Current Concepts of ARDS: A Narrative Review
Abstract
:1. Introduction
2. Definition
3. Diagnostic Evaluation
4. Patophysiology
5. Treatments
5.1. Non-Pharmacologic Interventions
5.1.1. Non-Invasive Ventilation
5.1.2. Invasive Mechanical Ventilation
5.1.3. Lung Recruitment
5.1.4. PEEP Selection
5.1.5. Tidal Volume Setting
5.1.6. Oxygen and Carbon Dioxide Target
5.1.7. Prone Positioning
5.1.8. Extracorporeal Assistance
5.2. Pharmacologic Interventions
5.2.1. Myoresolution
5.2.2. Inhaled Vasodilators
5.2.3. Corticosteroids
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ashbaugh, D.G.; Bigelow, D.B.; Petty, T.L.; Levine, B.E. Acute respiratory distress in adults. Lancet 1967, 2, 319–323. [Google Scholar] [CrossRef]
- Bernard, G.R.; Artigas, A.; Brigham, K.L.; Carlet, J.; Falke, K.; Hudson, L.; Lamy, M.; Legall, J.R.; Morris, A.; Spragg, R. The american-european consensus conference on ards. Definitions, mechanisms, relevant outcomes, and clinical trial coordination. Am. J. Respir. Crit. Care Med. 1994, 149, 818–824. [Google Scholar] [CrossRef] [PubMed]
- Ranieri, V.M.; Rubenfeld, G.D.; Thompson, B.T.; Ferguson, N.D.; Caldwell, E.; Fan, E.; Camporota, L.; Slutsky, A.S. Acute respiratory distress syndrome: The berlin definition. JAMA 2012, 307, 2526–2533. [Google Scholar] [PubMed]
- Montgomery, A.B. Early description of ards. Chest 1991, 99, 261–262. [Google Scholar] [CrossRef] [PubMed]
- Esteban, A.; Ferguson, N.D.; Meade, M.O.; Frutos-Vivar, F.; Apezteguia, C.; Brochard, L.; Raymondos, K.; Nin, N.; Hurtado, J.; Tomicic, V.; et al. Evolution of mechanical ventilation in response to clinical research. Am. J. Respir. Crit. Care Med. 2008, 177, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Rubenfeld, G.D.; Caldwell, E.; Peabody, E.; Weaver, J.; Martin, D.P.; Neff, M.; Stern, E.J.; Hudson, L.D. Incidence and outcomes of acute lung injury. N. Engl. J. Med. 2005, 353, 1685–1693. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Malinchoc, M.; Cartin-Ceba, R.; Venkata, C.V.; Kor, D.J.; Peters, S.G.; Hubmayr, R.D.; Gajic, O. Eight-year trend of acute respiratory distress syndrome: A population-based study in olmsted county, minnesota. Am. J. Respir. Crit. Care Med. 2011, 183, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Hejblum, G.; Chalumeau-Lemoine, L.; Ioos, V.; Boelle, P.Y.; Salomon, L.; Simon, T.; Vibert, J.F.; Guidet, B. Comparison of routine and on-demand prescription of chest radiographs in mechanically ventilated adults: A multicentre, cluster-randomised, two-period crossover study. Lancet 2009, 374, 1687–1693. [Google Scholar] [CrossRef]
- Pilon, C.S.; Leathley, M.; London, R.; McLean, S.; Phang, P.T.; Priestley, R.; Rosenberg, F.M.; Singer, J.; Anis, A.H.; Dodek, P.M. Practice guideline for arterial blood gas measurement in the intensive care unit decreases numbers and increases appropriateness of tests. Crit. Care Med. 1997, 25, 1308–1313. [Google Scholar] [CrossRef] [PubMed]
- Hebert, P.C.; Wells, G.; Blajchman, M.A.; Marshall, J.; Martin, C.; Pagliarello, G.; Tweeddale, M.; Schweitzer, I.; Yetisir, E. A multicenter, randomized, controlled clinical trial of transfusion requirements in critical care. Transfusion requirements in critical care investigators, canadian critical care trials group. N. Engl. J. Med. 1999, 340, 409–417. [Google Scholar] [CrossRef] [PubMed]
- Rosenthal, V.D.; Rodrigues, C.; Alvarez-Moreno, C.; Madani, N.; Mitrev, Z.; Ye, G.; Salomao, R.; Ulger, F.; Guanche-Garcell, H.; Kanj, S.S.; et al. Effectiveness of a multidimensional approach for prevention of ventilator-associated pneumonia in adult intensive care units from 14 developing countries of four continents: Findings of the international nosocomial infection control consortium. Crit. Care Med. 2012, 40, 3121–3128. [Google Scholar] [CrossRef] [PubMed]
- Neto, A.S.; Pereira, V.G.; Esposito, D.C.; Damasceno, M.C.; Schultz, M.J. Neuromuscular blocking agents in patients with acute respiratory distress syndrome: A summary of the current evidence from three randomized controlled trials. Ann. Intensive Care 2012, 2, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Calfee, C.S.; Ware, L.B.; Glidden, D.V.; Eisner, M.D.; Parsons, P.E.; Thompson, B.T.; Matthay, M.A. Use of risk reclassification with multiple biomarkers improves mortality prediction in acute lung injury. Crit. Care Med. 2011, 39, 711–717. [Google Scholar] [CrossRef] [PubMed]
- Ware, L.B.; Koyama, T.; Billheimer, D.D.; Wu, W.; Bernard, G.R.; Thompson, B.T.; Brower, R.G.; Standiford, T.J.; Martin, T.R.; Matthay, M.A. Prognostic and pathogenetic value of combining clinical and biochemical indices in patients with acute lung injury. Chest 2010, 137, 288–296. [Google Scholar] [CrossRef] [PubMed]
- Caironi, P.; Carlesso, E.; Cressoni, M.; Chiumello, D.; Moerer, O.; Chiurazzi, C.; Brioni, M.; Bottino, N.; Lazzerini, M.; Bugedo, G.; et al. Lung recruitability is better estimated according to the berlin definition of acute respiratory distress syndrome at standard 5 cm H2O rather than higher positive end-expiratory pressure: A retrospective cohort study. Crit. Care Med. 2015, 43, 781–790. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, N.D.; Fan, E.; Camporota, L.; Antonelli, M.; Anzueto, A.; Beale, R.; Brochard, L.; Brower, R.; Esteban, A.; Gattinoni, L.; et al. The berlin definition of ards: An expanded rationale, justification, and supplementary material. Intensive Care Med. 2012, 38, 1573–1582. [Google Scholar] [CrossRef] [PubMed]
- Piantadosi, C.A.; Schwartz, D.A. The acute respiratory distress syndrome. Ann. Intern. Med. 2004, 141, 460–470. [Google Scholar] [CrossRef] [PubMed]
- Chastre, J.; Trouillet, J.L.; Vuagnat, A.; Joly-Guillou, M.L.; Clavier, H.; Dombret, M.C.; Gibert, C. Nosocomial pneumonia in patients with acute respiratory distress syndrome. Am. J. Respir. Crit. Care Med. 1998, 157, 1165–1172. [Google Scholar] [CrossRef] [PubMed]
- De Roux, A.; Marcos, M.A.; Garcia, E.; Mensa, J.; Ewig, S.; Lode, H.; Torres, A. Viral community-acquired pneumonia in nonimmunocompromised adults. Chest 2004, 125, 1343–1351. [Google Scholar] [CrossRef] [PubMed]
- Jennings, L.C.; Anderson, T.P.; Beynon, K.A.; Chua, A.; Laing, R.T.; Werno, A.M.; Young, S.A.; Chambers, S.T.; Murdoch, D.R. Incidence and characteristics of viral community-acquired pneumonia in adults. Thorax 2008, 63, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Luyt, C.E.; Combes, A.; Trouillet, J.L.; Nieszkowska, A.; Chastre, J. Virus-induced acute respiratory distress syndrome: Epidemiology, management and outcome. Presse Med. 2011, 40, e561–e568. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.H.; Hong, S.B.; Ko, G.B.; Lee, Y.; Park, H.J.; Park, S.Y.; Moon, S.M.; Cho, O.H.; Park, K.H.; Chong, Y.P.; et al. Viral infection in patients with severe pneumonia requiring intensive care unit admission. Am. J. Respir. Crit. Care Med. 2012, 186, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Bautista, E.; Chotpitayasunondh, T.; Gao, Z.; Harper, S.A.; Shaw, M.; Uyeki, T.M.; Zaki, S.R.; Hayden, F.G.; Hui, D.S.; Kettner, J.D.; et al. Clinical aspects of pandemic 2009 influenza a (H1N1) virus infection. N. Engl. J. Med. 2010, 362, 1708–1719. [Google Scholar] [PubMed]
- Luyt, C.E.; Combes, A.; Deback, C.; Aubriot-Lorton, M.H.; Nieszkowska, A.; Trouillet, J.L.; Capron, F.; Agut, H.; Gibert, C.; Chastre, J. Herpes simplex virus lung infection in patients undergoing prolonged mechanical ventilation. Am. J. Respir. Crit. Care Med. 2007, 175, 935–942. [Google Scholar] [CrossRef] [PubMed]
- Papazian, L.; Doddoli, C.; Chetaille, B.; Gernez, Y.; Thirion, X.; Roch, A.; Donati, Y.; Bonnety, M.; Zandotti, C.; Thomas, P. A contributive result of open-lung biopsy improves survival in acute respiratory distress syndrome patients. Crit. Care Med. 2007, 35, 755–762. [Google Scholar] [CrossRef] [PubMed]
- Azoulay, E.; Lemiale, V.; Mokart, D.; Pene, F.; Kouatchet, A.; Perez, P.; Vincent, F.; Mayaux, J.; Benoit, D.; Bruneel, F.; et al. Acute respiratory distress syndrome in patients with malignancies. Intensive Care Med. 2014, 40, 1106–1114. [Google Scholar] [CrossRef] [PubMed]
- Gadsby, N.J.; Helgason, K.O.; Dickson, E.M.; Mills, J.M.; Lindsay, D.S.; Edwards, G.F.; Hanson, M.F.; Templeton, K.E. Molecular diagnosis of legionella infections—Clinical utility of front-line screening as part of a pneumonia diagnostic algorithm. J. Infect. 2016, 72, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Gibelin, A.; Parrot, A.; Maitre, B.; Brun-Buisson, C.; Mekontso-Dessap, A.; Fartoukh, M.; de Prost, N. Acute respiratory distress syndrome mimickers lacking common risk factors of the berlin definition. Intensive Care Med. 2016, 42, 164–172. [Google Scholar] [CrossRef] [PubMed]
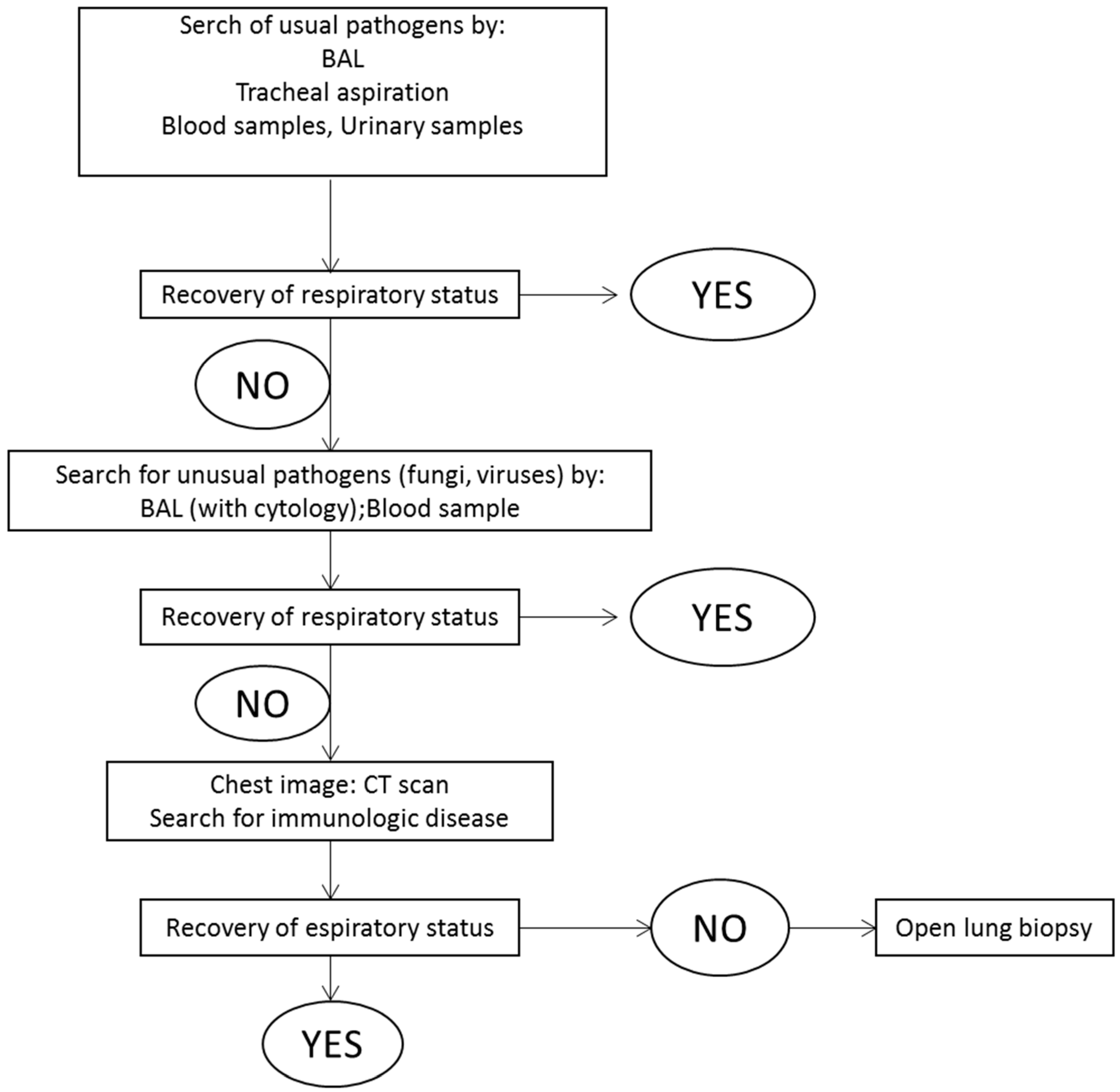
- Papazian, L.; Calfee, C.S.; Chiumello, D.; Luyt, C.E.; Meyer, N.J.; Sekiguchi, H.; Matthay, M.A.; Meduri, G.U. Diagnostic workup for ards patients. Intensive Care Med. 2016, 42, 674–685. [Google Scholar] [CrossRef] [PubMed]
- Palakshappa, J.A.; Meyer, N.J. Which patients with ards benefit from lung biopsy? Chest 2015, 148, 1073–1082. [Google Scholar] [CrossRef] [PubMed]
- Gattinoni, L.; Caironi, P.; Pelosi, P.; Goodman, L.R. What has computed tomography taught us about the acute respiratory distress syndrome? Am. J. Respir. Crit. Care Med. 2001, 164, 1701–1711. [Google Scholar] [CrossRef] [PubMed]
- Pelosi, P.; D’Andrea, L.; Vitale, G.; Pesenti, A.; Gattinoni, L. Vertical gradient of regional lung inflation in adult respiratory distress syndrome. Am. J. Respir. Crit. Care Med. 1994, 149, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Mead, J.; Takishima, T.; Leith, D. Stress distribution in lungs: A model of pulmonary elasticity. J. Appl. Physiol. 1970, 28, 596–608. [Google Scholar] [PubMed]
- Goodman, L.R.; Fumagalli, R.; Tagliabue, P.; Tagliabue, M.; Ferrario, M.; Gattinoni, L.; Pesenti, A. Adult respiratory distress syndrome due to pulmonary and extrapulmonary causes: Ct, clinical, and functional correlations. Radiology 1999, 213, 545–552. [Google Scholar] [CrossRef] [PubMed]
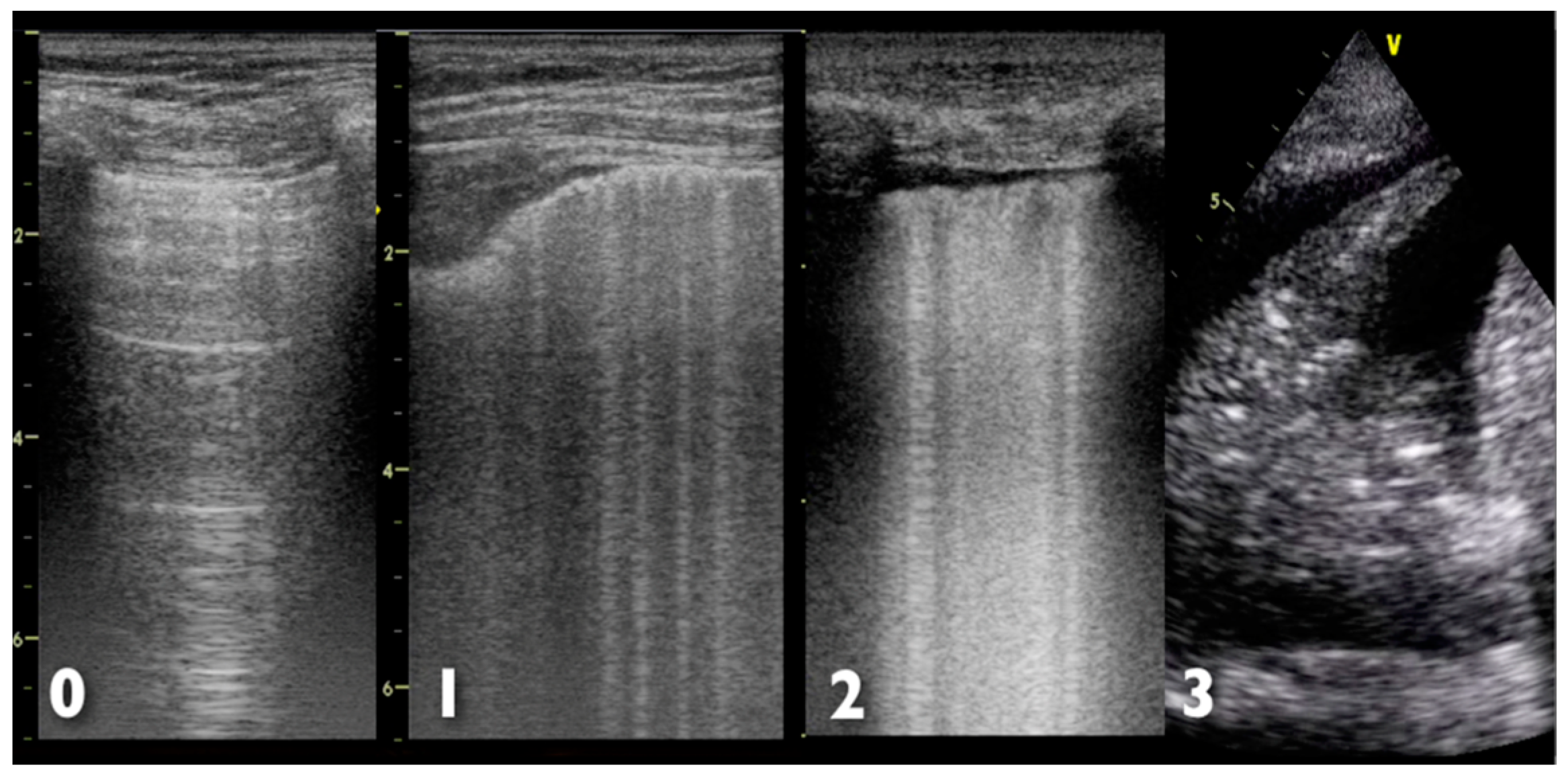
- Lichtenstein, D.A. Ultrasound in the management of thoracic disease. Crit. Care Med. 2007, 35, 250–261. [Google Scholar] [CrossRef] [PubMed]
- Volpicelli, G.; Elbarbary, M.; Blaivas, M.; Lichtenstein, D.A.; Mathis, G.; Kirkpatrick, A.W.; Melniker, L.; Gargani, L.; Noble, V.E.; Via, G.; et al. International evidence-based recommendations for point-of-care lung ultrasound. Intensive Care Med. 2012, 38, 577–591. [Google Scholar] [CrossRef] [PubMed]
- Lichtenstein, D.A.; Meziere, G.A. Relevance of lung ultrasound in the diagnosis of acute respiratory failure: The blue protocol. Chest 2008, 134, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Gargani, L.; Lionetti, V.; di Cristofano, C.; Bevilacqua, G.; Recchia, F.A.; Picano, E. Early detection of acute lung injury uncoupled to hypoxemia in pigs using ultrasound lung comets. Crit. Care Med. 2007, 35, 2769–2774. [Google Scholar] [CrossRef] [PubMed]
- Copetti, R.; Soldati, G.; Copetti, P. Chest sonography: A useful tool to differentiate acute cardiogenic pulmonary edema from acute respiratory distress syndrome. Cardiovasc. Ultrasound 2008, 6, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Bouhemad, B.; Liu, Z.H.; Arbelot, C.; Zhang, M.; Ferarri, F.; Le-Guen, M.; Girard, M.; Lu, Q.; Rouby, J.J. Ultrasound assessment of antibiotic-induced pulmonary reaeration in ventilator-associated pneumonia. Crit. Care Med. 2010, 38, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Peris, A.; Zagli, G.; Barbani, F.; Tutino, L.; Biondi, S.; di Valvasone, S.; Batacchi, S.; Bonizzoli, M.; Spina, R.; Miniati, M.; et al. The value of lung ultrasound monitoring in H1N1 acute respiratory distress syndrome. Anaesthesia 2010, 65, 294–297. [Google Scholar] [CrossRef] [PubMed]
- Martin, T.R. Lung cytokines and ards: Rogers. Mitchell lecture. Chest 1999, 116, 2–8. [Google Scholar] [CrossRef]
- Moine, P.; McIntyre, R.; Schwartz, M.D.; Kaneko, D.; Shenkar, R.; Le Tulzo, Y.; Moore, E.E.; Abraham, E. NF-κB regulatory mechanisms in alveolar macrophages from patients with acute respiratory distress syndrome. Shock 2000, 13, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Sadikot, R.T.; Blackwell, T.S. Bioluminescence imaging. Proc. Am. Thorac. Soc. 2005, 2, 537–540. [Google Scholar] [CrossRef] [PubMed]
- Aliaga, M.; Forel, J.M.; de Bourmont, S.; Jung, B.; Thomas, G.; Mahul, M.; Bisbal, M.; Nougaret, S.; Hraiech, S.; Roch, A.; et al. Diagnostic yield and safety of ct scans in ICU. Intensive Care Med. 2015, 41, 436–443. [Google Scholar] [CrossRef] [PubMed]
- Rocco, P.R.; Dos Santos, C.; Pelosi, P. Lung parenchyma remodeling in acute respiratory distress syndrome. Minerva Anestesiol. 2009, 75, 730–740. [Google Scholar] [PubMed]
- Meduri, G.U.; Annane, D.; Chrousos, G.P.; Marik, P.E.; Sinclair, S.E. Activation and regulation of systemic inflammation in ards: Rationale for prolonged glucocorticoid therapy. Chest 2009, 136, 1631–1643. [Google Scholar] [CrossRef] [PubMed]
- Mascheroni, D.; Kolobow, T.; Fumagalli, R.; Moretti, M.P.; Chen, V.; Buckhold, D. Acute respiratory failure following pharmacologically induced hyperventilation: An experimental animal study. Intensive Care Med. 1988, 15, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Dreyfuss, D.; Saumon, G. Ventilator-induced lung injury: Lessons from experimental studies. Am. J. Respir. Crit. Care Med. 1998, 157, 294–323. [Google Scholar] [CrossRef] [PubMed]
- Amato, M.B.; Meade, M.O.; Slutsky, A.S.; Brochard, L.; Costa, E.L.; Schoenfeld, D.A.; Stewart, T.E.; Briel, M.; Talmor, D.; Mercat, A.; et al. Driving pressure and survival in the acute respiratory distress syndrome. N. Engl. J. Med. 2015, 372, 747–755. [Google Scholar] [CrossRef] [PubMed]
- Hotchkiss, J.R., Jr.; Blanch, L.; Murias, G.; Adams, A.B.; Olson, D.A.; Wangensteen, O.D.; Leo, P.H.; Marini, J.J. Effects of decreased respiratory frequency on ventilator-induced lung injury. Am. J. Respir. Crit. Care Med. 2000, 161, 463–468. [Google Scholar] [CrossRef] [PubMed]
- Cressoni, M.; Gotti, M.; Chiurazzi, C.; Massari, D.; Algieri, I.; Amini, M.; Cammaroto, A.; Brioni, M.; Montaruli, C.; Nikolla, K.; et al. Mechanical power and development of ventilator-induced lung injury. Anesthesiology 2016, 124, 1100–1108. [Google Scholar] [CrossRef] [PubMed]
- Protti, A.; Andreis, D.T.; Milesi, M.; Iapichino, G.E.; Monti, M.; Comini, B.; Pugni, P.; Melis, V.; Santini, A.; Dondossola, D.; et al. Lung anatomy, energy load, and ventilator-induced lung injury. Intensive Care Med. Exp. 2015, 3, 34. [Google Scholar] [CrossRef] [PubMed]
- Gattinoni, L.; Tonetti, T.; Cressoni, M.; Cadringher, P.; Herrmann, P.; Moerer, O.; Protti, A.; Gotti, M.; Chiurazzi, C.; Carlesso, E.; et al. Ventilator-related causes of lung injury: The mechanical power. Intensive Care Med. 2016, 42, 1567–1575. [Google Scholar] [CrossRef] [PubMed]
- Ranieri, V.M.; Suter, P.M.; Tortorella, C.; de Tullio, R.; Dayer, J.M.; Brienza, A.; Bruno, F.; Slutsky, A.S. Effect of mechanical ventilation on inflammatory mediators in patients with acute respiratory distress syndrome: A randomized controlled trial. JAMA 1999, 282, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Gattinoni, L.; Pesenti, A. The concept of “baby lung”. Intensive Care Med. 2005, 31, 776–784. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, R.; Aggarwal, A.N.; Gupta, D. Role of noninvasive ventilation in acute lung injury/acute respiratory distress syndrome: A proportion meta-analysis. Respir. Care 2010, 55, 1653–1660. [Google Scholar] [PubMed]
- Lee, J.H.; Rehder, K.J.; Williford, L.; Cheifetz, I.M.; Turner, D.A. Use of high flow nasal cannula in critically ill infants, children, and adults: A critical review of the literature. Intensive Care Med. 2013, 39, 247–257. [Google Scholar] [CrossRef] [PubMed]
- Messika, J.; Ben Ahmed, K.; Gaudry, S.; Miguel-Montanes, R.; Rafat, C.; Sztrymf, B.; Dreyfuss, D.; Ricard, J.D. Use of high-flow nasal cannula oxygen therapy in subjects with ards: A 1-year observational study. Respir. Care 2015, 60, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Antonelli, M.; Conti, G.; Moro, M.L.; Esquinas, A.; Gonzalez-Diaz, G.; Confalonieri, M.; Pelaia, P.; Principi, T.; Gregoretti, C.; Beltrame, F.; et al. Predictors of failure of noninvasive positive pressure ventilation in patients with acute hypoxemic respiratory failure: A multi-center study. Intensive Care Med. 2001, 27, 1718–1728. [Google Scholar] [CrossRef] [PubMed]
- Frat, J.P.; Thille, A.W.; Mercat, A.; Girault, C.; Ragot, S.; Perbet, S.; Prat, G.; Boulain, T.; Morawiec, E.; Cottereau, A.; et al. High-flow oxygen through nasal cannula in acute hypoxemic respiratory failure. N. Engl. J. Med. 2015, 372, 2185–2196. [Google Scholar] [CrossRef] [PubMed]
- Gattinoni, L. Ultra-protective ventilation and hypoxemia. Crit. Care 2016, 20, 130. [Google Scholar] [CrossRef] [PubMed]
- Pelosi, P.; Goldner, M.; McKibben, A.; Adams, A.; Eccher, G.; Caironi, P.; Losappio, S.; Gattinoni, L.; Marini, J.J. Recruitment and derecruitment during acute respiratory failure: An experimental study. Am. J. Respir. Crit. Care Med. 2001, 164, 122–130. [Google Scholar] [CrossRef] [PubMed]
- Webb, H.H.; Tierney, D.F. Experimental pulmonary edema due to intermittent positive pressure ventilation with high inflation pressures. Protection by positive end-expiratory pressure. Am. Rev. Respir. Dis. 1974, 110, 556–565. [Google Scholar] [PubMed]
- Dreyfuss, D.; Soler, P.; Basset, G.; Saumon, G. High inflation pressure pulmonary edema. Respective effects of high airway pressure, high tidal volume, and positive end-expiratory pressure. Am. Rev. Respir. Dis. 1988, 137, 1159–1164. [Google Scholar] [CrossRef] [PubMed]
- Bowton, D.L.; Kong, D.L. High tidal volume ventilation produces increased lung water in oleic acid-injured rabbit lungs. Crit. Care Med. 1989, 17, 908–911. [Google Scholar] [CrossRef] [PubMed]
- Corbridge, T.C.; Wood, L.D.; Crawford, G.P.; Chudoba, M.J.; Yanos, J.; Sznajder, J.I. Adverse effects of large tidal volume and low peep in canine acid aspiration. Am. Rev. Respir. Dis. 1990, 142, 311–315. [Google Scholar] [CrossRef] [PubMed]
- Slutsky, A.S.; Tremblay, L.N. Multiple system organ failure. Is mechanical ventilation a contributing factor? Am. J. Respir. Crit. Care Med. 1998, 157, 1721–1725. [Google Scholar] [CrossRef] [PubMed]
- Gattinoni, L.; Pelosi, P.; Crotti, S.; Valenza, F. Effects of positive end-expiratory pressure on regional distribution of tidal volume and recruitment in adult respiratory distress syndrome. Am. J. Respir. Crit. Care Med. 1995, 151, 1807–1814. [Google Scholar] [CrossRef] [PubMed]
- Caironi, P.; Cressoni, M.; Chiumello, D.; Ranieri, M.; Quintel, M.; Russo, S.G.; Cornejo, R.; Bugedo, G.; Carlesso, E.; Russo, R.; et al. Lung opening and closing during ventilation of acute respiratory distress syndrome. Am. J. Respir. Crit. Care Med. 2010, 181, 578–586. [Google Scholar] [CrossRef] [PubMed]
- Brower, R.G.; Lanken, P.N.; MacIntyre, N.; Matthay, M.A.; Morris, A.; Ancukiewicz, M.; Schoenfeld, D.; Thompson, B.T. Higher versus lower positive end-expiratory pressures in patients with the acute respiratory distress syndrome. N. Engl. J. Med. 2004, 351, 327–336. [Google Scholar] [PubMed]
- Meade, M.O.; Cook, D.J.; Guyatt, G.H.; Slutsky, A.S.; Arabi, Y.M.; Cooper, D.J.; Davies, A.R.; Hand, L.E.; Zhou, Q.; Thabane, L.; et al. Ventilation strategy using low tidal volumes, recruitment maneuvers, and high positive end-expiratory pressure for acute lung injury and acute respiratory distress syndrome: A randomized controlled trial. JAMA 2008, 299, 637–645. [Google Scholar] [CrossRef] [PubMed]
- Cressoni, M.; Chiumello, D.; Carlesso, E.; Chiurazzi, C.; Amini, M.; Brioni, M.; Cadringher, P.; Quintel, M.; Gattinoni, L. Compressive forces and computed tomography-derived positive end-expiratory pressure in acute respiratory distress syndrome. Anesthesiology 2014, 121, 572–581. [Google Scholar] [CrossRef] [PubMed]
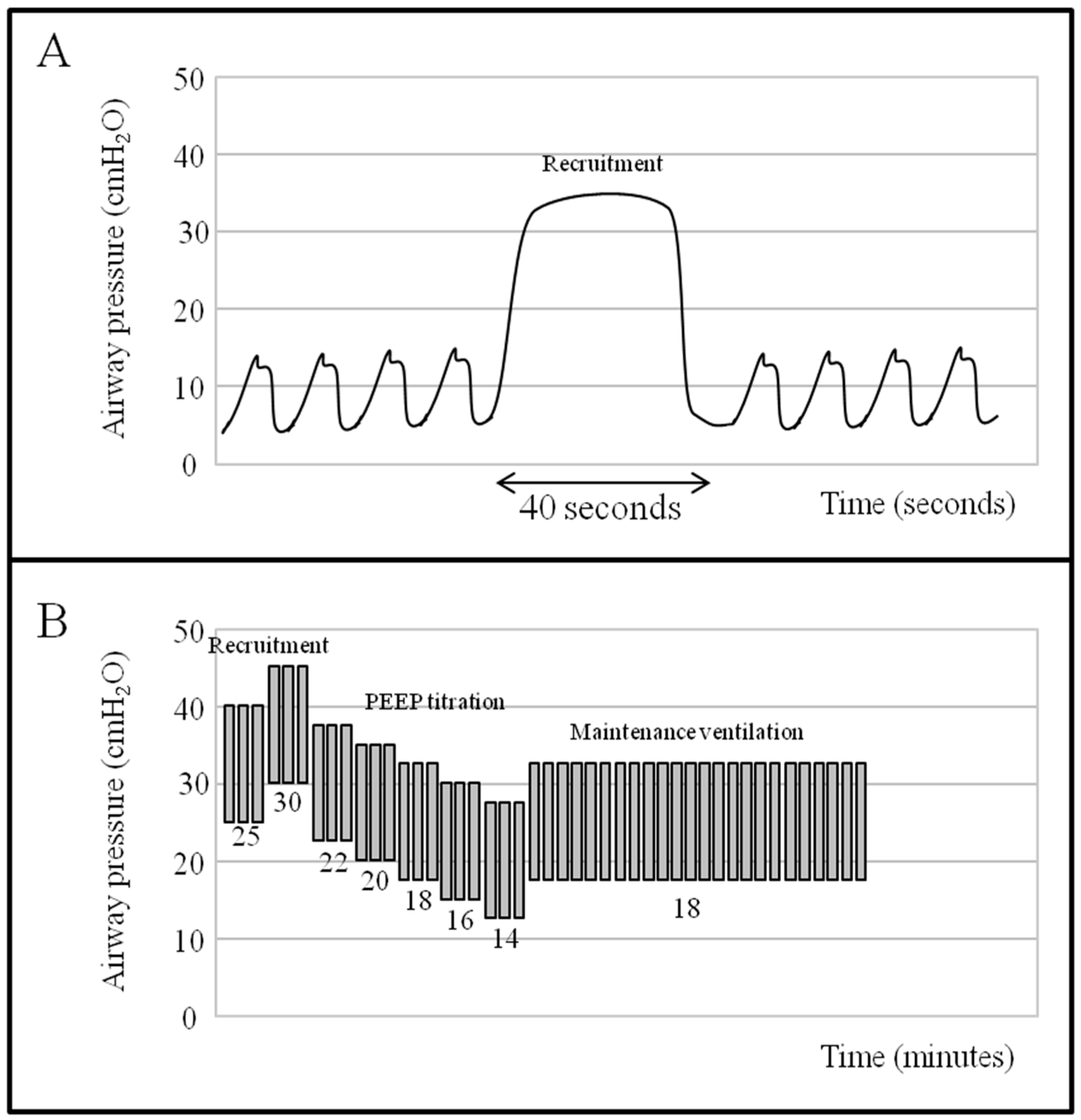
- Chiumello, D.; Algieri, I.; Grasso, S.; Terragni, P.; Pelosi, P. Recruitment maneuvers in acute respiratory distress syndrome and during general anesthesia. Minerva Anestesiol. 2016, 82, 210–220. [Google Scholar] [PubMed]
- Suzumura, E.A.; Figueiro, M.; Normilio-Silva, K.; Laranjeira, L.; Oliveira, C.; Buehler, A.M.; Bugano, D.; Passos-Amato, M.B.; Ribeiro-Carvalho, C.R.; Berwanger, O.; et al. Effects of alveolar recruitment maneuvers on clinical outcomes in patients with acute respiratory distress syndrome: A systematic review and meta-analysis. Intensive Care Med. 2014, 40, 1227–1240. [Google Scholar] [CrossRef] [PubMed]
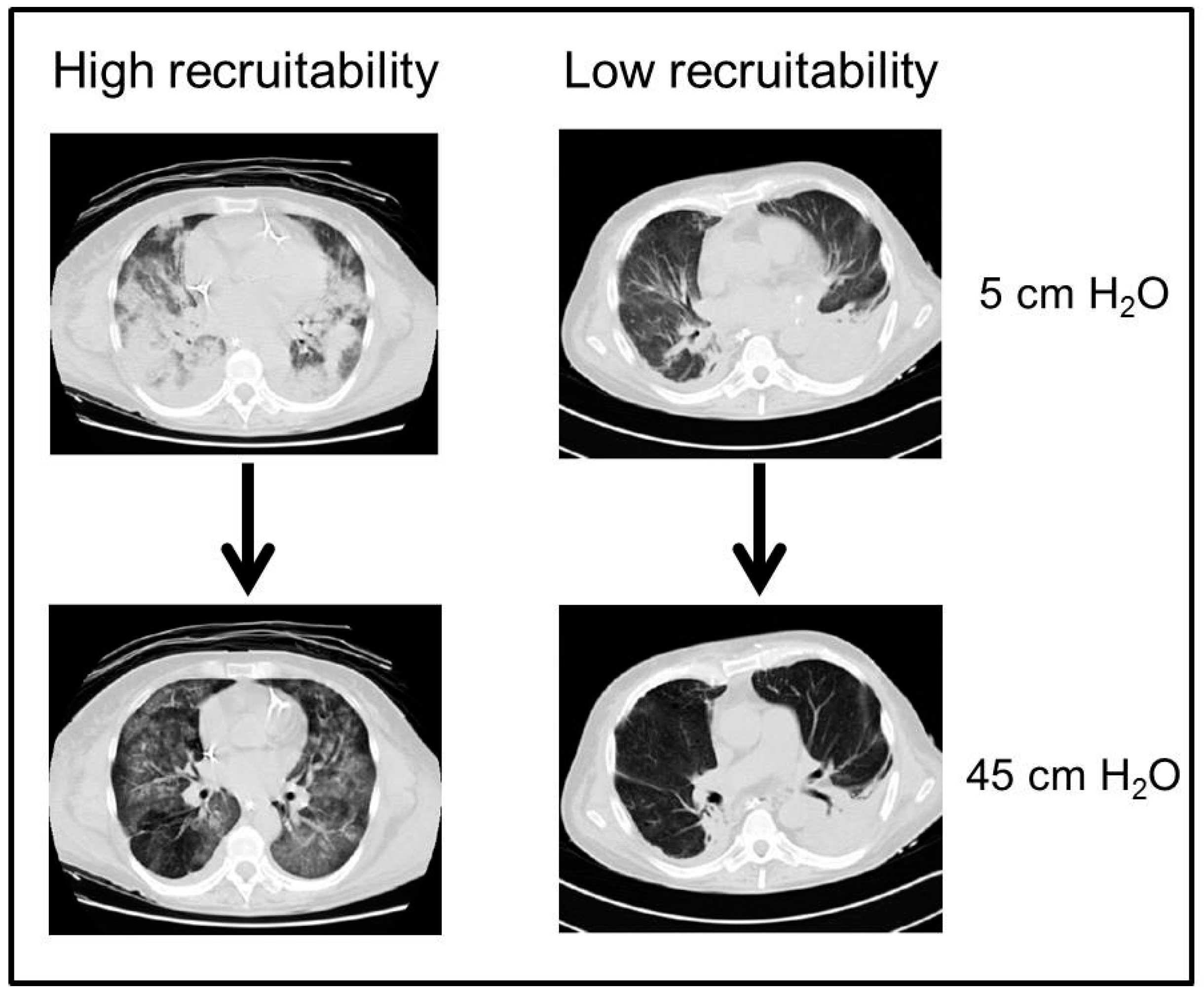
- Chiumello, D.; Marino, A.; Brioni, M.; Cigada, I.; Menga, F.; Colombo, A.; Crimella, F.; Algieri, I.; Cressoni, M.; Carlesso, E.; et al. Lung recruitment assessed by respiratory mechanics and computed tomography in patients with acute respiratory distress syndrome. What is the relationship? Am. J. Respir. Crit. Care Med. 2016, 193, 1254–1263. [Google Scholar] [CrossRef] [PubMed]
- Gattinoni, L.; Caironi, P.; Cressoni, M.; Chiumello, D.; Ranieri, V.M.; Quintel, M.; Russo, S.; Patroniti, N.; Cornejo, R.; Bugedo, G. Lung recruitment in patients with the acute respiratory distress syndrome. N. Engl. J. Med. 2006, 354, 1775–1786. [Google Scholar] [CrossRef] [PubMed]
- Chiumello, D.; Froio, S.; Bouhemad, B.; Camporota, L.; Coppola, S. Clinical review: Lung imaging in acute respiratory distress syndrome patients—An update. Crit. Care 2013, 17, 243. [Google Scholar] [CrossRef] [PubMed]
- Bouhemad, B.; Brisson, H.; Le-Guen, M.; Arbelot, C.; Lu, Q.; Rouby, J.J. Bedside ultrasound assessment of positive end-expiratory pressure-induced lung recruitment. Am. J. Respir. Crit. Care Med. 2011, 183, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Cressoni, M.; Cadringher, P.; Chiurazzi, C.; Amini, M.; Gallazzi, E.; Marino, A.; Brioni, M.; Carlesso, E.; Chiumello, D.; Quintel, M.; et al. Lung inhomogeneity in patients with acute respiratory distress syndrome. Am. J. Respir. Crit. Care Med. 2014, 189, 149–158. [Google Scholar] [CrossRef] [PubMed]
- Broccard, A.F.; Hotchkiss, J.R.; Kuwayama, N.; Olson, D.A.; Jamal, S.; Wangensteen, D.O.; Marini, J.J. Consequences of vascular flow on lung injury induced by mechanical ventilation. Am. J. Respir. Crit. Care Med. 1998, 157, 1935–1942. [Google Scholar] [CrossRef] [PubMed]
- Protti, A.; Cressoni, M.; Santini, A.; Langer, T.; Mietto, C.; Febres, D.; Chierichetti, M.; Coppola, S.; Conte, G.; Gatti, S.; et al. Lung stress and strain during mechanical ventilation: Any safe threshold? Am. J. Respir. Crit. Care Med. 2011, 183, 1354–1362. [Google Scholar] [CrossRef] [PubMed]
- Chiumello, D.; Cressoni, M.; Colombo, A.; Babini, G.; Brioni, M.; Crimella, F.; Lundin, S.; Stenqvist, O.; Gattinoni, L. The assessment of transpulmonary pressure in mechanically ventilated ARDS patients. Intensive Care Med. 2014, 40, 1670–1678. [Google Scholar] [CrossRef] [PubMed]
- Chiumello, D.; Langer, T.; Vecchi, V.; Luoni, S.; Colombo, A.; Brioni, M.; Froio, S.; Cigada, I.; Coppola, S.; Protti, A.; et al. Low-dose chest computed tomography for quantitative and visual anatomical analysis in patients with acute respiratory distress syndrome. Intensive Care Med. 2014, 40, 691–699. [Google Scholar] [CrossRef] [PubMed]
- Mercat, A.; Richard, J.C.; Vielle, B.; Jaber, S.; Osman, D.; Diehl, J.L.; Lefrant, J.Y.; Prat, G.; Richecoeur, J.; Nieszkowska, A.; et al. Positive end-expiratory pressure setting in adults with acute lung injury and acute respiratory distress syndrome: A randomized controlled trial. JAMA 2008, 299, 646–655. [Google Scholar] [CrossRef] [PubMed]
- Grasso, S.; Terragni, P.; Mascia, L.; Fanelli, V.; Quintel, M.; Herrmann, P.; Hedenstierna, G.; Slutsky, A.S.; Ranieri, V.M. Airway pressure-time curve profile (stress index) detects tidal recruitment/hyperinflation in experimental acute lung injury. Crit. Care Med. 2004, 32, 1018–1027. [Google Scholar] [CrossRef] [PubMed]
- Talmor, D.; Sarge, T.; Malhotra, A.; O’Donnell, C.R.; Ritz, R.; Lisbon, A.; Novack, V.; Loring, S.H. Mechanical ventilation guided by esophageal pressure in acute lung injury. N. Engl. J. Med. 2008, 359, 2095–2104. [Google Scholar] [CrossRef] [PubMed]
- Gattinoni, L.; Carlesso, E.; Caironi, P. Stress and strain within the lung. Curr. Opin. Crit. Care 2012, 18, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Wiedemann, H.P.; Arroliga, A.C.; Fisher, C.J.; Komara, J.J.; Perez, P.; Parsons, P.E.; Wolkin, R.; Welsh, C.; Fulkerson, W.J.; MacIntyre, N.; et al. Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. The acute respiratory distress syndrome network. N. Engl. J. Med. 2000, 342, 1301–1308. [Google Scholar]
- Petrucci, N.; de Feo, C. Lung protective ventilation strategy for the acute respiratory distress syndrome. Cochrane Database Syst. Rev. 2013. [Google Scholar] [CrossRef]
- Jaswal, D.S.; Leung, J.M.; Sun, J.; Cui, X.; Li, Y.; Kern, S.; Welsh, J.; Natanson, C.; Eichacker, P.Q. Tidal volume and plateau pressure use for acute lung injury from 2000 to present: A systematic literature review. Crit. Care Med. 2014, 42, 2278–2289. [Google Scholar] [CrossRef] [PubMed]
- Chiumello, D.; Carlesso, E.; Cadringher, P.; Caironi, P.; Valenza, F.; Polli, F.; Tallarini, F.; Cozzi, P.; Cressoni, M.; Colombo, A.; et al. Lung stress and strain during mechanical ventilation for acute respiratory distress syndrome. Am. J. Respir. Crit. Care Med. 2008, 178, 346–355. [Google Scholar] [CrossRef] [PubMed]
- Gattinoni, L.; Carlesso, E.; Cadringher, P.; Valenza, F.; Vagginelli, F.; Chiumello, D. Physical and biological triggers of ventilator-induced lung injury and its prevention. Eur. Respir. J. 2003, 47, 15–25. [Google Scholar] [CrossRef]
- Gattinoni, L.; Pelosi, P.; Suter, P.M.; Pedoto, A.; Vercesi, P.; Lissoni, A. Acute respiratory distress syndrome caused by pulmonary and extrapulmonary disease. Different syndromes? Am. J. Respir. Crit. Care Med. 1998, 158, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Chiumello, D.; Carlesso, E.; Brioni, M.; Cressoni, M. Airway driving pressure and lung stress in ards patients. Crit. Care 2016, 20, 276–286. [Google Scholar] [CrossRef] [PubMed]
- Chacko, B.; Peter, J.V.; Tharyan, P.; John, G.; Jeyaseelan, L. Pressure-controlled versus volume-controlled ventilation for acute respiratory failure due to acute lung injury (ALI) or acute respiratory distress syndrome (ARDS). Cochrane Database Syst. Rev. 2015. [Google Scholar] [CrossRef]
- Rittayamai, N.; Katsios, C.M.; Beloncle, F.; Friedrich, J.O.; Mancebo, J.; Brochard, L. Pressure-controlled vs. volume-controlled ventilation in acute respiratory failure: A physiology-based narrative and systematic review. Chest 2015, 148, 340–355. [Google Scholar] [CrossRef] [PubMed]
- Panwar, R.; Hardie, M.; Bellomo, R.; Barrot, L.; Eastwood, G.M.; Young, P.J.; Capellier, G.; Harrigan, P.W.; Bailey, M. Conservative versus liberal oxygenation targets for mechanically ventilated patients. A pilot multicenter randomized controlled trial. Am. J. Respir. Crit. Care Med. 2016, 193, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Hraiech, S.; Yoshida, T.; Papazian, L. Balancing neuromuscular blockade versus preserved muscle activity. Curr. Opin. Crit. Care 2015, 21, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Hickling, K.G.; Walsh, J.; Henderson, S.; Jackson, R. Low mortality rate in adult respiratory distress syndrome using low-volume, pressure-limited ventilation with permissive hypercapnia: A prospective study. Crit. Care Med. 1994, 22, 1568–1578. [Google Scholar] [CrossRef] [PubMed]
- Caironi, P. Driving pressure and intraoperative protective ventilation. Lancet Respir. Med. 2016, 4, 243–245. [Google Scholar] [CrossRef]
- Langer, M.; Mascheroni, D.; Marcolin, R.; Gattinoni, L. The prone position in ards patients. A clinical study. Chest 1988, 94, 103–107. [Google Scholar] [CrossRef] [PubMed]
- Piehl, M.A.; Brown, R.S. Use of extreme position changes in acute respiratory failure. Crit. Care Med. 1976, 4, 13–14. [Google Scholar] [CrossRef] [PubMed]
- Gattinoni, L.; Taccone, P.; Carlesso, E.; Marini, J.J. Prone position in acute respiratory distress syndrome. Rationale, indications, and limits. Am. J. Respir. Crit. Care Med. 2013, 188, 1286–1293. [Google Scholar] [CrossRef] [PubMed]
- Guerin, C.; Mancebo, J. Prone positioning and neuromuscular blocking agents are part of standard care in severe ards patients: Yes. Intensive Care Med. 2015, 41, 2195–2197. [Google Scholar] [CrossRef] [PubMed]
- Peek, G.J.; Mugford, M.; Tiruvoipati, R.; Wilson, A.; Allen, E.; Thalanany, M.M.; Hibbert, C.L.; Truesdale, A.; Clemens, F.; Cooper, N.; et al. Efficacy and economic assessment of conventional ventilatory support versus extracorporeal membrane oxygenation for severe adult respiratory failure (CESAR): A multicentre randomised controlled trial. Lancet 2009, 374, 1351–1363. [Google Scholar] [CrossRef]
- Ventetuolo, C.E.; Muratore, C.S. Extracorporeal life support in critically ill adults. Am. J. Respir. Crit. Care Med. 2014, 190, 497–508. [Google Scholar] [CrossRef] [PubMed]
- Siobal, M.S. Pulmonary vasodilators. Respir. Care 2007, 52, 885–899. [Google Scholar] [PubMed]
- Adhikari, N.K.; Dellinger, R.P.; Lundin, S.; Payen, D.; Vallet, B.; Gerlach, H.; Park, K.J.; Mehta, S.; Slutsky, A.S.; Friedrich, J.O. Inhaled nitric oxide does not reduce mortality in patients with acute respiratory distress syndrome regardless of severity: Systematic review and meta-analysis. Crit. Care Med. 2014, 42, 404–412. [Google Scholar] [CrossRef] [PubMed]
- Meduri, G.U.; Headley, A.S.; Golden, E.; Carson, S.J.; Umberger, R.A.; Kelso, T.; Tolley, E.A. Effect of prolonged methylprednisolone therapy in unresolving acute respiratory distress syndrome: A randomized controlled trial. JAMA 1998, 280, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Steinberg, K.P.; Hudson, L.D.; Goodman, R.B.; Hough, C.L.; Lanken, P.N.; Hyzy, R.; Thompson, B.T.; Ancukiewicz, M. Efficacy and safety of corticosteroids for persistent acute respiratory distress syndrome. N. Engl. J. Med. 2006, 354, 1671–1684. [Google Scholar] [PubMed]
- Bernard, G.R.; Luce, J.M.; Sprung, C.L.; Rinaldo, J.E.; Tate, R.M.; Sibbald, W.J.; Kariman, K.; Higgins, S.; Bradley, R.; Metz, C.A.; et al. High-dose corticosteroids in patients with the adult respiratory distress syndrome. N. Engl. J. Med. 1987, 317, 1565–1570. [Google Scholar] [CrossRef] [PubMed]
- Ha, M.; Kim, V.N. Regulation of microrna biogenesis. Nat. Rev. Mol. Cell Biol. 2014, 15, 509–524. [Google Scholar] [CrossRef] [PubMed]
- Foster, P.S.; Plank, M.; Collison, A.; Tay, H.L.; Kaiko, G.E.; Li, J.; Johnston, S.L.; Hansbro, P.M.; Kumar, R.K.; Yang, M.; et al. The emerging role of micrornas in regulating immune and inflammatory responses in the lung. Immunol. Rev. 2013, 253, 198–215. [Google Scholar] [CrossRef] [PubMed]
- Narute, P.; Seam, N.; Tropea, M.; Logun, C.; Cai, R.; Sun, J.; Shelhamer, J.H.; Meduri, G.U.; Suffredini, A.F. Temporal changes in microrna expression in blood leukocytes from patients with the acute respiratory distress syndrome. Shock 2016. [Google Scholar] [CrossRef] [PubMed]

| Characteristic | AECC Definition 1994 [2] | Berlin Definition 2012 [3] |
|---|---|---|
| Timing | Acute, without any specification | Maximum within a week after a trigger insult |
| Imaging | Chest X-ray with bilateral infiltrates | Chest X-ray or CT scan with bilateral infiltrates, not fully explained by effusion, lung collapse or nodules |
| Non-cardiogenic source of edema | Confirmation of non-elevated left atrial pressure | Respiratory failure not completely explained by excessive volume loading or cardiac failure |
| Classification | Based on PaO2/FiO2 | Based on PaO2/FiO2 calculated with PEEP ≥5 cmH2O |
| Acute lung injury: ≤300 | Mild: 201–300 | |
| ARDS: ≤200 | Moderate: 101–200 | |
| – | Severe: ≤100 | |
| Predisposing condition | Not specified | If none identified, then need to rule out cardiogenic edema with additional data |
| Bacteria | Virus | Fungi | Parasites |
|---|---|---|---|
| Streptococcus pneumoniae | Influenza A and B | Pneumocystis Jirovecii | Toxoplasma gondii |
| Haemophilus influenzae | Rhinoviruses | ||
| Enterobacteriaceae | RSV | ||
| Staphylococcus aureus | Parainfluenza viruses | ||
| Legionella pneumophila | Coronavirus | ||
| Clamydia pneumoniae | Enterovirus | Aspergillus fumigatus | |
| Mycoplasma pneumoniae | HSV | ||
| Pseudomonas aeruginosa | CMV | ||
| Acinetobacter baumannii | – | ||
| Stenotrophompnas maltophilia | – |
| Condition | Thoracic Ultrasound | Cardiac Ultrasound |
|---|---|---|
| ARDS | Bilateral B pattern | No change in ventricular function vs. previous examination |
| Non-uniform distribution | ||
| Pleural line abnormalities | No inferior vena cava dilation (diameter < 23 mm) | |
| Reduced in lung sliding | E/e’ ≤ 8 | |
| C pattern | – | |
| Cardiogenic Pulmonary Edema | Bilateral B pattern | New or worsening left ventricular disfunction |
| Uniform distribution | Inferior vena cava dilation (≥23 mm) | |
| Pleural effusion | E/e’ ≥ 14 | |
| Left-sided predominance | – |
| Non-Pharmacologic | Pharmacologic |
|---|---|
| Non-invasive ventilation | Myoresolution |
| Invasive mechanical ventilation | |
| Lung recruitment | Inhaled vasodilators |
| PEEP selection | |
| Tidal volume setting | Corticosteroids |
| Oxygen and Carbon Dioxide target | |
| Prone positioning | – |
| Extracorporeal assistance |
| Method | Characteristics |
|---|---|
| Lung Open Ventilation (LOV) study [72] | Setting PEEP as for the PEEP/FiO2 table of the lung open ventilation arm of LOV trial |
| ExPress [85] | Maintain an inspiratory plateau pressure between 28 and 30 cm H2O according to the increased recruitment strategy of the ExPress trial |
| Stress Index [86] | Obtain a stress index coefficient of 1 |
| Esophageal pressure [87] | Setting PEEP targeting an absolute end-expiratory transpulmonary pressure of 0–10 cm H2O |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Umbrello, M.; Formenti, P.; Bolgiaghi, L.; Chiumello, D. Current Concepts of ARDS: A Narrative Review. Int. J. Mol. Sci. 2017, 18, 64. https://doi.org/10.3390/ijms18010064
Umbrello M, Formenti P, Bolgiaghi L, Chiumello D. Current Concepts of ARDS: A Narrative Review. International Journal of Molecular Sciences. 2017; 18(1):64. https://doi.org/10.3390/ijms18010064
Chicago/Turabian StyleUmbrello, Michele, Paolo Formenti, Luca Bolgiaghi, and Davide Chiumello. 2017. "Current Concepts of ARDS: A Narrative Review" International Journal of Molecular Sciences 18, no. 1: 64. https://doi.org/10.3390/ijms18010064
APA StyleUmbrello, M., Formenti, P., Bolgiaghi, L., & Chiumello, D. (2017). Current Concepts of ARDS: A Narrative Review. International Journal of Molecular Sciences, 18(1), 64. https://doi.org/10.3390/ijms18010064









