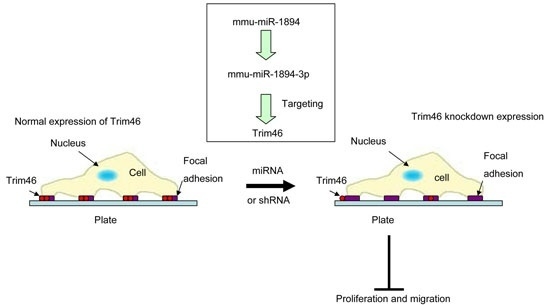
Mmu-miR-1894-3p Inhibits Cell Proliferation and Migration of Breast Cancer Cells by Targeting Trim46
Abstract
:1. Introduction
2. Results
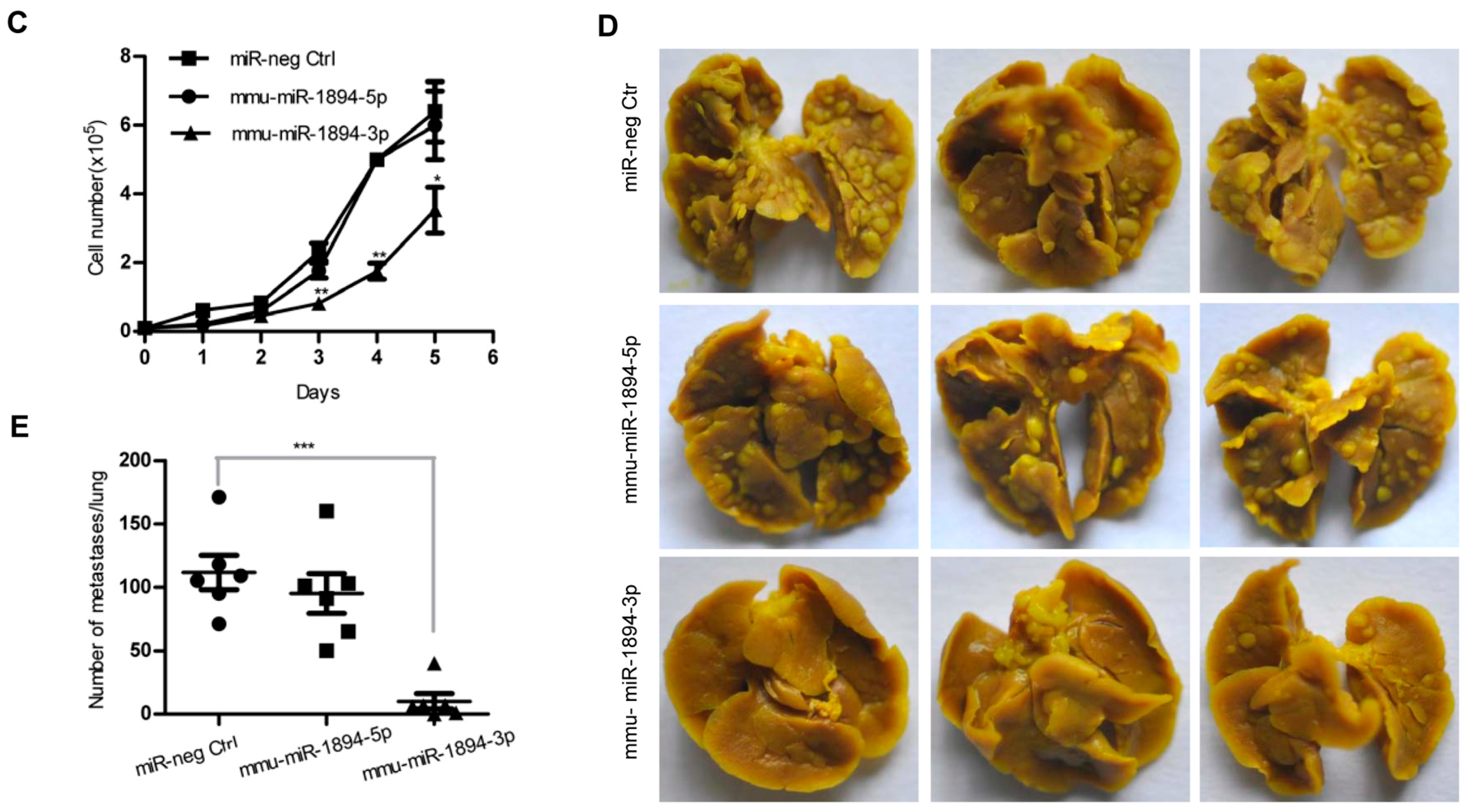
2.1. miR-1894 Is a Novel Metastasis Suppressor MicroRNA for Mouse Breast Cancer
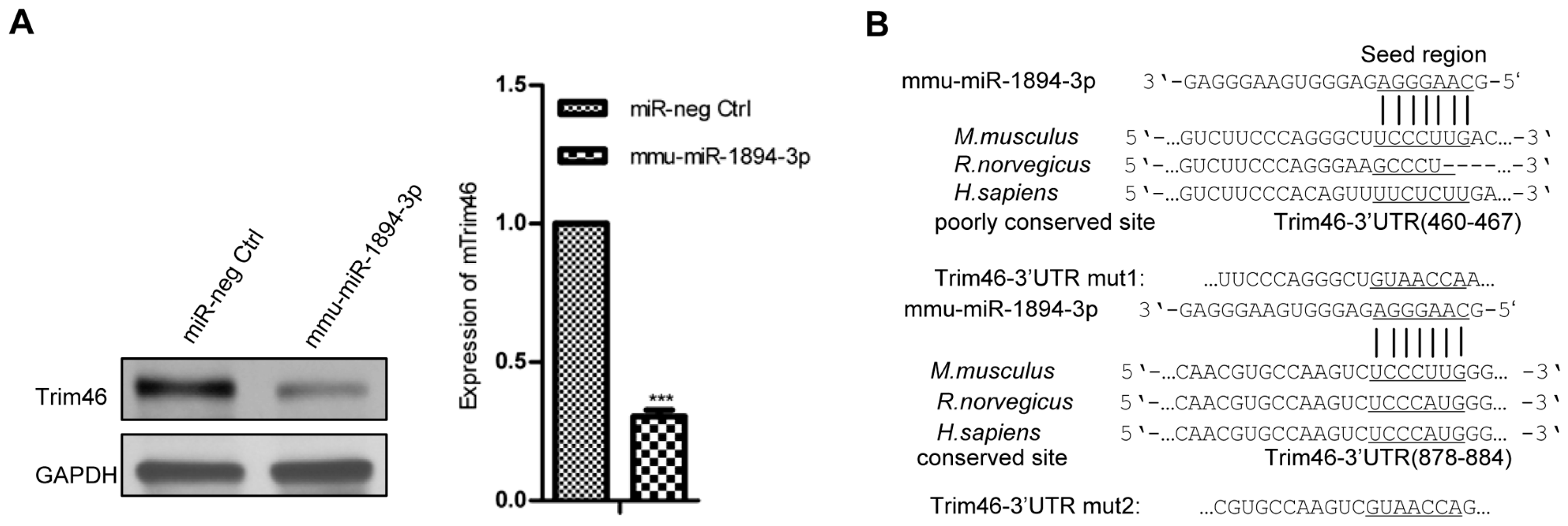
2.2. Mmu-miR-1894-3p Down-Regulated Trim46 by Binding to the Trim46 3′-Untranslated Region (UTR)
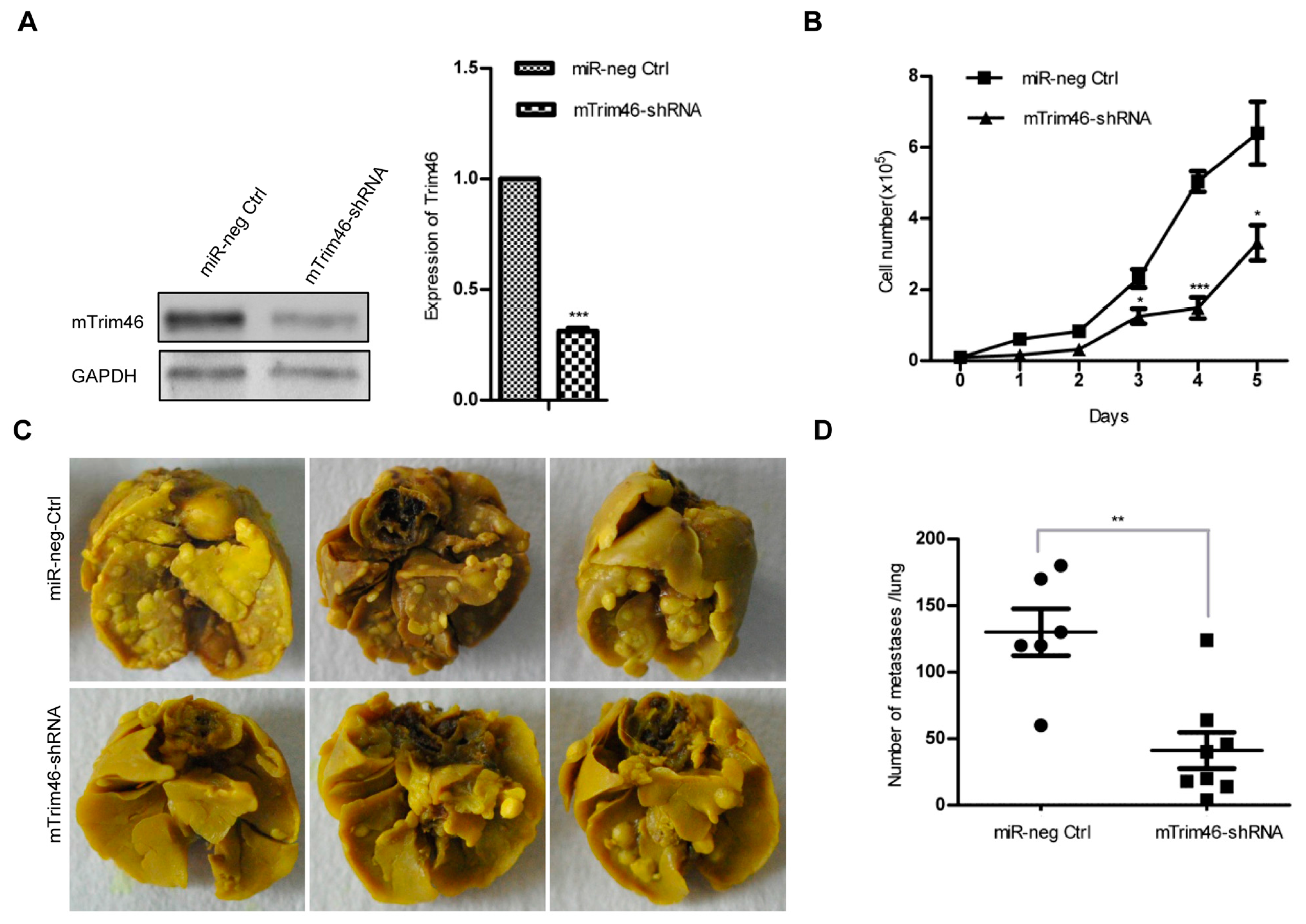
2.3. Trim46 Knockdown Inhibited Cell Proliferation and Metastasis in 4TO7 Mouse Breast Cancer Cells
2.4. Trim46 Was Involved in the Cell Proliferation and Migration of Human Breast Cancer Cells
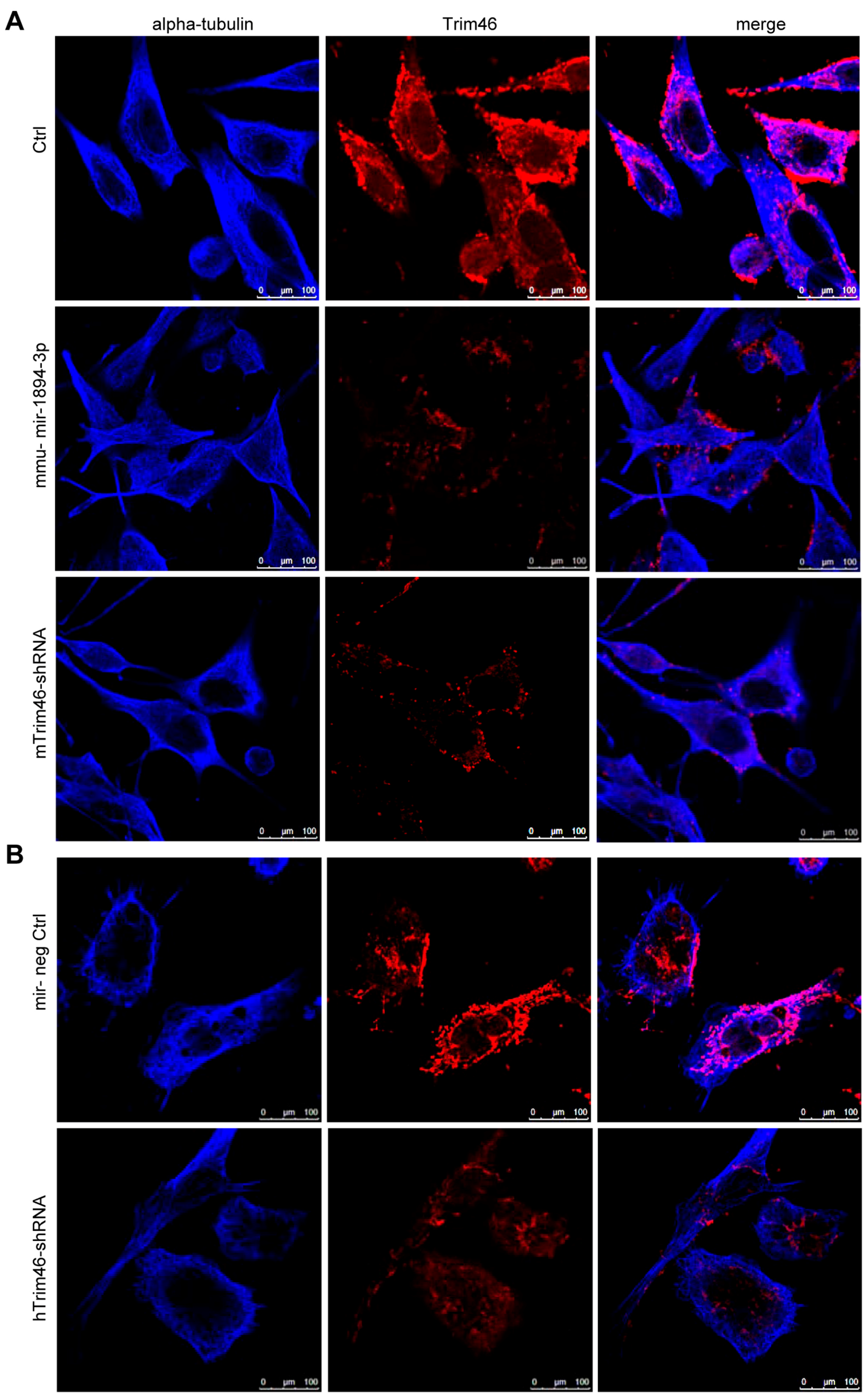
2.5. Trim46 Knockdown by Mmu-miR-1894-3p or shRNA Reduced Its Expression on the Cell Surface
3. Discussion
4. Materials and Methods
4.1. Constructs
4.2. Cell Culture
4.3. qRT-PCR
4.4. Western Blot
4.5. Target Prediction
4.6. Dual-Luciferase Reporter Assays
4.7. In vivo Metastasis Assays
4.8. RTCA Assays
4.9. Immunofluorescence
4.10. Wound Healing Assay
4.11. Statistical Analysis
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Siegel, R.; Ma, J.; Zou, Z.; Jemal, A. Cancer statistics, 2014. CA Cancer J. Clin. 2014, 64, 9–29. [Google Scholar] [PubMed]
- Helbig, G.; Christopherson, K.W., 2nd; Bhat-Nakshatri, P.; Kumar, S.; Kishimoto, H.; Miller, K.D.; Broxmeyer, H.E.; Nakshatri, H. NF-κB promotes breast cancer cell migration and metastasis by inducing the expression of the chemokine receptor CXCR4. J. Biol. Chem. 2003, 278, 21631–21638. [Google Scholar] [CrossRef] [PubMed]
- Serpico, D.; Molino, L.; di Cosimo, S. MicroRNAs in breast cancer development and treatment. Cancer Treat. Rev. 2014, 40, 595–604. [Google Scholar] [CrossRef] [PubMed]
- Shenouda, S.K.; Alahari, S.K. MicroRNA function in cancer: Oncogene or a tumor suppressor? Cancer Metastasis Rev. 2009, 28, 369–378. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Liu, J.; Wang, G. The role of microRNAs in human breast cancer progression. Tumor Biol. 2014, 35, 6235–6244. [Google Scholar] [CrossRef] [PubMed]
- Gyparaki, M.T.; Basdra, E.K.; Papavassiliou, A.G. MicroRNAs as regulatory elements in triple negative breast cancer. Cancer Lett. 2014, 354, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Sui, X.; Wang, X.; Han, W.; Li, D.; Xu, Y.; Lou, F.; Zhou, J.; Gu, X.; Zhu, J.; Zhang, C.; et al. MicroRNAs-mediated cell fate in triple negative breast cancers. Cancer Lett. 2015, 361, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Kozomara, A.; Griffiths-Jones, S. Mirbase: Annotating high confidence micrornas using deep sequencing data. Nucleic Acids Res. 2014, 42, D68–D73. [Google Scholar] [CrossRef] [PubMed]
- De Jong, M.; Maina, T. Of mice and humans: Are they the same?—Implications in cancer translational research. J. Nucl. Med. 2010, 51, 501–504. [Google Scholar] [CrossRef] [PubMed]
- HogenEsch, H.; Nikitin, A.Y. Challenges in pre-clinical testing of anti-cancer drugs in cell culture and in animal models. J. Control. Release 2012, 164, 183–186. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, K.; Seike, M.; Okano, T.; Matsuda, K.; Miyanaga, A.; Mizutani, H.; Noro, R.; Minegishi, Y.; Kubota, K.; Gemma, A. MiR-134/487b/655 cluster regulates TGF-β-induced epithelial-mesenchymal transition and drug resistance to gefitinib by targeting MAGI2 in lung adenocarcinoma cells. Mol. Cancer Ther. 2014, 13, 444–453. [Google Scholar] [CrossRef] [PubMed]
- Formosa, A.; Markert, E.K.; Lena, A.M.; Italiano, D.; Finazzi-Agro, E.; Levine, A.J.; Bernardini, S.; Garabadgiu, A.V.; Melino, G.; Candi, E. MicroRNAs, mir-154, mir-299-5p, mir-376a, mir-376c, mir-377, mir-381, mir-487b, mir-485-3p, mir-495 and mir-654-3p, mapped to the 14q32.31 locus, regulate proliferation, apoptosis, migration and invasion in metastatic prostate cancer cells. Oncogene 2014, 33, 5173–5182. [Google Scholar] [CrossRef] [PubMed]
- Krukovets, I.; Legerski, M.; Sul, P.; Stenina-Adognravi, O. Inhibition of hyperglycemia-induced angiogenesis and breast cancer tumor growth by systemic injection of microRNA-467 antagonist. FASEB J. 2015, 29, 3726–3736. [Google Scholar] [CrossRef] [PubMed]
- Persson, H.; Kvist, A.; Rego, N.; Staaf, J.; Vallon-Christersson, J.; Luts, L.; Loman, N.; Jonsson, G.; Naya, H.; Hoglund, M.; et al. Identification of new microRNAs in paired normal and tumor breast tissue suggests a dual role for the ERBB2/Her2 gene. Cancer Res. 2011, 71, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Li, L.P.; Wu, W.J.; Sun, D.Y.; Xie, Z.Y.; Ma, Y.C.; Zhao, Y.G. MiR-449a and CDK6 in gastric carcinoma. Oncol. Lett. 2014, 8, 1533–1538. [Google Scholar] [CrossRef] [PubMed]
- Lize, M.; Pilarski, S.; Dobbelstein, M. E2F1-inducible microRNA 449a/b suppresses cell proliferation and promotes apoptosis. Cell Death Differ. 2010, 17, 452–458. [Google Scholar] [CrossRef] [PubMed]
- Luo, W.T.; Huang, B.; Li, Z.X.; Li, H.Y.; Sun, L.M.; Zhang, Q.F.; Qiu, X.S.; Wang, E.H. MicroRNA-449a is downregulated in non-small cell lung cancer and inhibits migration and invasion by targeting c-Met. PLoS ONE 2013, 8, e54759. [Google Scholar] [CrossRef] [PubMed]
- Noonan, E.J.; Place, R.F.; Basak, S.; Pookot, D.; Li, L.C. MiR-449a causes Rb-dependent cell cycle arrest and senescence in prostate cancer cells. Oncotarget 2010, 1, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Ye, W.W.; Xue, J.S.; Zhang, Q.; Li, F.Y.; Zhang, W.; Chen, H.J.; Huang, Y.B.; Zheng, F.Y. MiR-449a functions as a tumor suppressor in endometrial cancer by targeting CDC25A. Oncol. Rep. 2014, 32, 1193–1199. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Song, Y.; Wang, Z.; Yue, Z.; Xu, H.; Xing, C.; Liu, Z. Altered expression of miR-148a and miR-152 in gastrointestinal cancers and its clinical significance. J. Gastrointest. Surg. 2010, 14, 1170–1179. [Google Scholar] [CrossRef] [PubMed]
- Nohata, N.; Hanazawa, T.; Enokida, H.; Seki, N. MicroRNA-1/133a and microRNA-206/133b clusters: Dysregulation and functional roles in human cancers. Oncotarget 2012, 3, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Benito, M.; Conchillo, A.; Garcia, M.A.; Vazquez, I.; Maicas, M.; Vicente, C.; Cristobal, I.; Marcotegui, N.; Garcia-Orti, L.; Bandres, E.; et al. Evi1 controls proliferation in acute myeloid leukaemia through modulation of miR-1-2. Br. J. Cancer 2010, 103, 1292–1296. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Martin, E.C.; Elliott, S.; Rhodes, L.V.; Antoon, J.W.; Fewell, C.; Zhu, Y.; Driver, J.L.; Jodari-Karimi, M.; Taylor, C.W.; Flemington, E.K.; et al. Preferential star strand biogenesis of pre-miR-24-2 targets PKC-α and suppresses cell survival in MCF-7 breast cancer cells. Mol. Carcinog. 2014, 53, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Huppi, K.; Pitt, J.; Wahlberg, B.; Caplen, N.J. Genomic instability and mouse microRNAs. Toxicol. Mech. Methods 2011, 21, 325–333. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, C.M.; Pestova, T.V.; Hellen, C.U.; Wagner, G. Structure and interactions of the translation initiation factor eIF1. EMBO J. 1999, 18, 2631–2637. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Chao, F.; Huang, B.; Liu, D.; Kim, J.; Huang, S. HOXC8 promotes breast tumorigenesis by transcriptionally facilitating cadherin-11 expression. Oncotarget 2014, 5, 2596–2607. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.K.; Lee, D.S.; Park, J.C. Nuclear factor I–C regulates E-cadherin via control of KLF4 in breast cancer. BMC Cancer 2015, 15, 113. [Google Scholar] [CrossRef] [PubMed]
- Vanharanta, S.; Marney, C.B.; Shu, W.P.; Valiente, M.; Zou, Y.L.; Mele, A.; Darnell, R.B.; Massague, J. Loss of the multifunctional RNA-binding protein RBM47 as a source of selectable metastatic traits in breast cancer. Elife 2014, 3. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.L.; Wang, Y.Y.; Lu, X.H.; Zhao, Z.L.; Zhu, L.H.; Chen, S.L.; Wu, Q.; Chen, C.J.; Wang, Z.W. miR-125b regulates epithelial-mesenchymal transition via targeting Sema4C in paclitaxel-resistant breast cancer cells. Oncotarget 2015, 6, 3268–3279. [Google Scholar] [CrossRef] [PubMed]
- Mitrovic, S.; Ben-Tekaya, H.; Koegler, E.; Gruenberg, J.; Hauri, H.P. The cargo receptors Surf4, endoplasmic reticulum-Golgi intermediate compartment (ERGIC)-53, and p25 are required to maintain the architecture of ERGIC and Golgi. Mol. Biol. Cell 2008, 19, 1976–1990. [Google Scholar] [CrossRef] [PubMed]
- Cambiaghi, V.; Giuliani, V.; Lombardi, S.; Marinelli, C.; Toffalorio, F.; Pelicci, P.G. TRIM proteins in cancer. Adv. Exp. Med. Biol. 2012, 770, 77–91. [Google Scholar] [PubMed]
- Ren, X.S.; Yin, M.H.; Zhang, X.; Wang, Z.; Feng, S.P.; Wang, G.X.; Luo, Y.J.; Liang, P.Z.; Yang, X.Q.; He, J.X.; et al. Tumor-suppressive microRNA-449a induces growth arrest and senescence by targeting E2F3 in human lung cancer cells. Cancer Lett. 2014, 344, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.Q.; Chen, Q.Y.; Qin, R.; Zhang, K.F.; Li, H. MicroRNA-449a reduces cell survival and enhances cisplatin-induced cytotoxicity via downregulation of NOTCH1 in ovarian cancer cells. Tumor Biol. 2014, 35, 12369–12378. [Google Scholar] [CrossRef] [PubMed]
- Vos, H.L.; Mockensturmwilson, M.; Rood, P.M.L.; Maas, A.M.C.E.; Duhig, T.; Gendler, S.J.; Bornstein, P. A tightly organized, conserved gene-cluster on mouse chromosome-3 (E3-F1). Mamm. Genome 1995, 6, 820–822. [Google Scholar] [CrossRef] [PubMed]
- Torok, M.; Etkin, L.D. Two B or not two B? Overview of the rapidly expanding B-box family of proteins. Differentiation 2001, 67, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Petrera, F.; Meroni, G. TRIM proteins in development. TRIM/RBCC Proteins 2012, 770, 131–141. [Google Scholar]
- Short, K.M.; Cox, T.C. Subclassification of the RBCC/TRIM superfamily reveals a novel motif necessary for microtubule binding. J. Biol. Chem. 2006, 281, 8970–8980. [Google Scholar] [CrossRef] [PubMed]
- Napolitano, L.M.; Meroni, G. TRIM family: Pleiotropy and diversification through homomultimer and heteromultimer formation. IUBMB Life 2012, 64, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Cox, T.C. The microtubule-associated C–I subfamily of TRIM proteins and the regulation of polarized cell responses. TRIM/RBCC Proteins 2012, 770, 105–118. [Google Scholar]
- Kitamura, K.; Tanaka, H.; Nishimune, Y. Haprin, a novel haploid germ cell-specific ring finger protein involved in the acrosome reaction. J. Biol. Chem. 2003, 278, 44417–44423. [Google Scholar] [CrossRef] [PubMed]
- Yoshigai, E.; Kawamura, S.; Kuhara, S.; Tashiro, K. TRIM36/haprin plays a critical role in the arrangement of somites during xenopus embryogenesis. Biochem. Biophys. Res. Commun. 2009, 378, 428–432. [Google Scholar] [CrossRef] [PubMed]
- Martin, S.K.; Kamelgarn, M.; Kyprianou, N. Cytoskeleton targeting value in prostate cancer treatment. Am. J. Clin. Exp. Urol. 2014, 2, 15–26. [Google Scholar] [PubMed]
- Oskarsson, T. Extracellular matrix components in breast cancer progression and metastasis. Breast 2013, 22 (Suppl. 2), S66–S72. [Google Scholar] [CrossRef] [PubMed]
- Uchil, P.D.; Pawliczek, T.; Reynolds, T.D.; Ding, S.Y.; Hinz, A.; Munro, J.B.; Huang, F.; Floyd, R.W.; Yang, H.T.; Hamilton, W.L.; et al. TRIM15 is a focal adhesion protein that regulates focal adhesion disassembly. J. Cell Sci. 2014, 127, 3928–3942. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Lv, W.; Zhang, J.H.; Lu, D.L. MiR-96 functions as a tumor suppressor gene by targeting NUAK1 in pancreatic cancer. Int. J. Mol. Med. 2014, 34, 1599–1605. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Yu, J.; Pan, X.; Li, Z.; Chen, Z.; Zhang, W.; Wang, B.; Yang, L.; Xu, H.; Zhang, G.; et al. HERG1 functions as an oncogene in pancreatic cancer and is downregulated by miR-96. Oncotarget 2014, 5, 5832–5844. [Google Scholar] [CrossRef] [PubMed]
- Sethupathy, P.; Megraw, M.; Hatzigeorgiou, A.G. A guide through present computational approaches for the identification of mammalian microRNA targets. Nat. Methods 2006, 3, 881–886. [Google Scholar] [CrossRef] [PubMed]



© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, L.; Li, X.; Dong, W.; Sun, C.; Guo, D.; Zhang, L. Mmu-miR-1894-3p Inhibits Cell Proliferation and Migration of Breast Cancer Cells by Targeting Trim46. Int. J. Mol. Sci. 2016, 17, 609. https://doi.org/10.3390/ijms17040609
Zhang L, Li X, Dong W, Sun C, Guo D, Zhang L. Mmu-miR-1894-3p Inhibits Cell Proliferation and Migration of Breast Cancer Cells by Targeting Trim46. International Journal of Molecular Sciences. 2016; 17(4):609. https://doi.org/10.3390/ijms17040609
Chicago/Turabian StyleZhang, Li, Xiaoying Li, Wei Dong, Caixian Sun, Deyu Guo, and Lianfeng Zhang. 2016. "Mmu-miR-1894-3p Inhibits Cell Proliferation and Migration of Breast Cancer Cells by Targeting Trim46" International Journal of Molecular Sciences 17, no. 4: 609. https://doi.org/10.3390/ijms17040609
APA StyleZhang, L., Li, X., Dong, W., Sun, C., Guo, D., & Zhang, L. (2016). Mmu-miR-1894-3p Inhibits Cell Proliferation and Migration of Breast Cancer Cells by Targeting Trim46. International Journal of Molecular Sciences, 17(4), 609. https://doi.org/10.3390/ijms17040609