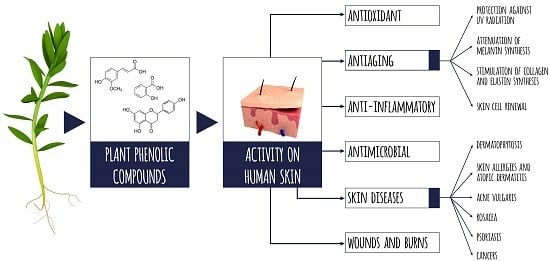
The Potential of Plant Phenolics in Prevention and Therapy of Skin Disorders
Abstract
:1. Introduction
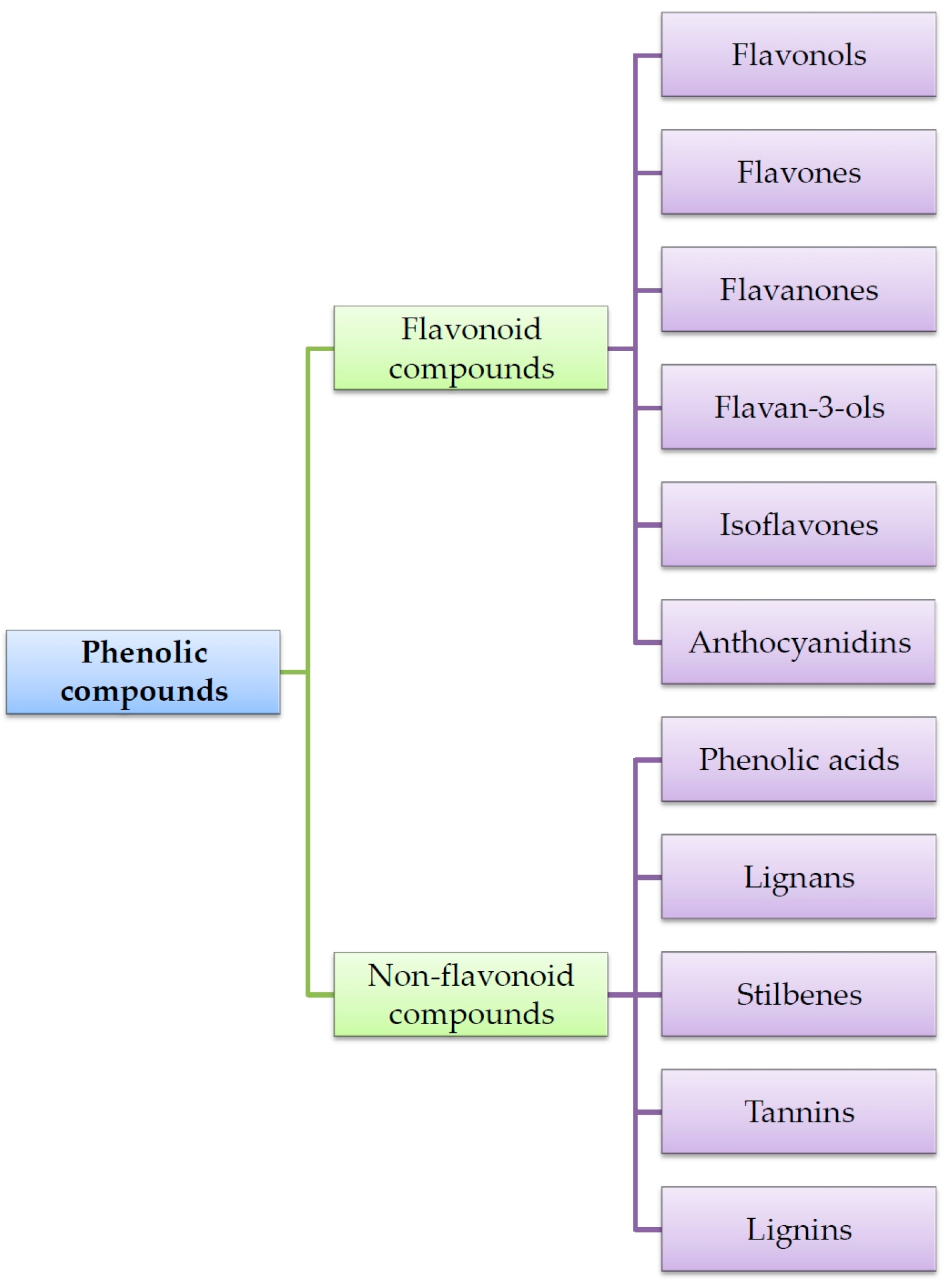
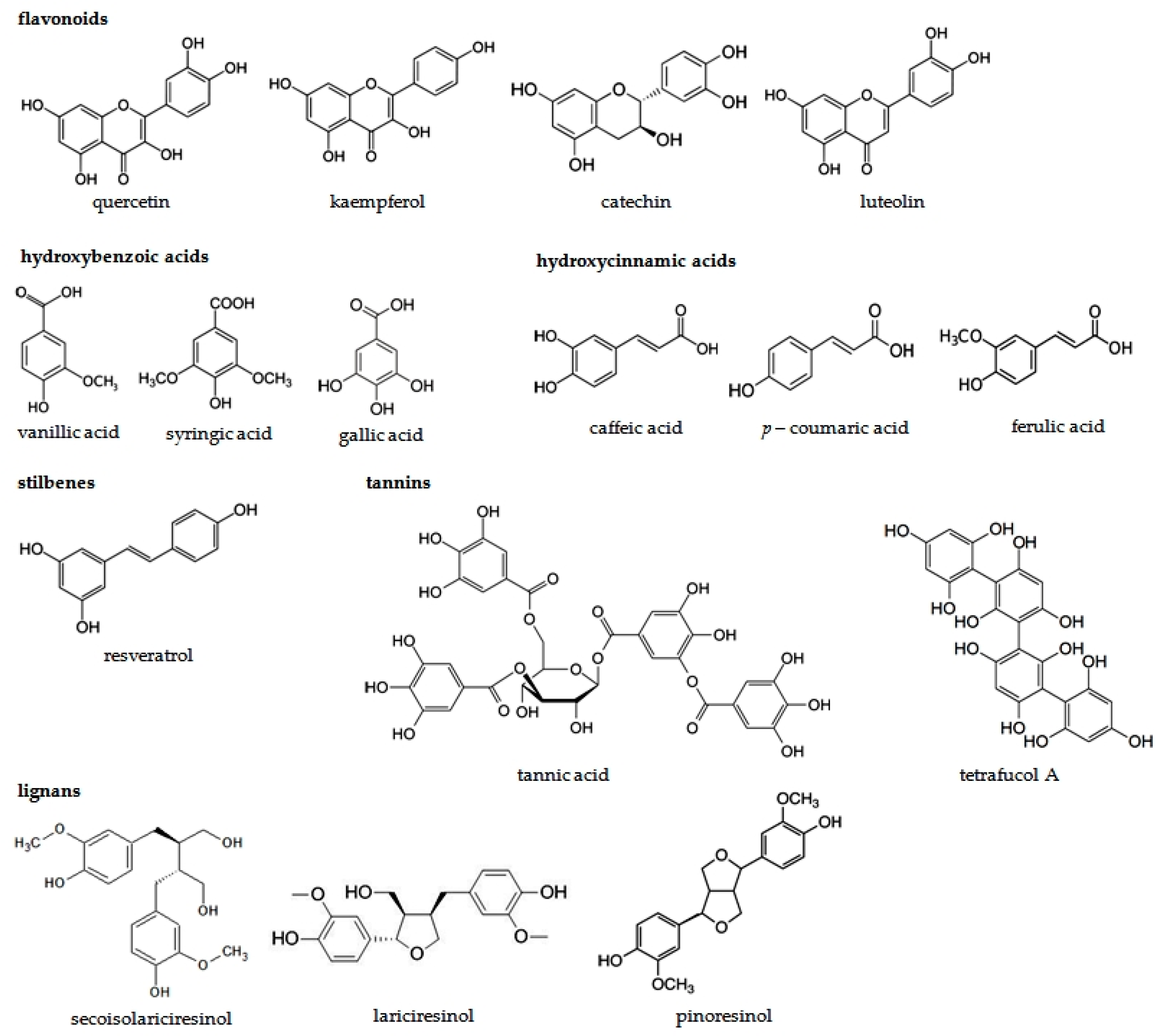
| Phenolic Compounds | Occurrence in Plants | References | |
|---|---|---|---|
| Flavonoids | Flavonols | Apples, oranges, grapefruits, black grapes, black elderberries, blueberries, cranberries, cabbage, lettuce, broccoli, radish, chives, onion paprica, chicory, green tea, red wine, Ginkgo biloba leaves, Morus alba leaves | [15,16,48,49,50,51,52,53] |
| Flavones | Selery, cayenne pepper, red paprica, parsley, thyme, lemon, rose hip, peppermint | [15,16,51,52,53,54] | |
| Flavanones | Tomatoes, mint, nigella seeds, citrus fruits (mainly oranges and grapefruits) | [15,16,51,52,53,54] | |
| Flavanols | Tea, red wine, chocolate, apples, kiwi | [15,16,51,52,53] | |
| Isoflavones | Soy, soy products, legumes | [15,16,51,52,53,55,56,57,58] | |
| Antocyjanidins | Cherries, strawberries, grapes, red wine, black currant, black elderberries, chokeberries, blueberries, red cabbage, rhubarb, radish, red onion | [15,16,51,52,53,59] | |
| Phenolic acids | Hydroxycinnamic acids | Apples, pears, plums, cherries, apricots, peachs, black currant, blueberries, Ginkgo biloba and Morus alba leaves, tobacco leaves, potatoes, spinach, lettuce, cabbage, bean, radish, potatoes, broccoli, curly kale, asparagus, olive oil, wine, coffee, citrus juice, grains | [20,23,30,60,61,62,63,64,65,66,67,68,69,70] |
| Hydroxybenzoic acids | Grapes, black currant, blackberries, lingon berries, strawberries, raspberries, onion, tea | [20,69,70,71,72] | |
| Tannins | Green and black tea, red wine | [32,33,48,73,74] | |
| Stilbens | Grapes, mulberries, peanuts, berries | [42,43,44,56,75,76,77,78,79,80] | |
| Lignans | Flaxseed, sunflower seeds, sezame seeds, grains, carrot, onion, chives, apples, cherries, blueberries, strawberries, nuts, tea, coffee | [34,35,38,39,43,81,82,83,84] | |
2. Health-Promoting Activity of Phenolic Compounds Based on Antioxidant, Anti-Inflammatory and Antimicrobial Mechanisms
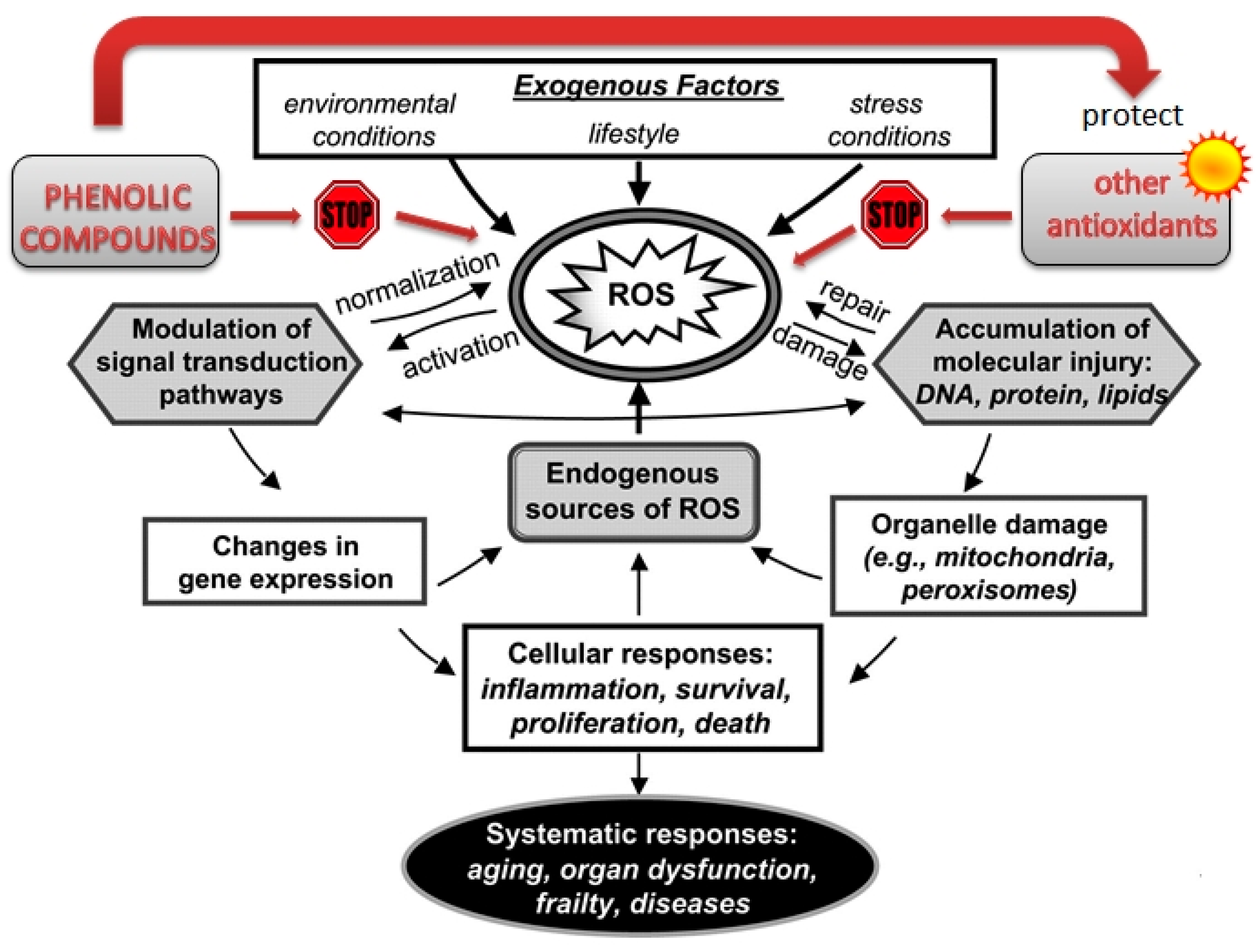
2.1. Antioxidant Activity
2.2. Anti-Inflammatory Properties
2.3. Antimicrobial Action
3. Anti-Aging Properties of Plant Phenolics
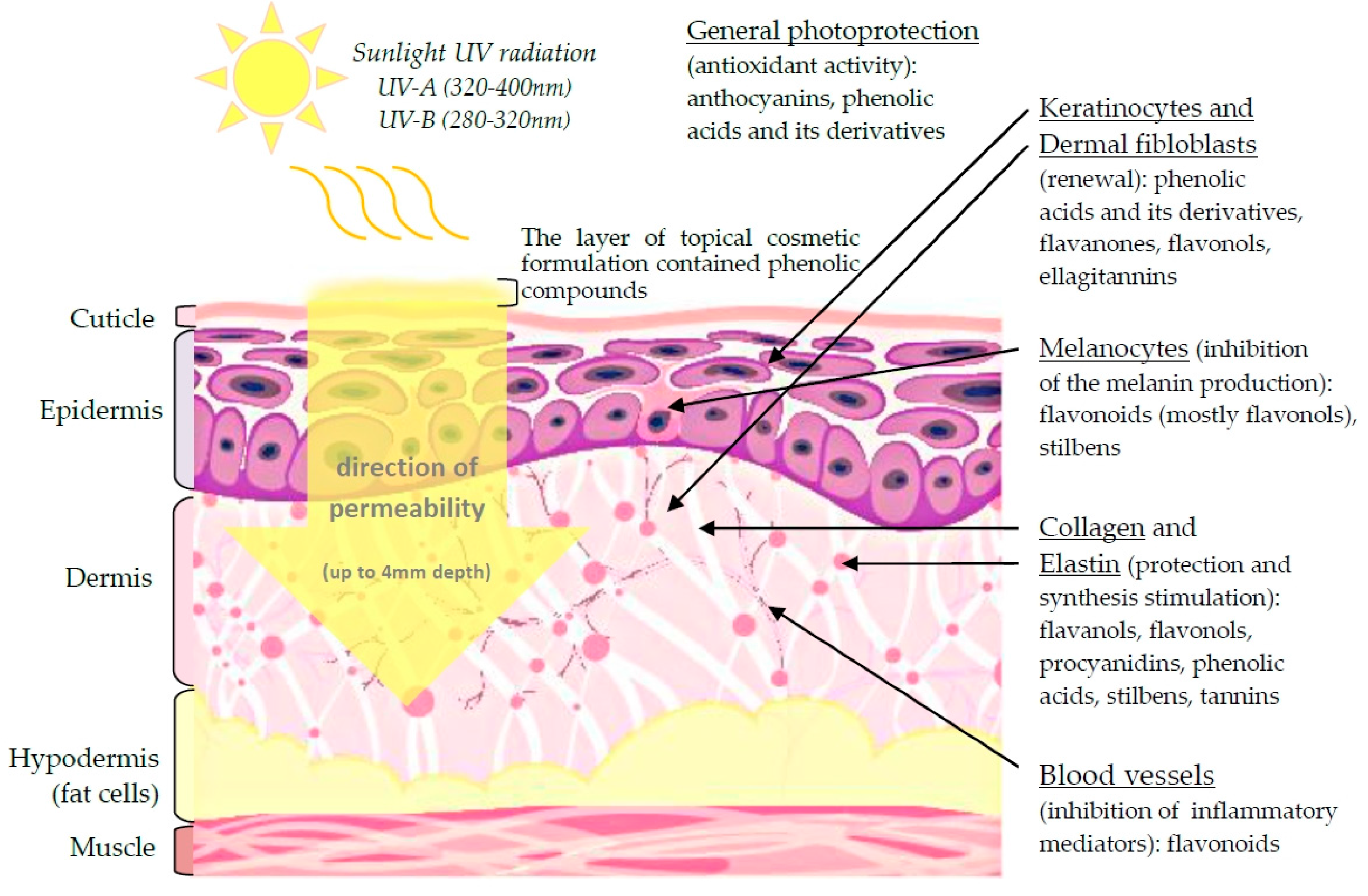
3.1. Skin Cell Renewal
| Analyzed Plant Material (Phenolic Compounds) | Expression of Skin-Related Genes up-(+) or down-(−) Regulation (%) | References | ||||
|---|---|---|---|---|---|---|
| cat a | KLF10 b | E2F4 c | ZFP36L1 d | CCL5 e | ||
| Poplar bud (phenolic acids, flavonoids, salicin) | +130 | +86 | +151 | +103 | −39 | [104] |
| Oak wood (ellagitannins) | +147 | +81 | +43 | +101 | −56 | [122] |
| Mate leaf (caffeoyl derivatives) | +228 | +83 | +44 | +70 | −46 | |
| Benjoin resin (phenolic acids) | +226 | +48 | +104 | +69 | −48 | |
3.2. Stimulation of Collagen and Elastin Synthesis
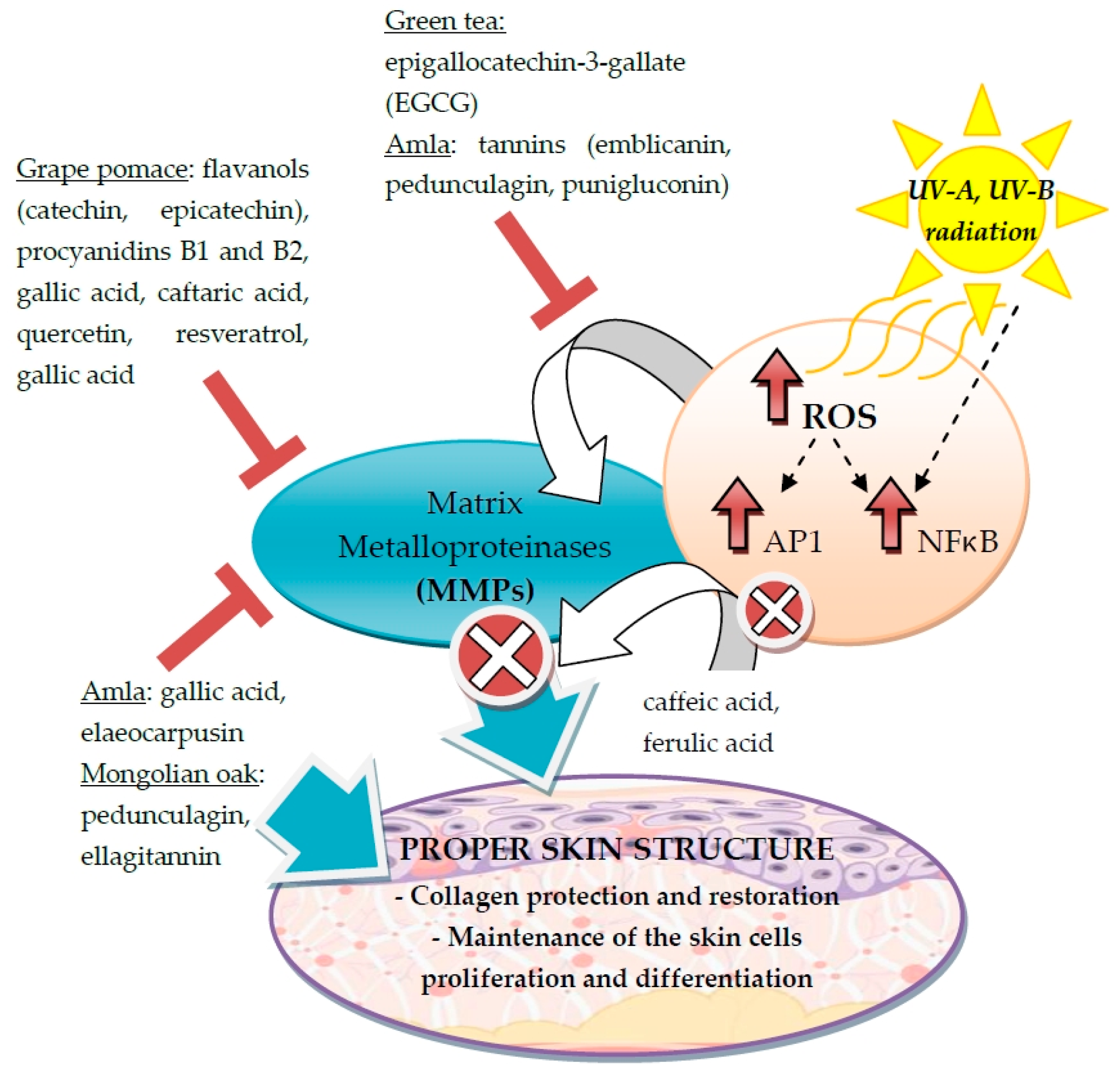
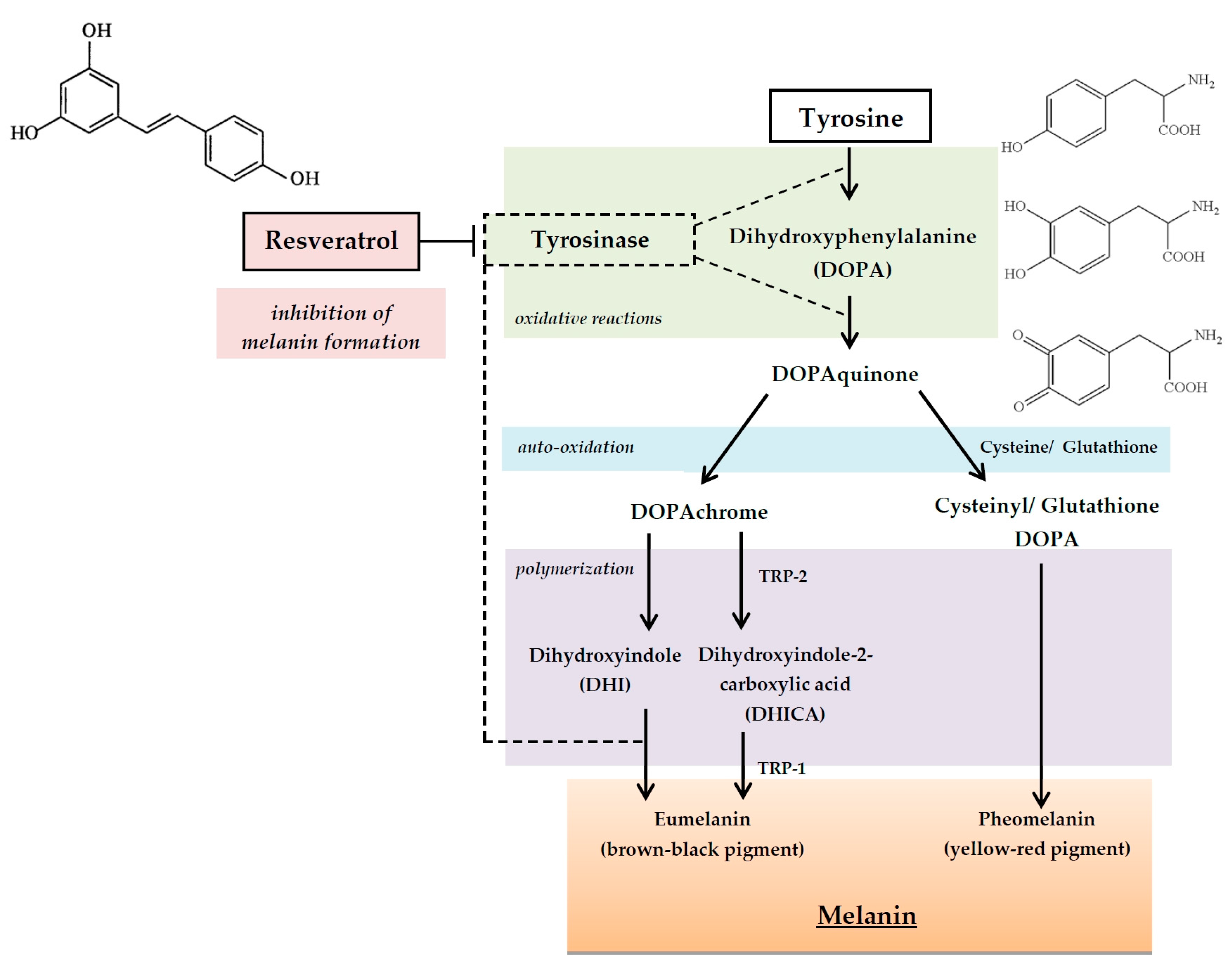
3.3. Attenuation of Melanin Production in Epidermis
3.4. Elimination of Oxidative Stress—Protection from UV Radiation
4. Skin Diseases
| Plant (Phenolic Compounds from Extract) | Object of Study | Protection from UV-Induced DNA Damage | Results | References |
|---|---|---|---|---|
| Castanea sativa (chlorogenic acid, ellagic acid, rutin, isoquercitrin, hyperoside) | Aneuploid human immortal keratinocyte cell line (HaCaT) | Direct antioxidant action (involving 1O2); Activation of nuclear factor erythroid-related factor-2 (NRF2) gene | Protection from oxidative damage; Reduction of the UV-induced damage in DNA | [144] |
| Blackberry Rubus adenotrichos (ellagitannins, cyanidin-3-glucoside) | Normal human epidermal keratinocytes (NHEK); 3D skin model | Increased UVB-mediated poly(ADP-ribose) polymerase cleavage; activation of caspases 3, 8 and 9 | Reduction of the DNA damage, including formation of cyclobutane pyrimidine dimers (CPDs) and 8-hydroxy-2′-deoxyguanosine (8-OHdG); severely modified cells were rapidly led to the apoptosis pathway | [146] |
| Honeyberry Lonicera caerulea (cyanidin-3-glucoside, caffeic acid, gallic acid) | HaCaT cell culture; Skin of albino SKH-1 hairless mice | Increased catalase activity and glutathione levels; activation of the caspases 3 and 9 | Reduction of the extent of DNA breakage; decreased generation of Reactive Oxygen/ Nitrogen Species (RONS) | [147,148] |
| Bilberry Vaccinium myrtillus (cyanidin and delphinidin derivatives) | HaCaT cell culture | Activation of the caspases 3 and 9 | [147] | |
| Morus australis (oxyresveratrol, kuwanon O) | Human primary epidermal keratinocytes culture (HEK) | Reduction of the H2O2-induced ROS formation; p53 activation | Cellular ROS inhibition; increased cell viability; reduction of 8-OHdG and CPDs formation | [149] |
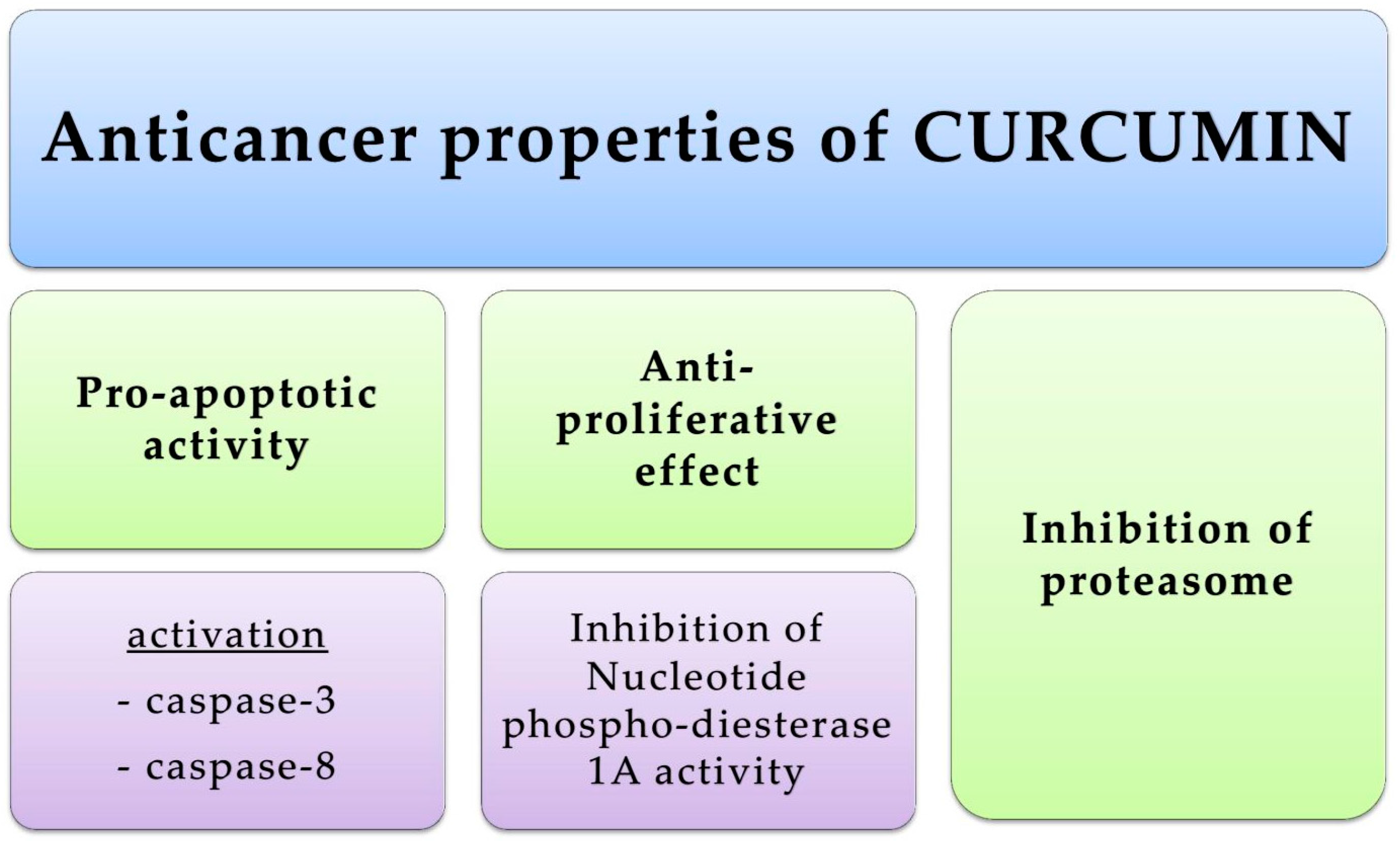
4.1. Skin Cancers

4.2. Psoriasis
4.3. Rosacea
4.4. Acne vulgaris
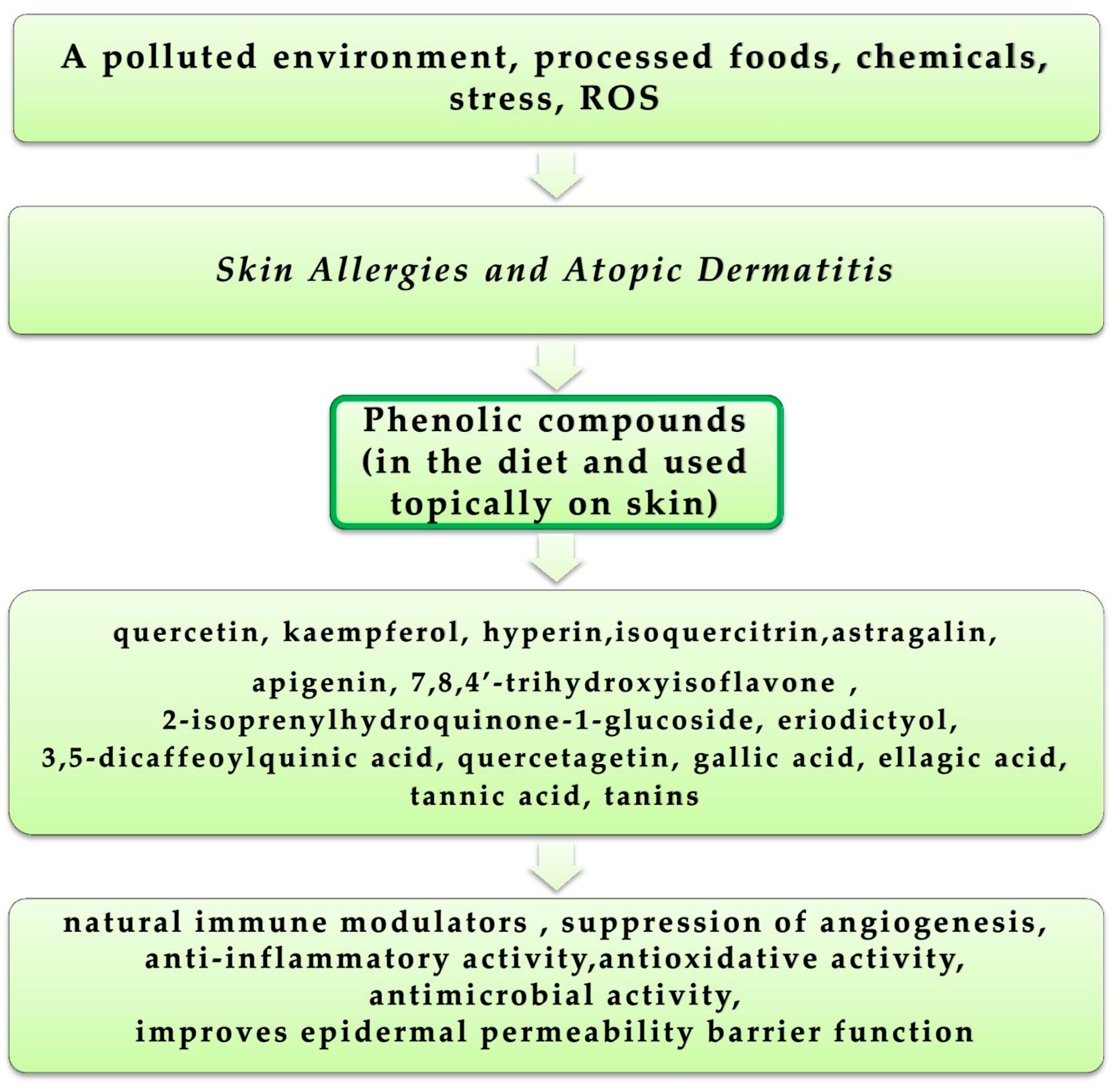
4.5. Skin Allergies and Atopic Dermatitis
4.6. Dermatophytosis
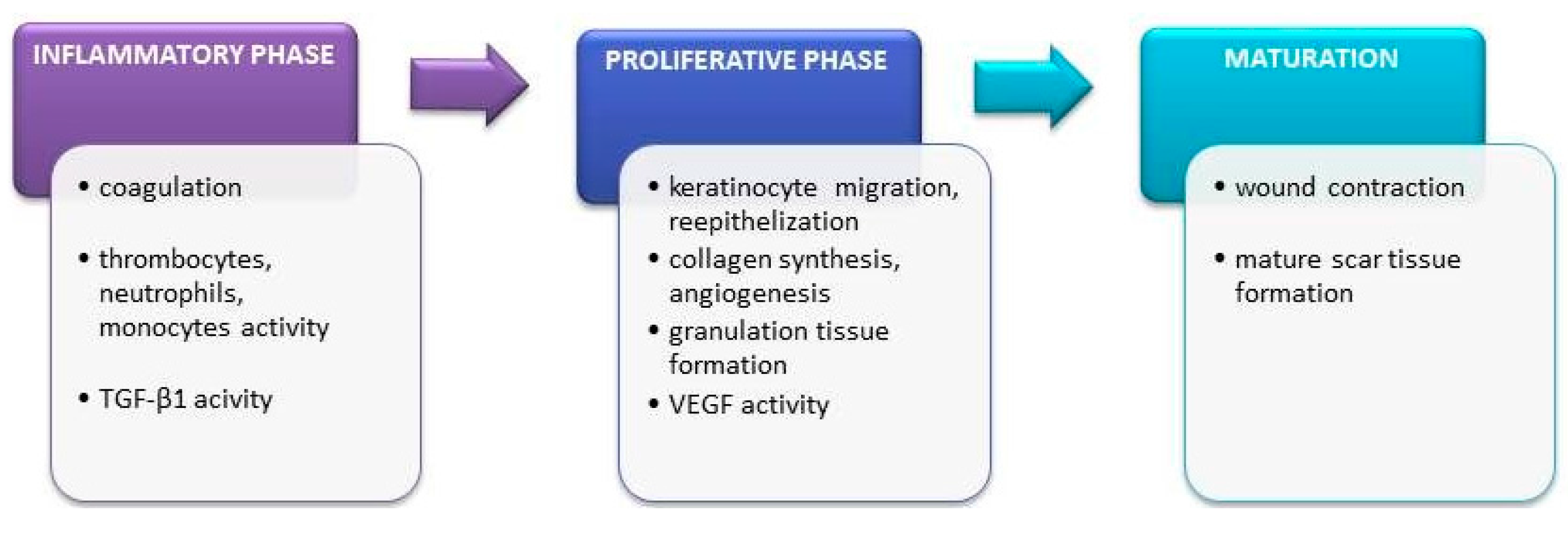
5. The Effect of Phenolic Compounds on Healing of Incised and Chronic Wounds and Burns
5.1. Wounds
5.1.1. Incised Wounds
5.1.2. Chronic Wounds
5.2. Burns
6. Conclusions
| Compound | Properties on Skin | References |
|---|---|---|
| Ellagic acid | Photoprotection, anti-allergic | [144,274] |
| Gallic acid | Photoprotection, maintaining skin homogeneity, anticarcinogenic, anti-acne, anti-allergic | [4,135,151,168,214,216,225,274] |
| Caffeic acid | Skin cells renewal, maintaining skin homogeneity, anticarcinogenic, Psoriasis treatment, anti-acne | [19,104,126,187,196,240] |
| Ferulic acid | Maintaining skin homogeneity, anti-acne | [19,104,126] |
| Quercetin | Maintaining skin homogeneity, melanogenesis inhibition, anticarcinogenic, Psoriasis treatment, anti-acne, anti-allergic, Atopic dermatitis treatment | [4,135,208,214,215,226,242,262,272,274] |
| Kaempferol | Melanogenesis inhibition, anti-acne, anti-allergic, Psoriasis treatment | [140,242,262] |
| Catechin, Epicatechin | Maintaining skin homogeneity, antibacterial, acceleration of wound healing, anticarcinogenic, Rosacea treatment, anti-acne, Dermatophytosis treatment | [4,124,223,224,252,263] |
| Tannins | Skin cells renewal, maintaining skin homogeneity, Atopic dermatitis treatment | [116,122,124,127,280] |
| Apigenin | Anticarcinogenic, anti-acne, atopic dermatitis, Dermatophytosis treatment | [226,281,288] |
| Luteolin | Antiinflammatory, anticarcinogenic, anti-acne | [155,165,206,261] |
| Vitexin | Antibacterial, acceleration of wound healing, anticarcinogenic | [9,116,166,167,217] |
| Isovitexin | Antibacterial, acceleration of wound healing, anticarcinogenic | [9,116,166,167,217] |
| Hyperin | Anticarcinogenic, anti-allergic | [208,274] |
| Myricetin, genistein , chlorogenic acid | Anticarcinogenic, anti-acne | [144,213,226,228,265] |
| Pinocembrin, pinobanksin, salicin | Skin cells renewal | [104] |
| Procyanidins B1, B2, caftaric acid, trans-resveratrol | Maintaining skin homogeneity | [4] |
| Brazilin, 4-O-methylsappanol, triacethyl resveratrol tetrahydroisoquinoline derivative | Melanogenesis inhibition | [132] |
| Rosmarinic acid | Photoprotection | [150] |
| Veratric acid, dihydrochalcone phloretin, afzelin | Antiinflammatory | [152,153,154] |
| Curcumin, syringic acid, pterostilbene furanocoumarins, eugenol | Anticarcinogenic | [162,163,164,222,229,234] |
| Capsaicin, rutin, isorhamnetin, benzoic compound, baicalin, isoorientin | Psoriasis treatment | [239,240,242,244,248] |
| Phenylpropenoic acids, saponosides | Rosacea treatment | [255] |
| Cinnamic acid, naringenin, α-mangostin, honokiol, magnolol ,thymol, 7-O-glucuronide, liquiritigenin 7-O-glucuronide, sakuranetin, genkwanin, arctigenin | Anti-acne | [260,265] |
| 3,5-dicaffeoylquinic acid, 3,5-dicaffeoylquinic acid methyl ester, isoquercitrin, astragalin, 2-isoprenylhydroquinone-1-glucoside | Anti-allergic | [274] |
| Quercetagetin, 7,8,4’-trihydroxyisoflavone, eriodictyol | Atopic dermatitis treatment | [278,279,283] |
| Coumarin | Acceleration of wound healing | [293] |
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Brohem, C.A.; Cardeal, L.B.; Tiago, M.; Soengas, M.S.; Barros, S.B.; Maria-Engler, S.S. Artificial skin in perspective: Concepts and applications. Pigment Cell Melanoma Res. 2011, 24, 35–50. [Google Scholar] [CrossRef] [PubMed]
- Metcalfe, A.D.; Ferguson, M.W. Tissue engineering of replacement skin: The crossroads of biomaterials, wound healing, embryonic development, stem cells and regeneration. J. R. Soc. Interface 2007, 4, 413–437. [Google Scholar] [CrossRef] [PubMed]
- Chioni, A.M.; Grose, R. Organotypic modelling as a means of investigating epithelial-stromal interactions during tumourigenesis. Fibrogenes. Tissue Repair 2008, 1, 8. [Google Scholar] [CrossRef] [PubMed]
- Wittenauer, J.; Mackle, S.; Sussmann, D.; Schweiggert-Weisz, U.; Carle, R. Inhibitory effects of polyphenols from grape pomace extract on collagenase and elastase activity. Fitoterapia 2015, 101, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Karim, A.A.; Azlan, A.; Ismail, A.; Hashim, P.; Abd Gani, S.S.; Zainudin, B.H.; Abdullah, N.A. Phenolic composition, antioxidant, anti-wrinkles and tyrosinase inhibitory activities of cocoa pod extract. BMC Complement. Altern. Med. 2014, 14, 381. [Google Scholar] [CrossRef] [PubMed]
- Danciu, C.; Vlaia, L.; Fetea, F.; Hancianu, M.; Coricovac, D.E.; Ciurlea, S.A.; Soica, C.M.; Marincu, I.; Vlaia, V.; Dehelean, C.A.; et al. Evaluation of phenolic profile, antioxidant and anticancer potential of two main representants of Zingiberaceae family against B164A5 murine melanoma cells. Biol. Res. 2015, 48, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Skorkowska-Telichowska, K.; Czemplik, M.; Kulma, A.; Szopa, J. The local treatment and available dressings designed for chronic wounds. J. Am. Acad. Dermatol. 2013, 68, e117–e126. [Google Scholar] [CrossRef] [PubMed]
- Del Rio, D.; Rodriguez-Mateos, A.; Spencer, J.P.; Tognolini, M.; Borges, G.; Crozier, A. Dietary (poly)phenolics in human health: Structures, bioavailability, and evidence of protective effects against chronic diseases. Antioxid. Redox Signal. 2013, 18, 1818–1892. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.Y.; Cai, Y.Z.; Zhang, Y. Natural phenolic compounds from medicinal herbs and dietary plants: Potential use for cancer prevention. Nutr. Cancer 2010, 62, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.; Mumper, R.J. Plant phenolics: Extraction, analysis and their antioxidant and anticancer properties. Molecules 2010, 15, 7313–7352. [Google Scholar] [CrossRef] [PubMed]
- Balasundrama, N.; Sundramb, K.; Sammana, S. Phenolic compounds in plants and agri-industrial by-products: Antioxidant activity, occurrence, and potential uses. Food Chem. 2006, 99, 191–203. [Google Scholar] [CrossRef]
- Okuda, T. Systematics and health effects of chemically distinct tannins in medicinal plants. Phytochemistry 2005, 66, 2012–2031. [Google Scholar] [CrossRef] [PubMed]
- Le Floch, A.; Jourdes, M.; Teissedre, P.L. Polysaccharides and lignin from oak wood used in cooperage: Composition, interest, assays: A review. Carbohydr. Res. 2015, 417, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Arct, J.; Pytkowska, K. Flavonoids as components of biologically active cosmeceuticals. Clin. Dermatol. 2008, 26, 347–357. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Scalbert, A.; Morand, C.; Remesy, C.; Jimenez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [PubMed]
- Aherne, S.A.; O’Brien, N.M. Dietary flavonols: Chemistry, food content, and metabolism. Nutrition 2002, 18, 75–81. [Google Scholar] [CrossRef]
- Castelluccio, C.; Paganga, G.; Melikian, N.; Bolwell, G.P.; Pridham, J.; Sampson, J.; Rice-Evans, C. Antioxidant potential of intermediates in phenylpropanoid metabolism in higher plants. FEBS Lett. 1995, 368, 188–192. [Google Scholar] [CrossRef]
- Saxena, M.; Saxena, J.; Pradhan, A. Flavonoids and phenolic acids as antioxidants in plants and human health. Int. J. Pharm. Sci. Rev. Res. 2012, 16, 130–134. [Google Scholar]
- Huang, C.J.; Zayas, J.F. Phenolic acids contribution to taste characteristic of corn germ protein flour products. J. Food Sci. 1991, 56, 1308–1310. [Google Scholar] [CrossRef]
- Fernández de Simón, B.; Hernández, T.; Estrella, I.; Gómez-Cordovés, C. Variation in phenol content in grapes during ripening: Low-molecular-weight phenols. Z. Lebensm. Unters. Forsch. 1992, 194, 351–354. [Google Scholar] [CrossRef]
- Herrmann, K. Occurrence and content of hydroxycinnamic and hydroxybenzoic acid compounds in foods. Crit. Rev. Food Sci. Nutr. 1989, 28, 315–347. [Google Scholar] [CrossRef] [PubMed]
- Ghasemzadeh, A.; Ghasemzadeh, N. Flavonoids and phenolic acids: Role and biochemical activity in plants and human. J. Med. Plants Res. 2011, 5, 6697–6703. [Google Scholar] [CrossRef]
- Breinholt, V. Desirable versus harmful levels of intake of flavonoids and phenolic acids. In Natural Antioxidants and Anticarcinogens in Nutrition, Health and Disease; Kumpulainen, J.T., Salonen, J.T., Eds.; The Royal Society of Chemistry: London, UK, 1999; pp. 93–99. [Google Scholar]
- Mattila, P.; Hellstrom, J. Phenolic acids in potatoes, vegetables, and some of their products. J. Food Compos. Anal. 2007, 20, 152–160. [Google Scholar] [CrossRef]
- Lima Mdos, S.; da Conceicao Prudencio Dutra, M.; Toaldo, I.M.; Correa, L.C.; Pereira, G.E.; de Oliveira, D.; Bordignon-Luiz, M.T.; Ninow, J.L. Phenolic compounds, organic acids and antioxidant activity of grape juices produced in industrial scale by different processes of maceration. Food Chem. 2015, 188, 384–392. [Google Scholar] [CrossRef] [PubMed]
- Monente, C.; Ludwig, I.A.; Irigoyen, A.; de Pena, M.P.; Cid, C. Assessment of total (free and bound) phenolic compounds in spent coffee extracts. J. Agric. Food Chem. 2015, 63, 4327–4334. [Google Scholar] [CrossRef] [PubMed]
- Bravo, L. Polyphenols: Chemistry, dietary sources, metabolism, and nutritional significance. Nutr. Rev. 1998, 56, 317–333. [Google Scholar] [CrossRef] [PubMed]
- Heleno, S.A.; Martins, A.; Queiroz, M.J.; Ferreira, I.C. Bioactivity of phenolic acids: Metabolites versus parent compounds: A review. Food Chem. 2015, 173, 501–513. [Google Scholar] [CrossRef] [PubMed]
- Farhoosh, R.; Johnny, S.; Asnaashari, M.; Molaahmadibahraseman, N.; Sharif, A. Structure-antioxidant activity relationships of o-hydroxyl, o-methoxy, and alkyl ester derivatives of p-hydroxybenzoic acid. Food Chem. 2016, 194, 128–134. [Google Scholar] [CrossRef] [PubMed]
- King, A.; Young, G. Characteristics and occurrence of phenolic phytochemicals. J. Am. Diet. Assoc. 1999, 99, 213–218. [Google Scholar] [CrossRef]
- Ashok, P.K.; Upadhyaya, K. Tannins are astringent. J. Pharmacogn. Phytochem. 2012, 1, 45–50. [Google Scholar]
- Okuda, T.; Ito, H. Tannins of constant structure in medicinal and food plants—hydrolyzable tannins and polyphenols related to tannins. Molecules 2011, 16, 2191. [Google Scholar] [CrossRef]
- Clinton, C. Plant tannins: A novel approach to the treatment of ulcerative colitis. Nat. Med. J. 2009, 1, 1–4. [Google Scholar]
- Lorenc-Kukula, K.; Zuk, M.; Kulma, A.; Czemplik, M.; Kostyn, K.; Skala, J.; Starzycki, M.; Szopa, J. Engineering flax with the GT family 1 Solanum sogarandinum Glycosyltransferase SsGT1 confers increased resistance to Fusarium infection. J. Agric. Food Chem. 2009, 57, 6698–6705. [Google Scholar] [CrossRef] [PubMed]
- Kurzer, M.S.; Xu, X. Dietary phytoestrogens. Annu. Rev. Nutr. 1997, 17, 353–381. [Google Scholar] [CrossRef] [PubMed]
- Sainvitu, P.; Nott, K.; Richard, G.; Blecker, C.; Jérôme, C.; Wathelet, J.P.; Paquot, M.; Deleu, M. Structure, properties and obtention routes of flaxseed lignan secoisolariciresinol, a review. Biotechnol. Agron. Soc. Environ. 2012, 16, 115–124. [Google Scholar]
- Slavova-Kazakova, A.; Karamac, M.; Kancheva, V.; Amarowicz, R. Antioxidant activity of flaxseed extracts in lipid systems. Molecules 2015, 21, 17. [Google Scholar] [CrossRef] [PubMed]
- Milder, I.E.; Arts, I.C.; van de Putte, B.; Venema, D.P.; Hollman, P.C. Lignan contents of dutch plant foods: A database including lariciresinol, pinoresinol, secoisolariciresinol and matairesinol. Br. J. Nutr. 2005, 93, 393–402. [Google Scholar] [CrossRef] [PubMed]
- Raffaelli, B.; Hoikkala, A.; Leppala, E.; Wahala, K. Enterolignans. J. Chromatogr. B 2002, 777, 29–43. [Google Scholar] [CrossRef]
- Smeds, A.I.; Eklund, P.C.; Sjöholm, R.E.; Willför, S.M.; Nishibe, S.; Deyama, T.; Holmbomet, B.R. Quantification of a broad spectrum of lignans in cereals, oilseeds, and nuts. J. Agric. Food Chem. 2007, 55, 1337–1346. [Google Scholar] [CrossRef] [PubMed]
- Patel, D.; Vaghasiya, J.; Pancholi, S.S.; Paul, A. Therapeutic potential of secoisolariciresinol diglucoside: A plant lignan. Int. J. Pharm. Sci. Drug Res. 2012, 4, 15–18. [Google Scholar]
- Stervbo, U.; Vang, O.; Bonnesen, C. A review of the content of the putative chemopreventive phytoalexin resveratrol in red wine. Food Chem. 2007, 2, 449–457. [Google Scholar] [CrossRef]
- Ososki, A.L.; Kennelly, E.J. Phytoestrogens: A review of the present state of research. Phytother. Res. 2003, 17, 845–869. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.M.; Wang, Z.R.; Hsieh, T.C.; Bruder, J.L.; Zou, J.G.; Huang, Y.Z. Mechanism of cardioprotection by resveratrol, a phenolic antioxidant present in red wine (Review). Int. J. Mol. Med. 2001, 8, 3–17. [Google Scholar] [CrossRef] [PubMed]
- Carter, L.G.; D’Orazio, J.A.; Pearson, K.J. Resveratrol and cancer: Focus on in vivo evidence. Endocr. Relat. Cancer 2014, 21, R209–R225. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.; Agrawal, M.; Dore, S. Neuroprotective properties and mechanisms of resveratrol in in vitro and in vivo experimental cerebral stroke models. ACS Chem. Neurosci. 2013, 4, 1151–1162. [Google Scholar] [CrossRef] [PubMed]
- Ma, T.; Tan, M.S.; Yu, J.T.; Tan, L. Resveratrol as a therapeutic agent for Alzheimer’s disease. Biomed. Res. Int. 2014, 2014, 350516. [Google Scholar] [CrossRef] [PubMed]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Radic. Biol. Med. 1996, 20, 933–956. [Google Scholar] [CrossRef]
- Alvarez, S.; Zaobornyj, T.; Actis-Goretta, L.; Fraga, C.G.; Boveris, A. Polyphenols and red wine as peroxynitrite scavengers: A chemiluminescent assay. Ann. N. Y. Acad. Sci. 2002, 957, 271–273. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Velez, M.; Martinez-Martinez, F.; del Valle-Ribes, C. The study of phenolic compounds as natural antioxidants in wine. Crit. Rev. Food Sci. Nutr. 2003, 43, 233–244. [Google Scholar] [CrossRef] [PubMed]
- Thilakarathna, S.H.; Rupasinghe, H.P. Flavonoid bioavailability and attempts for bioavailability enhancement. Nutrients 2013, 5, 3367–3387. [Google Scholar] [CrossRef] [PubMed]
- Harnly, J.M.; Doherty, R.F.; Beecher, G.R.; Holden, J.M.; Haytowitz, D.B.; Bhagwat, S.; Gebhardt, S. Flavonoid content of U.S. fruits, vegetables, and nuts. J. Agric. Food Chem. 2006, 54, 9966–9977. [Google Scholar] [CrossRef] [PubMed]
- Bhagwat, S.; Haytowitz, D.B.; Holden, J.M. USDA Database for the Flavonoid Content of Selected Foods. Available online: http://www.ars.usda.gov/SP2UserFiles/Place/80400525/Data/Flav/Flav_R03.pdf (accessed on 19 January 2016).
- Yao, L.H.; Jiang, Y.M.; Shi, J.; Tomas-Barberan, F.A.; Datta, N.; Singanusong, R.; Chen, S.S. Flavonoids in food and their health benefits. Plant Foods Hum. Nutr. 2004, 59, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Ibarreta, D.; Daxenberger, A.; Meyer, H.H. Possible health impact of phytoestrogens and xenoestrogens in food. Apmis 2001, 109, 161–184. [Google Scholar] [CrossRef] [PubMed]
- Cornwell, T.; Cohick, W.; Raskin, I. Dietary phytoestrogens and health. Phytochemistry 2004, 65, 995–1016. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Kumar, R.; Sabapathy, S.N.; Bawa, A.S. Functional and edible uses of soy protein products. Compr. Rev. Food Sci. Food Saf. 2008, 7, 14–28. [Google Scholar] [CrossRef]
- Cassidy, A. Isoflavones, lignans and stilbenes—origins, metabolism and potential importance to human health. J. Sci. Food Agric. 2000, 80, 1044–1062. [Google Scholar] [CrossRef]
- Sánchez-Moreno, C.; Cao, G.; Ou, B.; Prior, R.L. Anthocyanin and proanthocyanidin content in selected white and red wines. Oxygen radical absorbance capacity comparison with nontraditional wines obtained from highbush blueberry. J. Agric. Food Chem. 2003, 51, 4889–4896. [Google Scholar] [CrossRef] [PubMed]
- Kobus, J.; Flaczek, E.; Siger, A.; Nogala-Kałucka, M.; Korczak, J.; Pegg, R.B. Phenolic compounds and antioxidant activity of extracts of Ginkgo leaves. Eur. J. Lipid Sci. Technol. 2009, 111, 1150–1160. [Google Scholar] [CrossRef]
- Clifford, M.N. Chlorogenic acids and other cinnamates—nature, occurrence, dietary burden, adsorption and metabolism. J. Sci. Food. Agric. 2000, 80, 1033–1043. [Google Scholar] [CrossRef]
- Katsube, T.; Yamasaki, M.; Shiwaku, K.; Ishijima, T.; Matsumoto, I.; Abe, K.; Yamasaki, Y. Effect of flavonol glycoside in mulberry (Morus alba L.) leaf on glucose metabolism and oxidative stress in liver in diet-induced obese mice. J. Sci. Food Agric. 2010, 90, 2386–2392. [Google Scholar] [CrossRef] [PubMed]
- Antolovich, M.; Bedgood, D.R., Jr.; Bishop, A.G.; Jardine, D.; Prenzler, P.D.; Robards, K. LC-MS investigation of oxidation products of phenolic antioxidants. J. Agric. Food Chem. 2004, 52, 962–971. [Google Scholar] [CrossRef] [PubMed]
- Friedman, M. Chemistry, biochemistry and dietary role of potato polyphenols. J. Agric. Food Chem. 1997, 45, 1523–1540. [Google Scholar] [CrossRef]
- Woodring, P.J.; Edwards, P.A.; Chisholm, M.G. A hplc determination of nonflawonoid phenols in vidal blank wine using electrochemical detection. J. Agric. Food Chem. 1990, 38, 729–732. [Google Scholar] [CrossRef]
- Slavin, J.; Marquart, L.; Jakobs, D.J. Consumption of whole-grain food and decreased risk of cancer: Proposed mechanisms. Cereal Foods World 2000, 45, 54–58. [Google Scholar]
- Zielinski, H. Low molecular weight antioxidants in the cereal grains—A review. Pol. J. Food Nutr. Sci. 2002, 11, 3–9. [Google Scholar]
- Baublis, A.J.; Clydesdal, F.M.; Decker, E.A. Antioxidants in wheat-based breakfast cereals. Cereal Foods World 2000, 45, 71–74. [Google Scholar]
- Robbins, R.J. Phenolic acids in foods: An overview of analytical methodology. J. Agric. Food Chem. 2003, 51, 2866–2887. [Google Scholar] [CrossRef] [PubMed]
- Vinayagam, R.; Jayachandran, M.; Xu, B. Antidiabetic effects of simple phenolic acids: A comprehensive review. Phytother. Res. 2015. [Google Scholar] [CrossRef] [PubMed]
- Puupponen-Pimiä, R.; Nohynek, L.; Alakomi, H.L.; Oksman-Caldentey, K.M. Bioactive berry compounds-novel tools against human pathogens. Appl. Microbiol. Biotechnol. 2005, 67, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Bilyk, A.; Hicks, K.B.; Bills, D.D.; Sapers, G.M. Application of HPLC and dual wavelength detection to the analysis of phenolic compounds in apples. J. Liq. Chromatogr. 1988, 11, 2829–2841. [Google Scholar] [CrossRef]
- Ostrowska, J.; Luczaj, W.; Kasacka, I.; Rozanski, A.; Skrzydlewska, E. Green tea protects against ethanol-induced lipid peroxidation in rat organs. Alcohol 2004, 32, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Sang, S.; Tian, S.; Wang, H.; Stark, R.E.; Rosen, R.T.; Yang, C.S.; Ho, C.T. Chemical studies of the antioxidant mechanism of tea catechins: Radical reaction products of epicatechin with peroxyl radicals. Bioorg. Med. Chem. 2003, 11, 3371–3378. [Google Scholar] [CrossRef]
- Saiko, P.; Szakmary, A.; Jaeger, W.; Szekeres, T. Resveratrol and its analogs: Defense against cancer, coronary disease and neurodegenerative maladies or just a fad? Mutat. Res. 2008, 658, 68–94. [Google Scholar] [CrossRef] [PubMed]
- Dipak, K.D.; Maulik, N. Resveratrol in cardioprotection. A therapeutic promise of alternative medicine. Mol. Invent. 2006, 6, 36–47. [Google Scholar]
- Medina-Bolivar, F.; Condori, J.; Rimando, A.M.; Hubstenberger, J.; Shelton, K.; O’Keefe, S.F.; Bennett, S.; Dolan, M.C. Production and secretion of resveratrol in hairy root cultures of peanut. Phytochemistry 2007, 68, 1992–2003. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.S. Review: Quantitative extraction and analysis of grape and wine proanthocyanidins and stilbenes. Ciênc. Téc. Vitiv. 2005, 20, 59–89. [Google Scholar]
- Flamini, R.; Mattivi, F.; de Rosso, M.; Arapitsas, P.; Bavaresco, L. Advanced knowledge of three important classes of grape phenolics: Anthocyanins, stilbenes and flavonols. Int. J. Mol. Sci. 2013, 14, 19651–19669. [Google Scholar] [CrossRef] [PubMed]
- Vincenzi, S.; Tomasi, D.; Gaiotti, F.; Lovat, L.; Giacosa, S.; Torchio, F.; Río Segade, S.; Rolle, L. Comparative study of the resveratrol content of twenty-one Italian red grape varieties. S. Afr. J. Enol. Vitic. 2013, 34, 30–35. [Google Scholar]
- Murkies, A.L.; Wilcox, G.; Davis, S.R. Clinical review 92: Phytoestrogens. J. Clin. Endocrinol. Metab. 1998, 83, 297–303. [Google Scholar] [PubMed]
- Qiu, S.X.; Lu, Z.Z.; Luyengi, L.; Lee, S.K.; Pezzuto, J.M.; Farnsworth, M.R.; Thompson, L.U.; Fong, H.H.S. Isolation and characterisation of flaxseed (Linum usitatissimum ) constituents. Pharm. Biol. 1999, 37, 1–7. [Google Scholar] [CrossRef]
- Ide, T.; Kushiro, M.; Takahashi, Y.; Shinohara, K.; Fukuda, N.; Sirato-Yasumoto, S. Sesamin, a sesame lignan, as a potent serum lipid-lowering food component. Jpn. Agric. Res. Q. 2003, 37, 151–158. [Google Scholar] [CrossRef]
- Durazzo, A.; Zaccaria, M.; Polito, A.; Maiani, G.; Carcea, M. Lignan content in cereals, buckwheat and derived foods. Foods 2013, 2, 53–63. [Google Scholar] [CrossRef]
- Wade, L.G. Organic Chemistry, 8th ed.; Prentice Hall: Upper Saddle River, NJ, USA, 2012. [Google Scholar]
- Kefeli, V.I.; Kalevitch, M.V.; Borsari, B. Phenolic cycle in plants and environment. J. Cell. Mol. Biol. 2003, 2, 13–18. [Google Scholar]
- Naik, A.; Kalia, Y.N.; Guy, R.H. Transdermal drug delivery: Overcoming the skin’s barrier function. Pharm. Sci. Technol. Today 2000, 3, 318–326. [Google Scholar] [CrossRef]
- Arct, J.; Gronwald, M.; Kasiura, K. Possibilities for the prediction of an active substance penetration through epidermis. IFSCC Mag. 2001, 4, 179–183. [Google Scholar]
- Arct, J.; Oborska, A.; Mojski, M.; Binkowska, A.; Swidzikowska, B. Common cosmetic hydrophilic ingredients as penetration modifiers of flavonoids. Int. J. Cosmet. Sci. 2002, 24, 357–366. [Google Scholar] [CrossRef] [PubMed]
- Kogan, A.; Garti, N. Microemulsions as transdermal drug delivery vehicles. Adv. Colloid Interface Sci. 2006, 123, 369–385. [Google Scholar] [CrossRef] [PubMed]
- Korkina, L.G.; Pastore, S.; de Luca, C.; Kostyuk, V.A. Metabolism of plant polyphenols in the skin: Beneficial versus deleterious effects. Curr. Drug Metab. 2008, 9, 710–729. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, F.J.; Tukey, R.H.; Brunton, L.L.; Lazo, J.S.; Parker, K.L. Drug metabolism. In Goodman and Gilman’s the Pharmacological Basis of Therapeutics, 11th ed.; Brunton, L.L., Lazo, J.S., Parker, K.L., Eds.; Medical Publishing Division: New York, NY, USA, 2006; pp. 71–91. [Google Scholar]
- Morrow, D.I.J.; McCarron, P.A.; Woolfson, A.D.; Donnelly, R.F. Innovative strategies for enhancing topical and transdermal drug delivery. Open Drug Deliv. J. 2007, 1, 36–59. [Google Scholar] [CrossRef]
- Benson, H.A.E. Transdermal drug delivery: Penetration enhancement techniques. Curr. Drug. Deliv. 2005, 2, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Soto, M.; Falqué, E.; Domínguez, H. Relevance of natural phenolics from grape and derivative products in the formulation of cosmetics. Cosmetics 2015, 2, 259–276. [Google Scholar] [CrossRef]
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.; Mazur, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84. [Google Scholar] [CrossRef] [PubMed]
- Sies, H. Polyphenols and health: Update and perspectives. Arch. Biochem. Biophys. 2010, 501, 2–5. [Google Scholar] [CrossRef] [PubMed]
- Cai, H.; Xie, Z.; Liu, G.; Sun, X.; Peng, G.; Lin, B.; Liao, Q. Isolation, identification and activities of natural antioxidants from callicarpa kwangtungensis chun. PLoS ONE 2014, 9, e93000. [Google Scholar] [CrossRef] [PubMed]
- Yoo, H.G.; Lee, B.H.; Kim, W.; Lee, J.S.; Kim, G.H.; Chun, O.K.; Koo, S.I.; Kim, D.O. Lithospermum erythrorhizon extract protects keratinocytes and fibroblasts against oxidative stress. J. Med. Food 2014, 17, 1189–1196. [Google Scholar] [CrossRef] [PubMed]
- Samoylenko, A.; Hossain, J.A.; Mennerich, D.; Kellokumpu, S.; Hiltunen, J.K.; Kietzmann, T. Nutritional countermeasures targeting reactive oxygen species in cancer: From mechanisms to biomarkers and clinical evidence. Antioxid. Redox Signal. 2013, 19, 2157–2196. [Google Scholar] [CrossRef] [PubMed]
- Liaudanskas, M.; Viskelis, P.; Raudonis, R.; Kviklys, D.; Uselis, N.; Janulis, V. Phenolic composition and antioxidant activity of Malus domestica leaves. Sci. World J. 2014, 2014, 306217. [Google Scholar] [CrossRef] [PubMed]
- Alov, P.; Tsakovska, I.; Pajeva, I. Computational studies of free radical-scavenging properties of phenolic compounds. Curr. Top. Med. Chem. 2015, 15, 85–104. [Google Scholar] [CrossRef] [PubMed]
- Andjelković, M.; van Camp, J.; de Meulenaer, B.; Depaemelaere, G.; Socaciu, C.; Verloo, M.; Verhe, R. Iron-chelation properties of phenolic acids bearing catechol and galloyl groups. Food Chem. 2006, 98, 23–31. [Google Scholar] [CrossRef]
- Dudonne, S.; Poupard, P.; Coutiere, P.; Woillez, M.; Richard, T.; Merillon, J.M.; Vitrac, X. Phenolic composition and antioxidant properties of poplar bud (Populus Nigra) extract: Individual antioxidant contribution of phenolics and transcriptional effect on skin aging. J. Agric. Food Chem. 2011, 59, 4527–4536. [Google Scholar] [CrossRef] [PubMed]
- Sadowska-Bartosz, I.; Bartosz, G. Effect of antioxidants supplementation on aging and longevity. Biomed. Res. Int. 2014, 2014, 404680. [Google Scholar] [CrossRef] [PubMed]
- Pastore, S.; Potapovich, A.; Kostyuk, V.; Mariani, V.; Lulli, D.; de Luca, C.; Korkina, L. Plant Polyphenols Effectively Protect Hacat Cells from Ultraviolet C-Triggered Necrosis and Suppress Inflammatory Chemokine Expression. Ann. N. Y. Acad. Sci. 2009, 1171, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Rhein, L.D.; Fluhr, J.W. Aging Skin: Current and Future Therapeutic Strategies; Allured Business Media: Carol Stream, IL, USA, 2010; pp. 182–184, 225–240. [Google Scholar]
- Arct, J.; Bielenda, B.; Oborska, A.; Pytkowska, K. The tea and its cosmetic application. J. Appl. Cosmetol. 2003, 21, 117–127. [Google Scholar]
- Czemplik, M.; Zuk, M.; Kulma, A.; Kuc, S.; Szopa, J. GM flax as a source of effective antimicrobial compounds. Sci. Microb. Pathog. Commun. Curr. Res. Technol. Adv. 2011, 76, 39–47. [Google Scholar]
- Pinho, E.; Ferreira, I.C.; Barros, L.; Carvalho, A.M.; Soares, G.; Henriques, M. Antibacterial potential of northeastern portugal wild plant extracts and respective phenolic compounds. Biomed. Res. Int. 2014, 2014, 814590. [Google Scholar] [CrossRef] [PubMed]
- Zuk, M.; Dorotkiewicz-Jach, A.; Drulis-Kawa, Z.; Arendt, M.; Kulma, A.; Szopa, J. Bactericidal activities of GM flax seedcake extract on pathogenic bacteria clinical strains. BMC Biotechnol. 2014, 14, 70. [Google Scholar] [CrossRef] [PubMed]
- Anani, K.; Adjrah, Y.; Ameyapoh, Y.; Karou, S.D.; Agbonon, A.; de Souza, C.; Gbeassor, M. Effects of hydroethanolic extracts of Balanites aegyptiaca (L.) Delile (Balanitaceae) on some resistant pathogens bacteria isolated from wounds. J. Ethnopharmacol. 2015, 164, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Parisi, R.; Symmons, D.P.; Griffiths, C.E.; Ashcroft, D.M. Global epidemiology of psoriasis: A systematic review of incidence and prevalence. J. Investig. Dermatol. 2013, 133, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Langley, R.G.; Krueger, G.G.; Griffiths, C.E. Psoriasis: Epidemiology, clinical features, and quality of life. Ann. Rheum. Dis. 2005, 64 (Suppl. 2), ii18–ii23. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; He, M.; Zang, X.; Zhou, Y.; Qiu, T.; Pan, S.; Xu, X. A structure-activity relationship study of flavonoids as inhibitors of E. coli by membrane interaction effect. Biochim. Biophys. Acta BBA Biomembr. 2013, 1828, 2751–2756. [Google Scholar] [CrossRef] [PubMed]
- Kisseih, E.; Lechtenberg, M.; Petereit, F.; Sendker, J.; Zacharski, D.; Brandt, S.; Agyare, C.; Hensel, A. Phytochemical characterization and in vitro wound healing activity of leaf extracts from combretum mucronatum Schum. & Thonn.: Oligomeric procyanidins as strong inductors of cellular differentiation. J. Ethnopharmacol. 2015, 174, 628–636. [Google Scholar] [PubMed]
- Puizina-Ivić, N. Skin aging. Acta Dermatovenerol. Alp. Pannonica Adriat. 2008, 17, 47–54. [Google Scholar] [PubMed]
- Ramalingum, N.; Mahomoodally, M.F. The therapeutic potential of medicinal foods. Adv. Pharmacol. Sci. 2014, 2014, 354264. [Google Scholar] [CrossRef] [PubMed]
- Grether-Beck, S.; Marini, A.; Jaenicke, T.; Krutmann, J. French maritime pine bark extract (Pycnogenol®) effects on human skin: Clinical and molecular evidence. Skin Pharmacol. Physiol. 2015, 29, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, A.S.; Estanqueiro, M.; Oliveira, M.B.; Sousa Lobo, J.M. Main benefits and applicability of plant extracts in skin care products. Cosmetics 2015, 2, 48–65. [Google Scholar] [CrossRef]
- Koster, M.I. Making an Epidermis. Ann. N. Y. Acad. Sci. 2009, 1170, 7–10. [Google Scholar] [CrossRef] [PubMed]
- Dudonne, S.; Coutiere, P.; Woillez, M.; Merillon, J.M.; Vitrac, X. DNA macroarray study of skin aging-related genes expression modulation by antioxidant plant extracts on a replicative senescence model of human dermal fibroblasts. Phytother. Res. 2011, 25, 686–693. [Google Scholar] [CrossRef] [PubMed]
- Fujii, T.; Wakaizumi, M.; Ikami, T.; Saito, M. Amla (Emblica officinalis Gaertn.) extract promotes procollagen production and inhibits matrix metalloproteinase-1 in human skin fibroblasts. J. Ethnopharmacol. 2008, 119, 53–57. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, A.; Sarkar, J.; Chakraborti, T.; Chakraborti, S. Role of Spm-Cer-S1P signalling pathway in MMP-2 mediated U46619-induced proliferation of pulmonary artery smooth muscle cells: Protective role of epigallocatechin-3-gallate. Cell Biochem. Funct. 2015, 33, 463–477. [Google Scholar] [CrossRef] [PubMed]
- Quan, T.; Qin, Z.; Xia, W.; Shao, Y.; Voorhees, J.J.; Fisher, G.J. Matrix-degrading metalloproteinases in photoaging. J. Investig. Dermatol. Symp. Proc. 2009, 14, 20–24. [Google Scholar] [CrossRef] [PubMed]
- Pluemsamran, T.; Onkoksoong, T.; Panich, U. Caffeic acid and ferulic acid inhibit UVA-induced matrix metalloproteinase-1 through regulation of antioxidant defense system in keratinocyte HaCaT cells. Photochem. Photobiol. 2012, 88, 961–968. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.H.; Kim, D.H.; Oh, M.H.; Park, K.J.; Heo, J.H.; Lee, M.W. Inhibition of matrix metalloproteinase-1 and type-I procollagen expression by phenolic compounds isolated from the leaves of Quercus mongolica in ultraviolet-irradiated human fibroblast cells. Arch. Pharm. Res. 2015, 38, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Hah, Y.S.; Cho, H.Y.; Lim, T.Y.; Park, D.H.; Kim, H.M.; Yoon, J.; Kim, J.G.; Kim, C.Y.; Yoon, T.J. Induction of melanogenesis by rapamycin in human MNT-1 melanoma cells. Ann. Dermatol. 2012, 24, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.S.; Kuan, Y.D.; Chiu, K.H.; Wang, W.K.; Chang, F.H.; Liu, C.H.; Lee, C.H. The extract of rhodobacter sphaeroides inhibits melanogenesis through the MEK/ERK signaling pathway. Mar. Drugs 2013, 11, 1899–1908. [Google Scholar] [CrossRef] [PubMed]
- Parvez, S.; Kang, M.; Chung, H.S.; Cho, C.; Hong, M.C.; Shin, M.K.; Bae, H. Survey and mechanism of skin depigmenting and lightening agents. Phytother. Res. 2006, 20, 921–934. [Google Scholar] [CrossRef] [PubMed]
- Brenner, M.; Hearing, V.J. The protective role of melanin against uv damage in human skin. Photochem. Photobiol. 2008, 84, 539–549. [Google Scholar] [CrossRef] [PubMed]
- Mitani, K.; Takano, F.; Kawabata, T.; Allam, A.E.; Ota, M.; Takahashi, T.; Yahagi, N.; Sakurada, C.; Fushiya, S.; Ohta, T. Suppression of melanin synthesis by the phenolic constituents of sappanwood (Caesalpinia sappan). Planta Med. 2013, 79, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Rangkadilok, N.; Sitthimonchai, S.; Worasuttayangkurn, L.; Mahidol, C.; Ruchirawat, M.; Satayavivad, J. Evaluation of free radical scavenging and antityrosinase activities of standardized longan fruit extract. Food Chem. Toxicol. 2007, 45, 328–336. [Google Scholar] [CrossRef] [PubMed]
- Zucca, P.; Pintus, M.; Manzo, G.; Nieddu, M.; Steri, D.; Rinaldi, A.C. Antimicrobial, antioxidant and anti-tyrosinase properties of extracts of the mediterranean parasitic plant Cytinus hypocistis. BMC Res. Notes 2015, 8, 562. [Google Scholar] [CrossRef] [PubMed]
- Roh, H.J.; Noh, H.J.; Na, C.S.; Kim, C.S.; Kim, K.H.; Hong, C.Y.; Lee, K.R. Phenolic Compounds from the Leaves of Stewartia pseudocamellia Maxim. and their Whitening Activities. Biomol. Ther. Seoul 2015, 23, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Sugumaran, M. Comparative biochemistry of eumelanogenesis and the protective roles of phenoloxidase and melanin in insects. Pigment Cell Res. 2002, 15, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Boissy, R.E.; Manga, P. On the etiology of contact/occupational vitiligo. Pigment Cell Res. 2004, 17, 208–214. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Park, J.H.; Suh, H.J.; Lee, I.C.; Koh, J.; Boo, Y.C. Effects of resveratrol, oxyresveratrol, and their acetylated derivatives on cellular melanogenesis. Arch. Dermatol. Res. 2014, 306, 475–487. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.M.; Yun, J.; Lee, C.K.; Lee, H.; Min, K.R.; Kim, Y. Oxyresveratrol and hydroxystilbene compounds. Inhibitory effect on tyrosinase and mechanism of action. J. Biol. Chem. 2002, 277, 16340–16344. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.Y.; Liu, Q.; Kim, S.B.; Jo, Y.H.; Mo, E.J.; Yang, H.H.; Song, D.H.; Hwang, B.Y.; Lee, M.K. Characterization of Melanogenesis Inhibitory Constituents of Morus alba Leaves and Optimization of Extraction Conditions Using Response Surface Methodology. Molecules 2015, 20, 8730–8741. [Google Scholar] [CrossRef] [PubMed]
- Chao, H.C.; Najjaa, H.; Villareal, M.O.; Ksouri, R.; Han, J.; Neffati, M.; Isoda, H. Arthrophytum scoparium inhibits melanogenesis through the down-regulation of tyrosinase and melanogenic gene expressions in B16 melanoma cells. Exp. Dermatol. 2013, 22, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Demirkiran, O.; Sabudak, T.; Ozturk, M.; Topcu, G. Antioxidant and Tyrosinase Inhibitory Activities of Flavonoids from Trifolium nigrescens Subsp. petrisavi. J. Agric. Food Chem. 2013, 61, 12598–12603. [Google Scholar] [CrossRef] [PubMed]
- Godar, D.E. UV doses worldwide. Photochem. Photobiol. 2005, 81, 736–749. [Google Scholar] [CrossRef] [PubMed]
- Almeida, I.F.; Pinto, A.S.; Monteiro, C.; Monteiro, H.; Belo, L.; Fernandes, J.; Bento, A.R.; Duarte, T.L.; Garrido, J.; Bahia, M.F.; et al. Protective effect of C. sativa leaf extract against UV mediated-DNA damage in a human keratinocyte cell line. J. Photochem. Photobiol. B 2015, 144, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Godic, A.; Poljsak, B.; Adamic, M.; Dahmane, R. The role of antioxidants in skin cancer prevention and treatment. Oxid. Med. Cell. Longev. 2014, 2014, 860479. [Google Scholar] [CrossRef] [PubMed]
- Calvo-Castro, L.; Syed, D.N.; Chamcheu, J.C.; Vilela, F.M.; Perez, A.M.; Vaillant, F.; Rojas, M.; Mukhtar, H. Protective effect of tropical highland blackberry juice (Rubus adenotrichos Schltdl.) against UVB-mediated damage in human epidermal keratinocytes and in a reconstituted skin equivalent model. Photochem. Photobiol. 2013, 89, 1199–1207. [Google Scholar] [CrossRef] [PubMed]
- Svobodova, A.; Zdarilova, A.; Vostalova, J. Lonicera caerulea and Vaccinium myrtillus fruit polyphenols protect HaCaT keratinocytes against UVB-induced phototoxic stress and DNA damage. J. Dermatol. Sci. 2009, 56, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Vostalova, J.; Galandakova, A.; Palikova, I.; Ulrichova, J.; Dolezal, D.; Lichnovska, R.; Vrbkova, J.; Rajnochova Svobodova, A. Lonicera caerulea fruits reduce UVA-induced damage in hairless mice. J. Photochem. Photobiol. B 2013, 128, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Chen, F.; Wang, M. Photoprotective effects of oxyresveratrol and kuwanon O on DNA damage induced by UVA in human epidermal keratinocytes. Chem. Res. Toxicol. 2015, 28, 541–548. [Google Scholar] [CrossRef] [PubMed]
- Psotova, J.; Svobodova, A.; Kolarova, H.; Walterova, D. Photoprotective properties of prunella vulgaris and rosmarinic acid on human keratinocytes. J. Photochem. Photobiol. B 2006, 84, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Bulla, M.K.; Hernandes, L.; Baesso, M.L.; Nogueira, A.C.; Bento, A.C.; Bortoluzzi, B.B.; Serra, L.Z.; Cortez, D.A. Evaluation of Photoprotective potential and percutaneous penetration by photoacoustic spectroscopy of the schinus terebinthifolius raddi extract. Photochem. Photobiol. 2015, 91, 558–566. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.W.; Jung, E.; Kim, S.; Kim, J.H.; Kim, E.G.; Lee, J.; Park, D. Antagonizing Effects and Mechanisms of Afzelin against UVB-induced cell damage. PLoS ONE 2013, 8, e61971. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.W.; Jung, E.; Kim, S.; Lee, K.E.; Youm, J.K.; Park, D. Antagonist Effects of Veratric Acid against UVB-Induced Cell Damages. Molecules 2013, 18, 5405–5419. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.; Kum, H.; Ryu, D.; Kim, M.; Jung, E.; Park, D. Protective Effects of a New Phloretin Derivative against UVB-Induced Damage in Skin Cell Model and Human Volunteers. Int. J. Mol. Sci. 2014, 15, 18919–18940. [Google Scholar] [CrossRef] [PubMed]
- Wolfle, U.; Esser, P.R.; Simon-Haarhaus, B.; Martin, S.F.; Lademann, J.; Schempp, C.M. UVB-induced DNA damage, generation of reactive oxygen species, and inflammation are effectively attenuated by the flavonoid luteolin in vitro and in vivo. Free Radic. Biol. Med. 2011, 50, 1081–1093. [Google Scholar] [CrossRef] [PubMed]
- Lasithiotakis, K.G.; Petrakis, I.E.; Garbe, C. Cutaneous melanoma in the elderly: Epidemiology, prognosis and treatment. Melanoma Res. 2010, 20, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Erdmann, F.; Lortet-Tieulent, J.; Schuz, J.; Zeeb, H.; Greinert, R.; Breitbart, E.W.; Bray, F. International trends in the incidence of malignant melanoma 1953–2008—Are recent generations at higher or lower risk? Int. J. Cancer 2013, 132, 385–400. [Google Scholar] [CrossRef] [PubMed]
- Jemal, A.; Siegel, R.; Xu, J.; Ward, E. Cancer statistics, 2010. CA Cancer J. Clin. 2010, 60, 277–300. [Google Scholar] [CrossRef] [PubMed]
- Shoeb, M. Anticancer agents from medicinal plants Bangledesh. J. Pharmacol. 2006, 1, 35–41. [Google Scholar]
- Ouyang, L.; Luo, Y.; Tian, M.; Zhang, S.Y.; Lu, R.; Wang, J.H.; Kasimu, R.; Li, X. Plant natural products: From traditional compounds to new emerging drugs in cancer therapy. Cell Prolif. 2014, 47, 506–515. [Google Scholar] [CrossRef] [PubMed]
- Cragg, G.M.; Newman, D.J. Plants as a source of anti-cancer agents. J. Ethnopharmacol. 2005, 100, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Bush, J.A.; Cheung, K.J., Jr.; Li, G. Curcumin Induces Apoptosis in Human Melanoma Cells through a Fas Receptor/Caspase-8 Pathway Independent of p53. Exp. Cell Res. 2001, 271, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Zheng, M.; Ekmekcioglu, S.; Walch, E.T.; Tang, C.H.; Grimm, E.A. Inhibition of nuclear factor-κB and nitric oxide by curcumin induces G2/M cell cycle arrest and apoptosis in human melanoma cells. Melanoma Res. 2004, 14, 165–171. [Google Scholar] [CrossRef] [PubMed]
- Abusnina, A.; Keravis, T.; Yougbare, I.; Bronner, C.; Lugnier, C. Anti-proliferative effect of curcumin on melanoma cells is mediated by PDE1A inhibition that regulates the epigenetic integrator UHRF1. Mol. Nutr. Food Res. 2011, 55, 1677–1689. [Google Scholar] [CrossRef] [PubMed]
- Attoub, S.; Hassan, A.H.; Vanhoecke, B.; Iratni, R.; Takahashi, T.; Gaben, A.M.; Bracke, M.; Awad, S.; John, A.; Kamalboor, H.A.; et al. Inhibition of cell survival, invasion, tumor growth and histone deacetylase activity by the dietary flavonoid luteolin in human epithelioid cancer cells. Eur. J. Pharmacol. 2011, 651, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Tan, Z.; Zhang, Y.; Deng, J.; Zeng, G.; Zhang, Y. Purified vitexin compound 1 suppresses tumor growth and induces cell apoptosis in a mouse model of human choriocarcinoma. Int. J. Gynecol. Cancer 2012, 22, 360–366. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Liu, Y.E.; Cao, J.; Zeng, G.; Shen, C.; Li, Y.; Zhou, M.; Chen, Y.; Pu, W.; Potters, L.; et al. Vitexins, nature-derived lignan compounds, induce apoptosis and suppress tumor growth. Clin. Cancer Res. 2009, 15, 5161–5169. [Google Scholar] [CrossRef] [PubMed]
- Ji, B.C.; Hsu, W.H.; Yang, J.S.; Hsia, T.C.; Lu, C.C.; Chiang, J.H.; Yang, J.L.; Lin, C.H.; Lin, J.J.; Suen, L.J.; et al. Gallic acid induces apoptosis via caspase-3 and mitochondrion-dependent pathways in vitro and suppresses lung xenograft tumor growth in vivo. J. Agric. Food Chem. 2009, 57, 7596–7604. [Google Scholar] [CrossRef] [PubMed]
- Pisano, M.; Pagnan, G.; Loi, M.; Mura, M.E.; Tilocca, M.G.; Palmieri, G.; Fabbri, D.; Dettori, M.A.; Delogu, G.; Ponzoni, M.; et al. Antiproliferative and pro-apoptotic activity of eugenol-related biphenyls on malignant melanoma cells. Mol. Cancer 2007, 6, 8. [Google Scholar] [CrossRef] [PubMed]
- Kaur, G.; Athar, M.; Alam, M.S. Eugenol precludes cutaneous chemical carcinogenesis in mouse by preventing oxidative stress and inflammation and by inducing apoptosis. Mol. Carcinog. 2010, 49, 290–301. [Google Scholar] [CrossRef] [PubMed]
- Kim, G.C.; Choi, D.S.; Lim, J.S.; Jeong, H.C.; Kim, I.R.; Lee, M.H.; Park, B.S. Caspases-dependent apoptosis in human melanoma cell by eugenol. Korean J. Anat. 2006, 39, 245–253. [Google Scholar]
- Ghosh, R.; Nadiminty, N.; Fitzpatrick, J.E.; Alworth, W.L.; Slaga, T.J.; Kumar, A.P. Eugenol causes melanoma growth suppression through inhibition of E2F1 transcriptional activity. J. Biol. Chem. 2005, 280, 5812–5819. [Google Scholar] [CrossRef] [PubMed]
- Pal, D.; Banerjee, S.; Mukherjee, S.; Roy, A.; Panda, C.K.; Das, S. Eugenol restricts DMBA croton oil induced skin carcinogenesis in mice: Downregulation of c-Myc and H-ras, and activation of p53 dependent apoptotic pathway. J. Dermatol. Sci. 2010, 59, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Cunha, B.L.; Franca, J.P.; Moraes, A.A.; Chaves, A.L.; Gaiba, S.; Fontana, R.; Sacramento, C.K.; Ferreira, L.M.; Franca, L.P. Evaluation of antimicrobial and antitumoral activity of Garcinia mangostana L. (mangosteen) grown in Southeast Brazil. Acta Cir. Bras. 2014, 29 (Suppl. 2), S21–S28. [Google Scholar] [CrossRef]
- Teerasripreecha, D.; Phuwapraisirisan, P.; Puthong, S.; Kimura, K.; Okuyama, M.; Mori, H.; Kimura, A.; Chanchao, C. In vitro antiproliferative/cytotoxic activity on cancer cell lines of a cardanol and a cardol enriched from Thai Apis mellifera propolis. BMC Complement. Altern. Med. 2012, 12, 27. [Google Scholar] [CrossRef] [PubMed]
- Umthong, S.; Phuwapraisirisan, P.; Puthong, S.; Chanchao, C. In vitro antiproliferative activity of partially purified Trigona laeviceps propolis from thailand on human cancer cell lines. BMC Complement. Altern. Med. 2011, 11, 37. [Google Scholar] [CrossRef] [PubMed]
- Pratsinis, H.; Kletsas, D.; Melliou, E.; Chinou, I. Antiproliferative Activity of Greek Propolis. J. Med. Food 2010, 13, 286–290. [Google Scholar] [CrossRef] [PubMed]
- Szliszka, E.; Krol, W. Polyphenols isolated from propolis augment TRAIL-induced apoptosis in cancer cells. Evid. Based Complement. Alternat. Med. 2013, 2013, 731940. [Google Scholar] [CrossRef] [PubMed]
- Kubina, R.; Kabała-Dzik, A.; Bułdak, R.J.; Szaflarska-Stojko, E.; Tylka, P.; Bielec, B.; Stawiarska-Pięta, B. The in vitro proapoptotic activities evaluation of ethanolic extract of propolis. Post. Fitoter. 2011, 4, 232–237. [Google Scholar]
- Rajeshkumar, N.V.; Joy, K.L.; Kuttan, G.; Ramsewak, R.S.; Nair, M.G.; Kuttan, R. Antitumour and anticarcinogenic activity of Phyllanthusamarus extract. J. Ethnopharmacol. 2002, 81, 17–22. [Google Scholar] [CrossRef]
- Ren, W.; Qiao, Z.; Wang, H.; Zhu, L.; Zhang, L. Flavonoids: Promising anticancer agents. Med. Res. Rev. 2003, 23, 519–534. [Google Scholar] [CrossRef] [PubMed]
- Mailand, N.; Falck, J.; Lukas, C.; Syljuasen, R.G.; Welcker, M.; Bartek, J.; Lukas, J. Rapid destruction of human Cdc25A in response to DNA damage. Science 2000, 288, 1425–1429. [Google Scholar] [CrossRef] [PubMed]
- Madson, J.G.; Lynch, D.T.; Svoboda, J.; Ophardt, R.; Yanagida, J.; Putta, S.K.; Bowles, A.; Trempus, C.S.; Tennant, R.W.; Hansen, L.A. Erbb2 suppresses DNA damage-induced checkpoint activation and UV-induced mouse skin tumorigenesis. Am. J. Pathol. 2009, 174, 2357–2366. [Google Scholar] [CrossRef] [PubMed]
- Puri, N.; Eller, M.S.; Byers, H.R.; Dykstra, S.; Kubera, J.; Gilchrest, B.A. Telomere-based DNA damage responses: A new approach to melanoma. FASEB J. 2004, 18, 1373–1381. [Google Scholar] [CrossRef] [PubMed]
- Baek, S.J.; Kim, J.S.; Jackson, F.R.; Eling, T.E.; McEntee, M.F.; Lee, S.H. Epicatechin gallate-induced expression of NAG-1 is associated with growth inhibition and apoptosis in colon cancer cells. Carcinogenesis 2004, 25, 2425–2432. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.Q.; Jaganath, I.B.; Sekaran, S.D. Phyllanthus spp. Induces selective growth inhibition of PC-3 and MeWo human cancer cells through modulation of cell cycle and induction of apoptosis. PLoS ONE 2010, 5, e12644. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Fu, Y.; Malakhova, M.; Kurinov, I.; Zhu, F.; Yao, K.; Li, H.; Chen, H.; Li, W.; Lim do, Y.; et al. Caffeic acid directly targets ERK1/2 to attenuate solar UV-induced skin carcinogenesis. Cancer Prev. Res. 2014, 7, 1056–1066. [Google Scholar] [CrossRef] [PubMed]
- McCubrey, J.A.; Steelman, L.S.; Chappell, W.H.; Abrams, S.L.; Wong, E.W.; Chang, F.; Lehmann, B.; Terrian, D.M.; Milella, M.; Tafuri, A.; et al. Roles of the Raf/MEK/ERK pathway in cell growth, malignant transformation and drug resistance. Biochim. Biophys. Acta 2007, 1773, 1263–1284. [Google Scholar] [CrossRef] [PubMed]
- Dhillon, A.S.; Hagan, S.; Rath, O.; Kolch, W. MAP kinase signalling pathways in cancer. Oncogene 2007, 26, 3279–3290. [Google Scholar] [CrossRef] [PubMed]
- Ohori, M.; Kinoshita, T.; Okubo, M.; Sato, K.; Yamazaki, A.; Arakawa, H.; Nishimura, S.; Inamura, N.; Nakajima, H.; Neya, M.; et al. Identification of a selective ERK inhibitor and structural determination of the inhibitor-ERK2 complex. Biochem. Biophys. Res. Commun. 2005, 336, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Russo, A.E.; Torrisi, E.; Bevelacqua, Y.; Perrotta, R.; Libra, M.; McCubrey, J.A.; Spandidos, D.A.; Stivala, F.; Malaponte, G. Melanoma: Molecular pathogenesis and emerging target therapies (Review). Int. J. Oncol. 2009, 34, 1481–1489. [Google Scholar] [PubMed]
- Einspahr, J.G.; Calvert, V.; Alberts, D.S.; Curiel-Lewandrowski, C.; Warneke, J.; Krouse, R.; Stratton, S.P.; Liotta, L.; Longo, C.; Pellacani, G.; et al. Functional protein pathway activation mapping of the progression of normal skin to squamous cell carcinoma. Cancer Prev. Res. 2012, 5, 403–413. [Google Scholar] [CrossRef] [PubMed]
- Berwick, M.; Lachiewicz, A.; Pestak, C.; Thomas, N. Solar UV exposure and mortality from skin tumors. Adv. Exp. Med. Biol. 2008, 624, 117–124. [Google Scholar] [PubMed]
- De Gruijl, F.R. Skin cancer and solar UV radiation. Eur. J. Cancer 1999, 35, 2003–2009. [Google Scholar] [CrossRef]
- Huang, M.T.; Smart, R.C.; Wong, C.Q.; Conney, A.H. Inhibitory effect of curcumin, chlorogenic acid, caffeic acid, and ferulic acid on tumor promotion in mouse skin by 12-O-tetradecanoylphorbol-13-acetate. Cancer Res. 1988, 48, 5941–5946. [Google Scholar] [PubMed]
- Staniforth, V.; Chiu, L.T.; Yang, N.S. Caffeic acid suppresses UVB radiation-induced expression of interleukin-10 and activation of mitogen-activated protein kinases in mouse. Carcinogenesis 2006, 27, 1803–1811. [Google Scholar] [CrossRef]
- Wu, L.; Hsu, H.; Chen, Y.; Chiu, C.; Lin, Y.; Ho, J.A. Antioxidant and antiproliferative activities of red pitaya. Food Chem. 2006, 95, 319–327. [Google Scholar] [CrossRef]
- Montezano, A.C.; Touyz, R.M. Molecular mechanisms of hypertension—Reactive oxygen species and antioxidants: A basic science update for the clinician. Can. J. Cardiol. 2012, 28, 288–295. [Google Scholar] [CrossRef] [PubMed]
- Seifried, H.E.; Anderson, D.E.; Fisher, E.I.; Milner, J.A. A review of the interaction among dietary antioxidants and reactive oxygen species. J. Nutr. Biochem. 2007, 18, 567–579. [Google Scholar] [CrossRef] [PubMed]
- Kardeh, S.; Ashkani-Esfahani, S.; Alizadeh, A.M. Paradoxical action of reactive oxygen species in creation and therapy of cancer. Eur. J. Pharmacol. 2014, 735, 150–168. [Google Scholar] [CrossRef] [PubMed]
- Bauman, A.E. Updating the evidence that physical activity is good for health: An epidemiological review 2000–2003. J. Sci. Med. Sport 2004, 7, 6–19. [Google Scholar] [CrossRef]
- Chen, A.Y.; Chen, Y.C. A review of the dietary flavonoid, kaempferol on human health and cancer chemoprevention. Food Chem. 2013, 138, 2099–2107. [Google Scholar] [CrossRef] [PubMed]
- Doria, G.A.; Santos, A.R.; Bittencourt, L.S.; Bortolin, R.C.; Menezes, P.P.; Vasconcelos, B.S.; Souza, R.O.; Fonseca, M.J.; Santos, A.D.; Saravanan, S.; et al. Redox-active profile characterization of Remirea maritima extracts and its cytotoxic effect in mouse fibroblasts (L929) and melanoma (B16F10) cells. Molecules 2015, 20, 11699–11718. [Google Scholar] [CrossRef] [PubMed]
- Caruso, I.P.; Vilegas, W.; de Souza, F.P.; Fossey, M.A.; Cornelio, M.L. Binding of antioxidant flavone isovitexin to human serum albumin investigated by experimental and computational assays. J. Pharm. Biomed. Anal. 2014, 98, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Wu, W.; Li, D.; Xu, T.; Zhu, H.; Pan, D.; Zhu, S.; Liu, Y. Anti-oxidant and anti-apoptotic effects of luteolin on mice peritoneal macrophages stimulated by angiotensin II. Int. Immunopharmacol. 2014, 20, 346–351. [Google Scholar] [CrossRef] [PubMed]
- Horvathova, K.; Chalupa, I.; Sebova, L.; Tothova, D.; Vachalkova, A. Protective effect of quercetin and luteolin in human melanoma HMB-2 cells. Mutat. Res. 2005, 565, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.T.; Chen, C.T.; Chieng, K.T.; Huang, S.H.; Chiang, B.H.; Wang, L.F.; Kuo, H.S.; Lin, C.M. Inhibitory effects of a rice hull constituent on tumor necrosis factor α, prostaglandin E2, and cyclooxygenase-2 production in lipopolysaccharide-activated mouse macrophages. Ann. N. Y. Acad. Sci. 2005, 1042, 387–395. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kim, Y.J. Hyperin and quercetin modulate oxidative stress-induced melanogenesis. Biol. Pharm. Bull. 2012, 35, 2023–2027. [Google Scholar] [CrossRef] [PubMed]
- Panich, U.; Pluemsamran, T.; Tangsupa-a-nan, V.; Wattanarangsan, J.; Phadungrakwittaya, R.; Akarasereenont, P.; Laohapand, T. Protective effect of AVS073, a polyherbal formula, against UVA-induced melanogenesis through a redox mechanism involving glutathione-related antioxidant defense. BMC Complement. Altern. Med. 2013, 13, 159. [Google Scholar] [CrossRef] [PubMed]
- Rees, J.L. Genetics of hair and skin color. Annu. Rev. Genet. 2003, 37, 67–90. [Google Scholar] [CrossRef] [PubMed]
- Parvez, S.; Kang, M.; Chung, H.S.; Bae, H. Naturally occurring tyrosinase inhibitors: Mechanism and applications in skin health, cosmetics and agriculture industries. Phytother. Res. 2007, 21, 805–816. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Uyama, H. Tyrosinase inhibitors from natural and synthetic sources: Structure, inhibition mechanism and perspective for the future. Cell. Mol. Life Sci. 2005, 62, 1707–1723. [Google Scholar] [CrossRef]
- Li, H.R.; Habasi, M.; Xie, L.Z.; Aisa, H.A. Effect of chlorogenic acid on melanogenesis of B16 melanoma cells. Molecules 2014, 19, 12940–12948. [Google Scholar] [CrossRef] [PubMed]
- Fujii, T.; Saito, M. Inhibitory effect of quercetin isolated from rose hip (Rosa canina L.) against melanogenesis by mouse melanoma cells. Biosci. Biotechnol. Biochem. 2009, 73, 1989–1993. [Google Scholar] [CrossRef] [PubMed]
- Arung, E.T.; Furuta, S.; Ishikawa, H.; Kusuma, I.W.; Shimizu, K.; Kondo, R. Anti-melanogenesis properties of quercetin- and its derivative-rich extract from Allium cepa. Food Chem. 2011, 124, 1024–1028. [Google Scholar] [CrossRef]
- Su, T.R.; Lin, J.J.; Tsai, C.C.; Huang, T.K.; Yang, Z.Y.; Wu, M.O.; Zheng, Y.Q.; Su, C.C.; Wu, Y.J. Inhibition of melanogenesis by gallic acid: Possible involvement of the PI3K/Akt, MEK/ERK and Wnt/β-catenin signaling pathways in B16F10 cells. Int. J. Mol. Sci. 2013, 14, 20443–20458. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.C.; Hsieh, W.Y.; Niu, Y.L.; Chang, T.M. Inhibition of melanogenesis and antioxidant properties of Magnolia grandiflora L. Flower extract. BMC Complement. Altern. Med. 2012, 12, 72. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.C.; Chiu, S.H.; Chang, T.M. Inhibitory effect of [6]-gingerol on melanogenesis in B16F10 melanoma cells and a possible mechanism of action. Biosci. Biotechnol. Biochem. 2011, 75, 1067–1072. [Google Scholar] [CrossRef] [PubMed]
- Almond, J.B.; Cohen, G.M. The proteasome: A novel target for cancer chemotherapy. Leukemia 2002, 16, 433–443. [Google Scholar] [CrossRef] [PubMed]
- Kazi, A.; Urbizu, D.A.; Kuhn, D.J.; Acebo, A.L.; Jackson, E.R.; Greenfelder, G.P.; Kumar, N.B.; Dou, Q.P. A natural musaceas plant extract inhibits proteasome activity and induces apoptosis selectively in human tumor and transformed, but not normal and non-transformed, cells. Int. J. Mol. Med. 2003, 12, 879–887. [Google Scholar] [CrossRef] [PubMed]
- Jung, T.; Catalgol, B.; Grune, T. The proteasomal system. Mol. Asp. Med. 2009, 30, 191–296. [Google Scholar] [CrossRef] [PubMed]
- Orabi, K.Y.; Abaza, M.S.; El Sayed, K.A.; Elnagar, A.Y.; Al-Attiyah, R.; Guleri, R.P. Selective growth inhibition of human malignant melanoma cells by syringic acid-derived proteasome inhibitors. Cancer Cell Int. 2013, 13, 82. [Google Scholar] [CrossRef] [PubMed]
- Wan, S.B.; Chen, D.; Dou, Q.P.; Chan, T.H. Study of the green tea polyphenols catechin-3-gallate (CG) and epicatechin-3-gallate (ECG) as proteasome inhibitors. Bioorg. Med. Chem. 2004, 12, 3521–3527. [Google Scholar] [CrossRef] [PubMed]
- Osanai, K.; Landis-Piwowar, K.R.; Dou, Q.P.; Chan, T.H. A para-amino substituent on the D-ring of green tea polyphenol epigallocatechin-3-gallate as a novel proteasome inhibitor and cancer cell apoptosis inducer. Bioorg. Med. Chem. 2007, 15, 5076–5082. [Google Scholar] [CrossRef] [PubMed]
- Pettinari, A.; Amici, M.; Cuccioloni, M.; Angeletti, M.; Fioretti, E.; Eleuteri, A.M. Effect of polyphenolic compounds on the proteolytic activities of constitutive and immuno-proteasomes. Antioxid. Redox Signal. 2006, 8, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Daniel, K.G.; Chen, M.S.; Kuhn, D.J.; Landis-Piwowar, K.R.; Dou, Q.P. Dietary flavonoids as proteasome inhibitors and apoptosis inducers in human leukemia cells. Biochem. Pharmacol. 2005, 69, 1421–1432. [Google Scholar] [CrossRef] [PubMed]
- Dikshit, P.; Goswami, A.; Mishra, A.; Chatterjee, M.; Jana, N.R. Curcumin induces stress response, neurite outgrowth and prevent NF-κB activation by inhibiting the proteasome function. Neurotox Res 2006, 9, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Kazi, A.; Daniel, K.G.; Smith, D.M.; Kumar, N.B.; Dou, Q.P. Inhibition of the proteasome activity, a novel mechanism associated with the tumor cell apoptosis-inducing ability of genistein. Biochem. Pharmacol. 2003, 66, 965–976. [Google Scholar] [CrossRef]
- Mena, S.; Rodriguez, M.L.; Ponsoda, X.; Estrela, J.M.; Jaattela, M.; Ortega, A.L. Pterostilbene-induced tumor cytotoxicity: A lysosomal membrane permeabilization-dependent mechanism. PLoS ONE 2012, 7, e44524. [Google Scholar] [CrossRef] [PubMed]
- Ferrer, P.; Asensi, M.; Priego, S.; Benlloch, M.; Mena, S.; Ortega, A.; Obrador, E.; Esteve, J.M.; Estrela, J.M. Nitric oxide mediates natural polyphenol-induced Bcl-2 down-regulation and activation of cell death in metastatic B16 melanoma. J. Biol. Chem. 2007, 282, 2880–2890. [Google Scholar] [CrossRef] [PubMed]
- Mizushima, N. Autophagy: Process and function. Genes Dev. 2007, 21, 2861–2873. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.J.; Ho, C.T.; Wang, Y.J. Pterostilbene induces autophagy and apoptosis in sensitive and chemoresistant human bladder cancer cells. Mol. Nutr. Food Res. 2010, 54, 1819–1832. [Google Scholar] [CrossRef] [PubMed]
- Diffey, B. The contribution of medical physics to the development of psoralen photochemotherapy (PUVA) in the UK: A personal reminiscence. Phys. Med. Biol. 2006, 51, R229–R244. [Google Scholar] [CrossRef] [PubMed]
- Menichini, G.; Alfano, C.; Provenzano, E.; Marrelli, M.; Statti, G.A.; Menichini, F.; Conforti, F. Cachrys pungens jan inhibits human melanoma cell proliferation through photo-induced cytotoxic activity. Cell Prolif. 2012, 45, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Tram, N.T.; Le Son, H. Assessment of anti-psoriatic activity of Cassia fistula L. Extract incorporated cream. Br. J. Pharm. Res. 2015, 5, 370–378. [Google Scholar] [CrossRef]
- Rahman, M.; Alam, K.; Ahmad, M.Z.; Gupta, G.; Afzal, M.; Akhter, S.; Kazmi, I.; Jyoti; Ahmad, F.J.; Anwar, F. Classical to current approach for treatment of psoriasis: A review. Endocr. Metab. Immune Disord. Drug Targets 2012, 12, 287–302. [Google Scholar] [CrossRef] [PubMed]
- DiSepio, D.; Chandraratna, R.A.; Nagpal, S. Novel approaches for the treatment of psoriasis. Drug Discov. Today 1999, 4, 222–231. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the last 25 years. J. Nat. Prod. 2007, 70, 461–477. [Google Scholar] [CrossRef] [PubMed]
- Ellis, C.N.; Berberian, B.; Sulica, V.I.; Dodd, W.A.; Jarratt, M.T.; Katz, H.I.; Prawer, S.; Krueger, G.; Rex, I.H., Jr.; Wolf, J.E. A double-blind evaluation of topical capsaicin in pruritic psoriasis. J. Am. Acad. Dermatol. 1993, 29, 438–442. [Google Scholar] [CrossRef]
- Bhatti, M.Z.; Ali, A.; Ahmad, A.; Saeed, A.; Malik, S.A. Antioxidant and phytochemical analysis of Ranunculus arvensis L. Extracts. BMC Res. Notes 2015, 8, 279. [Google Scholar] [CrossRef] [PubMed]
- Campos, M.G.; Webby, R.F.; Markham, K.R.; Mitchell, K.A.; Da Cunha, A.P. Age-induced diminution of free radical scavenging capacity in bee pollens and the contribution of constituent flavonoids. J. Agric. Food Chem. 2003, 51, 742–745. [Google Scholar] [CrossRef] [PubMed]
- Noor, W.; Gul, R.; Ali, I.; Choudary, M.I. Isolation and antibacterial activity of the compound from Ranunculus repens L. J. Chem. Soc. Pak. 2006, 28, 272–274. [Google Scholar]
- Prieto, J.M.; Recio, M.C.; Giner, R.M.; Schinella, G.R.; Manez, S.; Rios, J.L. In vitro and in vivo effects of Ranunculus peltatus subsp. baudotii methanol extract on models of eicosanoid production and contact dermatitis. Phytother. Res. 2008, 22, 297–302. [Google Scholar] [PubMed]
- Botes, L.; van der Westhuizen, F.H.; Loots du, T. Phytochemical contents and antioxidant capacities of two Aloe greatheadii var. davyana extracts. Molecules 2008, 13, 2169–2180. [Google Scholar] [CrossRef] [PubMed]
- Musthaba, S.M.; Athar, M.T.; Kamal, Y.T.; Baboota, S.; Ali, J.; Ahmad, S. Fast analysis and validation of rutin in anti-psoriatic ayurvedic formulation by HPLC. J. Liq. Chromatogr. Relat. Technol. 2011, 34, 446–455. [Google Scholar] [CrossRef]
- Dhanabal, S.P.; Raj, B.A.; Muruganantham, N.; Praveen, T.K.; Raghu, P.S. Screening of Wrightia tinctoria leaves for anti psoriatic activity. Hygeia 2012, 4, 73–78. [Google Scholar]
- Chen, S.D.; Lu, C.J.; Zhao, R.Z. Identification and quantitative characterization of PSORI-CM01, a chinese medicine formula for psoriasis therapy, by liquid chromatography coupled with an LTQ orbitrap mass spectrometer. Molecules 2015, 20, 1594–1609. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Li, H.; Li, M. Effects of baicalin cream in two mouse models: 2,4-Dinitrofluorobenzene-induced contact hypersensitivity and mouse tail test for psoriasis. Int. J. Clin. Exp. Med. 2015, 8, 2128–2137. [Google Scholar] [PubMed]
- Maier, L. Rosacea: Advances in understanding pathogenesis and treatment. J. Clin. Investig. 2011, 1, 739–755. [Google Scholar] [CrossRef]
- Odom, R.B. The subtypes of rosacea: Implications for treatment. Cutis 2004, 73 (Suppl. 1), S9–S14. [Google Scholar]
- Gupta, A.K.; Chaudhry, M.M. Rosacea and its management: An overview. J. Eur. Acad. Dermatol. Venereol. 2005, 19, 273–285. [Google Scholar] [CrossRef] [PubMed]
- Domingo, D.S.; Camouse, M.M.; Hsia, A.H.; Matsui, M.; Maes, D.; Ward, N.L.; Cooper, K.D.; Baron, E.D. Anti-angiogenic effects of epigallocatechin-3-gallate in human skin. Int. J. Clin. Exp. Pathol. 2010, 3, 705–709. [Google Scholar] [PubMed]
- Detmar, M. The role of vegf and thrombospondins in skin angiogenesis. J. Dermatol. Sci. 2000, 24 (Suppl. 1), S78–S84. [Google Scholar] [CrossRef]
- Zhang, Q.; Tang, X.; Lu, Q.; Zhang, Z.; Rao, J.; Le, A.D. Green tea extract and (−)-epigallocatechin-3-gallate inhibit hypoxia- and serum-induced HIF-1α protein accumulation and VEGF expression in human cervical carcinoma and hepatoma cells. Mol. Cancer Ther. 2006, 5, 1227–1238. [Google Scholar] [CrossRef] [PubMed]
- Rigopoulos, D.; Kalogeromitros, D.; Gregoriou, S.; Pacouret, J.M.; Koch, C.; Fisher, N.; Bachmann, K.; Brown, M.; Schwarz, E.; Camel, E.; et al. Randomized placebo-controlled trial of a flavonoid-rich plant extract-based cream in the treatment of rosacea. J. Eur. Acad. Dermatol. Venereol. 2005, 19, 564–568. [Google Scholar] [CrossRef] [PubMed]
- Toyoda, M.; Morohashi, M. Pathogenesis of acne. Med. Electron. Microsc. 2001, 34, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, T.; Kokubo, R.; Sakaino, M. Antimicrobial activities of eucalyptus leaf extracts and flavonoids from Eucalyptus maculata. Lett. Appl. Microbiol. 2004, 39, 60–64. [Google Scholar] [CrossRef] [PubMed]
- Vijayalakshmi, A.; Tripura, A.; Ravichandiran, V. Development and evaluation of anti-acne products from Terminalia arjuna bark. Int. J. ChemTech. Res. 2011, 3, 320–327. [Google Scholar]
- Pothitirat, W.; Chomnawang, M.T.; Supabphol, R.; Gritsanapan, W. Free radical scavenging and anti-acne activities of mangosteen fruit rind extracts prepared by different extraction methods. Pharm. Biol. 2010, 48, 182–186. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.C.; Tsai, T.H.; Huang, C.J.; Li, Y.Y.; Chyuan, J.H.; Chuang, L.T.; Tsai, P.J. Inhibitory effects of wild bitter melon leaf extract on Propionibacterium acnes-induced skin inflammation in mice and cytokine production in vitro. Food Funct. 2015, 6, 2550–2560. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Lee, J.; Jung, E.; Park, Y.; Kim, K.; Park, B.; Jung, K.; Park, E.; Kim, J.; Park, D. In vitro antibacterial and anti-inflammatory effects of honokiol and magnolol against Propionibacterium sp. Eur. J. Pharmacol. 2004, 496, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Lim, Y.H.; Kim, I.H.; Seo, J.J. In vitro activity of kaempferol isolated from the impatiens balsamina alone and in combination with erythromycin or clindamycin against Propionibacterium acnes. J. Microbiol. 2007, 45, 473–477. [Google Scholar] [PubMed]
- Yoon, J.Y.; Kwon, H.H.; Min, S.U.; Thiboutot, D.M.; Suh, D.H. Epigallocatechin-3-gallate improves acne in humans by modulating intracellular molecular targets and inhibiting P. acnes. J. Investig. Dermatol. 2013, 133, 429–440. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, T.; Matsubara, Y.; Mizuhara, Y.; Sekiguchi, K.; Koseki, J.; Tsuchiya, K.; Nishimura, H.; Watanabe, J.; Kaneko, A.; Maemura, K.; et al. Plasma pharmacokinetics of polyphenols in a traditional japanese medicine, jumihaidokuto, which suppresses Propionibacterium acnes-induced dermatitis in rats. Molecules 2015, 20, 18031–18046. [Google Scholar] [CrossRef] [PubMed]
- Tohno, H.; Horii, C.; Fuse, T.; Okonogi, A.; Yomoda, S. Evaluation of estrogen receptor beta binding of pruni cortex and its constituents. Yakugaku Zasshi 2010, 130, 989–997. [Google Scholar] [CrossRef] [PubMed]
- Devereux, G.; Seaton, A. Diet as a risk factor for atopy and asthma. J. Allergy Clin. Immunol. 2005, 115, 1109–1117. [Google Scholar] [CrossRef] [PubMed]
- Shaheen, S.O.; Sterne, J.A.; Thompson, R.L.; Songhurst, C.E.; Margetts, B.M.; Burney, P.G. Dietary antioxidants and asthma in adults: Population-based case-control study. Am. J. Respir. Crit. Care Med. 2001, 164, 1823–1828. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Kouda, K.; Kotani, M.; Takeuchi, A.; Tabei, T.; Masamoto, Y.; Nakamura, H.; Takigawa, M.; Suemura, M.; Takeuchi, H.; et al. Vegetarian diet ameliorates symptoms of atopic dermatitis through reduction of the number of peripheral eosinophils and of PGE2 synthesis by monocytes. J. Physiol. Anthropol. Appl. Hum. Sci. 2001, 20, 353–361. [Google Scholar] [CrossRef]
- Kawai, M.; Hirano, T.; Higa, S.; Arimitsu, J.; Maruta, M.; Kuwahara, Y.; Ohkawara, T.; Hagihara, K.; Yamadori, T.; Shima, Y.; et al. Flavonoids and related compounds as anti-allergic substances. Allergol. Int. 2007, 56, 113–123. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T. Flavonoids for allergic diseases: Present evidence and future perspective. Curr. Pharm. Des. 2014, 20, 879–885. [Google Scholar] [CrossRef] [PubMed]
- Oku, H.; Ishiguro, K. Antipruritic and antidermatitic effect of extract and compounds of Impatiens balsamina L. In atopic dermatitis model NC mice. Phytother. Res. 2001, 15, 506–510. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Lim, S.J.; Kang, S.W.; Um, B.H.; Nho, C.W. Aceriphyllum rossii extract and its active compounds, quercetin and kaempferol inhibit IgE-mediated mast cell activation and passive cutaneous anaphylaxis. J. Agric. Food Chem. 2014, 62, 3750–3758. [Google Scholar] [CrossRef] [PubMed]
- Mehrotra, P.; Mishra, K.P.; Raman, G.; Banerjee, G. Differential regulation of free radicals (reactive oxygen and nitrogen species) by contact allergens and irritants in human keratinocyte cell line. Toxicol. Mech. Methods 2005, 15, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Fu, R.; Zhang, Y.; Peng, T.; Guo, Y.; Chen, F. Phenolic composition and effects on allergic contact dermatitis of phenolic extracts Sapium sebiferum (L.) Roxb. Leaves. J. Ethnopharmacol. 2015, 162, 176–180. [Google Scholar] [CrossRef] [PubMed]
- Bieber, T. Atopic dermatitis. N. Engl. J. Med. 2008, 358, 1483–1494. [Google Scholar] [CrossRef] [PubMed]
- Leung, D.Y.; Soter, N.A. Cellular and immunologic mechanisms in atopic dermatitis. J. Am. Acad. Dermatol. 2001, 44 (Suppl. 1), S1–S12. [Google Scholar] [CrossRef] [PubMed]
- Cabrini, D.A.; Moresco, H.H.; Imazu, P.; da Silva, C.D.; Pietrovski, E.F.; Mendes, D.A.; da Silveira Prudente, A.; Pizzolatti, M.G.; Brighente, I.M.; Otuki, M.F. Analysis of the potential topical anti-inflammatory activity of Averrhoa carambola L. in mice. Evid. Based Complement. Alternat. Med. 2011, 2011, 908059. [Google Scholar] [CrossRef] [PubMed]
- Tadros, S.H.; Sleem, A.A. Pharmacognostical and biological study of the stem and leaf of Avehrroa carambola L. Bull. Fac. Pharm. 2004, 42, 225–246. [Google Scholar]
- Park, S.J.; Lee, Y.H.; Lee, K.H.; Kim, T.J. Effect of eriodictyol on the development of atopic dermatitis-like lesions in ICR mice. Biol. Pharm. Bull. 2013, 36, 1375–1379. [Google Scholar] [CrossRef] [PubMed]
- Park, K.H.; Jeong, M.S.; Park, K.J.; Choi, Y.W.; Seo, S.J.; Lee, M.W. Topical application of Rosa multiflora root extract improves atopic dermatitis-like skin lesions induced by mite antigen in NC/Nga mice. Biol. Pharm. Bull. 2014, 37, 178–183. [Google Scholar] [CrossRef] [PubMed]
- Hou, M.; Sun, R.; Hupe, M.; Kim, P.L.; Park, K.; Crumrine, D.; Lin, T.K.; Santiago, J.L.; Mauro, T.M.; Elias, P.M.; et al. Topical apigenin improves epidermal permeability barrier homoeostasis in normal murine skin by divergent mechanisms. Exp. Dermatol. 2013, 22, 210–215. [Google Scholar] [CrossRef] [PubMed]
- Jung, M.K.; Hur, D.Y.; Song, S.B.; Park, Y.; Kim, T.S.; Bang, S.I.; Kim, S.; Song, H.K.; Park, H.; Cho, D.H. Tannic acid and quercetin display a therapeutic effect in atopic dermatitis via suppression of angiogenesis and TARC expression in Nc/Nga mice. J. Investig. Dermatol. 2010, 130, 1459–1463. [Google Scholar] [CrossRef] [PubMed]
- Kang, G.J.; Han, S.C.; Ock, J.W.; Kang, H.K.; Yoo, E.S. Anti-inflammatory effect of quercetagetin, an active component of immature Citrus unshiu, in HaCaT human keratinocytes. Biomol. Ther. 2013, 21, 138–145. [Google Scholar] [CrossRef] [PubMed]
- Weitzman, I.; Summerbell, R.C. The dermatophytes. Clin. Microbiol. Rev. 1995, 8, 240–259. [Google Scholar] [CrossRef]
- Park, B.J.; Taguchi, H.; Kamei, K.; Matsuzawa, T.; Hyon, S.H.; Park, J.C. In vitro antifungal activity of epigallocatechin 3-O-gallate against clinical isolates of dermatophytes. Yonsei Med. J. 2011, 52, 535–538. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, S.; Kanoya, Y.; Nagata, S. Effects of a foot bath containing green tea polyphenols on interdigital tinea pedis. Foot 2013, 23, 58–62. [Google Scholar] [CrossRef] [PubMed]
- Sharma, B.; Kumar, P.; Joshi, S.C. Topical treatment of dermatophytic lesion on mice (Mus musculus) model. Indian J. Microbiol. 2011, 51, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.; Kumar, P.; Joshi, S.C. Treatment of dermatophytosis by a new antifungal agent “apigenin”. Mycoses 2014, 57, 497–506. [Google Scholar] [CrossRef] [PubMed]
- Raut, J.S.; Shinde, R.B.; Chauhan, N.M.; Karuppayil, S.M. Phenylpropanoids of plant origin as inhibitors of biofilm formation by Candida albicans. J. Microbiol. Biotechnol. 2014, 24, 1216–1225. [Google Scholar] [CrossRef] [PubMed]
- Thang, P.T.; Patrick, S.; Teik, L.S.; Yung, C.S. Anti-oxidant effects of the extracts from the leaves of Chromolaena odorata on human dermal fibroblasts and epidermal keratinocytes against hydrogen peroxide and hypoxanthine—xanthine oxidase induced damage. Burns 2001, 27, 319–327. [Google Scholar] [CrossRef]
- Erik Klein, E.; Lukeš, V.; Cibulková, Z.; Aherne, S.A. On the energetics of phenol antioxidants activity. Petroleum Coal 2005, 47, 33–39. [Google Scholar]
- Furumoto, T.; Ozawa, N.; Inami, Y.; Toyoshima, M.; Fujita, K.; Zaiki, K.; Sahara, S.; Akita, M.; Kitamura, K.; Nakaoji, K.; et al. Mallotus philippinensis bark extracts promote preferential migration of mesenchymal stem cells and improve wound healing in mice. Phytomedicine 2014, 21, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Albaayit, S.F.; Abba, Y.; Rasedee, A.; Abdullah, N. Effect of Clausena excavata Burm. f. (Rutaceae) leaf extract on wound healing and antioxidant activity in rats. Drug Des. Dev. Ther. 2015, 9, 3507–3518. [Google Scholar]
- Pessoa, W.S.; Estevao, L.R.; Simoes, R.S.; Mendonca Fde, S.; Evencio-Luz, L.; Baratella-Evencio, L.; Florencio-Silva, R.; Sa, F.B.; Evencio-Neto, J. Fibrogenesis and epithelial coating of skin wounds in rats treated with angico extract (Anadenanthera colubrina var. Cebil). Acta Cir. Bras. 2015, 30, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Geethalakshmi, R.; Sakravarthi, C.; Kritika, T.; Arul Kirubakaran, M.; Sarada, D.V. Evaluation of antioxidant and wound healing potentials of Sphaeranthus amaranthoides Burm.f. Biomed. Res. Int. 2013, 2013, 607109. [Google Scholar] [CrossRef] [PubMed]
- Nyanhongo, G.S.; Sygmund, C.; Ludwig, R.; Prasetyo, E.N.; Guebitz, G.M. An antioxidant regenerating system for continuous quenching of free radicals in chronic wounds. Eur. J. Pharm. Biopharm. 2013, 83, 396–404. [Google Scholar] [CrossRef] [PubMed]
- Skorkowska-Telichowska, K.; Zuk, M.; Kulma, A.; Bugajska-Prusak, A.; Ratajczak, K.; Gasiorowski, K.; Kostyn, K.; Szopa, J. New dressing materials derived from transgenic flax products to treat long-standing venous ulcers—A pilot study. Wound Repair. Regen. 2010, 18, 168–179. [Google Scholar] [CrossRef] [PubMed]
- Skorkowska-Telichowska, K.; Kulma, A.; Zuk, M.; Czuj, T.; Szopa, J. The effects of newly developed linen dressings on decubitus ulcers. J. Palliat. Med. 2012, 15, 146–148. [Google Scholar] [CrossRef] [PubMed]
- Bahramsoltani, R.; Farzaei, M.H.; Rahimi, R. Medicinal plants and their natural components as future drugs for the treatment of burn wounds: An integrative review. Arch. Dermatol. Res. 2014, 306, 601–617. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Działo, M.; Mierziak, J.; Korzun, U.; Preisner, M.; Szopa, J.; Kulma, A. The Potential of Plant Phenolics in Prevention and Therapy of Skin Disorders. Int. J. Mol. Sci. 2016, 17, 160. https://doi.org/10.3390/ijms17020160
Działo M, Mierziak J, Korzun U, Preisner M, Szopa J, Kulma A. The Potential of Plant Phenolics in Prevention and Therapy of Skin Disorders. International Journal of Molecular Sciences. 2016; 17(2):160. https://doi.org/10.3390/ijms17020160
Chicago/Turabian StyleDziało, Magdalena, Justyna Mierziak, Urszula Korzun, Marta Preisner, Jan Szopa, and Anna Kulma. 2016. "The Potential of Plant Phenolics in Prevention and Therapy of Skin Disorders" International Journal of Molecular Sciences 17, no. 2: 160. https://doi.org/10.3390/ijms17020160
APA StyleDziało, M., Mierziak, J., Korzun, U., Preisner, M., Szopa, J., & Kulma, A. (2016). The Potential of Plant Phenolics in Prevention and Therapy of Skin Disorders. International Journal of Molecular Sciences, 17(2), 160. https://doi.org/10.3390/ijms17020160