3.1. Methods
The melting points were determined using a Mikroheiztisch Boetius (VEB Wägetechnik, Radebeul, DDR). Elemental analyses were carried out on an Elementar Vario EL III analyzer (elementar Analysensysteme GmbH, Hanau, Germany). Infrared spectra were recorded on a Perkin-Elmer type 16PC FT-IR spectrophotometer (Perkin-Elmer GmbH, Überlingen, GErmany) between 4000 and 400 cm
−1 as KBr-pellets (reference KBr). The measurements of the magnetic susceptibility were performed with a Magnetic Susceptibility Balance-Auto from Johnson Matthey GmbH (Matthey GmbH, Cambridge, UK) at room temperature, for diamagnetic correction the increment system of Pascal and Pacault [
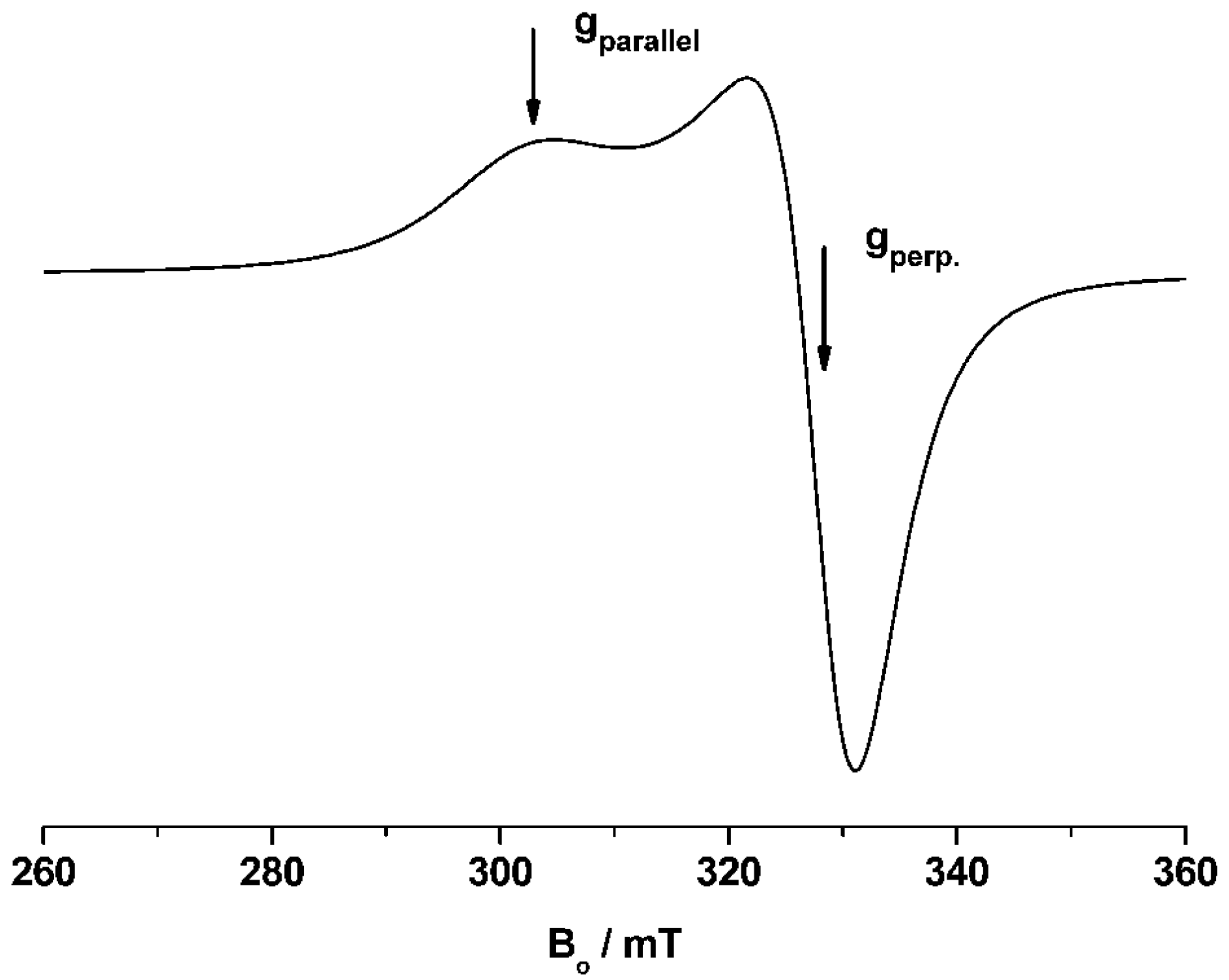
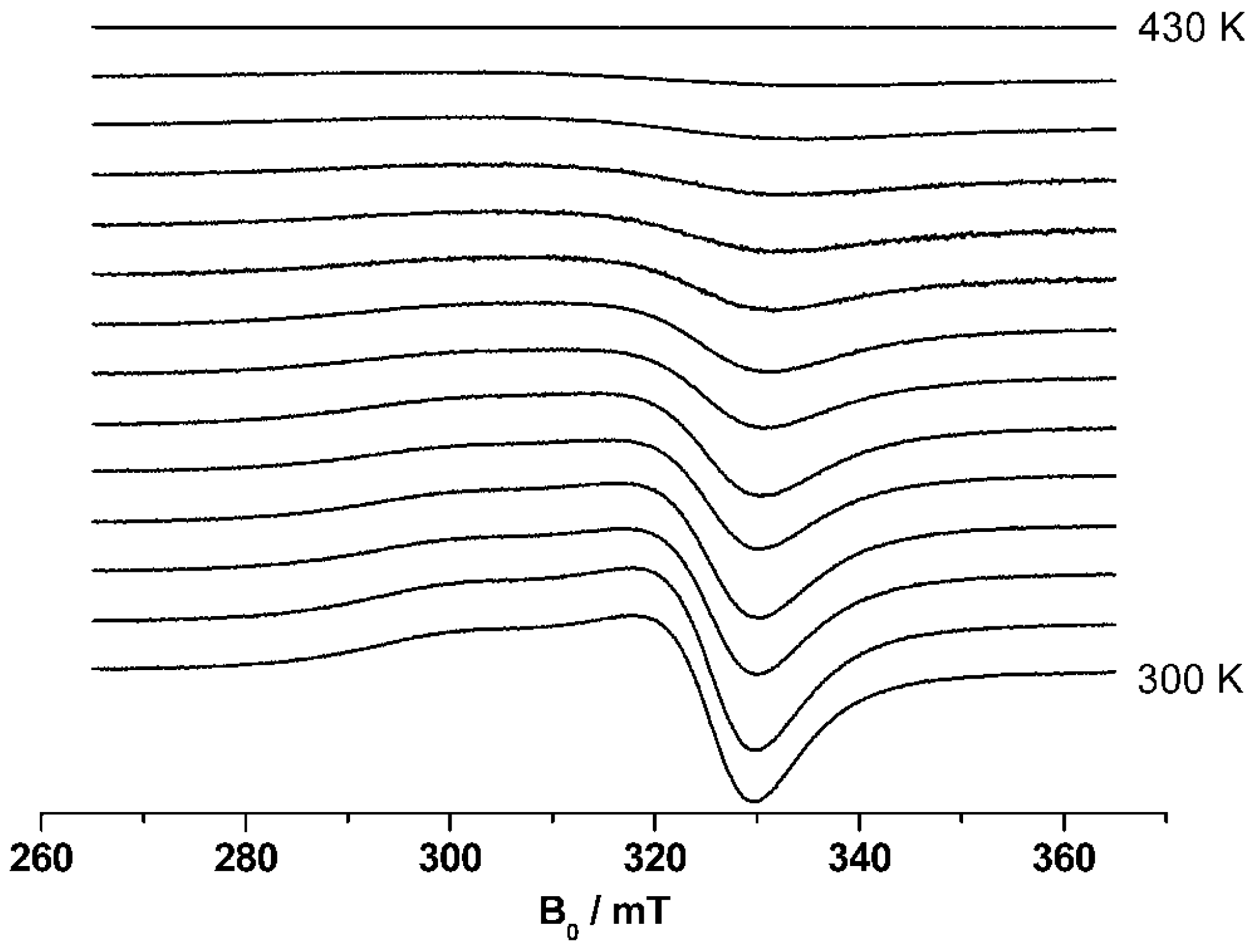
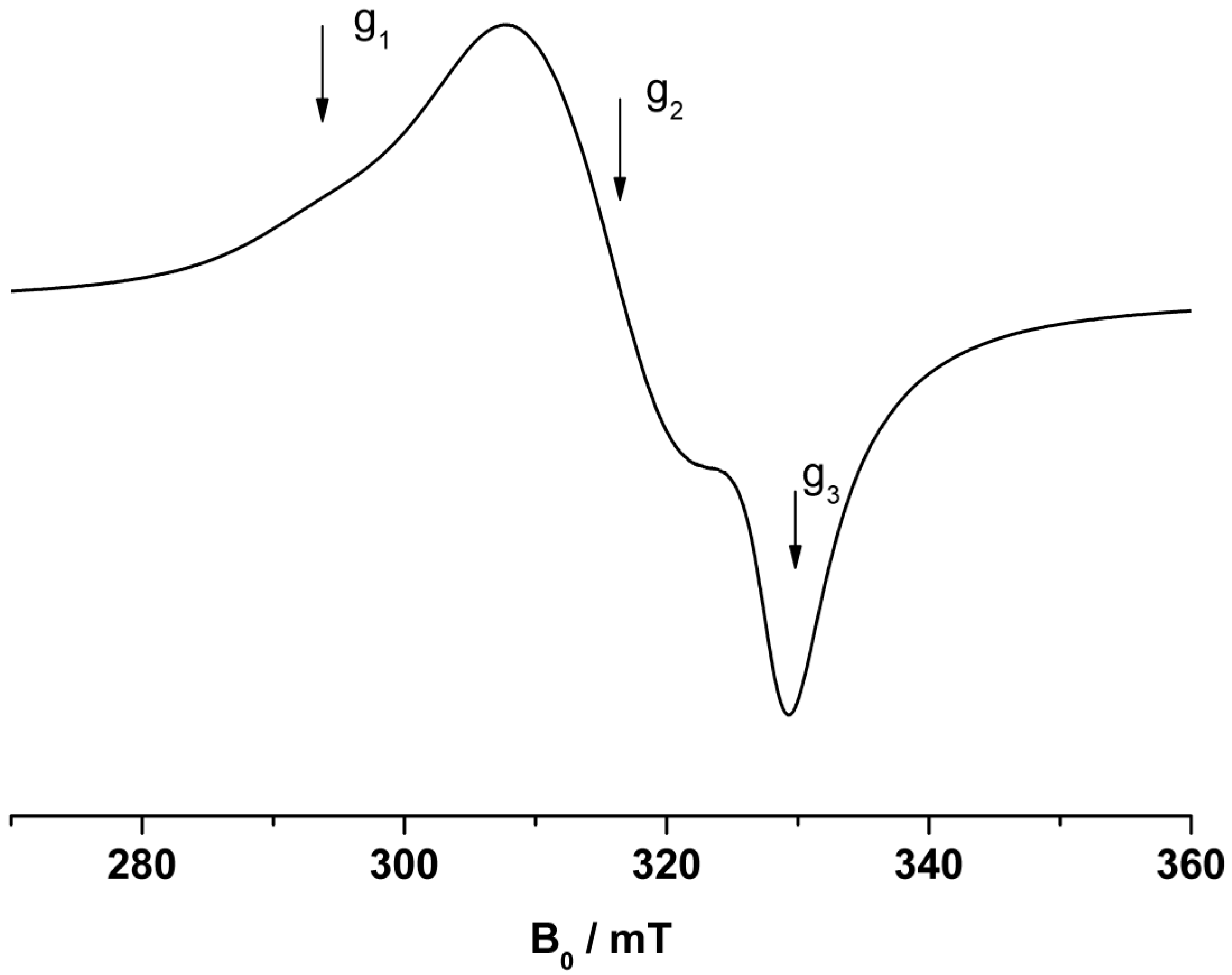
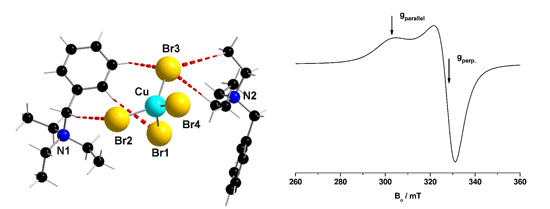
38] was applied. EPR spectra were recorded at 9.4 GHz (X-band) using a Bruker CW Elexsys E 500 spectrometer (Bruker BioSpin GmbH, Rheinstetten, Germany)
For X-ray structure determinations, the crystals were embedded in perfluoropolyalkylether oil and mounted on a glass fibre (
5) or within a MicroGripper (
8). For structure analysis of (
5), the intensity data were collected at 210 K using an Imaging Plate Diffraction System IPDS-2 (Stoe, Darmstadt, Germany) with graphite monochromatized Mo-
Kα radiation (λ = 0.71073 Å) at 50 kV and 40 mA. The data collection for (
8) was performed on a StadiVari diffractometer (Stoe, Darmstadt, Germany) equipped with a four-circle goniometer (open Eulerian cradle), a Genix Microfocus X-ray source (Mo) with a graded multilayer mirror and a Pilatus 200 K detector (Dectris, Baden-Daettwil, Switzerland). The data were corrected for absorption as well as for Lorentz polarization and extinction effects using the program X-Area (Stoe, 2004) [
39]. The structures were solved by direct methods using SHELXS-2013/1 [
40] and refined by full-matrix least squares on
F2 using the program SHELXL-2014/7 [
41]. All non-hydrogen atoms were refined anisotropically. The hydrogen atoms were calculated in their expected positions and refined with a riding model. CCDC 1459578 (
5) and CCDC 1459612 (
8) contain the supplementary crystallographic data for this paper. These data are provided free of charge by The Cambridge Crystallographic Centre (Cambridge, UK).
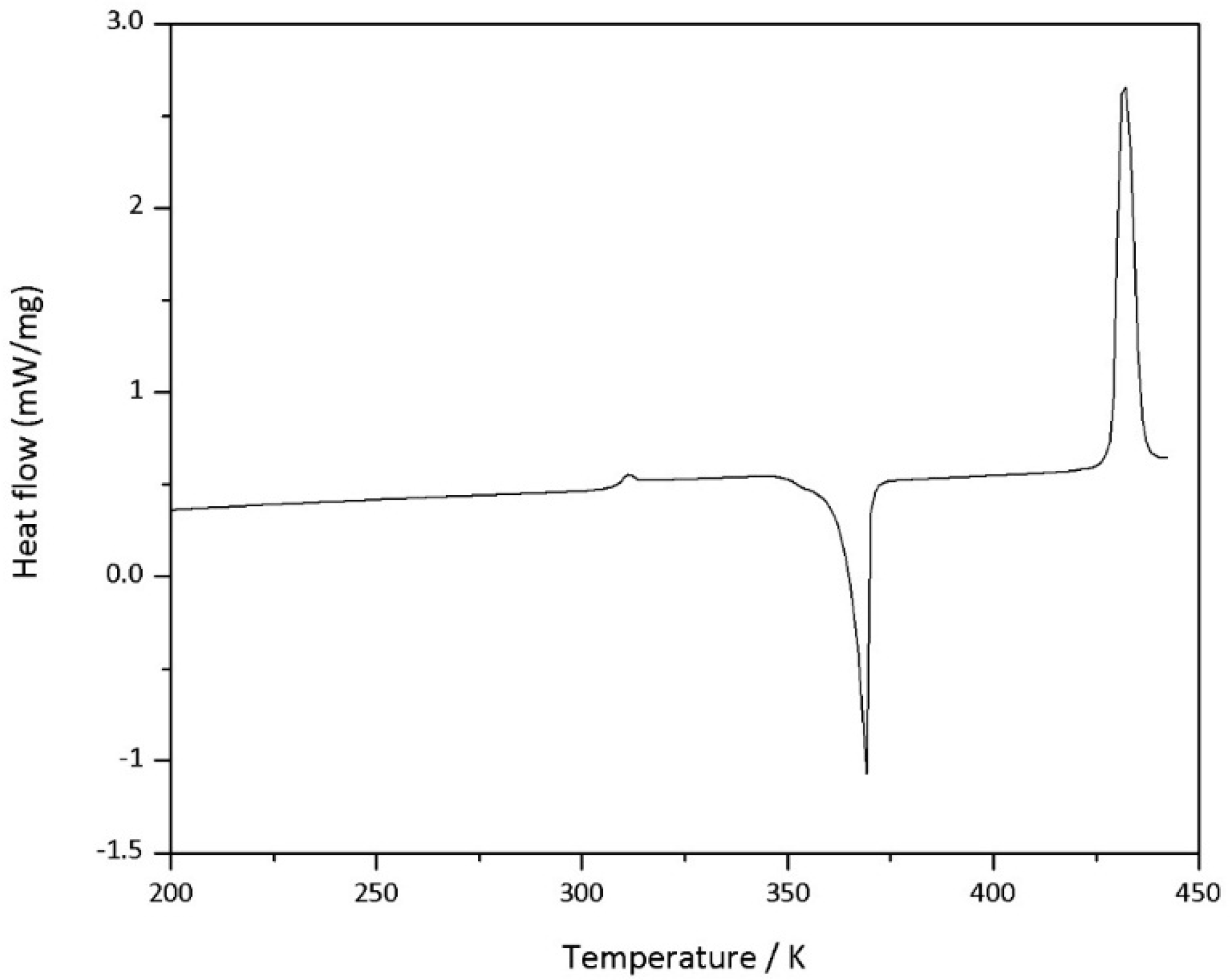
Differential scanning calorimetry (DSC) measurements were performed with a DSC 214 Polyma (Netzsch GmbH & Co. KG, Selb, Germany) by NETZSCH operating with a scan rate of 5–10 °C·min−1 under a nitrogen flow.
3.3. Syntheses
3.3.1. General Preparation
In general, tetrabromidocuprate(II) complexes can be achieved by different procedures [
42,
43,
44]. In the current work, the [CuBr
4]
2− moiety was synthesized according to a protocol by N. S. Gill and R. S. Nyholm [
44]: an ethanolic solution of a stoichiometric amount of CuBr
2 was added to the respective bromide salt of the cation dissolved in a minimum volume of ethanol. The reaction mixture was stirred for one hour at room temperature. The product was precipitated by evaporating the solvent.
3.3.2. Bis(tetraethylammonium)tetrabromidocuprate(II), (Et4N)2[CuBr4] (1)
Compound (
1) was synthesized according to an already published protocol [
36].
A solution of 1.5 mmol (0.32 g) of tetraethylammonium bromide in 3 mL of ethanol was mixed with a solution of 0.5 mmol (0.11 g) of copper(II) bromide in 10 mL ethanol. The solution was stirred one hour at room temperature. The solvent was removed and a violet powder was received, filtered off and dried.
Melting point: 241–242 °C. Yield: 0.22 g (67%). Elemental analysis calculated for C16H40N2CuBr4 (643.66): C 29.85, H 6.26, N 4.35 (%); found: C 29.80, H 6.20, N 4.34 (%). IR (KBr, cm−1): 3443 s, 2976 m, 2921 m, 2852 wm, 1628 wm, 1478 s, 1402 m, 1307 w, 1183 m, 1032 m, 1006 m, 793 m.
(IR: s = strong, ms = medium strong, m = medium, wm = weak medium, w = weak).
3.3.3. Bis(tetrabutylphosphonium)tetrabromidocuprate(II), (Bu4P)2[CuBr4] (2)
Solutions of 0.5 mmol (0.11 g) copper(II) bromide and 1.0 mmol (0.32 g) tetrabutylphosphonium bromide, each dissolved in 2 mL ethanol, were combined and stirred at room temperature for 1 h. The resulting violet precipitate is filtered off and dried.
Melting point: 49–52 °C. Yield: 0.38 g (84%). Elemental analysis calculated for C32H72P2CuBr4 (902.13): C 42.60, H 8.04 (%), found: C 42.08, H 8.16 (%). IR (KBr, cm−1): 2928 s, 2891 ms, 2870 ms, 1463 ms, 1407 m, 1375 m, 1237 w, 1098 m, 968 w, 920 m, 722 m. μeff = 1.5 B.M.
3.3.4. Bis(tetrahexylammonium)tetrabromidocuprate(II), (Hex4N)2[CuBr4] (3)
A solution of 1.0 mmol (0.22 g) CuBr2 in 5 mL ethanol was added to a solution of 2.0 mmol (0.87 g) tetrahexylammonium bromide in 5 mL ethanol. The mixed solution was stirred at room temperature for 1 h. The resulting violet precipitate was filtered off and dried.
Melting point: 93–95 °C. Yield: 0.58 g (53%). Elemental analysis calculated for C48H104N2CuBr4 (1092.51): C 52.77, H 9.60, N 2.56 (%), found: C 53.06, H 9.70, N 2.67 (%). IR (KBr, cm−1): 2928 s, 2553 m, 1634 w, 1480 m, 1383 mw, 1050 w, 728 w. μeff = 1.5 B.M.
3.3.5. Bis(benzyltrimethylammonium)tetrabromidocuprate(II), (BzlMe3N)2[CuBr4] (4)
Compound (
4) was also synthesized according to an already published procedure [
37].
To a solution of 0.5 mmol (0.11 g) of CuBr2 in 3.5 mL methanol a solution of 1.0 mmol (0.23 g) benzyltrimethylammonium bromide, dissolved in 1.5 mL methanol, was added. The mixture was stirred for one hour at room temperature. After a short while the complex precipitated as purple crystals.
Melting point: 173–175 °C. Yield: 0.35 g (72%). Elemental analysis calculated for C20H32N2CuBr4 (969.51): C 35.16, H 4.72, N 4.10 (%); found: C 35.09, H 4.71, N 4.17 (%). IR (KBr, cm−1): 3017 m, 1585 w, 1485 s, 1476 s, 1411 m, 1218 m, 989 m, 974 m, 887 s, 780 s, 703 s. μeff = 1.4 B.M.
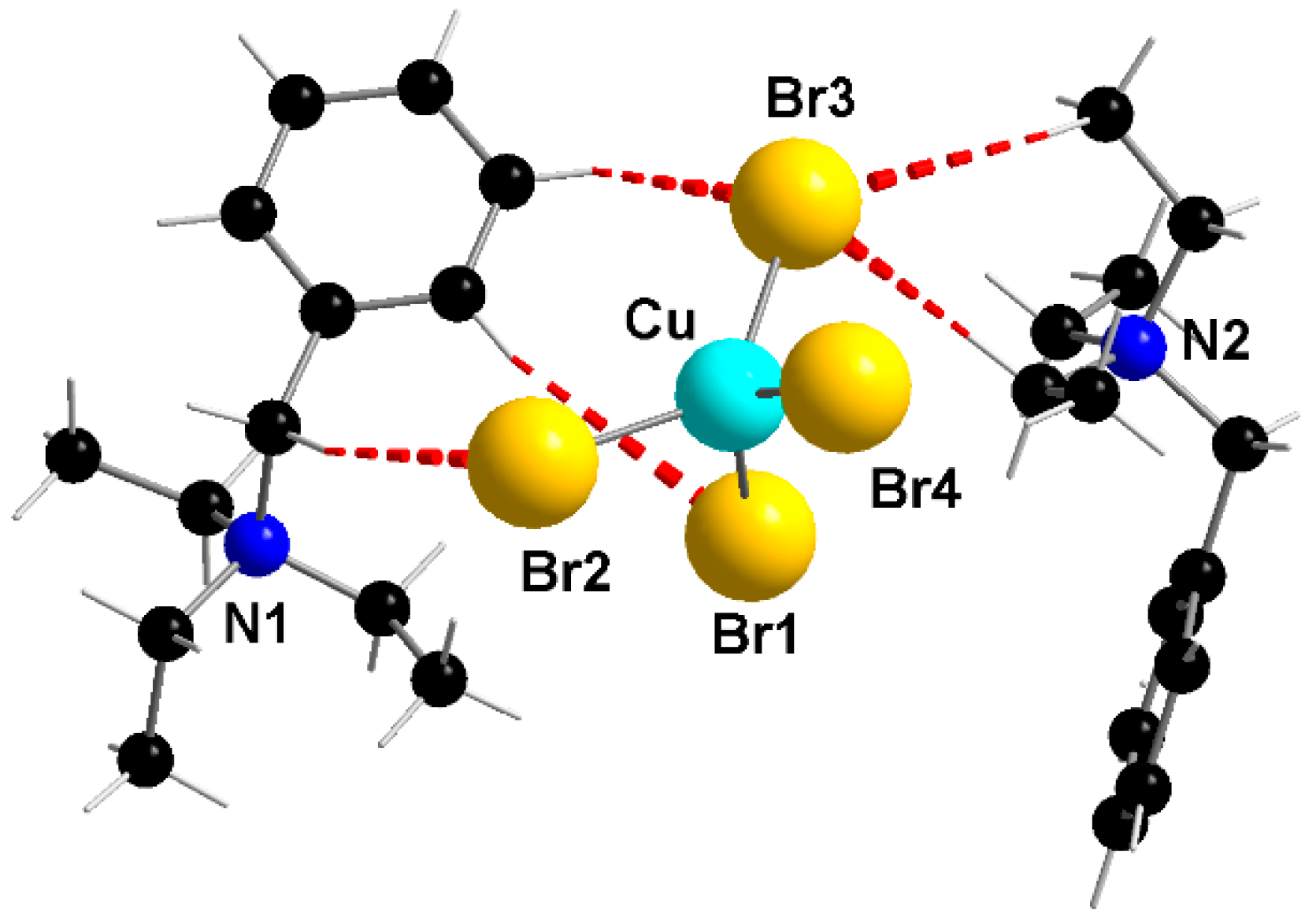
3.3.6. Bis(benzyltriethylammonium)tetrabromidocuprate(II), (BzlEt3N)2[CuBr4] (5)
0.5 mmol (0.11 g) of CuBr2 dissolved in 3 mL HBr was heated under reflux for 0.5 h. To this copper solution a solution of 1.0 mmol (0.27 g) BzlEt3NBr and 2 mL methanol was slowly added. The mixture was stirred for one hour at room temperature. The solvent was reduced and purple crystals were obtained by covering the remaining solution with N-hexane.
Melting point: 110–112 °C. Yield: 0.21 g (54%). Elemental analysis calculated for C26H44N2CuBr4 (767.7): C 40.67, H 5.78, N 3.65 (%); found: C 40.56, H 5.76, N 3.69 (%). IR (KBr, cm−1): 2983 m, 1583 w, 1450 s, 1402 ms, 1372 w, 1171 w, 1154 m, 1027 m, 1005 m, 787 m,756 s, 705 s. μeff = 1.6 B.M.
3.3.7. Bis(benzyltributylammonium)tetrabromidocuprate(II), (BzlBu3N)2[CuBr4] (6)
A solution of 1.0 mmol (0.22 g) CuBr2 in 5 mL ethanol was added to a solution of 2.0 mmol (0.71 g) benzyltributylammonium bromide in 5 mL ethanol. The mixed solution was stirred at room temperature for 1 h. The resulting violet precipitate was filtered off and dried.
Melting point: 58–60 °C. Yield: 0.59 g (63%). Elemental analysis calculated for C38H68N2CuBr4 (936.12): C 48.75, H 7.32, N 2.99 (%), found: C 48.90, H 7.48, N 3.12 (%). IR (KBr, cm−1): 2959 s, 2871 m, 1654 w, 1561 w, 1474 m, 1458 m, 1380 mw, 1212 w, 869 mw, 725 m, 704 m. μeff = 1.5 B.M.
3.3.8. Bis(ethyltriphenylphosphonium)tetrabromidocuprate(II), (EtPh3P)2[CuBr4] (7)
The synthesis of (EtPh3P)2[CuBr4] as follows: 0.5 mmol (0.11 g) copper(II) bromide and 1.0 mmol (0.37 g) EtPh3PBr solved even in 2 mL ethanol. The combined solutions are stirred for one hour at room temperature. A violet powder was obtained.
Melting point: 135–137 °C. Yield: 0.23 g (48%). Elemental analysis calculated for C40H40P2CuBr4 (964.90): C 49.74, H 4.17 (%), found: C 49.74, H 4.19 (%). IR (KBr, cm−1): 2922 m, 1586 m, 1483 m, 1438 s, 1113 s, 996 m, 780 w, 739 s, 691 s, 531 s, 510 s, 482 m. μeff = 1.6 B.M.
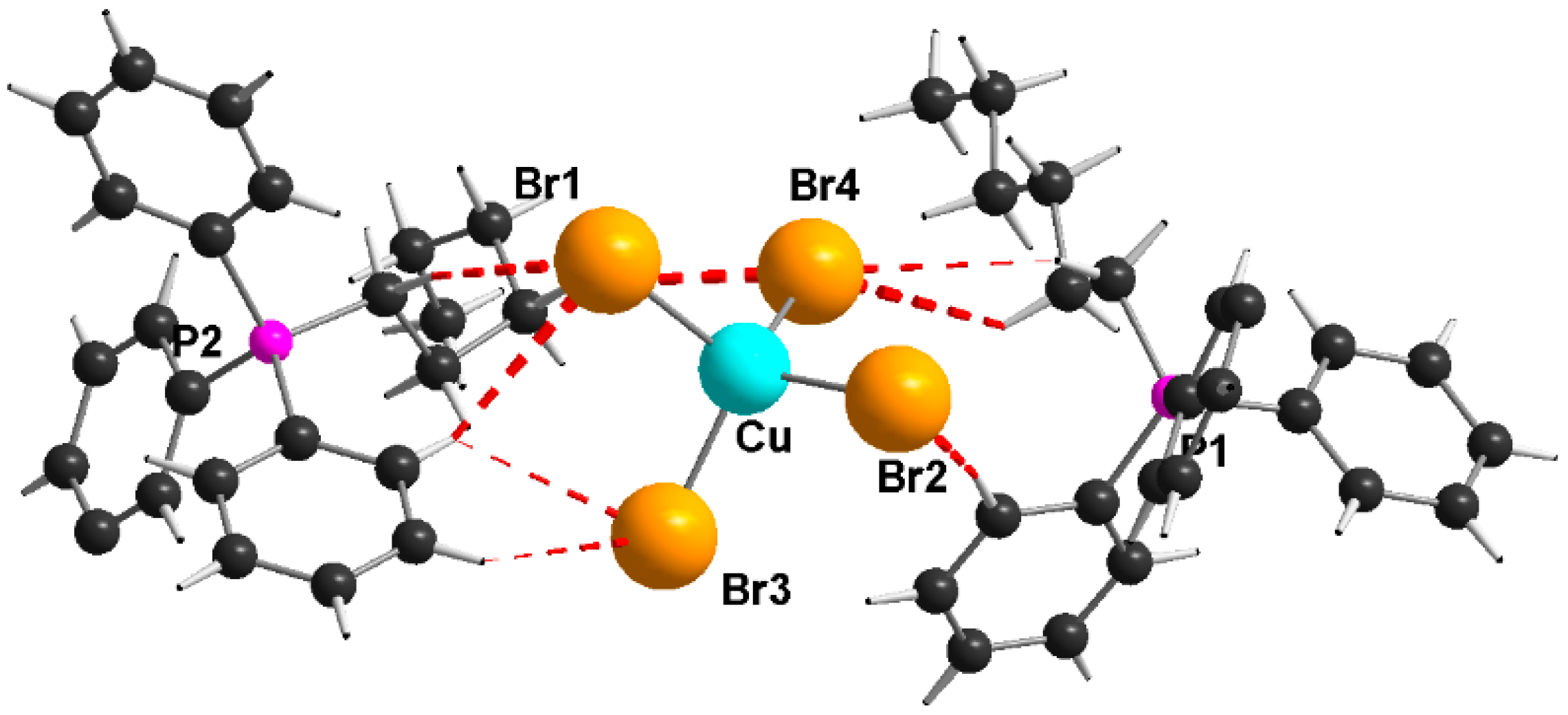
3.3.9. Bis(hexyltriphenylphosphonium)tetrabromidocuprate(II), (HexPh3P)2[CuBr4] (8)
A solution of 1.0 mmol (0.22 g) copper(II) bromide in 5 mL ethanol is added to 2.0 mmol (0.85 g) hexyltriphenylphosphonium bromide dissolved in 5 mL ethanol. The solution was stirred for 1 h at room temperature. The formed violet precipitate was filtered off and dried. Purple crystals were obtained by covering the remaining solution with n-hexane for slowly interdiffusion.
Melting point: 102–103 °C. Yield: 0.65 g (60%). Elemental analysis calculated for C48H56P2CuBr4 (1078.04): C 53.47, H 5.24 (%), found: C 53.05, H 4.97 (%). IR (KBr, cm−1): 2956 m, 2923 m, 2758 m, 1585 w, 1485 w, 1436 s, 1113 s, 724 s, 689 s, 533s, 498 ms. μeff = 1.6 B.M.
3.3.10. Bis(dodecyltrimethylammonium)tetrabromidocuprate(II), (C12H25Me3N)2[CuBr4] (9)
A solution of 1.0 mmol (0.22 g) of CuBr2 in 5 mL ethanol and a solution of 2.0 mmol (0.62 g) C12H25Me3NBr in 5 mL ethanol were combined and stirred at room temperature for 1 h. The resulting violet precipitate was filtered off and dried.
Melting point: 94–95 °C. Yield: 0.56 g (85%). Elemental analysis calculated for C30H48N2CuBr4 (662.23): C 42.89, H 8.16, N 3.34 (%), found: C 43.19, H 8.23, N 3.37 (%). IR (KBr, cm−1): 2922 ms, 2851 m, 1632 w, 1469 s, 966 ms, 909 m, 722 m. μeff = 1.5 B.M.
3.3.11. Bis(hexadecylpyridinium)tetrabromidocuprate(II), (C16-py)2[CuBr4] (10)
A solution of 0.5 mmol (0.11 g) copper(II) bromide in 10 mL ethanol is added to 1.0 mmol (0.40 g) hexadecylpyridinium bromide dissolved in 3 mL ethanol. The solution was stirred for 1 h at room temperature. The obtained violet precipitate was filtered off and dried.
Melting point: 73–74 °C. Yield: 0.15 g (30%). Elemental analysis calculated for C42H76N2CuBr4 (992.23): C 50.84, H 7.72, N 2.82 (%), found: C 47.67, H 7.20, N 2.95 (%). IR (KBr, cm−1): 3446 wm, 3053 w, 2918 s, 2850 m, 1633 wm, 1485 m, 1468 wm, 1175 w, 768 w, 721 w, 679 w. μeff = 1.6 B.M.
3.3.12. Bis(dihexadecyldimethylammonium)tetrabromidocuprate(II), (C16H33)2Me2N)2[CuBr4] (11)
A solution of 1.0 mmol (0.58 g) dihexadecyldimethylammonium bromide in 3 mL ethanol was slowly added to a solution of 0.5 mmol (0.11 g) copper(II) bromide in 3 mL ethanol. The mixture was stirred at room temperature for 1 h. The formed violet precipitate was filtered off and dried.
Melting point: 36–38 °C. Yield: 0.67 g (98%). Elemental analysis calculated for C68H144N2CuBr4 (1373.05): C 59.48, H 10.57, N 2.04 (%), found: C 59.11, H 10.89, N 2.15 (%). IR (KBr, cm−1): 2919 m, 2851 m, 1628 w, 1469 s, 1376 w, 1054 w, 990 w, 968 w, 878 w, 718 m.