Physical Characteristics of the Leaves and Latex of Papaya Plants Infected with the Papaya meleira Virus
Abstract
:1. Introduction
2. Results
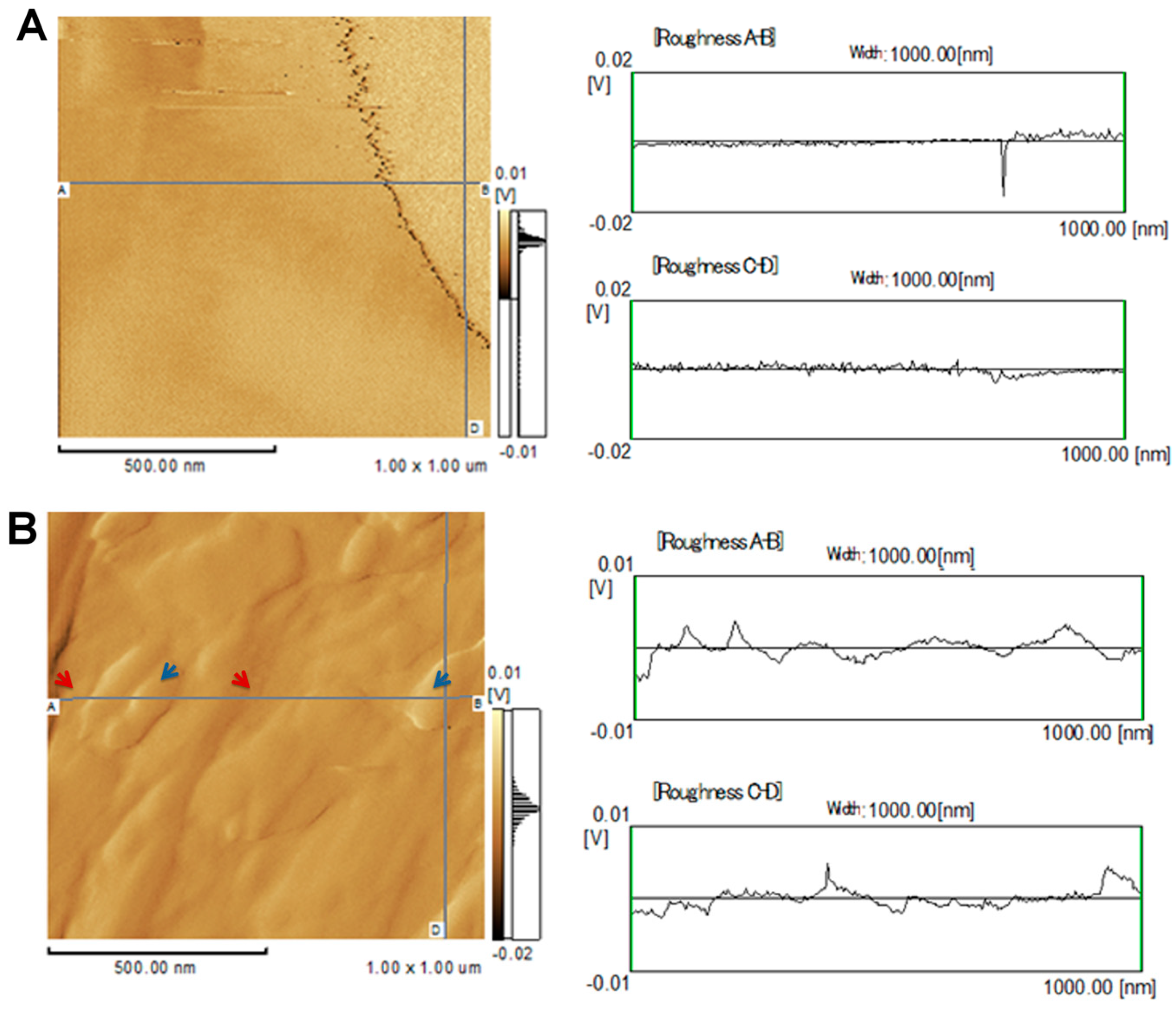
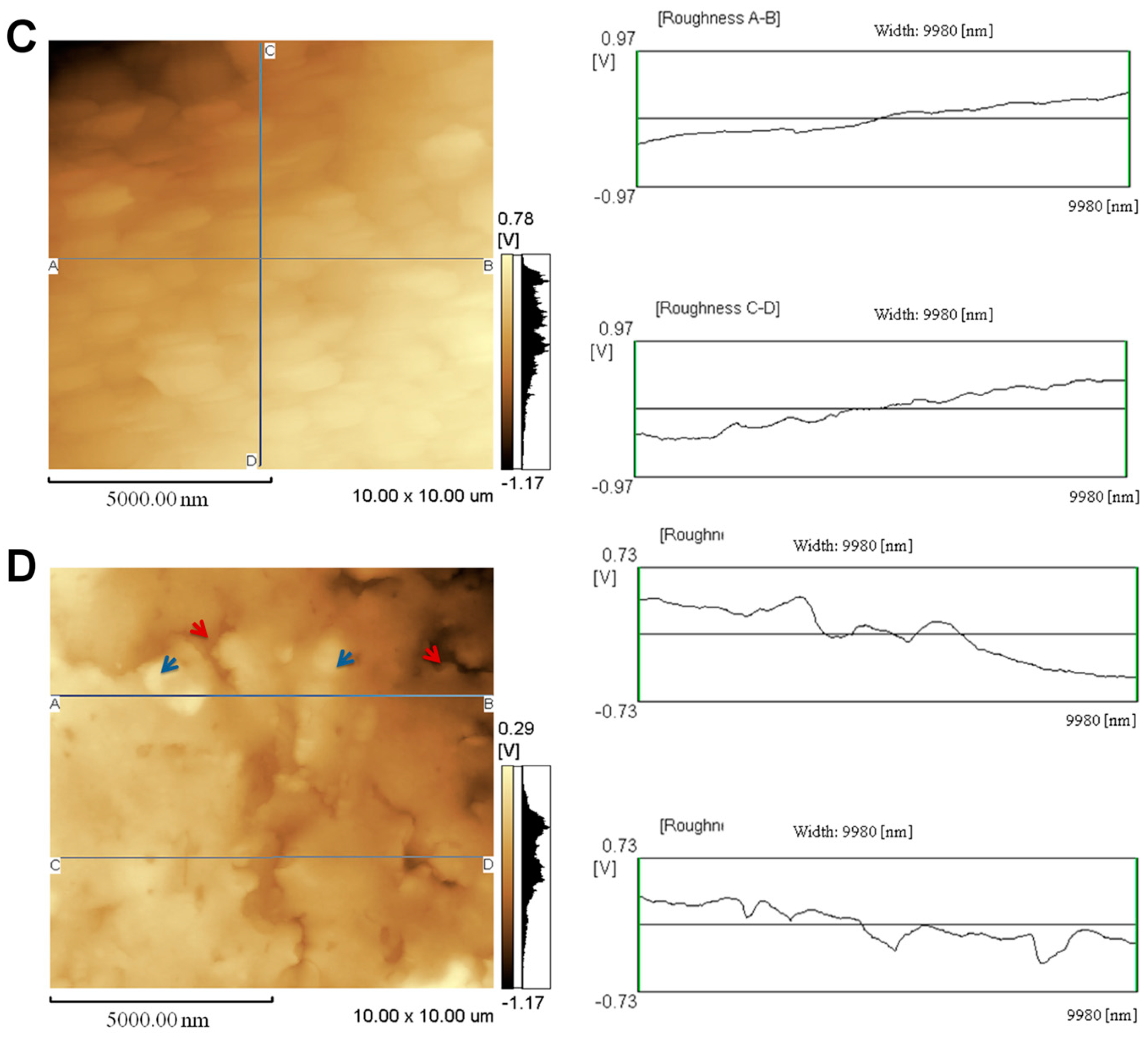
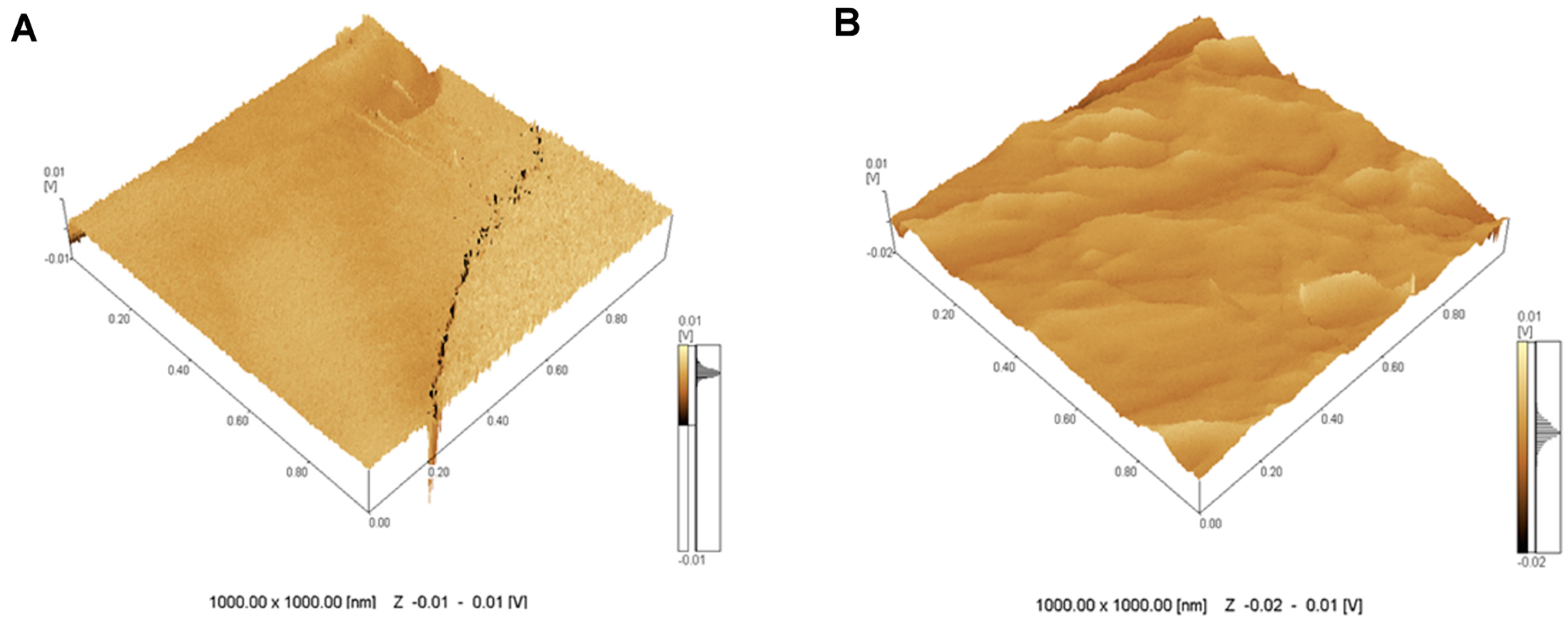
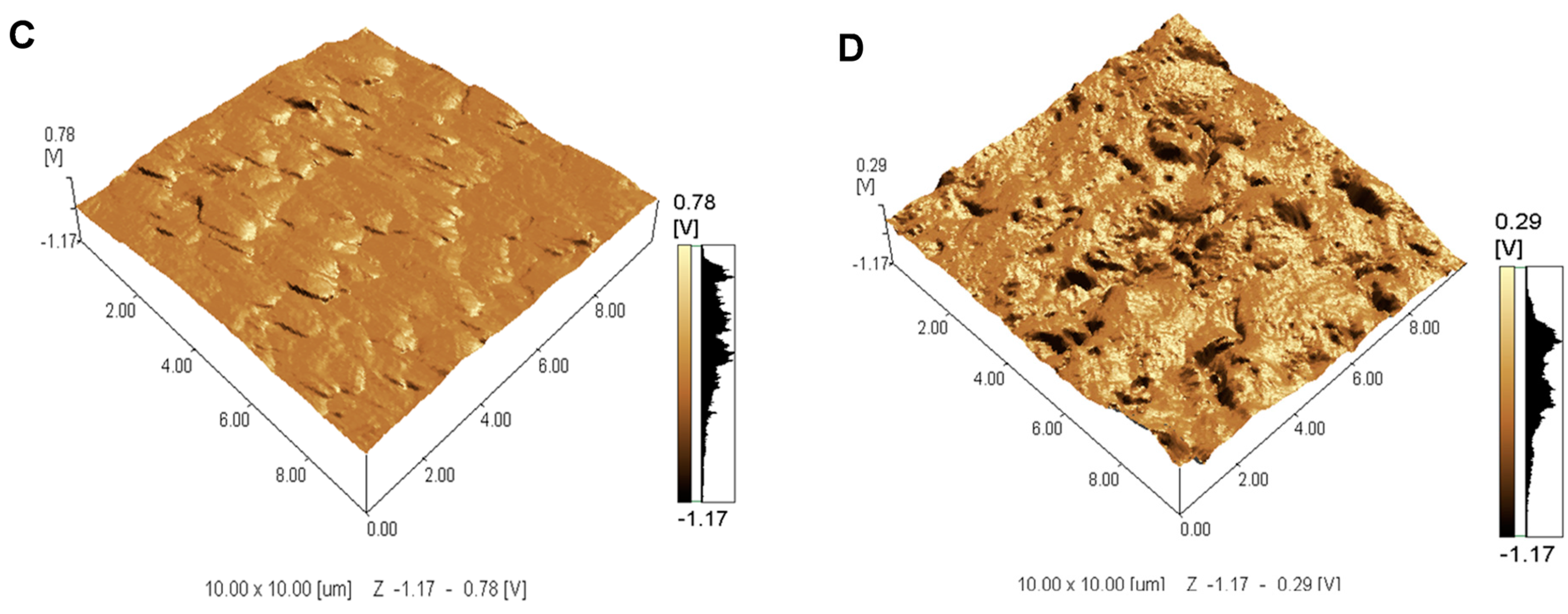
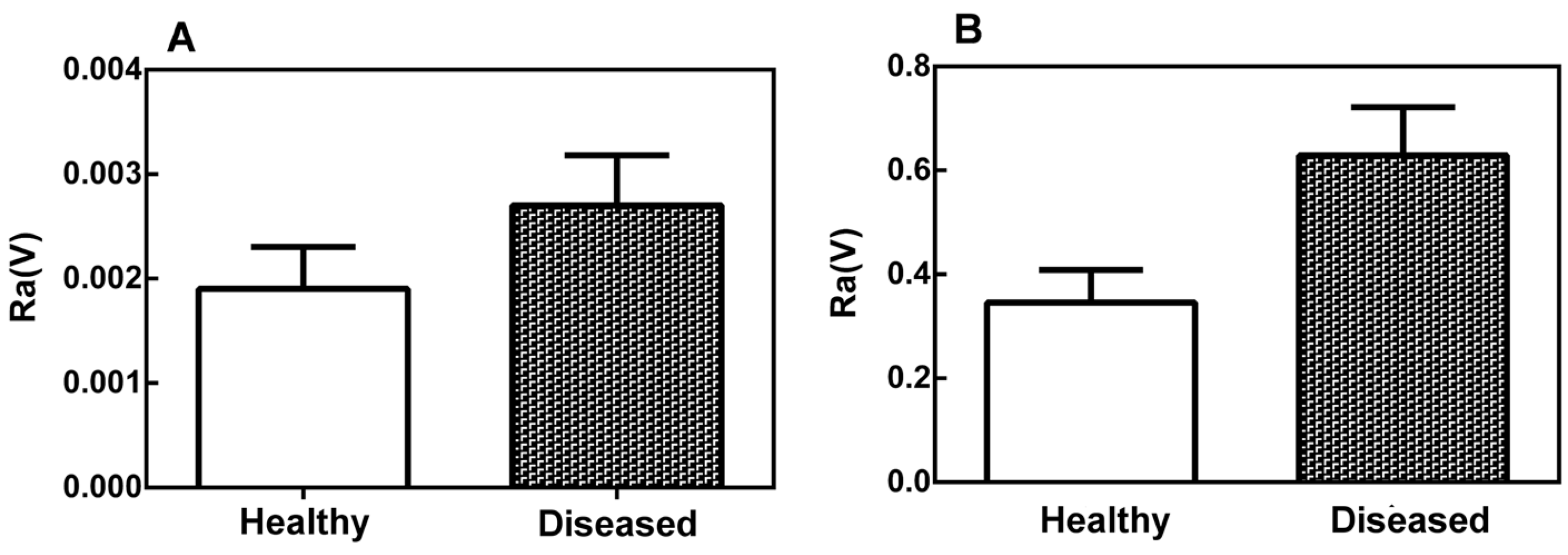
2.1. Topographical Analysis of the Leaf Midribs and Latex
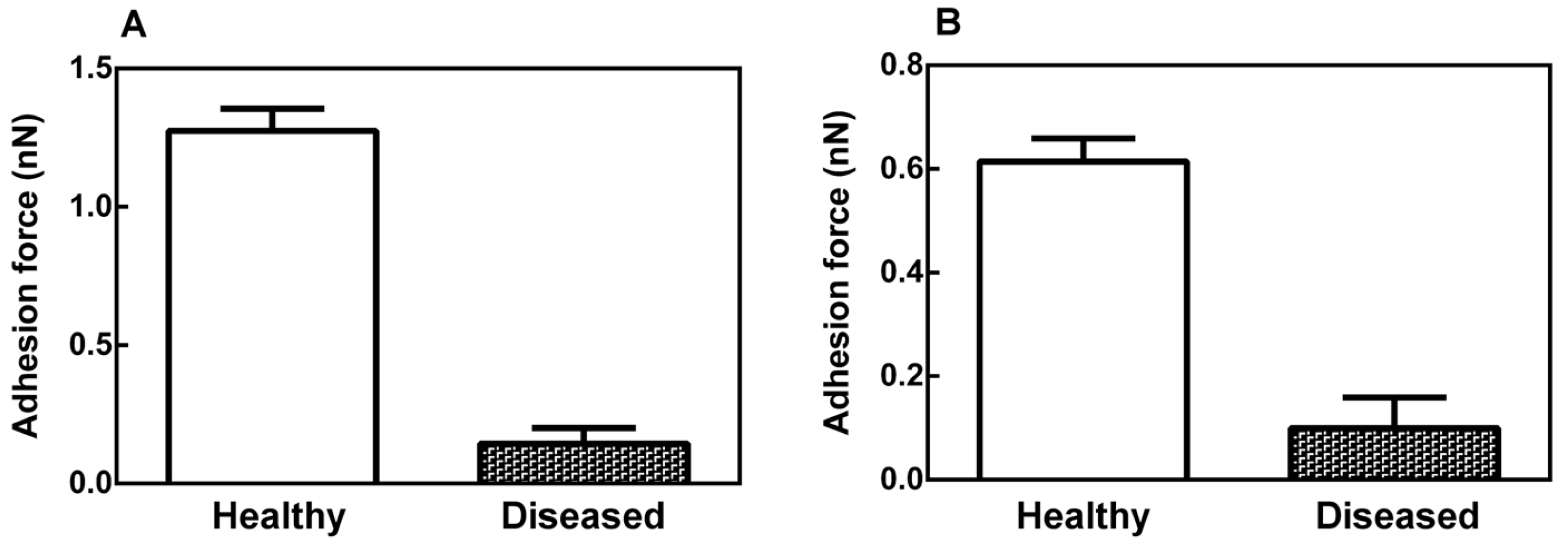
2.2. Mechanical Properties of the Leaf Midribs and Latex
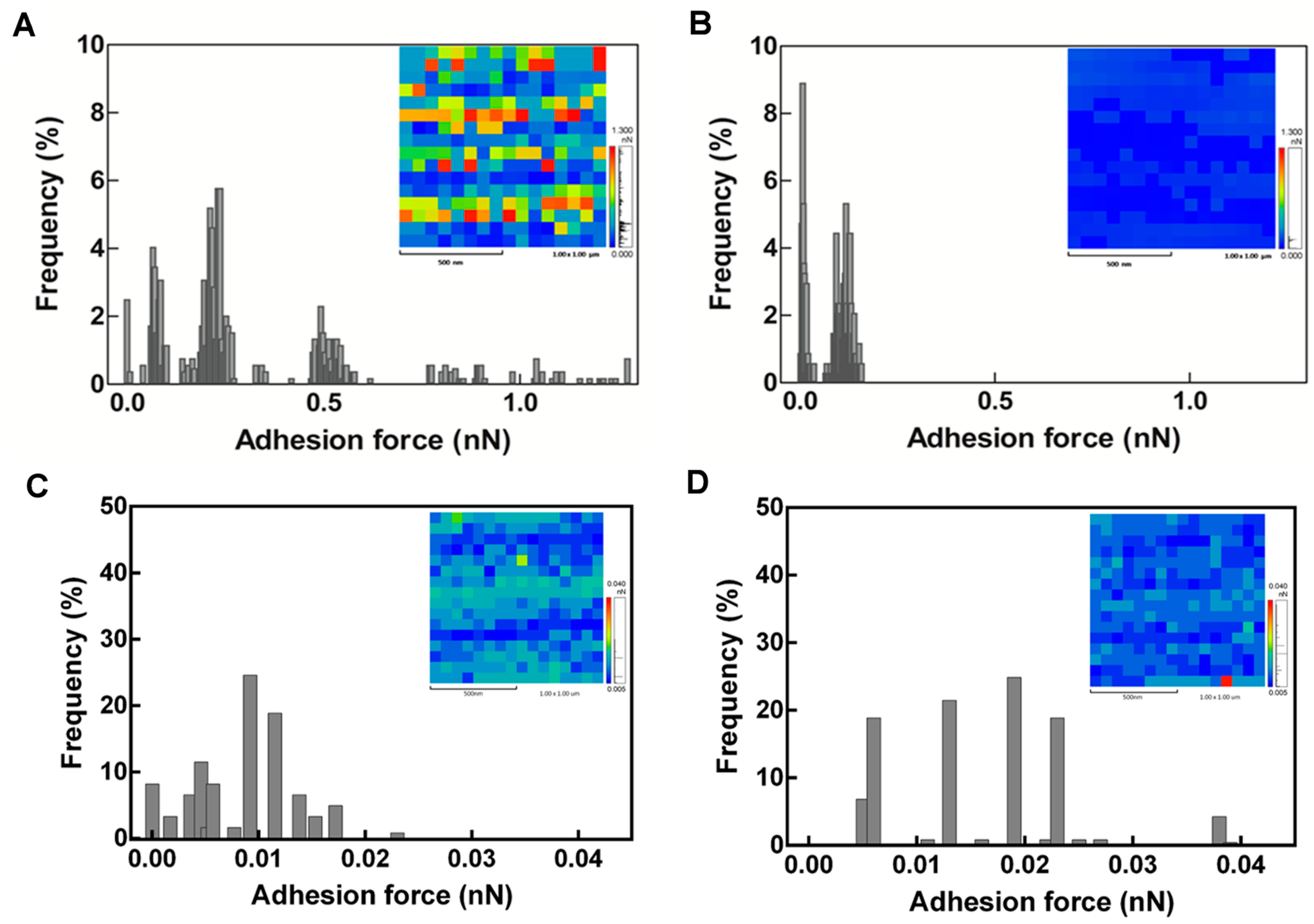
2.3. Latex Examination by Scanning Electron Microscopy (SEM)
3. Discussion
4. Materials and Methods
4.1. Leaf Midrib and Latex Collection
4.2. Image Acquisition
4.3. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| PMeV | Papaya Meleira Virus |
| AFM | Atomic force microscopy |
| SEM | Scanning electron microscopy |
| H2O2 | Hydrogen peroxide |
References
- FAOSTAT. FAO Statistics Division. Available online: http://faostat.fao.org/site/535/default.aspx#ancor (accessed on 25 August 2015).
- Abreu, P.M.V.; Gaspar, C.G.; Buss, D.S.; Ventura, J.A.; Ferreira, P.C.G.; Fernandes, P.M.B. Carica papaya microRNAs are responsive to Papaya meleira virus infection. PLoS ONE 2014, 9, e103401. [Google Scholar] [CrossRef] [PubMed]
- El Moussaoui, A.; Nijs, M.; Paul, C.; Wintjens, R.; Vincentelli, J.; Azarkan, M.; Looze, Y. Revisiting the enzymes stored in the laticifers of Carica papaya in the context of their possible participation in the plant defence mechanism. Cell. Mol. Life Sci. 2001, 58, 556–570. [Google Scholar] [CrossRef] [PubMed]
- Abreu, P.M.V.; Antunes, T.F.S.; Magaña-Álvarez, A.; Pérez-Brito, D.; Tapia-Tussell, R.; Ventura, J.A.; Fernandes, A.A.R.; Fernandes, P.M.B. A current overview of the Papaya meleira virus, an unusual plant virus. Viruses 2015, 7, 1853–1870. [Google Scholar] [CrossRef] [PubMed]
- Maciel-Zambolim, E.; Kunieda-Alonso, S.; Matsuoka, K.; De Carvalho, M.G.; Zerbini, F.M. Purification and some properties of Papaya meleira virus, a novel virus infecting papayas in Brazil. Plant Pathol. 2003, 52, 389–394. [Google Scholar] [CrossRef]
- Ventura, J.A.; Costa, H.; Tatagiba, J.D.S. Papaya diseases and integrated control. In Diseases of Fruits and Vegetables: Volume II. Klumer; Naqvi, S.A.M.H., Ed.; Academic Publishers: London, UK, 2004; pp. 201–268. [Google Scholar]
- Rodrigues, S.P.; da Cunha, M.; Ventura, J.A.; Fernandes, P.M.B. Effects of the Papaya meleira virus on papaya latex structure and composition. Plant Cell Rep. 2009, 28, 861–871. [Google Scholar] [CrossRef] [PubMed]
- Milani, P.; Gholamirad, M.; Traas, J.; Arnéodo, A.; Boudaoud, A.; Argoul, F.; Hamant, O. In vivo analysis of local wall stiffness at the shoot apical meristem in Arabidopsis using atomic force microscopy. Plant J. 2011, 67, 1116–1123. [Google Scholar] [CrossRef] [PubMed]
- Radotić, K.; Roduit, C.; Simonović, J.; Hornitschek, P.; Fankhauser, C.; Mutavdžić, D.; Steinbach, G.; Dietler, G.; Kasas, S. Atomic force microscopy stiffness tomography on living Arabidopsis thaliana cells reveals the mechanical properties of surface and deep cell-wall layers during growth. Biophys. J. 2012, 103, 386–394. [Google Scholar] [CrossRef] [PubMed]
- Lesniewska, E.; Adrian, M.; Klinguer, A.; Pugin, A. Cell wall modification in grapevine cells in response to UV stress investigated by atomic force microscopy. Ultramicroscopy 2004, 100, 171–178. [Google Scholar] [CrossRef] [PubMed]
- Aquije, G.M.F.V.; Zorzal, P.B.; Buss, D.S.; Ventura, J.A.; Fernandes, P.M.B.; Fernandes, A.A.R. Cell wall alterations in the leaves of fusariosis-resistant and susceptible pineapple cultivars. Plant Cell Rep. 2010, 29, 1109–1117. [Google Scholar] [CrossRef] [PubMed]
- Mahlein, A.K. Plant disease detection by imaging sensors—Parallels and specific demands for precision agriculture and plant phenotyping. Plant Dis. 2016, 100, 241–251. [Google Scholar] [CrossRef]
- Hagel, J.M.; Yeung, E.C.; Facchini, P.J. Got milk? The secret life of laticifers. Trends Plant Sci. 2008, 13, 631–639. [Google Scholar] [CrossRef] [PubMed]
- Jiménez, V.M.; Mora-Newcomer, E.; Gutiérrez-Soto, M.V. Biology of the Papaya Plant. In Genetics and Genomics of Papaya, Plant Genetics and Genomics; Ming, R., Moore, P.H., Eds.; Springer: New York, NY, USA, 2014; pp. 17–33. [Google Scholar]
- Gaboriaud, F.; Dufrêne, Y.F. Atomic force microscopy of microbial cells: Application to nanomechanical properties, surface forces and molecular recognition forces. Colloid Surf. B Biointerfaces 2007, 54, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Butt, H.-J.; Cappella, B.; Kappl, M. Force measurements with the atomic force microscope: Technique, interpretation and applications. Surf. Sci. Rep. 2005, 59, 1–152. [Google Scholar]
- Bessire, M.; Chassot, C.; Jacquat, A.C.; Humphry, M.; Borel, S.; Petétot, J.M.C.; Métraux, J.P.; Nawrath, C. A permeable cuticle in Arabidopsis leads to a strong resistance to Botrytis cinerea. EMBO J. 2007, 26, 2158–2168. [Google Scholar] [CrossRef] [PubMed]
- Reina-Pinto, J.J.; Yephremov, A. Surface lipids and plant defenses. Plant Physiol. Biochem. 2009, 47, 540–549. [Google Scholar] [CrossRef] [PubMed]
- Rosado, H.; Turner, D.R.; Foster, J.S.; Taylor, W.P. Impact of the β-lactam resistance modifier (−)-epicatechin gallate on the non-random distribution of phospholipids across the cytoplasmic membrane of Staphylococcus aureus. Int. J. Mol. Sci. 2015, 16, 1670–1672. [Google Scholar] [CrossRef] [PubMed]
- Rinne, P.L.H.; Boogaard, R.V.D.; Mensink, M.G.J.; Kopperud, C.; Kormelink, R.; Goldbach, R.; Schoot, C.V.D. Tobacco plants respond to the constitutive expression of the tospovirus movement protein NSM with a heat-reversible sealing of plasmodesmata that impairs development. Plant J. 2005, 43, 688–707. [Google Scholar] [CrossRef] [PubMed]
- Seisenberger, G.; Ried, M.U.; Endress, T.; Büning, H.; Hallek, M.; Bräuchle, C. Real-time single-molecule imaging of the infection pathway of an adeno-associated virus. Science 2001, 294, 1929–1932. [Google Scholar] [CrossRef] [PubMed]
- Hoang, K.; Lai, T.; Lin, C.; Chen, Y.; Liau, C. The chitinolytic activities of Streptomyces sp. TH-11. Int. J. Mol. Sci. 2011, 12, 56–65. [Google Scholar] [CrossRef] [PubMed]
- Yikmis, M.; Steinbuchel, A. Historical and recent achievements in the field of microbial degradation of natural and synthetic rubber. Appl. Environ. Microb. 2012, 78, 4543–4551. [Google Scholar] [CrossRef] [PubMed]
- Abreu, P.M.V.; Piccin, J.G.; Rodrigues, S.P.; Buss, D.S.; Ventura, J.A.; Fernandes, P.M.B. Molecular diagnosis of Papaya meleiravirus (PMeV) from leaf samples of Carica papaya L. using conventional and real-time RT-PCR. J. Virol. Methods 2012, 180, 11–17. [Google Scholar] [CrossRef] [PubMed]


© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Magaña-Álvarez, A.; Vencioneck Dutra, J.C.; Carneiro, T.; Pérez-Brito, D.; Tapia-Tussell, R.; Ventura, J.A.; Higuera-Ciapara, I.; Fernandes, P.M.B.; Fernandes, A.A.R. Physical Characteristics of the Leaves and Latex of Papaya Plants Infected with the Papaya meleira Virus. Int. J. Mol. Sci. 2016, 17, 574. https://doi.org/10.3390/ijms17040574
Magaña-Álvarez A, Vencioneck Dutra JC, Carneiro T, Pérez-Brito D, Tapia-Tussell R, Ventura JA, Higuera-Ciapara I, Fernandes PMB, Fernandes AAR. Physical Characteristics of the Leaves and Latex of Papaya Plants Infected with the Papaya meleira Virus. International Journal of Molecular Sciences. 2016; 17(4):574. https://doi.org/10.3390/ijms17040574
Chicago/Turabian StyleMagaña-Álvarez, Anuar, Jean Carlos Vencioneck Dutra, Tarcio Carneiro, Daisy Pérez-Brito, Raúl Tapia-Tussell, Jose Aires Ventura, Inocencio Higuera-Ciapara, Patricia Machado Bueno Fernandes, and Antonio Alberto Ribeiro Fernandes. 2016. "Physical Characteristics of the Leaves and Latex of Papaya Plants Infected with the Papaya meleira Virus" International Journal of Molecular Sciences 17, no. 4: 574. https://doi.org/10.3390/ijms17040574
APA StyleMagaña-Álvarez, A., Vencioneck Dutra, J. C., Carneiro, T., Pérez-Brito, D., Tapia-Tussell, R., Ventura, J. A., Higuera-Ciapara, I., Fernandes, P. M. B., & Fernandes, A. A. R. (2016). Physical Characteristics of the Leaves and Latex of Papaya Plants Infected with the Papaya meleira Virus. International Journal of Molecular Sciences, 17(4), 574. https://doi.org/10.3390/ijms17040574





