Sulfasalazine Treatment Suppresses the Formation of HLA-B27 Heavy Chain Homodimer in Patients with Ankylosing Spondylitis
Abstract
:1. Introduction
2. Results
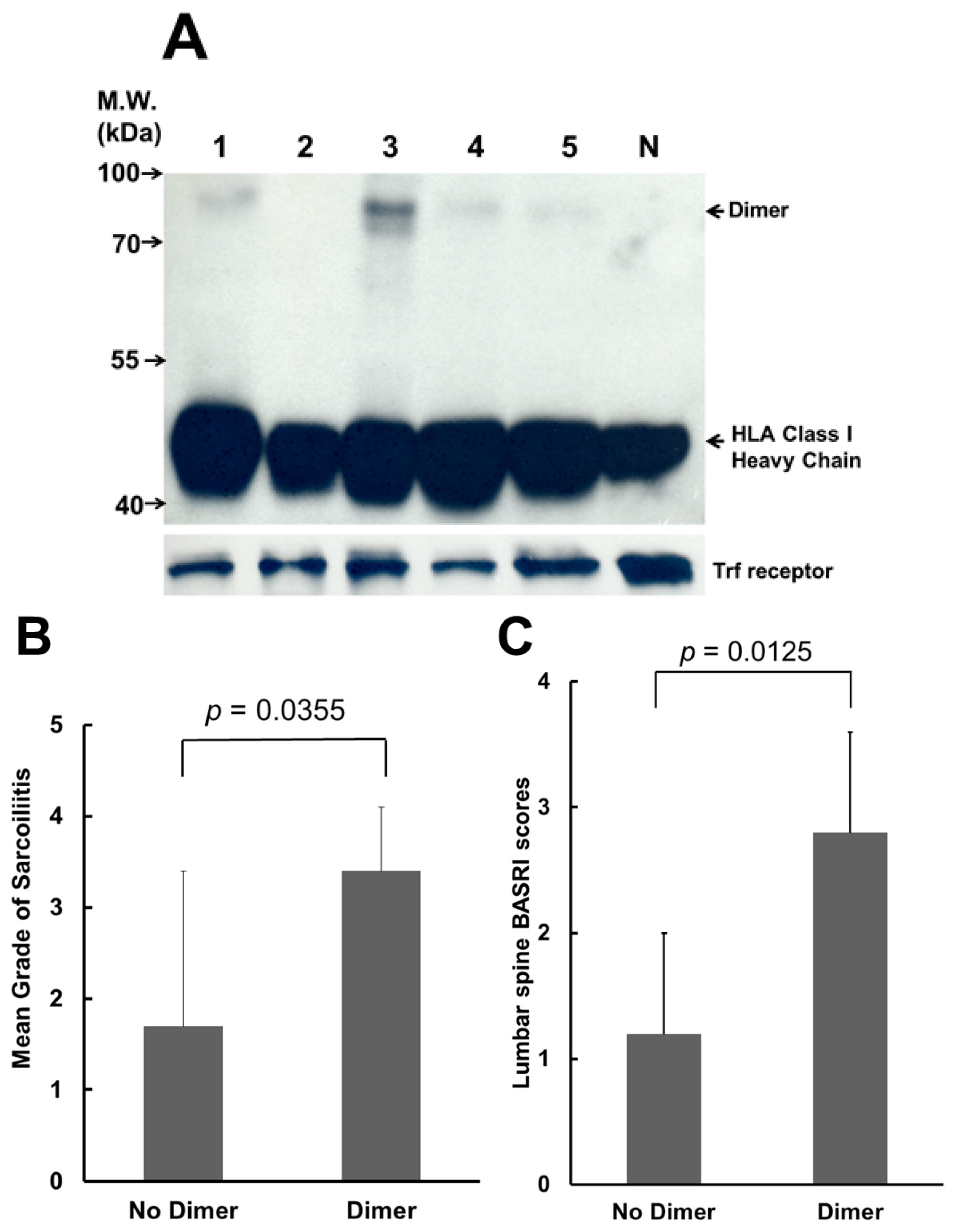
2.1. Most of Ankylosing Spondylitis (AS) Patients Display (B27-Heavy Chain (HC))2 on Their Peripheral Blood Mononuclear Cells (PBMCs), and the Mean Grades of Sacroiliitis and the Lumbar Spine Bath Ankylosing Spondylitis Radiology Index (BASRI) Scores Are Lower in AS Patients without (B27-HC)2 on Their PBMCs
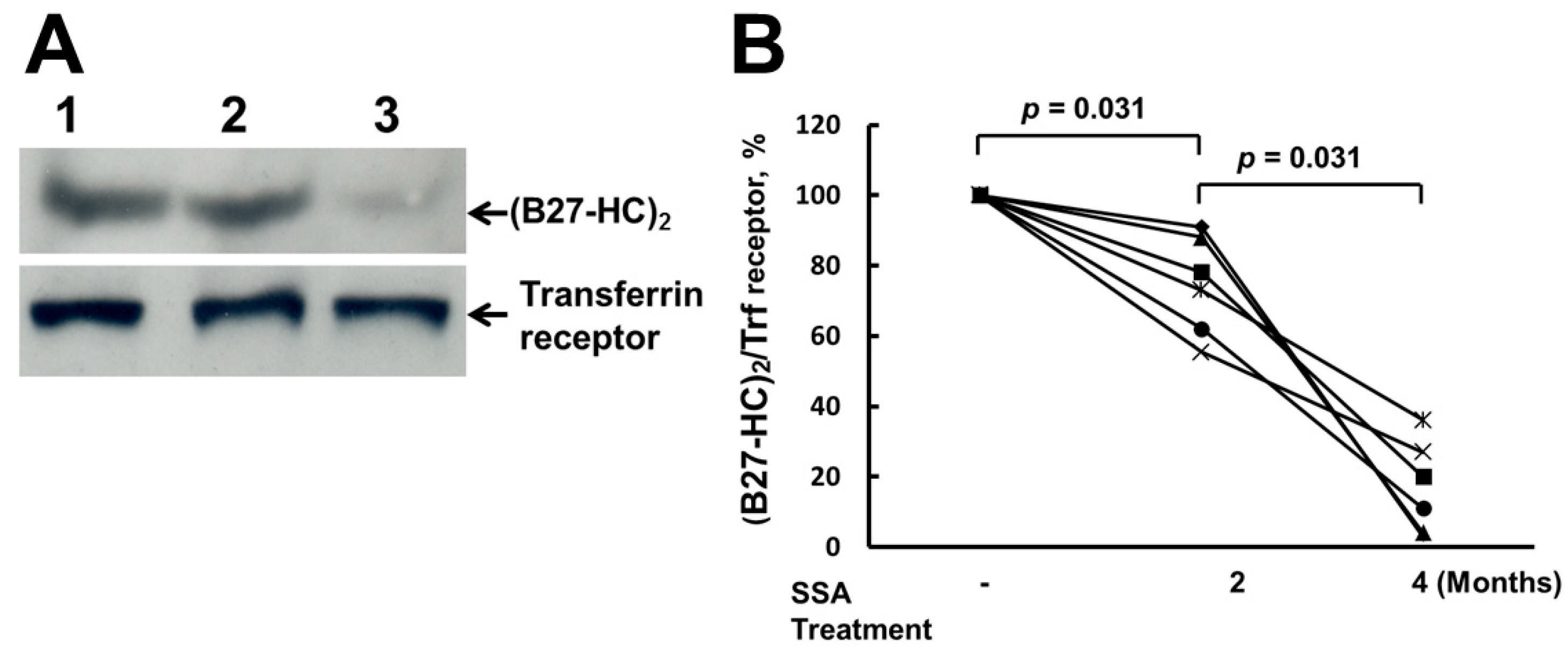
2.2. Sulfasalazine (SSA) Treatment Suppressed the Production of (B27-HC)2
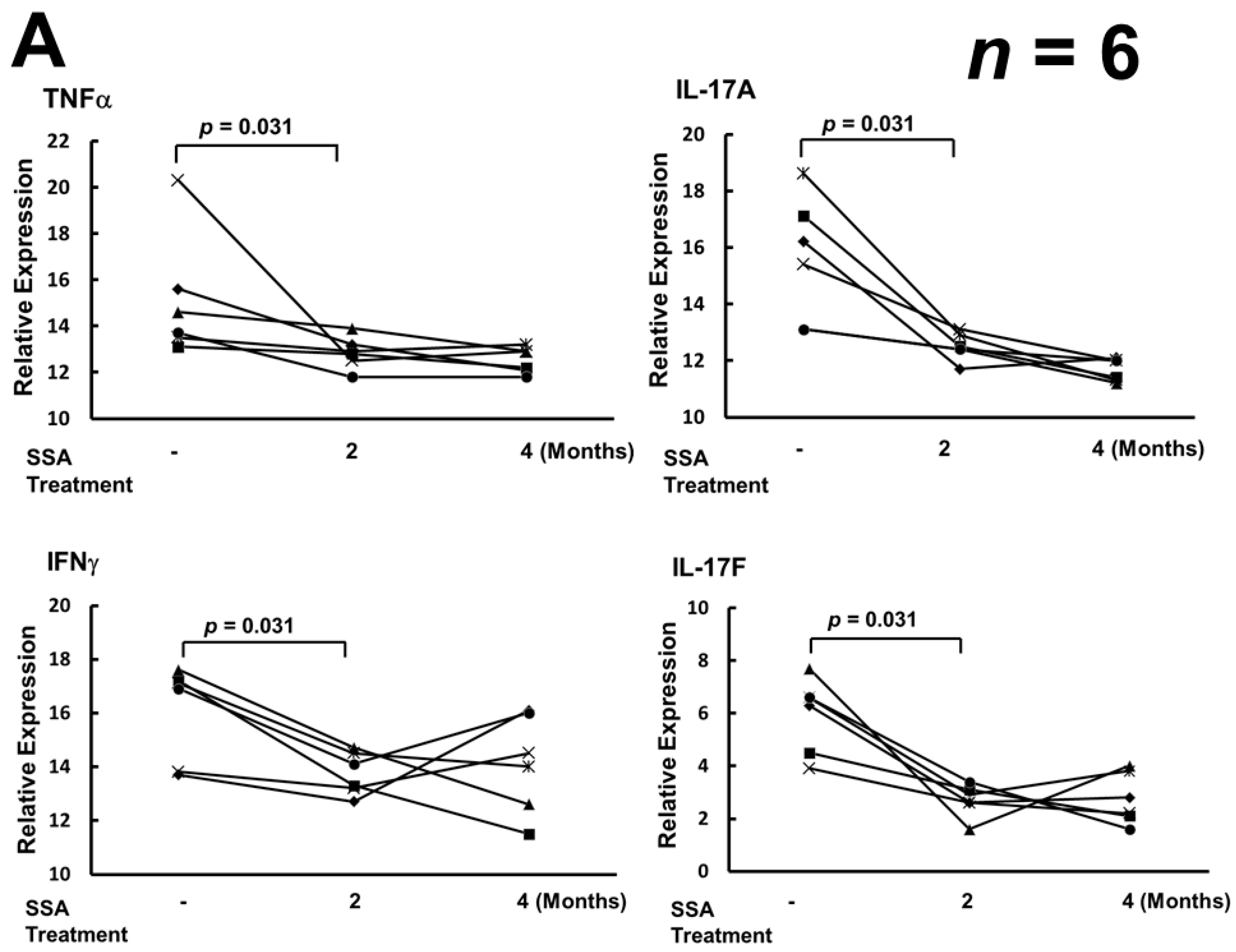
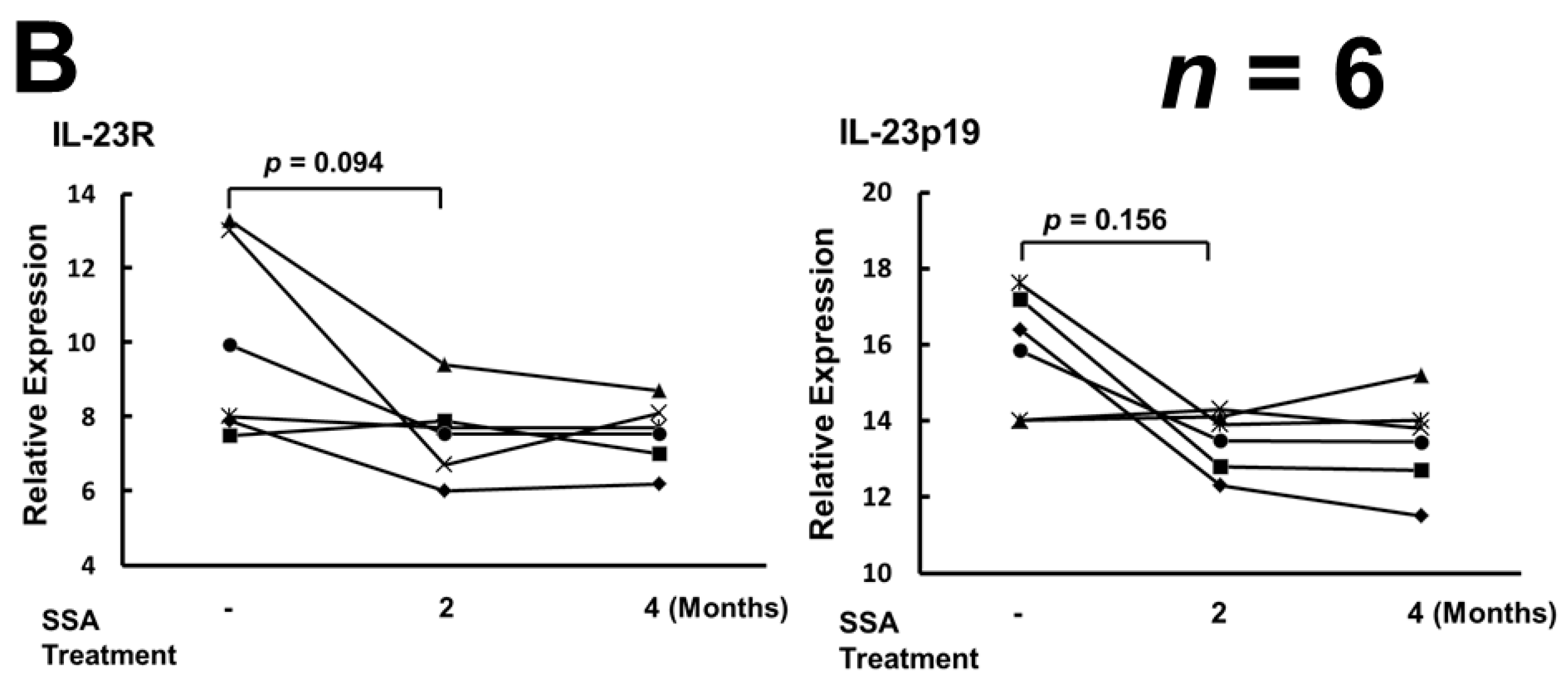
2.3. SSA Repressed the Expression of TNFα, IL-17A, IL-17F, and IFNγ mRNA, but Did Not Affect the Levels of mRNA of IL-23 and IL-23R
3. Discussion
4. Experimental Section
4.1. Materials
4.2. Patients
4.3. Western Blot Analysis
4.4. Analysis of Grade of Sacroiliitis and BASRI Scores
4.5. Real-Time PCR
4.6. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Colbert, R.A.; DeLay, M.L.; Klenk, E.I.; Layh-Schmitt, G. From HLA-B27 to spondyloarthritis: A journey through the ER. Immunol. Rev. 2010, 233, 181–202. [Google Scholar] [CrossRef] [PubMed]
- Melis, L; Elewaut, D. Immunopathogenesis of spondykoarthritis: Which cells drive disease? Arthritis Res. Ther. 2009, 11, 233–238. [Google Scholar]
- Smith, J.A.; Marker-Hermann, E.; Colbert, R.A. Pathogenesis of ankylosing spondylitis: Current concepts. Best Pract. Res. Clin. Rheumatol. 2006, 20, 571–591. [Google Scholar] [CrossRef] [PubMed]
- Reveille, J.D.; Arnett, F.C. Spondyloarthitis: Update on pathogenesis and management. Am. J. Med. 2005, 118, 592–603. [Google Scholar] [CrossRef] [PubMed]
- Caffrey, M.F.; James, D.C. Human lymphocyte antigen association in ankylosing spondylitis. Nature 1973, 242. [Google Scholar] [CrossRef]
- Brewerton, D.A.; Hart, F.D.; Nicholls, A.; Caffrey, M.; James, D.C.; Sturrock, R.D. Ankylosing spondylitis and HL-A 27. Lancet 1973, 1, 904–907. [Google Scholar] [CrossRef]
- Lopez-Larrea, C.; González, S.; Borra, J.M. The role of HLA-B27 polymorphism and molecular mimicry in spondyloarthropathy. Mol. Med. Today 1998, 4, 540–549. [Google Scholar] [CrossRef]
- Taurog, J.D.; Maika, S.D.; Satumtira, N.; Dorris, M.L.; McLean, I.L.; Yanagisawa, H.; Sayad, A.; Stagg, A.J.; Fox, G.M.; le O’Brein, A.; et al. Inflammatory disease in HLA-B27 transgenic rats. Immunol. Rev. 1999, 169, 209–223. [Google Scholar] [PubMed]
- May, E.; Dorris, M.L.; Satumtira, N.; Iqbal, I.; Rehman, M.I.; Lightfoot, E.; Taurog, J.D. CD8αβ T cells are not essential to the pathogenesis of arthritis or colitis in HLA-B27 transgenic rats. J. Immunol. 2003, 170, 1099–1105. [Google Scholar] [CrossRef] [PubMed]
- Taurog, J.D.; Dorris, M.L.; Satumtira, N.; Tran, T.M.; Sharma, R.; Dressel, R.; van den Brandt, J.; Reichardt, H.M. Spondylarthritis in HLA-B27/human β2-microglobulin-transgenic rats is not prevented by lack of CD8. Arthritis Rheumatol. 2009, 60, 1977–1984. [Google Scholar] [CrossRef] [PubMed]
- Allen, R.L.; O’Callaghan, C.A.; McMichael, A.J.; Bowness, P. HLA-B27 can form a novel β2-microglobulin-free heavy chain homodimer structure. J. Immunol. 1999, 162, 5045–5048. [Google Scholar] [PubMed]
- Kollnberger, S.; Bird, L.; Sun, M.Y.; Retiere, C.; Braud, V.M.; McMichael, A.; Bowness, P. Cell-surface expression and immune receptor recognition of HLA-B27 homodimers. Arthritis Rheumatol. 2002, 46, 2972–2982. [Google Scholar] [CrossRef] [PubMed]
- Dangoria, N.; DeLay, M.L.; Kingsbury, D.J.; Mear, J.P.; Uchanska-Ziegler, B.; Ziegler, A.; Colbert, R.A. HLA-B27 misfolding is associated with aberrant intermolecular disulfide bond formation (dimerization) in the endoplasmic reticulum. J. Biol. Chem. 2002, 277, 23459–23468. [Google Scholar] [CrossRef] [PubMed]
- Kollnberger, S.; Bird, L.A.; Roddis, M.; Hacquard-Bouder, C.; Kubagawa, H.; Bodmer, H.C.; Breban, M.; McMichael, A.J.; Bowness, P. HLA-B27 heavy chain homodimers are expressed in HLA-B27 transgenic rodent models of spondyloarthritis and are ligands for paired Ig-like receptors. J. Immunol. 2004, 173, 1699–1710. [Google Scholar] [CrossRef] [PubMed]
- Chan, A.T.; Kollnberger, S.D.; Wedderburn, L.R.; Bowness, P. Expansion and enchanced survival of natural killer cells expressing the killer immunoglobulin-like receptor KIR3DL2 inspondylarthritis. Arthritis Rheumatol. 2005, 52, 3586–3595. [Google Scholar] [CrossRef] [PubMed]
- Bowness, P.; Ridley, A.; Shaw, J.; Chan, A.T.; Wong-Baeza, I.; Fleming, M.; Cummings, F.; McMichael, A.; Kollnberger, S. Th17 cells expressing KIR3DL2+ and responsive to HLA-B27 homodimers are increased in ankylosing spondylitis. J. Immunol. 2011, 186, 2672–2680. [Google Scholar] [CrossRef] [PubMed]
- Hammer, R.E.; Maika, S.D.; Richardson, J.A.; Tang, J.P.; Taurog, J.D. Spontaneous inflammatory disease in transgenic rat expressing HLA-B27 and human β2m: An animal model of HLA-B27-associated human disorders. Cell 1990, 63, 1099–1112. [Google Scholar] [CrossRef]
- Pahl, H.L. Signal transduction from the endoplasmic reticulum to the cell nucleus. Physiol. Rev. 1999, 79, 683–701. [Google Scholar] [PubMed]
- Hotamisligil, G.S. Endoplasmic reticulum stress and the inflammatory basis of metabolic disease. Cell 2010, 140, 900–917. [Google Scholar] [CrossRef] [PubMed]
- Turner, M.J.; Sowder, D.P.; DeLay, M.L.; Mohapatra, R.; Bai, S.; Smith, J.A.; Brandewie, J.R.; Taurog, J.D.; Colbert, R.A. HLA-B27 misfolding in transgenic rats is associated with activation of the unfolded protein response. J. Immunol. 2005, 175, 2438–2448. [Google Scholar] [CrossRef] [PubMed]
- Turner, M.J.; DeLay, M.L.; Bai, S.; Klenk, E.; Colbert, R.A. HLA-B27 up-regulation causes accumulation of misfolded heavy chains and correlated with the magnitude of the unfolded protein response in transgenic rats. Arthritis Rheumatol. 2007, 56, 215–223. [Google Scholar] [CrossRef] [PubMed]
- DeLay, M.L.; Turner, M.J.; Klenk, E.I.; Smith, J.A.; Sowders, D.P.; Colbert, R.A. HLA-B27 misfolding and the unfolded protein response augment interleukin-23 production and are associated with Th17 activation in transgenic rats. Arthritis Rheumatol. 2009, 60, 2633–2643. [Google Scholar] [CrossRef] [PubMed]
- Wahl, C.; Liptay, S.; Adler, G.; Schmid, R.M. Sulfasalazine: A potent and specific inhibitor of nuclear factor κ B. J. Clin. Investig. 1998, 101, 1163–1174. [Google Scholar] [CrossRef] [PubMed]
- Weber, C.K.; Liptay, S.; Wirth, T.; Adler, G.; Schmid, R.M. Suppression of NF-κB activity by sulfasalazine is mediator by direct inhibition of IκB kinases α and β. Gastroenterology 2000, 119, 1209–1218. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.C.; Lu, M.C.; Li, C.; Huang, H.L.; Huang, K.Y.; Liu, S.Q.; Lai, N.S.; Huang, H.B. Targeted delivery of an antigenic peptide to the endoplasmic reticulum: Application for development of a peptide therapy for ankylosing spondylitis. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.C.; Huang, K.Y.; Lu, M.C.; Huang, H.L.; Liu, W.T.; Lee, W.C.; Liu, S.Q.; Huang, H.B.; Lai, N.S. Characterization of the recognition specificity of BH2, a monoclonal antibody prepared against the HLA-B27 heavy chain. Int. J. Mol. Sci. 2015, 16, 8142–8150. [Google Scholar] [CrossRef] [PubMed]
- Zeng, L.; Lindstrom, M.J.; Smith, J.A. Ankylosing spondylitis macrophage production of higher levels of interleukin-23 in response to lipopolysaccharide without induction of a significant unfolded protein response. Arthritis Rheumatol. 2011, 63, 3807–3817. [Google Scholar] [CrossRef] [PubMed]
- Olivieri, I.; van Tubergen, A.; Salvarani, C.; van der Linden, S. Seronegative pondyloarthritides. Best Pract. Res. Clin. Rheumatol. 2002, 16, 723–739. [Google Scholar] [CrossRef] [PubMed]
- Zochling, J.; Brandt, J.; Braun, J. The current concept of spondyloarthritis with special emphasis on undifferentiated spondyloarthritis. Rheumatology 2005, 44, 1483–1491. [Google Scholar] [CrossRef] [PubMed]
- Campbell, E.C.; Antoniu, A.N.; Powis, S.J. The multi-faceted nature of HLA class I dimer molecules. Immunology 2012, 136, 380–384. [Google Scholar] [CrossRef] [PubMed]
- Lynch, S.; Santos, S.G.; Campbell, E.C.; Nimmo, A.M.; Botting, C.; Prescott, A.; Antoniou, A.N.; Powis, S.J. Novel MHC class I structures on exosomes. J. Immunol. 2009, 183, 1884–1891. [Google Scholar] [CrossRef] [PubMed]
- Van der Linden, S.; Valkenburg, H.A.; Cats, A. Evaluation of diagnosis criteria for ankylosing spondylitis. A proposal for modification of the New York criteria. Arthritis Rheumatol. 1984, 27, 361–368. [Google Scholar] [CrossRef]
- Lu, M.C.; Lai, N.S.; Yu, H.C.; Hsieh, S.C.; Tung, C.H.; Yu, C.L. Nifedipine suppresses Th1/Th2 cytokine production and increased apoptosis of anti-CD3+ anti-CD28-activated mononuclear cells from patients with systemic lupus erythematosus via calcineurin pathway. Clin. Immunol. 2008, 129, 462–470. [Google Scholar] [CrossRef] [PubMed]
- Moll, J.M.; Wright, V. New York clinical criteria for ankylosing spondylitis. A statistical evaluation. Ann. Rheumatol. Dis. 1973, 32, 354–363. [Google Scholar] [CrossRef]
- MacKay, K.; Mack, C.; Brophy, S.; Calin, A. The Bath Ankylosing Spondylitis Radiology Index (BASRI): A new, validated approach to disease assessment. Arthritis Rheumatol. 1998, 41, 2263–2270. [Google Scholar] [CrossRef]
- Bechara, F.G.; Skrygan, M.; Kreuter, A.; Altmeyer, P.; Gambichler, T. Cytokine mRNA levels in human fat tissue after injection lipolysis with phosphatidylcholine and deoxycholate. Arch. Dermatol. Res. 2008, 300, 455–459. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Chu, Y.; Yang, X.; Gao, D.; Zhu, L.; Yang, X.; Wan, L.; Li, M. Th17 and natural Treg cell population dynamics in systemic lupus erythematosus. Arthritis Rheumatol. 2009, 60, 1472–1483. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.R.; Cho, M.L.; Kim, W.; Juhn, J.Y.; Hwang, S.Y.; Yoon, C.H.; Park, S.H.; Lee, S.H.; Kim, H.Y. Up-regulation of IL-23p19 expression in rheumatoid arthritis synovial fibroblasts by IL-17 through PI3-kinase-, NF-κB- and p38 MAPK-dependent signalling pathways. Rheumatology 2007, 46, 57–64. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, H.-C.; Lu, M.-C.; Huang, K.-Y.; Huang, H.-l.; Liu, S.-Q.; Huang, H.-B.; Lai, N.-S. Sulfasalazine Treatment Suppresses the Formation of HLA-B27 Heavy Chain Homodimer in Patients with Ankylosing Spondylitis. Int. J. Mol. Sci. 2016, 17, 46. https://doi.org/10.3390/ijms17010046
Yu H-C, Lu M-C, Huang K-Y, Huang H-l, Liu S-Q, Huang H-B, Lai N-S. Sulfasalazine Treatment Suppresses the Formation of HLA-B27 Heavy Chain Homodimer in Patients with Ankylosing Spondylitis. International Journal of Molecular Sciences. 2016; 17(1):46. https://doi.org/10.3390/ijms17010046
Chicago/Turabian StyleYu, Hui-Chun, Ming-Chi Lu, Kuang-Yung Huang, Hsien-lu Huang, Su-Qin Liu, Hsien-Bin Huang, and Ning-Sheng Lai. 2016. "Sulfasalazine Treatment Suppresses the Formation of HLA-B27 Heavy Chain Homodimer in Patients with Ankylosing Spondylitis" International Journal of Molecular Sciences 17, no. 1: 46. https://doi.org/10.3390/ijms17010046
APA StyleYu, H.-C., Lu, M.-C., Huang, K.-Y., Huang, H.-l., Liu, S.-Q., Huang, H.-B., & Lai, N.-S. (2016). Sulfasalazine Treatment Suppresses the Formation of HLA-B27 Heavy Chain Homodimer in Patients with Ankylosing Spondylitis. International Journal of Molecular Sciences, 17(1), 46. https://doi.org/10.3390/ijms17010046





