Elevated Expression of Calpain-4 Predicts Poor Prognosis in Patients with Gastric Cancer after Gastrectomy
Abstract
:1. Introduction
2. Results
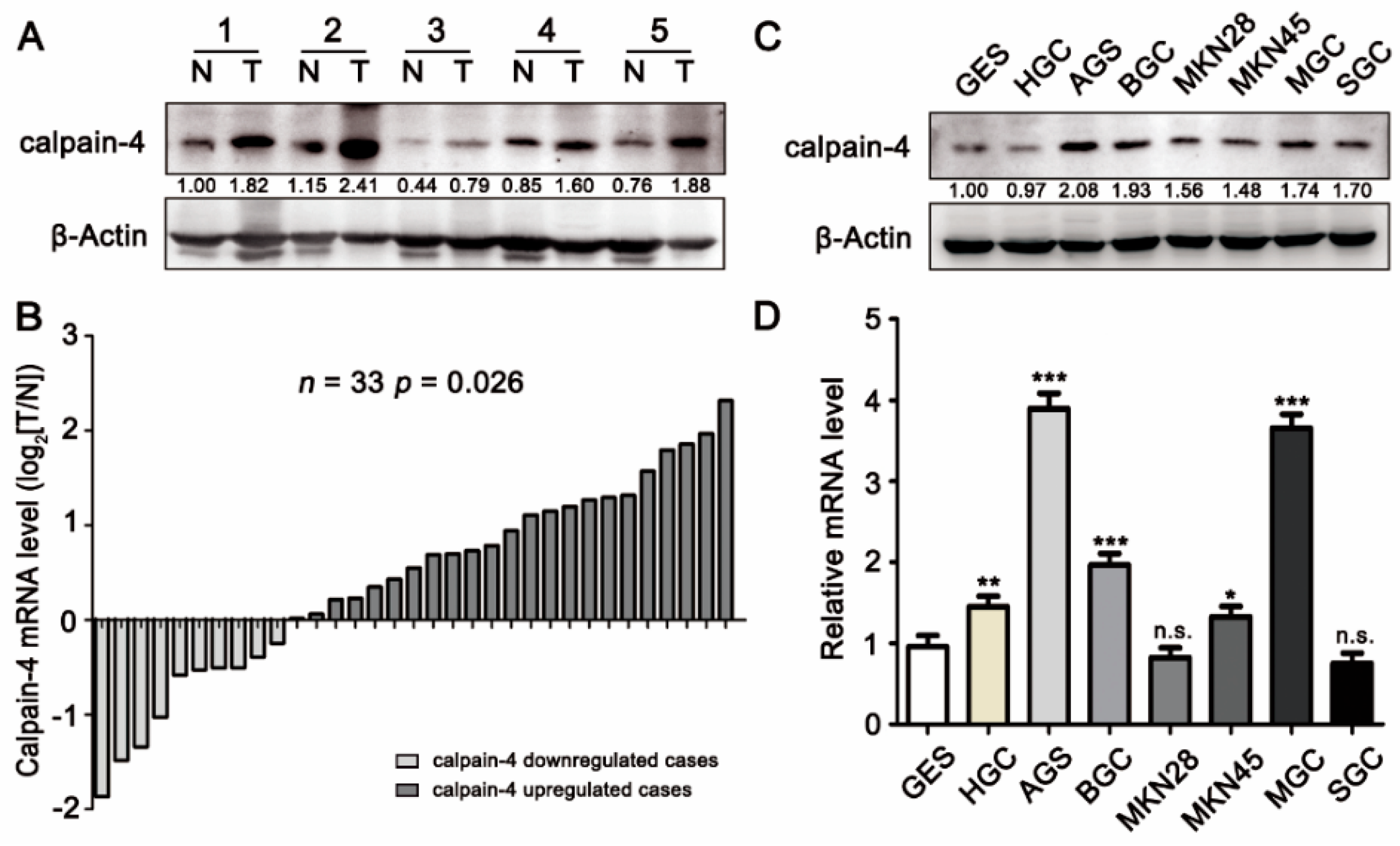
2.1. Expression of Calpain-4 Was High in Gastric Cancer
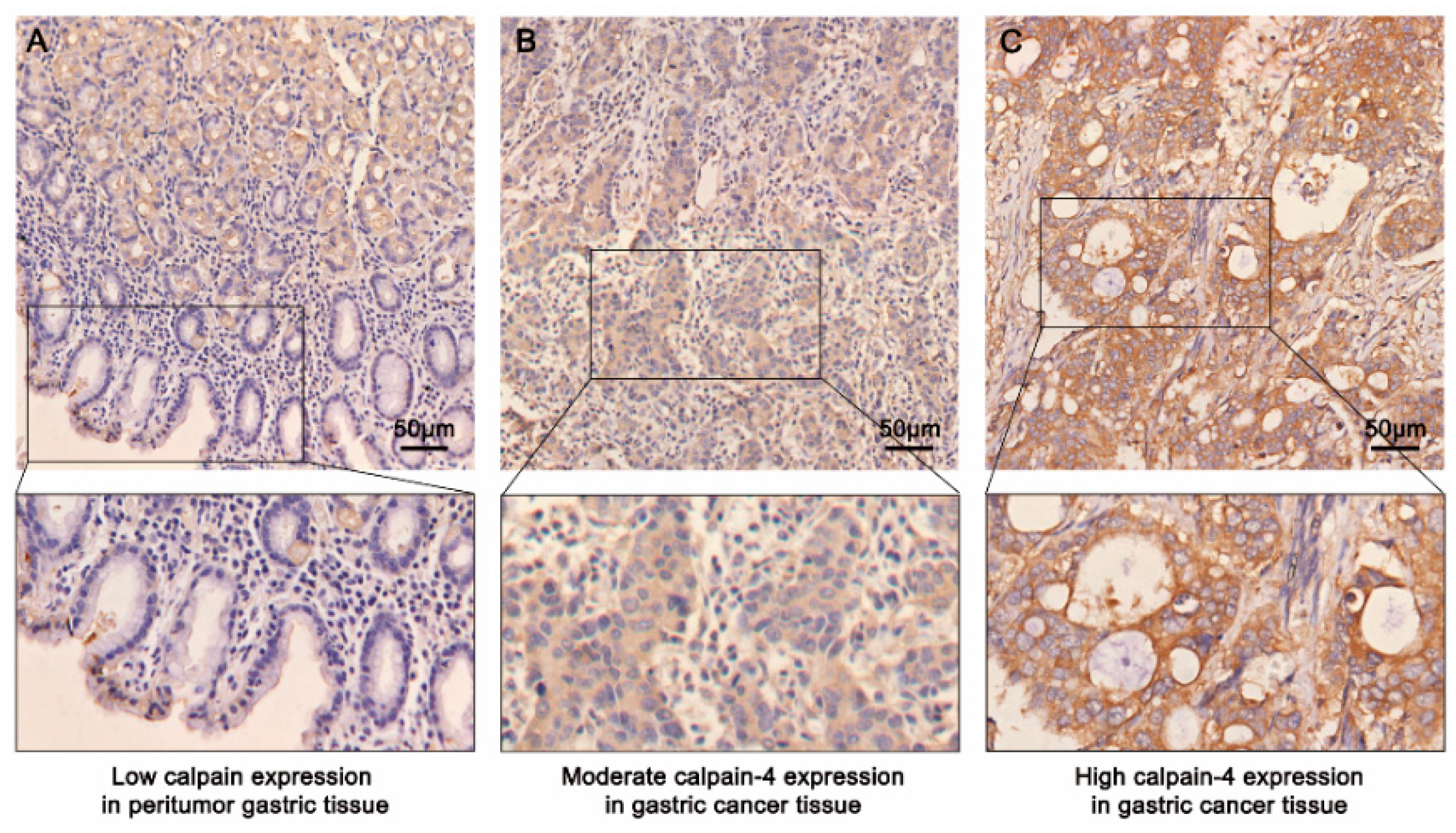
2.2. Immunohistochemical Findings and Association between Calpain-4 Expression and Clinicopathologic Characteristics in Patients with Gastric Cancer
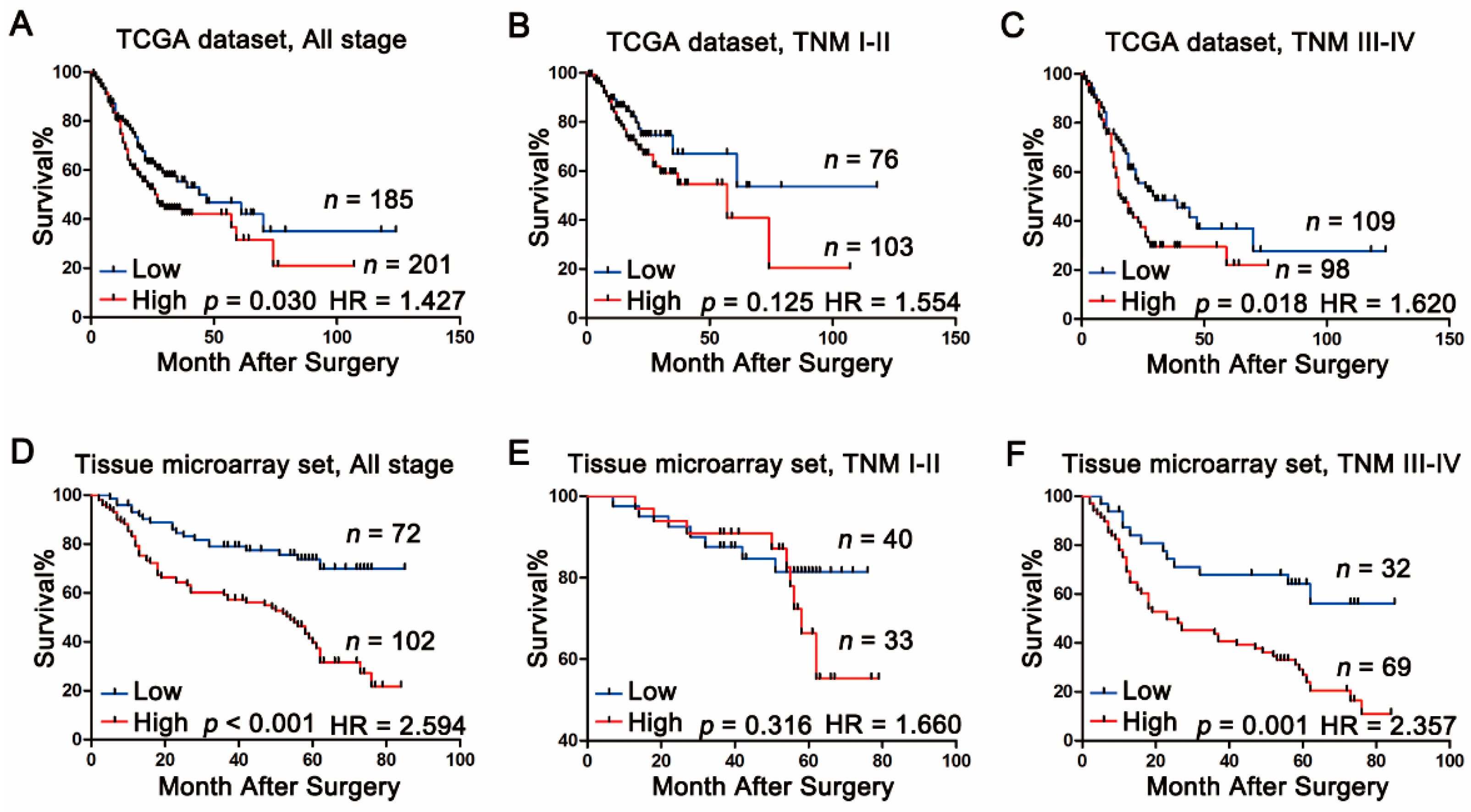
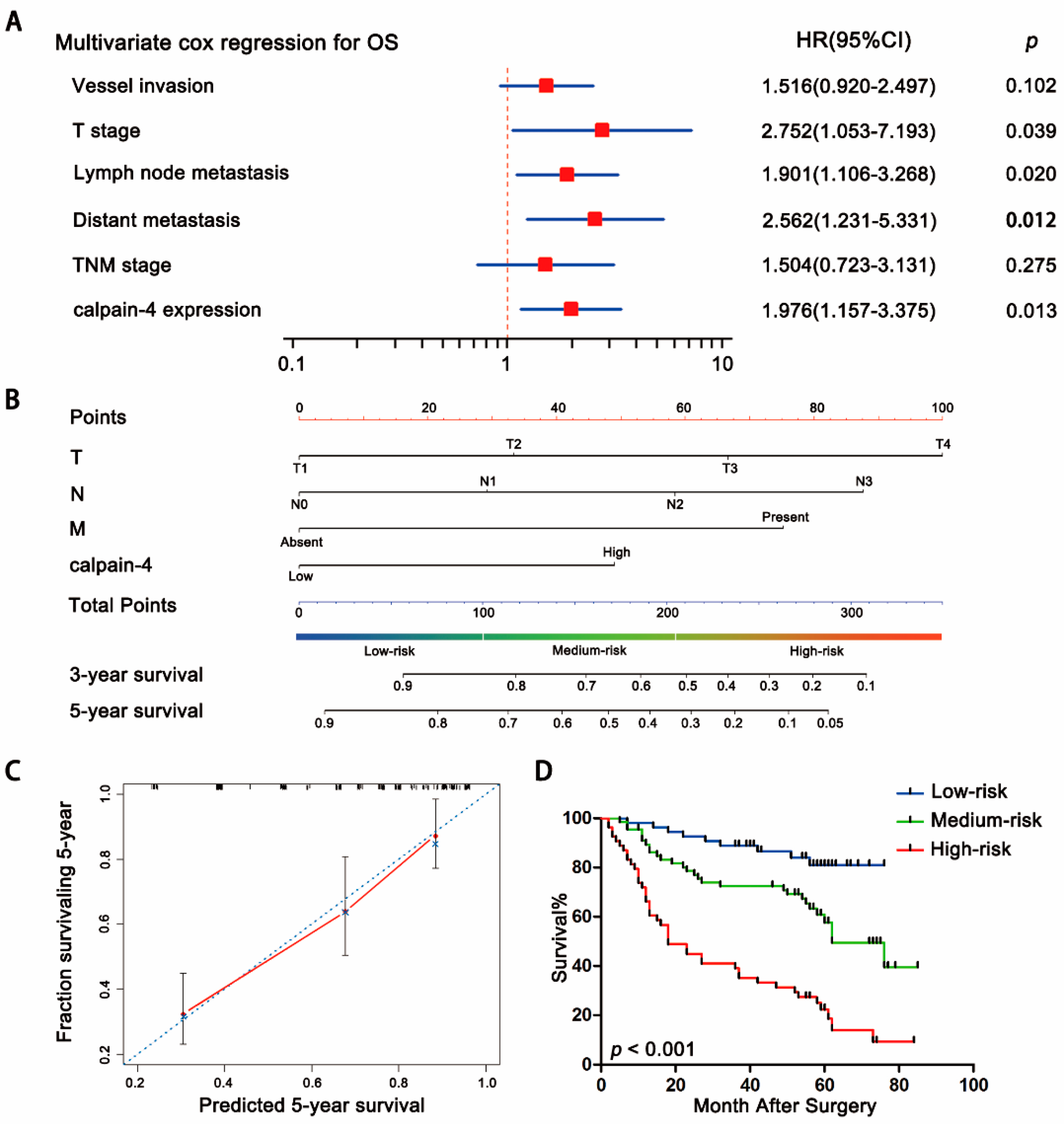
2.3. Association between Calpain-4 Expression and Prognosis of Patients with Gastric Cancer
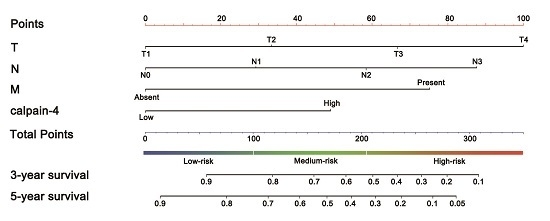
2.4. Predictive Nomogram for Overall Survival of Patients with Gastric Cancer
2.5. Knockdown of Calpain-4 Expression Reduced the Invasiveness of Gastric Cacner Cell in Vitro
3. Discussion
4. Materials and Methods
4.1. Primary Gastric Cancer Samples
4.2. Cell Lines
4.3. Western Blotting
4.4. Quantitative Real-Time PCR
4.5. Immunohistochemistry
4.6. TCGA Dataset
4.7. In Vitro Invasion Assays
4.8. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Torre, L.A.; Bray, F.; Siegel, R.L.; Ferlay, J.; Lortet-Tieulent, J.; Jemal, A. Global cancer statistics, 2012. CA Cancer J. Clin. 2015, 65, 87–108. [Google Scholar] [CrossRef] [PubMed]
- Mayer, R.J.; Venook, A.P.; Schilsky, R.L. Progress against GI cancer during the American Society of Clinical Oncology’s first 50 years. J. Clin. Oncol. 2014, 32, 1521–1530. [Google Scholar] [CrossRef] [PubMed]
- Edge, S.B.; Compton, C.C. The American Joint Committee on Cancer: The 7th edition of the AJCC cancer staging manual and the future of TNM. Ann. Surg. Oncol. 2010, 17, 1471–1474. [Google Scholar] [CrossRef] [PubMed]
- Lim, L.; Michael, M.; Mann, G.B.; Leong, T. Adjuvant therapy in gastric cancer. J. Clin. Oncol. 2005, 23, 6220–6232. [Google Scholar] [CrossRef] [PubMed]
- Stock, M.; Otto, F. Gene deregulation in gastric cancer. Gene 2005, 360, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Moretti, D.; Del Bello, B.; Allavena, G.; Maellaro, E. Calpains and cancer: Friends or enemies? Arch. Biochem. Biophys. 2014, 564, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Goll, D.E.; Thompson, V.F.; Li, H.; Wei, W.; Cong, J. The calpain system. Physiol. Rev. 2003, 83, 731–801. [Google Scholar] [CrossRef] [PubMed]
- Storr, S.J.; Carragher, N.O.; Frame, M.C.; Parr, T.; Martin, S.G. The calpain system and cancer. Nat. Rev. Cancer 2011, 11, 364–374. [Google Scholar] [CrossRef] [PubMed]
- Arthur, J.S.; Elce, J.S.; Hegadorn, C.; Williams, K.; Greer, P.A. Disruption of the murine calpain small subunit gene, Capn4: Calpain is essential for embryonic development but not for cell growth and division. Mol. Cell. Biol. 2000, 20, 4474–4481. [Google Scholar] [CrossRef] [PubMed]
- Shimada, M.; Greer, P.A.; McMahon, A.P.; Bouxsein, M.L.; Schipani, E. In vivo targeted deletion of calpain small subunit, Capn4, in cells of the osteoblast lineage impairs cell proliferation, differentiation, and bone formation. J. Biol. Chem. 2008, 283, 21002–21010. [Google Scholar] [CrossRef] [PubMed]
- Dourdin, N.; Bhatt, A.K.; Dutt, P.; Greer, P.A.; Arthur, J.S.; Elce, J.S.; Huttenlocher, A. Reduced cell migration and disruption of the actin cytoskeleton in calpain-deficient embryonic fibroblasts. J. Biol. Chem. 2001, 276, 48382–48388. [Google Scholar] [PubMed]
- Zhang, C.; Bai, D.S.; Huang, X.Y.; Shi, G.M.; Ke, A.W.; Yang, L.X.; Yang, X.R.; Zhou, J.; Fan, J. Prognostic significance of Capn4 overexpression in intrahepatic cholangiocarcinoma. PLoS ONE 2013, 8, e54619. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.J.; Qi, Z.X.; Hua, W.; Zhu, J.J.; Zhang, X.; Yao, Y.; Mao, Y. Increased expression of Capn4 is associated with the malignancy of human glioma. CNS Neurosci. Ther. 2014, 20, 521–527. [Google Scholar] [CrossRef] [PubMed]
- Dai, Z.; Zhou, S.L.; Zhou, Z.J.; Bai, D.S.; Xu, X.Y.; Fu, X.T.; Chen, Q.; Zhao, Y.M.; Zhu, K.; Yu, L.; et al. Capn4 contributes to tumour growth and metastasis of hepatocellular carcinoma by activation of the FAK-Src signalling pathways. J. Pathol. 2014, 234, 316–328. [Google Scholar] [CrossRef] [PubMed]
- Zheng, P.C.; Chen, X.; Zhu, H.W.; Zheng, W.; Mao, L.H.; Lin, C.; Liu, J.N.; Zheng, M. Capn4 is a marker of poor clinical outcomes and promotes nasopharyngeal carcinoma metastasis via nuclear factor-kappaB-induced matrix metalloproteinase 2 expression. Cancer Sci. 2014, 105, 630–638. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.; Xu, F.K.; Zhao, G.Y.; Lu, C.L.; Lin, Z.W.; Ding, J.Y.; Ge, D. Capn4 promotes non-small cell lung cancer progression via upregulation of matrix metalloproteinase 2. Med. Oncol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, Q.; Qian, X.; Cao, Y.; Fan, M.; Xu, X.; He, X. Capn4 mRNA level is correlated with tumour progression and clinical outcome in clear cell renal cell carcinoma. J. Int. Med. Res. 2014, 42, 282–291. [Google Scholar] [CrossRef] [PubMed]
- Turner, E.S.; Turner, J.R. Expanding the Lauren classification: A new gastric cancer subtype? Gastroenterology 2013, 145, 505–508. [Google Scholar] [CrossRef] [PubMed]
- Cristescu, R.; Lee, J.; Nebozhyn, M.; Kim, K.M.; Ting, J.C.; Wong, S.S.; Liu, J.; Yue, Y.G.; Wang, J.; Yu, K.; et al. Molecular analysis of gastric cancer identifies subtypes associated with distinct clinical outcomes. Nat. Med. 2015, 21, 449–456. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K.; Hata, S.; Kawabata, Y.; Sorimachi, H. Structure, activation, and biology of calpain. Diabetes 2004, 53, S12–S18. [Google Scholar] [CrossRef] [PubMed]
- Sorimachi, H.; Hata, S.; Ono, Y. Expanding members and roles of the calpain superfamily and their genetically modified animals. Exp. Anim. 2010, 59, 549–566. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, H.; Imajoh, S.; Kawashima, S.; Hayashi, H.; Suzuki, K. The small subunits of calcium dependent proteases with different calcium sensitivities are identical. J. Biochem. 1986, 99, 1525–1532. [Google Scholar] [PubMed]
- Shimada, M.; Mahon, M.J.; Greer, P.A.; Segre, G.V. The receptor for parathyroid hormone and parathyroid hormone-related peptide is hydrolyzed and its signaling properties are altered by directly binding the calpain small subunit. Endocrinology 2005, 146, 2336–2344. [Google Scholar] [CrossRef] [PubMed]
- Rosenberger, G.; Gal, A.; Kutsche, K. αPIX associates with calpain 4, the small subunit of calpain, and has a dual role in integrin-mediated cell spreading. J. Biol. Chem. 2005, 280, 6879–6889. [Google Scholar] [CrossRef] [PubMed]
- Peng, P.; Wu, W.; Zhao, J.; Song, S.; Wang, X.; Jia, D.; Shao, M.; Zhang, M.; Li, L.; Wang, L.; et al. Decreased expression of Calpain-9 predicts unfavorable prognosis in patients with gastric cancer. Sci. Rep. 2016. [Google Scholar] [CrossRef] [PubMed]
- Rios-Doria, J.; Kuefer, R.; Ethier, S.P.; Day, M.L. Cleavage of β-catenin by calpain in prostate and mammary tumor cells. Cancer Res. 2004, 64, 7237–7240. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Mendes, D.E.; Berkman, C.E. Prolonged androgen deprivation leads to overexpression of calpain 2: Implications for prostate cancer progression. Int. J. Oncol. 2014, 44, 467–472. [Google Scholar] [PubMed]
- Cuevas, B.D.; Abell, A.N.; Witowsky, J.A.; Yujiri, T.; Johnson, N.L.; Kesavan, K.; Ware, M.; Jones, P.L.; Weed, S.A.; DeBiasi, R.L.; et al. MEKK1 regulates calpain-dependent proteolysis of focal adhesion proteins for rear-end detachment of migrating fibroblasts. EMBO J. 2003, 22, 3346–3355. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Yin, J.; Wang, X.; Shao, M.; Duan, F.; Wu, W.; Peng, P.; Jin, J.; Tang, Y.; Ruan, Y.; et al. C-type lectin-like receptor 2 suppresses AKT signaling and invasive activities of gastric cancer cells by blocking expression of phosphoinositide 3-kinase subunits. Gastroenterology 2016. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Min, L.; Wang, X.; Zhao, J.; Chen, H.; Qin, J.; Chen, W.; Shen, Z.; Tang, Z.; Gan, Q.; et al. Loss of RACK1 promotes metastasis of gastric cancer by inducing a miR-302c/IL8 signaling loop. Cancer Res. 2015, 75, 3832–3841. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Qiu, P.; Ji, Y. TCGA-assembler: Open-source software for retrieving and processing TCGA data. Nat. Methods 2014, 11, 599–600. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.D.; Oshlack, A. A scaling normalization method for differential expression analysis of RNA-seq data. Genome Biol. 2010, 11, 25–34. [Google Scholar] [CrossRef] [PubMed]

| Calpain-4 Expression | ||||
|---|---|---|---|---|
| Variables | No. | Low | High | p-Value * |
| No. (%) | No. (%) | |||
| Gender | ||||
| Male | 112 | 45 (40.2) | 67 (59.8) | 0.666 |
| Female | 62 | 27 (43.5) | 35 (56.5) | |
| Age | ||||
| <60 | 84 | 32 (38.1) | 52 (61.9) | 0.395 |
| ≥60 | 90 | 40 (44.4) | 50 (55.6) | |
| Tumor site | ||||
| Cardia | 25 | 14 (56.0) | 11 (44.0) | 0.219 |
| Body | 38 | 13 (34.2) | 25 (65.8) | |
| Antrum | 111 | 45 (40.5) | 66 (59.5) | |
| Lauren type | ||||
| Intestinal | 109 | 47 (43.1) | 62 (56.9) | 0.808 |
| Mixture | 12 | 5 (41.7) | 7 (58.3) | |
| Diffuse | 53 | 20 (37.7) | 33 (62.3) | |
| Differentiation | ||||
| Well/Moderately | 34 | 17 (50.0) | 17 (50.0) | 0.255 |
| Poorly | 140 | 55 (39.3) | 85 (60.7) | |
| Vessel invasion | ||||
| Positive | 37 | 9 (24.3) | 28 (75.7) | 0.018 |
| Negative | 137 | 63 (46.0) | 74 (54.0) | |
| T stage | ||||
| T1–T2 | 43 | 23 (53.5) | 20 (46.5) | 0.063 |
| T3–T4 | 131 | 49 (37.4) | 82 (62.6) | |
| Lymph node metastasis | ||||
| Positive | 62 | 12 (19.4) | 50 (80.6) | <0.001 |
| Negative | 112 | 60 (53.6) | 52 (46.4) | |
| Distant metastasis | ||||
| M1 | 9 | 1 (11.1) | 8 (88.9) | 0.122 |
| M0 | 165 | 71 (43.0) | 94 (57.0) | |
| TNM stage | ||||
| I | 34 | 21 (61.8) | 13 (38.2) | 0.006 |
| II | 39 | 19 (48.7) | 20 (51.3) | |
| III | 92 | 31 (33.7) | 61 (66.3) | |
| IV | 9 | 1 (11.1) | 8 (88.9) | |
| Variables | Univariate | ||
|---|---|---|---|
| HR | 95% CI | p-Value | |
| Gender | |||
| Male vs. female | 0.866 | 0.541–1.385 | 0.547 |
| Age (years) | |||
| ≥60 vs. <60 | 1.436 | 0.923–2.235 | 0.108 |
| Tumor site | |||
| Cardia + body vs. antrum | 1.440 | 0.905–2.290 | 0.124 |
| Lauren type | |||
| Diffuse + mixture vs. intestinal | 1.029 | 0.642–1.651 | 0.905 |
| Differentiation | |||
| Poorly vs. well/moderately | 1.264 | 0.725–2.200 | 0.409 |
| Vessel invasion | |||
| Positive vs. negative | 1.861 | 1.052–3.293 | 0.033 |
| T stage | |||
| T3–T4 vs. T1–T2 | 2.923 | 1.791–4.772 | <0.001 |
| Lymph node metastasis | |||
| Positive vs. negative | 4.838 | 2.923–8.007 | <0.001 |
| Distant metastasis | |||
| M1 vs. M0 | 14.93 | 4.203–53.01 | <0.001 |
| TNM stage | |||
| III–IV vs. I–II | 3.376 | 2.166–5.261 | <0.001 |
| Calpain-4 expression | |||
| High vs. low | 2.594 | 1.664–4.043 | <0.001 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peng, P.; Min, L.; Song, S.; Zhao, J.; Li, L.; Yang, C.; Shao, M.; Zhang, M.; Wu, H.; Zhang, J.; et al. Elevated Expression of Calpain-4 Predicts Poor Prognosis in Patients with Gastric Cancer after Gastrectomy. Int. J. Mol. Sci. 2016, 17, 1612. https://doi.org/10.3390/ijms17101612
Peng P, Min L, Song S, Zhao J, Li L, Yang C, Shao M, Zhang M, Wu H, Zhang J, et al. Elevated Expression of Calpain-4 Predicts Poor Prognosis in Patients with Gastric Cancer after Gastrectomy. International Journal of Molecular Sciences. 2016; 17(10):1612. https://doi.org/10.3390/ijms17101612
Chicago/Turabian StylePeng, Peike, Lingqiang Min, Shushu Song, Junjie Zhao, Lili Li, Caiting Yang, Miaomiao Shao, Mingming Zhang, Hao Wu, Jie Zhang, and et al. 2016. "Elevated Expression of Calpain-4 Predicts Poor Prognosis in Patients with Gastric Cancer after Gastrectomy" International Journal of Molecular Sciences 17, no. 10: 1612. https://doi.org/10.3390/ijms17101612
APA StylePeng, P., Min, L., Song, S., Zhao, J., Li, L., Yang, C., Shao, M., Zhang, M., Wu, H., Zhang, J., Li, C., Wang, X., Wang, H., Qin, J., Ruan, Y., & Gu, J. (2016). Elevated Expression of Calpain-4 Predicts Poor Prognosis in Patients with Gastric Cancer after Gastrectomy. International Journal of Molecular Sciences, 17(10), 1612. https://doi.org/10.3390/ijms17101612