Identification of Resistance to Wet Bubble Disease and Genetic Diversity in Wild and Cultivated Strains of Agaricus bisporus
Abstract
:1. Introduction
2. Results
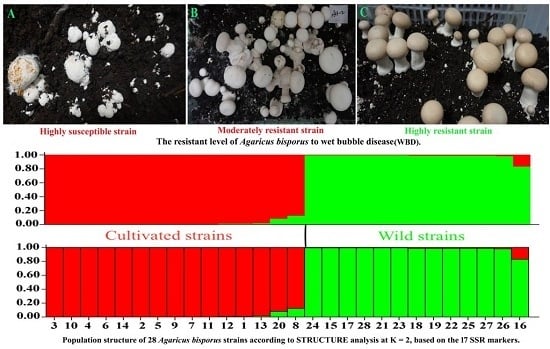
2.1. Identification of WBD Resistance in A. bisporus Strains
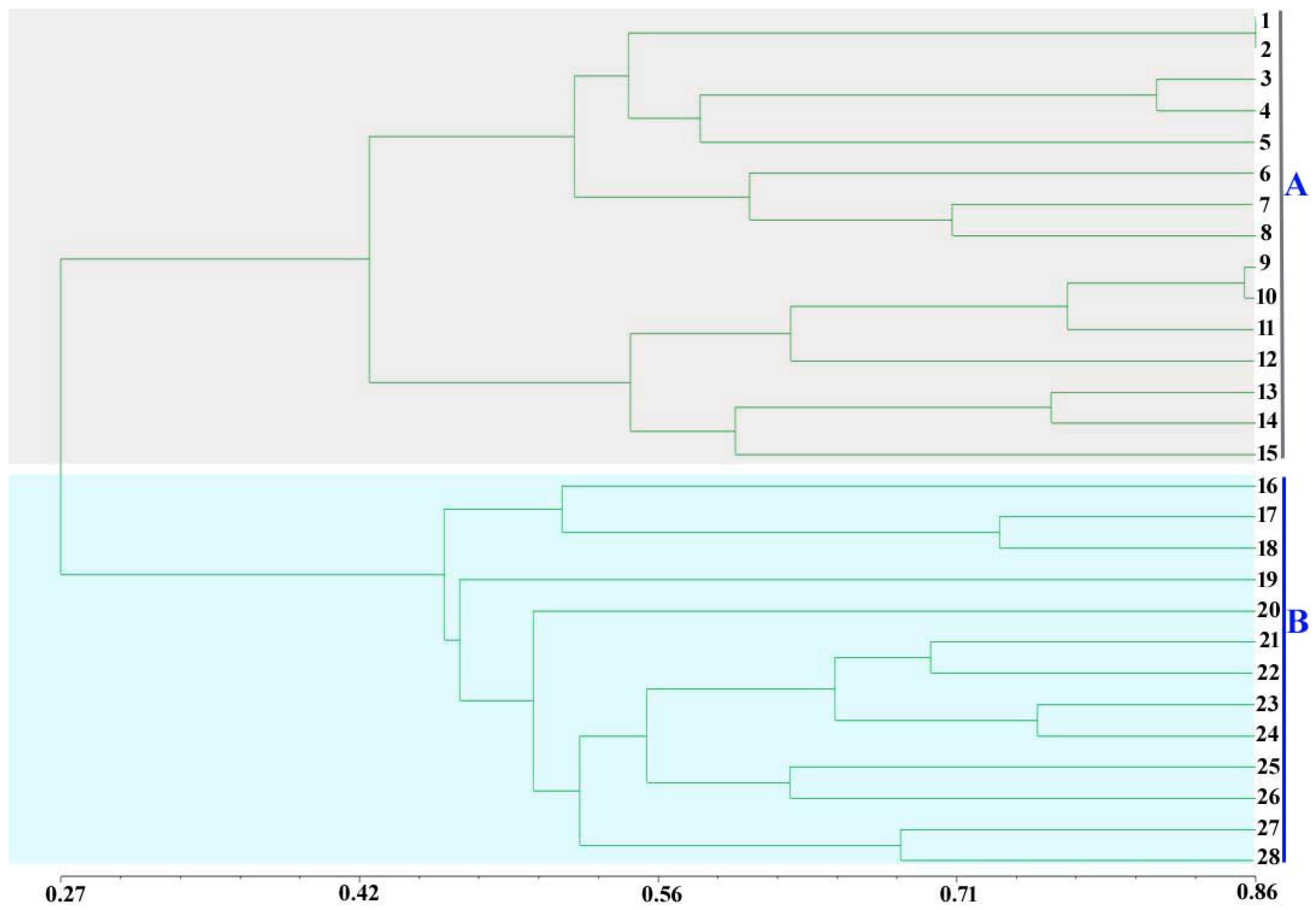
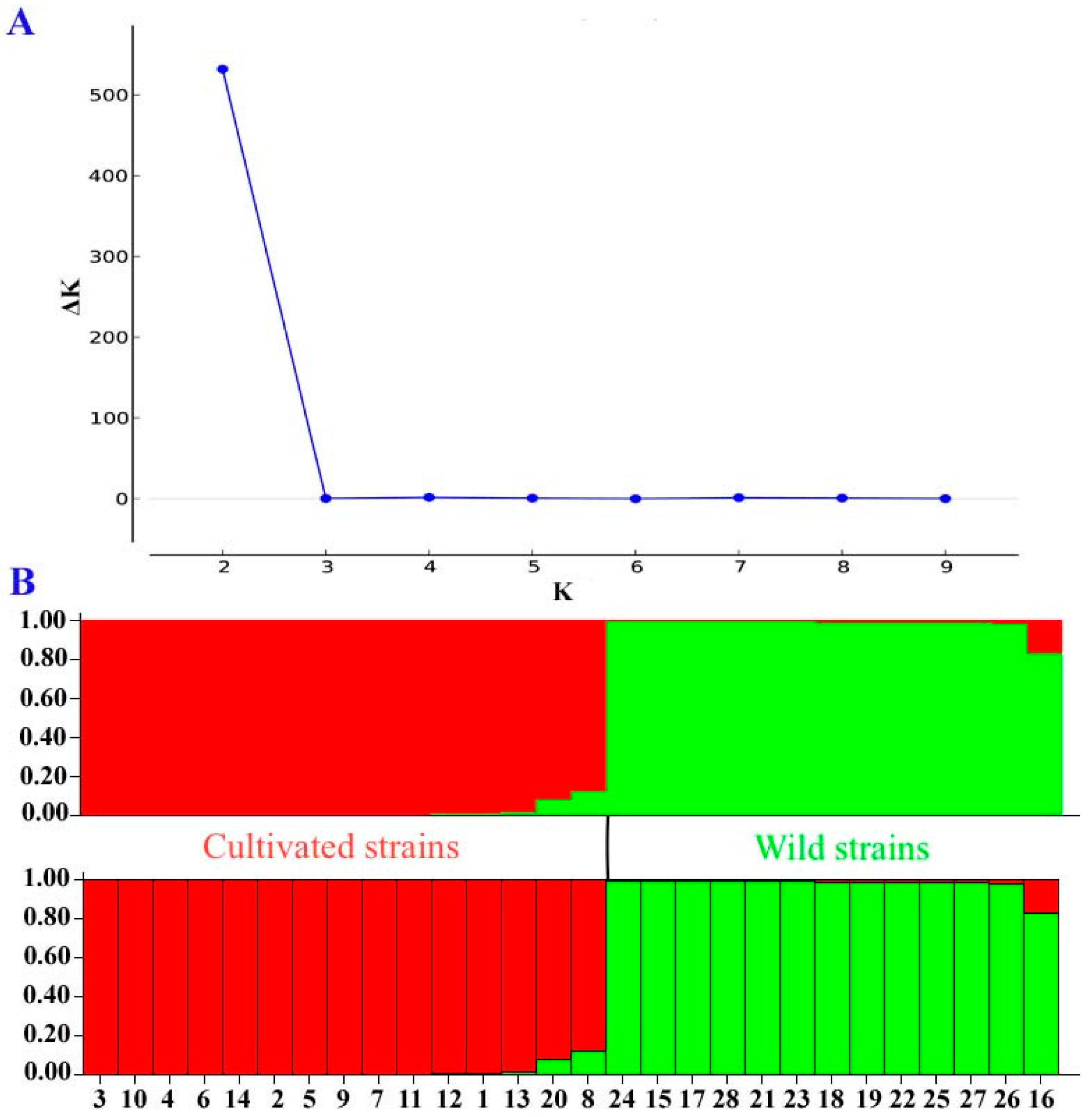
2.2. Assessment of Genetic Diversity in A. bisporus Using New SSR Markers
3. Discussion
4. Materials and Methods
4.1. M. perniciosa and A. bisporus Strains
4.2. Evaluation of A. bisporus Strains for Resistance to WBD under Controlled Environmental Conditions
4.3. Identification of the Pathogen Using Koch’s Postulates
4.4. Development of SSR Markers for A. bisporus
4.5. Identification of Genetic Diversity for the 28 A. bisporus Strains
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| WBD | Wet Bubble Disease |
| SSR | Simple Sequence Repeat |
| HS | High Susceptible |
| MR | Middle Resistant |
| HR | High Resistant |
| ITS | Internal Transcribed Spacers |
| PCR | Polymerase Chain Reaction |
| QTL | Quantitative Trait Locus |
| MISA | MIcroSAtellite |
References
- Glamočlija, J.; Soković, M.; LJALJEVIĆ-GRBIĆ, M.; Vukojević, J.; Milenković, I.; van Griensven, L. Morphological characteristics and mycelial compatibility of different Mycogone perniciosa isolates. J. Microsc. 2008, 232, 489–492. [Google Scholar] [CrossRef] [PubMed]
- Bueno, F.S.; Romão, A.; Wach, M.; Araùjo, W.; Azevedo, L. Variability in commercial and wild isolates of Agaricus species in Brazil. Mushroom Sci. 2008, 17, 135–145. [Google Scholar]
- Sonnenberg, A.; Johan, J.; Hendrickx, P.; Lavrijssen, B.; Wei, G.; Weijn, A.; Mes, J.; Savoie, J.; Foulongne-Oriol, M.; Largeteau, M. Breeding and strain protection in the button mushroom Agaricus bisporus, Mushroom biology and mushroom products. In Proceedings of the 7th International Conference on Mushroom Biology and Mushroom Products, Arcachon, France, 4–7 October 2011; Institute National de la Recherche Agronomique (INRA): Paris, France, 2011; Volume 1, pp. 7–15. [Google Scholar]
- Fletcher, J.; White, P.; Gaze, R. Mushrooms: Pest and Disease Control; Intercept Andover: Andover, UK, 1989. [Google Scholar]
- Largeteau, M.L.; Savoie, J.M. Microbially induced diseases of Agaricus bisporus: Biochemical mechanisms and impact on commercial mushroom production. Appl. Microbiol. Biotechnol. 2010, 86, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Kouser, S.; Ahmed, M.; Shah, S. Disease status and yield losses due to wet bubble disease (Mycogone perniciosa) associated with the cultivation of white button mushroom at different mushroom units of Kashmir valley. Plant Pathol. J. 2013, 12, 104–109. [Google Scholar]
- Fletcher, J.; Jaffe, B.; Muthumeenakshi, S.; Brown, A.; Wright, D. Variations in isolates of Mycogone perniciosa and in disease symptoms in Agaricus bisporus. Plant Pathol. 1995, 44, 130–140. [Google Scholar] [CrossRef]
- Umar, M.; Geels, F.; van Griensven, L. Pathology and pathogenesis of Mycogone perniciosa infection of Agaricus bisporus. Mushroom Sci. 2000, 15, 561–567. [Google Scholar]
- Sharma, S.; Kumar, S. Studies on wet bubble disease of white button mushrooms (Agaricus bisporus) caused by Mycogone perniciosa. Mushroom Sci. 2000, 15, 569–575. [Google Scholar]
- Ozaktan, T.B. Biological control of some important mushroom diseases in Turkey by fluorescent pseudomonads. In Science and Cultivation of Edible Fungi, Proceedings of the 15th International Congress on the Science and Cultivation of Edible Fungi, Maastricht, The Netherlands, 15–19 May 2000.
- Smith, F. Three diseases of cultivated mushrooms. Trans. Br. Mycol. Soc. 1924, 10, 81IN84–97IN85. [Google Scholar] [CrossRef]
- Wen, Z.Q.; Wang, Y.X.; Liu, X.R.; Xie, B.G. Population differetiaton of Mycogone perniciosa in Fujian, China. Mycosystema 2010, 29, 329–334. [Google Scholar]
- Zhou, C.; Li, D.; Chen, L.; Li, Y. Genetic diversity analysis of Mycogone perniciosa causing wet bubble disease of Agaricus bisporus in china using SRAP. J. Phytopathol. 2015, 164, 271–275. [Google Scholar] [CrossRef]
- Zhao, X.C.; Luo, Y.F.; Chen, Z.S. Occurrence and control of Mycogone pernicios Magn attacting Agaricus bisporus. Edible Fungi Jiangsu 1995, 16, 34–35. [Google Scholar]
- Kouser, S.; Shah, S. Isolation and identification of Mycogone perniciosa, causing wet bubble disease in Agaricus bisporus cultivation in Kashmir. Afr. J. Agric. Res. 2013, 8, 4804–4809. [Google Scholar]
- Kouser, S.; Shah, S.; Ahmed, M.; Shah, M.; Sheikh, P. Morphological characteristics of wet bubble disease (Mycogone perniciosa) isolated from button mushroom (Agaricus bisporus) and assessment of factors affecting disease development and spread. Afr. J. Microbiol. Res. 2015, 9, 185–193. [Google Scholar]
- Glamočlija, J.M.; Soković, M.D.; Ljaljević-Grbić, M.V.; Vukojević, J.B.; Milenković, I.M. Morpho-physiological characteristics and interactions of isolates of Mycogone perniciosa (magnus) delacr. Proc. Nat. Sci. 2007, 235–241. [Google Scholar] [CrossRef]
- Siwulski, M.; Sobieralski, K.; Górski, R.; Lisiecka, J.; Sas-Golak, I. Temperature and ph impact on the mycelium growth of Mycogone perniciosa and Verticillium fungicola isolates derived from polish and foreign mushroom growing houses. J. Plant Prot. Res. 2011, 51, 268–272. [Google Scholar] [CrossRef]
- Chakravarty, B. Trends in mushroom cultivation and breeding. Aust. J. Agric. Eng. 2011, 2, 102–109. [Google Scholar]
- Wang, Y.X. Study on Biological Characteristics and the Population Differentiation of Mycogone perniciosa Magn. Master's Thesis, Fujian Agriculture and Forestry University, Fuzhou, China, 2008. [Google Scholar]
- Sonnenberg, A.; van Griensven, L. Genetics and breeding of Agaricus bisporus. Mushroom Sci. 2000, 15, 25–39. [Google Scholar]
- Wang, Z.S.; Chen, M.Y.; Cai, Z.; Liao, J.H.; Li, H.R.; Guo, Z.J.; Lu, Z.H. DNA fingerprinting of genetic diversity of Agaricus bisporus. In Proceedings of the 7th International Conference on Mushroom Biology and Mushroom Products, Arcachon, France, 4–7 October 2011; Institute National de la Recherche Agronomique (INRA): Paris, France, 2011; Volume 1, pp. 1–8. [Google Scholar]
- Foulongne, O.M.; Murat, C.; Castanera, R.; Ramírez, L.; Sonnenberg, A.S. Genome-wide survey of repetitive DNA elements in the button mushroom Agaricus bisporus. Fungal Genet. Biol. 2013, 55, 6–21. [Google Scholar] [CrossRef] [PubMed]
- Kerrigan, R.W.; Ross, I.K. Allozymes of a wild Agaricus bisporus population: New alleles, new genotypes. Mycologia 1989, 433–443. [Google Scholar] [CrossRef]
- Kerrigan, R.W. Evidence of genetic divergence in two populations of Agaricus bisporus. Mycol. Res. 1990, 94, 721–733. [Google Scholar] [CrossRef]
- Loftus, M.; Moore, D.; Elliott, T. DNA polymorphisms in commercial and wild strains of the cultivated mushroom, Agaricus bisporus. Theor. Appl. Genet. 1988, 76, 712–718. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.S.; Guo, Z.J. Study on the biological characteristics of wild Agaricus bisporus strains from China. Mushroom Sci. 2008, XVII, 149–158. [Google Scholar]
- Chen, M.Y.; Liao, J.H.; Wang, B.; Li, H.R.; Lu, Z.H.; Guo, Z.J.; Can, D.F.; Wang, Z.S. DNA fingerprinting of ninety wild strains of Agaricus from China. Acta Edulis Fungi 2009, 16, 11–16. [Google Scholar]
- Olivier, J.; Mamoun, M.; Munsch, P. Standardization of a method to assess mushroom blotch resistance in cultivated and wild Agaricus bisporus strains. Can. J. Plant Pathol. 1997, 19, 36–42. [Google Scholar] [CrossRef]
- Moquet, F.; Desmerger, C.; Mamoun, M.; Ramos-Guedes-Lafargue, M.; Olivier, J.M. A quantitative trait locus of Agaricus bisporus resistance to pseudomonas tolaasii is closely linked to natural cap color. Fungal Genet. Biol. 1999, 28, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Rokni, N.; Goltapeh, M.E.; Shafeinia, A.; Safaie, N. Evaluation of genetic diversity among some commercial cultivars and Iranian wild strains of Agaricus bisporus by microsatellite markers. Bonaty 2016, 94, 9–13. [Google Scholar]
- Wang, Y.Z.; Cao, L.J.; Zhu, J.Y.; Wei, S.J. Development and characterization of novel microsatellite markers for the peach fruit moth Carposina sasakii (lepidoptera: Carposinidae) using next-generation sequencing. Int. J. Mol. Sci. 2016, 17, 362. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Xu, Y.; He, J.; Zhang, S.; Wang, Y.; Lu, P. Genetic diversity and population structure of Broomcorn Millet (Panicum miliaceum L.) cultivars and landraces in china based on microsatellite markers. Int. J. Mol. Sci. 2016, 17, 370. [Google Scholar] [CrossRef] [PubMed]
- Dutech, C.; Enjalbert, J.; Fournier, E.; Delmotte, F.; Barres, B.; Carlier, J.; Tharreau, D.; Giraud, T. Challenges of microsatellite isolation in fungi. Fungal Genet. Biol. 2007, 44, 933–949. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chen, M.; Wang, H.; Wang, J.F.; Bao, D. Microsatellites in the genome of the edible mushroom, Volvariella volvacea. Biol. Med. Res. Int. 2014, 2014, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Foulongne-Oriol, M.; Spataro, C.; Savoie, J.M. Novel microsatellite markers suitable for genetic studies in the white button mushroom Agaricus bisporus. Appl. Microbiol. Biol. 2009, 84, 1125–1135. [Google Scholar] [CrossRef] [PubMed]
- Callac, P. Breeding of edible fungi with emphasis on the variability among French genetic resources of Agaricus bisporus. Can. J. Bot. 1995, 73, 980–986. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. PCR Protoc. 1990, 18, 315–322. [Google Scholar]
- Fu, Y.P.; Liang, Y.; Dai, Y.T.; Yang, C.T.; Duan, M.Z.; Zhang, Z.; Hu, S.N.; Zhang, Z.W.; Li, Y. De novo sequencing and transcriptome analysis of Pleurotus eryngii subsp. tuoliensis (bailinggu) mycelia in response to cold stimulation. Molecules 2016, 21, 560. [Google Scholar] [CrossRef]
- Earl, D.A. Structure harvester: A website and program for visualizing structure output and implementing the evanno method. Conserv. Genet. Resour. 2012, 4, 359–361. [Google Scholar] [CrossRef]
- Evanno, G.; Regnaut, S.; Goudet, J. Detecting the number of clusters of individuals using the software structure: A simulation study. Mol. Ecol. 2005, 14, 2611–2620. [Google Scholar] [CrossRef] [PubMed]

| Strain Name | Original Reference | Origin | Pileus Color | Strain Types | Resistance Level a |
|---|---|---|---|---|---|
| CCMJ1009 | A15 | America | White | Cultivated strain | HS |
| CCMJ1013 | As2796 | Fujian, China | White | Cultivated strain | HS |
| CCMJ1020 | ZA | Germany | White | Cultivated strain | HS |
| CCMJ1021 | S130A | America | White | Cultivated strain | HS |
| CCMJ1022 | As4580 | Fujian, China | White | Cultivated strain | HS |
| CCMJ1028 | S46 | Fujian, China | White | Cultivated strain | HS |
| CCMJ1033 | C13 | America | Brown | Cultivated strain | HR |
| CCMJ1035 | 72 | America | White | Cultivated strain | HS |
| CCMJ1037 | U1 | The Netherlands | White | Cultivated strain | HS |
| CCMJ1038 | PSU310 | America | White | Cultivated strain | HS |
| CCMJ1039 | 126 | The Netherlands | White | Cultivated strain | MR |
| CCMJ1053 | M-1 | Spain | White | Cultivated strain | MR |
| CCMJ1106 | 2094 | Tibet, China | Brown | Wild stain | HR |
| CCMJ1109 | Ag23 | England | White | Cultivated strain | HS |
| CCMJ1110 | Dashan I | Shanxi, China | Brown | Wild stain | HS |
| CCMJ1343 | W192 | Fujian, China | White | Cultivated strain | HS |
| CCMJ1347 | T12387 | Yunnan, China | White | Wild stain | HR |
| CCMJ1350 | W1 | Sichuan, China | Brown | Wild stain | HS |
| CCMJ1351 | W2 | Sichuan, China | Brown | Wild stain | HR |
| CCMJ1352 | A12 | America | White | Cultivated strain | HS |
| CCMJ1360 | W3 | Sichuan, China | Brown | Wild stain | HR |
| CCMJ1361 | W4 | Sichuan, China | Brown | Wild stain | HR |
| CCMJ1363 | W5 | Sichuan, China | Brown | Wild stain | HR |
| CCMJ1369 | W6 | Sichuan, China | Brown | Wild stain | HR |
| CCMJ1372 | W7 | Sichuan, China | Brown | Wild stain | HR |
| CCMJ1377 | W8 | Sichuan, China | Brown | Wild stain | HR |
| CCMJ1381 | W9 | Sichuan, China | Brown | Wild stain | HR |
| CCMJ1384 | W10 | Sichuan, China | Brown | Wild stain | HS |
| Locus | SSR motif | Primer Sequence (5′–3′) | Ta (°C) | Na | Ne | Ho | He | PIC | I |
|---|---|---|---|---|---|---|---|---|---|
| AbSSR05 | (GATGAG)6 | F-CTCTGGGATATGGACGAGGA R-CCTCTTCACCTTGACCCTCA | 56 | 5 | 3.2197 | 0.5 | 0.702 | 0.635 | 1.3177 |
| AbSSR08 | (TGG)8 | F-GTAATGCTCCCGCTGTTGAT R-TCCGCTGTTCTTCCAACTCT | 56 | 4 | 2.3939 | 0.107 | 0.593 | 0.531 | 1.0785 |
| AbSSR10 | (CCA)8 | F-GAAGAATCACGGGTGAAGGA R-GAGGGCGATGTGACAGTTTT | 56 | 8 | 6.1455 | 0.423 | 0.854 | 0.816 | 1.905 |
| AbSSR14 | (TACC)6 | F-GGCAATCGGAAAGAACAAAA R-GCAGAGAACCATCCTCAACG | 56 | 7 | 4.2757 | 0.333 | 0.781 | 0.73 | 1.6081 |
| AbSSR15 | (TA)6 | F-GACTGCCTGATTGACGGATT R-TCCGACTCCGACATCCTATC | 56 | 6 | 4.2609 | 0.393 | 0.779 | 0.729 | 1.5613 |
| AbSSR17 | (CA)6 | F-GGACGAACTTATGCCGTGTT R-GGCACAGCCTGAGAGAGAAG | 56 | 2 | 1.6897 | 0 | 0.416 | 0.325 | 0.5983 |
| AbSSR18 | (GA)7 | F-CTCGAGTCGACGAAGGAAAC R-TCCTCGGTTTCGACTGTACC | 56 | 5 | 2.9418 | 0.786 | 0.672 | 0.611 | 1.2567 |
| AbSSR28 | (TC)12 | F-TGTCTGGTTTTGCTCACGTC R-TCAGCACACTTAATCGCACA | 56 | 4 | 2.5331 | 0.321 | 0.616 | 0.539 | 1.058 |
| AbSSR47 | (CA)8 | F-CATCGGAATCTGAGCTGTCA R-TGTGTCAAAAGTGGGTCCAA | 56 | 4 | 3.0519 | 0.308 | 0.686 | 0.607 | 1.1893 |
| AbSSR52 | (CAT)6 | F-TGGCTCTTTACAGCCTTGGT R-TGCAGATGTGGTAGGAGTTTTG | 56 | 6 | 3.3212 | 0.556 | 0.712 | 0.654 | 1.4004 |
| AbSSR75 | (CAA)7 | F-CGTCCAACATCAACGTCAAC R-GTGTACATCCCCTCGTCGTC | 56 | 6 | 5.0827 | 0.5 | 0.819 | 0.775 | 1.7012 |
| AbSSR85 | (CGT)5 | F-GACTGTTGACGTTTCGGGTT R-CAACGATGACCCGTTTTCTT | 56 | 3 | 2.1189 | 0 | 0.538 | 0.421 | 0.8165 |
| AbSSR87 | (CCT)6 | F-CAGTCGCACTCGAAATCGTA R-TTGTTGAGTGAGGCATCGAG | 56 | 5 | 1.6309 | 0.308 | 0.394 | 0.361 | 0.7984 |
| AbSSR89 | (CAT)7 | F-GATAGCTCCTGGTCACCGTC R-CTGGCTTCAAGAAGCGTACC | 56 | 6 | 4.0412 | 0.214 | 0.766 | 0.713 | 1.5111 |
| AbSSR111 | (GAG)12 | F-TGTCGATTGCGTCTTCTTTG R-CGCCTCGTTTCTCTACTTCG | 56 | 7 | 4.2151 | 0.536 | 0.777 | 0.728 | 1.6399 |
| AbSSR112 | (CAC)5 | F-TCACCCTCACTCAAACTCCC R-TCTCATCCGGTTCAACAACA | 56 | 3 | 2.8403 | 0 | 0.661 | 0.573 | 1.0695 |
| AbSSR159 | (GAA)6 | F-CGACCCATCATCAACTTCCT R-AACGAGGGAAAGGTCGATTT | 56 | 7 | 4.8246 | 0.143 | 0.807 | 0.763 | 1.697 |
| Mean | 5.1765 | 3.4463 | 0.3193 | 0.6807 | 0.6183 | 1.3063 | |||
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fu, Y.; Wang, X.; Li, D.; Liu, Y.; Song, B.; Zhang, C.; Wang, Q.; Chen, M.; Zhang, Z.; Li, Y. Identification of Resistance to Wet Bubble Disease and Genetic Diversity in Wild and Cultivated Strains of Agaricus bisporus. Int. J. Mol. Sci. 2016, 17, 1568. https://doi.org/10.3390/ijms17101568
Fu Y, Wang X, Li D, Liu Y, Song B, Zhang C, Wang Q, Chen M, Zhang Z, Li Y. Identification of Resistance to Wet Bubble Disease and Genetic Diversity in Wild and Cultivated Strains of Agaricus bisporus. International Journal of Molecular Sciences. 2016; 17(10):1568. https://doi.org/10.3390/ijms17101568
Chicago/Turabian StyleFu, Yongping, Xinxin Wang, Dan Li, Yuan Liu, Bing Song, Chunlan Zhang, Qi Wang, Meiyuan Chen, Zhiwu Zhang, and Yu Li. 2016. "Identification of Resistance to Wet Bubble Disease and Genetic Diversity in Wild and Cultivated Strains of Agaricus bisporus" International Journal of Molecular Sciences 17, no. 10: 1568. https://doi.org/10.3390/ijms17101568
APA StyleFu, Y., Wang, X., Li, D., Liu, Y., Song, B., Zhang, C., Wang, Q., Chen, M., Zhang, Z., & Li, Y. (2016). Identification of Resistance to Wet Bubble Disease and Genetic Diversity in Wild and Cultivated Strains of Agaricus bisporus. International Journal of Molecular Sciences, 17(10), 1568. https://doi.org/10.3390/ijms17101568