Characterizing Non-Tuberculous Mycobacteria Infection in Bronchiectasis
Abstract
:1. Introduction
2. Results
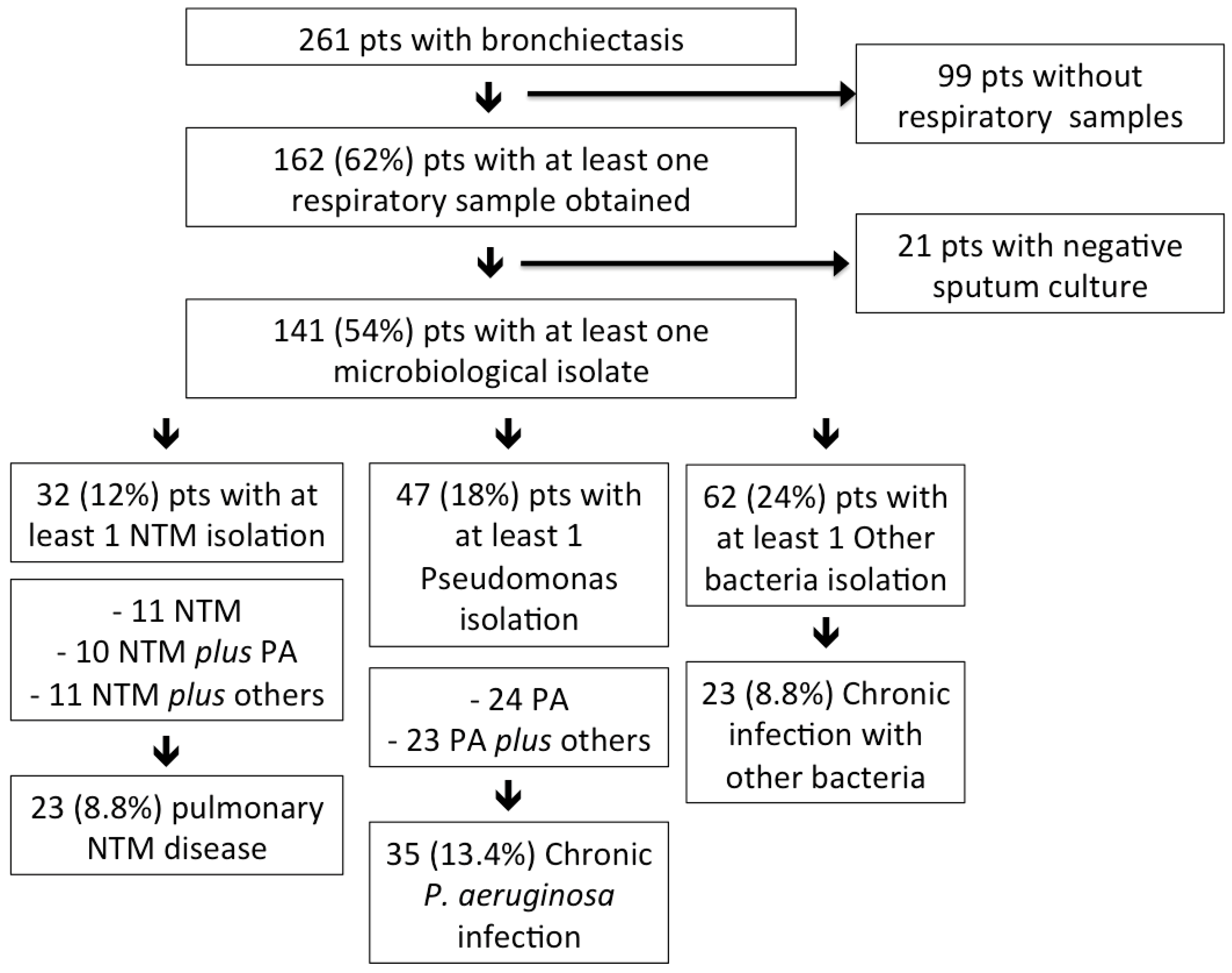
2.1. NTM Infection and pNTM Disease in Bronchiectasis
2.2. Characteristics of Bronchiectasis Patients with NTM
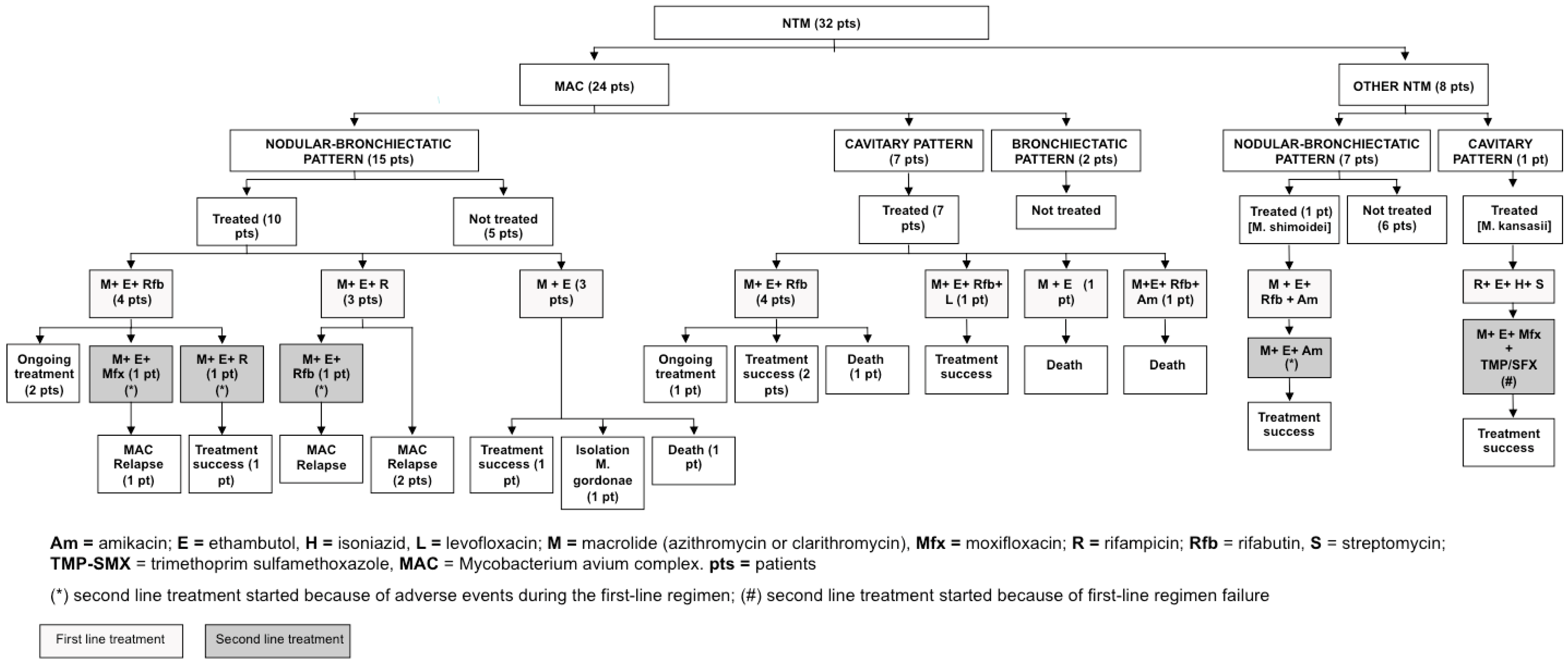
2.3. Treatment and Outcomes of Bronchiectasis Patients with pNTM
3. Discussion
4. Materials and Methods
4.1. Study Design
4.2. Data Collection and Microbiological Analysis
4.3. Study Definitions and Outcomes
4.4. Study Groups
4.5. Statistical Analysis
Author Contributions
Conflicts of Interest
Abbreviations
| NTM | non-tuberculous mycobacteria |
| pNTM | pulmonary NTM |
| ATS | American Thoracic Society |
| IDSA | Infectious Diseases Society of America |
| MAC | Mycobacterium avium complex |
| HRCT | high-resolution computed tomography |
| BAS | bronchial aspirate |
| BAL | bronchoalveolar lavage |
| BTS | British Thoracic Society |
| BSI | bronchiectasis severity index |
References
- Poppelwell, L.; Chalmers, J.D. Defining severity in non-cystic fibrosis bronchiectasis. Expert. Rev. Respir. Med. 2014, 8, 249–262. [Google Scholar] [CrossRef] [PubMed]
- Chalmers, J.D.; Aliberti, S.; Blasi, F. Management of bronchiectasis in adults. Eur. Respir. J. 2015, 45, 1446–1462. [Google Scholar] [CrossRef] [PubMed]
- Aliberti, S.; Lonni, S.; Dore, S.; McDonnell, M.J.; Goeminne, P.C.; Dimakou, K.; Fardon, T.C.; Rutherford, R.; Pesci, A.; Restrepo, M.I.; et al. Clinical phenotypes in adult patients with bronchiectasis. Eur. Respir. J. 2016, 47, 1113–1122. [Google Scholar] [CrossRef] [PubMed]
- McDonnell, M.J.; Jary, H.R.; Perry, A.; MacFarlane, J.G.; Hester, K.L.M.; Small, T.; Molyneux, C.; Perry, J.D.; Walton, K.E.; de Soyza, A. Non cystic fibrosis bronchiectasis: A longitudinal retrospective observational cohort study of Pseudomonas persistence and resistance. Respir. Med. 2015, 109, 716–726. [Google Scholar] [CrossRef] [PubMed]
- Finch, S.; McDonnell, M.J.; Abo-Leyah, H.; Aliberti, S.; Chalmers, J.D. A comprehensive analysis of the impact of Pseudomonas aeruginosa colonization on prognosis in adult bronchiectasis. Ann. Am. Thorac. Soc. 2015, 12, 1602–1611. [Google Scholar] [PubMed]
- Bonaiti, G.; Pesci, A.; Marruchella, A.; Lapadula, G.; Gori, A.; Aliberti, S. Nontuberculous mycobacteria in noncystic fibrosis bronchiectasis. BioMed Res. Int. 2015, 2015, 197950–197958. [Google Scholar] [CrossRef] [PubMed]
- Chu, H.; Zhao, L.; Xiao, H.; Zhang, Z.; Zhang, J.; Gui, T.; Gong, S.; Xu, L.; Sun, X. Prevalence of nontuberculous mycobacteria in patients with bronchiectasis: A meta-analysis. Arch. Med. Sci. 2014, 29, 661–668. [Google Scholar] [CrossRef] [PubMed]
- Griffith, D.E.; Aksamit, T.; Brown-Elliott, B.A.; Catanzaro, A.; Daley, C.; Gordin, F.; Holland, S.M.; Horsburgh, R.; Huitt, G.; Iademarco, M.F.; et al. An official ATS/IDSA statement: Diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. Am. J. Respir. Crit. Care Med. 2007, 175, 367–416. [Google Scholar] [CrossRef] [PubMed]
- Okumura, M.; Iwai, K.; Ogata, H.; Mizutani, S.; Yoshimori, K.; Itoh, K.; Nakajima, Y.; Kudoh, S. Pulmonary Mycobacterium avium complex (MAC) disease showing middle lobe syndrome—Pathological findings of 2 cases suggesting different mode of development. Kekkaku 2002, 77, 615–620. [Google Scholar] [PubMed]
- Aliberti, S.; Masefield, S.; Polverino, E.; de Soyza, A.; Loebinger, M.R.; Menendez, R.; Ringshausen, F.C.; Vendrell, M.; Powell, P.; Chalmers, J.D. Research priorities in bronchiectasis: A consensus statement from the EMBARC clinical research collaboration. Eur. Respir. J. 2016, 48, 632–647. [Google Scholar] [CrossRef] [PubMed]
- Máiz, L.; Girón, R.; Olveira, C.; Vendrell, M.; Nieto, R.; Martínez-García, M.A. Prevalence and factors associated with nontuberculous mycobacteria in non-cystic fibrosis bronchiectasis: A multicenter observational study. BMC Infect. Dis. 2016, 16, 437–444. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Cerón, E.; Prados, C.; Gómez-Carrera, L.; Cabanillas, J.J.; López-López, G.; Álvarez-Sala, R. Non-tuberculous mycobacterial infection in patients with non-cystic fibrosis bronchiectasias. Rev. Clín. Esp. 2012, 212, 127–130. [Google Scholar] [CrossRef] [PubMed]
- Fowler, S.J.; French, J.; Screaton, N.J.; Foweraker, J.; Condliffe, A.; Haworth, C.S.; Exley, A.R.; Bilton, D. Nontuberculous mycobacteria in bronchiectasis: Prevalence and patient characteristics. Eur. Respir. J. 2006, 28, 1204–1210. [Google Scholar] [CrossRef] [PubMed]
- Wickremasinghe, M.; Ozerovitch, L.J.; Davies, G.; Wodehouse, T.; Chadwick, M.V.; Abdallah, S.; Shah, P.; Wilson, R. Non-tuberculous mycobacteria in patients with bronchiectasis. Thorax 2005, 60, 1045–1051. [Google Scholar] [CrossRef] [PubMed]
- Mirsaeidi, M.; Hadid, W.; Ericsoussi, B.; Rodgers, D.; Sadikot, R.T. Non-tuberculous mycobacterial disease is common in patients with non-cystic fibrosis bronchiectasis. Int. J. Infect. Dis. 2013, 17, 1000–1004. [Google Scholar] [CrossRef] [PubMed]
- Zoumot, Z.; Boutou, A.K.; Gill, S.S.; van Zeller, M.; Hansell, D.M.; Wells, A.U.; Wilson, R.; Loebinger, M.R. Mycobacterium avium complex infection in non-cystic fibrosis bronchiectasis. Respirology 2014, 19, 714–722. [Google Scholar] [CrossRef] [PubMed]
- Pasteur, M.C.; Bilton, D.; Hill, A.T. British thoracic society bronchiectasis non-CF guideline group. British thoracic society guideline for non-CF bronchiectasis. Thorax 2010, 65, 1–58. [Google Scholar] [CrossRef] [PubMed]
- Koh, W.J.; Lee, K.S.; Kwon, O.J.; Jeong, Y.J.; Kwak, S.H.; Kim, T.S. Bilateral bronchiectasis and bronchiolitis at thin-section CT: Diagnostic implications in nontuberculous mycobacterial pulmonary infection. Radiology 2005, 235, 282–288. [Google Scholar] [CrossRef] [PubMed]
- Wallace, R.J.; Brown-Elliott, B.A.; McNulty, S.; Philley, J.V.; Killingley, J.; Wilson, R.W.; York, D.S.; Shepherd, S.; Griffith, D.E. Macrolide/Azalide therapy for nodular/bronchiectatic mycobacterium avium complex lung disease. Chest 2014, 146, 276–282. [Google Scholar] [CrossRef] [PubMed]
- Field, S.K.; Fisher, D.; Cowie, R.L. Mycobacterium avium complex pulmonary disease in patients without HIV infection. Chest 2004, 126, 566–581. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.B.; Jiang, R.H.; Li, L. Treatment outcomes for Mycobacterium avium complex: A systematic review and meta-analysis. Eur. J. Clin. Microbiol. Infect. Dis. 2014, 33, 347–358. [Google Scholar] [CrossRef] [PubMed]
- Park, I.K.; Olivier, K.N. Nontuberculous mycobacteria in cystic fibrosis and non-cystic fibrosis bronchiectasis. Semin. Respir. Crit. Care Med. 2015, 36, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Lonni, S.; Chalmers, J.D.; Goeminne, P.C.; McDonnell, M.J.; Dimakou, K.; de Soyza, A.; Polverino, E.; van de Kerkhove, C.; Rutherford, R.; Davison, J.; et al. Etiology of non-cystic fibrosis bronchiectasis in adults and its correlation to disease severity. Ann. Am. Thorac. Soc. 2015, 12, 1764–1770. [Google Scholar] [CrossRef] [PubMed]
- Chalmers, J.D.; Goeminne, P.; Aliberti, S.; McDonnell, M.J.; Lonni, S.; Davidson, J.; Poppelwell, L.; Salih, W.; Pesci, A.; Dupont, L.J.; et al. The bronchiectasis severity index. An international derivation and validation study. Am. J. Respir. Crit. Care Med. 2014, 189, 576–585. [Google Scholar] [CrossRef] [PubMed]
- McDonnell, M.J.; Aliberti, S.; Goeminne, P.C.; Dimakou, K.; Zucchetti, S.C.; Davidson, J.; Ward, C.; Laffey, J.G.; Finch, S.; Pesci, A.; et al. Multidimensional severity assessment in bronchiectasis: An analysis of seven European cohorts. Thorax 2016. [Google Scholar] [CrossRef] [PubMed]
- Pasteur, M.C.; Helliwell, S.M.; Houghton, S.J.; Webb, S.C.; Foweraker, J.E.; Coulden, R.A.; Flower, C.D.; Bilton, D.; Keogan, M.T. An investigation into causative factors in patients with bronchiectasis. Am. J. Respir. Crit. Care Med. 2000, 162, 1277–1284. [Google Scholar] [CrossRef] [PubMed]
| Non-Tuberculous Mycobacteria | ||
|---|---|---|
| Mycobacterium avium complex | 24 | |
| – | M. avium | 13 |
| – | M. intracellulare | 11 |
| M. gordonae | – | 4 |
| M. chelonae | – | 2 |
| M. kansasii | – | 1 |
| M. abscessus spp. | – | 1 |
| M. shimoidei | – | 1 |
| Variables | pNTM (n = 23) | Pseudomonas (n = 31) | Other Bacteria (n = 16) | p-Value |
|---|---|---|---|---|
| Demographics | – | – | – | – |
| Male, n (%) | 10 (44) | 15 (48) | 6 (38) | 0.79 |
| Age, median (IQR) | 70 (62–76) | 72 (66–76) | 60 (51–71) | 0.77 |
| BMI, median (IQR) | 20.6 (18.7–25.1) | 22.6 (21.6–25.2) | 21.4 (18.1–27.2) | 0.15 |
| BMI < 18.5, n (%) | 3 (19) | 4 (13) | 4 (27) | 0.68 |
| Prior tuberculosis, n (%) | 4 (22) | 3 (10) | 1 (6) | 0.39 |
| Either smoker or ex-smoker, n (%) | 11 (65) | 13 (42) | 6 (38) | 0.23 |
| Comorbidities, n (%) | – | – | – | – |
| Chronic obstructive pulmonary disease | 6 (35) | 13 (42) | 2 (13) | 0.76 |
| Asthma | 1 (6) | 3 (10) | 4 (25) | 1 |
| Sinusitis | 1 (6) | 5 (16) | 6 (38) | 0.65 |
| Cardiopathy | 4 (25) | 14 (45) | 7 (44) | 0.22 |
| Arterial hypertension | 6 (38) | 17 (55) | 6 (38) | 0.36 |
| Angina | 0 | 2 (7) | 1 (6) | 0.54 |
| Prior stroke | 0 | 1 (3) | 0 | 1 |
| Vasculopathy | 2 (13) | 4 (13) | 0 | 1 |
| Atrial fibrillation | 0 | 4 (13) | 0 | 0.28 |
| Valvulopathy | 1 (6) | 6 (19) | 2 (13) | 0.39 |
| Congestive heart failure | 0 | 2 (7) | 0 | 0.54 |
| Pulmonary hypertension | 0 | 2 (7) | 2 (13) | 0.54 |
| Diabetes | 3 (18) | 6 (19) | 1 (6) | 1 |
| Liver disease | 1 (6) | 1 (3) | 0 | 1 |
| Cirrhosis | 1 (6) | 1 (3) | 0 | 1 |
| Chronic renal failure | 0 | 2 (7) | 0 | 0.54 |
| Neurological disease | 0 | 2 (7) | 1 (6) | 0.54 |
| Rheumatological disease | 0 | 5 (16) | 1 (6) | 0.15 |
| Vasculitis | 0 | 1 (3) | 1 (6) | 1 |
| Gastroesophageal reflux disease | 5 (31) | 10 (32) | 8 (50) | 1 |
| Immuno-deficit | 0 | 1 (3) | 4 (25) | 1 |
| Solid cancer | 7 (39) | 6 (19) | 2 (13) | 0.18 |
| Haematological malignancy | 0 | 2 (7) | 1 (6) | 0.54 |
| Radiologic characteristics, n (%) | – | – | – | – |
| Cylindric | 13 (87) * | 16 (52) * | 12 (75) | 0.048 |
| Cystic | 2 (13) | 11 (38) | 3 (19) | 0.16 |
| Varicose | 0 | 2 (7) | 1 (6) | 0.54 |
| Tree-in-bud pattern | 13 (57) * | 7 (23) * | 7 (44) | 0.011 |
| Symptoms, n (%) | – | – | – | – |
| Daily cough | 9 (56) | 22 (71) | 8 (50) | 0.35 |
| Daily sputum | 5 (31) | 18 (58) | 10 (63) | 0.13 |
| Haemoptysis | 4 (25) | 4 (13) | 5 (31) | 0.42 |
| Dyspnoea | 9 (56) | 18 (58) | 9 (56) | 1 |
| Recurrent pneumonias | 1 (6) | 0 | 5 (31) | 0.34 |
| Weight loss | 6 (38) * | 3 (10) * | 3 (19) | 0.045 |
| Asthenia | 7 (44) | 16 (52) | 7 (44) | 0.76 |
| PFTs | – | – | – | – |
| FEV1 %, median (IQR) | 85 (56–100) | 61.5 (49–81) | 82 (52–100) | 0.074 |
| Laboratories collected during stable phase | – | – | – | |
| WBC × 103/mL, median (IQR) | 6.4 (5.4–7.7) | 7.4 (5.8–8.6) | 7.13 (5.48–9.61) | 0.19 |
| CRP mg/mL, median (IQR) | 0.57 (0.15–2.3) | 0.5 (0.26–1.38) | 0.97 (0.17–3.18) | 0.76 |
| Bronchiectasis Severity and Outcome | pNTM (n = 23) | Pseudomonas (n = 31) | Other Bacteria (n = 16) | p-Value |
|---|---|---|---|---|
| BSI, median (IQR) | 7 (6–10) * | 12 (9–15) * | 6 (4–9) | 0.004 |
| One-year exacerbations, median (IQR) | 0 (0–2) * | 1 (0.75–3.25) * | 0 (0–2) | 0.043 |
| One-year hospitalization (at least one/y), n (%) | 2 (9) | 5 (16) | 0 | 0.94 |
| One-year mortality, n (%) | 1 (4.3) | 1 (3.2) | 0 | 0.76 |
| Two-year mortality, n (%) | 2 (8.7) | 2 (6.5) | 0 | 1 |
| Three-year mortality, n (%) | 4 (17.4) | 4 (12.9) | 0 | 0.9 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Faverio, P.; Stainer, A.; Bonaiti, G.; Zucchetti, S.C.; Simonetta, E.; Lapadula, G.; Marruchella, A.; Gori, A.; Blasi, F.; Codecasa, L.; et al. Characterizing Non-Tuberculous Mycobacteria Infection in Bronchiectasis. Int. J. Mol. Sci. 2016, 17, 1913. https://doi.org/10.3390/ijms17111913
Faverio P, Stainer A, Bonaiti G, Zucchetti SC, Simonetta E, Lapadula G, Marruchella A, Gori A, Blasi F, Codecasa L, et al. Characterizing Non-Tuberculous Mycobacteria Infection in Bronchiectasis. International Journal of Molecular Sciences. 2016; 17(11):1913. https://doi.org/10.3390/ijms17111913
Chicago/Turabian StyleFaverio, Paola, Anna Stainer, Giulia Bonaiti, Stefano C. Zucchetti, Edoardo Simonetta, Giuseppe Lapadula, Almerico Marruchella, Andrea Gori, Francesco Blasi, Luigi Codecasa, and et al. 2016. "Characterizing Non-Tuberculous Mycobacteria Infection in Bronchiectasis" International Journal of Molecular Sciences 17, no. 11: 1913. https://doi.org/10.3390/ijms17111913
APA StyleFaverio, P., Stainer, A., Bonaiti, G., Zucchetti, S. C., Simonetta, E., Lapadula, G., Marruchella, A., Gori, A., Blasi, F., Codecasa, L., Pesci, A., Chalmers, J. D., Loebinger, M. R., & Aliberti, S. (2016). Characterizing Non-Tuberculous Mycobacteria Infection in Bronchiectasis. International Journal of Molecular Sciences, 17(11), 1913. https://doi.org/10.3390/ijms17111913








