Screening the Toxicity of Selected Personal Care Products Using Embryo Bioassays: 4-MBC, Propylparaben and Triclocarban
Abstract
:1. Introduction
2. Results
2.1. 4-MBC
2.2. Propylparaben
2.3. Triclocarban
3. Discussion
3.1. 4-MBC: An Ultra Violet Filter
3.2. Propylparaben: A Preservative
3.3. Triclocarban: Bactericide and Antifungal Agent
4. Material and Methods
4.1. Chemicals
4.2. Species Selection
4.2.1. Zebrafish
4.2.2. Sea Urchin
4.3. Fertilization and Embryo Collection
4.3.1. Zebrafish
4.3.2. Sea Urchin
4.4. Experimental Design and Embryo Bioassays
4.4.1. Experimental Solutions
4.4.2. Zebrafish
4.4.3. Sea Urchin
4.5. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| 4-MBC | 4-methylbenzylidene Camphor |
| DMSO | Dimethylsulfoxide |
| EC50 | 50%-Effective Concentration |
| EDCs | Endocrine Disrupting Chemicals |
| EE2 | Ethynilestradiol |
| EPA | US Environmental Protection Agency |
| hpf | hours post fertilization |
| LOEC | Lowest Observed Effect Concentration |
| NOEC | No Observed Effect Concentration |
| OECD | Organization for Economic Cooperation and Development |
| PCPs | Personal Care Products |
| PP | Propylparaben |
| PPCPs | Pharmaceuticals and Personal Care Products |
| TCC | Triclocarban |
| TCS | Triclosan |
| VTG | Vitellogenin |
| WWTPs | Wastewater Treatment Plants |
References
- Kim, J.W.; Ishibashi, H.; Yamauchi, R.; Ichikawa, N.; Takao, Y.; Hirano, M.; Koga, M.; Arizono, K. Acute toxicity of pharmaceutical and personal care products on freshwater crustacean (Thamnocephalus platyurus) and (Oryzias latipes). J. Toxicol. Sci. 2009, 34, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Li, X.; Zhu, S. Occurrence and distribution of selected pharmaceuticals and personal care products in aquatic environments: A comparative study of regions in China with different urbanization levels. Environ. Sci. Pollut. R 2012, 19, 2381–2389. [Google Scholar] [CrossRef] [PubMed]
- Lapworth, D.J.; Bran, N.; Stuart, M.E.; Ward, R.S. Emerging organic compounds in groundwater: A review of sources, fate and occurrence. Environ. Pollut. 2012, 163, 287–303. [Google Scholar] [CrossRef] [PubMed]
- Alygizakis, N.A.; Gago-Ferrero, P.; Borova, V.L.; Pavlidou, A.; Hatzianestis, I.; Thomaidis, N.S. Occurrence and spatial distribution of 158 pharmaceuticals, drugs of abuse and related metabolites in offshore seawater. Sci. Total Environ. 2016, 541, 1097–1105. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.Q.; Zhou, Z.; Patibandla, S.; Shu, X. Pharmaceutical removal from wastewater by ferrate (VI) and preliminar effluent toxicity assessments by the zebrafish embryo model. Microchem. J. 2013, 110, 239–245. [Google Scholar] [CrossRef]
- Kolpin, D.; Furlong, E.; Meyer, M.; Thurman, E.; Zaugg, S.; Barber, L.; Buxton, H. Pharmaceuticals, hormones, and other organic wastewater contaminants in U.S. Streams, 1999–2000: A national reconnaissance. Environ. Sci. Technol. 2002, 36, 1202–1211. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, B.; Paxéus, N.; Giudice, R.L.; Pollio, A.; Garric, J. Ecotoxicological impact of pharmaceuticals found in treated wastewaters: Study of carbamazepine, clofibric acid, and diclofenac. Ecotox. Environ. Saf. 2003, 55, 359–370. [Google Scholar] [CrossRef]
- Tran, N.H.; Hu, J.; Ong, S.L. Simultaneous determination of PPCPs, EDCs, and artificial sweeteners in environmental water samples using a single-step SPE coupled with HPLC-MS/MS and isotope dilution. Talanta 2013, 113, 82–92. [Google Scholar] [CrossRef] [PubMed]
- Bound, J.P.; Voulvoulis, N. Pharmaceuticals in the aquatic environment—A comparison of risk assessment strategies. Chemosphere 2004, 56, 1143–1155. [Google Scholar] [CrossRef] [PubMed]
- Moldovan, Z.; Chira, R.; Alder, A.C. Environmental exposure of pharmaceuticals and musk fragrances in the Somes River before and after upgrading the municipal wastewater treatment plant Cluj-Napoca, Romania. Environ. Sci. Pollut. R 2009, 16, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Brausch, J.M.; Rand, G.M. A review of personal care products in the aquatic environment: Environmental concentrations and toxicity. Chemosphere 2011, 82, 1518–1532. [Google Scholar] [CrossRef] [PubMed]
- Bossus, M.; Guler, Y.; Short, S.; Morrison, E.; Ford, A. Behavioral and transcriptional changes in the amphipod Echinogammarus marinus exposed to two antidepressants, fluoxetine and sertraline. Aquat. Toxicol. 2014, 151, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Roepke, T.; Snyder, M.; Cherr, G. Estradiol and endocrine disrupting compounds adversely affect development of sea urchin embryos at environmentally relevant concentrations. Aquat. Toxicol. 2005, 71, 155–173. [Google Scholar] [CrossRef] [PubMed]
- Witorsch, R.J.; Thomas, J.A. Personal care products and endocrine disruption: A critical review of the literature. Crit. Rev. Toxicol. 2010, 40, 1–30. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Shi, X.; Du, Y.; Zhou, B. Effects of xenoestrogens on the expression of vitellogenin (vtg) and cytochrome P450 aromatase (cyp19a and b) genes in zebrafish (Danio rerio) larvae. J. Environ. Sci. Heal. A 2011, 46, 960–967. [Google Scholar] [CrossRef] [PubMed]
- Nohynek, G.; Borgert, C.; Dietrichc, D.; Rozmand, K. Endocrine disruption: Fact or urban legend? Toxicol. Lett. 2013, 223, 295–305. [Google Scholar] [CrossRef] [PubMed]
- Castro, L.F.C.; Santos, M.M. To bind or not to bind: The taxonomic scope of nuclear receptor mediated endocrine disruption in invertebrate phyla. Environ. Sci. Technol. 2014, 48, 5361–5363. [Google Scholar] [CrossRef] [PubMed]
- Bachelot, M.; Li, Z.; Mubarn, D.; Gall, P.; Casells, C.; Fenet, H.; Gomez, E. Organic UV filter concentrations in marine mussels from French coastal regions. Sci. Total Environ. 2012, 420, 273–279. [Google Scholar] [CrossRef] [PubMed]
- Daughton, C.; Ternes, T. Pharmaceuticals and personal care products in the environment: Agents of subtle change? Environ. Health Persp. 1999, 107, 907–938. [Google Scholar] [CrossRef]
- González-Mariño, I.; Quintana, J.B.; Rodríguez, I.; Cela, R. Evaluation of the occurrence and biodegradation of parabens and halogenated by-products in wastewater by accurate-mass liquid chromatography-quadrupole-time-of-flight-mass spectrometry (LC-QTOF-MS). Water Res. 2011, 45, 6770–6780. [Google Scholar] [CrossRef] [PubMed]
- Albero, B.; Pérez, R.A.; Sánchez-Brunete, C.; Tadeo, J.L. Occurrence and analysis of parabens in municipal sewage sludge from wastewater treatment plants in Madrid (Spain). J. Hazard. Mater. 2012, 239–240, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Chalew, T.E.; Halden, R.U. Environmental exposure of aquatic and terrestrial biota to Triclosan and Triclocarban. J. Am. Water Works Assoc. 2009, 45, 4–13. [Google Scholar] [CrossRef] [PubMed]
- Balmer, M.E.; Buser, H.R.; Müller, M.D.; Poiger, T. Occurrence of some organic UV filters in wastewater, in surface waters, and in fish from Swiss Lakes. Environ. Sci. Technol. 2005, 39, 953–962. [Google Scholar] [CrossRef] [PubMed]
- Zucchi, S.; Blüthgen, N.; Ieronimo, A.; Fent, K. The UV-absorber benzophenone-4 alters transcripts of genes involved in hormonal pathways in zebrafish (Danio rerio) eleuthero-embryos and adult males. Toxicol. Appl. Pharm. 2011, 250, 137–146. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, D.; Sieratowicz, A.; Zielke, H.; Oetken, M.; Hollert, H.; Oehlmann, J. Ecotoxicological effect characterization of widely used organic UV filters. Environ. Pollut. 2012, 163, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Paredes, E.; Perez, S.; Rodil, R.; Quintana, J.B.; Beiras, R. Ecotoxicological evaluation of four UV filters using marine organisms from different trophic levels Isochrysis galbana, Mytilus galloprovincialis, Paracentrotus lividus, and Siriella armata. Chemosphere 2014, 104, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Schreurs, R.; Lanser, P.; Seinen, W.; van der Burg, B. Estrogenic activity of UV filters determined by an in vitro reporter gene assay and an in vivo transgenic zebrafish assay. Arch. Toxicol. 2002, 76, 257–261. [Google Scholar] [CrossRef] [PubMed]
- Schlumpf, M.; Durrer, S.; Faass, O.; Ehnes, C.; Fuestsch, M.; Gaille, C.; Henseler, M.; Hofkamp, L.; Maerkel, K.; Reolon, S.; et al. Developmental toxicity of UV filters and environmental exposure: A review. Int. J. Androl. 2008, 31, 144–151. [Google Scholar] [CrossRef] [PubMed]
- Nakata, H.; Murata, S.; Shinohara, R.; Filatreau, J.; Isobe, T.; Takahashi, J.; Tanabe, S. Occurrence and concentrations of persistent personal care products, organic UV filters, in the marine environment. Interdiscip. Stud. Environ. Chem. 2009, 1, 239–246. [Google Scholar]
- Li, W.V.; Tsui, M.P.; Chen, X.; Hui, M.N.; Jin, L.; Lam, R.H.; Yu, R.M.; Murphy, M.B.; Cheng, J.; Lam, P.K.; et al. Effects of 4-methylbenzylidene camphor (4-MBC) on neuronal and muscular development in Zebrafish (Danio. rerio) embryos. Environ. Sci. Pollut. R 2016, 23, 8275–8285. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, H.; Tamura, I.; Hirata, Y.; Kato, J.; Kagota, K.; Katsuki, S.; Yamamoto, A.; Kagami, Y.; Tatarazako, N. Aquatic toxicity and ecological risk assessment of seven parabens: Individual and additive approach. Sci. Total Environ. 2011, 410, 102–111. [Google Scholar] [CrossRef] [PubMed]
- Saco-Álvarez, L.; Beiras, R.; Durán, I.; Lorenzo, J.I. Methodological basis for the optimization of marine sea-urchin embryo test (SET) for the ecological assessment of coastal water quality. Ecotox. Environ. Saf. 2010, 73, 491–499. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, S.; Torres, T.; Martins, R.; Santos, M.M. Toxicity screening of diclofenac, propranolol, sertraline and simvastatin using Danio rerio and Paracentrotus lividus embryo bioassays. Ecotox. Environ. Saf. 2015, 114, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Dobbins, L.L.; Usenko, S.; Brain, R.A.; Brooks, B.W. Probabilistic ecological hazard assessment of parabens using Daphnia magna and Pimephales promelas. Environ. Toxicol. Chem. 2009, 28, 2744–2753. [Google Scholar] [CrossRef] [PubMed]
- Mikula, P.; Kruzikova, K.; Dobsikova, R.; Harustiakova, D. Influence of propylparaben on vitellogenesis and sex ratio in juvenile zebrafish (Danio rerio). Acta Vet. Brno 2009, 78, 319–326. [Google Scholar] [CrossRef]
- Gonzalez-Doncel, M.; Garcia-Maurino, J.E.; Segundo, L.S.; Beltran, E.M.; Satre, S.; Torija, C.F. Embryonic exposure of medaka (Oryzias latipes) to propylparaben: Effects on early development and post-hatching growth. Environ. Pollut. 2014, 184, 360–369. [Google Scholar] [CrossRef] [PubMed]
- Nipa, M. Finished Product Specification on Nipasol M. Reference No. P–P1; Nipa Laboratories Ltd.: Mid Glamorgan, UK, 1992. [Google Scholar]
- Madsen, T.; Boyd, H.B.; Nylen, D.; Pedersen, A.R.; Petersen, G.I.; Simonsen, F. Environmental and health assessment of substances in household detergents and cosmetic detergent products. Dan. Environ. Protect. Agency Rep. 2011. Available online: http://www.xn--miljstyrelsen-enb.dk/udgiv/publications/2001/87-7944-596-9/pdf/87-7944-597-7.pdf (accessed on 31 July 2016). [Google Scholar]
- Bjerregaard, P.; Andersen, D.N.; Pedersen, K.L.; Pedersen, S.N.; Korsgaard, B. Estrogenic effect of propylparaben (propylhydroxybenzoate) in rainbow trout Oncorhynchus mykiss after exposure via food and water. Comp. Biochem. Phys. C 2003, 136, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Schebb, N.H.; Flores, I.; Kurobe, T.; Franze, B.; Ranganathan, A.; Hammock, B.D.; Teh, S. Bioconcentration, metabolism and excretion of triclocarban in larval Quart Medaka (Oryzias latipes). Aquat. Toxicol. 2011, 105, 448–454. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Ki, C.A.; Gee, N.A.; Ahmed, M.I.; Duleba, A.J.; Zhao, L.; Gee, S.J.; Hammock, B.D.; Lasley, B.L. Triclocarban enhances testosterone action: A new type of endocrine disruptor? Endocrinology 2008, 149, 1173–1179. [Google Scholar] [CrossRef] [PubMed]
- Giudice, B.D.; Young, T.M. The antimicrobial triclocarban stimulates embryo production in the freshwater mudsnail Potamopyrgus antipodarum. Environ. Toxicol. Chem. 2010, 29, 966–970. [Google Scholar] [CrossRef] [PubMed]
- Schultz, M.M.; Bartell, S.E. Effects of Triclosan and Triclocarban, two ubiquitous environmental contaminats, on anatomy, physiology and behavior of the fathead minnow (Pimephales promelas). Arch. Environ. Contam. Toxicol. 2012, 63, 114–124. [Google Scholar] [CrossRef] [PubMed]
- Chow, W.; Chan, W.K.; Chan, K.M. Toxicity assessment and vitellogenin expression in zebrafish (Danio rerio) embryos and larvae acutely exposed to bisphenol A, endosulfan, heptachlor, methoxychlor and tetrabromobisphenol A. J. Appl. Toxicol. 2012, 33, 670–678. [Google Scholar] [CrossRef] [PubMed]
- Soares, J.; Filipe, L.; Castro, C.; Reis-Henriques, M.A.; Monteiro, N.M.; Santos, M.M. Zebrafish (Danio rerio) life-cycle exposure to chronic low doses of ethinylestradiol modulates p53 gene transcription within the gonads, but not NER pathways. Ecotoxicolgy 2012, 21, 1513–1522. [Google Scholar] [CrossRef] [PubMed]
- Segner, H. Zebrafish (Danio rerio) as a model organism for investigating endocrine disruption. Comp. Biochem. Phys. C 2009, 149, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Soares, J.; Coimbra, A.; Reis-Henriques, M.; Monteiro, N.; Vieira, M.; Oliveira, J.; Guedes-Dias, P.; Fontaínhas-Fernandes, A.; Parra, S.; Carvalho, A.; et al. Disruption of zebrafish (Danio rerio) embryonic development after full life-cycle parental exposure to low levels of ethinylestradiol. Aquat. Toxicol. 2009, 95, 330–338. [Google Scholar] [CrossRef] [PubMed]
- Bellas, J.; Beiras, R.; Mariño-Balsa, J.; Fernández, N. Toxicity of organic compounds to marine invertebrate embryos and larvae: A comparison between the sea urchin embryogenesis bioassay and alternative test species. Ecotoxicolgy 2005, 14, 337–353. [Google Scholar] [CrossRef]
- Zaroogian, G.E.; Pesh, G.; Morrison, G. Formulation of an artificial seawater medium suitable for oyster larvae development. Am. Zool. 1969, 9, 1144. [Google Scholar]
- Chapman, G.; Denton, D.; Lazorchak, J. Short-Term Methods for Estimating the Chronic Toxicity of Effluents and Receiving Waters to West Coast Marine and Estuarine Organisms, 1st ed.U.S. EPA–United States Environmental Protection Agency: Washington, DC, USA, 1995. Available online: https://cfpub.epa.gov/si/si_public_record_report.cfm?dirEntryId=46584 (accessed on 13 October 2016).
- Kimmel, C.; Ballard, W.; Kimmel, S.; Ullmann, B.; Schilling, T. Stages of embryonic development of the zebrafish. Dev. Dyn. 1995, 203, 253–310. [Google Scholar] [CrossRef] [PubMed]
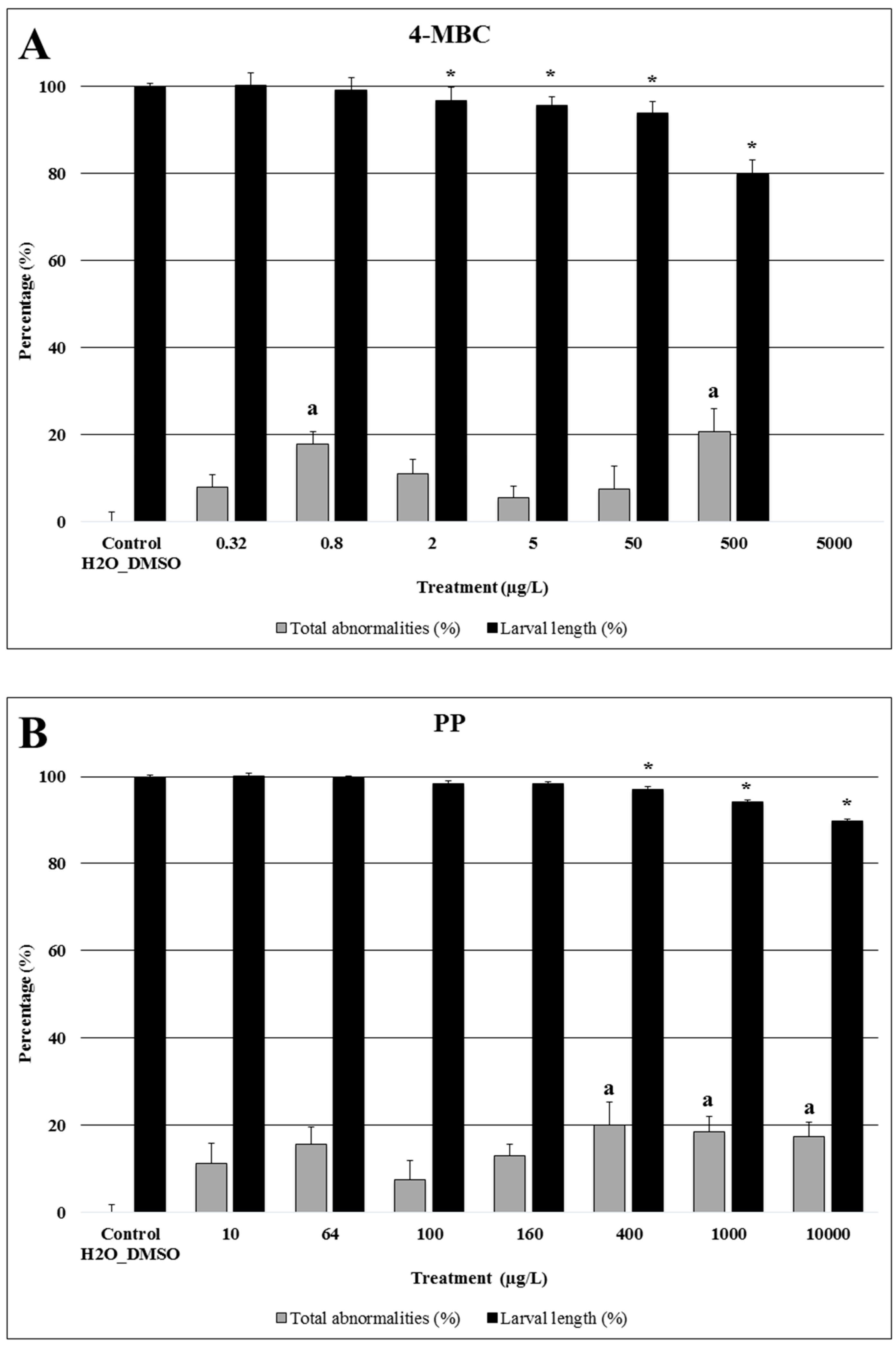
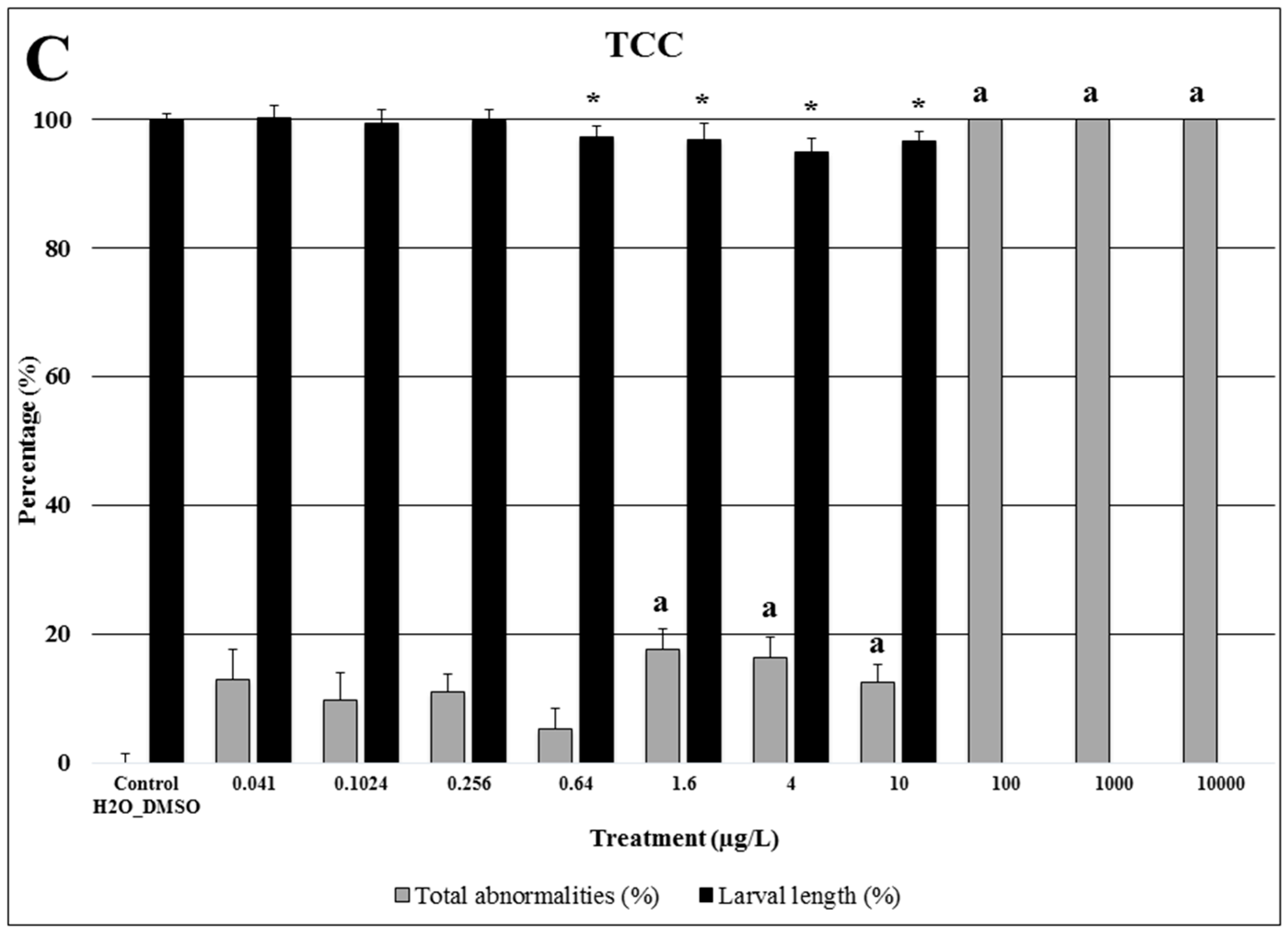

| Compound | Endpoints | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Treatment (µg/L) | Mortalitiy Rate | Hatching Rate | Abnormal Cellular Growth | 75%-Epiboly | Head Abnormalities | Eyes Abnormalities | Yolk-sac Abnormalities | Tail Abnormalities | Pericardial Edema | Heart Rate | Total Abnormalities | Abnormal Muscular Contractions | |
| 4-MBC | Controls | 1.88 ± 1.0 | 95.0 ± 1.58 | 1.25 ± 0.85 | 90.0 ± 6.26 | 1.04 ± 1.04 | 1.04 ± 1.04 | 1.04 ± 1.04 | 2.08 ± 1.42 | 1.04 ± 1.04 | 126.75 ± 4.46 | 2.08 ± 1.42 | 0.0 ± 0.0 |
| 5 | 7.5 ± 4.12 | 87.5 ± 3.66 | 7.5 ± 4.12 | 91.25 ± 3.98 | 4.17 ± 2.72 | 4.17 ± 2.72 | 4.17 ± 2.72 | 6.25 ± 3.05 | 2.08 ± 2.08 | 123.5 ± 3.58 | 6.25 ± 3.05 | 0.0 ± 0.0 | |
| 50 | 6.25 ± 2.63 | 92.5 ± 3.13 | 3.75 ± 2.63 | 95.0 ± 2.67 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 6.25 ± 4.38 | 0.0 ± 0.0 | 120.0 ± 4.21 | 6.25 ± 4.38 | 0.0 ± 0.0 | |
| 500 | 5.0 ± 2.67 | 81.25 ± 4.79 | 3.75 ± 1.82 | 92.5 ± 3.13 | 4.17 ± 2.72 | 4.17 ± 2.72 | 4.17 ± 2.72 | 10.42 ± 3.05 | 2.08 ± 2.08 | 121.0 ± 3.53 | 10.42 ± 3.05 | 35.42 ± 11.55 * | |
| 5000 | 2.5 ± 1.64 | 38.75 ± 7.66 * | 1.25 ± 1.25 | 92.5 ± 4.91 | 4.17 ± 2.72 | 2.08 ± 2.08 | 2.08 ± 2.08 | 8.33 ± 4.45 | 2.08 ± 2.08 | 95.0 ± 3.0 * | 10.42 ± 4.38 | 85.42 ± 3.78 * | |
| PP | Controls | 3.13 ± 0.95 | 92.19 ± 1.89 | 1.56 ± 0.65 | 97.81 ± 0.74 | 1.56 ± 0.87 | 1.56 ± 0.87 | 1.56 ± 0.87 | 3.13 ± 1.17 | 2.08 ± 0.99 | 143.0 ± 2.02 | 3.13 ± 1.17 | – |
| 10 | 2.5 ± 1.64 | 87.5 ± 4.11 | 1.25 ± 1.25 | 96.25 ± 1.83 | 2.08 ± 2.08 | 2.08 ± 2.08 | 2.08 ± 2.08 | 2.08 ± 2.08 | 2.08 ± 2.08 | 144 ± 5.01 | 2.08 ± 2.08 | – | |
| 100 | 1.25 ± 1.25 | 90.0 ± 3.78 | 1.25 ± 1.25 | 98.75 ± 1.25 | 0.0 ± 0.0 | 0.0 ± 0.0 | 2.08 ± 2.08 | 0.0 ± 0.0 | 2.08 ± 2.08 | 146.0 ± 3.38 | 2.08 ± 2.08 | – | |
| 1000 | 4.37 ± 1.57 | 85.68 ± 5.91 | 1.25 ± 0.85 | 94.49 ± 1.76 | 3.13 ± 1.68 | 2.08 ± 1.42 | 2.08 ± 1.42 | 2.08 ± 1.42 | 2.08 ± 1.42 | 139.75 ± 3.36 | 4.17 ± 1.86 | – | |
| 3500 | 3.75 ± 1.83 | 66.25 ± 8.22 | 1.25 ± 1.25 | 96.25 ± 1.82 | 95.83 ± 2.73 * | 89.58 ± 5.4 * | 58.33 ± 7.04 * | 91.67 ± 3.15 * | 91.67 ± 4.45 * | 124.5 ± 6.16 * | 100 ± 0.0 * | – | |
| 6000 | 1.25 ± 1.25 | 18.75 ± 3.98 * | 1.25 ± 1.25 | 91.25 ± 3.98 | 100 ± 0.0 * | 95.83 ± 4.17 * | 83.33 ± 8.33 * | 93.75 ± 6.25 * | 100 ± 0.0 * | 67.5 ± 7.98 * | 100 ± 0.0 * | – | |
| 8500 | 21.25 ± 7.89 | 2.5 ± 2.5 * | 1.25 ± 1.25 | 87.5 ± 4.53 * | 100 ± 0.0 * | 100 ± 0.0 * | 100 ± 0.0 * | 100 ± 0.0 * | 100 ± 0.0 * | 70.29 ± 6.47 * | 100 ± 0.0 * | – | |
| 10,000 | 100 ± 0.0 * | 0.0 ± 0.0 * | 0.0 ± 0.0 | 87.5 ± 3.13 * | – | – | – | – | – | – | – | – | |
| TCC | Controls | 2.19 ± 0.87 | 96.88 ± 0.95 | 1.56 ± 0.79 | 97.81 ± 0.87 | 0.52 ± 0.52 | 0.52 ± 0.52 | 0.0 ± 0.0 | 0.0 ± 0.0 | 1.58 ± 0.88 | 130.5 ± 3.53 | 0.52 ± 0.52 | – |
| 10 | 1.25 ± 1.25 | 98.75 ± 1.25 | 1.25 ± 1.25 | 98.75 ± 1.25 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 4.25 ± 2.78 | 118.5 ± 4.07 | 0.0 ± 0.0 | – | |
| 100 | 6.25 ± 1.8 | 91.11 ± 2.26 | 1.68 ± 1.01 | 93.61 ± 2.08 | 2.08 ± 1.42 | 2.08 ± 1.42 | 2.08 ± 1.42 | 3.12 ± 1.68 | 4.23 ± 1.89 | 128.0 ± 4.29 | 3.13 ± 1.68 | – | |
| 350 | 75.0 ± 12.81 * | 91.25 ± 4.79 | 0.0 ± 0.0 | 97.5 ± 1.64 | 5.56 ± 5.56 | 0.0 ± 0.0 | 0.0 ± 0.0 | 33.33 ± 33.33 | 0.0 ± 0.0 | 72.0 ± 48.0 | 33.33 ± 33.33 | – | |
| 600 | 95.0 ± 5.0 * | 92.5 ± 3.66 | 1.25 ± 1.25 | 97.5 ± 1.64 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 16.67 ± 16.67 | 0.0 ± 0.0 | – | 16.67 ± 16.67 | – | |
| 850 | 100 ± 0.0 * | 93.75 ± 2.63 | 1.25 ± 1.25 | 98.75 ± 1.25 | – | – | – | – | – | – | – | – | |
| 1000 | 100 ± 0.0 * | 95.0 ± 2.76 | 0.0 ± 0.0 | 100 ± 00 | – | – | – | – | – | – | – | – | |
| 10,000 | 100 ± 0.0 * | 83.75 ± 5.32 | 0.0 ± 0.0 | 98.75 ± 1.25 | – | – | – | – | – | – | – | – | |
| Compound | Zebrafish | Sea Urchin | Maximal Concentration | ||||
|---|---|---|---|---|---|---|---|
| – | NOEC | LOEC | NOEC | LOEC | WWTPs Influents | WWTPs Effluents | Surface Water |
| 4-MBC | 50 | 500 | 0.32 | 0.8 | 6.5 [23] | 2.7 [23] | * 0.799 [25] |
| PP | 1000 | 3500 | 160 | 400 | 2.8 [20] | 0.021[20] | 0.207 [31] |
| TCC | 100 | 350 | 0.256 | 0.64 | 50 [22] | >10 [11] | 6.75 [11] |
| Zebrafish | Sea Urchin | ||||
|---|---|---|---|---|---|
| 4-MBC | PP | TCC | 4-MBC | PP | TCC |
| 5000 | 10,000 | 10,000 | 5000 | 10,000 | 10,000 |
| 500 | 8500 | 1000 | 500 | 1000 | 1000 |
| 50 | 6000 | 850 | 50 | 400 | 100 |
| 5 | 3500 | 600 | 5 | 160 | 10 |
| – | 1000 | 350 | 2 | 100 | 4 |
| – | 100 | 100 | 0.8 | 64 | 1.6 |
| – | 10 | 10 | 0.32 | 10 | 0.64 |
| – | – | – | – | – | 0.256 |
| – | – | – | – | – | 0.1024 |
| Endpoint | 8 hpf | 32 hpf | 80 hpf |
|---|---|---|---|
| Mortality rate | ✓ | ✓ | ✓ |
| 75% of epiboly stage | ✓ | – | – |
| Abnormal cell growth | ✓ | – | – |
| Head abnormalities | – | ✓ | ✓ |
| Tail abnormalities | – | ✓ | ✓ |
| Eyes abnormalities | – | ✓ | ✓ |
| Yolk-sac abnormalities | – | ✓ | ✓ |
| Pericardial edema | – | ✓ | ✓ |
| Heart rate | – | ✓ | ✓ |
| Hatching rate | – | – | ✓ |
| (1) Muscular involuntary contractions | – | – | ✓ |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Torres, T.; Cunha, I.; Martins, R.; Santos, M.M. Screening the Toxicity of Selected Personal Care Products Using Embryo Bioassays: 4-MBC, Propylparaben and Triclocarban. Int. J. Mol. Sci. 2016, 17, 1762. https://doi.org/10.3390/ijms17101762
Torres T, Cunha I, Martins R, Santos MM. Screening the Toxicity of Selected Personal Care Products Using Embryo Bioassays: 4-MBC, Propylparaben and Triclocarban. International Journal of Molecular Sciences. 2016; 17(10):1762. https://doi.org/10.3390/ijms17101762
Chicago/Turabian StyleTorres, Tiago, Isabel Cunha, Rosário Martins, and Miguel M. Santos. 2016. "Screening the Toxicity of Selected Personal Care Products Using Embryo Bioassays: 4-MBC, Propylparaben and Triclocarban" International Journal of Molecular Sciences 17, no. 10: 1762. https://doi.org/10.3390/ijms17101762
APA StyleTorres, T., Cunha, I., Martins, R., & Santos, M. M. (2016). Screening the Toxicity of Selected Personal Care Products Using Embryo Bioassays: 4-MBC, Propylparaben and Triclocarban. International Journal of Molecular Sciences, 17(10), 1762. https://doi.org/10.3390/ijms17101762









