Molecular Dynamic Studies of the Complex Polyethylenimine and Glucose Oxidase
Abstract
:1. Introduction
2. Results and Discussion
2.1. Docking Results
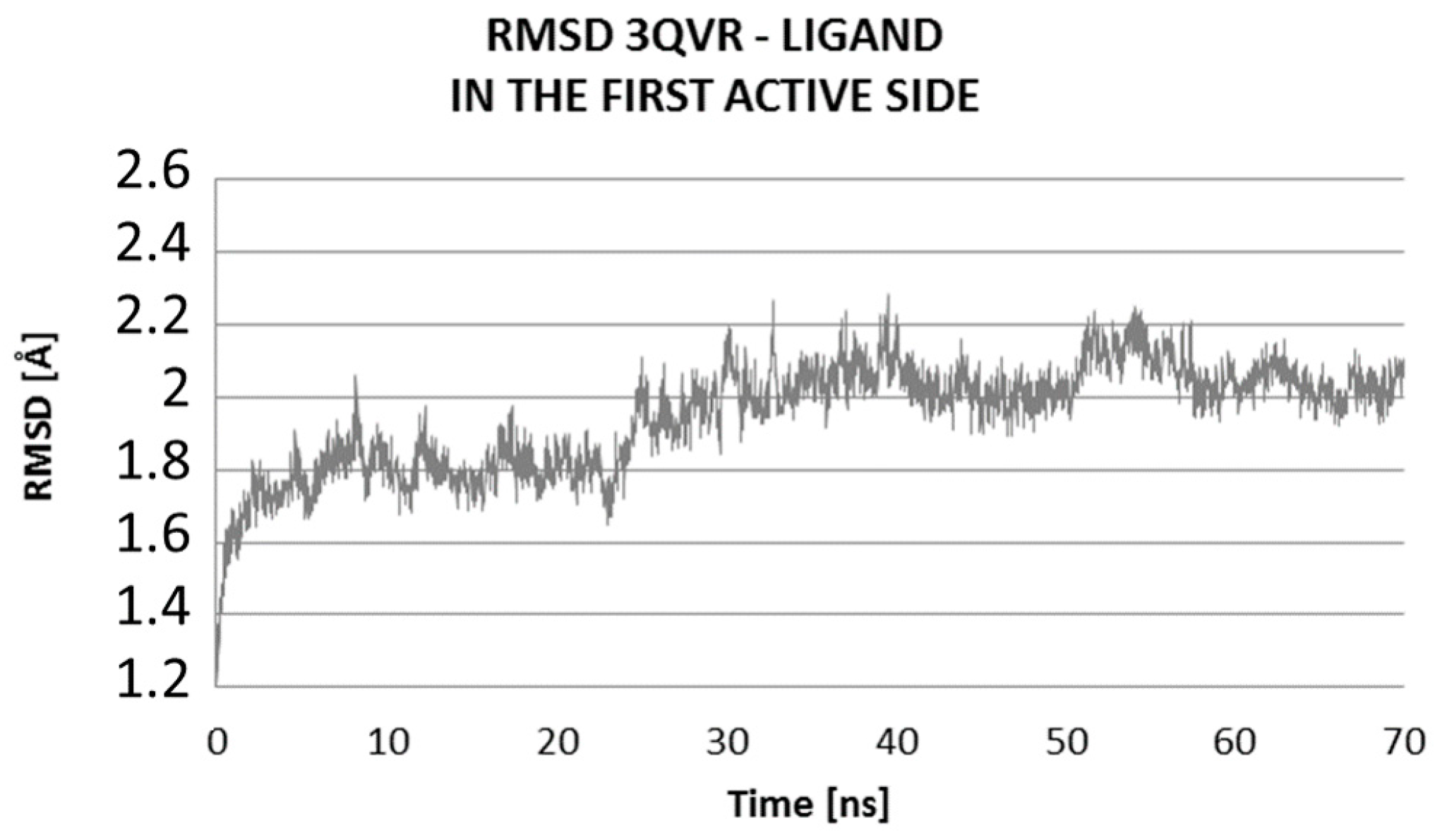
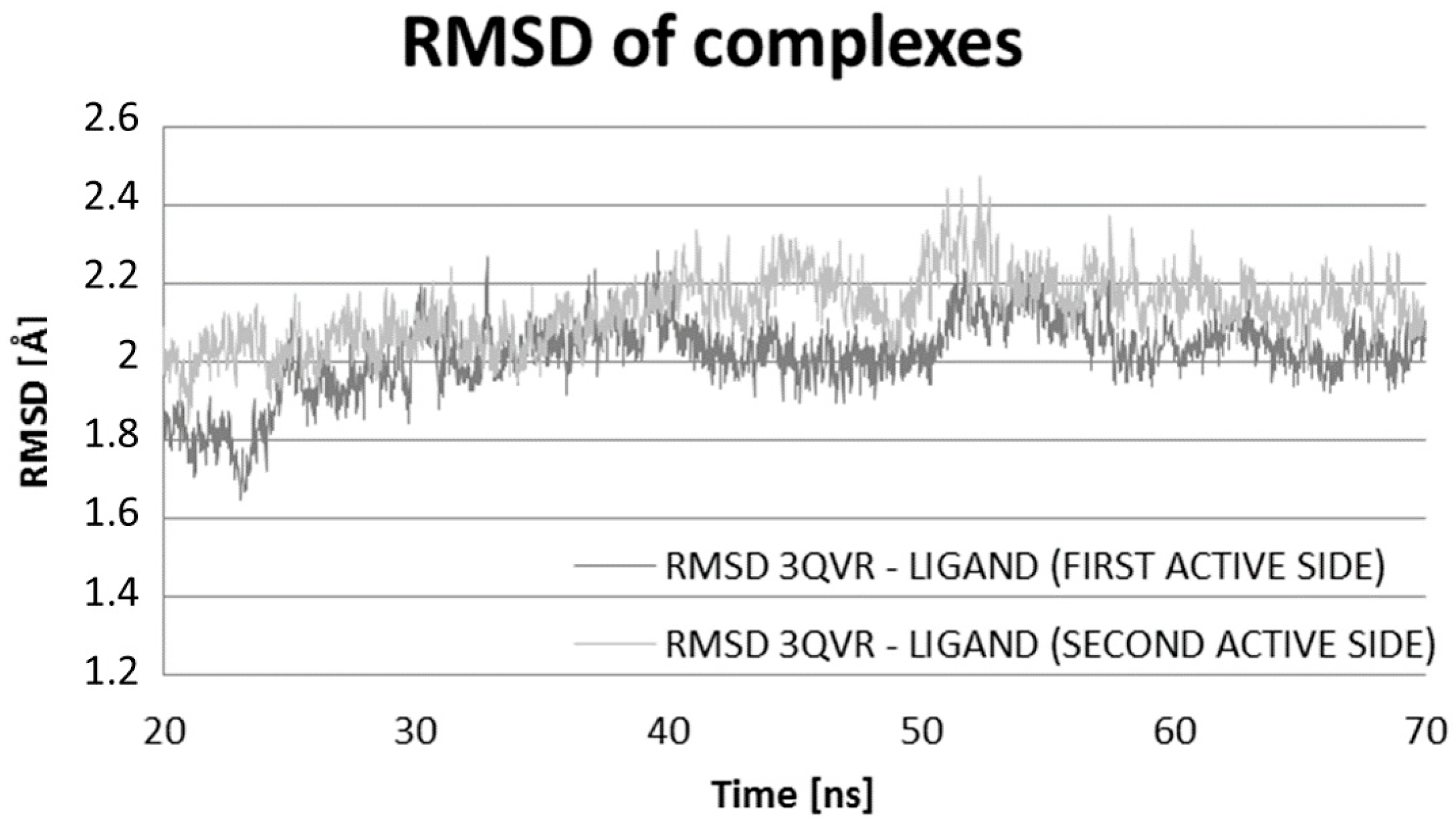
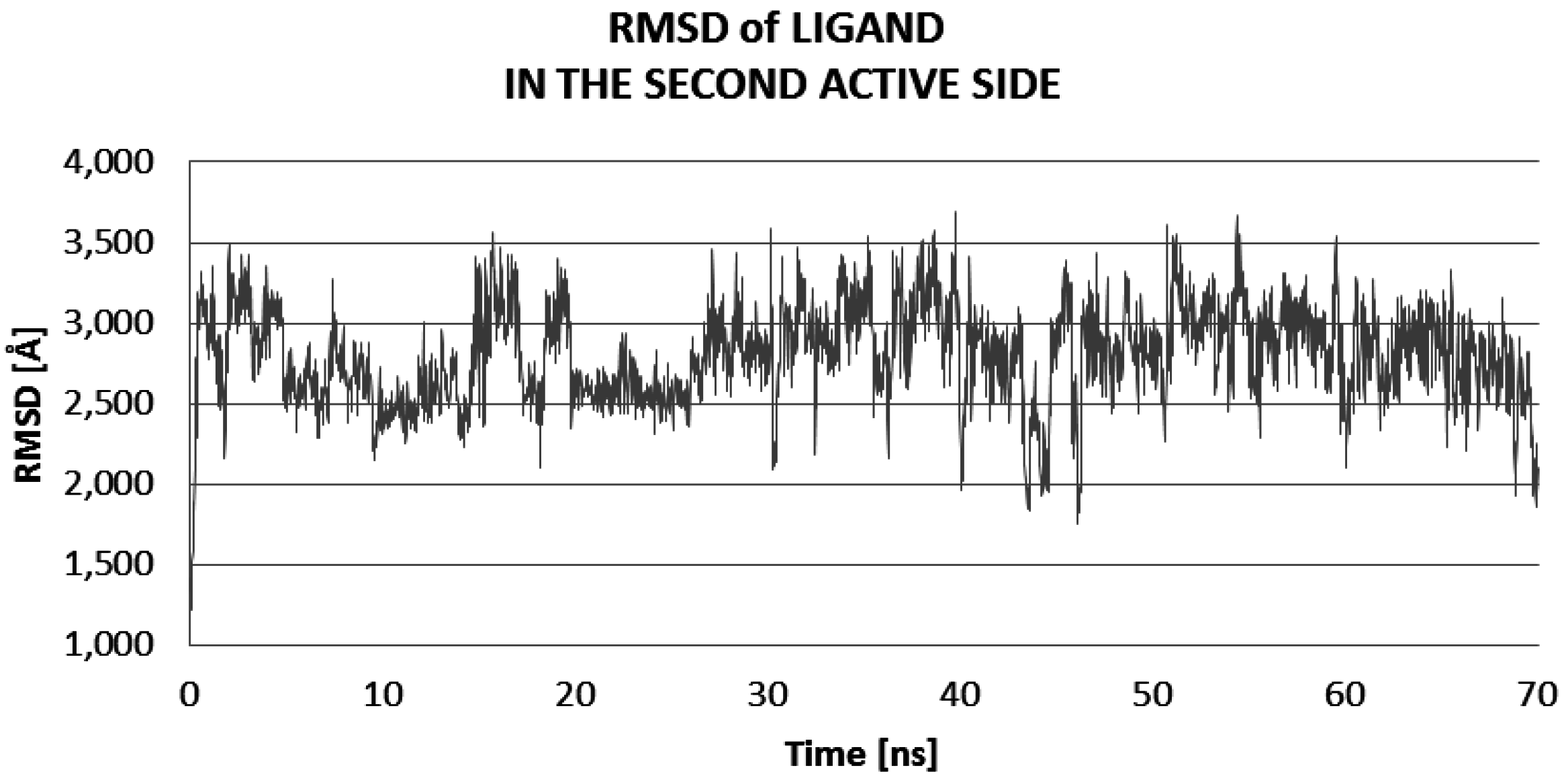
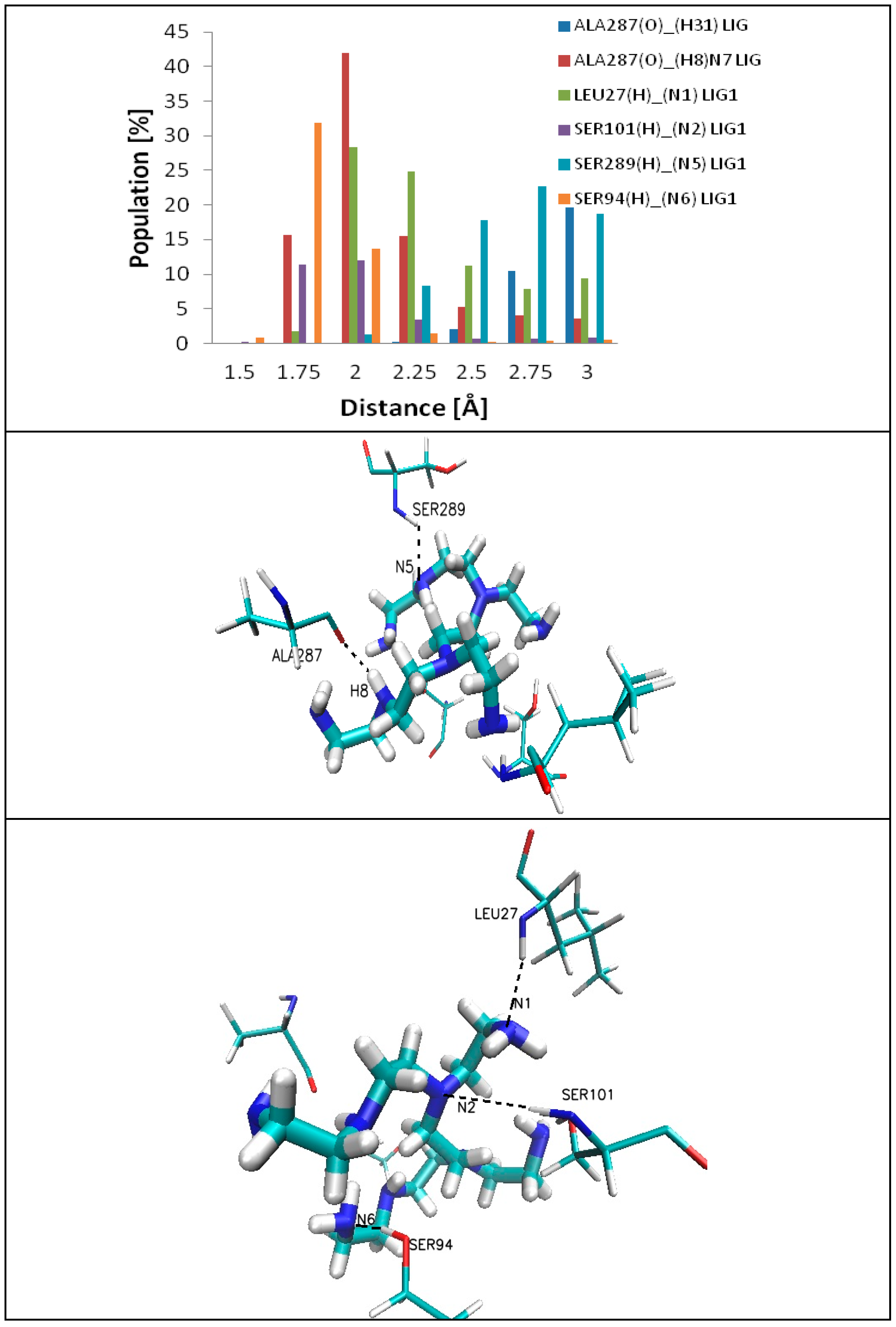
2.2. Molecular Dynamic Results
- ALA287(O)….(H31-PEI)LIG1, in 30% of all conformations, electrostatically interacts with the hydrogen atom H31 (of the ligand chain) to form a hydrogen bond of low strength;
3. Materials and Methods
4. Conclusions
Supplementary Materials
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sittiwet, C.; Srisa-ard, M.; Baimark, Y. Immobilization of Glucose oxidase and Peroxidase onto Nanoporous Chitosan-based Films. Malays. Polym. J. 2010, 5, 108–116. [Google Scholar]
- Homma, T.; Ichimura, T.; Kondo, M.; Kuwahara, T.; Shimomura, M. Covalent immobilization of glucose oxidase on the film prepared by electrochemical polymerization of N-phenylglycine for amperometric glucose sensing. Eur. Polym. J. 2014, 51, 130–135. [Google Scholar] [CrossRef]
- Shimomura, M.; Kojima, N.; Oshima, K.; Yamauchi, T.; Miyauchi, S. Covalent Immobilization of Glucose Oxidase on film prepared by electrochemical copolymerization of Thiophene-3-acetic acid and 3-Methylthiophene for Glucose sensing. Polym. J. 2001, 33, 629–631. [Google Scholar] [CrossRef]
- RCSB PDB. GOx—Molecule of the Month. Available online: http://www.rcsb.org/pdb/home/home.do (accessed on 23 December 2008).
- Pekel, N.; Salih, B.; Guven, O. Enhancement of stability of glucose oxidase by immobilization onto metal ion-chelated poly(N-vinyl imidazole) hydrogels. J. Biomater. Sci. Polym. Ed. 2005, 16, 253–266. [Google Scholar] [CrossRef] [PubMed]
- Karachevtsev, V.A.; Glamazda, A.Y.U.; Zarudnev, E.S.; Karachevtsev, M.V.; Leontiev, V.S.; Linnik, A.S.; Lytvyn, O.S.; Plokhotnichenko, A.M.; Stepanian, S.G. Glucose oxidase immobilization onto carbon nanotube networking. Ukr. J. Phys. 2012, 57, 700–709. [Google Scholar]
- Sharma, K.P.; Zhang, Y.; Thomas, M.R.; Brogan, A.P.S.; Perriman, A.W.; Mann, S. Self-organization of glucose oxidase-polymer surfactant nano-constructs in solvent-free soft solids and liquids. J. Phys. Chem. B 2014, 118, 11573–11580. [Google Scholar] [CrossRef] [PubMed]
- Poplawska, M.; Bystrzejewski, M.; Grudzinski, I.P.; Cywinska, M.A.; Ostapko, J.; Cieszanowski, A. Immobilization of gamma globulins and polyclonal antibodies of class IgG onto carbon-encapsulated iron nanoparticles functionalized with various surface linkers. Carbon 2014, 74, 180–194. [Google Scholar] [CrossRef]
- Karajanagi, S.S.; Vertegel, A.A.; Kane, R.S.; Dordick, J.S. Structure and function of enzymes adsorbed onto single-walled carbon Nanotubes. Langmuir 2004, 20, 11594–11599. [Google Scholar] [CrossRef] [PubMed]
- Feng, W.; Ji, P. Enzymes immobilized on carbon Nanotubes. Biotechnol. Adv. 2011, 29, 889–895. [Google Scholar] [CrossRef] [PubMed]
- Vicent, M.J.; Duncan, R. Polymer conjugates: Nanosized medicines for treating cancer. Trends Biotechnol. 2006, 24, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Kasprzak, A.; Popławska, M.; Bystrzejewski, M.; Łabędź, O.; Grudziński, I.P. Conjugation of polyethylenimine and its derivatives to carbon-encapsulated iron nanoparticles. RSC Adv. 2015, 5, 85556–85567. [Google Scholar] [CrossRef]
- Herlem, G.; Lakard, B.; Fahys, B. Recent research developments in electroanalytical chemistry. Trivandrum 2001, 3, 21–33. [Google Scholar]
- Herlem, G.; Lakard, B. Ab initio study of the electronic and structural properties of the crystalline polyethyleneimine polymer. J. Chem. Phys. 2004, 120, 9376–9382. [Google Scholar] [CrossRef] [PubMed]
- Padilla-Martíneza, S.G.; Martínez-Jothar, L.; Sampedro, J.G.; Tristan, F.; Pérez, E. Enhanced thermal stability and pH behavior of glucose oxidaseon electrostatic interaction with polyethylenimine. Int. J. Biol. Macromol. 2015, 75, 453–459. [Google Scholar] [CrossRef] [PubMed]
- Todde, G.; Hovmoller, S.; Laaksonen, A.; Mocci, F. Penicillium amagasakiense: Characterization of the transition state of its denaturation from molecular dynamics simulations. Proteins 2014, 82, 2353–2363. [Google Scholar] [CrossRef] [PubMed]
- Cleland, W.W.; Kreevoy, M.M. Low-barrier hydrogen bonds and enzymic catalysis. Science 1994, 264, 1887–1890. [Google Scholar] [CrossRef] [PubMed]
- Berman, H.M.; Henrick, K.; Nakamura, H. Announcing the worldwide Protein Data Bank. Nat. Struct. Mol. Biol. 2003, 10, 980. [Google Scholar] [CrossRef] [PubMed]
- PubChem database. Available online: https://pubchem.ncbi.nlm.nih.gov/search (accessed on 5 October 2014).
- Bayly, C.I.; Cieplak, P.; Cornell, W.D.; Kollman, P.A. A well-behaved electrostatic potential based method using charge restraints for deriving atomic charges: the RESP model. J. Phys. Chem. 1993, 97, 10269–10280. [Google Scholar] [CrossRef]
- Adelman, S.A.; Doll, J.D. Generalized Langevin equation approach for atom/solid surface scattering: General formulation for classical scattering off harmonic solids. J. Chem. Phys. 1976, 64, 2375–2388. [Google Scholar] [CrossRef]
- Ryckaert, J.P.; Ciccotti, G.; Berendsen, H.J.C. Numerical integration of the Cartesian equations of motion of a system with constraints: molecular dynamics of n-Alkanes. J. Comput. Phys. 1977, 23, 327–341. [Google Scholar] [CrossRef]
- Humphrey, W.; Dalke, A.; Schulten, K. VMD: Visual molecular dynamics. J. Mol. Graph. 1996, 14, 33–38. [Google Scholar] [CrossRef]
- Miller, B.R.; McGee, T.D.; Swails, J.M.; Homeyer, N.; Gohlke, H.; Roitberg, A.E. MMPBSA.py: An efficient program for End-State free energy calculations. J. Chem. Theory Comput. 2012, 8, 3314–3321. [Google Scholar] [CrossRef] [PubMed]
- Case, D.A.; Darden, T.A.; Cheatham, T.E., III; Simmerling, C.L.; Wang, J.; Duke, R.E.; Luo, R.; Walker, R.C.; Zhang, W.; Merz, K.M.; et al. AMBER 11. Available online: http://ambermd.org (accessed on 11 February 2016).
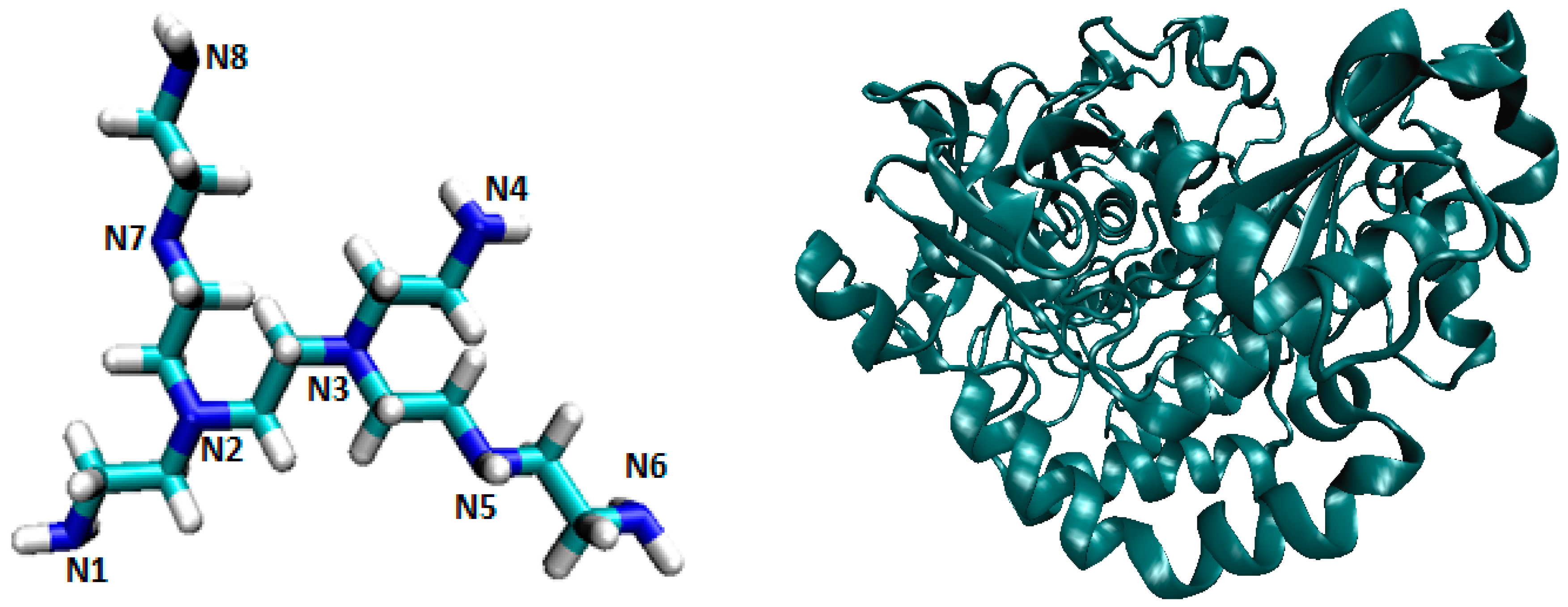



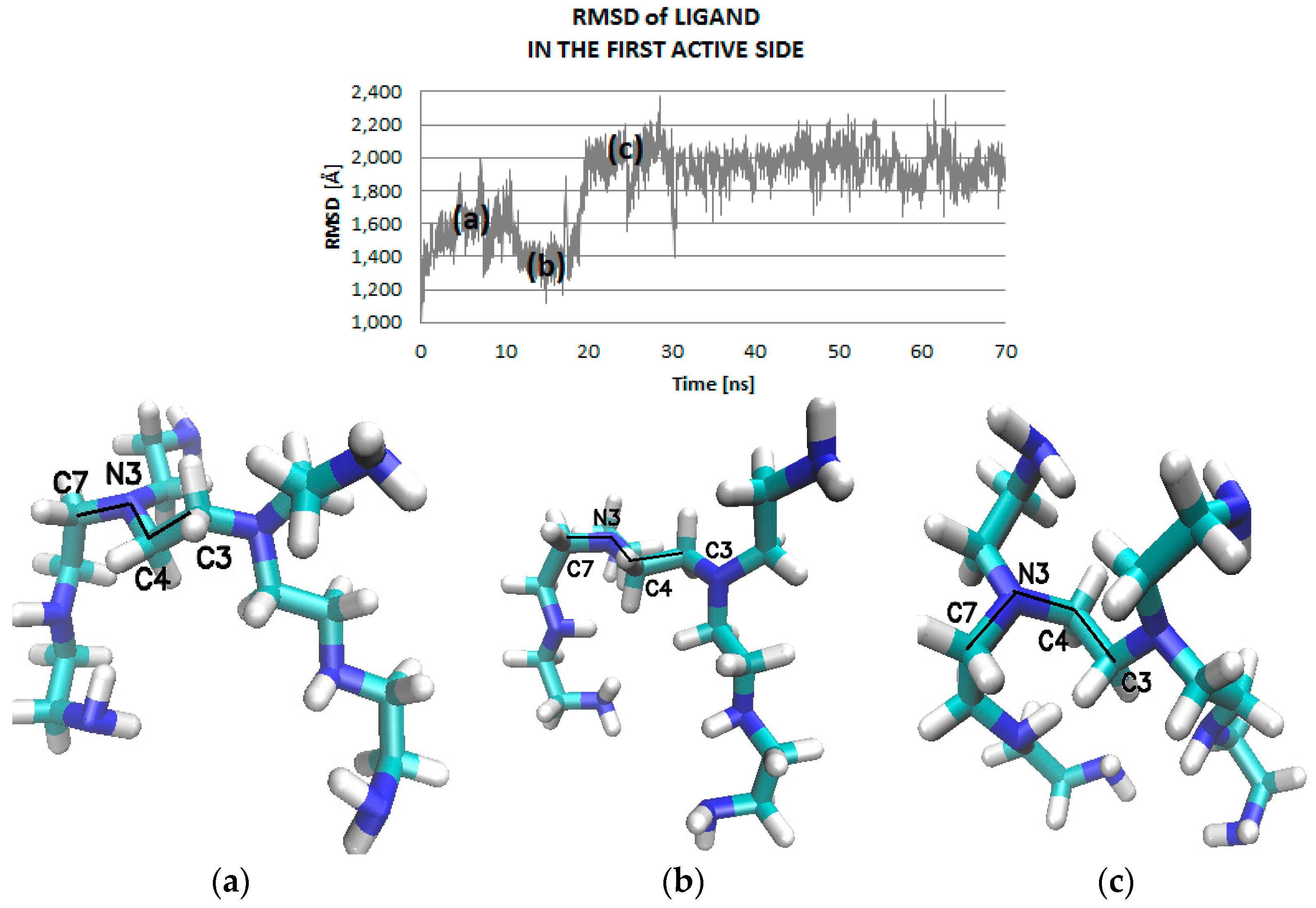

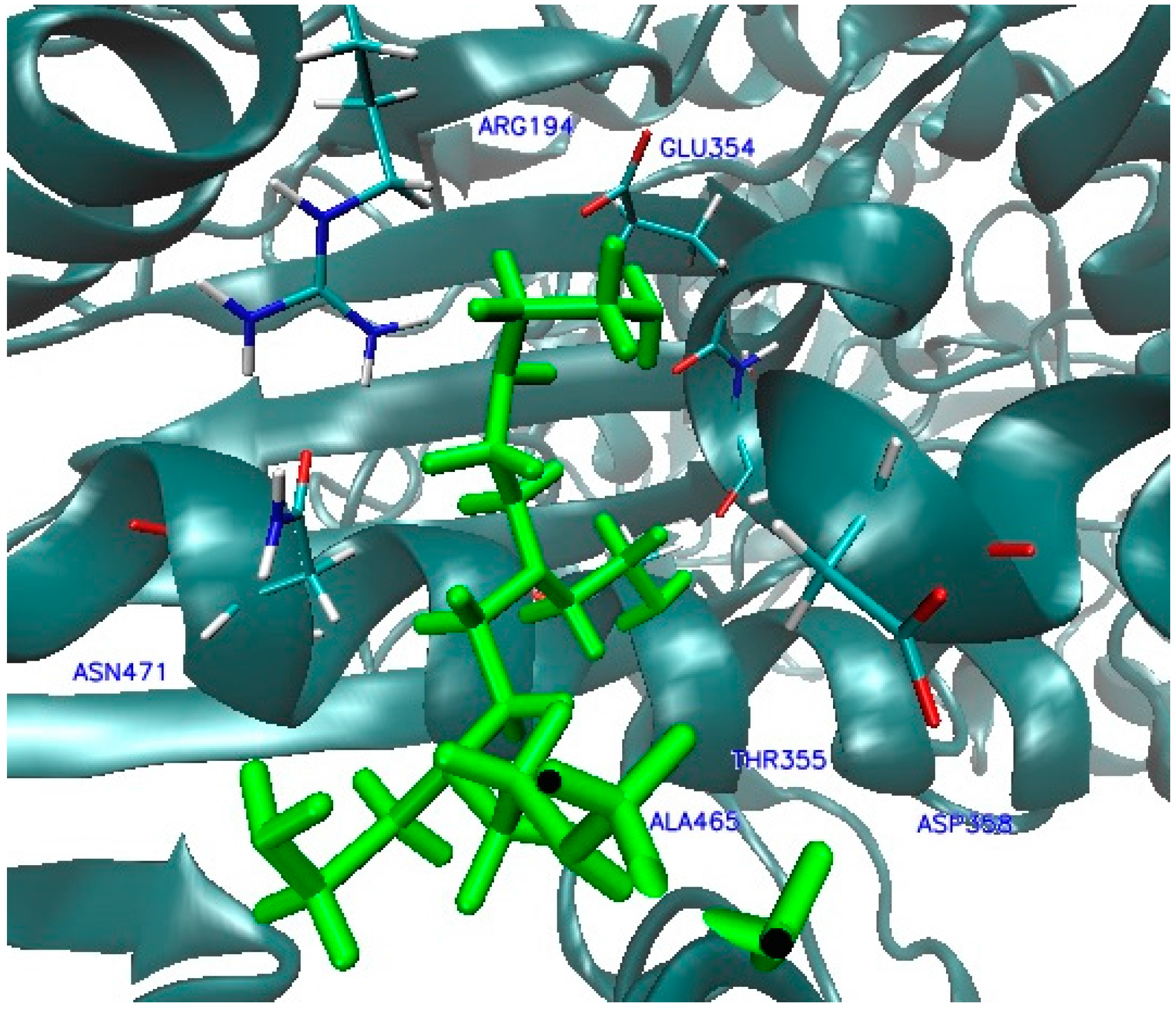

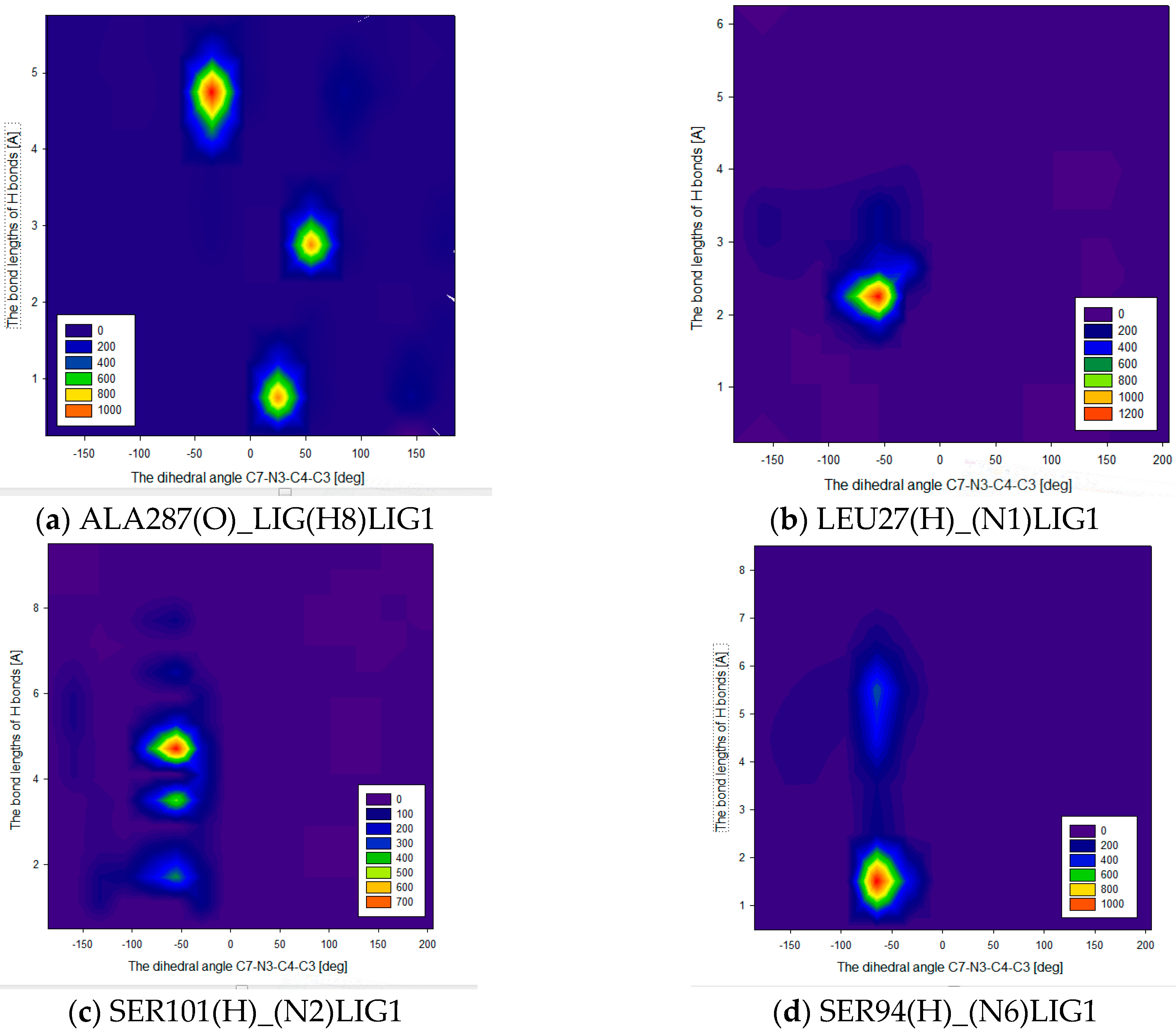
| Ligand Binding Site | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | Best Docking Energy (kcal/mol) |
|---|---|---|---|---|---|---|---|---|---|---|
| LIG1 | −5.8 | −5.8 | −5.6 | −5.6 | −5.6 | −5.5 | −5.5 | −5.5 | −5.5 | −5.8 |
| LIG2 | −4.5 | −4.3 | −4.2 | −4.2 | −4.2 | −4.2 | −4.2 | −4.1 | −4.1 | −4.5 |
| LIG3 | −3.8 | −3.8 | −3.7 | −3.6 | −3.6 | −3.6 | −3.6 | −3.6 | −3.5 | −3.8 |
| LIG4 | −3.9 | −3.4 | −2.7 | −1.9 | −1.5 | −1.2 | × | × | × | −3.9 |
| Ligand Binding Site | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | Docked Energy (kcal/mol) |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| 2 | −0.6 | −0.4 | −0.2 | 0.0 | 0.0 | 0.1 | 0.2 | 0.3 | 0.4 | −0.6 |
| 3 | 0.8 | 1.6 | 2.4 | 2.7 | 3.0 | × | × | × | × | 0.8 |
| Ligand Close to LIG1 of 3QVR | Ligand Close to LIG2 of 3QVR | Complex Ligand–Enzyme at LIG1 Site | Complex Ligand–Enzyme at LIG2 Site | |
|---|---|---|---|---|
| RMSD (Å) | 1.84 | 2.78 | 1.95 | 2.05 |
| SD | 0.24 | 0.30 | 0.14 | 0.16 |
| Hydrogen Bonds at LIG1 | Hydrogen Bond Length (Å) | Population (Among All Conformations) (%) | Hydrogen Bonds at LIG2 | Hydrogen Bond Length (Å) | Population (Among All Conformations) (%) |
|---|---|---|---|---|---|
| ALA287(O)_(H31)LIG1 | 2.25 | 0.03 | ASN471(H21)_(N2)LIG2 | 2.75 | 2.7 |
| 2.5 | 2.11 | ASP358(O1)_(H7)N6LIG2 | 1.75 | 0.17 | |
| 2.75 | 10.48 | 2 | 0.42 | ||
| 3 | 19.62 | GLU354(O1)_(H9)N8LIG2 | 2 | 0.4 | |
| ALA287(O)_(H8)N7LIG1 | 1.75 | 15.7 | 2.25 | 0.6 | |
| 2 | 41.9 | 2.5 | 0.4 | ||
| 2.25 | 15.5 | 2.75 | 0.9 | ||
| 2.5 | 5.2 | 3 | 1.3 | ||
| 2.75 | 4.0 | GLU354(O1)_(H10)N8LIG2 | 1.75 | 0.1 | |
| 3 | 3.6 | 2 | 0.9 | ||
| LEU27(H)_(N1)LIG1 | 1.7 | 1.8 | 2.25 | 2.5 | |
| 2 | 28.3 | 2.5 | 2.4 | ||
| 2.25 | 24.8 | 2.75 | 2.6 | ||
| 2.5 | 11.2 | 3 | 5.8 | ||
| 2.75 | 7.8 | GLU354(O2)_(H9)N8LIG2 | 1.75 | 0.1 | |
| 3 | 9.5 | 2 | 1.3 | ||
| SER101(H)_(N4)LIG1 | 1.5 | 0.14 | 2.25 | 1.5 | |
| 1.7 | 11.31 | 2.5 | 1.5 | ||
| 2 | 11.94 | 2.75 | 1.4 | ||
| 2.25 | 3.48 | 3 | 1.7 | ||
| 2.5 | 0.63 | GLU354(O2)_(H10)N8LIG2 | 1.75 | 0.09 | |
| 2.75 | 0.60 | 2 | 0.45 | ||
| 3 | 0.86 | 2.25 | 0.63 | ||
| SER94(H)_(N6)LIG1 | 1.5 | 0.9 | 2.5 | 1.16 | |
| 1.7 | 31.9 | 2.75 | 1.16 | ||
| 2 | 13.7 | 3 | 1.7 | ||
| 2.25 | 1.4 | ||||
| 2.5 | 0.3 | ||||
| 2.75 | 0.4 | ||||
| 3 | 0.5 | ||||
| SER289(H)_(N5)LIG1 | 2 | 1.2 | |||
| 2.25 | 8.3 | ||||
| 2.5 | 17.7 | ||||
| 2.75 | 22.7 | ||||
| 3 | 18.8 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szefler, B.; Diudea, M.V.; Putz, M.V.; Grudzinski, I.P. Molecular Dynamic Studies of the Complex Polyethylenimine and Glucose Oxidase. Int. J. Mol. Sci. 2016, 17, 1796. https://doi.org/10.3390/ijms17111796
Szefler B, Diudea MV, Putz MV, Grudzinski IP. Molecular Dynamic Studies of the Complex Polyethylenimine and Glucose Oxidase. International Journal of Molecular Sciences. 2016; 17(11):1796. https://doi.org/10.3390/ijms17111796
Chicago/Turabian StyleSzefler, Beata, Mircea V. Diudea, Mihai V. Putz, and Ireneusz P. Grudzinski. 2016. "Molecular Dynamic Studies of the Complex Polyethylenimine and Glucose Oxidase" International Journal of Molecular Sciences 17, no. 11: 1796. https://doi.org/10.3390/ijms17111796
APA StyleSzefler, B., Diudea, M. V., Putz, M. V., & Grudzinski, I. P. (2016). Molecular Dynamic Studies of the Complex Polyethylenimine and Glucose Oxidase. International Journal of Molecular Sciences, 17(11), 1796. https://doi.org/10.3390/ijms17111796








