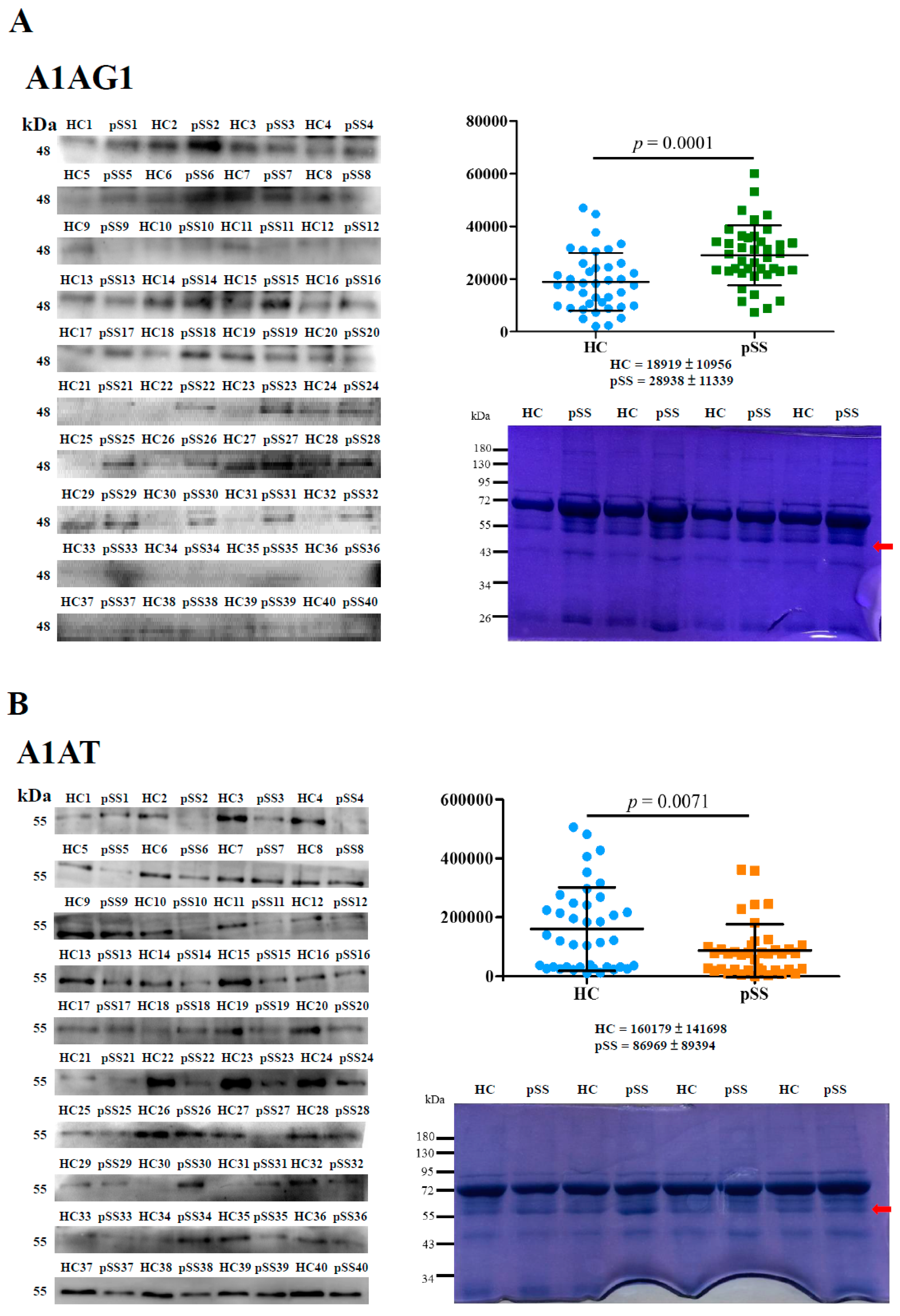
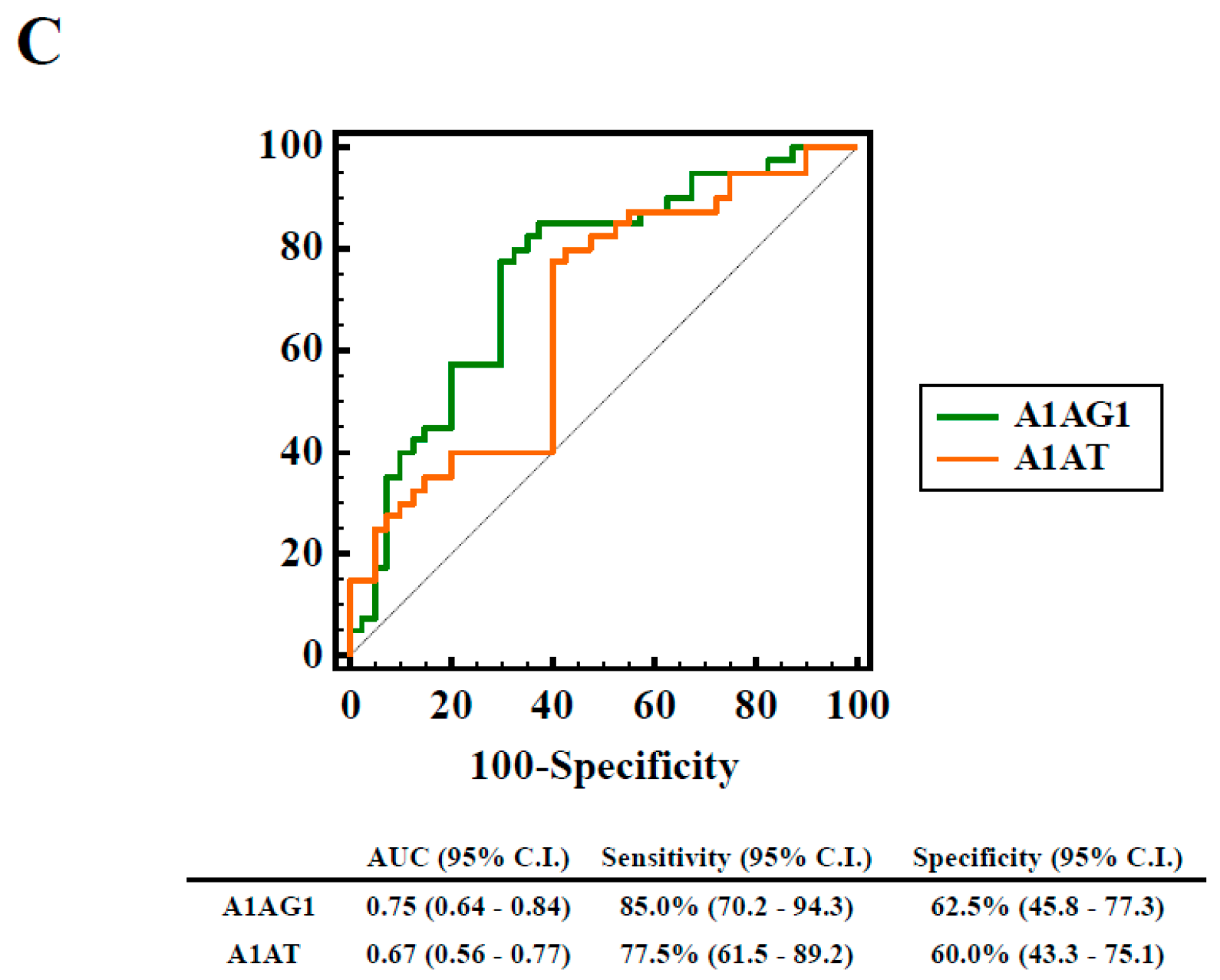
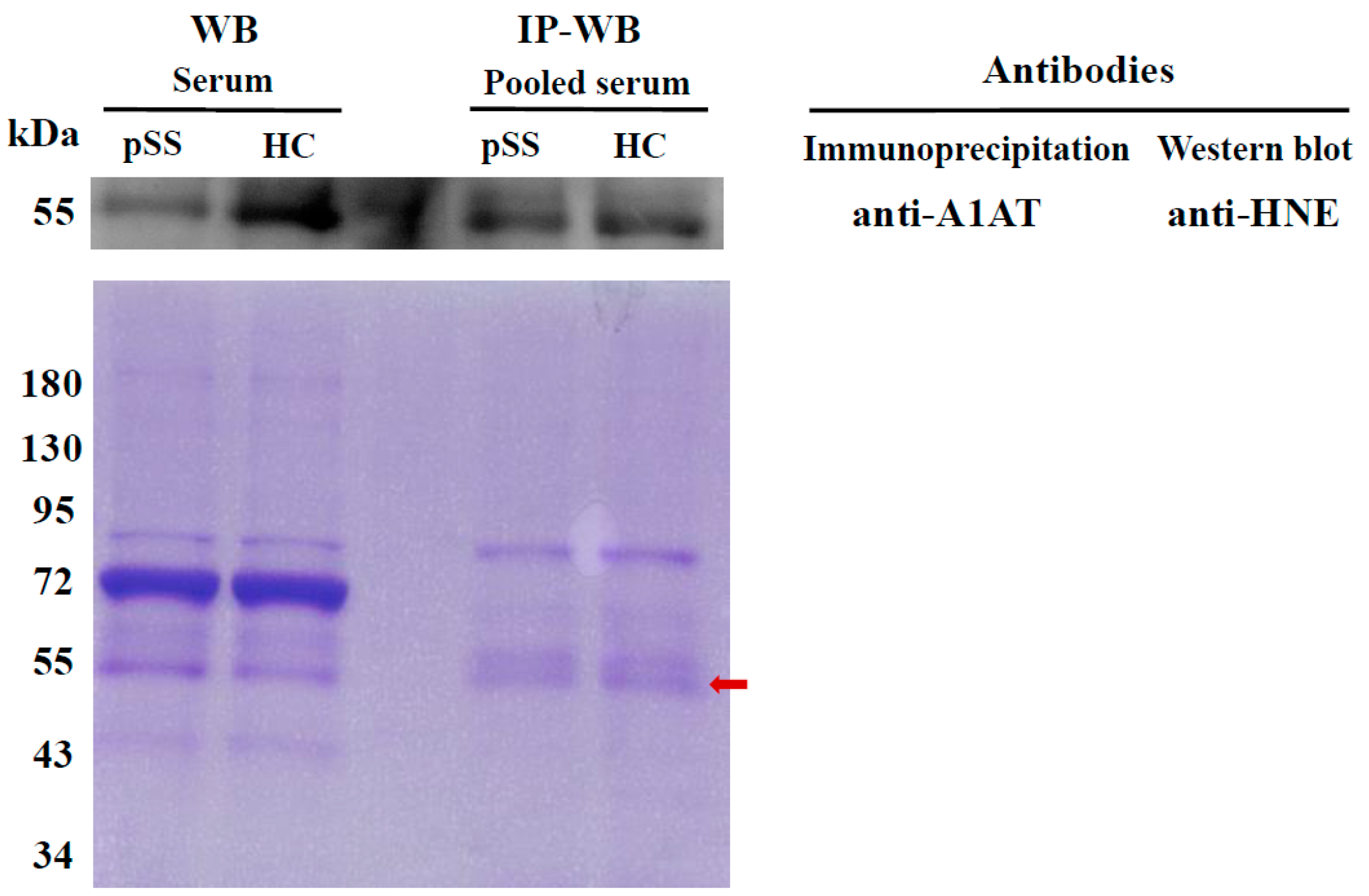
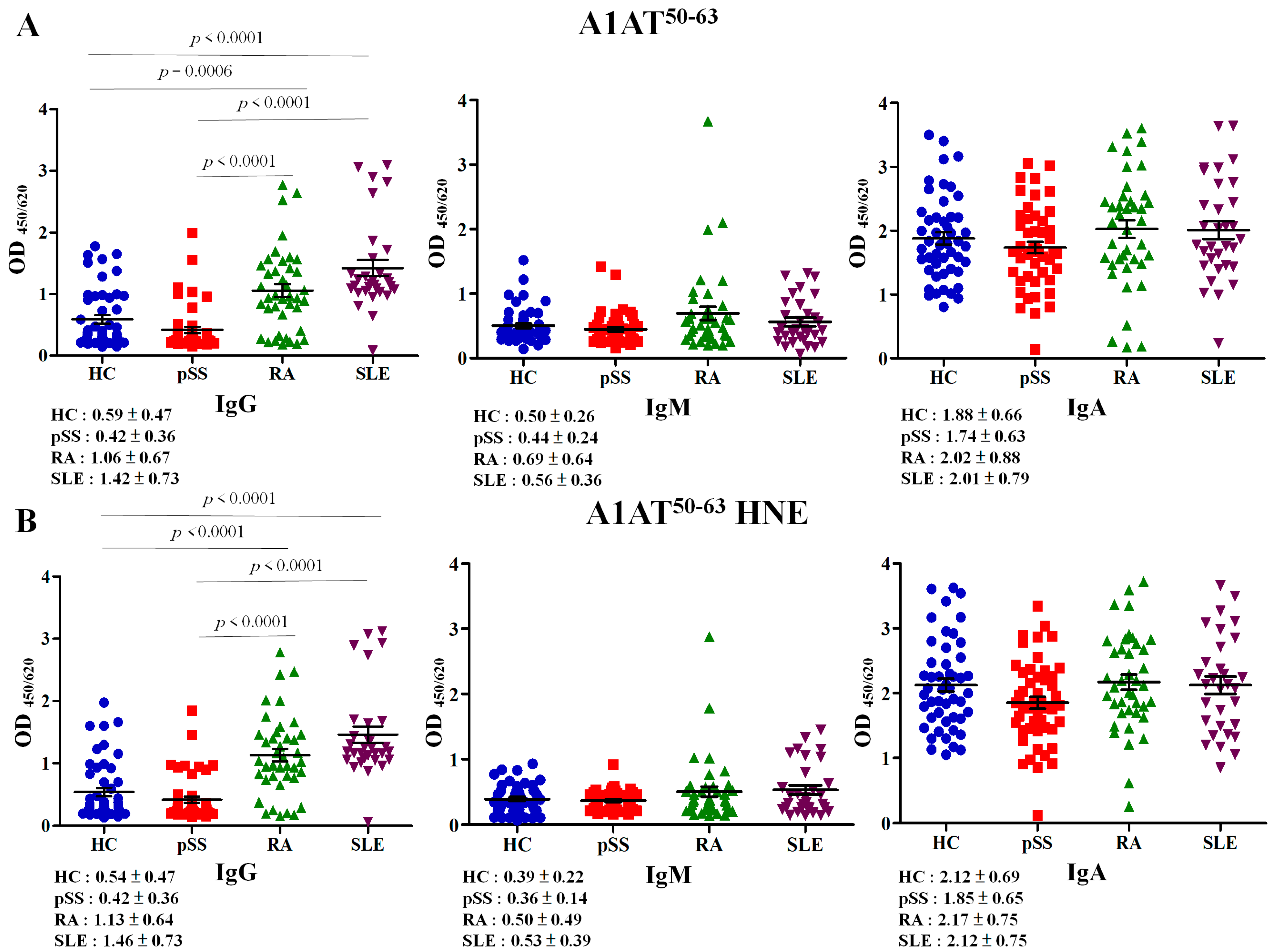
| Protein ID | Symbol | Protein Name | Normalized Average Spectral Counts (Mean ± RSD%) | −10lgp | Fold Change a | |
|---|---|---|---|---|---|---|
| Normal | pSS | |||||
| P02763 | A1AG1 | α-1-acid glycoprotein 1 | 1,528,333 ± 1.76 | 3,740,000 ± 6.64 | 23.5 | ↑ 2.4 |
| P00738 | HPT | Haptoglobin | 730,666,667 ±11.29 | 1,223,333,333 ± 40.04 | 44.0 | ↑ 1.7 |
| P69905 | HBA | Hemoglobin subunit α | 56,900,000 ± 32.80 | 87,200,000 ± 9.86 | 32.4 | ↑ 1.5 |
| P00739 | HPTR | Haptoglobin-related protein | 10,410,000 ± 3.53 | 15,366,667 ± 4.97 | 30.4 | ↑ 1.5 |
| P01781 | HV320 | Ig heavy chain V-III region GAL | 3,339,000 ± 1.48 | 4,910,000 ± 15.60 | 22.1 | ↑ 1.5 |
| P68871 | HBB | Hemoglobin subunit beta | 47,033,333 ± 19.59 | 63,500,000 ± 17.09 | 26.5 | ↑ 1.4 |
| P01860 | IGHG3 | Ig gamma-3 chain C region | 6,816,667 ± 10.72 | 8,310,000 ± 5.22 | 23.6 | ↑ 1.2 |
| O75636 | FCN3 | Ficolin-3 | 8,266,667 ± 3.99 | 8,640,000 ± 3.09 | 27.2 | ↓ 1.0 |
| P01857 | IGHG1 | Ig gamma-1 chain C region | 161,000,000 ± 10.64 | 163,000,000 ± 7.53 | 21.6 | ↓ 1.0 |
| P01591 | IGJ | Immunoglobulin J chain | 18,366,667 ± 5.63 | 18,300,000 ± 12.91 | 21.1 | ↓ 1.0 |
| P02749 | APOH | Beta-2-glycoprotein 1 | 15,363,333 ± 3.01 | 15,266,667 ± 3.27 | 25.6 | ↓ 1.0 |
| P02745 | C1QA | Complement C1q subcomponent subunit A | 5,913,333 ± 3.85 | 5,410,000 ± 7.56 | 21.6 | ↓ 1.1 |
| P20742 | PZP | Pregnancy zone protein | 3,450,000 ± 7.97 | 3,130,000 ± 7.19 | 25.9 | ↓ 1.1 |
| P06681 | CO2 | Complement C2 | 2,213,333 ± 4.01 | 1,906,667 ± 11.73 | 20.6 | ↓ 1.2 |
| P13671 | CO6 | Complement component C6 | 4,256,667 ± 6.68 | 3,483,333 ± 9.82 | 34.0 | ↓ 1.2 |
| P04003 | C4BPA | C4b-binding protein α chain | 38,466,667 ± 14.64 | 31,133,333 ± 7.59 | 24.3 | ↓ 1.2 |
| P04196 | HRG | Histidine-rich glycoprotein | 30,800,000 ± 5.97 | 24,233,333 ± 18.20 | 24.9 | ↓ 1.3 |
| P05090 | APOD | Apolipoprotein D | 6,350,000 ± 5.43 | 4,936,667 ± 10.03 | 26.4 | ↓ 1.3 |
| P01024 | CO3 | Complement C3 | 175,666,667 ± 14.62 | 135,000,000 ± 9.04 | 36.3 | ↓ 1.3 |
| P08697 | A2AP | α-2-antiplasmin | 2,643,333 ± 9.92 | 1,880,000 ± 10.03 | 23.8 | ↓ 1.4 |
| P10909 | CLUS | Clusterin | 20,466,667 ± 12.03 | 14,500,000 ± 11.06 | 31.1 | ↓ 1.4 |
| P00734 | THRB | Prothrombin | 27,733,333 ± 16.53 | 19,566,667 ± 16.23 | 27.0 | ↓ 1.4 |
| P09871 | C1S | Complement C1s subcomponent | 5,006,667 ± 6.47 | 3,423,333 ± 3.91 | 36.7 | ↓ 1.5 |
| P00736 | C1R | Complement C1r subcomponent | 5,373,333 ± 9.76 | 3,450,000 ± 4.28 | 37.2 | ↓ 1.6 |
| P02647 | APOA1 | Apolipoprotein A-I | 36,466,667 ± 33.11 | 21,866,667 ± 16.56 | 44.8 | ↓ 1.7 |
| P02655 | APOC2 | Apolipoprotein C-II | 1,356,667 ± 53.91 | 785,000 ± 4.70 | 39.7 | ↓ 1.7 |
| P05160 | F13B | Coagulation factor XIII B chain | 431,667 ± 5.81 | 228,667 ± 3.58 | 21.7 | ↓ 1.9 |
| P01009 | A1AT | α-1-antitrypsin | 50,366,667 ± 8.66 | 19,766,667 ± 29.04 | 72.6 | ↓ 2.5 |
| Risk Factor | Cut-Off | HC, n | pSS, n | Age-Adjusted Logistic Regression Model a | Power | |
|---|---|---|---|---|---|---|
| ORs (95% CI) | p Value | |||||
| HNE-protein adduct | ≥2.884 | 29 | 43 | 1.0 | 0.708 | |
| <2.884 | 20 | 6 | 4.887 (1.744–13.637) | 0.003 | ||
| Serum A1AT | ≥123,574.37 | 19 | 7 | 1.0 | 0.726 | |
| <123,574.37 | 21 | 33 | 3.910 (1.384–11.035) | 0.010 | ||
| Anti-A1AT50–63 IgG | ≥0.407 | 21 | 9 | 1.0 | ||
| <0.407 | 28 | 40 | 3.360 (1.336–8.451) | 0.010 | 0.802 | |
| Anti-A1AT50–63 HNE IgG | ≥0.350 | 22 | 13 | 1.0 | ||
| <0.350 | 27 | 36 | 2.263 (0.967–5.299) | 0.060 | 0.500 | |
| Anti-A1AT50–63 IgA | ≥1.796 | 25 | 19 | 1.0 | ||
| <1.796 | 24 | 30 | 1.650 (0.738–3.687) | 0.222 | 0.232 | |
| Anti-A1AT50–63 HNE IgA | ≥1.903 | 28 | 21 | 1.0 | ||
| <1.903 | 21 | 28 | 1.786 (0.801–3.986) | 0.157 | 0.293 | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, Y.-S.; Pan, C.-H.; Chang, C.-C.; Tsai, K.-L.; Chou, H.-W.; Chen, J.-H.; Lin, S.-H.; Lu, Y.-Y.; Tai, C.-C.; Lin, Y.-F.; et al. Low Levels of IgG Recognizing the α-1-Antitrypsin Peptide and Its Association with Taiwanese Women with Primary Sjögren’s Syndrome. Int. J. Mol. Sci. 2017, 18, 2750. https://doi.org/10.3390/ijms18122750
Chang Y-S, Pan C-H, Chang C-C, Tsai K-L, Chou H-W, Chen J-H, Lin S-H, Lu Y-Y, Tai C-C, Lin Y-F, et al. Low Levels of IgG Recognizing the α-1-Antitrypsin Peptide and Its Association with Taiwanese Women with Primary Sjögren’s Syndrome. International Journal of Molecular Sciences. 2017; 18(12):2750. https://doi.org/10.3390/ijms18122750
Chicago/Turabian StyleChang, Yu-Sheng, Chih-Hong Pan, Che-Chang Chang, Kai-Leun Tsai, Han-Wen Chou, Jin-Hua Chen, Sheng-Hong Lin, Yi-Ying Lu, Chih-Chun Tai, Yi-Fang Lin, and et al. 2017. "Low Levels of IgG Recognizing the α-1-Antitrypsin Peptide and Its Association with Taiwanese Women with Primary Sjögren’s Syndrome" International Journal of Molecular Sciences 18, no. 12: 2750. https://doi.org/10.3390/ijms18122750
APA StyleChang, Y.-S., Pan, C.-H., Chang, C.-C., Tsai, K.-L., Chou, H.-W., Chen, J.-H., Lin, S.-H., Lu, Y.-Y., Tai, C.-C., Lin, Y.-F., & Lin, C.-Y. (2017). Low Levels of IgG Recognizing the α-1-Antitrypsin Peptide and Its Association with Taiwanese Women with Primary Sjögren’s Syndrome. International Journal of Molecular Sciences, 18(12), 2750. https://doi.org/10.3390/ijms18122750





