Herbal Hepatotoxicity: Clinical Characteristics and Listing Compilation
Abstract
:1. Introduction
2. Data Sources and Searches
2.1. Search Terms
2.2. Data Extraction
3. Variability of Herbal Product Types
4. Herbal Product Quality
4.1. Plant Identification and Its Parts
4.2. Misidentifications, Adulterants, and Impurities
4.2.1. Misidentifications
4.2.2. Adulterants and Unlabeled Contaminants
4.2.3. Impurities
5. Pathogenetic Aspects of Herb Induced Liver Injury (HILI)
5.1. General Considerations
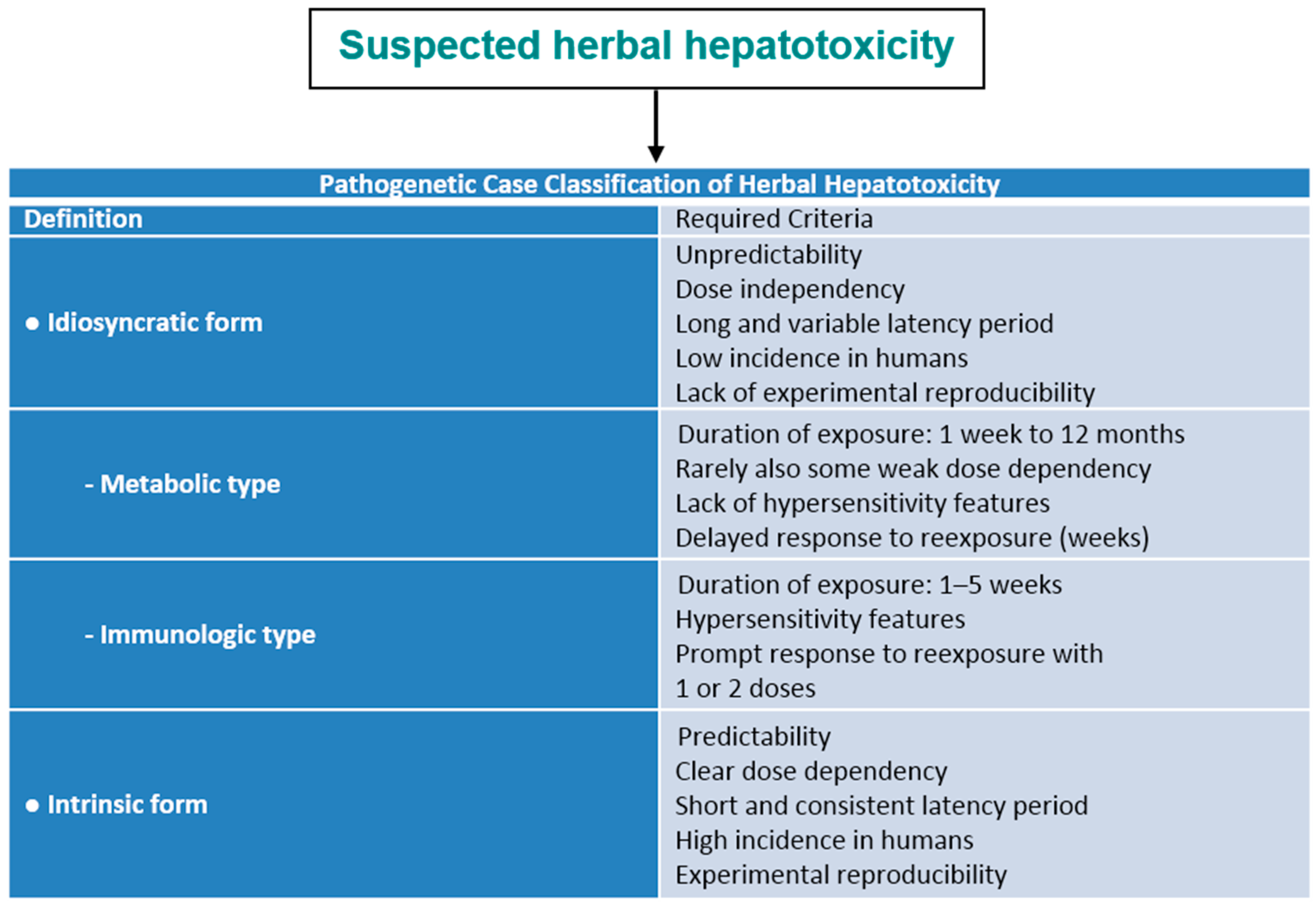
5.2. Idiosyncratic and Intrinsic HILI
5.2.1. Idiosyncratic HILI Type
5.2.2. Intrinsic HILI Type
6. Clinical Features of HILI
7. Progress in Developing Valid Diagnostic Biomarkers
8. HILI Case Criteria
9. Liver Injury versus Adaptation
10. HILI Case Classification
11. RUCAM as the Most Used Method to Assess Causality
12. Reexposure
13. Herbs with Established Causality for HILI
14. Questionable and Lacking Causality
15. Alternative Diagnoses
16. Confounding Variables
17. Listing Compilation of Published Reports with Potentially Hepatotoxic Herbs
18. HILI Outcome and Management
18.1. Natural Course and Discontinuation of Herbal Use
18.2. Severe HILI and Hy’s Law
18.3. Adaptation
19. HILI Outbreaks
20. Regulatory Issues
21. Conclusions
Conflicts of Interest
References
- Tu, Y.; Ni, M.Y.; Zhong, Y.R.; Li, L.N.; Cui, S.L.; Zhang, M.Q.; Qang, W.Z.; Hi, Z.; Li, X.T. Studies on the constituents of Artemisia annua Part II. Planta Medica 1982, 44, 143–145. [Google Scholar]
- Tu, Y. The development of new antimalarial drugs: Qinghaosu and dihydro-qinghaosu. Chin. Med. J. 1999, 112, 976–977. [Google Scholar]
- Efferth, T.; Romero, M.R.; Wolf, D.G.; Stamminger, T.; Marin, J.J.G.; Marschall, M. The antiviral activities of artemisinin and artesunate. Clin. Infect. Dis. 2008, 47, 804–808. [Google Scholar] [CrossRef] [PubMed]
- Maridass, M.; de Britto, A.J. Origin of plant derived medicines. Ethnobot Leaflets 2008, 12, 373–387. [Google Scholar]
- Pelkonen, O.; Xu, O.; Fan, T.P. Why is research on herbal medicinal products important and how can we improve quality? J. Tradit. Complement. Med. 2014, 4, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Ekor, M. The growing use of herbal medicines: Issues relating to adverse reactions and challenges in monitoring safety. Front. Pharmacol. 2014, 4, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Wolff, A.; Frenzel, C.; Eickhoff, A.; Schulze, J. Herbal traditional Chinese medicine and its evidence base in gastrointestinal disorders. World J. Gastroenterol. 2015, 21, 4466–4490. [Google Scholar] [PubMed]
- Teschke, R.; Eickhoff, A. Herbal hepatotoxicity in traditional and modern medicine: Actual key issues and new encouraging steps. Front. Pharmacol. 2015, 6, 72. [Google Scholar] [CrossRef] [PubMed]
- Calitz, C.; du Plessis, L.; Gouws, C.; Steyn, D.; Steenekamp, J.; Muller, C.; Hamman, S. Herbal hepatotoxicity: Current status, examples, and challenges. Expert Opin. Drug Metab. Toxicol. 2015, 11, 1551–1565. [Google Scholar] [CrossRef] [PubMed]
- Zhao, P.; Wang, C.; Liu, W.; Chen, G.; Liu, X.; Wang, X.; Wang, B.; Yu, L.; Sun, Y.; Liang, X.; et al. Causes and outcomes of acute liver failure in China. PLoS ONE 2013, 8, e80991. [Google Scholar] [CrossRef] [PubMed]
- Avigan, M.I.; Mozersky, R.P.; Seeff, L.B. Scientific and regulatory perspectives in herbal and dietary supplement associated hepatotoxicity in the United States. Int. J. Mol. Sci. 2016, 17, 331. [Google Scholar] [CrossRef] [PubMed]
- Danan, G.; Teschke, R. RUCAM in drug and herb induced liver injury: The update. Int. J. Mol. Sci. 2016, 17, 14. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Zhang, L.; Melzer, L.; Schulze, J.; Eickhoff, A. Green tea extract and the risk of drug-induced liver injury. Expert Opin. Drug Metab. Toxicol. 2014, 10, 1663–1676. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Yan, J.; Liu, X.; Ye, Z.; Yang, X.; Meyboom, R.; Chan, K.; Shaw, D.; Duez, P. Pharmacovigilance practice and risk control of Traditional Chinese Medicine drugs in China: Current status and future perspective. J. Ethnopharmacol. 2012, 140, 519–525. [Google Scholar]
- Teschke, R.; Schwarzenboeck, A.; Eickhoff, A.; Frenzel, C.; Wolff, A.; Schulze, J. Clinical and causality assessment in herbal hepatotoxicity. Expert Opin. Drug Saf. 2013, 12, 339–366. [Google Scholar] [CrossRef] [PubMed]
- Health Products and Food Branch Marketed Health Products Directorate Canadian Adverse Reaction Newsletter Editorial Team. Available online: http://www.hc-sc.gc.ca/dhp-mps/medeff/bulletin/carn-bcei_v20n1-eng.php#a1t1 (accessed on 15 March 2016).
- Ko, R.J. Adulterants in Asian patent medicines. N. Engl. J. Med. 1998, 339, 847. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.F.; Wen, K.C.; Hsiao, M.L. Adulteration by synthetic therapeutic substances of traditional Chinese medicines in Taiwan. J. Clin. Pharmacol. 1997, 37, 344–350. [Google Scholar] [CrossRef] [PubMed]
- Espinoza, E.O.; Mann, M.J.; Bleasdell, B. Arsenic and mercury in traditional Chinese herbal balls. N. Engl. J. Med. 1995, 333, 803–804. [Google Scholar] [CrossRef] [PubMed]
- Gertner, E.; Marshall, P.S.; Filandrinos, D.; Potek, A.S.; Smith, T.M. Complications resulting from the use of Chinese herbal medications containing undeclared prescription drugs. Arthritis. Rheum. 1995, 38, 614–617. [Google Scholar] [CrossRef] [PubMed]
- Kang-Yum, E.; Oransky, S.H. Chinese patent medicine as a potential source of mercury poisoning. Vet. Hum. Toxicol. 1992, 34, 235–238. [Google Scholar] [PubMed]
- Navarro, V.J.; Bonkovsky, H.L.; Hwang, S.I.; Vega, M.; Barnhart, H.; Serrano, J. Catechins in dietary supplements and hepatotoxicity. Dig. Dis. Sci. 2013, 58, 2682–2690. [Google Scholar] [CrossRef] [PubMed]
- Mazzanti, G.; Menniti-Ippolito, F.; Moro, P.A.; Cassetti, F.; Raschetti, R.; Santuccio, C.; Mastrangelo, S. Hepatotoxicity from green tea: A review of the literature and two unpublished cases. Eur. J. Clin. Pharmacol. 2009, 65, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Sarma, D.N.; Barrett, M.L.; Chavez, M.L.; Gardiner, P.; Ko, R.; Mahady, G.B.; Marles, R.J.; Pellicore, L.S.; Giancaspro, G.I.; Low Dog, T. Safety of green tea extract: A systematic review by the US Pharmacopeia. Drug Saf. 2008, 31, 469–484. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.L.; Yu, G.Y.; Chen, J. Clinical analysis of hepatic veno-occlusive disease induced by Sedum aizoon. Zhongguo Zhong Yao Za Zhi 2008, 33, 2402–2404. (In Chinese) [Google Scholar] [PubMed]
- Lin, G.; Wang, J.Y.; Li, N.; Li, M.; Gao, H.; Ji, Y.; Zhang, F.; Wang, H.; Zhou, Y.; Ye, Y.; et al. Hepatic sinusoidal obstruction syndrome associated with consumption of Gynura segetum. J. Hepatol. 2011, 54, 666–673. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.S.; Xiao, S.S.; He, J.F. Analysis of alkaloids in Sedum aizoon and establishment of hepatic veno-occlusive model in mice. Chin. J. Integr. Trad. Western Med. Dig. 2006, 14, 311–313. [Google Scholar]
- Gao, H.; Li, N.; Wang, J.Y.; Zhang, S.C.; Lin, G. Definitive diagnosis of hepatic sinusoidal obstruction syndrome induced by pyrrolizidine alkaloids. J. Dig. Dis. 2012, 13, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.Y.; Gao, H. Tusanqi and hepatic sinusoidal obstruction syndrome. J. Dig. Dis. 2014, 15, 105–107. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Wolff, A.; Frenzel, C.; Schulze, J.; Eickhoff, A. Herbal hepatotoxicity: A tabular compilation of reported cases. Liver Int. 2012, 32, 1543–1556. [Google Scholar] [CrossRef] [PubMed]
- Dai, H.F.; Gao, Y.; Yang, M.; Yu, C.H.; Gu, Z.Y.; Chen, W.X. Hepatic veno-occlusive disease induced by Gynura segetum: Report of two cases. Hepatobiliary Pancreat. Dis. Int. 2006, 5, 406–408. [Google Scholar] [PubMed]
- Kumana, C.R.; Ng, M.; Lin, H.J.; Ko, W.; Wu, P.C.; Todd, D. Hepatic veno-occlusive disease due to toxic alkaloid in herbal tea. Lancet 1983, 322, 1360–1361. [Google Scholar] [CrossRef]
- Kumana, C.R.; Ng, M.; Lin, H.J.; Ko, W.; Wu, P.C.; Todd, D. Herbal tea induced hepatic veno-occlusive disease: Quantification of toxic alkaloid exposure in adults. Gut 1985, 26, 101–104. [Google Scholar] [CrossRef] [PubMed]
- Culvenor, C.C.J.; Edgar, J.A.; Smith, L.W.; Kumana, C.R.; Lin, H.J. Heliotropium lasiocarpum Fisch and Mey identified as cause of veno-occlusive disease due to herbal tea. Lancet 1986, 1, 978. [Google Scholar] [CrossRef]
- Ernst, E. Heavy metals in traditional Indian remedies. Eur. J. Clin. Pharmacol. 2002, 57, 891–896. [Google Scholar] [CrossRef] [PubMed]
- Ernst, E. Review article. Methodological aspects of Traditional Chinese Medicine (TCM). Ann. Acad. Med. Singap. 2006, 35, 773–774. [Google Scholar] [PubMed]
- Saper, R.B.; Phillips, R.S.; Sehgal, A.; Khouri, N.; Davis, R.B.; Paquin, J.; Thuppil, V.; Kalses, S.N. Lead, mercury, and arsenic in US- and Indian-manufactured Ayurvedic medicines sold via the internet. JAMA 2008, 300, 915–923. [Google Scholar] [CrossRef] [PubMed]
- Bunchorntavakul, C.; Reddy, K.R. Review article: Herbal and dietary supplement hepatotoxicity. Aliment Pharmacol. Ther. 2013, 37, 3–17. [Google Scholar] [CrossRef] [PubMed]
- Dantuluri, S.; North-Lewis, P.; Karthik, S.V. Gotu Kola induced hepatotoxicity in a child—Need for causation with alternative remedies. Dig. Liver Dis. 2011, 43, 500. [Google Scholar] [CrossRef] [PubMed]
- Jorge, O.A.; Jorge, A.D. Hepatotoxicity associated with the ingestion of Centella asiatica. Rev. Esp. Enferm. Dig. 2005, 97, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Bahre, R. Severe hepatotoxicity by Indian Ayurvedic herbal products, A structured causality assessment. Ann. Hepatol. 2009, 8, 258–266. [Google Scholar] [PubMed]
- Fleig, W.W.; Morgan, M.Y.; Holzer, M.A. European multicenter study group. The ayurvedic drug Liv.52 in patients with alcoholic cirrhosis. Results of a prospective, randomized, double blind, placebo-controlled clinical trial. J. Hepatol. 1997, 126, 127. [Google Scholar]
- Adachi, M.; Saito, H.; Kobayashi, H.; Horie, Y.; Kato, S.; Yoshioka, M.; Ishii, H. Hepatic injury in 12 patients taking the herbal loss aids Chaso and Onshido. Ann. Intern. Med. 2003, 139, 488–492. [Google Scholar] [CrossRef] [PubMed]
- Lau, G.; Lo, D.S.; Yao, Y.J.; Leong, H.T.; Chan, C.L.; Chu, S.S. A fatal case of hepatic failure possibly induced by nitrosofenfluramine: A case report. Med. Sci. Law 2004, 44, 252–263. [Google Scholar] [CrossRef] [PubMed]
- Kanda, T.; Yokosuka, O.; Okada, O.; Suzuki, Y.; Saisho, H. Severe hepatotoxicity associated with Chinese diet product “Ohnshidou-Genbi-Kounou”. J. Gastroenterol. Hepatol. 2003, 18, 354–355. [Google Scholar] [CrossRef] [PubMed]
- Kanda, T.; Yokosuka, O.; Tada, M.; Kurihara, T.; Yoshida, S.; Suzuki, Y.; Nagao, K.; Saisho, H. N-nitroso-fenfluramine hepatotoxicity resembling chronic hepatitis. J. Gastroenterol. Hepatol. 2003, 18, 999–1000. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R. Traditional Chinese Medicine induced liver injury. J. Clin. Translat. Hepatol. 2014, 2, 80–94. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Schulze, J.; Eickhoff, A.; Wolff, A.; Frenzel, C. Review Article: Mysterious Hawaii liver disease case—Naproxen overdose as cause rather than OxyELITE Pro? J. Liver Clin. Res. 2015, 2, 1013. [Google Scholar]
- Teschke, R.; Schwarzenboeck, A.; Frenzel, C.; Schulze, J.; Eickhoff, A.; Wolff, A. The mystery of the Hawaii liver disease cluster in summer 2013: A pragmatic and clinical approach to solve the problem. Ann. Hepatol. 2016, 15, 91–119. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Eickhoff, A. The Honolulu Liver disease cluster at the Medical Center: Its mysteries and challenges. Int. J. Mol. Sci. 2016, 17, 476. [Google Scholar] [CrossRef] [PubMed]
- Ernst, E. Adulteration of Chinese herbal medicines with synthetic drugs: A systematic review. J. Int. Med. 2002, 252, 107–113. [Google Scholar] [CrossRef]
- Efferth, T.; Kaina, B. Toxicities by herbal medicines with emphasis to Traditional Chinese medicine. Curr. Drug Metab. 2011, 12, 989–996. [Google Scholar] [CrossRef] [PubMed]
- Shaw, D. Toxicological risks of Chinese herbs. Planta Med. 2010, 76, 2012–2018. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Qiu, S.X.; Xuan, T.D.; Lebot, V. Kava and kava hepatotoxicity: Requirements for novel experimental, ethnobotanical, and clinical studies based on a review of the evidence. Phytother. Res. 2011, 25, 1262–1274. [Google Scholar] [CrossRef] [PubMed]
- Rowe, A.; Ramzan, I. Letter to the Editor: Are mould hepatotoxins responsible for kava hepatotoxicity? Phytother. Res. 2012, 26, 1768–1770. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Sarris, J.; Lebot, V. Contaminant hepatotoxins as culprits for kava hepatotoxicity—Fact or fiction? Phytother. Res. 2013, 27, 472–474. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Qiu, S.X.; Lebot, V. Herbal hepatotoxicity by kava: Update on pipermethystine, flavokavain B, and mould hepatotoxins as primarily assumed culprits. Dig. Liver Dis. 2011, 43, 676–681. [Google Scholar] [CrossRef] [PubMed]
- Lebot, V. The quality of kava consumed in the South Pacific. HerbalGram 2006, 71, 34–37. [Google Scholar]
- Schmidt, M. Quality criteria for kava. HerbalGram 2007, 73, 45–49. [Google Scholar]
- Teschke, R.; Lebot, V. Proposal for a Kava Quality Standardization Code. Food Chem. Toxicol. 2011, 49, 2503–2516. [Google Scholar] [CrossRef] [PubMed]
- Sarris, J.; Adams, J.; Wardle, J.L. Editorial: Time for a reassessment of the use of kava in anxiety? Complement. Ther. Med. 2009, 17, 121–122. [Google Scholar] [CrossRef] [PubMed]
- Sarris, J.; Teschke, R.; Stough, C.; Scholey, A.; Schweitzer, I. Re-introduction of kava (Piper methysticum) to the EU: Is there a way forward? Planta Med. 2011, 77, 107–110. [Google Scholar] [CrossRef] [PubMed]
- Sarris, J.; Kavanagh, D.J. Kava and St. John’s wort: Current evidence for use in mood and anxiety disorders. J. Altern. Complement. Med. 2009, 15, 827–836. [Google Scholar] [CrossRef] [PubMed]
- Sarris, J.; Kavanagh, D.J.; Byrne, G.; Bone, K.M.; Adams, J.; Deed, G. The Kava Anxiety Depression Spectrum Study (KADSS): A randomized, placebo-controlled crossover trial using an aqueous extract of Piper methysticum. Psychopharmacology 2009, 205, 399–407. [Google Scholar] [CrossRef] [PubMed]
- Sarris, J.; Adams, J.; Kavanagh, D. An explorative qualitative analysis of participants’ experience of using kava versus placebo in an RCT. Aust. J. Med. Herbalism. 2010, 22, 12–16. [Google Scholar]
- Sarris, J.; LaPorte, E.; Schweitzer, I. Kava: A comprehensive review of efficacy, safety, and psychopharmacology. Aust. N. Z. J. Psychiatry 2011, 45, 36–44. [Google Scholar] [CrossRef] [PubMed]
- Sarris, J.; Panossian, A.; Schweitzer, I.; Stough, C.; Scholey, A. Herbal medicine for depression, anxiety and insomnia: A review of psychopharmacology and clinical evidence. Eur. Neuropsychopharmacol. 2011, 21, 841–860. [Google Scholar] [CrossRef] [PubMed]
- Sarris, J.; Stough, C.; Bousman, C.A.; Wahid, T.; Murray, G.; Teschke, R.; Savage, K.M.; Dowell, A.; Ng, C.; Schweitzer, I. Kava in the treatment of generalized anxiety disorder: A double-blind, randomized, placebo-controlled study. J. Clin. Psychiatry 2013, 33, 643–648. [Google Scholar] [CrossRef] [PubMed]
- Savage, K.M.; Stough, C.K.; Byrne, G.J.; Scholey, A.; Bousman, C.; Murphy, J.; Macdonald, P.; Suo, C.; Hughes, M.; Thomas, S.; et al. Kava for the Treatment of Generalised Anxiety Disorder (K-GAD): Study protocol for a randomized controlled trial. Trials 2015, 16, 493. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Peng, J.H.; Hu, Y.Y. Chinese herbal medicine-induced liver injury. J. Clin. Transl. Hepatol. 2014, 2, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Larrey, D.; Faure, S. Herbal medicine hepatotoxicity: A new step with development of specific biomarkers. J. Hepatol. 2011, 54, 599–601. [Google Scholar] [CrossRef] [PubMed]
- Larrey, D.; Vial, T.; Pauwels, A.; Castot, A.; Biour, M.; David, M.; Michel, H. Hepatitis after germander (Teucrium chamaedrys) administration: Another instance of herbal medicine hepatotoxicity. Ann. Intern. Med. 1992, 117, 129–132. [Google Scholar] [CrossRef] [PubMed]
- Urban, T.J.; Daly, A.K.; Aithal, G.P. Genetic basis of drug-induced liver injury: Present and future. Semin. Liver Dis. 2014, 34, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Andrade, R.J.; Robles, M.; Ulzurrun, E.; Lucena, M.I. Drug-induced liver injury: Insights from genetic studies. Pharmacogenomics 2009, 10, 1467–187. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Borlak, J.; Tong, W. High lipophilicity and high daily dose of oral medications are associated with significant risk for drug-induced liver injury. Hepatology 2013, 58, 388–396. [Google Scholar] [CrossRef] [PubMed]
- Lammert, C.; Einarsson, S.; Saha, C.; Niklasson, A.; Bjornsson, E.; Chalasani, N. Relationship between daily dose of oral medications and idiosyncratic drug-induced liver injury: Search for signals. Hepatology 2008, 47, 2003–2009. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, H.J. Hepatotoxicity; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 1999. [Google Scholar]
- Teschke, R.; Frenzel, C.; Glass, X.; Schulze, J.; Eickhoff, A. Greater Celandine hepatotoxicity: A clinical review. Ann. Hepatol. 2012, 11, 838–848. [Google Scholar] [PubMed]
- Teschke, R.; Glass, X.; Schulze, J.; Eickhoff, A. Suspected Greater Celandine hepatotoxicity: Liver specific causality evaluation of published case reports from Europe. Eur. J. Gastroenterol. Hepatol. 2012, 24, 270–280. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R. Kava hepatotoxicity: A clinical review. Ann. Hepatol. 2010, 9, 251–265. [Google Scholar] [PubMed]
- Teschke, R.; Schwarzenboeck, A.; Hennermann, K.H. Kava hepatotoxicity: A clinical survey and critical analysis of 26 suspected cases. Eur. J. Gastroenterol. Hepatol. 2008, 20, 1182–1193. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Gaus, W.; Loew, D. Kava extracts: Safety and risks including rare hepatotoxicity. Phytomedicine 2003, 10, 440–446. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Schwarzenboeck, A.; Akinci, A. Kava hepatotoxicity: A European view. N. Z. Med. J. 2008, 121, 90–98. [Google Scholar] [PubMed]
- Teschke, R.; Wolff, A. Kava hepatotoxicity: Regulatory data selection and causality assessment. Dig. Liver Dis. 2009, 41, 891–901. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Schulze, J. Risk of kava hepatotoxicity and the FDA consumer advisory. J. Am. Med. Assoc. 2010, 304, 2174–2175. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R. Kava hepatotoxicity: Pathogenetic aspects and prospective considerations. Liver Int. 2010, 30, 1270–1279. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R. Special report. Kava and the risk of liver toxicity: Past, current, and future. Am. Herb. Prod. Assoc. 2011, 26, 1–17. [Google Scholar]
- Teschke, R.; Wolff, A. Regulatory causality evaluation methods applied in kava hepatotoxicity: Are they appropriate? Regul. Toxicol. Pharmacol. 2011, 59, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Olsen, L.R.; Grillo, M.P.; Skonberg, C. Constituents in kava extracts potentially involved in hepatotoxicity: A review. Chem. Res. Toxicol. 2011, 24, 992–1002. [Google Scholar] [CrossRef] [PubMed]
- Martin, A.C.; Johnston, E.; Xing, C.; Hegeman, A.D. Measuring the chemical and cytotoxic variability of commercially available kava (Piper methysticum G. Forster). PLoS ONE 2014, 9, e111572. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M. German court ruling reverses kava ban; German regulatory authority appeals decision. HerbalGram 2014, 103, 38–42. [Google Scholar]
- Kuchta, K.; Schmidt, M.; Nahrstedt, A. German kava ban lifted by court: The alleged hepatotoxicity of kava (Piper methysticum) as a case of ill-defined herbal drug identity, lacking quality control, and misguided regulatory politics. Planta Med. 2015, 81, 1647–1653. [Google Scholar] [CrossRef] [PubMed]
- Verwaltungsgericht Köln, 7 K 2197/12. Available online: http://www.justiz.nrw.de/nrwe/ovgs/vg_koeln/j2014/7_K_2197_12_Urteil_20140520.html (accessed on 15 March 2016).
- BfArM. Bundesinstitut für Arzneimittel und Medizinprodukte, Bonn. Federal Institute for Drugs and Medicinal Products in Germany. Rejection of Drug Risks, Step II. As related to: Kava-Kava (Piper methysticum)-containing, and kavain-containing drugs, including homeopathic preparations with a final concentration up to, and including D4. 14 June 2002. Available online: http://www.spc.int/cis/documents/02_0714_BfArM_Kava_Removal.pdf (accessed on 15 March 2016).
- Fu, P.P.; Xia, Q.; Lin, G.; Chou, M.W. Pyrrolizidine alkaloids—Genotoxicity, metabolism enzymes, metabolic activation, and mechanisms. Drug Metab. Rev. 2004, 36, 1–55. [Google Scholar] [CrossRef] [PubMed]
- Roulet, M.; Laurini, R.; Rivier, L.; Calame, A. Hepatic veno-occlusive disease in newborn infant of a woman drinking herbal tea. J. Pediatr. 1988, 112, 433–436. [Google Scholar] [CrossRef]
- Lee, C.H.; Wang, J.D.; Chen, P.C. Risk of liver injury associated with Chinese herbal products containing Radix bupleuri in 639,779 patients with hepatitis B virus infection. PLoS ONE 2011, 6, e16064. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, Z.; Liu, X.; Pan, R. Review on the toxic effects of radix Bupleuri. Curr. Opin. Complement. Altern. Med. 2014, 1, 3–7. [Google Scholar]
- Lei, X.; Chen, J.; Ren, J.; Li, Y.; Zhai, J.; Mu, W.; Zhang, L.; Zheng, W.; Tian, G.; Shang, H. Liver damage associated with Polygonum multiflorum Thunb.: A systematic review of case reports and case series. Evid. Based Complement. Altern. Med. 2015, 4597949. [Google Scholar] [CrossRef]
- Mazzanti, G.; di Soto, A.; Vitalone, A. Hepatotoxicity of green tea: An update. Arch. Toxicol. 2015, 89, 1175–1191. [Google Scholar] [CrossRef] [PubMed]
- Chau, T.N.; Cheung, W.I.; Ngan, T.; Lin, J.; Lee, K.W.S.; Poon, W.T.; Leung, V.K.S.; Mak, T.; Tse, M.L.; the Hong Kong Herb-Induced Liver Injury Network (HK-HILIN). Causality assessment of herb-induced liver injury using multidisciplinary approach and the Roussel Uclaf Causality assessment Method (RUCAM). Clin. Toxicol. 2011, 49, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Qin, S.; Wang, K. Biomarkers of drug-induced liver injury. Curr. Biomark. Find 2013, 3, 1–9. [Google Scholar]
- Yang, X.; Salminen, W.F.; Schnackenberg, L.K. Current and emerging biomarkers of hepatotoxicity. Curr. Biomark. Find 2012, 2, 43–55. [Google Scholar]
- Chalasani, N.; Björnsson, E. Risk factors for idiosyncratic drug-induced liver injury. Gastroenterology 2010, 138, 2246–2259. [Google Scholar] [CrossRef] [PubMed]
- Mahady, G.B.; Low Dog, T.; Barrett, M.L.; Chavez, M.L.; Gardiner, P.; Ko, R.; Marles, R.J.; Pellicore, L.S.; Giancaspro, G.I.; Sarma, D.N. United States Pharmacopeia review of the black cohosh case reports of hepatotoxicity. Menopause 2008, 15, 628–638. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Bahre, R.; Genthner, A.; Fuchs, J.; Schmidt-Taenzer, W.; Wolff, A. Suspected black cohosh hepatotoxicity—Challenges and pitfalls of causality assessment. Maturitas 2009, 63, 302–314. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R. Black cohosh and suspected hepatotoxicity—Inconsistencies, confounding variables, and prospective use of a diagnostic causality algorithm: A critical review. Menopause 2010, 17, 426–440. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Schulze, J. Suspected herbal hepatotoxicity: Requirements for appropriate causality assessment by the US Pharmacopeia. Drug Saf. 2012, 12, 1091–1097. [Google Scholar] [CrossRef]
- Teschke, R.; Schmidt-Taenzer, W.; Wolff, A. Spontaneous reports of assumed herbal hepatotoxicity by black cohosh: Is the liver unspecific Naranjo scale precise enough to ascertain causality? Pharmacoepidemiol. Drug Saf. 2011, 20, 567–582. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Schwarzenboeck, A.; Schmidt-Taenzer, W.; Wolff, A.; Hennermann, K.H. Herb induced liver injury presumably caused by black cohosh: A survey of initially purported cases and herbal quality specifications. Ann. Hepatol. 2011, 11, 249–259. [Google Scholar]
- Liss, G.; Lewis, J.H. Drug-induced liver injury: What was new in 2008? Expert Opin. Drug Metab. Toxicol. 2009, 5, 843–860. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Assessments of the Risk of Hepatotoxicity with Kava Products; WHO Document Production Services: Geneva, Switzerland, 2007. [Google Scholar]
- BfArM, Bundesinstitut für Arzneimittel und Medizinprodukte (German regulatory agency). Bekanntmachung. Abwehr von Gefahren durch Arzneimittel, Stufe II, Anhörung: Schöllkraut-haltige Arzneimittel zur innerlichen Anwendung. 6 May 2005. Available online: http://www.bfarm.de/cae/servlet/contentblob/1014620/publicationFile/66198/schoellkraut-anhoerung_050505.pdf (accessed on 15 March 2016).
- Schmidt, M.; Morgan, M.; Bone, K.; McMillan, J. Kava: A risk-benefit assessment. In The Essential Guide to Herbal Safety; Mills, M., Bone, K., Eds.; Elsevier Churchill Livingstone: St. Louis, MS, USA, 2005; pp. 155–221. [Google Scholar]
- DCGMA, Drug Commission of the German Medical Association (Arzneimittelkommission der Deutschen Ärzteschaft). Hepatitis in connection with Umckaloabo®. Deutsches Ärzteblatt 2011, 108, C1399–C1400. (In German) [Google Scholar]
- Teschke, R.; Frenzel, C.; Schulze, J.; Eickhoff, A. Spontaneous reports of primarily suspected herbal hepatotoxicity by Pelargonium sidoides: Was causality adequately ascertained? Regul. Toxicol. Pharmacol. 2012, 63, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Frenzel, C.; Schulze, J.; Eickhoff, A. Suspected herbal hepatotoxicity: The pharmacovigilance dilemma with disputed and obsolete evaluation methods. Regul. Toxicol. Pharmacol. 2012, 64, 343–344. [Google Scholar] [CrossRef]
- Teschke, R.; Frenzel, C.; Wolff, A.; Herzog, J.; Glass, X.; Schulze, J.; Eickhoff, A. Initially purported hepatotoxicity by Pelargonium sidoides: The dilemma of pharmacovigilance and proposals for improvements. Ann. Hepatol. 2012, 11, 500–512. [Google Scholar] [PubMed]
- Teschke, R.; Frenzel, C.; Schulze, J.; Schwarzenboeck, A.; Eickhoff, A. Herbalife hepatotoxicity: Evaluation of cases with positive reexposure tests. World J. Hepatol. 2013, 5, 353–363. [Google Scholar] [CrossRef] [PubMed]
- Melchart, D.; Linde, K.; Weidenhammer, W.; Hager, S.; Shaw, D.; Bauer, R. Liver enzyme elevations in patients treated with traditional Chinese medicine. JAMA 1999, 282, 28–29. [Google Scholar] [CrossRef] [PubMed]
- Au, J.S.; Navarro, V.J.; Rossi, S. Review article: Drug induced liver injury—Its pathophysiology and evolving diagnostic tools. Aliment. Pharmacol. Ther. 2011, 34, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Dalton, H.R.; Fellows, H.J.; Stableforth, W.; Joseph, M.; Thurairajah, P.H.; Warshow, U.; Hazeldine, S.; Remnarace, R.; Ijaz, S.; Hussaini, S.H.; et al. The role of hepatitis E virus testing in drug-induced liver injury. Aliment. Pharmacol. Ther. 2007, 26, 1429–1435. [Google Scholar] [CrossRef] [PubMed]
- Davern, T.J.; Chalasani, N.; Fontana, R.J.; Hayashi, P.H.; Protiva, P.; Kleiner, D.E.; Engle, R.E.; Nguyen, H.; Emerson, S.U.; Purcell, R.H.; et al. Acute hepatitis E infection accounts for some cases of suspected drug-induced liver injury. Gastroenterology 2011, 141, 1665–1672. [Google Scholar] [CrossRef] [PubMed]
- Hoofnagle, J.H.; Nelson, K.E.; Purcell, R.H. Review article: Hepatitis E. N. Engl. J. Med. 2012, 367, 1237–1244. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Andrade, R. Editorial. Drug-induced liver injury: Expanding our knowledge by enlarging population analysis with prospective and scoring causality assessment. Gastroenterology 2015, 148, 1271–1273. [Google Scholar] [CrossRef] [PubMed]
- Chen, E.Y.; Baum, K.; Collins, W.; Löve, A.; Merz, M.; Olafsson, S.; Björnsson, E.S.; Lee, W.M. Hepatitis E masquerading as drug–induced liver injury. Hepatology 2012, 56, 2420–2423. [Google Scholar] [CrossRef] [PubMed]
- Bénichou, C.; Danan, G.; Flahault, A. Causality assessment of adverse reactions to drugs—II. An original model for validation of drug causality assessment methods: Case reports with positive rechallenge. J. Clin. Epidemiol. 1993, 46, 1331–1336. [Google Scholar] [CrossRef]
- Teschke, R.; Zhang, L.; Long, H.; Schwarzenboeck, A.; Schmidt-Taenzer, W.; Genthner, A.; Wolff, A.; Frenzel, C.; Schulze, J.; Eickhoff, A. Traditional Chinese Medicine and herbal hepatotoxicity: A tabular compilation of reported cases. Ann. Hepatol. 2015, 14, 7–19. [Google Scholar] [PubMed]
- Teschke, R.; Genthner, A.; Wolff, A.; Frenzel, C.; Schulze, J.; Eickhoff, A. Herbal hepatotoxicity: Analysis of cases with initially reported positive reexposure tests. Dig. Liver Dis. 2014, 46, 264–269. [Google Scholar] [CrossRef] [PubMed]
- National Institutes of Health (NIH) and LiverTox: Drug record. Ba Jiao Lian (Dysosma pleianthum). Last updated 19 May 2014. Available online: http://livertox.nih.gov/BaJiaoLian.htm (accessed on 15 March 2016).
- Naser, B.; Schnitker, J.; Minkin, M.J.; de Arriba, S.G.; Nolte, K.U.; Osmers, R. Hepatotoxicity suspected by black cohosh: No evidence by metaanalysis of randomized controlled clinical trials for isopropanolic black cohosh extract. Menopause 2011, 18, 366–375. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Schulze, J.; Schwarzenboeck, A.; Eickhoff, A.; Frenzel, C. Herbal hepatotoxicity: Suspected cases assessed for alternative causes. Eur. J. Gastroenterol. Hepatol. 2013, 25, 1093–1098. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Frenzel, C.; Wolff, A.; Eickhoff, A.; Schulze, J. Drug induced liver injury: Accuracy of diagnosis in published reports. Ann. Hepatol. 2014, 13, 248–255. [Google Scholar] [PubMed]
- Navarro, V.J.; Barnhart, H.; Bonkovsky, H.L.; Davern, T.; Fontana, R.J.; Grant, L.; Reddy, K.R.; Seeff, L.B.; Serrano, J.; Averell, H.; et al. Liver injury from herbals and dietary supplements in the U.S. Drug-Induced Liver Injury Network. Hepatology 2014, 60, 1399–1408. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Glass, X.; Schulze, J. Herbal hepatotoxicity by Greater Celandine (Chelidonium majus): Causality assessment of 22 spontaneous reports. Regul. Toxicol. Pharmacol. 2011, 61, 282–291. [Google Scholar] [CrossRef] [PubMed]
- Haller, C.A.; Dyer, J.E.; Ko, R.; Olson, K.R. Making a diagnosis of herbal-related toxic hepatitis. West J. Med. 2002, 176, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Estes, J.D.; Stolpman, D.; Olyaei, A.; Corless, C.L.; Ham, J.M.; Schwartz, J.M.; Orloff, S. High prevalence of potentially hepatotoxic herbal supplement use in patients with fulminant hepatic failure. Arch. Surg. 2003, 138, 852–858. [Google Scholar] [CrossRef] [PubMed]
- Talari, P.; Talari, G.; Gundareddy, V.; Vemula, P.; Barmecha, J. Acute Hepatitis Associated with the Chinese Herbal Product bai shi wan. Available online: http://www.toothpicks.info/rc2/posters/2010/Internal_Medicine/Talari_P_Acute_hepatitis_associated.pdf (accessed on 15 March 2016).
- Perharic-Walton, L.; Murray, V. Toxicity of Chinese herbal remedies. Lancet 1992, 340, 674. [Google Scholar] [PubMed]
- Kane, J.A.; Kane, S.P.; Jain, S. Hepatitis induced by traditional Chinese herbs: Possible toxic components. Gut 1995, 36, 146–147. [Google Scholar] [CrossRef] [PubMed]
- Vautier, G.; Spiller, R.C. Safety of complementary medicines should be monitored. BMJ 1995, 311, 633. [Google Scholar] [CrossRef] [PubMed]
- Yuen, M.F.; Tam, S.; Fung, J.; Wong, D.K.H.; Wong, B.C.Y.; Lai, C.L. Traditional Chinese Medicine causing hepatotoxicity in patients with chronic hepatitis B infection: A 1-year prospective study. Aliment. Pharmacol. Ther. 2006, 24, 1179–1186. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.H.; Kim, J.I.; Jeong, K.H.; Ko, K.H.; Ko, P.G.; Hwang, S.W.; Kim, E.M.; Kim, S.H.; Lee, H.Y.; Lee, B.S. Clinical characteristics of 159 cases of acute toxic hepatitis. Korean J. Hepatol. 2008, 14, 483–492, (abstract in English, article in Korean). [Google Scholar] [CrossRef] [PubMed]
- Sohn, C.H.; Cha, M.I.; Oh, B.J.; Yeo, W.H.; Lee, J.H.; Kim, W.; Lim, K.S. Liver transplantation for acute toxic hepatitis due to herbal medicines and preparations. J. Korean Soc. Clin. Toxicol. 2008, 6, 110–116, (abstract in English, article in Korean). [Google Scholar]
- Cortez, E.; Boulger, C.; Bernard, A. Ban Tu Wan hepatotoxicity. BMJ Case Rep. 2012. [Google Scholar] [CrossRef] [PubMed]
- Sangsuwan, C.; Udompanthurak, S.; Vannasaeng, S.; Thamlikitkul, V. Randomized controlled trial of Tinospora crispa for additional therapy in patients with type 2 diabetes mellitus. J. Med. Assoc. Thai. 2004, 87, 543–546, (abstract in English, article in Thai). [Google Scholar] [PubMed]
- Motoyama, H.; Enomoto, M.; Yasuda, T.; Fujii, H.; Kobayashi, S.; Iwai, S.; Morikawa, H.; Takeda, T.; Tamori, A.; Sakaguchi, H.; et al. Drug-induced liver injury caused by a herbal medicine, bofu-tsu-sho-san. Nihon Shokakibyo Gakkai Zasshi 2008, 105, 1234–1239, (abstract in English, article in Japanese). [Google Scholar] [PubMed]
- Hwang, S.H.; Park, J.A.; Jang, Y.S.; Lee, K.M.; Lee, D.S.; Ahn, B.M.; Lee, E.H. Case of acute cholestatic hepatitis caused by the seeds of Psoralea-corylifolia. Korean J. Hepatol. 2001, 7, 341–344, (abstract in English, article in Korean). [Google Scholar]
- Nam, S.W.; Baek, J.T.; Lee, D.S.; Kang, S.B.; Ahn, B.M.; Chung, K.W. A case of acute cholestatic hepatitis associated with the seeds of Psoralea corylifolia (Boh-Gol-Zhee). Clin. Toxicol. 2005, 43, 589–591. [Google Scholar] [CrossRef]
- Cheung, W.I.; Tse, M.L.; Ngan, T.; Lin, J.; Lee, W.K.; Poon, W.T.; Mak, T.W.; Leung, V.K.S.; Chau, T.N. Liver injury associated with the use of Fructus Psoraleae (Bol-gol-zhee or Bu-gu-zhi) and its related propriety medicine. Clin. Toxicol. 2009, 47, 683–685. [Google Scholar] [CrossRef] [PubMed]
- Chau, T.N. Drug-induced liver injury: An update. Hong Kong Med. Diary 2008, 13, 23–26. [Google Scholar]
- Lin, T.J.; Tsai, M.S.; Chiou, N.M.; Deng, J.F.; Chiu, N.Y. Hepatotoxicity caused by Breynia officinalis. Vet. Hum. Toxicol. 2002, 44, 87–88. [Google Scholar] [PubMed]
- Lin, T.J.; Su, C.C.; Lan, C.K.; Jiang, D.D.; Tsai, J.L.; Tsai, M.S. Acute poisonings with Breynia officinalis—An outbreak of hepatotoxicity. J. Toxicol. Clin. Toxicol. 2003, 41, 591–594. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, E.M.; McLean, C.A.; Cheng, E.S.; Blanc, P.D.; Somberg, K.A.; Ferrell, L.D.; Lake, J.R. Chinese herbal medicine, fulminant hepatitis, and liver transplantation. Am. J. Gastroenterol. 1996, 91, 2647–2648. [Google Scholar] [PubMed]
- Kamiyama, T.; Nouchi, T.; Kojima, S.; Murata, N.; Ikeda, T.; Sato, C. Autoimmune hepatitis triggered by administration of an herbal medicine. Am. J. Gastroenterol. 1997, 92, 703–704. [Google Scholar] [PubMed]
- Björnsson, E.S.; Bergmann, O.M.; Björnsson, H.K.; Kvaran, R.B.; Olafsson, S. Incidence, presentation and outcomes in patients with drug-induced liver injury in the general population of Iceland. Gastroenterology 2013, 144, 1419–1425. [Google Scholar] [CrossRef] [PubMed]
- Verucchi, G.; Calza, L.; Attard, L.; Chiodo, F. Acute hepatitis induced by traditional Chinese herbs used in the treatment of psoriasis. J. Gastroenterol. Hepatol. 2002, 17, 1342–1343. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Yim, H.J.; Ahn, J.H.; Kim, J.H.; Kim, J.N.; Yoon, I.; Kim, D.I.; Lee, H.S.; Lee, S.W.; Choi, J.H. Two cases of toxic hepatitis caused by arrowroot juice. Korean J. Hepatol. 2009, 15, 504–509, (abstract in English, article in Korean). [Google Scholar] [CrossRef] [PubMed]
- Bae, S.H.; Kim, D.H.; Bae, Y.S.; Lee, K.J.; Kim, D.W.; Yoon, J.B.; Hong, J.H.; Kim, S.H. Toxic hepatitis associated with Polygoni multiflori. Korean J. Hepatol. 2010, 16, 182–186, (abstract in English, article in Korean). [Google Scholar] [CrossRef] [PubMed]
- Gono, Y.; Odaguchi, H.; Hayasaki, T.; Suzuki, K.; Oikawa, T.; Muranushi, A.; Akahoshi, T.; Hanawa, T. Clinical analysis of cases with drug-induced liver injury for Kampo medicine. Kampo Med. 2010, 61, 828–833, (abstract in English, article in Japanese). [Google Scholar] [CrossRef]
- Linnebur, S.A.; Rapacchietta, O.C.; Vejar, M. Hepatotoxicity associated with Chinese skullcap contained in Move Free Advanced dietary supplement: Two case reports and review of the literature. Pharmacotherapy 2010, 750, 258e–262e. [Google Scholar] [CrossRef]
- Yang, L.; Aronsohn, A.; Hart, J.; Jensen, D. Herbal hepatotoxicity from Chinese skullcap: A case report. World J. Hepatol. 2012, 4, 231–233. [Google Scholar] [CrossRef] [PubMed]
- Dhanasekaran, R.; Owens, V.; Sanchez, W. Chinese skullcap in Move Free arthritis supplement causes drug induced liver injury and pulmonary infiltrates. Case Rep. Hepatol. 2013, 965092. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.S.; Choi, H.S.; Yun, T.J.; Lee, K.G.; Seo, Y.S.; Yeon, J.E.; Byun, K.S.; Um, S.H.; Kim, C.D.; Ryu, H.S. A case of acute cholestatic hepatitis induced by Corydalis speciosa Max. Korean J. Hepatol. 2009, 15, 517–523. (In Korean) [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Ryu, S.L.; Shim, J.W.; Kim, D.S.; Shim, J.Y.; Park, M.S.; Jung, H.L. A pediatric case of toxic hepatitis induced by Hovenia dulcis. Pediatr. Gastroenterol. Hepatol. Nutr. 2012, 15, 111–116. [Google Scholar] [CrossRef]
- Woolf, G.M.; Petrovic, L.M.; Rojter, S.E.; Wainwright, S.; Villamil, F.G.; Katkov, W.N.; Michieletti, P.; Wanless, I.R.; Stermitz, F.R.; Beck, J.J.; et al. Acute hepatitis associated with the Chinese herbal product Jin Bu Huan. Ann. Intern. Med. 1994, 121, 729–735. [Google Scholar] [CrossRef] [PubMed]
- Horowitz, R.S.; Feldhaus, K.; Dart, R.C.; Stermitz, F.R.; Beck, J.J. The clinical spectrum of Jin Bu Huan toxicity. Arch Intern. Med. 1996, 156, 899–903. [Google Scholar] [CrossRef] [PubMed]
- Picciotti, A.; Campo, N.; Brizzolara, R.; Giusto, R.; Guido, G.; Sinelli, N.; Lapertosa, G.; Celle, G. Chronic hepatitis induced by Jin Bu Huan. J. Hepatol. 1998, 28, 165–167. [Google Scholar] [CrossRef]
- Divinsky, M. Case report: Jin Bu Huan—Not so benign herbal medicine. Can Fam. Phys. 2002, 48, 1640–1642. [Google Scholar]
- Inoue, H.; Yamazaki, S.; Shimizu, M.; Uozki, H.; Goto, T.; Ohnishi, S.; Koike, K. Liver injury induced by the Japanese herbal drug kamishoyosan. Gastroenterol. Hepatol. 2011, 7, 692–695. [Google Scholar]
- Garcia-Moran, S.; Saez-Royuela, F.; Gento, E.; Lopez Morante, A.; Arias, L. Acute hepatitis associated with Camellia tea and Orthosiphon stamineus ingestion. Gastroenterol. Hepatol. 2004, 27, 559–560. [Google Scholar] [CrossRef]
- Peyrin-Biroulet, L.; Petitpain, N.; Kalt, P.; Ancel, D.; Petit-Laurent, F.; Trechot, P.; Barraud, H.; Bronowicki, J.P. Probable hepatotoxicity from epigallocatecol gallate used for Phytotherapy. Gastroenterol. Clin. Biol. 2004, 28, 404–406. (In French) [Google Scholar] [CrossRef]
- Gloro, R.; Hourmand-Ollivier, I.; Mosquet, B.; Mosquet, L.; Rousselot, P.; Salamé, E.; Piquet, M.A.; Dao, T. Fulminant hepatitis during self-medication with hydroalcoholic extract of green tea. Eur. J. Gastroenterol. Hepatol. 2005, 17, 1135–1137. [Google Scholar] [CrossRef] [PubMed]
- Javaid, A.; Bonkovsky, H.L. Hepatotoxicity due to extracts of Chinese green tea (Camellia sinensis): A growing concern. J. Hepatol. 2006, 45, 334–335. [Google Scholar] [CrossRef] [PubMed]
- Jimenez-Saenz, M.; Martinez-Sanchez M del, C. Acute hepatitis associated with the use of green tea infusions. J. Hepatol. 2006, 44, 616–617. [Google Scholar] [CrossRef] [PubMed]
- Bonkovsky, H.L. Hepatotoxicity associated with supplements containing Chinese green tea (Camellia sinensis). Ann. Intern. Med. 2006, 144, 68–71. [Google Scholar] [CrossRef] [PubMed]
- Molinari, M.; Watt, K.D.; Kruszyna, T.; Nelson, R.; Walsh, M.; Huang, W.Y.; Nashan, B.; Peltekian, K. Acute liver failure induced by green tea extracts: Case reports and review of the literature. Liver Transpl. 2006, 12, 1892–1895. [Google Scholar] [CrossRef] [PubMed]
- Björnsson, E.; Olsson, R. Serious adverse liver reactions associated with herbal weight loss supplements. J. Hepatol. 2007, 47, 295–297. [Google Scholar] [CrossRef] [PubMed]
- García-Cortés, M.; Borraz, Y.; Lucena, M.I.; Peláez, G.; Salmerón, J.; Diago, M.; Martínez-Sierra, M.C.; Navarro, J.M.; Planas, R.; Soria, M.J.; et al. Liver injury induced by “natural remedies”: An analysis of cases submitted to the Spanish Liver Toxicity Registry. Rev. Esp. Enferm. Dig. 2008, 100, 688–695. [Google Scholar] [PubMed]
- Rohde, J.; Jacobsen, C.; Kromann-Andersen, H. Toxic hepatitis triggered by green tea. Ugeskr Laeger 2011, 173, 205–206, (abstract in English, article in Danish). [Google Scholar] [PubMed]
- Nadir, A.; Agrawal, S.; King, P.D.; Marshall, J.B. Acute hepatitis associated with the use of a Chinese herbal product, ma-huang. Am. J. Gastroenterol. 1996, 91, 1436–1438. [Google Scholar] [PubMed]
- Borum, M.L. Fulminant exacerbation of autoimmune hepatitis after the use of Ma Huang. Am. J. Gastroenterol. 2001, 96, 1654–1655. [Google Scholar] [CrossRef] [PubMed]
- Skoulidis, F.; Alexander, G.J.; Davies, S.E. Ma huang associated acute liver failure requiring liver transplantation. Eur. J. Gastroenterol. Hepatol. 2005, 17, 581–584. [Google Scholar] [CrossRef] [PubMed]
- Reuben, A.; Koch, D.G.; Lee, W.M.; the Acute Liver Failure Study Group. Drug-induced acute liver failure: Results of a U.S. multicenter, prospective study. Hepatology 2010, 52, 2065–2076. [Google Scholar] [CrossRef] [PubMed]
- Aiba, T.; Takahashi, T.; Suzuki, K.; Okoshi, S.; Nomoto, M.; Uno, K.; Aoyagi, Y. Liver injury induced by a Japanese herbal medicine, sairei-to (TJ-114, Bupleurum and Hoelen combination, Chai-Ling-Tang). J. Gastroenterol. Hepatol. 2007, 22, 762–763. [Google Scholar] [CrossRef] [PubMed]
- Tsuda, T.; Yashiro, S.; Gamo, Y.; Watanabe, K.; Hoshino, T.; Oikawa, T.; Hanawa, T. Discrepancy between clinical course and drug-induced lymphocyte stimulation tests in a case of saireito-induced liver injury accompanied by Sjögren syndrome. J. Altern. Complement. Med. 2010, 16, 501–505. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.Y.; Cai, J.T.; Du, Q. Hepatic veno-occlusive disease associated with the use of Gynura segetum. Eur. J. Intern. Med. 2007, 18, 609. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Liang, X.S.; Li, C.Z. Sinusoidal obstruction syndrome associated with the ingestion of Gynura root. Clin. Toxicol. 2010, 48, 962–964. [Google Scholar] [CrossRef] [PubMed]
- Cárdenas, A.; Restrepo, J.C.; Sierra, F.; Correa, G. Acute hepatitis due to shen-min: A herbal product derived from Polygonum multiflorum. J. Clin. Gastroenterol. 2006, 40, 629–632. [Google Scholar] [CrossRef] [PubMed]
- But, P.P.H.; Tomlinson, B.; Lee, K.L. Hepatitis related to the Chinese medicine Shou-wu-pian manufactured from Polygonum multiflorum. Vet. Hum. Toxicol. 1996, 38, 280–282. [Google Scholar] [PubMed]
- Park, G.J.H.; Mann, S.P.; Ngu, M.C. Acute hepatitis induced by Shou-Wu-Pian, a herbal product derived from Polygonum multiflorum. J. Gastroenterol. Hepatol. 2001, 16, 115–117. [Google Scholar] [CrossRef] [PubMed]
- Battinelli, L.; Daniele, C.; Mazzanti, G.; Mastroianni, C.M.; Lichtner, M.; Coletta, S.; Costantini, S. New case of acute hepatitis following the consumption of Shou Wu Pian, a Chinese herbal product derived from Polygonum multiflorum. Ann. Intern. Med. 2004, 140, 587–588. [Google Scholar]
- Panis, B.; Wong, D.R.; Hooymans, P.M.; De Smet, P.A.G.M.; Rosias, P.R. Recurrent toxic hepatitis in a Caucasian girl related to the use of Shou-Wu-Pian, a Chinese herbal preparation. J. Pediat. Gastroenterol. Nutr. 2005, 41, 256–258. [Google Scholar] [CrossRef]
- Laird, A.R.; Ramchandani, N.; deGoma, E.M.; Avula, B.; Khan, I.A.; Gesundheit, N. Acute hepatitis associated with the use of an herbal supplement (Polygonum multiflorum) mimicking iron-overload syndrome. Clin. Gastroenterol. 2008, 42, 861–862. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, M.; Kasajima, S.; Nakamura, Y.; Shouzushima, M.; Nagatani, N.; Takinishi, A.; Taguchi, A.; Fujita, M.; Niimi, A.; Misaka, R.; et al. Toxic hepatitis induced by Show-Wu-Pian, a Chinese herbal preparation. Intern. Med. 2010, 49, 1537–1540. [Google Scholar] [CrossRef] [PubMed]
- Valente, G.; Sanges, M.; Campione, S.; Bellevicine, C.; de Franchis, G.; Sollazzo, R.; Matera, D.; Cimino, L.; Vecchione, R.; D’Arienzo, A. Herbal hepatotoxicity: A case of difficult interpretation. Eur. Rev. Med. Pharmacol. Sci. 2010, 14, 865–870. [Google Scholar] [CrossRef]
- Jung, K.A.; Min, H.J.; Yoo, S.S.; Kim, H.J.; Choi, S.N.; Ha, C.Y.; Kim, H.J.; Kim, T.H.; Jung, W.T.; Lee, O.J.; et al. Drug-induced liver injury: Twenty five cases of acute hepatitis following ingestion of Polygonum multiflorum Thun. Gut Liver 2011, 5, 493–499. [Google Scholar] [CrossRef] [PubMed]
- Banarova, A.; Koller, T.; Payer, J. Toxic hepatitis induced by Polygonum multiflorum. Vnitr Lek 2012, 58, 958–962, (abstract in English, article in Slovak). [Google Scholar] [PubMed]
- Cohen, S.M.; Heywood, E.; Pillai, A.; Ahn, J. Hepatotoxicity associated with the use of White Flood, a nutritional supplement. Pract. Gastroenterol. 2012, 10, 45–48. [Google Scholar]
- Itoh, S.; Marutani, K.; Nishijima, T.; Matsuo, S.; Itabashi, M. Liver injuries induced by herbal medicine, Syo-saiko-to (xiao-chai-hu-tang). Dig. Dis. Sci. 1995, 40, 1845–1848. [Google Scholar] [CrossRef] [PubMed]
- Hsu, L.M.; Huang, Y.S.; Tsay, S.H.; Chang, F.Y.; Lee, S.D. Acute hepatitis induced by Chinese hepatoprotective herb xiao-chai-hu-tang. J. Chin. Med. Assoc. 2006, 69, 86–88. [Google Scholar] [CrossRef]
- Rabe, C.; Musch, A.; Schirrmacher, P.; Kruis, W.; Hoffmann, R. Acute hepatitis induced by an aloe vera preparation: A case report. World J. Gastroenterol. 2005, 11, 303–304. [Google Scholar] [CrossRef] [PubMed]
- Kanat, O.; Ozet, A.; Ataegin, S. Aloe vera-induced acute toxic hepatitis in a healthy young man. Eur. J. Intern. Med. 2006, 17, 589. [Google Scholar] [CrossRef] [PubMed]
- Bottenberg, M.M.; Wall, G.C.; Harvey, R.L.; Habib, S. Oral Aloe vera-induced hepatitis. Ann. Pharmacother. 2007, 41, 1740–1743. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.N.; Kim, D.J.; Kim, Y.M.; Kim, B.H.; Sohn, K.M.; Choi, M.J.; Choi, Y.H. Aloe-induced toxic hepatitis. J. Korean Med. Sci. 2010, 25, 492–495. [Google Scholar] [CrossRef] [PubMed]
- Smith, L.W.; Culvenor, C.C.J. Plant sources of hepatotoxic pyrrolizidine alkaloids. J. Nat. Prod. 1981, 44, 129–152. [Google Scholar] [CrossRef] [PubMed]
- Nadir, A.; Reddy, D.; van Thiel, D.H. Cascara sagrada-induced intrahepatic cholestasis causing portal hypertension: Case report and review of herbal hepatotoxicity. Am. J. Gastroenterol. 2000, 95, 3634–3637. [Google Scholar] [CrossRef] [PubMed]
- Katz, M.; Saibil, F. Herbal hepatitis: Subacute hepatic necrosis secondary to chaparral leaf. J. Clin. Gastroenterol. 1990, 12, 203–206. [Google Scholar] [CrossRef] [PubMed]
- Centers of Disease Control and Prevention. Chaparral-induced toxic hepatitis California and Texas 1992. J. Am. Med. Ass. 1992, 268, 3295–3298. [Google Scholar]
- Smith, B.C.; Desmond, P.V. Acute hepatitis induced by the ingestion of the herbal medication chaparral. Aust. N. Z. J. Med. 1993, 23, 526. [Google Scholar] [CrossRef] [PubMed]
- Alderman, S.; Goldfarb, S.; Malone, D.G. Cholestatic hepatitis after ingestion of chaparral leaf: Confirmation by endoscopic retrograde cholangio-pancreatography and liver biopsy. J. Clin. Gastroenterol. 1994, 19, 242–247. [Google Scholar] [CrossRef] [PubMed]
- Batchelor, W.B.; Heathcote, J.; Wanless, I.R. Chaparral-induced hepatic injury. Am. J. Gastroenterol. 1995, 90, 831–833. [Google Scholar] [PubMed]
- Gordon, D.W.; Rosenthal, G.; Hart, J.; Sirota, S.; Baker, A.L. Chaparral ingestion: The broadening spectrum of liver injury caused by herbal medications. J. Am. Med. Assoc. 1995, 273, 489–490. [Google Scholar] [CrossRef]
- Sheikh, N.M.; Philen, R.M.; Love, L.A. Chaparral-associated hepatotoxicity. Arch. Intern. Med. 1997, 157, 913–919. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M.; Ohkuma, S.; McDermott, W.V.; Trey, C.; Huxtable, R.J. Hepatic venoocclusive disease associated with the consumption of pyrrolizidine-containing dietary supplements. Gastroenterology 1985, 88, 1050–1054. [Google Scholar] [PubMed]
- Weston, C.F.M.; Cooper, B.T.; Davies, J.D.; Levine, D.F. Veno-occlusive disease of the liver secondary to ingestion of comfrey. Br. Med. J. 1987, 295, 183. [Google Scholar] [CrossRef]
- Bach, N.; Thung, S.N.; Schaffner, F. Comfrey herb tea-induced hepatic veno-occlusive disease. Am. J. Med. 1989, 87, 97–99. [Google Scholar] [CrossRef]
- Ridker, P.M.; McDermott, W.V. Comfrey herbs tea and hepatic veno-occlusive disease. Lancet 1989, 333, 657–658. [Google Scholar] [CrossRef]
- Miskelly, F.G.; Goodyer, L.I. Hepatic and pulmonary complications of herbal medicine (letter). Postgrad Med. J. 1992, 68, 935. [Google Scholar] [CrossRef] [PubMed]
- Georgia, M. Hepatotoxicity due to Atractylis gummifera. Clin. Toxicol. 1988, 26, 487–493. [Google Scholar] [CrossRef]
- McDonnell, W.M.; Bhattacharya, R.; Halldorson, J.B. Fulminant hepatic failure after use of the herbal weight-loss supplement Exilis. Ann. Intern. Med. 2009, 151, 673–674. [Google Scholar] [CrossRef] [PubMed]
- Mostefa-Kara, N.; Pauwels, A.; Pines, E.; Biour, M.; Levy, V.G. Fatal hepatitis after herbal tea (letter). Lancet 1992, 340, 674. [Google Scholar] [CrossRef]
- Dao, T.; Peytier, A.; Galateau, F.; Valla, A. Chronic hepatitis due to germander. Gastroenterol. Clin. Biol. 1993, 17, 614–615. [Google Scholar]
- Mattéi, A.; Rucay, P.; Samuel, D.; Feray, C.; Michel, R.; Bismuth, H. Liver transplantation for acute liver failure after herbal medicine (Teucrium polium) administration (letter). J. Hepatol. 1995, 22, 597. [Google Scholar] [CrossRef]
- Laliberté, L.; Villeneuve, J.P. Hepatitis after the use of germander, a herbal remedy. Can. Med. Assoc. J. 1996, 154, 1689–1692. [Google Scholar]
- Starakis, I.; Siagris, D.; Leonidou, L.; Mazakopakis, E.; Tsamandas, A.; Karatza, C. Hepatitis caused by the herbal remedyTeucrium polium L. Eur. J. Gastroenterol. Hepatol. 2006, 18, 681–683. [Google Scholar] [CrossRef] [PubMed]
- Strahl, S.; Ehret, V.; Dahm, H.H.; Maier, K.P. Necrotising hepatitis after taking herbal remedies. Dtsch Med. Wschr. 1998, 123, 1410–1414. (In German) [Google Scholar] [CrossRef] [PubMed]
- Greving, I.; Meister, V.; Monnerjahn, C.; Müller, K.M.; May, B. Chelidonium majus: A rare reason for severe hepatotoxic reaction. Pharmacoepidemiol. Drug Saf. 1998, 7, S66–S69. [Google Scholar] [CrossRef]
- Benninger, J.; Schneider, H.T.; Schuppan, D.; Kirchner, T.; Hahn, E.G. Acute hepatitis induced by Greater Celandine (Chelidonium majus). Gastroenterology 1999, 117, 1234–1237. [Google Scholar] [CrossRef]
- Crijns, A.P.; de Smet, P.A.; van den Heuvel, M.; Schot, B.W.; Haagsma, E.B. Acute hepatitis after use of herbal preparation with greater celandine (Chelidonium majus). Ned. Tijdschr. Geneeskd 2002, 146, 124–128. (In Dutch) [Google Scholar] [PubMed]
- Hardeman, E.; van Overbeke, L.; Ilegems, S.; Ferrante, M. Acute hepatitis induced by greater celandine (Chelidonium majus). Acta Gastroenterol. Belg. 2003, 71, 281–282. [Google Scholar]
- Stickel, F.; Pöschl, G.; Seitz, K.H.; Waldherr, R.; Hahn, E.G.; Schuppan, D. Acute hepatitis induced by Greater Celandine (Chelidonium majus). Scand. J. Gastroenterol. 2003, 38, 565–568. [Google Scholar] [CrossRef] [PubMed]
- Rifai, K.; Flemming, P.; Manns, M.P.; Trautwein, C. Severe drug hepatitis caused by Chelidonium majus. Internist 2006, 47, 749–751. (In German) [Google Scholar] [CrossRef] [PubMed]
- Conti, E.; De Checchi, G.; Mencarelli, R.; Pinato, S.; Rovere, P. Lycopodium similiaplex-induced acute hepatitis: A case report. Eur. J. Gastroenterol. Hepatol. 2008, 20, 469–471. [Google Scholar] [CrossRef] [PubMed]
- Moro, P.A.; Cassetti, F.; Giugliano, G.; Falce, M.T.; Mazzanti, G.; Menniti-Ippolito, F.; Raschetti, R.; Santuccio, C. Hepatitis from Greater celandine (Chelidonium majus L.): Review of literature and report of a new case. J. Ethnopharmacol. 2009, 124, 328–332. [Google Scholar] [CrossRef] [PubMed]
- Tarantino, G.; Di Minno, M.N.D.; Pezullo, M.G.; Pezzullo, L.S.; Milone, F.; Milone, M.; Capone, D. Drug-induced liver injury due to “natural products” used for weight loss: A case report. World J. Gastroenterol. 2009, 15, 2414–2417. [Google Scholar] [CrossRef] [PubMed]
- European Medicines Agency (EMA). Assessment report on Chelidonium majus L., herba. Draft. 25 November 2010. Available online: http://www.ema.europa.eu/docs/en_GB/document_library/Public_statement/2011/01/WC500100940.pdf (accessed on 15 March 2016).
- Duenas Sadornil, C.; Fabregas Piugtio, S.; Durandez, R. Hepatotoxicity due to Camelia sinensis. Med. Clin. 2004, 122, 677–678. (In Spanish) [Google Scholar]
- Abu el Wafa, Y.; Benaventa Fernandez, A.; Talavera Fabuel, A.; Perez Ramos, M.A.; Ramos-Clemente, J.I. Acute hepatitis induced by Camellia sinensis (green tea). An. Med. Interna 2005, 22, 298. (In Spanish) [Google Scholar] [PubMed]
- Martinez-Sierra, C.; Rendon Unceta, P.; Martin Herrera, L. Acute hepatitis after green tea ingestion. Med. Clin. 2006, 127, 119. [Google Scholar] [CrossRef]
- Federico, A.; Tiso, A.; Loguercio, C. A case of hepatotoxicity caused by green tea. Free Radic. Biol. Med. 2007, 43, 474. [Google Scholar] [CrossRef] [PubMed]
- Verheist, X.; Burvenich, P.; Van Sassenbroeck, D.; Gabiel, C.; Lootens, M.; Baert, D. Acute hepatitis after treatment for hair loss with oral green tea extracts (Camellia sinensis). Acta Gastroenterol. Belg. 2009, 72, 262–264. [Google Scholar]
- Stillman, A.S.; Huxtable, R.; Consroe, P.; Kohnen, P.; Smith, S. Hepatic veno-occlusive disease due to pyrrolizidine (Senecio) poisoning in Arizona. Gastroenterology 1977, 73, 349–352. [Google Scholar] [PubMed]
- Fox, D.W.; Hart, M.C.; Bergeson, P.S.; Jarrett, P.B.; Stillman, A.E.; Huxtable, R.J. Pyrrolizidine (Senecio) intoxication mimicking Reye syndrome. J. Pediatr. 1978, 93, 980–982. [Google Scholar] [CrossRef]
- Dara, L.; Hewett, J.; Lim, J.K. Hydroxycut hepatotoxicity: A case series and review of liver toxicity from herbal weight loss supplements. World J. Gastroenterol. 2008, 14, 6999–7004. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, M.; Marbet, U.A.; Hurni, A.; Bianchi, L.; Göldi, H. Rezidiv einer medikamentös-toxischen Hepatitis. Schweiz Med. Forum 2005, 5, 147–148. [Google Scholar]
- Duque, J.M.; Ferreiro, J.; Salgueiro, E.; Manso, G. Hepatotoxicity associated with the consumption of herbal slimming products. Med. Clin. 2007, 128, 238–239. [Google Scholar]
- Elinav, E.; Pinsker, G.; Safadi, R.; Pappo, O.; Bromberg, M.; Anis, E.; Keinan-Boker, L.; Broide, E.; Ackerman, Z.; Nitzan Kaluski, D.; et al. Association between consumption of Herbalife nutritional supplements and acute hepatotoxicity. J. Hepatol. 2007, 47, 514–520. [Google Scholar] [CrossRef] [PubMed]
- Schoepfer, A.M.; Engel, A.; Fattinger, K.; Marbet, U.A.; Criblez, D.; Reichen, J.; Zimmermann, A.; Oneta, C.M. Herbal does not mean innocuous: Ten cases of severe hepatotoxicity associated with dietary supplements from Herbalife products. J. Hepatol. 2007, 47, 521–526. [Google Scholar] [CrossRef] [PubMed]
- Chao, S.; Anders, M.; Turbay, M.; Olaiz, E.; Mc Cormack, L.; Mastai, R. Toxic hepatitis by consumption of Herbalife products: A case report. Acta Gastroenterol. Latinoam 2008, 38, 274–277. [Google Scholar] [PubMed]
- Manso, G.; López-Rivaz, L.; Duque, J.M.; Salgueiro, E. Spanish reports of hepatotoxicity associated with Herbalife. J. Hepatol. 2008, 49, 289–290. Author reply 290–292. [Google Scholar] [CrossRef] [PubMed]
- Stickel, F.; Droz, S.; Patsenker, E.; Bögli-Stuber, K.; Aebi, B.; Leib, S.L. Severe hepatotoxicity following ingestion of Herbalife nutritionally supplements contaminated with Bacillus subtilis. J. Hepatol. 2009, 50, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Jóhannsson, M.; Ormarsdóttir, S.; Olafsson, S. Hepatotoxicity associated with the use of Herbalife. Laeknabladid 2010, 96, 167–172. [Google Scholar] [PubMed]
- Chen, G.C.; Ramanathan, V.S.; Law, D.; Funchain, P.; Chen, G.C.; French, S.; Shlopov, B.; Eysselein, V.; Chung, D.; Reicher, S.; et al. Acute liver injury induced by weight-loss herbal supplements. World J. Hepatol. 2010, 2, 410–415. [Google Scholar] [CrossRef] [PubMed]
- Appelhans, K.; Smith, C.; Bejar, E.; Henig, Y.S. Revisiting acute liver injury associated with Herbalife products. World J. Hepatol. 2011, 3, 275–277. [Google Scholar] [CrossRef] [PubMed]
- Manso, G.; López-Rivas, L.; Salgueiro, M.E.; Duque, J.M.; Jimeno, F.J.; Andrade, R.J.; Lucena, M.I. Continuous reporting of new cases in Spain supports the relationship between Herbalife products and liver injury. Pharmacoepidemiol. Drug Saf. 2011, 20, 1080–1087. [Google Scholar] [CrossRef] [PubMed]
- Appelhans, K.; Frankos, V.; Shao, A. Misconceptions regarding association between Herbalife products and liver related case reports in Spain. Pharmacoepidemiol. Drug Saf. 2012, 21, 333–334. [Google Scholar] [CrossRef] [PubMed]
- Stevens, T.; Qadri, A.; Zein, N.N. Two patients with acute liver injury associated with use of the herbal weight-loss supplement hydroxycut. Ann. Intern. Med. 2005, 14, 477–478. [Google Scholar] [CrossRef]
- Jones, F.J.; Andrews, A.H. Acute liver injury associated with the herbal supplement hydroxycut in a soldier deployed to Iraq. Am. J. Gastroenterol. 2007, 102, 2357–2358. [Google Scholar] [CrossRef] [PubMed]
- Shim, M.; Saab, S. Severe hepatotoxicity due to Hydroxycut: A case report. Dig. Dis. Sci. 2009, 54, 406–408. [Google Scholar] [CrossRef] [PubMed]
- Fong, T.L.; Klontz, K.C.; Canas-Coto, A.; Casper, S.J.; Durazo, F.A.; Davern, T.J.; Hayashi, P.; Lee, W.M.; Seeff, L.B. Hepatotoxicity due to Hydroxycut: A case series. Am. J. Gastroenterol. 2010, 105, 1561–1566. [Google Scholar] [CrossRef] [PubMed]
- Wainwright, J.; Schonland, M.M. Toxic hepatitis in black patients in Natal. S. Afr. Med. J. 1977, 51, 571–573. [Google Scholar] [PubMed]
- Wainwright, J.; Schonland, M.M.; Candy, H.A. Toxicity of Callilepis laureola. S. Afr. Med. J. 1977, 52, 313–315. [Google Scholar] [PubMed]
- Popat, A.; Shear, N.H.; Malkiewicz, I.; Stewart, M.J.; Steenkamp, V.; Thomson, S.; Neuman, M.G. The toxicity of Callilepis laureola, a South African traditional herbal medicine. Clin. Biochem. 2001, 34, 229–236. [Google Scholar] [CrossRef]
- Fraquelli, M.; Colli, A.; Cocciolo, M.; Conte, D. Adult syncytial giant cell chronic hepatitis due to herbal remedy. J. Hepatol. 2000, 33, 505–508. [Google Scholar] [CrossRef]
- Brauer, R.B.; Pfab, R.; Becker, K.; Berger, H.; Stangl, M. Fulminantes Leberversagen nach Einnahme des pflanzlichen Heilmittels Kava-Kava. Z Gastroenterol. 2001, 39, 30, (abstract in German). [Google Scholar]
- Escher, M.; Desmeules, J.; Giostra, E.; Mentha, G. Hepatitis associated with kava, a herbal remedy for anxiety. Br. Med. J. 2001, 322, 139. [Google Scholar] [CrossRef]
- Kraft, M.; Spahn, T.W.; Menzel, J.; Senninger, N.; Dietl, K.H.; Herbst, H.; Domschke, W.; Lerch, M.M. Fulminant liver failure after administration of the herbal antidepressant Kava-Kava. Dtsch Med. Wschr. 2001, 126, 970–972. (In German) [Google Scholar] [CrossRef] [PubMed]
- Russmann, S.; Lauterburg, B.H.; Helbling, A. Kava hepatotoxicity. Ann. Intern. Med. 2001, 135, 68–69. [Google Scholar] [CrossRef] [PubMed]
- Saß, M.; Schnabel, S.; Kröger, J.; Liebe, S.; Schareck, W.D. Akutes Leberversagen durch Kava Kava—eine seltene Indikation zur Lebertransplantation. Z. Gastroenterol. 2001, 39, 29. (In German) [Google Scholar]
- Bujanda, L.; Palacios, A.; Silvariño, R.; Sánchez, A.; Muñoz, C. Hepatitis aguda icterica secundaria a kava. Gastroenterol. Hepatol. 2002, 25, 434–435. [Google Scholar] [CrossRef]
- Denham, A.; McIntyre, M.; Whitehouse, J. Kava—The unfolding story: Report on a work-in-progress. J. Altern. Complement. Med. 2002, 8, 237–263. [Google Scholar] [CrossRef] [PubMed]
- Weise, B.; Wiese, M.; Plötner, A.; Ruf, B.R. Toxic hepatitis after intake of kava-kava. Verdauungskrankheiten 2002, 4, 166–169. (In German) [Google Scholar]
- Gow, P.J.; Connelly, N.J.; Hill, R.L.; Crowley, P.; Angus, P.W. Fatal fulminant hepatic failure induced by a natural therapy containing kava. Med. J. Aust. 2003, 178, 442–443. [Google Scholar] [PubMed]
- Humberston, C.L.; Akhtar, J.; Krenzelok, E.P. Acute hepatitis induced by kava kava. J. Toxicol. Clin. Toxicol. 2003, 41, 109–113. [Google Scholar] [CrossRef] [PubMed]
- Russmann, S.; Barguil, Y.; Cabalion, P.; Kritsanida, M.; Duhet, D.; Lauterburg, B.H. Hepatic injury due to traditional aqueous extracts of kava root in New Caledonia. Eur. J. Gastroenterol. Hepatol. 2003, 15, 1033–1036. [Google Scholar] [CrossRef] [PubMed]
- Schulze, J.; Raasch, W.; Siegers, C.P. Toxicity of kava pyrones, drug safety and precautions–A case study. Phytomedicine 2003, 10, 68–73. [Google Scholar] [CrossRef] [PubMed]
- Stickel, F.; Baumüller, H.M.; Seitz, K.; Vasilakis, D.; Seitz, G.; Seitz, H.K.; Schuppan, D. Hepatitis induced by kava (Piper methysticum rhizoma). J. Hepatol. 2003, 39, 62–67. [Google Scholar] [CrossRef]
- Christl, S.U.; Seifert, A. Seeler D: Toxic hepatitis after consumption of traditional kava preparation. Int. Soc. Travel. Med. 2009, 16, 55–56. [Google Scholar] [CrossRef] [PubMed]
- McGee, J.; Patrick, R.S.; Wood, C.B.; Blumgart, L.H. A case of veno-occlusive disease of the liver in Britain associated with herbal tea consumption. J. Clin. Pathol. 1976, 29, 788–794. [Google Scholar] [CrossRef] [PubMed]
- Harvey, J.; Colin-Jones, D.G. Mistletoe hepatitis. Br. Med. J. 1981, 282, 186–187. [Google Scholar] [CrossRef]
- Hyde, F.F. Mistletoe hepatitis. Br. Med. J. 1981, 282, 739. [Google Scholar] [CrossRef]
- Colin-Jones, D.G.; Harvey, J. Mistletoe hepatitis. Br. Med. J. 1982, 284, 744–745. [Google Scholar] [CrossRef]
- Farnsworth, N.R.; Loub, W.D. Mistletoe hepatitis. Br. Med. J. 1981, 283, 1058. [Google Scholar] [CrossRef]
- Stirpe, F. Mistletoe hepatitis. Br. Med. J. 1983, 1, 29. [Google Scholar]
- Millonig, G.; Stadlmann, S.; Vogel, W. Herbal hepatotoxicity: Acute hepatitis caused by a Noni preparation (Morinda citrifolia). Eur. J. Gastroenterol. Hepatol. 2005, 17, 445–447. [Google Scholar] [CrossRef] [PubMed]
- Stadlbauer, V.; Fickert, P.; Lackner, C.; Schmerlaib, J.; Krisper, P.; Trauner, M.; Stauber, R.E. Hepatotoxicity of NONI juice: Report of two cases. World J. Gastroenterol. 2005, 11, 4758–4760. [Google Scholar] [CrossRef] [PubMed]
- Yüce, B.; Gülberg, V.; Diebold, J.; Gerbes, A.L. Hepatitis induced by Noni juice from Morinda citrifolia: A rare cause of hepatotoxicity or the tip of a iceberg? Digestion 2006, 73, 167–170. [Google Scholar] [PubMed]
- López-Cepero Andrada, J.M.; Lerma Castilla, S.; Fernandèz Olvera, M.D.; Amaya Vidal, A. Hepatotoxicity caused by a Noni (Morinda citrifolia) preparation. Espanola de Patologia Digestiva 2007, 99, 179–181. (In Spanish) [Google Scholar]
- Stadlbauer, V.; Weiss, S.; Payer, F.; Stauber, R.E. Herbal does not at all mean innocuous: The sixth case of hepatotoxicity associated with Morinda citrifolia (Noni). Am. J. Gastroenterol. 2008, 103, 2406–2407. [Google Scholar] [PubMed]
- Yu, E.L.; Sivagnanam, M.; Ellis, L.; Huang, J.S. Acute hepatotoxicity after ingestion of Morinda citrifolia (Noni Berry) juice in a 14-year-old boy. J. Pedriatr. Gastroenterol. Nutr. 2011, 52, 222–224. [Google Scholar] [CrossRef] [PubMed]
- Altan, E.; Bitik, B.; Kalpkci, Y.; Dogan, E.; Altundag, K. Probable hepatotoxicity related to Nerium oleander extract in a patient with metastatic synovial sarcoma of the knee. J. Altern. Complement. Med. 2009, 15, 113. [Google Scholar] [CrossRef] [PubMed]
- Mokhobo, K.P. Herb use and necrodegenerative hepatitis. S. Afr. Med. J. 1976, 50, 1096–1099. [Google Scholar] [PubMed]
- Vallance, W.B. Pennyroyal poisoning: A fatal case. Lancet 1955, 266, 850–851. [Google Scholar] [CrossRef]
- Sullivan, J.B.; Rumack, B.H.; Thomas, H.; Peterson, R.G.; Bryson, P. Pennyroyal oil poisoning and hepatotoxicity. J. Am. Med. Assoc. 1979, 242, 2873–2874. [Google Scholar] [CrossRef]
- Anderson, I.B.; Mullen, W.H.; Meeker, J.E.; Khojasteh-Bakht, S.C.; Oishi, S.; Nelson, S.D.; Blanc, P.D. Pennyroyal toxicity: Measurement of toxic metabolite levels in two cases and review o the literature. Ann. Intern. Med. 1996, 124, 726–734. [Google Scholar] [CrossRef] [PubMed]
- Bakerink, J.A.; Gospe, S.M.; Dimand, R.J.; Eldridge, M.W. Multiple organ failure after ingestion of pennyroyal oil from herbal tea in two infants. Pediatrics 1996, 98, 944–947. [Google Scholar] [PubMed]
- Joshi, D.; Cross, T.J.S.; Wong, V.S. Acute drug induced hepatitis secondary to a weight loss product purchased over the internet. Nutr. J. 2007, 6. [Google Scholar] [CrossRef] [PubMed]
- Roselle, H.; Ekatan, A.; Tzeng, J.; Sapienza, M.; Kocher, J. Symptomatic hepatitis associated with the use of herbal Red Yeast Rice. Ann. Intern. Med. 2008, 149, 516–517. [Google Scholar] [CrossRef] [PubMed]
- Larrey, D. Hepatotoxicity of herbal remedies. J. Hepatol. 1997, 26, 47–51. [Google Scholar] [CrossRef]
- Lapi, F.; Gallo, E.; Giocaliere, E.; Vietri, M.; Baronti, R.; Pieraccini, G.; Tafi, A.; Menniti-Ippolito, F.; Mugelli, A.; Firenzuoli, F.; et al. Acute liver damage due to Serenoa repens: A case report. Br. J. Clin. Pharmacol. 2010, 69, 558–560. [Google Scholar] [CrossRef] [PubMed]
- MacGregor, F.B.; Abernethy, V.E.; Dahabra, S.; Cobden, I.; Hayes, P.C. Hepatotoxicity of herbal remedies. Br. Med. J. 1989, 299, 1156–1157. [Google Scholar] [CrossRef]
- Caldwell, S.H.; Feeley, J.W.; Wieboldt, T.F.; Featherston, P.L.; Dickson, R.C. Acute hepatitis with use of over-the-counter herbal remedies. Va Med. Q. 1994, 121, 31–33. [Google Scholar] [PubMed]
- Hullar, T.E.; Sapers, B.L.; Ridker, P.M.; Judkins, R.L.; Huth, T.S.; Farraye, F.A. Herbal toxicity and fatal hepatic failure (letter). Am. J. Med. 1999, 106, 267–268. [Google Scholar] [PubMed]
- Beuers, U.; Spengler, U.; Pape, G.R. Hepatitis after chronic abuse of senna. Lancet 1991, 337, 372–373. [Google Scholar] [CrossRef]
- Seybold, U.; Landauer, L.; Hillebrand, S.; Goebel, F.D. Senna-induced hepatitis in a poor metabolizer. Ann. Intern. Med. 2004, 141, 650–651. [Google Scholar] [CrossRef] [PubMed]
- Vanderperren, B.; Rizzo, M.; Angenot, L.; Haufroid, V.; Jadoul, M.; Hantson, P. Acute liver failure with renal impairment related to the abuse of Senna anthraquinone glycosides. Ann. Pharmacother. 2005, 39, 1353–1357. [Google Scholar] [CrossRef] [PubMed]
- Mennecier, D.; Saloum, T.; Dourthe, P.M.; Bronstein, J.A.; Thiolet, C.; Farret, O. Acute hepatitis after phytotherapy. Présse Med. 1999, 28, 966. [Google Scholar] [PubMed]
- De Smet, P.A.G.M.; van den Eertwegh, A.J.M.; Lesterhuis, W.; Stricker, B.H.C. Hepatotoxicity associated with herbal tablets. BMJ 1996, 313, 92. [Google Scholar] [CrossRef] [PubMed]
- Takegoshi, K.; Tohyama, T.; Okuda, K.; Suzuki, K.; Ohta, G. A case of Venoplant-induced hepatic injury. Gastroenterol. Jpn. 1986, 21, 62–65. [Google Scholar] [PubMed]
- Mathieu, N.; Bouallegue, L.; Mognol, P.; Vallot, T.; Soule, J.C. Hepatic toxicity probably due to X-elles in phytotherapy. Gastroenterol. Clin. Biol. 2005, 29, 1188–1189. [Google Scholar] [CrossRef]
- Hayashi, P.H.; Fontana, R.J.; Chalasani, N.P.; Stolz, A.A.; Talwalkar, J.A.; Navarro, V.J.; Lee, W.M.; Davern, T.J.; Kleiner, D.E.; Gu, J.; et al. Under-reporting of poor adherence to monitoring guidelines for severe cases of Isoniazid hepatotoxicity. Clin. Gastroenterol. Hepatol. 2015, 13, 1676–1682. [Google Scholar] [CrossRef] [PubMed]
- Chalasani, N.P.; Hayashi, P.H.; Bonkovsky, H.L.; Navarro, V.J.; Lee, W.M.; Fontana, R.J. ACG Clinical guideline: The diagnosis and management of idiosyncratic drug-induced liver injury. Am. J. Gastroenterol. 2014, 109, 950–966. [Google Scholar] [CrossRef] [PubMed]
- Mohabbat, O.; Younos, M.S.; Merzad, A.A.; Srivastava, R.N.; Sediq, G.G.; Aram, G.N. An outbreak of hepatic veno-occlusive disease in north-western Afghanistan. Lancet 1976, 308, 269–271. [Google Scholar] [CrossRef]
- Kakar, F.; Akbarian, Z.; Leslie, T.; Mustafa, M.L.; Watson, J.; van Egmond, H.P.; Omar, M.F.; Mofleh, J. An outbreak of hepatic veno-occlusive disease in western Afghanistan associated with exposure to wheat flour contaminated with pyrrolizidine alkaloids. J. Toxicol. 2010, 313280. [Google Scholar] [CrossRef] [PubMed]
- Tandon, R.K.; Tandon, B.N.; Tandon, H.D. Study of an epidemic of venoocclusive disease in India. Gut 1976, 17, 849–855. [Google Scholar] [CrossRef] [PubMed]
- Tandon, B.N.; Tandon, H.D.; Tandon, R.K.; Narndranathan, M.; Joshi, Y.K. An epidemic of veno-occlusive disease of the liver in central India. Lancet 1976, 308, 271–272. [Google Scholar] [CrossRef]
| Item | Product Quality Specifications |
|---|---|
| Herbal product declarations | Declaration of the manufacturer with address, phone and fax number, e-mail Expiration date of the herbal drug and herbal supplement Batch number Correct labelling of all ingredients |
| Herbal product definitions | Definition of plant family, subfamily, species, subspecies, and variety Definition of plant part Definition of used solvents and solubilizers |
| Herbal quality standards | Exclusion of impurities, adulterants, and misidentifications Minimum or lack of batch to batch variability Minimum or lack of product to product variability Lack of variety to variety variability Current Good Agricultural Practices (cGAPs) Current Good Manufacturing Practices (cGMPs) Regulatory surveillance |
| Consumer information | Detailed recommendation for indication and contraindication Advice for daily dose and maximum use duration |
| Chinese Name | Scientific Name | Tentative Hepatotoxic Components |
|---|---|---|
| Ai Ye | Artemisia argyi | Volatile oil |
| Bi Ma Zi | Rhicinus communis | Ricin, toxic proteins |
| Cang Shan | Xanthium | Glycosides (kaurene), diterpenoids |
| Chang Shan | Dichor febrifuga Lour | Alkaloids (dichroine) |
| He Huan Pi | Albizia julibrissin | Glycosides (saponine) |
| He Shou Wu | Polygonum multiflorum | Anthraquinones |
| Huang Yao Zi | Discorea bulbifera L | Glycosides (steroids, diosgenin), diterpenoids-lactones |
| Ku Lian Zi | Melia azedarach | Glycosides (tetranortriterpenoids) |
| Lei Gong Teng | Tripterygium wilfordii hook F | Glycosides (tripterygium), diterpenoid-lactones |
| Qian Li Guang | Senecio scandens | Pyrrolizidine alkaloids |
| Shan Lu | Phytolacca acinosa Roxb. | Alkaloids (phytolaccine) |
| Xiang Si Zi | Abrus Precatorius | Abrin |
| Items | Clinical Characteristics of HILI by Greater Celandine (GC) |
|---|---|
| ● Background | 1. Clinical characterization of HILI by GC as a specific disease entity was feasible and based on 16 patients with liver disease and high causality levels for GC; |
| ● RUCAM | 2. RUCAM based causality for GC was graded highly probable in 4 patients and probable in 12 patients; |
| ● Comedication causality assessment by RUCAM | 3. Among these 16 patients, there was an additional RUCAM based causality for comedication with curcuma graded as possible, for comedication with Lycopodium serratum graded as probable, and for biliary disease graded as possible; |
| ● Positive reexposure result | 4. The existence of GC HILI has been verified by a positive reexposure test in two patients; |
| ● Age and gender | 5. Ages of the 16 patients ranged from 32 to 69 years with an average of 54.7 years, and the ratio of females: males was 10: 6; |
| ● High comedication rate | 6. Comedication with synthetic or herbal drugs and dietary supplements and herbal mixtures was used in the majority of assessable cases; |
| ● Chelidonine dose adherence | 7. On average, the patients used 10 mg chelidonine daily, with no reported overdose in any of the cases; |
| ● Variable treatment duration | 8. Treatment duration was 3 weeks to 9 months with an average of 2.4 months; |
| ● Continued GC use despite symptoms | 9. Latency period until first symptoms was 3 weeks to 4.5 months with an average of 1.7 months, which was considerably shorter than the treatment length; |
| ● Jaundice as major symptom of GC induced HILI | 10. Jaundice was the most frequently reported symptom, rarely also weakness, anorexia, nausea, vomiting, abdominal pains, dark urine, pale stools, and itching; |
| ● Hepatocellular liver injury | 11. High serum activities are found for ALT but not for ALP, suggestive of hepatocellular liver injury in patients with GC HILI; |
| ● Liver histology | 12. Histology showed predominantly liver cell necrosis and hepatitis; |
| ● Favorable outcome | 13. Outcome was favorable in all 16 patients, with lack of both acute liver failure and requirement of a liver transplant; |
| ● Good prognosis despite continued GC intake | 14. In one patient, good prognosis was sustained even after 7 months of continued GC use despite presence of emerging GC HILI; |
| ● Idiosyncratic liver injury with its metabolic subgroup | 15. GC HILI usually represents the hepatocellular and idiosyncratic type of liver injury with its metabolic subgroup, characterized as acute clinical course; |
| ● Individual culprits still undetermined | 16. The underlying mechanism(s) leading to GC hepatotoxicity as well as possible culprit(s) are still unknown; |
| ● Safety concern | 17. In cases of liver disease, causality for GC was verified and creates concern regarding safety of patients; |
| ● GC HILI likely with low incidence | 18. Lacking valid epidemiologic data, but incidence of GC HILI is likely low. |
| Conditions | Symptoms |
|---|---|
| ● Herbal use for overall 9 months, with full daily dose as recommended for the initial 7 months. | Well-being during first 6 months of treatment with Indian Ayurvedic herbs for her vitiligo. Pruritus as first symptom after herbal treatment for 6 months, with subsequent symptoms step by step: Loss of appetite, fatigue, nausea, and vomiting several times per week. |
| ● After herbal use for 7 months, dose reduction to 50% for the remaining 2 months of treatment. Dose reduction was the patient’s decision. The clinic in India, which distributed this herbal medicine via internet, denied an association with the described symptoms. | Dark urine after use of herbs for 7 months. Reduced daily dose led to disappearance of pruritus but other symptoms persisted. Light-colored stool as new symptom appearing 8 months after initiation of the herbal treatment. |
| ● First presentation to her PCP 9 months after initiation of herbal treatment | Jaundice was objectively confirmed |
| ● Discontinuation of herbal treatment 9 months after treatment initiation and 3 months following emerging pruritus as the first symptom | Hospital admission the other day and discontinuation of the herbal treatment as HILI was suspected. Clinical evaluation with exclusion of alternative diagnoses including HEV. Causality assessment by RUCAM that provided a probable causality. Following cessation of herbal treatment, clinical symptoms and LTs rapidly improved to complete recovery. This case is best described as idiosyncratic HILI of the metabolic type. |
| Items | Clinical Characteristics of Kava Hepatotoxicity |
|---|---|
| ● Positive reexposure result | 1. The existence of kava hepatotoxicity has been verified by a positive reexposure test; |
| ● RUCAM | 2. RUCAM based causality for kava ± comedication was graded probable in 4 patients; |
| 3. In nine patients and thus in the majority of case, causality for kava ± comedication was possible and hence weak; | |
| ● Kava products | 4. Kava hepatotoxicity may be caused by traditional aqueous kava extracts, commercial ethanolic and acetonic kava extracts, and kava-herbs mixtures; |
| ● Risk factors | 5. Daily overdose of kavalactones and prolonged treatment were common phenomena in patients with kava hepatotoxicity and considered as risk factors, although it occurred also with normal doses under recommended therapy duration of 3 months or less; |
| 6. Synthetic or herbal drugs and dietary supplements including herbal ones were comedicated with kava in the majority of cases and considered as risk factors; | |
| 7. Additional risk factors included non-adherence to regulatory treatment recommendations, but not extraction media or solubilizers, used for the manufacturing of the kava drug; | |
| ● Age and Gender | 8. The ages of the 14 patients with a probable causality for kava ± comedication or a highly probable causality for kava ranged from 14 to 60 years, and the ratio of females: males was 6:1; |
| ● Hepatocellular injury | 9. High serum activities were found for ALT but not for ALP, suggesting a hepatocellular injury type in kava hepatotoxicity; |
| ● Liver histology | 10. Histology showed predominantly liver cell necrosis and hepatitis; |
| ● Pathogenetic type of liver injury | 11. Depending on the kava product used, kava hepatotoxicity represents primarily the idiosyncratic type of injury with the metabolic subtype but secondarily also the intrinsic type of injury; the intrinsic and thereby predictable type of hepatotoxicity applies to those patients who might have used one of the few extracts containing kava of inappropriate quality with toxic properties, or who took kava with daily overdose or prolonged; |
| ● Incidence | 12. Valid epidemiology data are lacking, and the incidence of kava hepatotoxicity cannot be accurately calculated, but appears to be low. |
| Conditions | Results |
|---|---|
| ● Cohort | n = 116 |
| ● Gender | Males 57 Females 56 (NA 3) |
| ● Age | 17–76 years |
| ● Ascites | 115/116 cases |
| ● Hepatomegaly | 104/113 cases |
| ● Jaundice | 95/113 cases |
| ● ALT elevation | 47/60 cases (NA 56 cases) |
| ● AST elevation | 50/58 cases (NA 58 cases) |
| ● Outcome | Recovery 75 cases Chronicity 27 cases Death 11 cases (NA 3 cases) |
| Items | RUCAM |
|---|---|
| ● Time frame of latency period (score) | + |
| ● Time frame of dechallenge (score) | + |
| ● Recurrent ALT or ALP increase (score) | + |
| ● Definition of risk factors (score) | + |
| ● All comedications (score) | + |
| ● Individual comedication (score) | + |
| ● Search for individual alternative causes (score) | + |
| ● Verified exclusion of specific alternative causes (score) | + |
| ● All specifically assessed HAV, HBV, HCV, HEV (score) | + |
| ● All specifically assessed CMV, EBV, HSV, VZV (score) | + |
| ● Evaluation of cardiac hepatopathy (score) | + |
| ● Liver and biliary tract imaging (score) | + |
| ● Color Doppler sonography of liver vessels (score) | + |
| ● Prior known hepatotoxicity (score) | + |
| ● Search for unintended reexposure (score) | + |
| ● Definition of unintended reexposure (score) | + |
| ● Qualified criteria of unintended reexposure (score) | + |
| ● Laboratory hepatotoxicity criteria | + |
| ● Laboratory hepatotoxicity pattern | + |
| ● Hepatotoxicity specific method | + |
| ● Structured, liver related method | + |
| ● Quantitative, liver related method | + |
| ● Validated method (gold standard) | + |
| ● Worlwide use | + |
| ● Use by international registries | + |
| ● Use by regulatory agencies | + |
| ● Use by HILI case reports | + |
| ● Use by HILI case series | + |
| ● Transparent documentation | + |
| Reexposure Test Result | Hepatocellular Injury | Cholestatic or Mixed Liver Injury | ||
|---|---|---|---|---|
| ALTb | ALTr | ALPb | ALPr | |
| ● Positive | <5N | ≥2ALTb | <2N | ≥2ALPb |
| ● Negative | <5N | <2ALTb | <2N | <2ALPb |
| ● Negative | ≥5N | ≥2ALTb | ≥2N | ≥2ALPb |
| ● Negative | ≥5N | <2ALTb | ≥2N | <2ALPb |
| ● Uninterpretable | <5N | n.a. | <2N | n.a. |
| ● Uninterpretable | n.a. | ≥2ALTb | n.a. | ≥2ALTb |
| ● Uninterpretable | n.a. | n.a. | n.a. | n.a. |
| Herbal TCM | RUCAM Based Causality | Reexposure Based Causality |
|---|---|---|
| ● Bai Xian Pi | + | − |
| ● Bo He | + | − |
| ● Ci Wu Jia | + | − |
| ● Chuan Lian Zi | + | − |
| ● Da Huang | + | − |
| ● Gan Cao | + | − |
| ● Ge Gen | + | − |
| ● Ho Shou Wu | + | − |
| ● Huang Qin | − | − |
| ● Hwang Geun Cho | − | + |
| ● Ji Gu Cao | − | + |
| ● Ji Xue Cao | − | − |
| ● Jin Bu Huan | + | + |
| ● Jue Ming Zi | + | − |
| ● Jiguja | + | − |
| ● Kudzu | − | − |
| ● Ling Yang Qing Fei Keli | + | − |
| ● Lu Cha | + | + |
| ● Ma Huang | − | + |
| ● Polygonum multiflorum | + | + |
| ● Rhen Shen | + | − |
| ● Shou Wu Pian | + | + |
| ● Shan Chi | + | − |
| ● Shen Min | + | − |
| ● Syo Saiko To | + | + |
| ● Xiao Chai Hu Tang | − | + |
| ● Zexie | + | − |
| ● Zhen Chu Cao | + | − |
| Problematic Items as Confounding Variables for Causality Assessment in some Cases | References for few HILI Cases as Examples |
|---|---|
| ● Problematic plant authentication | [14,16,22,25,26,27,28,29,105,106,107] |
| ● Unspecified plant part | [107] |
| ● Lacking herbal product identification | [106,107] |
| ● Unidentified indication of herbal treatment | [107] |
| ● Unassessed daily dosage | [107] |
| ● Unidentified date of product start | [107] |
| ● Unidentified date of product stop | [107] |
| ● Unclear time to onset | [106,107] |
| ● Unconfirmed herbal product purchase | [49,50] |
| ● Unconfirmed herbal product use | [49,50] |
| ● Unassessed comedication by drugs | [48,49,50] |
| ● Use of many herbal dietary supplements | [49,50,107] |
| ● Unreported initial ALT value | [107] |
| ● Unreported initial ALP value | [107,135] |
| ● Unconsidered HAV | [50,107,135] |
| ● Unconsidered HBV | [50,99,106,107,135] |
| ● Unconsidered HCV | [50,99,106,107,135] |
| ● Unconsidered HEV | [48,49,50,99,106,107] |
| ● Unconsidered CMV | [50,99,106,107] |
| ● Unconsidered EBV | [48,49,50,99,106,107] |
| ● Unconsidered HSV | [48,49,50,99,106,107] |
| ● Unconsidered VZV | [50,99,106,107] |
| ● Incomplete imaging data | [99,106,107] |
| ● Ignored hepatotoxicity criteria | [107] |
| ● Causality assessment by non-RUCAM | [24,94,99,105,107,112,120] |
| Chinese Name | Botanical Names, Ingredients |
|---|---|
| ● Ai Ye | Artemisia argyi |
| ● An Shu Ling | Lycopodium serratum or rarely, Corydalis species, Panax ginseng, Pseudo ginseng, or two species of Stephania |
| ● Bai Fang | Angelica sinensis, Cyperus rotundus, Ginseng, Ligusticum wallichii, Paeonia alba, Rehmannia glutinosa |
| ● Bai Shi Wan | Atractylis, Carthamus tinctorius, Dalbergia odorifera, Dioscorea bulbifera, Glycyrrhiza, Lithospermum erythrorhizon, Paeonia suffruticosa, Polygonum multiflorum, Psoralea corylifolia, Salvia miltiorrhiza; Endoconcha sepiae, Ganoderma lucidum (mushroom) |
| ● Bi Ma Zi | Rhicinus communis, Chaenomeles, Codonopsis pilosula, Notopterygium, Polygonum multiflorum, Rehmannia, Schisandra |
| ● Bo He | Mentha haplocalyx |
| ● Bo Ye Qing Niu Dan | Tinospora crispa |
| ●Bofu Tsu Sho San | Angelica, Atractylis, Cnidium, Gardenia, Ephedra, Forsythia, Glycyrrhhiza, Gypsum fibrosum, Ledebouriella, Mentha, Paeonia, Platycodon, Rheum, Schizonepeta, Scutellaria, Zingiber; Kadinum (talcum powder), sodium sulfuricum |
| ● Boh Gol Zhee | Psoralea corylifolia |
| ● Cang Er Zi | Xanthium sibiricum |
| ● Chang Shan | Dichora febrifuga Lour |
| ● Chai Hu | Bupleurum falcatum |
| ● Chaso | Camellia sinensis, Cassia tora (syn. Senna), Crataegus, N-nitroso-fenfluramine |
| ● Chi R Yun | Breynia officinalis |
| ● Chinese herbal mixtures (various) | Dictamnus dasycarpus, Gentiana scabra, Hedyotis diffusa, Paeonia suffructicosa, Paris polyphylla, Rehmannia glutinosa, Smilax glabra, Sophora subprostrata; Angelica sinensis, Bupleurum chinese, Dictamnus dasycarpus, Paeonia suffructiosa, Philodendron chinese, Saposhnikovia divaricata,Shisandra chinesis, Shizonepeta tenuifolia, Tribulus terrestris; Cocculus trilobus, Dictamnus dasycarpus, Eurysolen gracilis, Glycyrrhiza, Lophatherum, Paeonia, Potentilla, Rehmannia glutinosa; Alisma plantago aquatica, Artemisia capillaris, Bupleurum, Chrysanthemum morifolium, Circuma, Gardenia jasminoidis, Gentiana scabra, Glycyrrhiza, Magnolia, Paeonia, Plantago asiatica, Saussurea lappa |
| ● Chuan Lian Zi | Melia toosendan |
| ● Ci Wu Jia | Acanthopanax senticosus |
| ● Da Chai Hu Tang | Bupleurum falcatum, Ginseng, Glycyrrhiza glabra, Pinellia, Scutellaria, Zingiber officinale, Zizyphus jujuba |
| ● Da Huang | Rheum palmatum |
| ● Du Huo | Angelica archangelica |
| ● Fu Fang Qing Dai Wan | Angelica dahurica, Isatis indigotica (Indigo naturalis), Massa medicata fermentata (yeast), Salvia milthiorrhiza, Smilax glabra |
| ● Gan Cao | Glycyrrhiza uralensis, syn. Liquorice |
| ● Ge Gen | Pueraria lobata, syn. Arrowroot |
| ● He Huan Pi | Albizia julibrissin |
| ● Ho Shou Wu | Polygonum multiflorum, syn. He Shou Wu |
| ● Hu Bohe You | Mentha pulegium, syn. Pennyroyal oil |
| ● Hu Zhang | Polygonum cuspidatum |
| ● Huang Qin | Scutellaria baicalensis |
| ● Huang Yao Zi | Dioscorea bulbifera |
| ● Hwang Geun Cho | Corydalis speciosa |
| ● Ji Gu Cao | Abrus cantoniensis |
| ● Ji Ji | Chloranthus serratus |
| ● Ji Xue Cao | Centella asiatica, syn. Gotu Kola |
| ● Jiguja | Hovenia dulcis |
| ● Jin Bu Huan | Lycopodium serratum or rarely, Corydalis species, Panax ginseng, Pseudo ginseng, or two species of Stephania |
| ● Jue Ming Zi | Cassia obtusifolia, syn. Senna |
| ● Kamishoyosan | Angelica sinensis, Atractylodes racea, Bupleurum falcatum, Gardenia, Glycyrrhiza glabra, Mentha haplocalyx, Moutan, Paeonia alba, Sclerotium Poriae Cocos, Zingiber officinale |
| ● Kudzu | Pueraria thunbergiana |
| ● Ku Lian Zi | Melia azedarach |
| ● Lei Gong Teng | Tripterygium wilfordii Hook |
| ● Long Dan Xie Gan Tang | Acebia, Alisma, Angelica sinensis, Bupleurum, Gardenia, Gentiana, Glycyrrhiza, Plantago, Rehmannia, Scutellaria |
| ● Lu Cha | Camellia sinensis, syn. Chinese green tea |
| ● Ma Huang | Ephedra sinica |
| ● Mao Guo Tian Jie Cai | Heliotropium lasiocarpum |
| ● Onshido | Aloe, Camellia sinensis, Crataegus, Gynostemma pentaphyllum makino, Raphanus; N-nitroso-fenfluramine |
| ● Qian Li Guang | Senecio scandens |
| ● Ren Shen | Panax ginseng |
| ● Sairei To | Alisma, Atractylis, Bupleurum, Cinnamomum, Ginseng, Glycyrrhiza, Pinellia, Polyporus, Poria, Scutellaria, Zingiber, Zizyphus |
| ● Shan Chi | Gynura segetum |
| ● Shang Lu | Phytolacca acinosa |
| ● Shen Min | Black cohosh, Burdock, Cayenne pepper, Ginkgo biloba, Horse chestnut, Piper nigrum, Polygonum multiflorum, uva ursi; biotin, collagen (hydrolyzed), niacin, pantothenic acid, silica (from plant sources), soy isoflavones, vitamin A, vitamin B6 |
| ● Shi Can | Teucrium chamaedrys, syn. Germander |
| ● Shi Liu Pi | Pericarpium granati |
| ● ShouWu Pian | Achyranthes bidentata, Cuscuta chinensis, Eclipta prostrata, Ligustrum lucidum, Lonicera japonica, Morus alba, Polygonum multiflorum, Psoralea corylifolia, Rehmannia glutinosa, Rosa aevigat, Sesemum indicum, Siegesbeckia orientalis |
| ● Tian Hua Fen | Trichosanthes kirilowii |
| ● White flood | Qian Ceng Ta (Huperzia serrata), Wu Zhu Yu Evodia rutaecarpa); beet root, caffein, cocoa bean, vinpocetine (from Vinca plant); acesulfame potassium, calcium silicate, carnitine tartrate, Carno-Syn® beta-alanine, citrulline, cryptoxanthin, folic acid, gamma-aminobutyric acid (GABA), glucuronolactone, selenium, L-norvaline, L-tyrosine, lutein, malic acid, ornithine, potassium gluconate, sucralose, sugar cane, watermelon flavor, zeaxanthin |
| ● Wu Bei Zi | Galla chinensis |
| ● Xi Shu | Camptotheca acuminata |
| ● Xian Si Zi | Abrus Precatorius |
| ● Xiao Chai Hu Tang | Bupleurum falcatum, Ginseng, Glycyrrhiza glabra, Pinellia tuber, Scutellaria baicalensis, Zingiber officinale, Zizyphus jujuba |
| ● Yin Chen Hao | Artemisia capillaris |
| ● Zexie | Alisma orientalis |
| ● Zhen Chu Cao | Phyllanthus urinaria |
| Search Terms | Botanical Names, Ingredients |
|---|---|
| ● Acacia catechu | see Ayurvedic herbs |
| ● Aloe | Aloe perfoliata var. vera |
| ● Amorphophallus Konjac | see Hydroxycut® |
| ● Arrowroot | Maranta aruninacea or Tacca leontopetaloides |
| ● Atractylis gummifera | see Distaff thistle |
| ● Ayurvedic herbs | Psoralea corylifolia, Acacia catechu, Eclipta alba or Bacopa monnieri, Vetivexia zizaniodis |
| ● Babchi | Psoralea corylifolia, see also Ayurvedic herbs |
| ● Bacopa monnieri | see Ayurvedic herbs |
| ● Boronia Sm. | see Pro-Lean® |
| ● Buchu tea | Agathosma betulina, Agathosma crenulata |
| ● Bush tea | Crotalaria species |
| ● Callilepis laureola | see Impila |
| ● Camellia sinensis | see green tea, Exolise®, Hydroxycut®, X-elles® |
| ● Cascara sagrada | Rhamnus purshianus |
| ● Cassia angustifolia | see Senna |
| ● Centella asiatica | see Gotu Kola, Pro-Lean® |
| ● Chamaerops humilis | see Saw Palmetto |
| ●Chaparral syn. Creosot | Larrea tridentata, Larrea divariatica |
| ● Chelidonium majus | see Greater Celandine, Lycopodium similiaplex® |
| ● Chlorophora species | see Kambala tea |
| ● Chrysanthemum leucanthemum | see Oxeye Daisy |
| ● Citrus aurantium | see X-elles® |
| ● Citrus paradisum | see X-elles® |
| ● Cyrana scolymus | see X-elles® |
| ● Cola nitida | see Pro-Lean® |
| ● Coltsfoot | Tussilago farfara |
| ● Comfrey | Symphytum officinale, Symphytum asperum, Symphytum uplandicum |
| ● Compositae species | see Indian herbs |
| ● Creosot | see Chaparral |
| ● Crotalaria species | see Bush tea, Rattlebox |
| ● Cyperus | see Pro-Lean® |
| ● Distaff thistle | Atractylis gummifera |
| ● Eclipta alba | see Ayurvedic herbs |
| ● Emblica officinalis | see Isabgol |
| ● Ephedra species | Ephedra californica, Ephedra sinica |
| ● Exolise® | Garcinia cambogia, Gymnema sylvestre, White kidney bean, Camellia sinensis, l-Carnitine fumarate, Calcium, Magnesium chelate, Chromium chelate, Conjugated linoleic acid, Chitosan |
| ● Fallopia multiflora | see Pro-Lean® |
| ● Foeniculum amare | see Herbalife® |
| ● Fucus vesiculosus | see Pro-Lean® |
| ● Garcinia cambogia | see Exilis®, Herbalife®, Hydroxycut® |
| ● Germander | Teucrium chamaedrys, Teucrium polium |
| ● Ginkgo biloba | seePro-Lean® |
| ● Ginseng | see Bai Fang, Dai Saiko To, Pro-Lean®, Xiao Chai Hu Tang |
| ● Glycyrrhiza glabra | See Dai Saiko To, Xiao Chai Hu Tang |
| ● Gotu Kola | Centella asiatica |
| ● Greater Celandine | Chelidonium majus, see also Lycopodium similiaplex® |
| ● Green tea | Camellia sinensis, see also Lu Cha |
| ● Groundsel syn. Senecio | Senecio longilobus, Senecio species |
| ● Guaraná | Paullinia cupana |
| ● Gymnema sylvestre | see Exilis ®, Hydroxycut® |
| ● Hawthorn | see Crataegus |
| ● Hedeoma pulegoides | see Pennyroyal |
| ● Heliotropium | Heliotropium eichwaldii, Heliotropium species |
| ● Herbalife® | Solidaginis gigantea, Ilex paraguariensis, Petroselinum crispum, Garcinia cambogia, Spiraea, Matricaria chamomilla, Liquiritia, Foeniculum amare, Humulus lupulus, Chromium, and various other ingredients |
| ● Horse chestnut | see Venencapsan®, Venoplant® |
| ● Humulus lupulus | see Herbalife® |
| ● Hydroxycut® | Camellia sinensis, Gymnema sylvestre, Amorphophallus Konjac, Paullinia cupana, Garcinia cambogia, Caffeine, α-Lipoic acid, l-Carnitine, Calcium, Potassium, Chromium |
| ● Ilex paraguariensis | see Herbalife®, Maté |
| ● Impila | Callilepis laureola |
| ● Indian herbs | Compositae species |
| ● Iroko | see Kambala Tea |
| ● Isabgol | Plantago ovata, Emblica officinalis |
| ● Kambala Tea syn. Iroko | Chlorophora excelsa, Chlorophora regia |
| ● Kava | Piper methysticum |
| ● Larrea divariatica | see Chaparral |
| ● Larrea tridentata | see Chaparral |
| ● Leucanthemum vulgare | see Oxeye Daisy |
| ● Liquiritia | see Herbalife® |
| ● Lycopodium serratum | see Lycopodium similiaplex®, Wolf’s foot clubmass |
| ● Lycopodium similiaplex® | Lycopodium serratum, Chelidonium majus |
| ● Maranta aruninacea | see Arrowroot |
| ● Maté | Ilex paraguariensis |
| ● Mentha pulegium | see Pennyroyal |
| ● Mistletoe | Viscum album |
| ● Monascus purpureus | see Red Yeast Rice |
| ● Morinda citrifolium | see Noni |
| ● Nerium oleander | see Oleander |
| ● Noni | Morinda citrifolium |
| ● Oleander | Nerium oleander |
| ● Oxeye Daisy | Leucanthemum vulgare, Chrysanthemum leucanthemum |
| ● Paullinia cupana | see Guaraná, Hydroxycut®, Pro-Lean® |
| ● Pennyroyal | Mentha pulegium, Hedeoma pulegoides |
| ● Petroselinum crispum | see Herbalife® |
| ● Petroselinum sativum | see X-elles® |
| ● Piper methysticum | see Kava |
| ● Phaseolus vulgaris | see Exilis® |
| ● Plantago ovata | see Isabgol |
| ● Pro-Lean® | Ma Huang, Paullinia cupana, Cola nitida, Centella asiatica, Salix alba, Ginkgo biloba, Fucus vesiculosus, Boronia Sm., Ginseng, Fallopia multiflora, Cyperus, Bee pollen, Caffeine, l-Tyrosine, Chromium, Vanadium, Magnesium salicylat, Folic acid, Vitamin B12, and various other ingredients |
| ● Psoralea corylifolia | see Ayurvedic herbs |
| ● Pyrrolizidine alkaloid containing herbs | see Bush tea, see Comfrey, see Groundsel, see Heliotropium species, see Indian herbs, see Maté, see Rattlebox |
| ● Rattlebox syn. Crotalaria | Crotalaria species |
| ● Red Yeast Rice | Monascus purpureus |
| ● Rhamnus purshianu | see Cascara sagrada |
| ● Rooibos tea | Aspalathus linearis |
| ● Salix alba | see Pro-Lean® |
| ● Sassafra | Sassafras albidum |
| ● Saw Palmetto | Serenoa serpens, Chamaerops humilis |
| ● Skullcap | Scutellaria lateriflora, Scutellaria species |
| ● Scutellaria species | see Skullcap |
| ● Senecio | see Groundsel |
| ● Senna | Cassia angustifolia |
| ● Serenoa serpens | see Saw Palmetto |
| ● Solidaginis gigantea | see Herbalife® |
| ● Spiraea | see Herbalife® |
| ● Stonecrop | Sedum aizoon |
| ● Symphytum | see Comfrey |
| ● Tacca leontopetaloides | see Arrowroot |
| ● Teucrium | see Germander |
| ● Tussilago farfara | see Coltsfoot |
| ● Valerian | Valeriana officinalis |
| ● Valeriana officinalis | see Valerian |
| ● Venencapsan® | Aesculus hippocastanum, Chelidonium majus, Melilotus officinalis, Milfoil, Silybum Adans., Taraxacum officinale |
| ● Venoplant® | Aesculus hippocastanum |
| ● Vetivexia zizaniodis | see Ayurvedic herbs |
| ● Viscum album | see Mistletoe |
| ● Wolf’s foot clubmass | Lycopodium serratum |
| ● X-elles® | Petroselinum sativum, Citrus aurantium, Citrus paradisum, Cyrana scolymus, Camellia sinensis |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Frenzel, C.; Teschke, R. Herbal Hepatotoxicity: Clinical Characteristics and Listing Compilation. Int. J. Mol. Sci. 2016, 17, 588. https://doi.org/10.3390/ijms17050588
Frenzel C, Teschke R. Herbal Hepatotoxicity: Clinical Characteristics and Listing Compilation. International Journal of Molecular Sciences. 2016; 17(5):588. https://doi.org/10.3390/ijms17050588
Chicago/Turabian StyleFrenzel, Christian, and Rolf Teschke. 2016. "Herbal Hepatotoxicity: Clinical Characteristics and Listing Compilation" International Journal of Molecular Sciences 17, no. 5: 588. https://doi.org/10.3390/ijms17050588
APA StyleFrenzel, C., & Teschke, R. (2016). Herbal Hepatotoxicity: Clinical Characteristics and Listing Compilation. International Journal of Molecular Sciences, 17(5), 588. https://doi.org/10.3390/ijms17050588






