Advances of Proteomic Sciences in Dentistry
Abstract
:1. Introduction
2. Dental Hard Tissue Proteomics
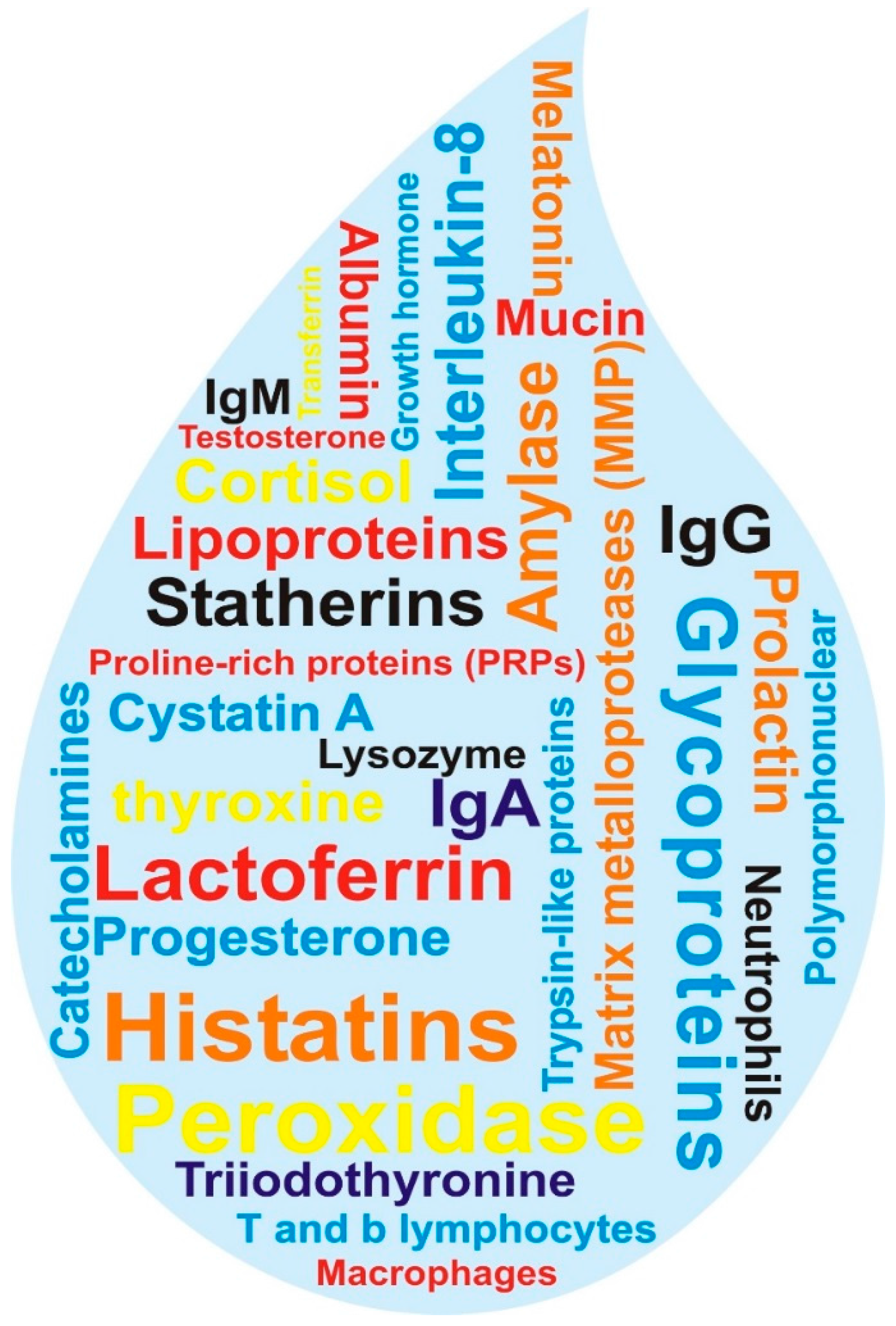
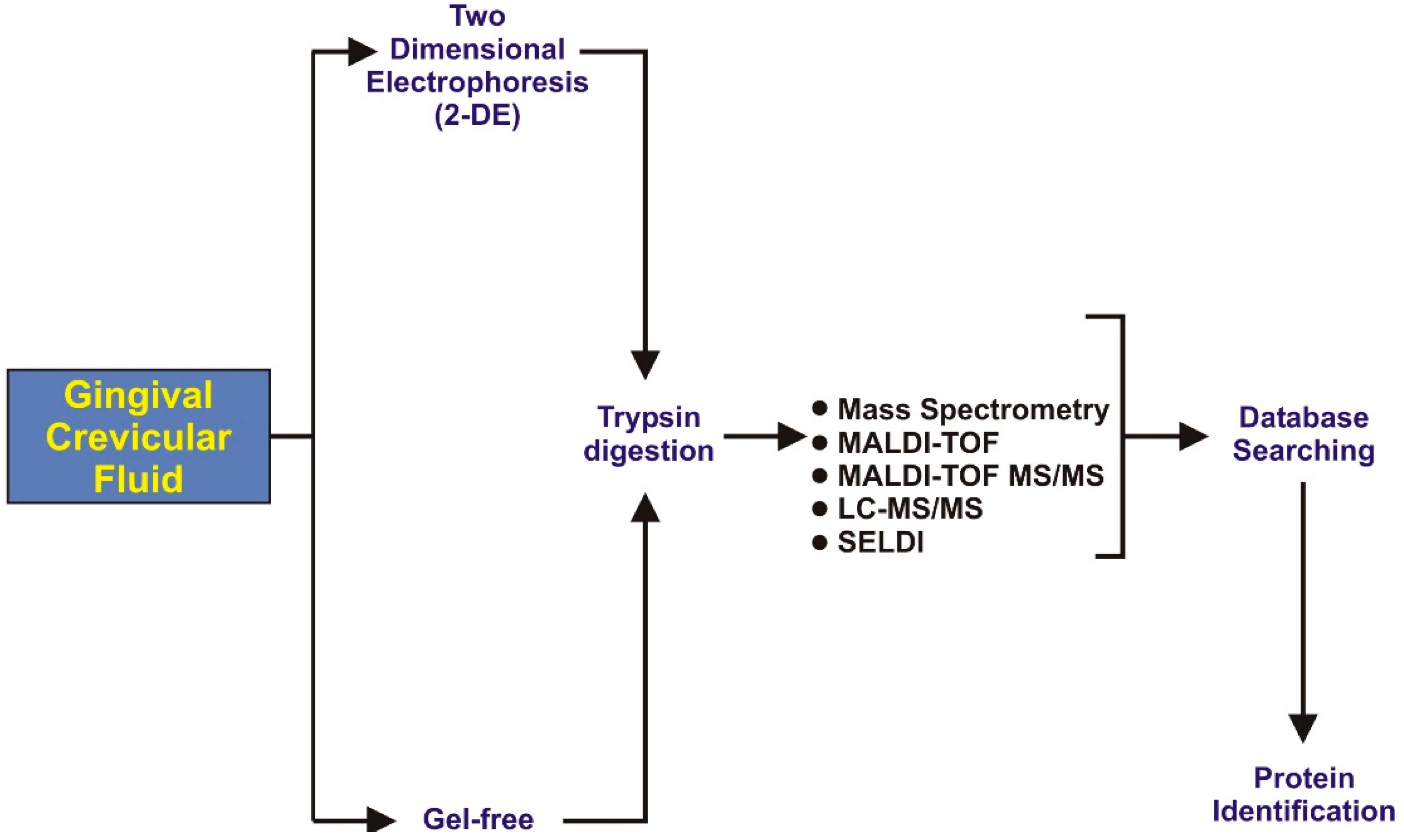
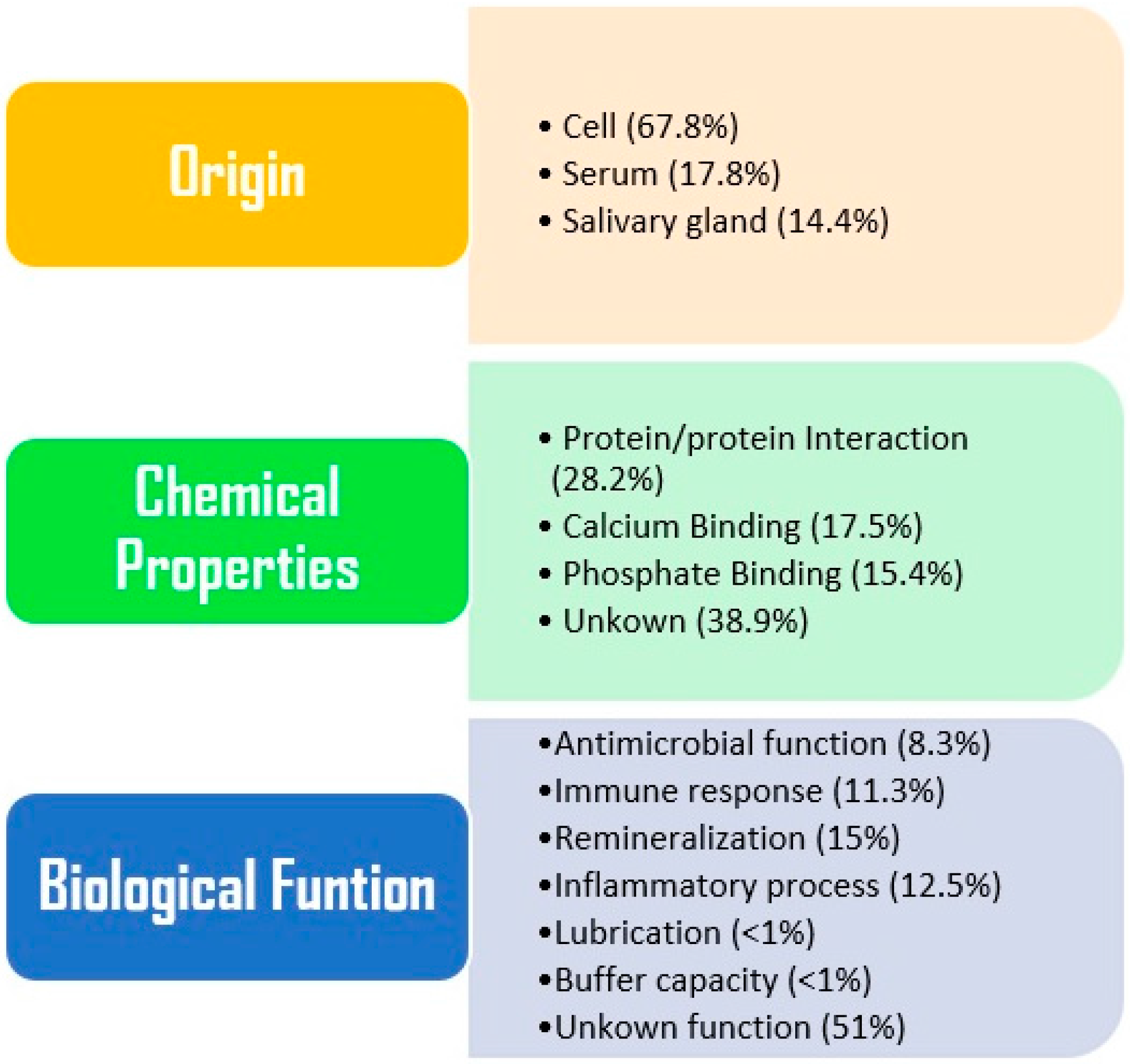
3. Oral Fluid Proteomics
4. Dental Soft Tissue Proteomics
5. Dental Materials Proteomics
6. Conclusions
Author Contributions
Conflicts of Interest
Abbreviations
| BGLAP | Bone Gamma Carboxyglutamate Protein |
| cDNA | Complementary Deoxyribonucleic Acid |
| CHAD | Chondroadherin |
| C-HPP | Chromosome-Centric Human Proteome Project |
| CsA | Cyclosporin A |
| DNAse | Deoxyribonuclease |
| EMD | Enamel Matrix Derivative |
| ESI | Electrospray Ionization |
| FN | Fibronectin |
| FNA | Fine Needle Aspiration |
| HGF | Hepatocyte Growth Factor |
| GCF | Gingival Crevicular Fluid |
| GVHD | Graft Versus Host Disease |
| HA | Hydroxyapatite |
| HPP | Human Proteome Project |
| LC/MS | Liquid chromatography/mass spectrometry |
| LTQ-FT | Linear Ion Trap Mass Spectrometer |
| LUM | Lumican |
| mRNA | messenger Ribonucleic Acid |
| MS | Mass-Spectrometry |
| MALDI | Matrix-Assisted Laser Desorption/Ionization |
| PDL | Periodontal Ligament |
| PEEK | Polyetheretherketone |
| R-PCR | Realtime-Polymerase Chain Reaction |
| SDS-PAGE | Sodium dodecyl sulfate polyacrylamide gel electrophoresis |
| TAILS | Terminal Amine Isotropic Labelling of Substrates |
| TNN | Tenascin |
| USWS | Unstimulated Whole-Mouth Saliva |
| VIM | Vimentin |
| VTN | Vitronectin |
| WS | Whole-Mouth saliva |
| WB | Western Blotting |
| 2-DE | 2-Dimensional Electrophoresis |
References
- Bhattacharyya, M. Protein structure and function: Looking through the network of side-chain interactions. Curr. Protein Pept. Sci. 2015, 17, 4–25. [Google Scholar] [CrossRef]
- Alberts, B. The cell as a collection of protein machines: Preparing the next generation of molecular biologists. Cell 1998, 92, 291–294. [Google Scholar] [CrossRef]
- Murzin, A.G.; Brenner, S.E.; Hubbard, T.; Chothia, C. SCOP: A structural classification of proteins database for the investigation of sequences and structures. J. Mol. Biol. 1995, 247, 536–540. [Google Scholar] [CrossRef]
- Luscombe, N.M. Amino acid-base interactions: A three-dimensional analysis of protein-DNA interactions at an atomic level. Nucleic Acids Res. 2001, 29, 2860–2874. [Google Scholar] [CrossRef] [PubMed]
- Nussinov, R.; Wolfson, H.J. Efficient detection of three-dimensional structural motifs in biological macromolecules by computer vision techniques. Proc. Natl. Acad. Sci. USA 1991, 88, 10495–10499. [Google Scholar] [CrossRef] [PubMed]
- Wilkins, M. Proteomics data mining. Exp. Rev. Proteom. 2009, 6, 599–603. [Google Scholar] [CrossRef] [PubMed]
- Altelaar, A.F.M.; Munoz, J.; Heck, A.J.R. Next-generation proteomics: Towards an integrative view of proteome dynamics. Nat. Rev. Genet. 2013, 14, 35–48. [Google Scholar] [CrossRef] [PubMed]
- Latterich, M.; Abramovitz, M.; Leyland-Jones, B. Proteomics: New technologies and clinical applications. Eur. J. Cancer 2008, 44, 2737–2741. [Google Scholar] [CrossRef] [PubMed]
- De Vries, S.; Bonvin, A. How proteins get in touch: Interface prediction in the study of biomolecular complexes. Curr. Protein Pept. Sci. 2008, 9, 394–406. [Google Scholar] [CrossRef] [PubMed]
- Chiappelli, F.; Covani, U.; Giacomelli, L. Proteomics as it pertains to oral pathologies and dental research. Bioinformation 2010, 5, 277. [Google Scholar] [CrossRef]
- Hubbard, M.J.; Faught, M.J.; Carlisle, B.H.; Stockwell, P.A. ToothPrint, a proteomic database for dental tissues. Proteomics 2001, 1, 132–135. [Google Scholar] [CrossRef]
- Jágr, M.; Eckhardt, A.; Pataridis, S.; Broukal, Z.; Dušková, J.; Mikšík, I. Proteomics of human teeth and saliva. Physiol. Res. 2014, 63, S141–S154. [Google Scholar] [PubMed]
- Rezende, T.M.B.; Lima, S.M.F.; Petriz, B.A.; Silva, O.N.; Freire, M.S.; Franco, O.L. Dentistry proteomics: From laboratory development to clinical practice. J. Cell. Physiol. 2013, 228, 2271–2284. [Google Scholar] [CrossRef] [PubMed]
- Amado, F.M.L.; Ferreira, R.P.; Vitorino, R. One decade of salivary proteomics: Current approaches and outstanding challenges. Clin. Biochem. 2013, 46, 506–517. [Google Scholar] [CrossRef] [PubMed]
- Cabras, T.; Iavarone, F.; Manconi, B.; Olianas, A.; Sanna, M.T.; Castagnola, M.; Messana, I. Top-down analytical platforms for the characterization of the human salivary proteome. Bioanalysis 2014, 6, 563–581. [Google Scholar] [CrossRef] [PubMed]
- Seema, S.; Krishnan, M.; Harith, A.K.; Sahai, K.; Iyer, S.R.; Arora, V.; Tripathi, R.P. Laser ionization mass spectrometry in oral squamous cell carcinoma. J. Oral Pathol. Med. 2014, 43, 471–483. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Arellano, M.; Boontheung, P.; Wang, J.; Zhou, H.; Jiang, J.; Elashoff, D.; Wei, R.; Loo, J.A.; Wong, D.T. Salivary proteomics for oral cancer biomarker discovery. Clin. Cancer Res. 2008, 14, 6246–6252. [Google Scholar] [CrossRef] [PubMed]
- Bencharit, S.; Altarawneh, S.K.; Baxter, S.S.; Carlson, J.; Ross, G.F.; Border, M.B.; Mack, C.R.; Byrd, W.C.; Dibble, C.F.; Barros, S.; et al. Elucidating role of salivary proteins in denture stomatitis using a proteomic approach. Mol. BioSyst. 2012, 8, 3216–3223. [Google Scholar] [CrossRef] [PubMed]
- Baldini, C.; Giusti, L.; Ciregia, F.; da Valle, Y.; Giacomelli, C.; Donadio, E.; Sernissi, F.; Bazzichi, L.; Giannaccini, G.; Bombardieri, S.; et al. Proteomic analysis of saliva: A unique tool to distinguish primary Sjögren’s syndrome from secondary Sjögren’s syndrome and other sicca syndromes. Arthritis Res. Ther. 2011, 13, R194. [Google Scholar] [CrossRef] [PubMed]
- Baldini, C.; Laura, G.; Laura, B.; Antonio, L.; Stefano, B. Proteomic analysis of the saliva: A clue for understanding primary from secondary Sjögren’s syndrome? Autoimmun. Rev. 2008, 7, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Devic, I.; Shi, M.; Schubert, M.M.; Lloid, M.; Izutsu, K.T.; Pan, C.; Missaghi, M.; Morton, T.H.; Mancl, L.A.; Zhang, J.; et al. Proteomic analysis of saliva from patients with oral chronic graft-versus-host disease. Biol. Blood Marrow Transplant. 2014, 20, 1048–1055. [Google Scholar] [CrossRef] [PubMed]
- Fonteles, C.S.R.; dos Santos, C.F.; da Silva Alves, K.S.; de Miranda Mota, A.C.; Damasceno, J.X.; Fonteles, M.C. Comparative proteomic analysis of human whole saliva of children with protein-energy undernutrition. Nutrition 2012, 28, 744–748. [Google Scholar] [CrossRef] [PubMed]
- Dowling, P.; Robert, W.; Paula, M.; Michael, H.; Aongus, C.; Martin, C. Analysis of the saliva proteome from patients with head and neck squamous cell carcinoma reveals differences in abundance levels of proteins associated with tumour progression and metastasis. J. Proteom. 2008, 71, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Rao, P.V.; Reddy, A.P.; Lu, X.; Dasari, S.; Krishnaprasad, A.; Biggs, E.; Roberts, C.T.; Nagalla, S.R. Proteomic identification of salivary biomarkers of type-2 diabetes. J. Proteome Res. 2009, 8, 239–245. [Google Scholar] [CrossRef] [PubMed]
- Border, M.B.; Schwartz, S.; Carlson, J.; Dibble, C.F.; Kohltfarber, H.; Offenbacher, S.; Buse, J.B.; Bencharit, S. Exploring salivary proteomes in edentulous patients with type 2 diabetes. Mol. BioSyst. 2012, 8, 1304–1310. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Gao, P.; Wang, X.; Duan, Y. Investigation and identification of potential biomarkers in human saliva for the early diagnosis of oral squamous cell carcinoma. Clin. Chim. Acta 2014, 427, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Yan, G.; Huang, W.; Xue, H.; Jia, Y.; Yang, D. [Relationship between dental caries and salivary proteome by electrospray ionization ion-trap tandem mass spectrometry in children aged 6 to 8 years]. Hua Xi Kou Qiang Yi Xue Za Zhi 2014, 32, 297–302. [Google Scholar] [PubMed]
- Ellias, M.F.; Zainal Ariffin, S.H.; Karsani, S.A.; Abdul Rahman, M.; Senafi, S.; Megat Abdul Wahab, R. Proteomic analysis of saliva identifies potential biomarkers for orthodontic tooth movement. Sci. World J. 2012, 2012, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Shu, R.; Luo, L.-J.; Ge, L.-H.; Xie, Y.-F. Initial comparison of proteomic profiles of whole unstimulated saliva obtained from generalized aggressive periodontitis patients and healthy control subjects. J. Periodontal Res. 2009, 44, 636–644. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, L.D.R.; Soares, M.R.; Nogueira, F.C.S.; Garcia, C.; Camisasca, D.R.; Domont, G.; Feitosa, A.C.R.; Pereira, D.D.A.; Zingali, R.B.; Alves, G. Comparative proteomic analysis of whole saliva from chronic periodontitis patients. J. Proteom. 2010, 73, 1334–1341. [Google Scholar] [CrossRef] [PubMed]
- Rangé, H.; Léger, T.; Huchon, C.; Ciangura, C.; Diallo, D.; Poitou, C.; Meilhac, O.; Bouchard, P.; Chaussain, C. Salivary proteome modifications associated with periodontitis in obese patients. J. Clin. Periodontol. 2012, 39, 799–806. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, L.D.R.; Soares, M.R.; Nogueira, F.C.S.; Garcia, C.H.S.; Camisasca, D.R.; Domont, G.; Feitosa, A.C.R.; Pereira, D.A.; Zingali, R.B.; Alves, G. Analysis of the salivary proteome in gingivitis patients. J. Periodontal Res. 2011, 46, 599–606. [Google Scholar] [CrossRef] [PubMed]
- Salmon, C.R.; Tomazela, D.M.; Ruiz, K.G.S.; Foster, B.L.; Paes Leme, A.F.; Sallum, E.A.; Somerman, M.J.; Nociti, F.H. Proteomic analysis of human dental cementum and alveolar bone. J. Proteom. 2013, 91, 544–555. [Google Scholar] [CrossRef] [PubMed]
- Donadio, E.; Giusti, L.; Seccia, V.; Ciregia, F.; da Valle, Y.; Dallan, I.; Ventroni, T.; Giannaccini, G.; Sellari-Franceschini, S.; Lucacchini, A. New insight into benign tumours of major salivary glands by proteomic approach. PLoS ONE 2013, 8, e71874. [Google Scholar] [CrossRef] [PubMed]
- Huynh, A H.S.; Veith, P.D.; McGregor, N.R.; Adams, G.G.; Chen, D.; Reynolds, E.C.; Ngo, L.H.; Darby, I.B. Gingival crevicular fluid proteomes in health, gingivitis and chronic periodontitis. J. Periodontal Res. 2015, 50, 637–649. [Google Scholar]
- Park, E.-S.; Cho, H.-S.; Kwon, T.-G.; Jang, S.-N.; Lee, S.-H.; An, C.-H.; Shin, H.-I.; Kim, J.-Y.; Cho, J.-Y. Proteomics analysis of human dentin reveals distinct protein expression profiles. J. Proteome Res. 2009, 8, 1338–1346. [Google Scholar] [CrossRef] [PubMed]
- Eckhardt, A.; Jágr, M.; Pataridis, S.; Mikšík, I. Proteomic analysis of human tooth pulp: Proteomics of human tooth. J. Endod. 2014, 40, 1961–1966. [Google Scholar] [CrossRef] [PubMed]
- Reichenberg, E.; Redlich, M.; Cancemi, P.; Zaks, B.; Pitaru, S.; Fontana, S.; Pucci-Minafra, I.; Palmon, A. Proteomic analysis of protein components in periodontal ligament fibroblasts. J. Periodontol. 2005, 76, 1645–1653. [Google Scholar] [CrossRef] [PubMed]
- Siqueira, W.; Zhang, W.; Helmerhorst, E.J.; Gygi, S.; Oppenheim, F.G. Identification of protein components in in vivo human acquired enamel pellicle using LC-ESI-MS/MS. J. Proteome Res. 2007, 6, 2152–2160. [Google Scholar] [CrossRef] [PubMed]
- Khurshid, Z.; Naseem, M.; Sheikh, Z.; Najeeb, S.; Shahab, S.; Zafar, M.S. Oral antimicrobial peptides: Types and role in the oral cavity. Saudi Pharm. J. 2015. [Google Scholar] [CrossRef]
- He, L.H.; Swain, M.V. Understanding the mechanical behaviour of human enamel from its structural and compositional characteristics. J. Mech. Behav. Biomed. Mater. 2008, 1, 18–29. [Google Scholar] [CrossRef] [PubMed]
- Hubbard, M.J.; Kon, J.C. Proteomic analysis of dental tissues. J. Chromatogr. B 2002, 771, 211–220. [Google Scholar] [CrossRef]
- Hubbard, M.J.; McHugh, N.J.; Mangum, J.E. Exclusion of all three calbindins from a calcium-ferry role in rat enamel cells. Eur. J. Oral Sci. 2011, 119, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Hubbard, M.J.; McHugh, N.J.; Carne, D.L. Isolation of ERp29, a novel endoplasmic reticulum protein, from rat enamel cells. Eur. J. Biochem. 2000, 267, 1945–1957. [Google Scholar] [CrossRef] [PubMed]
- Hubbard, M.J. Abundant calcium homeostasis machinery in rat dental enamel cells. Up-regulation of calcium store proteins during enamel mineralization implicates the endoplasmic reticulum in calcium transcytosis. Eur. J. Biochem. 1996, 239, 611–623. [Google Scholar] [CrossRef] [PubMed]
- Hubbard, M.J. Calbindin28kDa and calmodulin are hyperabundant in rat dental enamel cells. Identification of the protein phosphatase calcineurin as a principal calmodulin target and of a secretion-related role for calbindin28kDa. Eur. J. Biochem. 1995, 230, 68–79. [Google Scholar] [CrossRef] [PubMed]
- Moradian-Oldak, J.; Goldberg, M. Amelogenin supra-molecular assembly in vitro compared with the architecture of the forming enamel matrix. Cells Tissues Organs 2005, 181, 202–218. [Google Scholar] [CrossRef] [PubMed]
- Bartlett, J.D.; Bernhard, G.; Michel, G.; Janet, M.-O.; Michael, L.P.; Malcolm, L.S.; Xin, W.; Shane, N.W.; Yan, L.Z. Protein–protein interactions of the developing enamel matrix. Curr. Top. Dev. Biol. 2006, 74, 57–115. [Google Scholar] [PubMed]
- Dusevich, V.; Xu, C.; Wang, Y.; Walker, M.P.; Gorski, J.P. Identification of a protein-containing enamel matrix layer which bridges with the dentine-enamel junction of adult human teeth. Arch. Oral Biol. 2012, 57, 1585–1594. [Google Scholar] [CrossRef] [PubMed]
- Vymetal, J.; Slabý, I.; Spahr, A.; Vondrásek, J.; Lyngstadaas, S.P. Bioinformatic analysis and molecular modelling of human ameloblastin suggest a two-domain intrinsically unstructured calcium-binding protein. Eur. J. Oral Sci. 2008, 116, 124–134. [Google Scholar] [CrossRef] [PubMed]
- Wald, T.; Bednárová, L.; Osička, R.; Pachl, P.; Sulc, M.; Lyngstadaas, S.P.; Slaby, I.; Vondrášek, J. Biophysical characterization of recombinant human ameloblastin. Eur. J. Oral Sci. 2011, 119, 261–269. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.J.; Scheven, B.A.; Takahashi, Y.; Ferracane, J.L.; Shelton, R.M.; Cooper, P.R. Dentine as a bioactive extracellular matrix. Arch. Oral Biol. 2012, 57, 109–121. [Google Scholar] [CrossRef] [PubMed]
- Petersson, U.; Hultenby, K.; Wendel, M. Identification, distribution and expression of osteoadherin during tooth formation. Eur. J. Oral Sci. 2003, 111, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Jágr, M.; Eckhardt, A.; Pataridis, S.; Mikšík, I. Comprehensive proteomic analysis of human dentin. Eur. J. Oral Sci. 2012, 120, 259–268. [Google Scholar] [CrossRef] [PubMed]
- Robey, P.G. Vertebrate mineralized matrix proteins: Structure and function. Connect. Tissue Res. 1996, 35, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Bosshardt, D.D.; Selvig, K.A. Dental cementum: The dynamic tissue covering of the root. Periodontol. 2000 1997, 13, 41–75. [Google Scholar] [CrossRef] [PubMed]
- Hammarström, L.; Alatli, I.; Fong, C.D. Origins of cementum. Oral Dis. 1996, 2, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Campisi, G.; Fede, O.D.; Roccia, P.; Nicola, F.D.; Falaschini, S.; Muzio, L. Lo Saliva: Its value as a biological matrix and current methods of sampling. Eur. J. Inflamm. 2006, 4, 11–19. [Google Scholar]
- Bassim, C.W.; Ambatipudi, K.S.; Mays, J.W.; Edwards, D.A.; Swatkoski, S.; Fassil, H.; Baird, K.; Gucek, M.; Melvin, J.E.; Pavletic, S.Z. Quantitative salivary proteomic differences in oral chronic graft-versus-host disease. J. Clin. Immunol. 2012, 32, 1390–1399. [Google Scholar] [CrossRef] [PubMed]
- Chianeh, Y.R.; Prabhu, K. Biochemical markers in saliva of patients with oral squamous cell carcinoma. Asian Pac. J. Trop. Dis. 2014, 4, S33–S40. [Google Scholar] [CrossRef]
- Levine, M. Susceptibility to dental caries and the salivary proline-rich proteins. Int. J. Dent. 2011, 2011, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Xiao, H.; Karlan, S.; Zhou, H.; Gross, J.; Elashoff, D.; Akin, D.; Yan, X.; Chia, D.; Karlan, B.; et al. Discovery and preclinical validation of salivary transcriptomic and proteomic biomarkers for the non-invasive detection of breast cancer. PLoS ONE 2010, 5, e15573. [Google Scholar] [CrossRef] [PubMed]
- Lamster, I.B. Evaluation of components of gingival crevicular fluid as diagnostic tests. Ann. Periodontol. 1997, 2, 123–137. [Google Scholar] [CrossRef] [PubMed]
- Tsuchida, S.; Satoh, M.; Umemura, H.; Sogawa, K.; Kawashima, Y.; Kado, S.; Sawai, S.; Nishimura, M.; Kodera, Y.; Matsushita, K.; et al. Proteomic analysis of gingival crevicular fluid for discovery of novel periodontal disease markers. Proteomics 2012, 12, 2190–2202. [Google Scholar] [CrossRef] [PubMed]
- Tsuchida, S.; Satoh, M.; Sogawa, K.; Kawashima, Y.; Kado, S.; Ishige, T.; Beppu, M.; Sawai, S.; Nishimura, M.; Kodera, Y.; et al. Application of proteomic technologies to discover and identify biomarkers for periodontal diseases in gingival crevicular fluid: A review. PROTEOM.-Clin. Appl. 2014, 8, 232–240. [Google Scholar] [CrossRef]
- Kunimatsu, K. Cathepsins B, H and L activities in gingival crevicular fluid from chronic adult periodontitis patients and exprimental gingivitis subjects. J. Periodontal Res. 1990, 25, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Dannies, P.S. Protein degradation in health and disease. Ciba foundation symposium 75 (new series). Yale J. Biol. Med. 1982, 55, 75. [Google Scholar]
- Baliban, R.C.; Sakellari, D.; Li, Z.; DiMaggio, P.A.; Garcia, B.A.; Floudas, C.A. Novel protein identification methods for biomarker discovery via a proteomic analysis of periodontally healthy and diseased gingival crevicular fluid samples. J. Clin. Periodontol. 2012, 39, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Ngo, L.H.; Darby, I.B.; Veith, P.D.; Locke, A.G.; Reynolds, E.C. Mass spectrometric analysis of gingival crevicular fluid biomarkers can predict periodontal disease progression. J. Periodontal Res. 2013, 48, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Silva-Boghossian, C.M.; Colombo, A.P.V.; Tanaka, M.; Rayo, C.; Xiao, Y.; Siqueira, W.L. Quantitative proteomic analysis of gingival crevicular fluid in different periodontal conditions. PLoS ONE 2013, 8, e75898. [Google Scholar] [CrossRef] [PubMed]
- Carneiro, L.G.; Venuleo, C.; Oppenheim, F.G.; Salih, E. Proteome data set of human gingival crevicular fluid from healthy periodontium sites by multidimensional protein separation and mass spectrometry. J. Periodontal Res. 2012, 47, 248–262. [Google Scholar] [CrossRef] [PubMed]
- Carneiro, L.G.; Nouh, H.; Salih, E. Quantitative gingival crevicular fluid proteome in health and periodontal disease using stable isotope chemistries and mass spectrometry. J. Clin. Periodontol. 2014, 41, 733–747. [Google Scholar] [CrossRef] [PubMed]
- Rody, W.J., Jr.; Holliday, L.S.; McHugh, K.P.; Wallet, S.M.; Spicer, V.; Krokhin, O. Mass spectrometry analysis of gingival crevicular fluid in the presence of external root resorption. Am. J. Orthod. Dentofac. Orthop. 2014, 145, 787–798. [Google Scholar] [CrossRef] [PubMed]
- Kinney, J.S.; Morelli, T.; Oh, M.; Braun, T.M.; Ramseier, C.A.; Sugai, J.V.; Giannobile, W.V. Crevicular fluid biomarkers and periodontal disease progression. J. Clin. Periodontol. 2014, 41, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Siqueira, W.L.; Custodio, W.; McDonald, E.E. New insights into the composition and functions of the acquired enamel pellicle. J. Dent. Res. 2012, 91, 1110–1118. [Google Scholar] [CrossRef] [PubMed]
- Vukosavljevic, D.; Custodio, W.; Buzalaf, M.A.R.; Hara, A.T.; Siqueira, W.L. Acquired pellicle as a modulator for dental erosion. Arch. Oral Biol. 2014, 59, 631–638. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.H.; Zimmerman, J.N.; Custodio, W.; Xiao, Y.; Basiri, T.; Hatibovic-Kofman, S.; Siqueira, W.L. Proteomic evaluation of acquired enamel pellicle during in vivo formation. PLoS ONE 2013, 8, e67919. [Google Scholar] [CrossRef] [PubMed]
- Lendenmann, U.; Grogan, J.; Oppenheim, F.G. Saliva and dental pellicle—A review. Adv. Dent. Res. 2000, 14, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Reich, M.; Hannig, C.; Al-Ahmad, A.; Bolek, R.; Kummerer, K. A comprehensive method for determination of fatty acids in the initial oral biofilm (pellicle). J. Lipid Res. 2012, 53, 2226–2230. [Google Scholar] [CrossRef] [PubMed]
- Siqueira, W.L.; Helmerhorst, E.J.; Zhang, W.; Salih, E.; Oppenheim, F.G. Acquired enamel pellicle and its potential role in oral diagnostics. Ann. N. Y. Acad. Sci. 2007, 1098, 504–509. [Google Scholar] [CrossRef] [PubMed]
- Mayhall, C.W. Concerning the composition and source of the acquired enamel pellicle of human teeth. Arch. Oral Biol. 1970, 15, 1327–1341. [Google Scholar] [CrossRef]
- Armstrong, W.G. Amino-acid composition of the acquired pellicle of human tooth enamel. Nature 1966, 210, 197–198. [Google Scholar] [CrossRef] [PubMed]
- Siqueira, W.L.; Margolis, H.C.; Helmerhorst, E.J.; Mendes, F.M.; Oppenheim, F.G. Evidence of intact histatins in the in vivo acquired enamel pellicle. J. Dent. Res. 2010, 89, 626–630. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, J.N.; Custodio, W.; Hatibovic-Kofman, S.; Lee, Y.H.; Xiao, Y.; Siqueira, W.L. Proteome and peptidome of human acquired enamel pellicle on deciduous teeth. Int. J. Mol. Sci. 2013, 14, 920–934. [Google Scholar] [CrossRef] [PubMed]
- Yu, T.; Volponi, A.A.; Babb, R.; An, Z.; Sharpe, P.T. Stem cells in tooth development, growth, repair, and regeneration. Curr. Top. Dev. Biol. 2015, 115, 187–212. [Google Scholar] [PubMed]
- Mjör, I.A.; Sveen, O.B.; Heyeraas, K.J. Pulp-dentin biology in restorative dentistry. Part 1: Normal structure and physiology. Quintessence Int. 2001, 32, 427–446. [Google Scholar] [PubMed]
- Farges, J.-C.; Alliot-Licht, B.; Renard, E.; Ducret, M.; Gaudin, A.; Smith, A.J.; Cooper, P.R. Dental pulp defence and repair mechanisms in dental caries. Mediat. Inflamm. 2015, 2015, 230251. [Google Scholar] [CrossRef] [PubMed]
- Robertson, A.; Lundgren, T.; Andreasen, J.O.; Dietz, W.; Hoyer, I.; Norén, J.G. Pulp calcifications in traumatized primary incisors. A morphological and inductive analysis study. Eur. J. Oral Sci. 1997, 105, 196–206. [Google Scholar] [CrossRef] [PubMed]
- Yamazoe, T.; Aoki, K.; Simokawa, H.; Ohya, K.; Takagi, Y. Gene expression of bone matrix proteins in a calcified tissue appeared in subcutaneously transplanted rat dental pulp. J. Med. Dent. Sci. 2002, 49, 57–66. [Google Scholar] [PubMed]
- Zafar, M.S.; Khurshid, Z.; Almas, K. Oral tissue engineering progress and challenges. Tissue Eng. Regen. Med. 2015, 12, 387–397. [Google Scholar] [CrossRef]
- Sandanayake, N.S.; Sinclair, J.; Andreola, F.; Chapman, M.H.; Xue, A.; Webster, G.J.; Clarkson, A.; Gill, A.; Norton, I.D.; Smith, R.C.; et al. A combination of serum leucine-rich α-2-glycoprotein 1, CA19–9 and interleukin-6 differentiate biliary tract cancer from benign biliary strictures. Br. J. Cancer 2011, 105, 1370–1378. [Google Scholar] [CrossRef] [PubMed]
- Pääkkönen, V.; Ohlmeier, S.; Bergmann, U.; Larmas, M.; Salo, T.; Tjäderhane, L. Analysis of gene and protein expression in healthy and carious tooth pulp with cDNA microarray and two-dimensional gel electrophoresis. Eur. J. Oral Sci. 2005, 113, 369–379. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Wu, L.; Ling, J.; Liu, L.; Liu, S.; Liu, W.; Li, M.; Xiao, Y. Differentially expressed protein profile of human dental pulp cells in the early process of odontoblast-like differentiation in vitro. J. Endod. 2008, 34, 1077–1084. [Google Scholar] [CrossRef] [PubMed]
- Tete, S.; Mastrangelo, F.; Scioletti, A.P.; Tranasi, M.; Raicu, F.; Paolantonio, M.; Stuppia, L.; Vinci, R.; Gherlone, E.; Ciampoli, C.; et al. Microarray expression profiling of human dental pulp from single subject. Clin. Investig. Med. 2008, 31, 55–61. [Google Scholar]
- McLachlan, J.L.; Smith, A.J.; Bujalska, I.J.; Cooper, P.R. Gene expression profiling of pulpal tissue reveals the molecular complexity of dental caries. Biochim. Biophys. Acta Mol. Basis Dis. 2005, 1741, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Eckhard, U.; Marino, G.; Abbey, S.R.; Tharmarajah, G.; Matthew, I.; Overall, C.M. The human dental pulp proteome and N-terminome: Levering the unexplored potential of semitryptic peptides enriched by TAILS to identify missing proteins in the human proteome project in underexplored tissues. J. Proteome Res. 2015, 14, 3568–3582. [Google Scholar] [CrossRef] [PubMed]
- Paik, Y.-K.; Hancock, W.S. Uniting ENCODE with genome-wide proteomics. Nat. Biotechnol. 2012, 30, 1065–1067. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Wei, X.; Ling, J.; Liu, L.; Liu, S.; Li, M.; Xiao, Y. Early osteogenic differential protein profile detected by proteomic analysis in human periodontal ligament cells. J. Periodontal Res. 2009, 44, 645–656. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Wei, X.; Ling, J.; Liu, L. A differential expression proteomic study of human periodontal ligament cell during osteogenic differentiation. Zhong Hua Kou Qiang Yi Xue Za Zhi 2009, 44, 212–216. [Google Scholar]
- Khurshid, Z.; Zafar, M.; Qasim, S.; Shahab, S.; Naseem, M.; AbuReqaiba, A. Advances in nanotechnology for restorative dentistry. Materials 2015, 8, 717–731. [Google Scholar] [CrossRef]
- Sheikh, Z.; Najeeb, S.; Khurshid, Z.; Verma, V.; Rashid, H.; Glogauer, M. Biodegradable materials for bone repair and tissue engineering applications. Materials 2015, 8, 5744–5794. [Google Scholar] [CrossRef]
- Najeeb, S.; Zafar, M.S.; Khurshid, Z.; Siddiqui, F. Applications of polyetheretherketone (PEEK) in oral implantology and prosthodontics. J. Prosthodont. Res. 2016, 60, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Matinlinna, J.P.; Zeeshan, S.; Mohamed-Nur, A.; Nader, H.; Mohammad, A.J.; Zohaib, K. Barrier membranes for periodontal guided tissue regeneration applications. In Handbook of Oral Biomaterials; Pan Stanford Publishing: Boca Raton, FL, USA, 2014; pp. 605–636. [Google Scholar]
- Najeeb, S.; Khurshid, Z.; Matinlinna, J.P.; Siddiqui, F.; Nassani, M.Z.; Baroudi, K. Nanomodified peek dental implants: Bioactive composites and surface modification—A review. Int. J. Dent. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Najeeb, S.; Khurshid, Z.; Zafar, M.S.; Ajlal, S. Applications of light amplification by stimulated emission of radiation (lasers) for restorative dentistry. Med. Princ. Pract. 2016, 25, 201–211. [Google Scholar] [CrossRef] [PubMed]
- Naseem, M.; Khurshid, Z.; Khan, H.A.; Niazi, F.; Shahab, S.; Zafar, M.S. Oral health challenges in pregnant women: Recommendations for dental care professionals. Saudi J. Dent. Res. 2015. [Google Scholar] [CrossRef]
- Łagocka, R.; Jakubowska, K.; Chlubek, D.; Buczkowska-Radlińska, J. Elution study of unreacted TEGDMA from bulk-fill composite (SDR™ Dentsply) using HPLC. Adv. Med. Sci. 2015, 60, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Miletic, V.; Santini, A.; Trkulja, I. Quantification of monomer elution and carbon–carbon double bonds in dental adhesive systems using HPLC and micro-Raman spectroscopy. J. Dent. 2009, 37, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Boyan, B.D.; Weesner, T.C.; Lohmann, C.H.; Andreacchio, D.; Carnes, D.L.; Dean, D.D.; Cochran, D.L.; Schwartz, Z. Porcine fetal enamel matrix derivative enhances bone formation induced by demineralized freeze dried bone allograft in vivo. J. Periodontol. 2000, 71, 1278–1286. [Google Scholar] [CrossRef] [PubMed]
- Derhami, K.; Zheng, J.; Li, L.; Wolfaardt, J.F.; Scott, P.G. Proteomic analysis of human skin fibroblasts grown on titanium: Novel approach to study molecular biocompatibility. J. Biomed. Mater. Res. 2001, 56, 234–244. [Google Scholar] [CrossRef]
- Koin, P.J.; Kilislioglu, A.; Zhou, M.; Drummond, J.L.; Hanley, L. Analysis of the degradation of a model dental composite. J. Dent. Res. 2008, 87, 661–665. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.Y.; Kang, G.C.; Jeong, Y.J.; Kim, S.H.; Kwak, Y.G.; Kim, W.J. Proteomic analysis in cyclosporin a-induced overgrowth of human gingival fibroblasts. Biol. Pharm. Bull. 2009, 32, 1480–1485. [Google Scholar] [CrossRef] [PubMed]
- Taiyoji, M.; Shitomi, Y.; Taniguchi, M.; Saitoh, E.; Ohtsubo, S. Identification of proteinaceous inhibitors of a cysteine proteinase (an Arg-specific gingipain) from Porphyromonas gingivalis in rice grain, using targeted-proteomics approaches. J. Proteome Res. 2009, 8, 5165–5174. [Google Scholar] [CrossRef] [PubMed]
- Zilm, P.S.; Bartold, P.M. Proteomic identification of proteinase inhibitors in the porcine enamel matrix derivative, EMD®. J. Periodontal Res. 2011, 46, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Dorkhan, M.; Chávez de Paz, L.E.; Skepö, M.; Svensäter, G.; Davies, J.R. Effects of saliva or serum coating on adherence of Streptococcus oralis strains to titanium. Microbiology 2012, 158, 390–397. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; An, M.; Wang, Q.; Liu, X.; Lai, W.; Zhao, X.; Wei, S.; Ji, J. Quantitative proteomic analysis of human osteoblast-like MG-63 cells in response to bioinert implant material titanium and polyetheretherketone. J. Proteom. 2012, 75, 3560–3573. [Google Scholar] [CrossRef] [PubMed]
- Oakley, M.; Barthel, D.; Bykov, Y.; Garibaldi, J.; Burke, E.; Krasnogor, N.; Hirst, J. Search strategies in structural bioinformatics. Curr. Protein Pept. Sci. 2008, 9, 260–274. [Google Scholar] [CrossRef] [PubMed]
- Khurshid, Z.; Shariq, N.; Maria, M.; Syed, F.M.; Syed, Q.R.; Sana, Z.; Farshid, S.; Muhammad, S.Z. Histatin peptides: Pharmacological functions and its applications in dentistry. Saudi Pharm. J. 2016. [Google Scholar] [CrossRef]
| Sample | Disease Condition | Proteomic Tools | Identified Markers | References |
|---|---|---|---|---|
| Whole mouth saliva (WS) | Oral squamous cell carcinoma | Shotgun proteome analysis, Western blotting (WB) and Enzyme Linked Immuno-Sorbent Assay (ELISA) | MRP14, M2BP, CD59, catalase, profilin, M2BP, involucrin, histone H1, S100A12, and S100P | [17] |
| WS | Denture stomatitis | Surface-Enhanced Laser Desorption/Ionization (SELDI) time-of-flight-(TOF)/ mass spectrometry (MS), liquid chromatography (LC)- Matrix-Assisted Laser Desorption/Ionization (MALDI)-TOF-MS | Statherin, desmocollin-2, kininogen-1, carbonic anhydrase-6, cystatin SN, cystatin c, peptidyl-prolyl cis-trans isomerase and immunoglobulin fragments | [18] |
| WS | Primary Sjögren’s syndrome | two dimensional electrophoresis (2-DE), MALDI-TOF/MS, WB and ELISA | Carbonic anhydrase VI, α-amylases precursor, epidermal fatty acid binding protein (E-FABP), macroglobulin (b-2), immunoglobulin k light chain (IGK-light chain) and glyceraldehydes-3-phosphate dehydrogenase (G3PDH) | [19] |
| WS | Secondary Sjögren’s syndrome | 2-DE, MALDI-TOF-MS, WB and ELISA | Decrease ↓ Proline rich proteins (PRPs), ↓ Cystatin C, ↓ Lysozyme C and histatin, Increase ↑ Kallikrein and defensins | [20] |
| WS | GVHD | Tandem MS & ELISA | IL-1 receptor antagonist and cystatin B | [21] |
| WS | Protein-energy undernutrition | 2-DE Gel and Image Master two dimensional 2D | Cyclic-dependent protein kinase | [22] |
| WS | Squamous cell carcinoma (head and neck region) | C4 Reverse Phase-High Pressure Liquid Chromatography (RP-HPLC), and LC-MS/MS | MRP14, Profilin, CD59, catalase and M2BP | [23] |
| WS | Diabetes (type-2) | 2D-LC-MS/MS, WB and ELISA | α-1-antitrypsin (A1AT), α-2 macroglobulin (A2MG), transthyretin (TTR), salivary α -amylase (AMYS), cystatin C (Cys-C) | [24] |
| WS | Edentulous patient with type-2 diabetes | 2D-LC-MS/MS | Serum amyloid-A and glyceraldehyde-3-phosphate dehydrogenase are increased, serotransferrin and amylase, palate, lung and nasal epithelium associated proteins (PLUNC) are reduced | [25] |
| Unstimulated whole mouth saliva (USWS) | Squamous cell carcinoma (oral mucosa) | Ultra-Pressure Liquid Chromatography (UPLC-MS), Hydrophilic Interaction Liquid Chromatography (HILIC) | l-carnitine, choline, betaine and pipecolinic acid | [26] |
| Saliva (parotid glands) | Caries | HPLC-MS/MS | matrix metalloproteinase-9 (MMP9), mucin-7 (MUC7), lactotransferrin (LTF), carbonic anhydrase 6 (CA6), azurocidin (AZU), and cold agglutinin | [27] |
| WS | Orthodontic tooth movement | 2-DE, MALDI-TOF/tandem mass spectrometry (TEM) | Protein S100-A9, CRISP-3, Immunoglobulin J chain and Ig α-1 chain C region | [28] |
| USWS | Aggressive periodontitis | 2-DE/HPLC–Electrospray Ionization (ESI)-MS | Increase in serum albumin, immunoglobulin Ig γ2, α2 chain C region, zinc-α2 glycoprotein, salivary α-amylase and vitamin D-binding proteins. Decrease in lactotransferrin, carbonic anhydrase 6, elongation factor 2, 14-3-3 sigma, short palate, lung and nasal epithelium carcinoma-associated protein-2 | [29] |
| USWS | Periodontitis chronic | 2-DE/MALDI-TOF/TOF MS and nLC-Q-TOF | Rise in serum albumin, hemoglobin, immunoglobulin and α-amylase | [30] |
| WS | Periodontitis in obese patient | SELDI-TOF-MS | Albumin, haemoglobin (α and β chains) and α-defensins (1, 2 & 3) | [31] |
| USWS | Gingivitis | 2-DE/MALDI-TOF/TOF MS and nLC-Q-TOF | Zymogen granule protein-16 homolog B mucin, S100-A9, histatin, proline-rich-protein, 3, lipocalin-1 precursor, carbonic anhydrase 6, prolactin-induced protein, cystatin, keratins | [32] |
| Dental cementum | - | Nano-Acuity HPLC and LTQ-FT ultra | Osteomodulin (OMD), biglycan (BGN), insulin-like growth factor II (IGF2), pigment epithelium-derived factor (SERPINF1) and POSTN | [33] |
| Fine Needle Aspiration (FNA) fluid | Parotid gland tumor (Benign origin) | Nano LC-ESI-MS/MS and LTQ-Qrbitrap velos analysis and Western blot analysis | Ig γ-1 and kappa chain and Ig α-1 chain C regions, S100A9, macrophage capping proteins, apolipoprotein E and α crystalline B chain, annexin (A1 and A4) | [34] |
| Gingival crevicular fluids (GCF) | Gingivitis and chronic periodontitis | 2-DE-LC-ESI-MS and Nano-LC-ESI-MS | Fibronectin, keratin, neutrophil, defensin3, Immunoglobulins, lactotransferrin precursor, 14-3-3 protein ζ/δ and α-actinin | [35] |
| Dentin | - | LC-MS/MS | Biglycan, osteoglycin, osteopontin, osteocalcin, asporin, lumican, mimecan, DSPP and SOD3 | [36] |
| Dental pulp | - | 2-DE, Nano-LCMS/MS | 342 proteins identified | [37] |
| Periodontal fibers (PDL) | - | 2-DE, MALDI-TOF, Western blot, | 117 proteins identified | [38] |
| Acquired enamel pellicle (AEP) | - | LC-ESI-MS/MS | 130 proteins identified | [39] |
| Author | Sample Collection Sites | Collection Method | Proteomic Tool | Number of Identified Proteins | Outcome of Study | Reference |
|---|---|---|---|---|---|---|
| Baliban et al. | Collected from pre-selected sites with probing depth >6 mm and <8 mm in periodontitis patients and for periodontaly health from mesio-buccal sites of first molar | Filter strips (Periopapers®, Interstate Drug. Exchange, Amityville, NY, USA) | Protein digest with trypsin, HPLC, fragmented analysis with tandem mass spectrometry (MS/MS) | 432 human proteins identified (120 new) | Study identified novel biomarkers from GCF of periodontaly healthy and chronic periodontitis patients | [68] |
| Tsuchida et al. | Labial side of maxillary incisors without crown and restoration | Absorbent paper points (ZIPPERER®, Munich, Germany) | 2-DE, Sodium-Dodecyl-Sulfate-Polyacrylamide Gel Electrophoresis (SDS-PAGE), WB analysis, HPLC with LTQ-XL, HPLC with LTQ-Orbitrap XL, LC-MS/MS | 327 proteins identified | SOD1 and DCD were significantly increase ↑ in GCF of periodontal patients | [64] |
| Carneiro et al. | Healthy gingival sulcus of the second and third molar teeth | Periopapers®, USA | Trypsin digested followed by nano-flow liquid chromatography electrospray ionization tandem mass spectrometry (LC-ESI-MS/MS) analysis and ELISA for human albumin analysis | 199 proteins identified | Provide proteins analysis of healthy periodontium and explore GCF composition with new groups of proteins specific to GCF microenvironment | [71] |
| Ngo et al. | Five deepest sites and molar sites except mesial surface were excluded | Microcaps (glass micocapillary tubes); Drummed Scientific, Brookmall, PA, USA | Matrix-assisted laser desposition/ionization time-of-flight (MALDI-TOF) mass spectrometry (MS) | GCF mass spectra could be best for analyzing attachment loss and diagnosis of periodontal diseases | [69] | |
| Carina, et al. | Chronic Periodontitis patients sample were taken from different sites (5 deep sites, 5 shallow sites with gingivitis, and 4 without bleeding on probing sites) | Periopaper strip (ProFlow Inc., Amityville, NY, USA) | Reversed- phase (RP) LC-ESi-MS/MS and ELISA | 230 proteins identified | Concluded marked differences in GCF proteomics in different disease profiles | [70] |
| Carneiro et al. | The pre-selected specific sites with moderate and severe chronic periodontal disease were defined by pocket depth of 5–7 mm (24 patients) and >7 mm (16 patients) | Periopaper strips (Oraflow, Plainview, NY, USA) | SDS-PAGE, Isotope-Coded-Affinity-Tag (ICAT) labelling, mTRAQ labelling, Nano-LC-ESI-MS/MS, Human Albumin ELISA Kit, and S100-A9 protein quantification by ELISA | 238 proteins Identified | Innovative approach concluded the novel changes in host and microbial derived GCF proteome of periodontal patients | [72] |
| Rody Jr et al. | Collected from a deciduous second molar with radiographic evidence of root resorption on 1 quadrant (experimental site) and from the permanent first molar on the contralateral quadrant (control site) in the same jaw | Periopaper strips (Oraflow, Plainview, NY, USA) | One dimensional LC-MS and Two dimensional (2D) LC-MS | 2789 proteins in control group and 2421 proteins in root resorption group | Mass spectrometry is useful tool for analyzing external root resorption | [73] |
| Kinney et al. | Collection from the mesio-buccal aspect of each site (tooth) for up-to 28 teeth per patient | Methylcellulose strip (Pro Flow, Inc., Amityville, NY, USA) | ELISA and Quantibody Human Cytokine Array (HCA) | This method offer improved patient monitoring and disease control | [74] | |
| Huynh et al. | Collection were chosen based on how well they represented the healthy, gingivitis and chronic periodontitis inclusion criteria | Glass-microcapillary tube (Drummond Scientific, Brookmall, PA, USA) | One dimensional Gel-Electrophoresis and Nano-LC-ESI-MS | 121 proteins identified | Concluded various biomarkers which differentiate between healthy periodontium, gingivitis and chronic periodontitis | [35] |
| Author Name | Title of Study | Outcomes | References |
|---|---|---|---|
| Boyan et al. | Porcine fetal enamel matrix derivative enhances bone formation induced by demineralized freeze dried bone allograft in vivo | Emdogain contains a number of low-molecular-weight proteins (mainly amelogenins), associated with cementogenesis and osteogenesis during tooth development | [109] |
| Derhami et al. | Proteomic analysis of human skin fibroblasts grown on titanium: Novel approach to study molecular biocompatibility | Gain a better understanding of the molecular basis of biocompatibility of human skin fibroblast on titanium | [110] |
| Koin et al. | Analysis of the degradation of a model dental composite | Liquid chromatography mass spectrometry (LC-MS) found leaching of intact BisGMA and several degradation products that contained the bisphenol A moiety from the overlayer into distilled water after 2 weeks of aging | [111] |
| Jung et al. | Proteomic analysis in cyclosporin A (CsA)-induced overgrowth of human gingival fibroblast (HGF) | The CsA-treated HGF demonstrated that Prx 1 may play a crucial role in the HGF proliferation induced by CsA and proteomic analysis data provide an efficient approach in understanding the mechanisms of HGF proliferation by CsA | [112] |
| Taiyoji et al. | Identification of proteinaceous inhibitors of a cysteine proteinase (an Arg-specific gingipain) from Porphyromonas gingivalis in rice grain, using targeted-proteomics approaches | These results suggest that these rice proteins may be useful as nutraceutical ingredients for the prevention and management of periodontal diseases | [113] |
| Haigh et al. | Alterations in the salivary proteome associated with periodontitis | Results highlight the predominant involvement of S100 proteins in the host response during periodontitis | [113] |
| Zilm and Bartold et al. | Proteomic identification of proteinase inhibitors in the porcine enamel matrix derivative, EMD® | Enamel matrix derivatives (EMD) contains a number of high-molecular-weight compounds which include the proteinase inhibitors, fetuin A and α1-antichymotrypsin | [114] |
| Dorkhan et al. | Effects of saliva or serum coating on adherence of Streptococcus oralis strains to titanium | The adherence of LA11 and 89C strain to the moderately rough surfaces coated with saliva was more than twice that seen on the smooth saliva coated surfaces. This clearly demonstrates that surface topography is, at least to some degree, maintained in the presence of a saliva coating | [115] |
| Zhao et al. | Quantitative proteomic analysis of human osteoblast-like MG-63 cells in response to bio-inert implant material titanium and polyetheretherketone (PEEK) | Titanium and polyetheretherketone (PEEK) induces similar response in osteoblast proteome and PEEK causing worse proliferation was related to mRNA processing | [116] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khurshid, Z.; Zohaib, S.; Najeeb, S.; Zafar, M.S.; Rehman, R.; Rehman, I.U. Advances of Proteomic Sciences in Dentistry. Int. J. Mol. Sci. 2016, 17, 728. https://doi.org/10.3390/ijms17050728
Khurshid Z, Zohaib S, Najeeb S, Zafar MS, Rehman R, Rehman IU. Advances of Proteomic Sciences in Dentistry. International Journal of Molecular Sciences. 2016; 17(5):728. https://doi.org/10.3390/ijms17050728
Chicago/Turabian StyleKhurshid, Zohaib, Sana Zohaib, Shariq Najeeb, Muhammad Sohail Zafar, Rabia Rehman, and Ihtesham Ur Rehman. 2016. "Advances of Proteomic Sciences in Dentistry" International Journal of Molecular Sciences 17, no. 5: 728. https://doi.org/10.3390/ijms17050728
APA StyleKhurshid, Z., Zohaib, S., Najeeb, S., Zafar, M. S., Rehman, R., & Rehman, I. U. (2016). Advances of Proteomic Sciences in Dentistry. International Journal of Molecular Sciences, 17(5), 728. https://doi.org/10.3390/ijms17050728









