A Unique TGFB1-Driven Genomic Program Links Astrocytosis, Low-Grade Inflammation and Partial Demyelination in Spinal Cord Periplaques from Progressive Multiple Sclerosis Patients
Abstract
:1. Introduction
2. Results
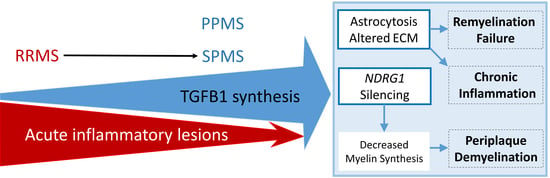
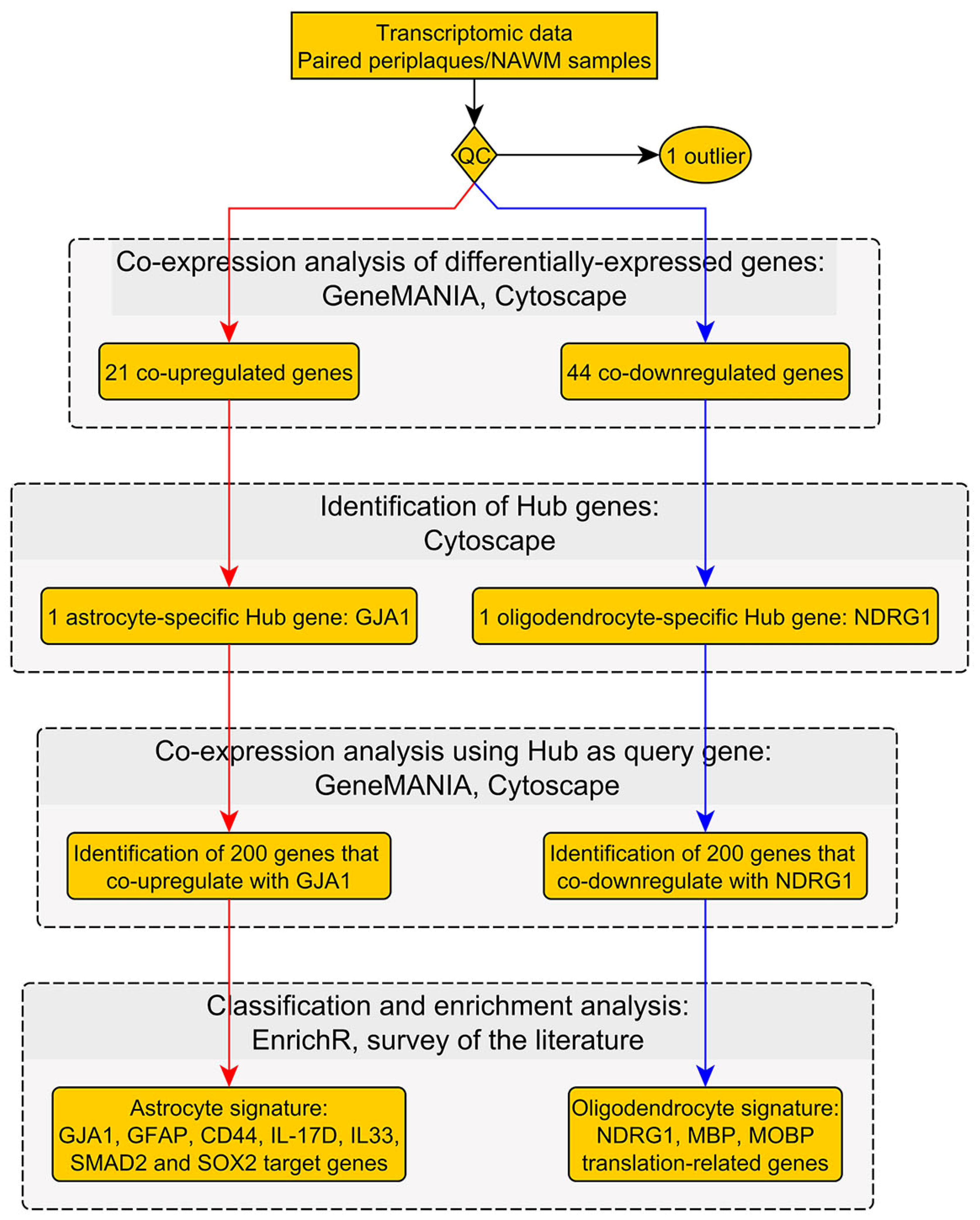
2.1. Workflow and Results Summary
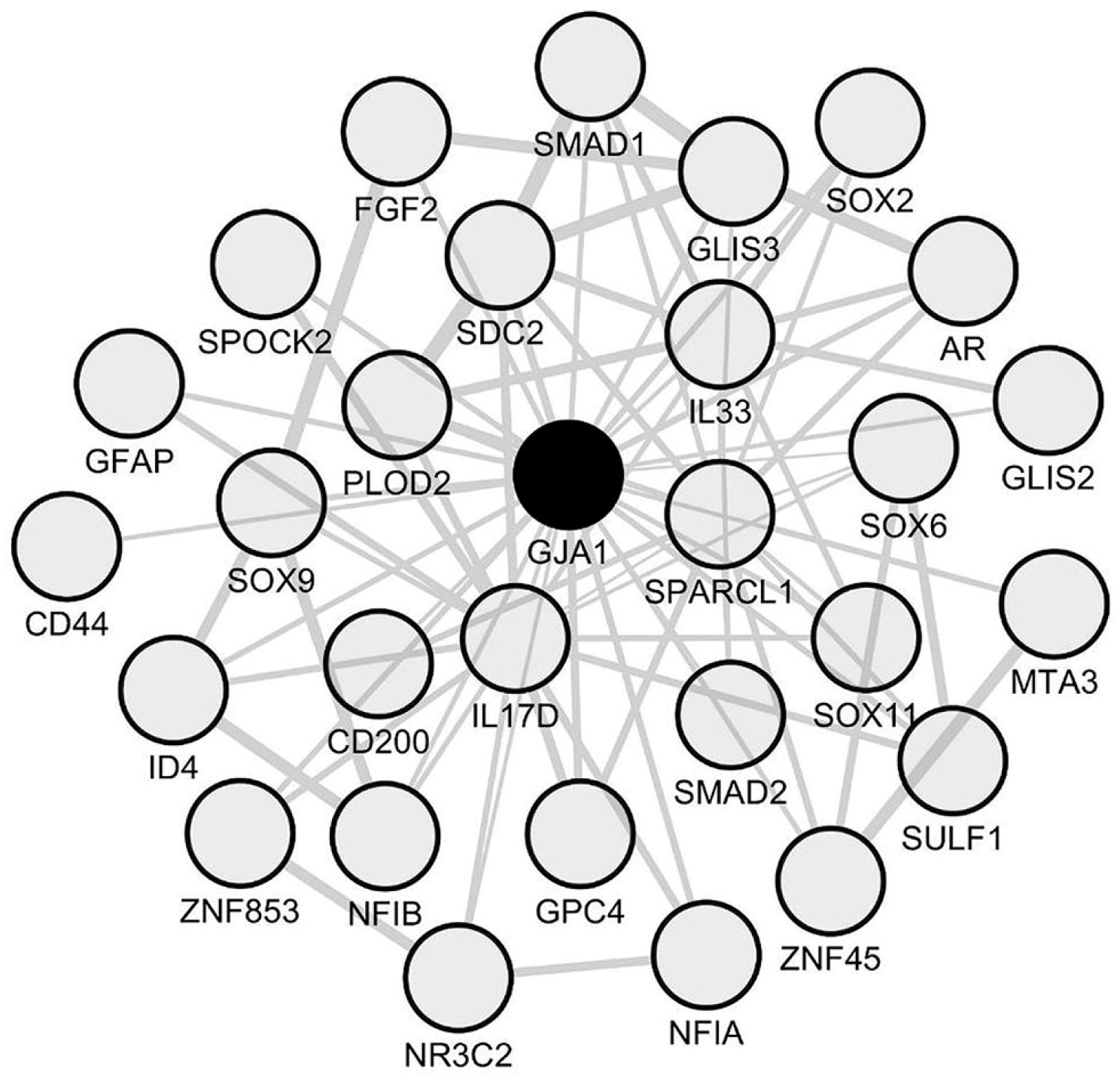
2.2. Identification of Cx43/GJA1 as an Upregulated Astrocyte-Related Hub Gene in Spinal Cord Periplaques
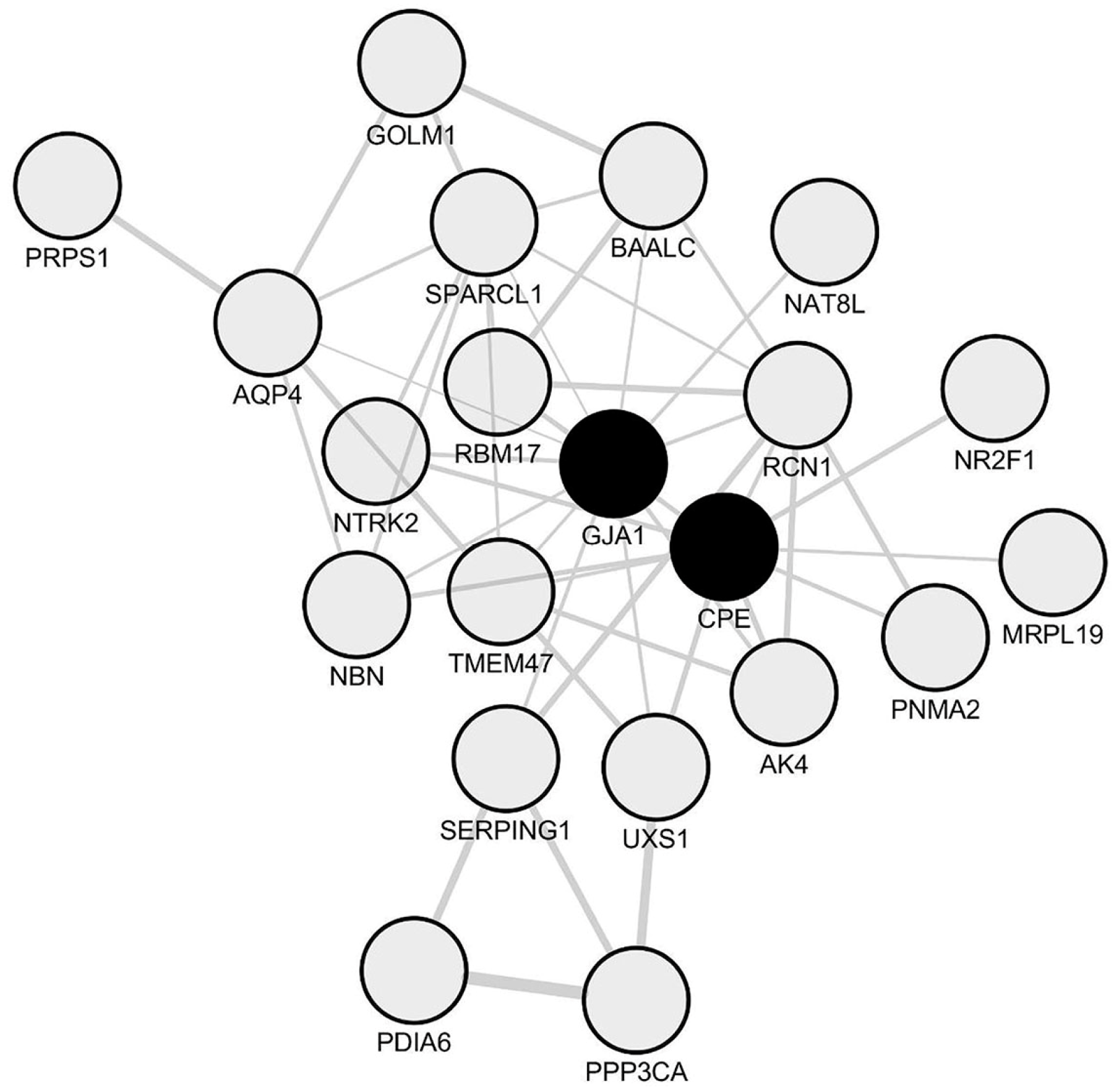
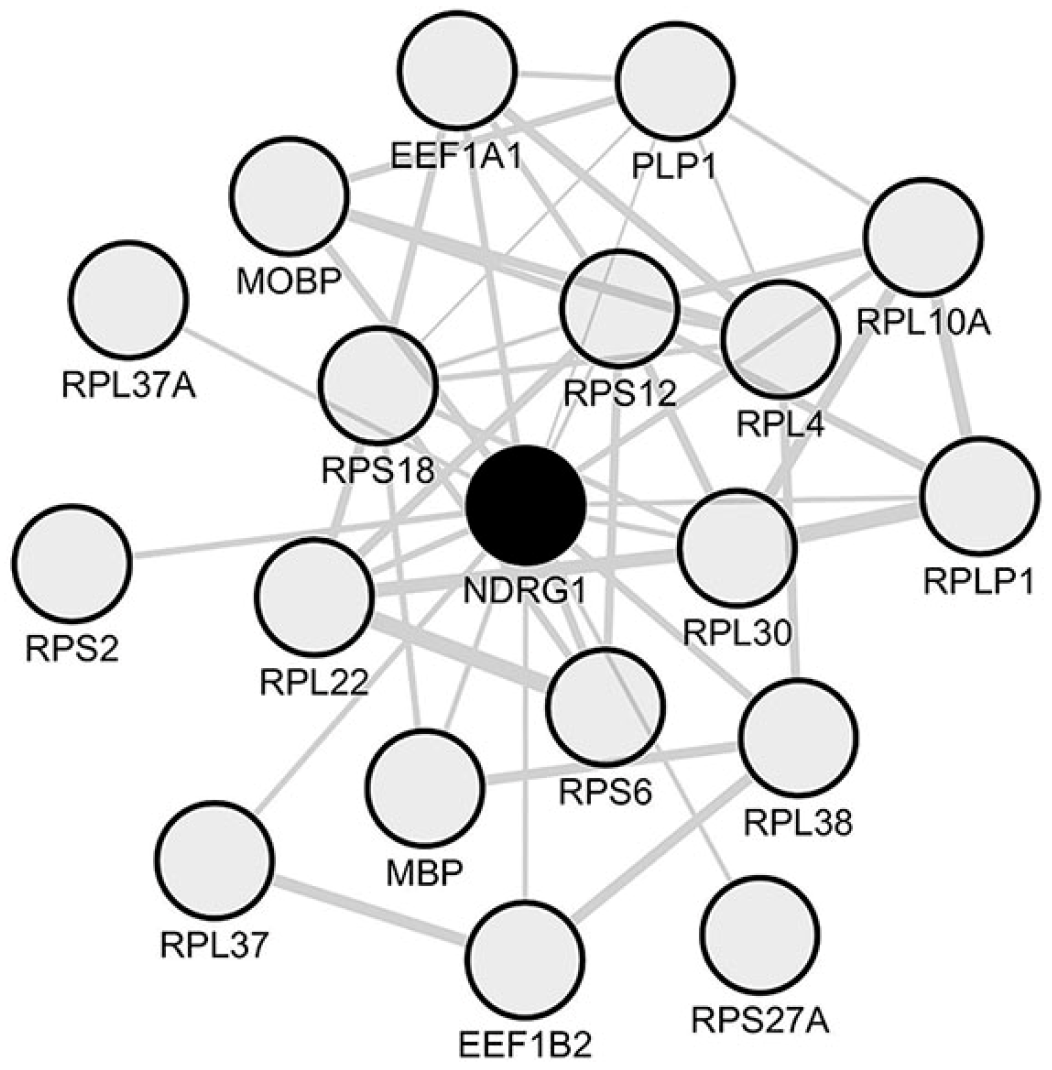
2.3. Identification of NDRG1 as a Downregulated Oligodendrocyte-Related Hub Gene in Spinal Cord Periplaques
2.4. A Unique Set of Genes Co-Upregulate with CX43/GJA1 in Periplaques of MS Spinal Cords
2.5. A Unique Set of Genes Co-Express with NDRG1 in Periplaque Areas of MS Spinal Cords
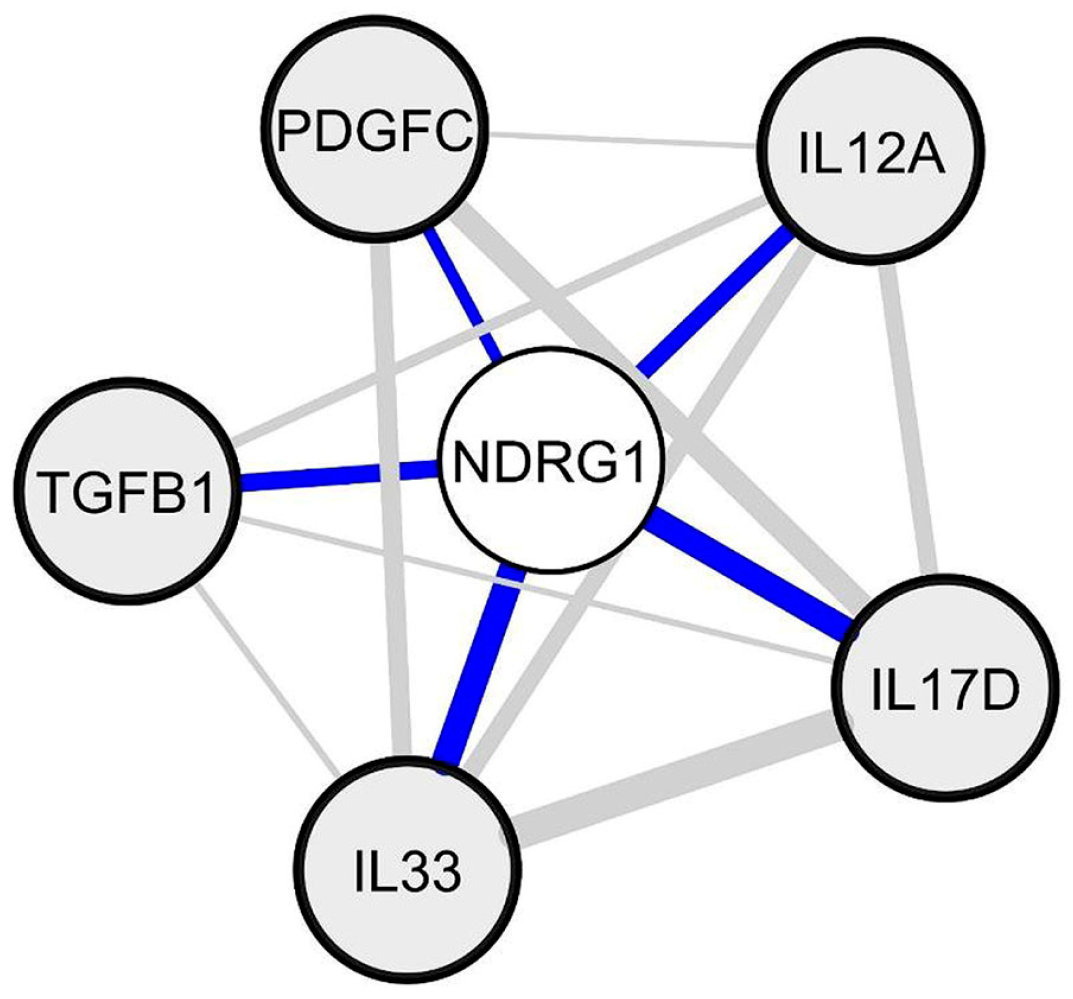
2.6. Identification of Candidate Soluble Factors that May Trigger NDRG1 Silencing in Periplaque Areas of MS Spinal Cords
2.7. Assessment in Normal Human Astrocytes of Genes Associated with the Identified Cx43/GJA1 Module
2.8. Assessment in Normal Human Oligodendrocytes of Genes Associated with the Identified NDRG1 Module
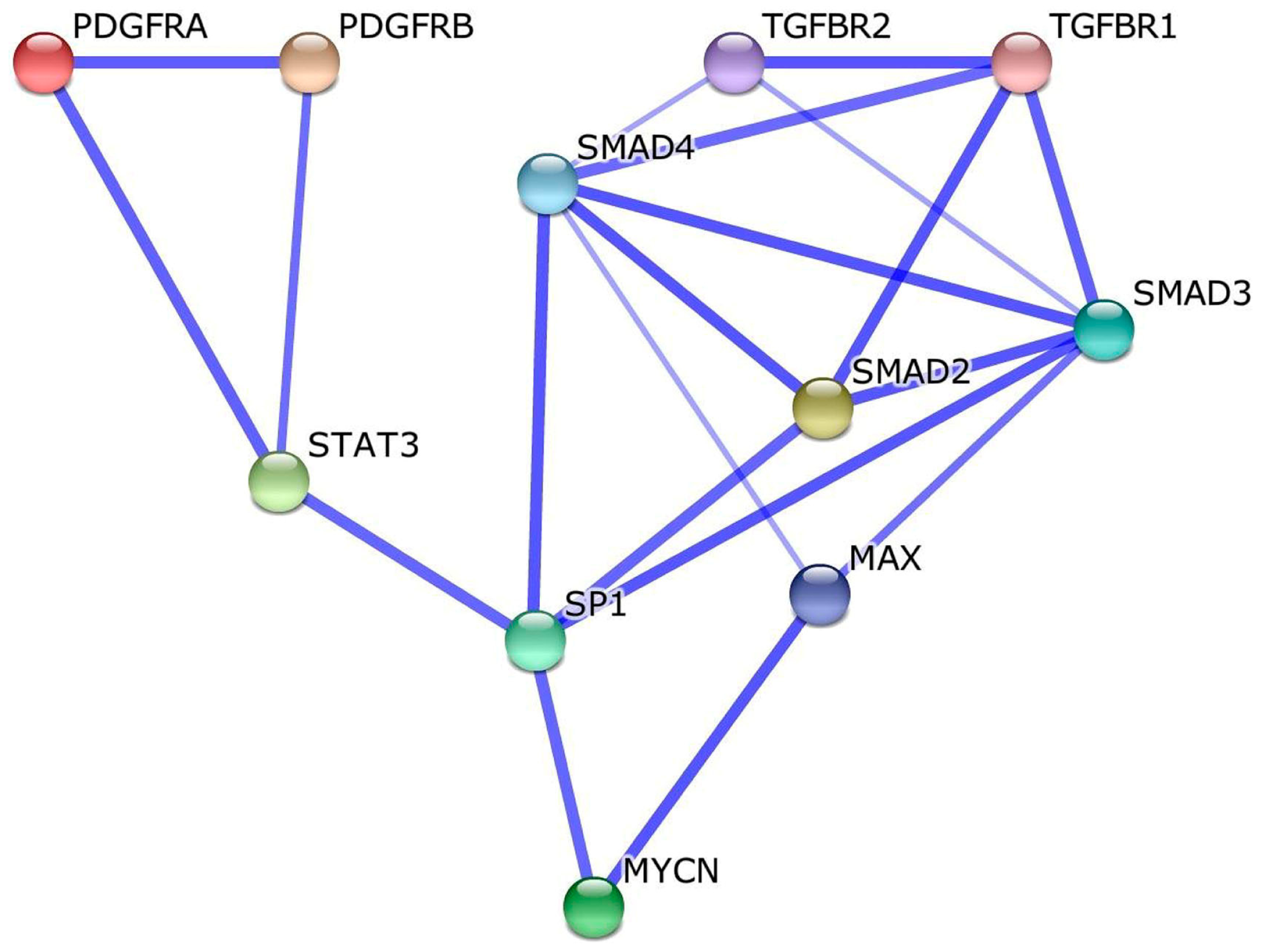
2.9. Identification of a Molecular Pathway Linking MYCN ((MYCN proto-oncogene, bHLH transcription factor) to PDGFC and TGFB1
3. Discussion
4. Materials and Methods
4.1. Transcriptomic Data
4.2. Generation of a Global Co-Expression Network
4.3. Connectivity Analysis of Gene Co-Expression Networks
4.4. Identification and Analysis of the Cx43/GJA1 Co-Upregulated Gene Network
4.5. Identification and Analysis of the NDRG1 Co-Downregulated Gene Network
4.6. Identification of aNetwork of NDRG1 Inversely Correlated Genes Coding for Cytokines/Chemokines or Growth Factors
4.7. Meta-Analysis of MYCN Protein Interactants
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| MS | Multiple sclerosis |
| RRMS | Relapsing-remitting multiple sclerosis |
| SPMS | Secondary progressive multiple sclerosis |
| PPMS | Primary progressive multiple sclerosis |
| NAWM | Normal-appearing white matter |
References
- Compston, A.; Coles, A. Multiple sclerosis. Lancet 2002, 359, 1221–1231. [Google Scholar] [CrossRef]
- Antel, J.; Antel, S.; Caramanos, Z.; Arnold, D.L.; Kuhlmann, T. Primary progressive multiple sclerosis: Part of the MS disease spectrum or separate disease entity? Acta. Neuropathol. 2012, 123, 627–638. [Google Scholar] [CrossRef] [PubMed]
- Ciccarelli, O.; Barkhof, F.; Bodini, B.; de Stefano, N.; Golay, X.; Nicolay, K.; Pelletier, D.; Pouwels, P.J.W.; Smith, S.A.; et al. Pathogenesis of multiple sclerosis: Insights from molecular and metabolic imaging. Lancet Neurol. 2014, 13, 807–822. [Google Scholar] [CrossRef]
- Larochelle, C.; Uphaus, T.; Prat, A.; Zipp, F. Secondary progression in multiple sclerosis: Neuronal exhaustion or distinct pathology? Trends Neurosci. 2016, 39, 325–339. [Google Scholar] [CrossRef] [PubMed]
- Lassmann, H.; van Horssen, J.; Mahad, D. Progressive multiple sclerosis: Pathology and pathogenesis. Nat. Rev. Neurol. 2012, 8, 647–656. [Google Scholar] [CrossRef] [PubMed]
- Kearney, H.; Miller, D.H.; Ciccarelli, O. Spinal cord MRI in multiple sclerosis—Diagnostic, prognostic and clinical value. Nat. Rev. Neurol. 2015, 11, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Kearney, H.; Altmann, D.R.; Samson, R.S.; Yiannakas, M.C.; Wheeler-Kingshott, C.A.M.; Ciccarelli, O.; Miller, D.H. Cervical cord lesion load is associated with disability independently from atrophy in MS. Neurology 2015, 84, 367–373. [Google Scholar] [CrossRef] [PubMed]
- Lieury, A.; Chanal, M.; Androdias, G.; Reynolds, R.; Cavagna, S.; Giraudon, P.; Confavreux, C.; Nataf, S. Tissue remodeling in periplaque regions of multiple sclerosis spinal cord lesions. Glia 2014, 62, 1645–1658. [Google Scholar] [CrossRef] [PubMed]
- Zeis, T.; Graumann, U.; Reynolds, R.; Schaeren-Wiemers, N. Normal-appearing white matter in multiple sclerosis is in a subtle balance between inflammation and neuroprotection. Brain 2007, 131, 288–303. [Google Scholar] [CrossRef] [PubMed]
- Melief, J.; de Wit, S.J.; van Eden, C.G.; Teunissen, C.; Hamann, J.; Uitdehaag, B.M.; Swaab, D.; Huitinga, I. HPA axis activity in multiple sclerosis correlates with disease severity, lesion type and gene expression in normal-appearing white matter. Acta Neuropathol. 2013, 126, 237–249. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, C.; Mirakhur, M.; Kirk, J.; Farrell, M.; McQuaid, S. Up-regulation of osteopontin and alphaBeta-crystallin in the normal-appearing white matter of multiple sclerosis: An immunohistochemical study utilizing tissue microarrays. Neuropathol. Appl. Neurobiol. 2005, 31, 292–303. [Google Scholar] [CrossRef] [PubMed]
- Seewann, A.; Vrenken, H.; van der Valk, P.; Blezer, E.L.A.; Knol, D.L.; Castelijns, J.A.; Polman, C.H.; Pouwels, P.J.W.; Barkhof, F.; Geurts, J.J.G. Diffusely abnormal white matter in chronic multiple sclerosis: Imaging and histopathologic analysis. Arch. Neurol. 2009, 66, 601–609. [Google Scholar] [CrossRef] [PubMed]
- Mellergård, J.; Tisell, A.; Dahlqvist Leinhard, O.; Blystad, I.; Landtblom, A.-M.; Blennow, K.; Olsson, B.; Dahle, C.; Ernerudh, J.; Lundberg, P.; et al. Association between change in normal appearing white matter metabolites and intrathecal inflammation in natalizumab-treated multiple sclerosis. PLoS ONE 2012, 7, e44739. [Google Scholar] [CrossRef] [PubMed]
- Trotter, J.L.; Wegescheide, C.L.; Garvey, W.F.; Tourtellotte, W.W. Studies of myelin proteins in multiple sclerosis brain tissue. Neurochem. Res. 1984, 9, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Johnson, D.; Sato, S.; Quarles, R.H.; Inuzuka, T.; Brady, R.O.; Tourtellotte, W.W. Quantitation of the myelin-associated glycoprotein in human nervous tissue from controls and multiple sclerosis patients. J. Neurochem. 1986, 46, 1086–1093. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, D.; Bandaru, V.V.R.; Calabresi, P.A.; Nath, A.; Haughey, N.J. A defect of sphingolipid metabolism modifies the properties of normal appearing white matter in multiple sclerosis. Brain 2008, 131, 3092–3102. [Google Scholar] [CrossRef] [PubMed]
- Huynh, J.L.; Garg, P.; Thin, T.H.; Yoo, S.; Dutta, R.; Trapp, B.D.; Haroutunian, V.; Zhu, J.; Donovan, M.J.; Sharp, A.J.; et al. Epigenome-wide differences in pathology-free regions of multiple sclerosis-affected brains. Nat. Neurosci. 2014, 17, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Markoullis, K.; Sargiannidou, I.; Schiza, N.; Hadjisavvas, A.; Roncaroli, F.; Reynolds, R.; Kleopa, K.A. Gap junction pathology in multiple sclerosis lesions and normal-appearing white matter. Acta Neuropathol. 2012, 123, 873–886. [Google Scholar] [CrossRef] [PubMed]
- Echaniz-Laguna, A.; Degos, B.; Bonnet, C.; Latour, P.; Hamadouche, T.; Lévy, N.; Leheup, B. NDRG1-linked Charcot-Marie-Tooth disease (CMT4D) with central nervous system involvement. Neuromuscul. Disord. 2007, 17, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Pietiainen, V.; Vassilev, B.; Blom, T.; Wang, W.; Nelson, J.; Bittman, R.; Back, N.; Zelcer, N.; Ikonen, E. NDRG1 functions in LDL receptor trafficking by regulating endosomal recycling and degradation. J. Cell Sci. 2013, 126, 3961–3971. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Lin, A.H.; Masliah, E.; Wyss-Coray, T. Bioluminescence imaging of Smad signaling in living mice shows correlation with excitotoxic neurodegeneration. Proc. Natl. Acad. Sci. USA 2006, 103, 18326–18331. [Google Scholar] [CrossRef] [PubMed]
- Schachtrup, C.; Ryu, J.K.; Helmrick, M.J.; Vagena, E.; Galanakis, D.K.; Degen, J.L.; Margolis, R.U.; Akassoglou, K. Fibrinogen triggers astrocyte scar formation by promoting the availability of active TGF- after vascular damage. J. Neurosci. 2010, 30, 5843–5854. [Google Scholar] [CrossRef] [PubMed]
- Gomes, W.A.; Mehler, M.F.; Kessler, J.A. Transgenic overexpression of BMP4 increases astroglial and decreases oligodendroglial lineage commitment. Dev. Biol. 2003, 255, 164–177. [Google Scholar] [CrossRef]
- Fuller, M.L.; DeChant, A.K.; Rothstein, B.; Caprariello, A.; Wang, R.; Hall, A.K.; Miller, R.H. Bone morphogenetic proteins promote gliosis in demyelinating spinal cord lesions. Ann. Neurol. 2007, 62, 288–300. [Google Scholar] [CrossRef] [PubMed]
- Wanner, I.B.; Anderson, M.A.; Song, B.; Levine, J.; Fernandez, A.; Gray-Thompson, Z.; Ao, Y.; Sofroniew, M.V. Glial scar borders are formed by newly proliferated, elongated astrocytes that interact to corral inflammatory and fibrotic cells via STAT3-dependent mechanisms after spinal cord injury. J. Neurosci. 2013, 33, 12870–12886. [Google Scholar] [CrossRef] [PubMed]
- Robel, S.; Berninger, B.; Götz, M. The stem cell potential of glia: Lessons from reactive gliosis. Nat. Rev. Neurosci. 2011, 12, 88–104. [Google Scholar] [CrossRef] [PubMed]
- Brun, M.; Coles, J.E.; Monckton, E.A.; Glubrecht, D.D.; Bisgrove, D.; Godbout, R. Nuclear factor I regulates brain fatty acid-binding protein and glial fibrillary acidic protein gene expression in malignant glioma cell lines. J. Mol. Biol. 2009, 391, 282–300. [Google Scholar] [CrossRef] [PubMed]
- Hussain, R.; Ghoumari, A.M.; Bielecki, B.; Steibel, J.; Boehm, N.; Liere, P.; Macklin, W.B.; Kumar, N.; Habert, R.; Mhaouty-Kodja, S.; et al. The neural androgen receptor: A therapeutic target for myelin repair in chronic demyelination. Brain 2013, 136, 132–146. [Google Scholar] [CrossRef] [PubMed]
- Acaz-Fonseca, E.; Avila-Rodriguez, M.; Garcia-Segura, L.M.; Barreto, G.E. Regulation of astroglia by gonadal steroid hormones under physiological and pathological conditions. Prog. Neurobiol. 2016, 144, 5–26. [Google Scholar] [CrossRef] [PubMed]
- Coers, S.; Tanzer, L.; Jones, K.J. Testosterone treatment attenuates the effects of facial nerve transection on glial fibrillary acidic protein (GFAP) levels in the hamster facial motor nucleus. Metab. Brain Dis. 2002, 17, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Barreto, G.; Veiga, S.; Azcoitia, I.; Garcia-Segura, L.M.; Garcia-Ovejero, D. Testosterone decreases reactive astroglia and reactive microglia after brain injury in male rats: Role of its metabolites, oestradiol and dihydrotestosterone. Eur. J. Neurosci. 2007, 25, 3039–3046. [Google Scholar] [CrossRef] [PubMed]
- Arevalo, M.A.; Santos-Galindo, M.; Acaz-Fonseca, E.; Azcoitia, I.; Garcia-Segura, L.M. Gonadal hormones and the control of reactive gliosis. Horm. Behav. 2013, 63, 216–221. [Google Scholar] [CrossRef] [PubMed]
- Giatti, S.; Rigolio, R.; Romano, S.; Mitro, N.; Viviani, B.; Cavaletti, G.; Caruso, D.; Garcia-Segura, L.M.; Melcangi, R.C. Dihydrotestosterone as a protective agent in chronic experimental autoimmune encephalomyelitis. Neuroendocrinology 2015, 101, 296–308. [Google Scholar] [CrossRef] [PubMed]
- Lachmann, A.; Xu, H.; Krishnan, J.; Berger, S.I.; Mazloom, A.R.; Ma’ayan, A. ChEA: Transcription factor regulation inferred from integrating genome-wide ChIP-X experiments. Bioinformatics 2010, 26, 2438–2444. [Google Scholar] [CrossRef] [PubMed]
- Fabregat, A.; Sidiropoulos, K.; Garapati, P.; Gillespie, M.; Hausmann, K.; Haw, R.; Jassal, B.; Jupe, S.; Korninger, F.; McKay, S.; et al. The reactome pathway knowledgebase. Nucleic Acids Res. 2016, 44, D481–D487. [Google Scholar] [CrossRef] [PubMed]
- Koning, N.; Swaab, D.F.; Hoek, R.M.; Huitinga, I. Distribution of the immune inhibitory molecules CD200 and CD200R in the normal central nervous system and multiple sclerosis lesions suggests neuron–glia and glia–glia interactions. J. Neuropathol. Exp. Neurol. 2009, 68, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Gabel, S.; Koncina, E.; Dorban, G.; Heurtaux, T.; Birck, C.; Glaab, E.; Michelucci, A.; Heuschling, P.; Grandbarbe, L. Inflammation promotes a conversion of astrocytes into neural progenitor cells via NF-κB activation. Mol. Neurobiol. 2016, 53, 5041–5055. [Google Scholar] [CrossRef] [PubMed]
- Allen, N.J.; Bennett, M.L.; Foo, L.C.; Wang, G.X.; Chakraborty, C.; Smith, S.J.; Barres, B.A. Astrocyte glypicans 4 and 6 promote formation of excitatory synapses via GluA1 AMPA receptors. Nature 2012, 486, 410–414. [Google Scholar] [CrossRef] [PubMed]
- Higginson, J.R.; Thompson, S.M.; Santos-Silva, A.; Guimond, S.E.; Turnbull, J.E.; Barnett, S.C. Differential sulfation remodelling of heparan sulfate by extracellular 6-O-sulfatases regulates fibroblast growth factor-induced boundary formation by glial cells: Implications for glial cell transplantation. J. Neurosci. 2012, 32, 15902–15912. [Google Scholar] [CrossRef] [PubMed]
- Lively, S.; Moxon-Emre, I.; Schlichter, L.C. SC1/hevin and reactive gliosis after transient ischemic stroke in young and aged rats. J. Neuropathol. Exp. Neurol. 2011, 70, 913–929. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, V.; Bell, G.W.; Nam, J.-W.; Bartel, D.P. Predicting effective microRNA target sites in mammalian mRNAs. Elife 2015, 4. [Google Scholar] [CrossRef] [PubMed]
- Gould, R.M.; Freund, C.M.; Barbarese, E. Myelin-associated oligodendrocytic basic protein mRNAs reside at different subcellular locations. J. Neurochem. 1999, 73, 1913–1924. [Google Scholar] [PubMed]
- Torvund-Jensen, J.; Steengaard, J.; Reimer, L.; Fihl, L.B.; Laursen, L.S. Transport and translation of MBP mRNA is regulated differently by distinct hnRNP proteins. J. Cell Sci. 2014, 127, 1550–1564. [Google Scholar] [CrossRef] [PubMed]
- Müller, C.; Bauer, N.M.; Schäfer, I.; White, R. Making myelin basic protein-from mRNA transport to localized translation. Front. Cell. Neurosci. 2013, 7, 169. [Google Scholar] [CrossRef] [PubMed]
- Seiberlich, V.; Bauer, N.G.; Schwarz, L.; Ffrench-Constant, C.; Goldbaum, O.; Richter-Landsberg, C. Downregulation of the microtubule associated protein Tau impairs process outgrowth and myelin basic protein mRNA transport in oligodendrocytes. Glia 2015, 63, 1621–1635. [Google Scholar] [CrossRef] [PubMed]
- Colman, D.R.; Kreibich, G.; Frey, A. B.; Sabatini, D.D. Synthesis and incorporation of myelin polypeptides into CNS myelin. J. Cell Biol. 1982, 95, 598–608. [Google Scholar] [CrossRef] [PubMed]
- Gould, R.M.; Freund, C.M.; Palmer, F.; Feinstein, D.L. Messenger RNAs located in myelin sheath assembly sites. J. Neurochem. 2000, 75, 1834–1844. [Google Scholar] [CrossRef] [PubMed]
- Basha, O.; Barshir, R.; Sharon, M.; Lerman, E.; Kirson, B.F.; Hekselman, I.; Yeger-Lotem, E. The TissueNet v.2 database: A quantitative view of protein-protein interactions across human tissues. Nucleic Acids Res. 2017, 45, D427–D431. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Sloan, S.A.; Clarke, L.E.; Caneda, C.; Plaza, C.A.; Blumenthal, P.D.; Vogel, H.; Steinberg, G.K.; Edwards, M.S.B.; Li, G.; et al. Purification and characterization of progenitor and mature human astrocytes reveals transcriptional and functional differences with mouse. Neuron 2016, 89, 37–53. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Chen, K.; Sloan, S.A.; Bennett, M.L.; Scholze, A.R.; O’Keeffe, S.; Phatnani, H.P.; Guarnieri, P.; Caneda, C.; Ruderisch, N.; et al. An RNA-sequencing transcriptome and splicing database of glia, neurons, and vascular cells of the cerebral cortex. J. Neurosci. 2014, 34, 11929–11947. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Sloan, S.; Barres, B.A. Purification of functional human astrocytes. U.S. Patent Application No. 15/158,404, 24 November 2016. [Google Scholar]
- Li, J.; Kretzner, L. The growth-inhibitory NDRG1 gene is a Myc negative target in human neuroblastomas and other cell types with overexpressed N- or C-myc. Mol. Cell. Biochem. 2003, 250, 91–105. [Google Scholar] [CrossRef] [PubMed]
- Shimono, A.; Okuda, T.; Kondoh, H. N-myc-dependent repression of ndr1, a gene identified by direct subtraction of whole mouse embryo cDNAs between wild type and N-myc mutant. Mech. Dev. 1999, 83, 39–52. [Google Scholar] [CrossRef]
- Gherardi, S.; Valli, E.; Erriquez, D.; Perini, G. MYCN-mediated transcriptional repression in neuroblastoma: The other side of the coin. Front. Oncol. 2013, 3, 42. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Chen, S.; Zhang, W.; Zhang, J.; Liu, X.; Shi, H.; Che, H.; Wang, W.; Li, F.; Yao, L. Human differentiation-related gene NDRG1 is a Myc downstream-regulated gene that is repressed by Myc on the core promoter region. Gene 2008, 417, 5–12. [Google Scholar] [CrossRef] [PubMed]
- Ellen, T.P.; Ke, Q.; Zhang, P.; Costa, M. NDRG1, a growth and cancer related gene: Regulation of gene expression and function in normal and disease states. Carcinogenesis 2008, 29, 2–8. [Google Scholar] [CrossRef] [PubMed]
- Ambrosio, S.; Amente, S.; Saccà, C.D.; Capasso, M.; Calogero, R.A.; Lania, L.; Majello, B. LSD1 mediates MYCN control of epithelial-mesenchymal transition through silencing of metastatic suppressor NDRG1 gene. Oncotarget 2017, 8, 3854–3869. [Google Scholar] [CrossRef] [PubMed]
- John, G.R.; Shankar, S.L.; Shafit-Zagardo, B.; Massimi, A.; Lee, S.C.; Raine, C.S.; Brosnan, C.F. Multiple sclerosis: Re-expression of a developmental pathway that restricts oligodendrocyte maturation. Nat. Med. 2002, 8, 1115–1121. [Google Scholar] [CrossRef] [PubMed]
- Zwain, I.H.; Yen, S.S.C. Neurosteroidogenesis in astrocytes, oligodendrocytes, and neurons of cerebral cortex of rat brain. Endocrinology 1999, 140, 3843–3852. [Google Scholar] [CrossRef] [PubMed]
- Allolio, B.; Arlt, W. DHEA treatment: Myth or reality? Trends Endocrinol. Metab. 2002, 13, 288–294. [Google Scholar] [CrossRef]
- Luu-The, V.; Labrie, F. The Intracrine Sex Steroid Biosynthesis Pathways. In Progress in Brain Research; 2010; Volume 181, pp. 177–192. [Google Scholar]
- Bauman, D.R.; Steckelbroeck, S.; Penning, T.M. The roles of aldo-keto reductases in steroid hormone action. Drug News Perspect. 2004, 17, 563–578. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.-Y.; Huang, K.-E.; Chang, S.Y.; Ma, W.-L.; Lin, W.-J.; Chang, C. Differential modulation of androgen receptor-mediated transactivation by Smad3 and tumor suppressor Smad4. J. Biol. Chem. 2002, 277, 43749–43756. [Google Scholar] [CrossRef] [PubMed]
- Chipuk, J.E.; Cornelius, S.C.; Pultz, N.J.; Jorgensen, J.S.; Bonham, M.J.; Kim, S.-J.; Danielpour, D. The androgen receptor represses transforming growth factor- signaling through interaction with Smad3. J. Biol. Chem. 2002, 277, 1240–1248. [Google Scholar] [CrossRef] [PubMed]
- Hayes, S.A.; Zarnegar, M.; Sharma, M.; Yang, F.; Peehl, D.M.; ten Dijke, P.; Sun, Z. SMAD3 represses androgen receptor-mediated transcription. Cancer Res. 2001, 61, 2112–2118. [Google Scholar] [PubMed]
- Wang, H.; Song, K.; Sponseller, T.L.; Danielpour, D. Novel function of androgen receptor-associated protein 55/Hic-5 as a negative regulator of Smad3 signaling. J. Biol. Chem. 2005, 280, 5154–5162. [Google Scholar] [CrossRef] [PubMed]
- Van der Poel, H.G. Androgen receptor and TGFbeta1/Smad signaling are mutually inhibitory in prostate cancer. Eur. Urol. 2005, 48, 1051–1058. [Google Scholar] [CrossRef] [PubMed]
- Song, K.; Wang, H.; Krebs, T.L.; Wang, B.; Kelley, T.J.; Danielpour, D. DHT selectively reverses Smad3-mediated/TGF-β-induced responses through transcriptional down-regulation of Smad3 in prostate epithelial cells. Mol. Endocrinol. 2010, 24, 2019–2029. [Google Scholar] [CrossRef] [PubMed]
- Saddawi-Konefka, R.; Seelige, R.; Gross, E.T.E.; Levy, E.; Searles, S.C.; Washington, A.; Santosa, E.K.; Liu, B.; O’Sullivan, T.E.; Harismendy, O.; et al. Nrf2 Induces IL-17D to mediate tumor and virus surveillance. Cell Rep. 2016, 16, 2348–2358. [Google Scholar] [CrossRef] [PubMed]
- Seelige, R.; Washington, A.; Bui, J.D. The ancient cytokine IL-17D is regulated by Nrf2 and mediates tumor and virus surveillance. Cytokine 2017, 91, 10–12. [Google Scholar] [CrossRef] [PubMed]
- Broxmeyer, H.E.; Starnes, T.; Ramsey, H.; Cooper, S.; Dahl, R.; Williamson, E.; Hromas, R. The IL-17 cytokine family members are inhibitors of human hematopoietic progenitor proliferation. Blood 2006, 108, 770. [Google Scholar] [CrossRef] [PubMed]
- Starnes, T.; Broxmeyer, H.E.; Robertson, M.J.; Hromas, R. Cutting edge: IL-17D, a novel member of the IL-17 family, stimulates cytokine production and inhibits hemopoiesis. J. Immunol. 2002, 169, 642–646. [Google Scholar] [CrossRef] [PubMed]
- Allan, D.; Fairlie-Clarke, K.J.; Elliott, C.; Schuh, C.; Barnett, S.C.; Lassmann, H.; Linnington, C.; Jiang, H.-R. Role of IL-33 and ST2 signalling pathway in multiple sclerosis: Expression by oligodendrocytes and inhibition of myelination in central nervous system. Acta Neuropathol. Commun. 2016, 4, 75. [Google Scholar] [CrossRef] [PubMed]
- Natarajan, C.; Yao, S.-Y.; Sriram, S. TLR3 agonist poly-IC induces IL-33 and promotes myelin repair. PLoS ONE 2016, 11, e0152163. [Google Scholar] [CrossRef] [PubMed]
- Gadani, S.P.; Smirnov, I.; Smith, A.T.; Overall, C.C.; Kipnis, J. Characterization of meningeal type 2 innate lymphocytes and their response to CNS injury. J. Exp. Med. 2016, 214, jem.20161982. [Google Scholar] [CrossRef] [PubMed]
- Molofsky, A.B.; Savage, A.K.; Locksley, R.M. Interleukin-33 in tissue homeostasis, injury, and inflammation. Immunity 2015, 42, 1005–1019. [Google Scholar] [CrossRef] [PubMed]
- Griesenauer, B.; Paczesny, S. The ST2/IL-33 axis in immune cells during inflammatory diseases. Front. Immunol. 2017, 8, 475. [Google Scholar] [CrossRef] [PubMed]
- Gjaltema, R.A.F.; de Rond, S.; Rots, M.G.; Bank, R.A. Procollagen lysyl hydroxylase 2 expression is regulated by an alternative downstream transforming growth factor β-1 activation mechanism. J. Biol. Chem. 2015, 290, 28465–28476. [Google Scholar] [CrossRef] [PubMed]
- Remst, D.F.G.; Blom, A.B.; Vitters, E.L.; Bank, R.A.; van den Berg, W.B.; Blaney Davidson, E.N.; van der Kraan, P.M. Gene expression analysis of murine and human osteoarthritis synovium reveals elevation of transforming growth factor β-responsive genes in osteoarthritis-related fibrosis. Arthritis Rheumatol. 2014, 66, 647–656. [Google Scholar] [CrossRef] [PubMed]
- Remst, D.F.G.; Blaney Davidson, E.N.; Vitters, E.L.; Bank, R.A.; van den Berg, W.B.; van der Kraan, P.M. TGF-β induces Lysyl hydroxylase 2b in human synovial osteoarthritic fibroblasts through ALK5 signaling. Cell Tissue Res. 2014, 355, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Van der Slot, A.J.; Zuurmond, A.-M.; Bardoel, A.F.J.; Wijmenga, C.; Pruijs, H.E.H.; Sillence, D.O.; Brinckmann, J.; Abraham, D.J.; Black, C.M.; Verzijl, N.; et al. Identification of PLOD2 as telopeptide Lysyl hydroxylase, an important enzyme in fibrosis. J. Biol. Chem. 2003, 278, 40967–40972. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Reinhardt, D.P.; Batmunkh, C.; Lindenmaier, W.; Far, R.K.-K.; Notbohm, H.; Hunzelmann, N.; Brinckmann, J. Functional diversity of lysyl hydroxylase 2 in collagen synthesis of human dermal fibroblasts. Exp. Cell Res. 2006, 312, 3485–3494. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Terajima, M.; Yang, Y.; Sun, L.; Ahn, Y.-H.; Pankova, D.; Puperi, D.S.; Watanabe, T.; Kim, M.P.; Blackmon, S.H.; et al. Lysyl hydroxylase 2 induces a collagen cross-link switch in tumor stroma. J. Clin. Investig. 2015, 125, 1147–1162. [Google Scholar] [CrossRef] [PubMed]
- Trinchieri, G. Interleukin-12 and the regulation of innate resistance and adaptive immunity. Nat. Rev. Immunol. 2003, 3, 133–146. [Google Scholar] [CrossRef] [PubMed]
- Campbell, J.S.; Hughes, S.D.; Gilbertson, D.G.; Palmer, T.E.; Holdren, M.S.; Haran, A.C.; Odell, M.M.; Bauer, R. L.; Ren, H.-P.; Haugen, H.S.; et al. Platelet-derived growth factor C induces liver fibrosis, steatosis, and hepatocellular carcinoma. Proc. Natl. Acad. Sci. USA 2005, 102, 3389–3394. [Google Scholar] [CrossRef] [PubMed]
- Martin, I.V.; Borkham-Kamphorst, E.; Zok, S.; van Roeyen, C.R.C.; Eriksson, U.; Boor, P.; Hittatiya, K.; Fischer, H.-P.; Wasmuth, H.E.; Weiskirchen, R.; et al. Platelet-derived growth factor (PDGF)-C neutralization reveals differential roles of PDGF receptors in liver and kidney fibrosis. Am. J. Pathol. 2013, 182, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Zhang, F.; Tang, Z.; Liu, Y.; Li, X. PDGF-C: A new performer in the neurovascular interplay. Trends Mol. Med. 2013, 19, 474–486. [Google Scholar] [CrossRef] [PubMed]
- Sedel, F.; Bernard, D.; Mock, D.M.; Tourbah, A. Targeting demyelination and virtual hypoxia with high-dose biotin as a treatment for progressive multiple sclerosis. Neuropharmacology 2016, 110, 644–653. [Google Scholar] [CrossRef] [PubMed]
- Sedel, F.; Papeix, C.; Bellanger, A.; Touitou, V.; Lebrun-Frenay, C.; Galanaud, D.; Gout, O.; Lyon-Caen, O.; Tourbah, A. High doses of biotin in chronic progressive multiple sclerosis: A pilot study. Mult. Scler. Relat. Disord. 2015, 4, 159–169. [Google Scholar] [CrossRef] [PubMed]
- De Gramont, A.; Faivre, S.; Raymond, E. Novel TGF- β inhibitors ready for prime time in onco-immunology. Oncoimmunology 2017, 6, e1257453. [Google Scholar] [CrossRef] [PubMed]
- Den Hollander, M.W.; Bensch, F.; Glaudemans, A.W.J.M.; Oude Munnink, T.H.; Enting, R.H.; den Dunnen, W.F.A.; Heesters, M.A.A.M.; Kruyt, F.A.E.; Lub-de Hooge, M.N.; Cees de Groot, J.; et al. TGF- antibody uptake in recurrent high-grade glioma imaged with 89Zr-fresolimumab PET. J. Nucl. Med. 2015, 56, 1310–1314. [Google Scholar] [CrossRef] [PubMed]
- Rice, L.M.; Padilla, C.M.; McLaughlin, S.R.; Mathes, A.; Ziemek, J.; Goummih, S.; Nakerakanti, S.; York, M.; Farina, G.; Whitfield, M.L.; et al. Fresolimumab treatment decreases biomarkers and improves clinical symptoms in systemic sclerosis patients. J. Clin. Investig. 2015, 125, 2795–2807. [Google Scholar] [CrossRef] [PubMed]
- Wyss-Coray, T.; Borrow, P.; Brooker, M.J.; Mucke, L. Astroglial overproduction of TGF-beta 1 enhances inflammatory central nervous system disease in transgenic mice. J. Neuroimmunol. 1997, 77, 45–50. [Google Scholar] [CrossRef]
- Luo, J.; Ho, P.P.; Buckwalter, M.S.; Hsu, T.; Lee, L.Y.; Zhang, H.; Kim, D.-K.; Kim, S.-J.; Gambhir, S.S.; Steinman, L.; et al. Glia-dependent TGF-β signaling, acting independently of the TH17 pathway, is critical for initiation of murine autoimmune encephalomyelitis. J. Clin. Investig. 2007, 117, 3306–3315. [Google Scholar] [CrossRef] [PubMed]
- Huber, W.; Carey, V.J.; Gentleman, R.; Anders, S.; Carlson, M.; Carvalho, B.S.; Bravo, H.C.; Davis, S.; Gatto, L.; Girke, T.; et al. Orchestrating high-throughput genomic analysis with Bioconductor. Nat. Methods 2015, 12, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Warde-Farley, D.; Donaldson, S.L.; Comes, O.; Zuberi, K.; Badrawi, R.; Chao, P.; Franz, M.; Grouios, C.; Kazi, F.; Lopes, C.T.; et al. The GeneMANIA prediction server: Biological network integration for gene prioritization and predicting gene function. Nucleic Acids Res. 2010, 38, W214–W220. [Google Scholar] [CrossRef] [PubMed]
- Montojo, J.; Zuberi, K.; Rodriguez, H.; Kazi, F.; Wright, G.; Donaldson, S.L.; Morris, Q.; Bader, G.D. GeneMANIA Cytoscape plugin: Fast gene function predictions on the desktop. Bioinformatics 2010, 26, 2927–2928. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.-M.; Liu, T.; Liu, C.-J.; Song, S.; Zhang, X.; Liu, W.; Jia, H.; Xue, Y.; Guo, A.-Y. AnimalTFDB 2.0: A resource for expression, prediction and functional study of animal transcription factors. Nucleic Acids Res. 2015, 43, D76–D81. [Google Scholar] [CrossRef] [PubMed]
- Kuleshov, M.V.; Jones, M.R.; Rouillard, A.D.; Fernandez, N.F.; Duan, Q.; Wang, Z.; Koplev, S.; Jenkins, S. L.; Jagodnik, K.M.; Lachmann, A.; et al. Enrichr: A comprehensive gene set enrichment analysis web server 2016 update. Nucleic Acids Res. 2016, 44, W90–W97. [Google Scholar] [CrossRef] [PubMed]
- Szklarczyk, D.; Franceschini, A.; Wyder, S.; Forslund, K.; Heller, D.; Huerta-Cepas, J.; Simonovic, M.; Roth, A.; Santos, A.; Tsafou, K.P.; et al. STRING v10: Protein-protein interaction networks, integrated over the tree of life. Nucleic Acids Res. 2015, 43, D447–D452. [Google Scholar] [CrossRef] [PubMed]
- Orii, N.; Ganapathiraju, M.K. Wiki-Pi: A web-server of annotated human protein-protein interactions to aid in discovery of protein function. PLoS ONE 2012, 7, e49029. [Google Scholar] [CrossRef] [PubMed]
- Chatr-aryamontri, A.; Oughtred, R.; Boucher, L.; Rust, J.; Chang, C.; Kolas, N.K.; O’Donnell, L.; Oster, S.; Theesfeld, C.; Sellam, A.; et al. The BioGRID interaction database: 2017 update. Nucleic Acids Res. 2017, 45, D369–D379. [Google Scholar] [CrossRef] [PubMed]
- Keshava Prasad, T.S.; Goel, R.; Kandasamy, K.; Keerthikumar, S.; Kumar, S.; Mathivanan, S.; Telikicherla, D.; Raju, R.; Shafreen, B.; Venugopal, A.; et al. Human protein reference database-2009 supdate. Nucleic Acids Res. 2009, 37, D767–D772. [Google Scholar] [CrossRef] [PubMed]


| Hub Genes | |
| LHPP | Phospholysine phosphohistidine inorganic pyrophosphate phosphatase |
| NDRG1 | N-myc downstream regulated 1 |
| PAQR6 | Progestin and adipoQ receptor family member VI |
| PEA15 | Phosphoprotein enriched in astrocytes 15 |
| RASSF2 | Ras association (RalGDS/AF-6) domain family member 2 |
| Oligodendrocyte-Related Genes | |
| NDRG1 | N-myc downstream regulated 1 |
| MOBP | Myelin-associated oligodendrocyte basic protein |
| MBP | Myelin basic protein |
| PLP1 | Proteolipid protein 1 |
| OLIG1 | Oligodendrocyte transcription factor 1 |
| SIRT2 | Sirtuin (silent mating type information regulation 2 homolog) 2 (Saccharomyces cerevisiae) |
| FA2H | Fatty acid 2-hydroxylase |
| Transcription Factors | |
| AR | Androgen receptor |
| NR3C2 | Nuclear receptor subfamily 3 group C member 2 (mineralocorticoid receptor) |
| SMAD1 | SMAD family member 1 |
| SMAD2 | SMAD family member 2 |
| SOX2 | SRY (sex determining region Y)-box 2 |
| SOX6 | SRY (sex determining region Y)-box 6 |
| SOX9 | SRY (sex determining region Y)-box 9 |
| SOX11 | SRY (sex determining region Y)-box 11 |
| ZNF45 | Zinc finger protein 175 |
| ZNF853 | Zinc finger protein 853 |
| GLIS2 | GLIS family zinc finger 2 |
| GLIS3 | GLIS family zinc finger 3 |
| NFIA | Nuclear factor I/A |
| NFIB | Nuclear factor I/B |
| ID4 | Inhibitor of DNA binding 4, dominant negative helix-loop-helix protein |
| MTA3 | Metastasis associated 1 family, member 3 |
| Cytokines | |
| IL33 | Interleukin 33 |
| IL17D | Interleukin 17D |
| ECM-Related Genes | |
| SPARCL1 | SPARC-like 1 (hevin) |
| SDC2 | Syndecan 2 |
| PLOD2 | Procollagen-lysine, 2-oxoglutarate 5-dioxygenase 2 |
| GPC4 | Glypican 4 |
| SULF1 | Sulfatase 1 |
| SPOCK2 | SPARC/osteonectin, cwcv and kazal-like domains proteoglycan (testican) 2 |
| Others | |
| GFAP | Glial fibrillary acidic protein |
| CD44 | CD44 molecule (Indian blood group) |
| CD200 | CD200 molecule |
| FGF2 | Fibroblast growth factor 2 (basic) |
| Myelin-Related Genes | |
| MBP | Myelin basic protein |
| MOBP | Myelin-associated oligodendrocyte basic protein |
| PLP1 | Proteolipid protein 1 |
| Genes Involved in the “Eukaryotic Translation Elongation” Pathway (Reactome 2016, p = 1.37 × 10−9) | |
| EEF1A1 | Eukaryotic translation elongation factor 1 α 1 |
| EEF1B2 | Eukaryotic translation elongation factor 1 β 2 |
| RPLP1 | Ribosomal protein, large, P1 |
| RPL4 | Ribosomal protein L4 |
| RPL10A | Ribosomal protein L10a |
| RPL22 | Ribosomal protein L22 |
| RPL30 | Ribosomal protein L30 |
| RPL37 | Ribosomal protein L37 |
| RPL37A | Ribosomal protein L37a |
| RPL38 | Ribosomal protein L38 |
| RPS2 | Ribosomal protein S2 |
| RPS6 | Ribosomal protein S6 |
| RPS12 | Ribosomal protein S12 |
| RPS18 | Ribosomal protein S18 |
| RPS27A | Ribosomal protein S27A |
| Gene Symbol | Astrocytes (Mean FPKM) | SD | Other CNS Cells (Mean FPKM) | SD | Fold Change | p-Value |
|---|---|---|---|---|---|---|
| FGF2 | 15.9 | 4.1 | 0.9 | 0.6 | 16.9 | 5.40 × 10−5 |
| GLIS3 | 5.5 | 1.7 | 0.3 | 0.3 | 16.1 | 5.29 × 10−5 |
| SDC2 | 34.0 | 9.7 | 1.8 | 1.6 | 18.5 | 5.50 × 10−5 |
| IL33 | 17.4 | 10.4 | 0.4 | 0.5 | 46.1 | 5.04 × 10−5 |
| GFAP | 119.7 | 113.1 | 8.5 | 11.9 | 14.2 | 5.91 × 10−5 |
| SOX9 | 50.5 | 18.6 | 2.2 | 1.9 | 22.7 | 5.52 × 10−5 |
| SPARCL1 | 1913.8 | 576.8 | 104.0 | 137.6 | 18.4 | 1.47 × 10−5 |
| IL17D | 21.7 | 4.8 | 1.9 | 1.7 | 11.2 | 5.52 × 10−5 |
| ID4 | 22.5 | 7.8 | 0.4 | 0.4 | 51.3 | 5.20 × 10−5 |
| GPC4 | 3.5 | 1.0 | 0.2 | 0.2 | 17.4 | 4.25 × 10−5 |
| Gene Symbol | Astrocytes (Mean FPKM) | SD | Other CNS Cells (Mean FPKM) | SD | Fold Change | p-Value |
|---|---|---|---|---|---|---|
| IL17D | 21.7 | 4.8 | 1.9 | 1.7 | 11.2 | 5.52 × 10−5 |
| IL33 | 17.4 | 10.4 | 0.4 | 0.5 | 46.1 | 5.04 × 10−5 |
| IL1B | 7.0 | 5.9 | 219.4 | 344.3 | 0.0 | 0.002 |
| IL6 | 2.1 | 3.1 | 8.2 | 9.6 | 0.3 | 0.095 |
| IL9 | 0.7 | 0.7 | 0.2 | 0.1 | 4.3 | 0.0049 |
| IL18 | 0.7 | 0.6 | 14.1 | 20.1 | 0.0 | 0.01 |
| IL1A | 0.6 | 0.4 | 28.9 | 49.9 | 0.0 | 0.01 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nataf, S.; Barritault, M.; Pays, L. A Unique TGFB1-Driven Genomic Program Links Astrocytosis, Low-Grade Inflammation and Partial Demyelination in Spinal Cord Periplaques from Progressive Multiple Sclerosis Patients. Int. J. Mol. Sci. 2017, 18, 2097. https://doi.org/10.3390/ijms18102097
Nataf S, Barritault M, Pays L. A Unique TGFB1-Driven Genomic Program Links Astrocytosis, Low-Grade Inflammation and Partial Demyelination in Spinal Cord Periplaques from Progressive Multiple Sclerosis Patients. International Journal of Molecular Sciences. 2017; 18(10):2097. https://doi.org/10.3390/ijms18102097
Chicago/Turabian StyleNataf, Serge, Marc Barritault, and Laurent Pays. 2017. "A Unique TGFB1-Driven Genomic Program Links Astrocytosis, Low-Grade Inflammation and Partial Demyelination in Spinal Cord Periplaques from Progressive Multiple Sclerosis Patients" International Journal of Molecular Sciences 18, no. 10: 2097. https://doi.org/10.3390/ijms18102097
APA StyleNataf, S., Barritault, M., & Pays, L. (2017). A Unique TGFB1-Driven Genomic Program Links Astrocytosis, Low-Grade Inflammation and Partial Demyelination in Spinal Cord Periplaques from Progressive Multiple Sclerosis Patients. International Journal of Molecular Sciences, 18(10), 2097. https://doi.org/10.3390/ijms18102097