Physical, Structural, Barrier, and Antifungal Characterization of Chitosan–Zein Edible Films with Added Essential Oils
Abstract
:1. Introduction
2. Results and Discussion
2.1. Color, Transparency, and Optical Properties
2.2. Physical Properties
Mechanical Properties
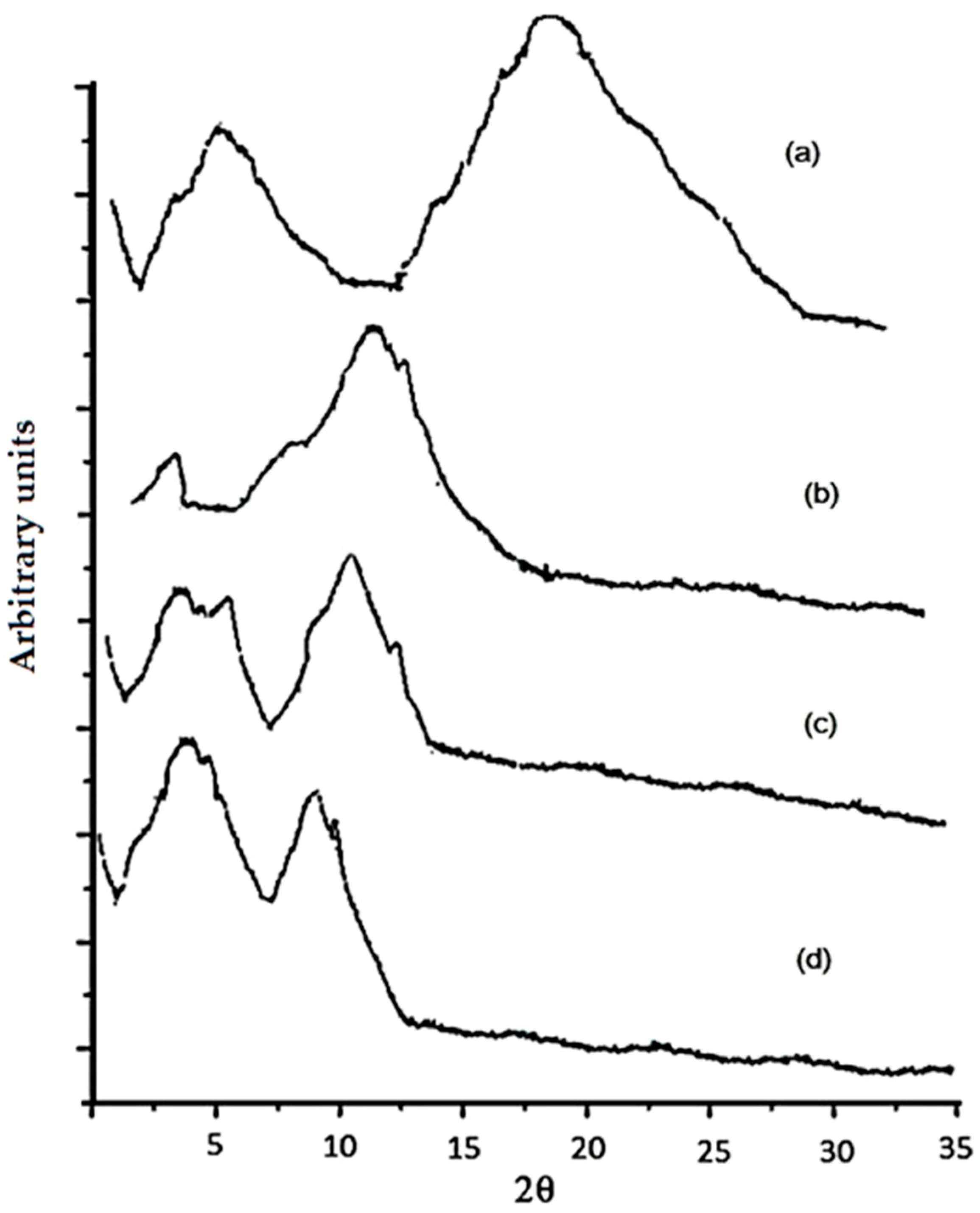
2.3. X-ray Diffraction
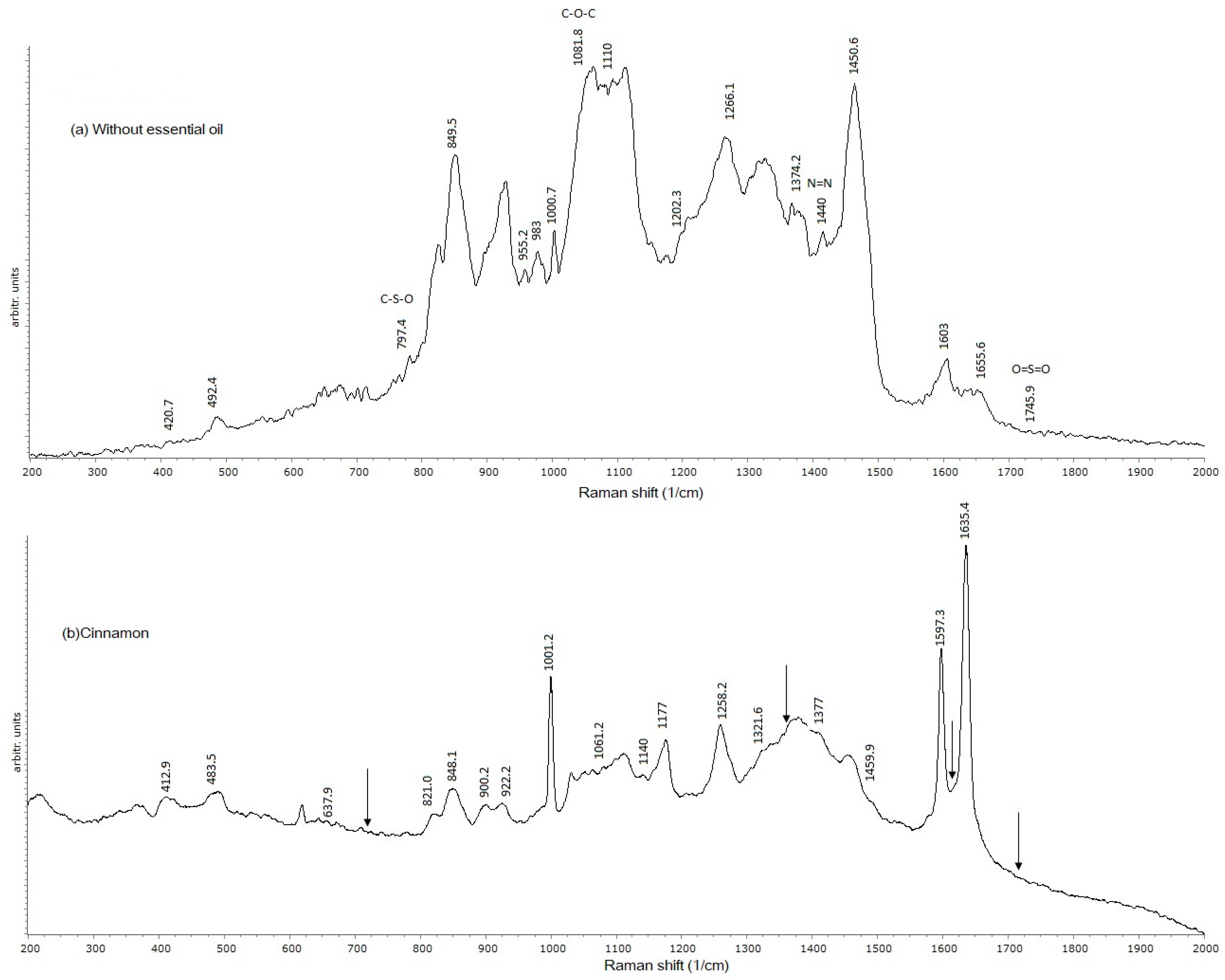
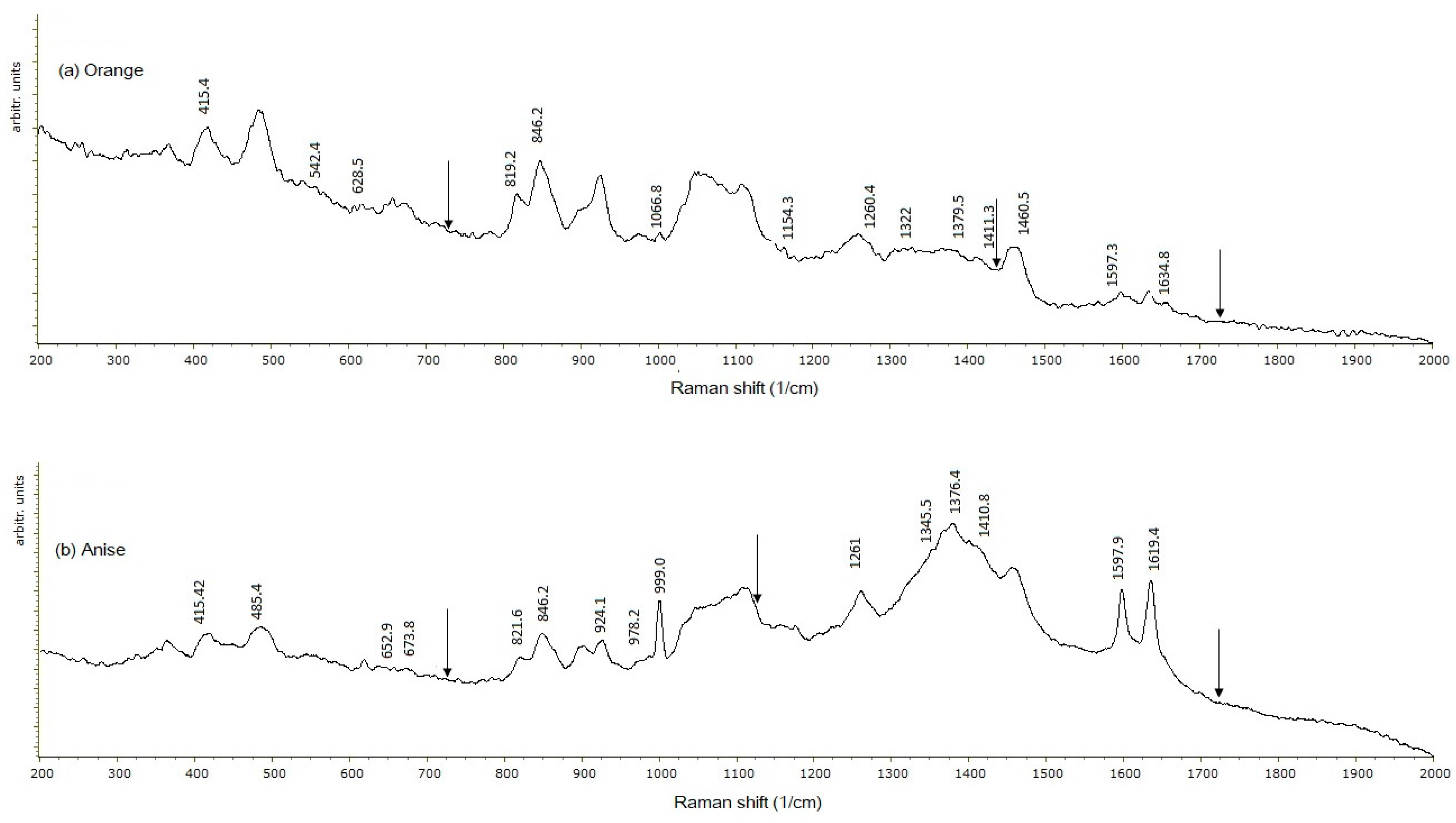
2.4. Raman Spectroscopy
2.5. Antifungal Activity
3. Materials and Methods
3.1. Materials
3.2. Films Preparation
3.3. Physical Characterization
3.3.1. Color, Transparency, and Optical Properties
3.3.2. Atomic Force Microscopy (AFM)
3.3.3. Water Vapor Permeability
3.3.4. Mechanical Properties
3.4. Spectroscopic Evaluation
3.4.1. X-ray Diffraction
3.4.2. Raman Spectroscopy
3.5. Microbiological Analysis
3.6. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Licciardello, F. Packaging, Blessing in Disguise. Review on Its Diverse Contribution to Food Sustainability. Trends Food Sci. Technol. 2017, 65, 32–39. [Google Scholar] [CrossRef]
- Ganiari, S.; Choulitoudi, E.; Oreopoulou, V. Edible and Active Films and Coatings as Carriers of Natural Antioxidant for Lipid Food. Trends Food Sci. Technol. 2017, 68, 70–82. [Google Scholar] [CrossRef]
- Ribeiro-Santos, R.; Andrade, M.; Ramos de Melo, N.; Sanches-Silva, A. Use of Essential Oils in Active Food Packaging: Recent Advances and Future Trends. Trends Food Sci. Technol. 2017, 61, 132–140. [Google Scholar] [CrossRef]
- Zhang, S.; Zhang, M.; Fang, Z.; Liu, Y. Preparation and Characterization of Blended Cloves/Cinnamon Essential Oil Nanoemulsions. LWT-Food Sci. Technol. 2017, 75, 316–322. [Google Scholar] [CrossRef]
- Pina-Pérez, M.; Martínez-López, A.; Rodrigo, D. Cinnamon Antimicrobial Effect Against Salmonella typhimurium Cells Treated by Pulsed Electric Fields (PEF) in Pasteurized Skim Milk beverage. Int. Food Res. J. 2012, 48, 777–783. [Google Scholar] [CrossRef]
- Noshirvani, N.; Ghanbarzadeh, B.; Garrat, C.; Rezaei, M.; Hashemi, M. Cinnamon and Ginger Essential Oils to Improve Antifungal, Physical and Mechanical Properties of Chitosan-Carboxymethyl Cellulose Films. Food Hydrocoll. 2017, 70, 36–45. [Google Scholar] [CrossRef]
- Matan, N.; Matan, N. Antifungal Activities of Anise Oil, Lime Oil and Tangerine Oil against Molds on Rubberwood (Hevea brasiliensis). Int. Biodeterior. Biodegrad. 2008, 62, 75–78. [Google Scholar] [CrossRef]
- Kosalec, I.; Pepeljnjak, S.; Kuš, S. Antifungal activity of fluid extract and essential oil from anise fruits (Pimpinella anisum L., Apiaceae). Acta Pharm. 2005, 5, 377–385. [Google Scholar]
- Matan, N. Antimicrobial activity of edible film incorporated with essential oils to preserve dried fish (Decapterus maruadsi). Int. Food Res. J. 2012, 19, 1733–1738. [Google Scholar]
- Acar, Ü.; Kesbic, O.S.; Yilmaz, S.; Gültepe, N.; Türker, A. Evaluation of the Effects of Essential Oil Extracted from Sweet Orange Peel (Citrus sinensis) on Growth Rate of Tilapia (Oreochromis mossambicus) and Possible Disease Resistance Against Streptococcus iniae. Aquaculture 2015, 437, 282–286. [Google Scholar] [CrossRef]
- Velázquez-Nuñez, M.; Ávila-Sosa, R.; Palou, E.; López-Malo, A. Antifungal Activity of Orange (Citrus sinensis var. Valencia) Peel Essential Oil. Food Control 2013, 31, 1–4. [Google Scholar] [CrossRef]
- Evageliou, V.; Saliari, D. Limonene Encapsulation in Freeze Dried Gellan Systems. Food Chem. 2017, 223, 72–75. [Google Scholar] [CrossRef] [PubMed]
- Duncan, T. Applications of Nanotechnology in Food Packaging and Food Safety: Barrier Materials, Antimicrobials and Sensors. J. Colloid Interface Sci. 2011, 363, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Chandrasekaran, R. X-ray Diffraction of Food Polysaccharides. Adv. Food Nutr. Res. 1998, 42, 131–210. [Google Scholar] [CrossRef] [PubMed]
- Coso, R.; Solís, J. Relation between Nonlinear Refractive Index and Third-Order Susceptibility in Absorbing Media. J. Opt. Soc. Am. B 2004, 21, 640–644. [Google Scholar] [CrossRef]
- Loftalian, A.; Jandaghian, A.; Saghafifar, H.; Mohajerani, E. Young’s Modulus Measurement Based on Surface Plasmon Resonance. Opt. Laser Technol. 2017, 94, 248–252. [Google Scholar] [CrossRef]
- Bonilla, J.; Vargas, M.; Atarés, L.; Chiralt, A. Physical Properties of Chitosan-Basil Essential Oil Edible Films as Affected by Oil Content and Homogenization Conditions. Procedia Food Sci. 2011, 1, 50–56. [Google Scholar] [CrossRef]
- Escamilla-García, M.; Calderón-Domínguez, G.; Chanona-Pérez, J.J.; Farrera-Rebollo, R.; Andraca-Adame, L.A.; Arzate-Vázquez, I.; Mendez-Mendez, J.V.; Moreno-Ruíz, L. Physical and Structural Characterisation of Zein and Chitosan Edible Films Using Nanotechnology Tools. Int. J. Biol. Macromol. 2013, 61, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Lu, Y.; Cui, H.; Jia, X.; Bai, M.Y. Factors Affecting the Physical Properties of Edible Composite Film Prepared from Zein and Wheat Gluten. Molecules 2012, 17, 3794–3804. [Google Scholar] [CrossRef] [PubMed]
- Atarés, L.; Bonilla, J.; Chiralt, A. Characterization of Sodium Caseinate-Based Edible Films Incorporated with Cinnamon or Ginger Essential Oils. J. Food Eng. 2010, 100, 678–687. [Google Scholar] [CrossRef]
- Aguirre, A.; Borneo, R.; León, A. Antimicrobial, Mechanical and Barrier Properties of Triticale Protein Films Incorporated with Oregano Essential Oil. Food Biosci. 2013, 1, 2–9. [Google Scholar] [CrossRef]
- Nisar, T.; Wang, Z.C.; Yang, X.; Tian, Y.; Iqbal, M.; Guo, Y. Characterization of Citrus Pectin Films Integrated with Clove Bud Essential Oil: Physical, Thermal, Barrier, Antioxidant and Antibacterial Properties. Int. J. Biol. Macromol. 2017, in press. [Google Scholar] [CrossRef] [PubMed]
- Acosta, S.; Chiralt, A.; Santamarina, P.; Rosello, J.; González-Martínez, C.; Cháfer, M. Antifungal Films Based on Starch-Gelatin Blend, Containing Essential Oils. Food Hydrocoll. 2016, 61, 233–240. [Google Scholar] [CrossRef]
- Peng, Y.; Li, Y. Combined Effects of Two Kinds of Essential Oils on Physical, Mechanical and Structural Properties of Chitosan Films. Food Hydrocoll. 2014, 36, 287–293. [Google Scholar] [CrossRef]
- Rocha Bastos, M.S.; Laurentino, L.S.; Canuto, K.; Guabiraba Mendes, L.; Mota Matin, C.; Frota Silva, S.; Ferro Furtado, R. Physical and Mechanical Testing of Essential Oil-Embedded Cellulose Ester Films. Polym. Test. 2016, 49, 156–161. [Google Scholar] [CrossRef]
- Zinoviadou, K.; Koutsoumanis, K.; Biliaderis, C. Biopolymer-Based Films as Carriers of Antimicrobial Agents. Procedia Food Sci. 2011, 1, 190–196. [Google Scholar] [CrossRef]
- Ghasemlou, M.; Khodaiyan, F.; Oromiehie, A. Rheological and Structural Characterisation of Film-Forming Solutions and Biodegradable Edible Film Made from Kefiran as Affected by Various Plasticizer Types. Int. J. Biol. Macromol. 2011, 49, 814–821. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-González, L.; Vargas, M.; González-Martínez, C.; Chiralt, A.; Cháfer, M. Use of Essential Oils in Bioactive Edible Coatings. Food Eng. Rev. 2011, 3, 1–16. [Google Scholar] [CrossRef]
- Jahed, E.; Khaledebed, M.A.; Almasi, H.; Hasanzadeh, R. Physicochemical Properties of Carum copticum Essential Oil Loaded Chitosan Films Coating Organic Nanoreinforcements. Carbohydr. Polym. 2017, 164, 325–338. [Google Scholar] [CrossRef] [PubMed]
- Gizem-Gezer, P.; Logan-Liu, G.; Kokini, J.L. Development of a Biodegradable Sensor Platform from Gold Coated Zein Nanofotonic Films to Detect Peanut Allergen, Ara h1, Using Surface enhanced Raman Spectroscopy. Talanta 2016, 150, 224–232. [Google Scholar] [CrossRef] [PubMed]
- Zając, A.; Hanuza, J.; Wandas, M.; Dymińska, L. Determination of N-acetylation Degree in Chitosan Using Raman Spectroscopy. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2015, 134, 114–120. [Google Scholar] [CrossRef] [PubMed]
- Gao, B.; Wan, M.; Men, J.; Zhang, Y. Aerobic Selective Oxidation of Benzyl Alcohols to Benzaldehyde Catalyzed by Bidentate Shift Base Dioxonolybdenum (VI) Complex Immobilized on CPS Microspheres. Appl. Catal. A 2012, 439–440, 156–162. [Google Scholar] [CrossRef]
- Meng, X.; Edgar, K. Synthesis of Amide-Functionalized Cellulose Esters by Olefin Cross-Metathesis. Carbohydr. Polym. 2015, 132, 565–573. [Google Scholar] [CrossRef] [PubMed]
- Berhe, D.; Engelsen, S.; Hviid, M.; Lametsch, R. Raman Spectroscopy Study of Effect of the Cooking Temperature and Time on Meat Proteins. Int. Food Res. J. 2014, 66, 123–131. [Google Scholar] [CrossRef]
- Baschieri, A.; Ajvazi, M.D.; Folifack-Tonfack, J.L.; Valgimigli, L.; Amorati, R. Explaining the Antioxidant Activity of Some Common Non-Phenolic Components of Essential Oils. Food Chem. 2017, 232, 656–663. [Google Scholar] [CrossRef] [PubMed]
- Ferdes, M.; Al Juhami, F.; Özcan, M.M.; Ghafoor, K. Inhibitory Effect of Some Plant Essential Oils on Growth of Aspergillus niger, Aspergillus oryzae, Mucor pusillus and Fusarium oxysporum. S. Afr. J. Bot. 2017, in press. [Google Scholar] [CrossRef]
- Aly, S.; Sabry, B.; Shaheen, M.; Hathout, A. Assessment of Antimycotoxigenic and Antioxidant Activity of Star Anse (Illicium verum) in Vitro. J. Saudi Soc. Agric. Sci. 2016, 15, 20–27. [Google Scholar] [CrossRef]
- Ribes, S.; Fuentes, A.; Talens, P.; Barat, J.M.; Ferrari, G.; Donsì, F. Influence of Emulsifier Type on Antifungal Activity of Cinnamon Leaf, Lemon, and Bergamot Oil Nanoemulsions against Aspergillus niger. Food Control 2017, 73, 784–795. [Google Scholar] [CrossRef]
- Van Long, N.N.; Dantigny, P. Fungal Contamination in Packaged Foods. Antimicrobial Food Packaging; Academic Press: Oxford, UK, 2016; pp. 45–59. ISBN 978-12-800723-5. [Google Scholar]
- Gao, P.; Wang, F.; Gu, F.; Ning, J.; Liang, J.; Li, N.; Ludescher, R.D. Preparation and Characterization of Zein Thermo-Modified Starch Films. Carbohydr. Polym. 2017, 157, 1254–1260. [Google Scholar] [CrossRef] [PubMed]
- Abugoch, L.; Tapia, C.; Villamán, M.; Yazdani-Pedram, M.; Díaz-Dosque, M. Characterization of Quinoa Protein Chitosan Blend Edible Film. Food Hydrocoll. 2011, 25, 879–886. [Google Scholar] [CrossRef]
- Murray, C.; Dutcher, J. Effect of Changes in Relative Humidity and Temperature on Ultrathin Chitosan Films. Biomacromolecules 2006, 7, 3460–3465. [Google Scholar] [CrossRef] [PubMed]
- Standard Test Method for Density and Relative Density (Specific Gravity) of Liquids by Bingham Pycnometer; ASTM International: West Conshohocken, PA, USA, 2012; ASTM D1217–12. [CrossRef]
- Ghanbarzadeh, B.; Oromiehi, A. Thermal and Mechanical Behavior of Laminated Protein Films. J. Food Eng. 2009, 90, 517–524. [Google Scholar] [CrossRef]
- American Society for Testing and Materials. Standard Test Method for Water Vapor Transmission of Materials. In ASTM Book of Standards; American Society for Testing and Materials: West Conshohocken, PA, USA, 1980; ASTM E96-80. [Google Scholar] [CrossRef]
- Alvarado-González, J.; Chanona-Pérez, J.J.; Welti-Chanes, J.; Calderón-Domínguez, G.; Arzate-Vázquez, I.; Gutierrez López, G.F.; Pacheco-Alcalá, L. Optical, Microstructural, Functional and Nanomechanical Properties of Aloe vera Gel/Gellan Gum Edible Films. Rev. Mex. Ing. Quim. 2012, 11, 193–210. [Google Scholar]
- Gennadios, A.; Weller, C.; Gooding, C. Measurement Errors in Water Vapor Permeability of Highly Permeable, Hydrophilic Edible Films. J. Food Eng. 1994, 21, 395–400. [Google Scholar] [CrossRef]
- Smith, W.E.; Dent, G. Modern Raman Spectroscopy—A Practical Approach; John Wiley & Sons: West Sussex, UK, 2005; pp. 11–31. [Google Scholar]
- Boonruang, L.; Kerddonfag, N.; Chinsnrikul, W.; Mitcham, E.J.; Chohenchob, V. Antifungl Effrect of Poly(Lactic Acid) Films Containing thymol and R-(−)-Carvone against Anthractnose Pathogens Isolated from Avocado and Citrus. Food Control 2017, 78, 85–93. [Google Scholar] [CrossRef]
| Edible Film | % T | a* | b* | η |
|---|---|---|---|---|
| Without oil | 88.4 ± 0.7 a | −1.34 ± 0.02 a | 12.4 ± 2.2 a | 1.45 ± 0.02 a |
| Anise | 72.6 ± 1.4 b | −1.10 ± 0.01 b | 22.6 ± 1.3 b | 1.35 ± 0.02 b |
| Orange | 71.6 ± 1.1 b,c | −1.70 ± 0.02 c | 27.8 ± 1.2 c | 1.55 ± 0.01 a,c |
| Cinnamon | 69.3 ± 1.1 c | −1.42 ± 0.01 d | 28.8 ± 1.3 c | 1.50 ± 0.02 c |
| Edible Film | e (µm) | ρ (g cm−3) | WVP (g mm h−1 m−2 kPa−1) | Rq (nm) | Ra (nm) |
|---|---|---|---|---|---|
| Without oil | 20.02 ± 1.45 a | 1.33 ± 0.02 a | 2.92 ± 0.16 a | 13.94 ± 0.09 a | 12.63 ± 0.06 a |
| Anise | 23.92 ± 0.92 b | 1.72 ± 0.01 b | 1.21 ± 0.10 b | 11.63 ± 0.14 b | 9.12 ± 0.12 b |
| Orange | 21.43 ± 0.51 c | 1.42 ± 0.02 a | 1.62 ± 0.02 c | 7.24 ± 0.11 c | 5.64 ± 0.16 c |
| Cinnamon | 22.54 ± 0.32 d | 1.54 ± 0.02 c | 1.53 ± 0.20 b,c | 3.62 ± 0.12 d | 2.84 ± 0.09 d |
| Edible Film | Hardness (MPa) | Elastic Modulus (MPa) |
|---|---|---|
| Without oil | 5.91 ± 0.41 a | 66 ± 6 a |
| Anise | 104.33 ± 3.22 b | 22 ± 3 b |
| Orange | 57.51 ± 2.43 c | 20 ± 2 b |
| Cinnamon | 25.54 ± 1.85 d | 24 ± 3 b |
| Edible Film | Average Diameter (cm) | |
|---|---|---|
| Penicillium sp. | Rhizopus sp. | |
| Without essential oil | 0 | 0 |
| Anise essential oil | 2.2 ± 0.2 a | 1.5 ± 0.3 a |
| Cinnamon essential oil | 1.9 ± 0.3 a | 1.7 ± 0.3 a |
| Orange essential oil | 0.7 ± 0.1 b | 0.6 ± 0.2 b |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Escamilla-García, M.; Calderón-Domínguez, G.; Chanona-Pérez, J.J.; Mendoza-Madrigal, A.G.; Di Pierro, P.; García-Almendárez, B.E.; Amaro-Reyes, A.; Regalado-González, C. Physical, Structural, Barrier, and Antifungal Characterization of Chitosan–Zein Edible Films with Added Essential Oils. Int. J. Mol. Sci. 2017, 18, 2370. https://doi.org/10.3390/ijms18112370
Escamilla-García M, Calderón-Domínguez G, Chanona-Pérez JJ, Mendoza-Madrigal AG, Di Pierro P, García-Almendárez BE, Amaro-Reyes A, Regalado-González C. Physical, Structural, Barrier, and Antifungal Characterization of Chitosan–Zein Edible Films with Added Essential Oils. International Journal of Molecular Sciences. 2017; 18(11):2370. https://doi.org/10.3390/ijms18112370
Chicago/Turabian StyleEscamilla-García, Monserrat, Georgina Calderón-Domínguez, Jorge J. Chanona-Pérez, Angélica G. Mendoza-Madrigal, Prospero Di Pierro, Blanca E. García-Almendárez, Aldo Amaro-Reyes, and Carlos Regalado-González. 2017. "Physical, Structural, Barrier, and Antifungal Characterization of Chitosan–Zein Edible Films with Added Essential Oils" International Journal of Molecular Sciences 18, no. 11: 2370. https://doi.org/10.3390/ijms18112370
APA StyleEscamilla-García, M., Calderón-Domínguez, G., Chanona-Pérez, J. J., Mendoza-Madrigal, A. G., Di Pierro, P., García-Almendárez, B. E., Amaro-Reyes, A., & Regalado-González, C. (2017). Physical, Structural, Barrier, and Antifungal Characterization of Chitosan–Zein Edible Films with Added Essential Oils. International Journal of Molecular Sciences, 18(11), 2370. https://doi.org/10.3390/ijms18112370







