Effect of Chromatin Structure on the Extent and Distribution of DNA Double Strand Breaks Produced by Ionizing Radiation; Comparative Study of hESC and Differentiated Cells Lines
Abstract
:1. Introduction
2. Results
2.1. hESC Have Less Heterochromatin than Partially and Fully Differentiated Cells
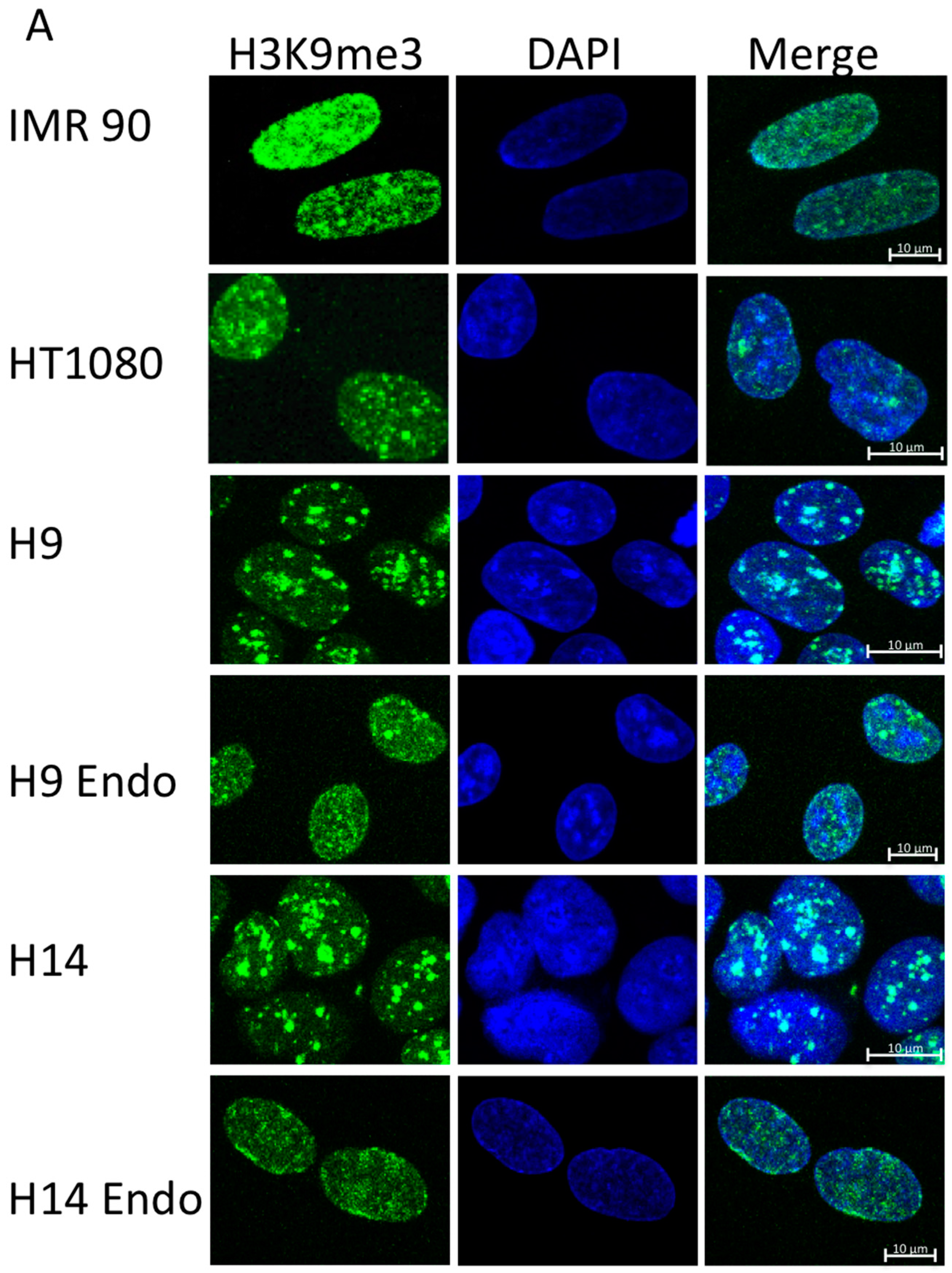
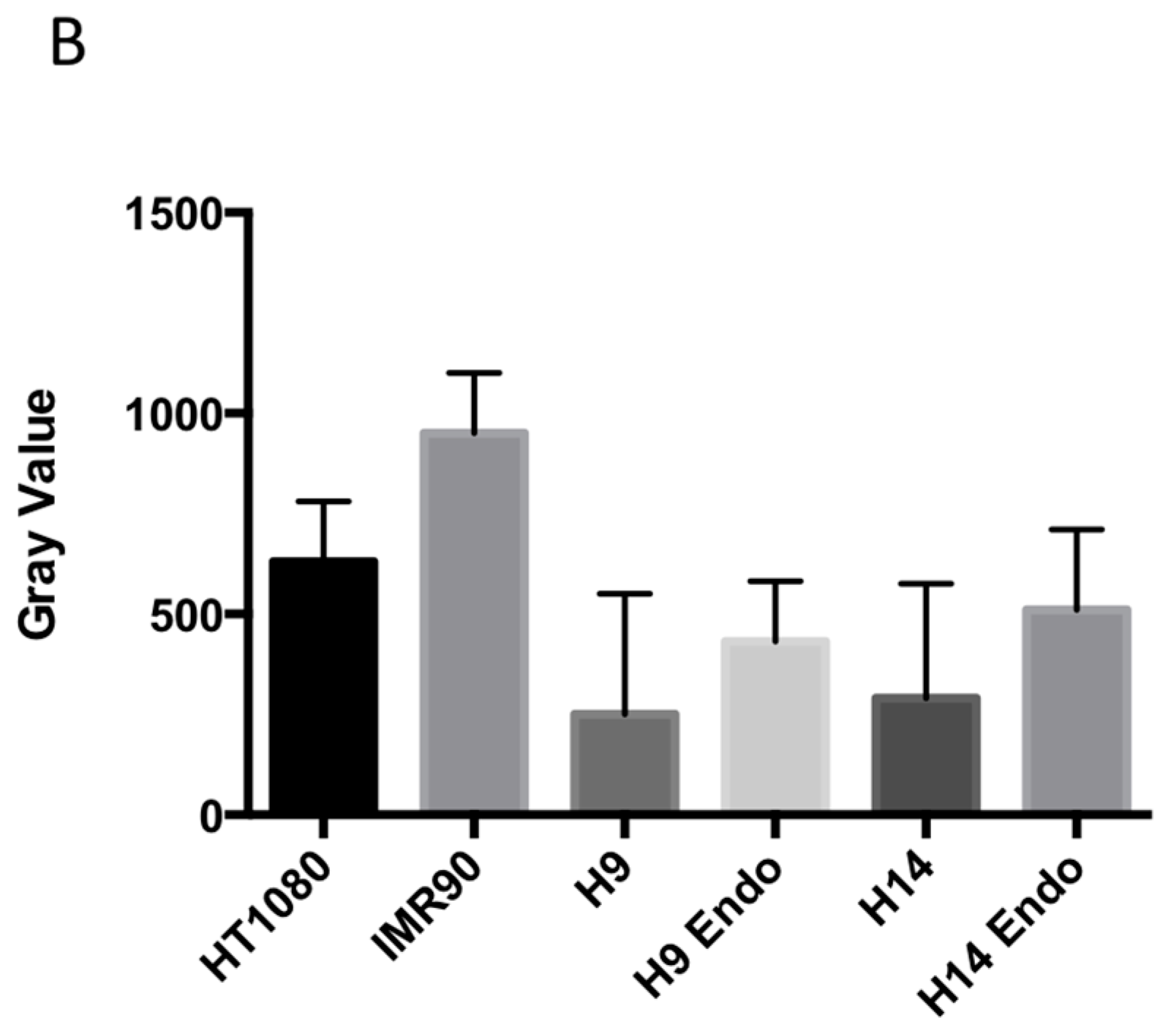
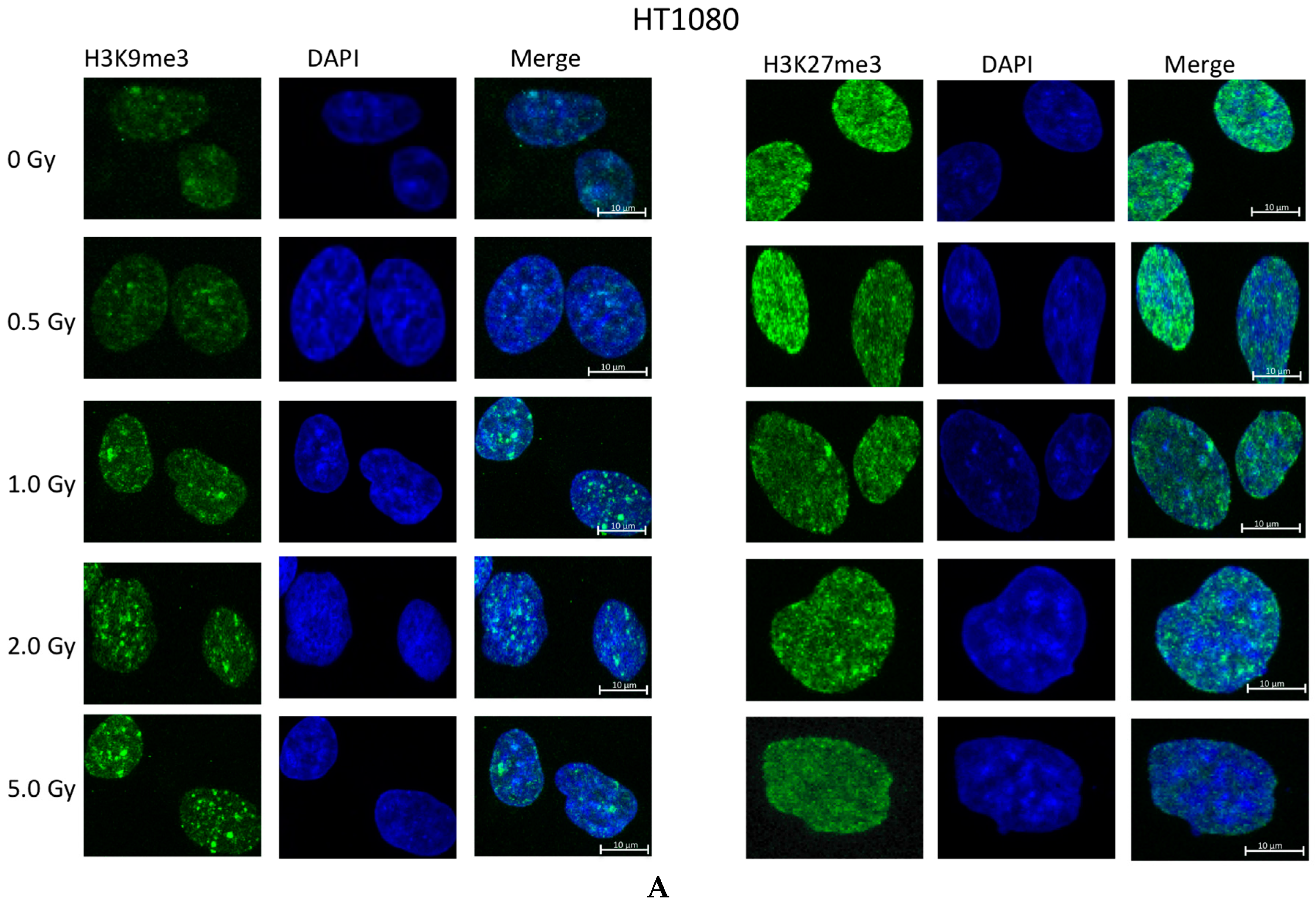
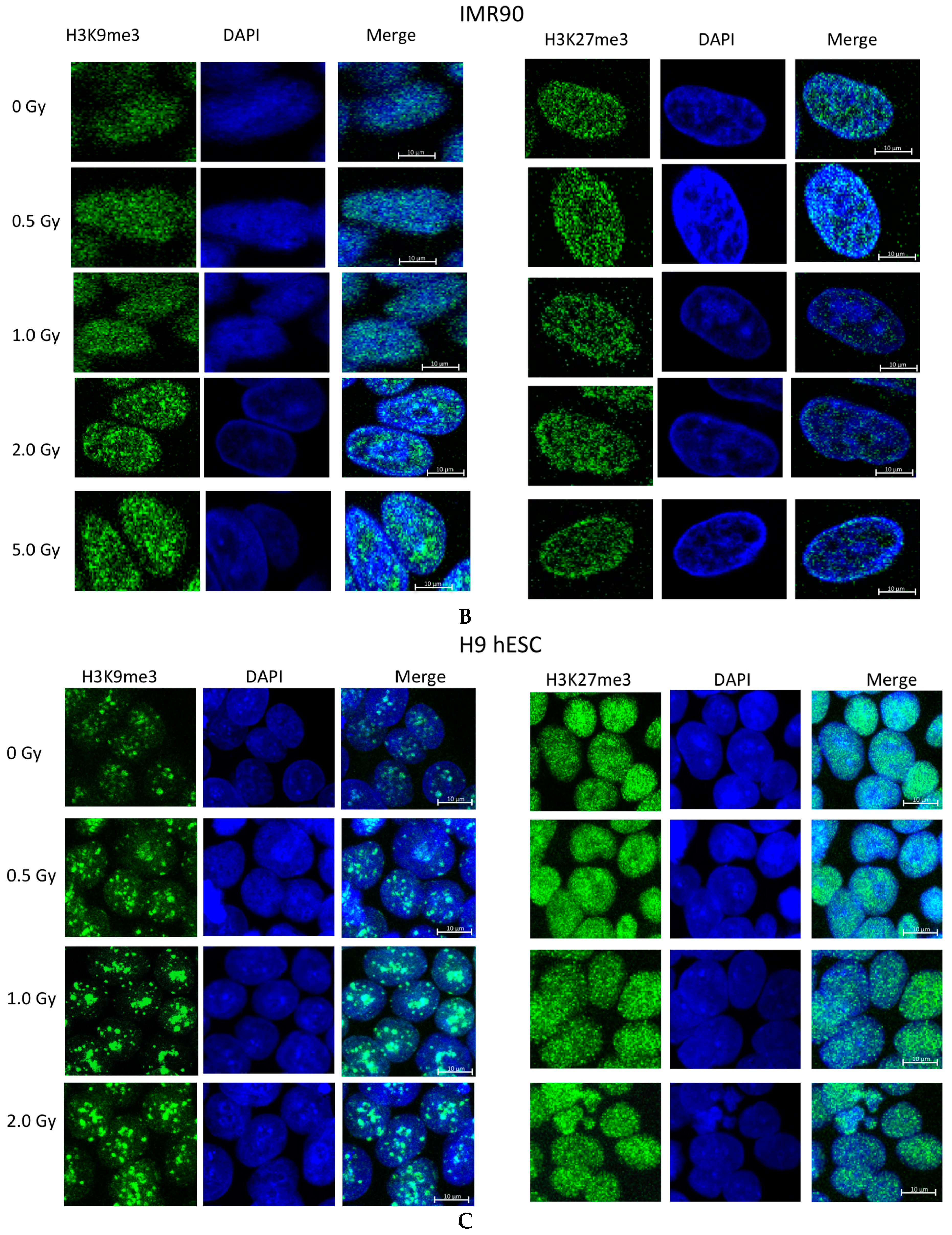
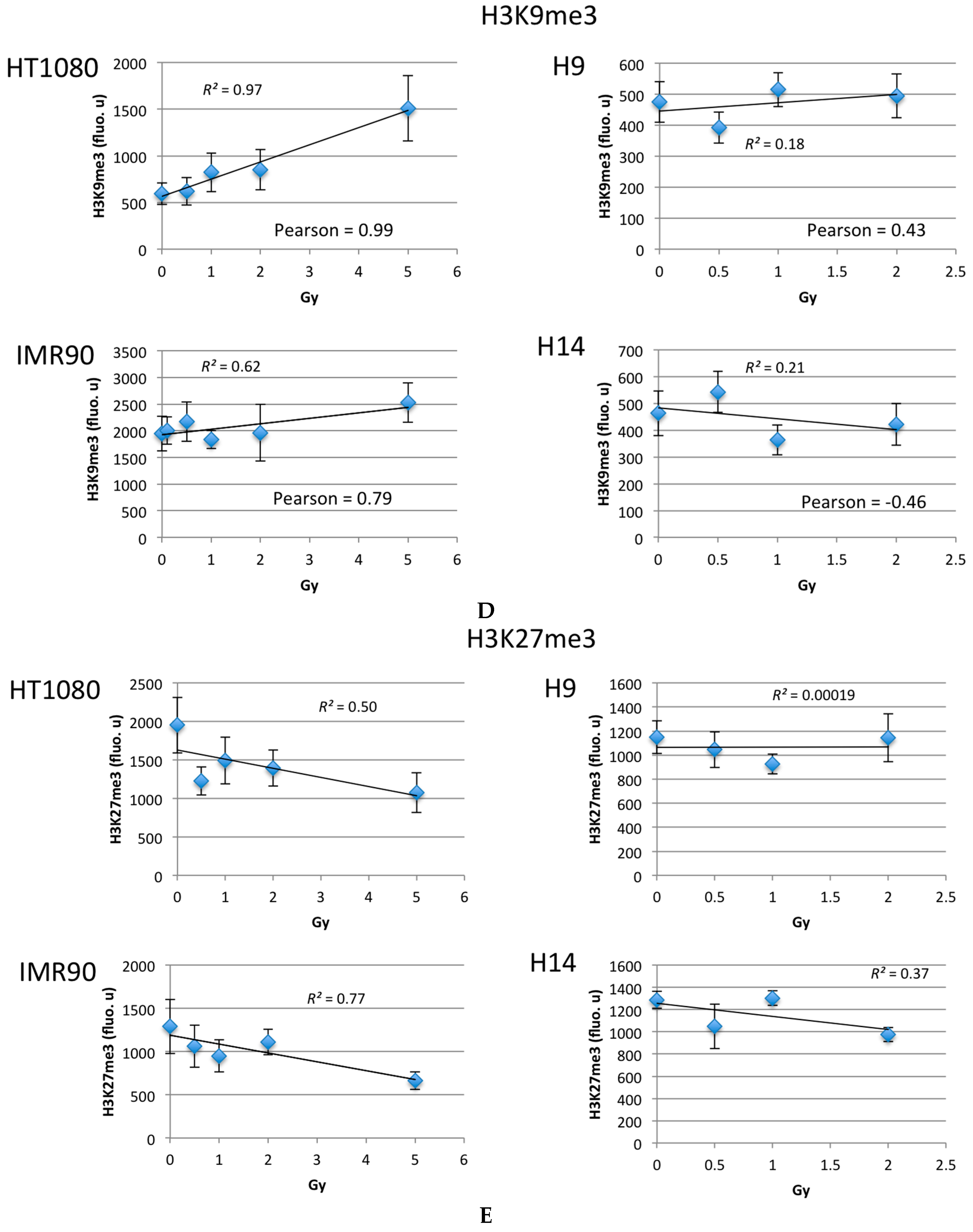
2.2. Ionizing Radiation Dose Dependent Change in Heterochromatin Staining
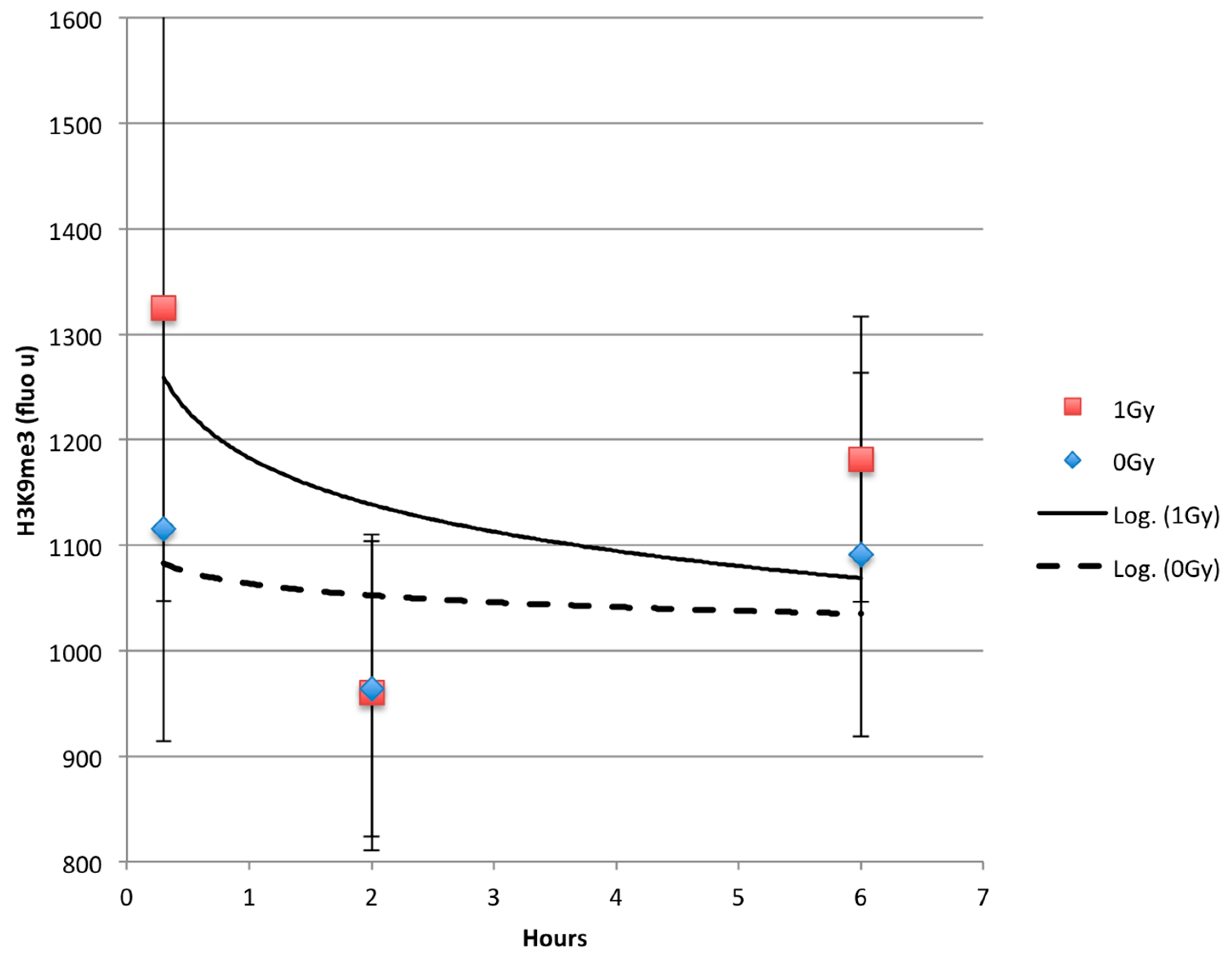
2.3. Time Dependent Recovery of HT1080 Cells after Exposure to Ionizing Radiation
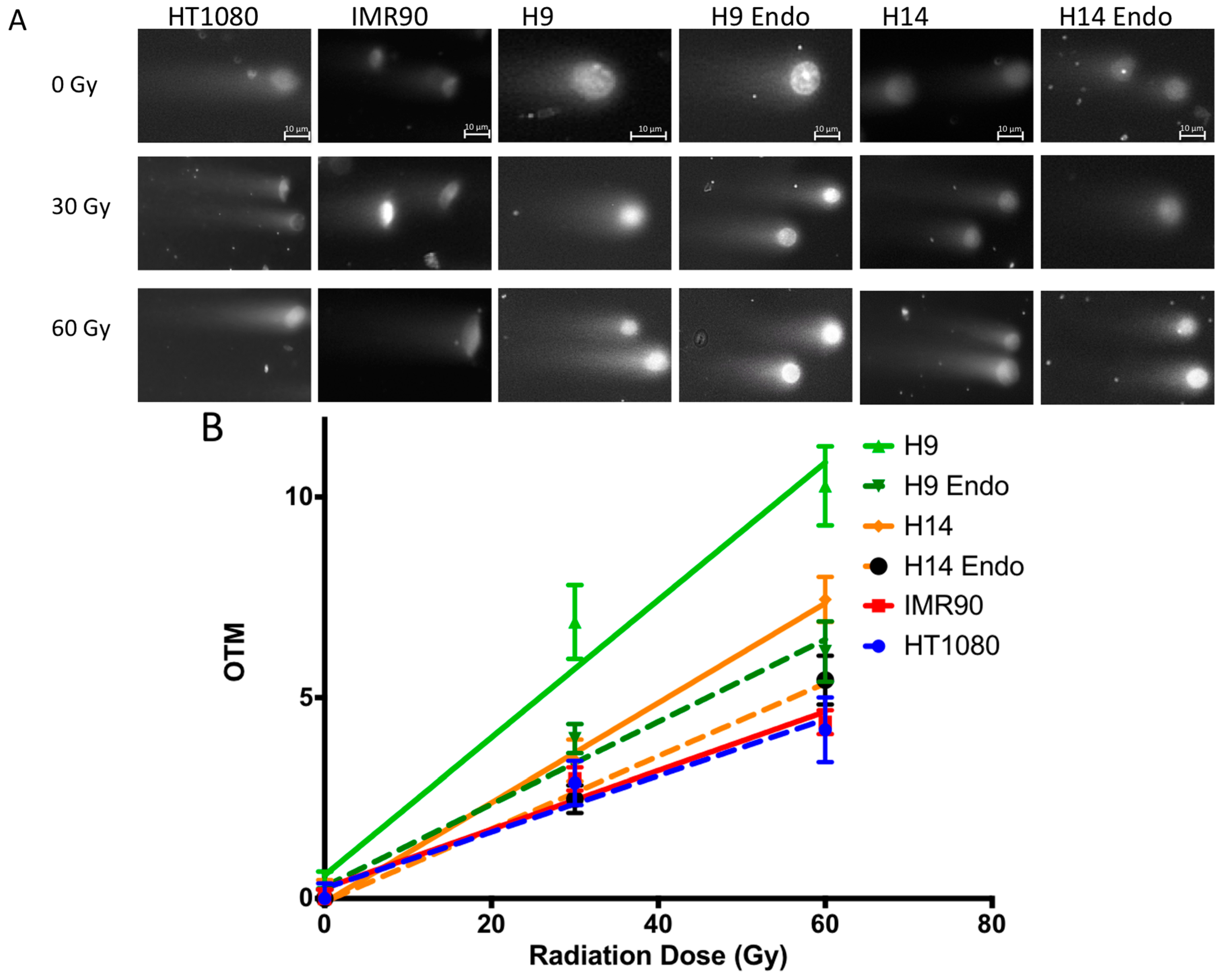
2.4. hESC Show More Double Strand Breaks after Exposure to High Doses of Ionizing Radiation
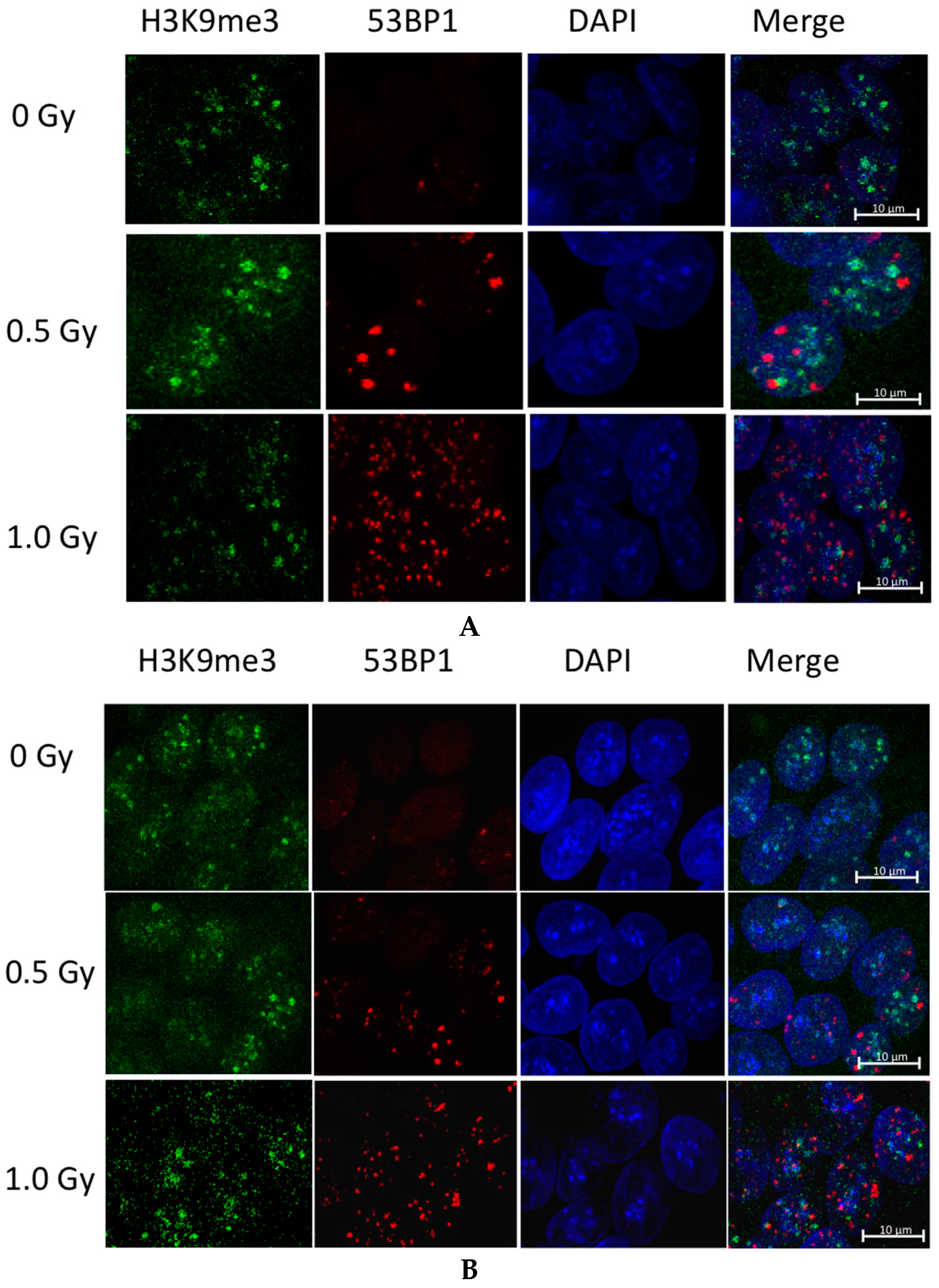
2.5. hESC Show Comparable Numbers of Radiation Induced Foci between Cell Lines after Exposure to Low Doses of IR

3. Discussion
3.1. hESC Have Less Heterochromatin than Lineage-Committed Cells
3.2. Global Changes in Chromatin Methylation after Exposure to IR
4. Experimental Section
4.1. Cell Culture
4.2. Directed Endoderm Differentiation
4.3. Irradiation
4.4. Neutral Comet Assay
4.5. Immunostaining and Imaging
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Elia, M.C.; Bradley, M.O. Influence of chromatin structure on the induction of DNA double strand breaks by ionizing radiation. Cancer Res. 1992, 52, 1580–1586. [Google Scholar] [PubMed]
- Radulescu, I.; Elmroth, K.; Stenerlow, B. Chromatin organization contributes to non-randomly distributed double-strand breaks after exposure to high-let radiation. Radiat. Res. 2004, 161, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Magnander, K.; Hultborn, R.; Claesson, K.; Elmroth, K. Clustered DNA damage in irradiated human diploid fibroblasts: Influence of chromatin organization. Radiat. Res. 2010, 173, 272–282. [Google Scholar] [CrossRef] [PubMed]
- Storch, K.; Eke, I.; Borgmann, K.; Krause, M.; Richter, C.; Becker, K.; Schrock, E.; Cordes, N. Three-dimensional cell growth confers radioresistance by chromatin density modification. Cancer Res. 2010, 70, 3925–3934. [Google Scholar] [CrossRef] [PubMed]
- Meshorer, E.; Misteli, T. Chromatin in pluripotent embryonic stem cells and differentiation. Nat. Rev. Mol. Cell Biol. 2006, 7, 540–546. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; Urata, S.M.; Aguilera, J.A.; Perry, C.C.; Milligan, J.R. Modeling the influence of histone proteins on the sensitivity of DNA to ionizing radiation. Radiat. Res. 2012, 177, 152–163. [Google Scholar] [CrossRef] [PubMed]
- Chinnaiyan, P.; Vallabhaneni, G.; Armstrong, E.; Huang, S.M.; Harari, P.M. Modulation of radiation response by histone deacetylase inhibition. Int. J. Radiat. Oncol. Biol. Phys. 2005, 62, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Camphausen, K. Enhanced radiation-induced cell killing and prolongation of γH2AX foci expression by the histone deacetylase inhibitor MS-275. Cancer Res. 2004, 64, 316–321. [Google Scholar] [CrossRef] [PubMed]
- De Schutter, H.; Nuyts, S. Radiosensitizing potential of epigenetic anticancer drugs. Anti-Cancer Agents Med. Chem. 2009, 9, 99–108. [Google Scholar] [CrossRef]
- Bartova, E.; Krejci, J.; Harnicarova, A.; Kozubek, S. Differentiation of human embryonic stem cells induces condensation of chromosome territories and formation of heterochromatin protein 1 foci. Differ. Res. Biol. Divers. 2008, 76, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Clarke, M.F.; Fuller, M. Stem cells and cancer: Two faces of eve. Cell 2006, 124, 1111–1115. [Google Scholar] [CrossRef] [PubMed]
- Oh, I.H.; Humphries, R.K. Concise review: Multidimensional regulation of the hematopoietic stem cell state. Stem Cells 2012, 30, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Stancheva, I. Revisiting heterochromatin in embryonic stem cells. PLoS Genet. 2011, 7, e1002093. [Google Scholar] [CrossRef] [PubMed]
- Hawkins, R.D.; Hon, G.C.; Lee, L.K.; Ngo, Q.; Lister, R.; Pelizzola, M.; Edsall, L.E.; Kuan, S.; Luu, Y.; Klugman, S.; et al. Distinct epigenomic landscapes of pluripotent and lineage-committed human cells. Cell Stem Cell 2010, 6, 479–491. [Google Scholar] [CrossRef] [PubMed]
- Onyshchenko, M.I.; Panyutin, I.G.; Panyutin, I.V.; Neumann, R.D. Stimulation of cultured H9 human embryonic stem cells with thyroid stimulating hormone does not lead to formation of thyroid-like cells. Stem Cells Int. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Lai, P.P.; Taylor, Y.C. Differential radioprotection of cultured human diploid fibroblasts and fibrosarcoma cells by WR1065. Int. J. Radiat. Oncol. Biol. Phys. 1992, 24, 713–719. [Google Scholar] [CrossRef]
- Olive, P.L.; Banath, J.P.; Durand, R.E. Heterogeneity in radiation-induced DNA damage and repair in tumor and normal cells measured using the “comet” assay. Radiat. Res. 1990, 122, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Olive, P.L.; Banath, J.P. The comet assay: A method to measure DNA damage in individual cells. Nat. Protoc. 2006, 1, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Harfouche, G.; Martin, M.T. Response of normal stem cells to ionizing radiation: A balance between homeostasis and genomic stability. Mutat. Res. 2010, 704, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Gladstone, M.; Su, T.T. Radiation responses and resistance. Int. Rev. Cell Mol. Biol. 2012, 299, 235–253. [Google Scholar] [PubMed]
- Schuster-Bockler, B.; Lehner, B. Chromatin organization is a major influence on regional mutation rates in human cancer cells. Nature 2012, 488, 504–507. [Google Scholar] [CrossRef] [PubMed]
- Rea, S.; Eisenhaber, F.; O’Carroll, D.; Strahl, B.D.; Sun, Z.W.; Schmid, M.; Opravil, S.; Mechtler, K.; Ponting, C.P.; Allis, C.D.; et al. Regulation of chromatin structure by site-specific histone H3 methyltransferases. Nature 2000, 406, 593–599. [Google Scholar] [PubMed]
- Peters, A.H.; O’Carroll, D.; Scherthan, H.; Mechtler, K.; Sauer, S.; Schofer, C.; Weipoltshammer, K.; Pagani, M.; Lachner, M.; Kohlmaier, A.; et al. Loss of the Suv39h histone methyltransferases impairs mammalian heterochromatin and genome stability. Cell 2001, 107, 323–337. [Google Scholar] [CrossRef]
- Goodarzi, A.A.; Jeggo, P.; Lobrich, M. The influence of heterochromatin on DNA double strand break repair: Getting the strong, silent type to relax. DNA Repair 2010, 9, 1273–1282. [Google Scholar] [CrossRef] [PubMed]
- Jakob, B.; Splinter, J.; Conrad, S.; Voss, K.O.; Zink, D.; Durante, M.; Lobrich, M.; Taucher-Scholz, G. DNA double-strand breaks in heterochromatin elicit fast repair protein recruitment, histone H2AX phosphorylation and relocation to euchromatin. Nucleic Acids Res. 2011, 39, 6489–6499. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, C.; Hayward, R.L.; Gilbert, N. Global chromatin fibre compaction in response to DNA damage. Biochem. Biophys. Res. Commun. 2011, 414, 820–825. [Google Scholar] [CrossRef] [PubMed]
- Tjeertes, J.V.; Miller, K.M.; Jackson, S.P. Screen for DNA-damage-responsive histone modifications identifies H3K9Ac and H3K56Ac in human cells. EMBO J. 2009, 28, 1878–1889. [Google Scholar] [CrossRef] [PubMed]
- Loh, Y.H.; Zhang, W.; Chen, X.; George, J.; Ng, H.H. Jmjd1a and Jmjd2c histone H3 Lys 9 demethylases regulate self-renewal in embryonic stem cells. Genes Dev. 2007, 21, 2545–2557. [Google Scholar] [CrossRef] [PubMed]
- Rebuzzini, P.; Pignalosa, D.; Mazzini, G.; di Liberto, R.; Coppola, A.; Terranova, N.; Magni, P.; Redi, C.A.; Zuccotti, M.; Garagna, S. Mouse embryonic stem cells that survive γ-rays exposure maintain pluripotent differentiation potential and genome stability. J. Cell. Physiol. 2012, 227, 1242–1249. [Google Scholar] [CrossRef] [PubMed]
- Sokolov, M.V.; Panyutin, I.V.; Onyshchenko, M.I.; Panyutin, I.G.; Neumann, R.D. Expression of pluripotency-associated genes in the surviving fraction of cultured human embryonic stem cells is not significantly affected by ionizing radiation. Gene 2010, 455, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Wilson, K.D.; Sun, N.; Huang, M.; Zhang, W.Y.; Lee, A.S.; Li, Z.; Wang, S.X.; Wu, J.C. Effects of ionizing radiation on self-renewal and pluripotency of human embryonic stem cells. Cancer Res. 2010, 70, 5539–5548. [Google Scholar] [CrossRef] [PubMed]
- Ayrapetov, M.K.; Gursoy-Yuzugullu, O.; Xu, C.; Xu, Y.; Price, B.D. DNA double-strand breaks promote methylation of histone H3 on Lysine 9 and transient formation of repressive chromatin. Proc. Natl. Acad. Sci. USA 2014, 111, 9169–9174. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Venkatesh, P.; Panyutin, I.V.; Remeeva, E.; Neumann, R.D.; Panyutin, I.G. Effect of Chromatin Structure on the Extent and Distribution of DNA Double Strand Breaks Produced by Ionizing Radiation; Comparative Study of hESC and Differentiated Cells Lines. Int. J. Mol. Sci. 2016, 17, 58. https://doi.org/10.3390/ijms17010058
Venkatesh P, Panyutin IV, Remeeva E, Neumann RD, Panyutin IG. Effect of Chromatin Structure on the Extent and Distribution of DNA Double Strand Breaks Produced by Ionizing Radiation; Comparative Study of hESC and Differentiated Cells Lines. International Journal of Molecular Sciences. 2016; 17(1):58. https://doi.org/10.3390/ijms17010058
Chicago/Turabian StyleVenkatesh, Priyanka, Irina V. Panyutin, Evgenia Remeeva, Ronald D. Neumann, and Igor G. Panyutin. 2016. "Effect of Chromatin Structure on the Extent and Distribution of DNA Double Strand Breaks Produced by Ionizing Radiation; Comparative Study of hESC and Differentiated Cells Lines" International Journal of Molecular Sciences 17, no. 1: 58. https://doi.org/10.3390/ijms17010058
APA StyleVenkatesh, P., Panyutin, I. V., Remeeva, E., Neumann, R. D., & Panyutin, I. G. (2016). Effect of Chromatin Structure on the Extent and Distribution of DNA Double Strand Breaks Produced by Ionizing Radiation; Comparative Study of hESC and Differentiated Cells Lines. International Journal of Molecular Sciences, 17(1), 58. https://doi.org/10.3390/ijms17010058





