RUCAM in Drug and Herb Induced Liver Injury: The Update
Abstract
1. Introduction
2. Data Sources and Searches
2.1. Search Terms
2.2. Data Extraction
3. A General View Back to the Original RUCAM
3.1. Nomenclature of RUCAM
| Operational Information on the Updated RUCAM | Ref. |
|---|---|
| 1. RUCAM affords prospective use, since retrospective scoring is less accurate. | [8,15] |
| 2. RUCAM is to be calculated individually for each co-administered product. | [8,15] |
| 3. RUCAM is conceptualized primarily for idiosyncratic, not for intrinsic reactions. | [8] |
| 4. RUCAM excludes cases with onset of hepatic injury before start of product use. | [8] |
| 5. RUCAM is applicable only for acute liver injury, not for preexisting chronic liver disease. | [8] |
| 6. RUCAM cannot correctly assess when ALP is elevated for non-hepatic reasons. | [8] |
| Items for Hepatocellular Injury | Score | Result |
|---|---|---|
| 1. Time to onset from the beginning of the drug/herb | ||
| ● 5–90 days (rechallenge: 1–15 days) | +2 | □ |
| ● <5 or >90 days (rechallenge: >15 days) | +1 | □ |
| Alternative: Time to onset from cessation of the drug/herb | ||
| ● ≤15 days (except for slowly metabolized chemicals: >15 days) | +1 | □ |
| 2. Course of ALT after cessation of the drug/herb | ||
| Percentage difference between ALT peak and N | ||
| ● Decrease ≥ 50% within 8 days | +3 | □ |
| ● Decrease ≥ 50% within 30 days | +2 | □ |
| ● No information or continued drug use | 0 | □ |
| ● Decrease ≥ 50% after the 30th day | 0 | □ |
| ● Decrease < 50% after the 30th day or recurrent increase | −2 | □ |
| 3. Risk factors | ||
| ● Alcohol use (current drinks/d: >2 for women, >3 for men) | +1 | □ |
| ● Alcohol use (current drinks/d: ≤2 for women, ≤3 for men) | 0 | □ |
| ● Age ≥ 55 years | +1 | □ |
| ● Age < 55 years | 0 | □ |
| 4. Concomitant drug(s)/herb(s) | ||
| ● None or no information | 0 | □ |
| ● Concomitant drug/herb with incompatible time to onset | 0 | □ |
| ● Concomitant drug/herb with compatible or suggestive time to onset | −1 | □ |
| ● Concomitant drug/herb known as hepatotoxin and with compatible or suggestive time to onset delete marking right side above | −2 | □ |
| ● Concomitant drug/herb with evidence for its role in this case (positive rechallenge or validated test) | −3 | □ |
| 5. Search for alternative causes | Tick if negative | Tick if not done |
| Group I (7 causes) | ||
| ● HAV: Anti-HAV-IgM | □ | □ |
| ● Hepatobiliary sonography / colour Doppler | □ | □ |
| ● HCV: Anti-HCV, HCV-RNA | □ | □ |
| ● HEV: Anti-HEV-IgM, anti-HEV-IgG, HEV-RNA | □ | □ |
| ● Hepatobiliary sonography/colour Doppler sonography of liver vessels/ endosonography/CT/MRC | □ | □ |
| ● Alcoholism (AST/ALT ≥ 2) | □ | □ |
| ● Acute recent hypotension history (particularly if underlying heart disease) | □ | □ |
| Group II (5 causes) | ||
| ● Complications of underlying disease(s) such as sepsis, metastatic malignancy, autoimmune hepatitis, chronic hepatitis B or C, primary biliary cholangitis or sclerosing cholangitis, genetic liver diseases | □ | □ |
| ● Infection suggested by PCR and titer change for | ||
| ● CMV (anti-CMV-IgM, anti-CMV-IgG) | □ | □ |
| ● EBV (anti-EBV-IgM, anti-EBV-IgG) | □ | □ |
| ● HSV (anti-HSV-IgM, anti-HSV-IgG) | □ | □ |
| ● VZV (anti-VZV-IgM, anti-VZV-IgG) | □ | □ |
| Evaluation of groups I and II | ||
| ● All causes-groups I and II—reasonably ruled out | +2 | □ |
| ● The 7 causes of group I ruled out | +1 | □ |
| ● 6 or 5 causes of group I ruled out | 0 | □ |
| ● Less than 5 causes of group I ruled out | -2 | □ |
| ● Alternative cause highly probable | -3 | □ |
| 6. Previous hepatotoxicity of the drug/herb | ||
| ● Reaction labelled in the product characteristics | +2 | □ |
| ● Reaction published but unlabelled | +1 | □ |
| ● Reaction unknown | 0 | □ |
| 7. Response to unintentional reexposure | ||
| ● Doubling of ALT with the drug/herb alone, provided ALT below 5N before reexposure | +3 | □ |
| ● Doubling of ALT with the drug(s)/herb(s) already given at the time of first reaction | +1 | □ |
| ● Increase of ALT but less than N in the same conditions as for the first administration | −2 | □ |
| ● Other situations | 0 | □ |
| Total score for the case | □ | |
| Items for Cholestatic or Mixed Liver Injury | Score | Result |
|---|---|---|
| 1. Time to onset from the beginning of the drug/herb | ||
| ● 5–90 days (rechallenge: 1–90 days) | +2 | □ |
| ● <5 or >90 days (rechallenge: >90 days) | +1 | □ |
| Alternative: Time to onset from cessation of the drug/herb | ||
| ● (except for slowly metabolized chemicals: ≤30 days) | +1 | □ |
| 2. Course of ALP after cessation of the drug/herb | ||
| Percentage difference between ALP peak and N | ||
| ● Decrease ≥ 50% within 180 days | +2 | □ |
| ● Decrease < 50% within 180 days | +1 | □ |
| ● No information, persistence, increase, or continued drug/herb use | 0 | □ |
| 3. Risk factors | ||
| ● Alcohol use current drinks/d: >2 for women, >3 for men) | +1 | □ |
| ● Alcohol use (current drinks/d: ≤2 for women, ≤3 for men) | 0 | □ |
| ● Pregnancy | +1 | □ |
| ● Age ≥ 55 years | +1 | □ |
| ● Age < 55 years | 0 | □ |
| 4. Concomitant use of drug(s)/herb(s) | ||
| ● None or no information | 0 | □ |
| ● Concomitant drug/herb with incompatible time to onset | 0 | □ |
| ● Concomitant drug/herb with compatible or suggestive time to onset | −1 | □ |
| ● Concomitant drug/herb known as hepatotoxin and with compatible or suggestive time to onset | −2 | □ |
| ● Concomitant drug/herb with evidence for its role in this case (positive rechallenge or validated test) | −3 | □ |
| 5. Search for alternative causes | Tick if negative | Tick if not done |
| Group I (7 causes) | ||
| ● HAV: Anti-HAV-IgM | □ | □ |
| ● HBV: HBsAg, anti-HBc-IgM, HBV-DNA | □ | □ |
| ● HCV: Anti-HCV, HCV-RNA | □ | □ |
| ● HEV: Anti-HEV-IgM, anti-HEV-IgG, HEV-RNA | □ | □ |
| ● Hepatobiliary sonography/colour Doppler sonography of liver vessels/endosonography/CT/MRC | □ | □ |
| ● Alcoholism (AST/ ALT ≥ 2) | □ | □ |
| ● Acute recent hypotension history (particularly if underlying heart disease) | □ | □ |
| Group II (5 causes) | ||
| ● Complications of underlying disease(s) such as sepsis, metastatic malignancy, autoimmune hepatitis, chronic hepatitis B or C, primary biliary cholangitis or sclerosing cholangitis, genetic liver diseases | □ | □ |
| ● Infection suggested by PCR and titer change for | ||
| ● CMV (anti-CMV-IgM, anti-CMV-IgG) | □ | □ |
| ● EBV (anti-EBV-IgM, anti-EBV-IgG) | □ | □ |
| ● HSV (anti-HSV-IgM, anti-HSV-IgG) | □ | □ |
| ● VZV (anti-VZV-IgM, anti-VZV-IgG) | □ | □ |
| Evaluation of group I and II | ||
| ● All causes—groups I and II—reasonably ruled out | +2 | □ |
| ● The 7 causes of group I ruled out | +1 | □ |
| ● 6 or 5 causes of group I ruled out | 0 | □ |
| ● Less than 5 causes of group I ruled out | −2 | □ |
| ● Alternative cause highly probable | −3 | □ |
| 6. Previous hepatotoxicity of the drug/herb | ||
| ● Reaction labelled in the product characteristics | +2 | □ |
| ● Reaction published but unlabelled | +1 | □ |
| ● Reaction unknown | 0 | □ |
| 7. Response to unintentional reexposure | ||
| ● Doubling of ALP with the drug/herb alone, provided ALP below 2N before reexposure | +3 | □ |
| ● Doubling of ALP with the drugs(s)/herbs(s) already given at the time of first reaction | +1 | □ |
| ● Increase of ALP but less than N in the same conditions as for the first administration | −2 | □ |
| ● Other situations | 0 | □ |
| Total score for the case | □ | |
3.2. Precursor Versions
4. Original RUCAM
4.1. Consensus Meetings
4.2. Case Definition
4.3. Liver Injury Classification
4.4. Elements with Individual Scoring
4.5. Sensitivity, Specificity, and Predictive Values
4.6. Shortcomings
5. Updated RUCAM
| Differential Diagnosis | Diagnostic Parameters | Diagnostic Exclusion Done for Patient’s Assessment | ||
|---|---|---|---|---|
| Yes | No | Partial | ||
| ● Hepatitis A virus (HAV) | Anti-HAV-IgM | □ | □ | □ |
| ● Hepatitis B virus (HBV) | HBV-DNA, anti-HBc-IgM | □ | □ | □ |
| ● Hepatitis C virus (HCV) | HCV-RNA, anti-HCV | □ | □ | □ |
| ● Hepatitis E virus (HEV) | HEV-RNA, titer change for anti-HEV-IgM/anti-HEV-IgG | □ | □ | □ |
| ● Cytomegalovirus (CMV) | CMV-PCR, titer change for anti-CMV-IgM/anti-CMV-IgG | □ | □ | □ |
| ● Epstein Barr virus (EBV) | EBV-PCR, titer change for anti-EBV-IgM/anti-EBV-IgG | □ | □ | □ |
| ● Herpes simplex virus (HSV) | HSV-PCR, titer change for anti-HSV-IgM/anti-HSV-IgG | □ | □ | □ |
| ● Varicella zoster virus (VZV) | VZV-PCR, titer change for anti-VZV-IgM/anti-VZV-IgG | □ | □ | □ |
| ● Other virus infections | Specific serology of Adenovirus, Coxsackie-B-Virus, Echovirus, Measles virus, Rubella virus, Flavivirus, Arenavirus, Filovirus, Parvovirus, HIV, and others | □ | □ | □ |
| ● Other infectious diseases | Specific assessment of bacteria, fungi, parasites, worms, and others | □ | □ | □ |
| ● Autoimmune hepatitis (AIH) type I | Gamma globulins, ANA, SMA, AAA, SLA/LP, Anti-LSP, Anti-ASGPR | □ | □ | □ |
| ● Autoimmune hepatitis (AIH) type II | Gamma globulins, Anti-LKM-1 (CYP 2D6), Anti-LKM-2 (CYP 2C9), Anti-LKM-3 | □ | □ | □ |
| ● Primary biliary cholangitis (PBC) | AMA, Anti PDH-E2 | □ | □ | □ |
| ● Primary sclerosing cholangitis (PSC) | p-ANCA, MRC | □ | □ | □ |
| ● Autoimmune cholangitis (AIC) | ANA, SMA | □ | □ | □ |
| ● Overlap syndromes | See AIH, PBC, PSC, and AIC | □ | □ | □ |
| ● Non alcoholic steatohepatitis (NASH) | BMI, insulin resistance, hepatomegaly, echogenicity of the liver | □ | □ | □ |
| ● Alcoholic liver disease (ALD) | Patient’s history, clinical and laboratory assessment, other alcoholic disease(s) | □ | □ | □ |
| ● Drug induced liver injury (DILI) or herb induced liver injury (HILI) | Patient’s history, clinical and laboratory assessment, sonography, use of the updated RUCAM | □ | □ | □ |
| ● Cocaine, ecstasy and other amphetamines | Toxin screening | □ | □ | □ |
| ● Rare intoxications | Toxin screening for household and occupational toxins | □ | □ | □ |
| ● Hereditary hemochromatosis | Serum ferritin, total iron-binding capacity, genotyping for C2824 and H63D mutation, hepatic iron content | □ | □ | □ |
| ● Wilson disease | Copper excretion (24 h urine), ceruloplasmin in serum, free copper in serum, Coombs-negative hemolytic anemia, hepatic copper content, Kayser-Fleischer-ring, neurologic-psychiatric work-up, genotyping | □ | □ | □ |
| ● Porphyria | Porphobilinogen in urine, total porphyrines in urine | □ | □ | □ |
| ● α1—Antitrypsin deficiency | α1—Antitrypsin in serum | □ | □ | □ |
| ● Biliary diseases | Clinical and laboratory assessment, hepatobiliary sonography, MRC | □ | □ | □ |
| ● Pancreatic diseases | Clinical and laboratory assessment, sonography, CT, MRT | □ | □ | □ |
| ● Celiac disease | TTG antibodies, endomysium antibodies, duodenal biopsy | □ | □ | □ |
| ● Anorexia nervosa | Clinical context | □ | □ | □ |
| ● Parenteral nutrition | Clinical context | □ | □ | □ |
| ● Cardiopulmonary diseases | Cardiopulmonary assessment of congestive heart disease, myocardial infarction, cardiomyopathy, cardiac valvular dysfunction, pulmonary embolism, pericardial diseases, arrhythmia, hemorrhagic shock, and various other conditions | □ | □ | □ |
| ● Addison’s disease | Plasma cortisol | □ | □ | □ |
| ● Thyroid diseases | TSH basal, T4, T3 | □ | □ | □ |
| ● Grand mal seizures | Clinical context of epileptic seizure (duration > 30 min) | □ | □ | □ |
| ● Heat stroke | Shock, hyperthermia | □ | □ | □ |
| ● Polytrauma | Shock, liver injury | □ | □ | □ |
| ● Systemic diseases | Specific assessment of sarcoidosis, amyloidosis, metastatic tumor, sepsis, and others | □ | □ | □ |
| ● Other diseases | Clinical context | □ | □ | □ |
| Reexposure Test Result | Hepatocellular Injury | Cholestatic or Mixed Liver Injury | ||
|---|---|---|---|---|
| ALTb | ALTr | ALPb | ALPr | |
| ● Positive | <5N | ≥2ALTb | <2N | ≥2ALPb |
| ● Negative | <5N | <2ALTb | <2N | <2ALPb |
| ● Negative | ≥5N | ≥2ALTb | ≥2N | ≥2ALPb |
| ● Negative | ≥5N | <2ALTb | ≥2N | <2ALPb |
| ● Uninterpretable | <5N | n.a. | <2N | n.a. |
| ● Uninterpretable | n.a. | ≥2ALTb | n.a. | ≥2ALPb |
| ● Uninterpretable | n.a. | n.a. | n.a. | n.a. |
5.1. Prospective Use
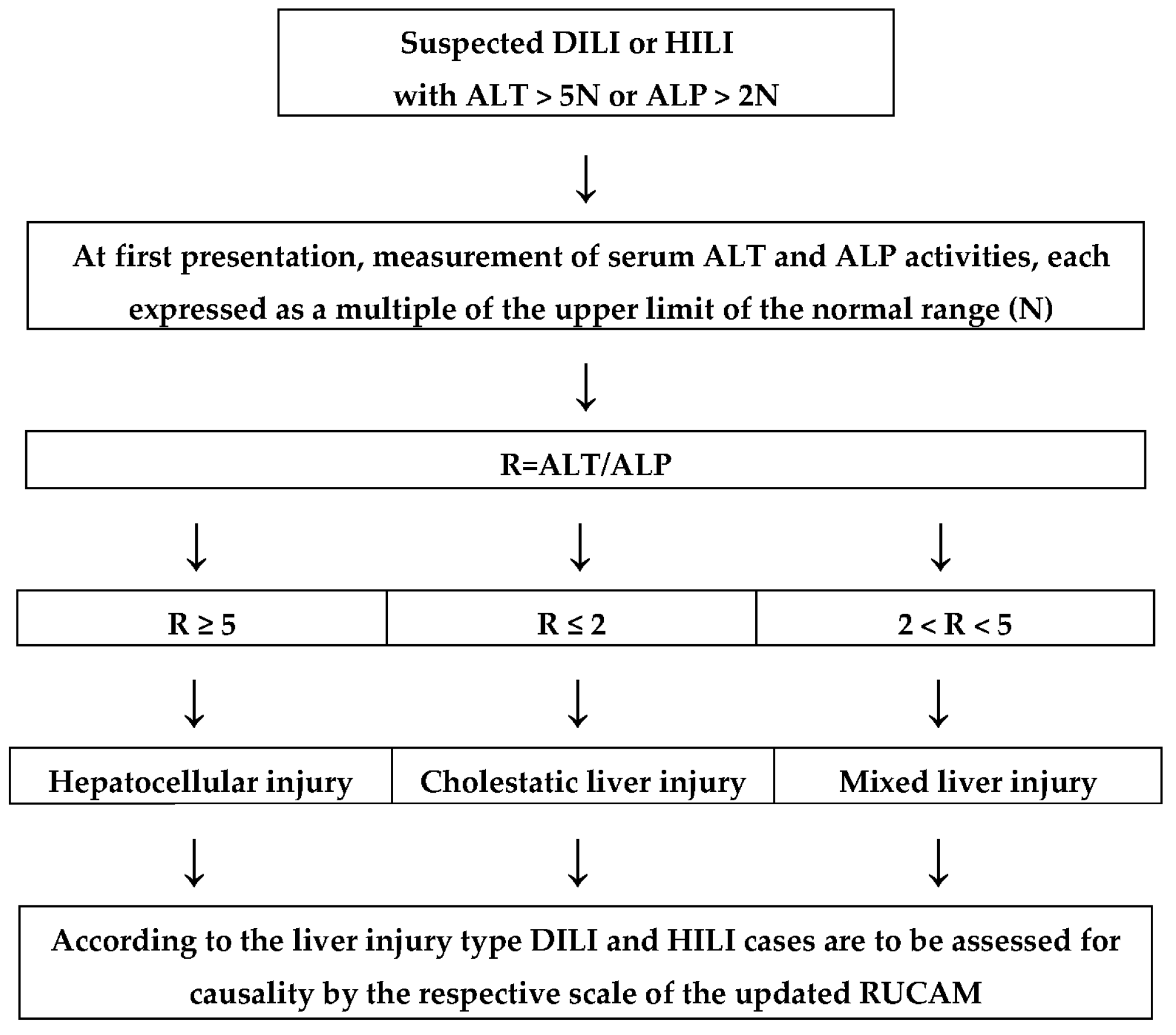
5.2. Case Classification
5.2.1. Liver Injury Criteria
5.2.2. Liver Injury Pattern
5.3. Core Items
5.3.1. Time to Onset from the Beginning of the Drug/Herb Administration
5.3.2. Course of ALT or ALP after Cessation of Drug/Herb
5.3.3. Risk Factors
Alcohol Use
Age
Pregnancy
5.3.4. Concomitant Drug(s) and Herb(s)
5.3.5. Search for Alternative Causes
5.3.6. Previous Hepatotoxicity
5.3.7. Response to Unintentional Reexposure
5.3.8. Final Scores
6. Validity of Updated RUCAM
6.1. Valid Causality Assessment
6.2. Correct Diagnoses
6.3. Missed Diagnoses

7. Updated RUCAM with Its Strengths and Challenges
7.1. Sophisticated Diagnostic Approach
7.2. Strengths
7.3. Mandatory Systematic Documentation for Data Transparency
7.4. Robust Framework by Sequential Assessment
7.5. Open for Worldwide Use
7.6. Limitations
| Items | RUCAM | MV | DILIN | Naranjo | WHO | Ad Hoc |
|---|---|---|---|---|---|---|
| ● Time frame of latency period (score) | + | + | 0 | 0 | 0 | 0 |
| ● Time frame of dechallenge (score) | + | + | 0 | 0 | 0 | 0 |
| ● Recurrent ALT or ALP increase (score) | + | 0 | 0 | 0 | 0 | 0 |
| ● Definition of risk factors (score) | + | 0 | 0 | 0 | 0 | 0 |
| ● All comedications (score) | + | 0 | 0 | + | 0 | 0 |
| ● Individual comedication (score) | + | 0 | 0 | 0 | 0 | |
| ● Search for individual alternative causes (score) | + | + | 0 | 0 | 0 | 0 |
| ● Verified exclusion of specific alternative causes (score) | + | + | 0 | 0 | 0 | 0 |
| ● All specifically assessed HAV, HBV, HCV, HEV (score) | + | 0 | 0 | 0 | 0 | 0 |
| ● All specifically assessed CMV, EBV, HSV, VZV (score) | + | 0 | 0 | 0 | 0 | 0 |
| ● Evaluation of cardiac hepatopathy (score) | + | + | 0 | 0 | 0 | 0 |
| ● Liver and biliary tract imaging (score) | + | 0 | 0 | 0 | 0 | 0 |
| ● Color Doppler sonography of liver vessels (score) | + | 0 | 0 | 0 | 0 | 0 |
| ● Prior known hepatotoxicity (score) | + | + | 0 | + | 0 | 0 |
| ● Search for unintended reexposure (score) | + | + | 0 | + | 0 | 0 |
| ● Definition of unintended reexposure (score) | + | 0 | 0 | 0 | 0 | 0 |
| ● Qualified criteria of unintended reexposure (score) | + | 0 | 0 | 0 | 0 | 0 |
| ● Laboratory hepatotoxicity criteria | + | + | + | 0 | 0 | 0 |
| ● Laboratory hepatotoxicity pattern | + | + | + | 0 | 0 | 0 |
| ● Hepatotoxicity specific method | + | + | + | 0 | 0 | 0 |
| ● Structured, liver related method | + | + | 0 | 0 | 0 | 0 |
| ● Quantitative, liver related method | + | + | 0 | 0 | 0 | 0 |
| ● Validated method (gold standard) | + | 0 | 0 | 0 | 0 | 0 |
8. RUCAM and the Other Causality Assessment Methods
8.1. Liver Specific Methods
8.1.1. MV Scale
8.1.2. TTK Scale
8.1.3. DILIN Method
8.2. Liver Unspecific Methods
8.2.1. Naranjo Scale
8.2.2. WHO Method
8.2.3. Ad Hoc Approach
9. RUCAM and Its International Use
| Cases | Suspected Products | Country or Region | Group/Agency | Year | First Author |
|---|---|---|---|---|---|
| DILI | Multiple synthetic drugs | Spain Europe | Spanish Group for the Study of the Drug-Induced Liver Disease, Malaga | 2005 | Andrade [1] |
| DILI | Multiple synthetic drugs | Spain Europe | Spain Hepatotoxicity Registry, Grupo de Estudio Para las Hepatopatías Asociadas a Medicamentos, Malaga | 2006 | Andrade [2] |
| HILI, DILI | Various herbal TCM, synthetic drugs | Singapore Asia | National University of Singapore | 2006 | Wai [79] |
| HILI | Lu Cha | Sweden Europe | Swedish Adverse Drug Reactions Advisory Committee | 2007 | Björnsson [80] |
| HILI | Black cohosh | Various countries Europe | European Medicines Agency | 2007 | EMA [81] |
| HILI | Herbs | Spain Europe | Spanish Liver Toxicity Registry | 2008 | García-Cortés [82] |
| DILI HILI | Multiple synthetic drugs, few herbs | Spain Europe | Spanish Group for the Study of Drug-induced Liver Disease | 2008 | García-Cortés [76] |
| DILI | Flucloxacillin | UK, other countries | DILIGEN Study & International SAE Consortium | 2009 | Daly [83] |
| DILI | Synthetic drugs | Serbia Europe | Medicines and Medical Devices Agency of Serbia, Belgrade | 2010 | Miljkovic [84] |
| HILI | Polygonum multiflorum | Korea Asia | Gyeongsang National University School of Medicine, Jinju/Sungkyunkwan University School of Medicine, Changwon | 2011 | Jung [85] |
| HILI | Various herbal TCM | Hong Kong | Hong Kong Herb-Induced Liver Injury Network (HK-HILIN), Hong Kong | 2011 | Chau [86] |
| DILI | Multiple synthetic drugs | Spain, other countries | Spanish DILI Registry, EUDRAGENE, DILIN, DILIGEN, and International SAEC. | 2011 | Lucena [87] |
| DILI | Synthetic drugs | Serbia Europe | Medicines and Medical Devices Agency of Serbia, Belgrade | 2011 | Miljkovic [88] |
| DILI | Statins | Iceland/Sweden Europe | National University Hospital Reykjavik/ University of Gothenburg/Swedish Adverse Drug Reactions Advisory Committee (SADRAC) | 2012 | Björnsson [5] |
| DILI | Various synthetic drugs (expected) | Spain Latin America | Spanish-Latin American Network on drug induced liver Injury, in progress | 2012 | Bessone [89] |
| DILI | Flupirtine | Germany Europe | Drug Commission of the German Medical Association | 2012 | Stammschulte [90] |
| HILI | Some Herbalife® products | USA, other countries | Various registries and groups | 2013 | Halegoua de Marzio [91] |
| DILI | Flupirtine | Germany Europe | Berlin Case-control Surveillance Study, German drug reaction reporting database | 2014 | Douros [92] |
| DILI | Anabolic and androgenic steroids | Spain, Latin America | Spanish DILI Registry and Spanish-Latin-American DILI Network | 2015 | Robles-Diaz [93] |
| DILI | Multiple synthetic drugs | Germany Europe | Berlin Case-control Surveillance Study | 2015 | Douros [94] |
| HILI DILI | Multiple dietary supplements and synthetic drugs | USA | Hawaii Department of Health | 2015 | Johnston [95] |
| Cases | Products | Country/Region | Year | First Author |
|---|---|---|---|---|
| DILI | Various synthetic drugs | France Europe | 1993 | Danan [8] |
| DILI | Various synthetic drugs | France Europe | 1993 | Bénichou [9] |
| DILI | Ketoprofen | France Europe | 1998 | Flamenbaum 96] |
| DILI | NSAIDs | Europe Europe | 2003 | Lucena [97] |
| HILI | Kava | Germany Europe | 2003 | Stickel [98] |
| DILI | Various synthetic drugs | Japan Asia | 2003 | Masumotuo [99] |
| DILI | Multiple synthetic drugs | Spain Europe | 2004 | Andrade [100] |
| DILI | Pioglitazone | France Europe | 2004 | Arotcarena [101] |
| DILI | Ximelagatran | USA, France, Sweden | 2005 | Lee W [102] |
| HILI | Ji Xue Cao | Argentina South America | 2005 | Jorge [103] |
| HILI | Lu Cha | France Europe | 2005 | Gloro [104] |
| DILI | Amoxicillin, Amoxicillin/Clavulanate | USA | 2005 | Fontana [105] |
| DILI | Various synthetic drugs | Sweden Europe | 2006 | De Valle [106] |
| HILI | Bo He, Chuan Lian Zi, and various other herbal TCM | Korea Asia | 2006 | Yuen [107] |
| HILI | Lu Cha | Spain Europe | 2006 | Jimenez-Saenz [108] |
| HILI | Polygonum multiflorum | Columbia South America | 2006 | Cárdenas [109] |
| DILI | Rofecoxib | Canada North America | 2006 | Yan [110] |
| DILI | Antibiotics | UK Europe | 2007 | Hussaini [111] |
| DILI | Atomoxetine | USA | 2007 | Stojanovski [112] |
| DILI | Various synthetic drugs | Sweden Europe | 2007 | Björnsson [113] |
| DILI | Flavoxate | Italy Europe | 2007 | Rigato[114] |
| HILI | Kava | Germany Europe | 2008 | Teschke [115] |
| HILI | Bai Xian Pi, Kudzu, Lu Cha, Yin Chen Hao | Korea Asia | 2008 | Kang [116] |
| HILI | Bai Xian Pi, Ci Wu Jia, Shou Wu Pian, Yin Chen Hao | Korea Asia | 2008 | Sohn [117] |
| DILI | Albedazole | Korea Asia | 2008 | Choi [118] |
| HILI | Indian Ayurvedic herbs | Germany Europe | 2009 | Teschke [119] |
| HILI | Green tea (Camellia sinensis) | Italy Europe | 2009 | Mazzanti [120] |
| HILI | Herbalife | Switzerland Europe | 2009 | Stickel [121] |
| HILI | Corydalis speciosa | Korea Asia | 2009 | Kang [122] |
| DILI | Black cohosh | Germany Europe | 2009 | Teschke [123] |
| HILI | Black cohosh | Germany Europe | 2009 | Teschke {124] |
| HILI | Ge Gen | Korea Asia | 2009 | Kim [125] |
| DILI | Montelukast | India Asia | 2009 | Harugeri [126] |
| DILI | Nimesulide | Italy Europe | 2010 | Licata [127] |
| DILI | Tadalafil | Morocco Africa | 2010 | Essaid [128] |
| HILI | Herbalife | Iceland Europe | 2010 | Jóhannsson [129] |
| HILI | H Shou Wu | Korea Asia | 2010 | Bae [130] |
| DILI | Antimicrobial agents | Thailand Asia | 2010 | Treeprasertsuk [131] |
| HILI | Aloe | Korea Asia | 2010 | Yang [132] |
| DILI | Cephalexin | USA | 2010 | Singla [133] |
| HILI | Kava | Germany Europe | 2010 | Teschke [134] |
| HILI | Gynura segetum | Hong Kong Asia | 2011 | Lin [135] |
| DILI | Amiodarone | Israel Europe | 2011 | Gluck[136] |
| DILI | Paracetamol | Spain Europe | 2011 | Sabaté [137] |
| HILI | Greater Celandine | Germany Europe | 2011 | Teschke [73] |
| HILI | Black cohosh | Germany Europe | 2011 | Teschke [60] |
| HILI | Pelargonium sidoides | Germany Europe | 2012 | Teschke [70] |
| DILI | Statins | Sweden Europe | 2012 | Björnsson [5] |
| DILI | Various dietary supplements | Iran Asia | 2012 | Timcheh-Hariri [138] |
| HILI | Greater Celandine | Germany Europe | 2012 | Teschke [139] |
| DILI | Etifoxine | France Europe | 2012 | Moch [140] |
| HILI | Juguju | Korea Asia | 2012 | Kim [141] |
| HILI | Gynura segetum | Hong Kong Asia | 2012 | Gao [142] |
| DILI | Varenicline | USA | 2012 | Sprague [143] |
| DILI HILI | Multiple synthetic drugs and herbs | KoreaAsia | 2012 | Suk [144] |
| DILI | Multiple synthetic drugs | China Asia | 2012 | Hou [145] |
| DILI | Etravirine | USA | 2012 | Nabha [146] |
| HILI | Pelargonium sidoides | Germany Europe | 2012 | Teschke [71] |
| DILI | Crizotinib | France Europe | 2013 | Ripault [147] |
| DILI | Methylprednisolone | France Europe | 2013 | Carrier [148] |
| DILI | Albendazole | Colombia South America | 2013 | Ríos [149] |
| HILI | Herbalife | Germany Europe | 2013 | Teschke [59] |
| DILI | Ibandronate | Belgium Europe | 2013 | Goossens [150] |
| DILI | Bosentan | USA | 2013 | Markova [151] |
| DILI | Cyproterone acetate | Italy Europe | 2013 | Abenavoli [152] |
| DILI | Various synthetic drugs | Iceland Europe | 2013 | Björnsson [153] |
| DILI | NSAID (investigational) | USA | 2013 | Marumoto [154] |
| HILI | Black cohosh | USA | 2014 | Adnan [155] |
| DILI | Volatile anesthetics | Australia | 2014 | Lin [156] |
| DILI | Multiple synthetic drugs | USA | 2014 | Cheetham [157] |
| DILI | Rivaroxaban | Switzerland Europe | 2014 | Russmann [158] |
| DILI | Daptomycin | USA | 2014 | Bohm [159] |
| DILI | Anastrazole | UK Europe | 2014 | Saiful-Islam [160] |
| HILI | Greater Celandine | Korea Asia | 2014 | Im [161] |
| DILI | Various synthetic drugs | USA | 2014 | Lim [162] |
| DILI HILI | Multiple synthetic drugs and herbal TCM | China Asia | 2014 | Hao [163] |
| DILI | Pomalidomide | USA | 2014 | Veluswamy [164] |
| DILI | Amoxicillin | USA | 2014 | Lin [165] |
| HILI | Various dietary supplements | USA | 2014 | Roytman [51] |
| DILI | Sofosbuvir | UK Europe | 2015 | Dyson J [166] |
| DILI HILI | Multiple synthetic drugs and dietary supplements | Germany Europe | 2015 | Teschke [16] |
| HILI | Lesser Celandine | Turkey Europe | 2015 | Yilmaz [167] |
| HILI | Green tea (Camellia sinensis) | Italy Europe | 2015 | Mazzanti [168] |
| DILI | Ipimimumab | Australia | 2015 | Tauquer [169] |
| DILI | Meloxicam | Korea Asia | 2015 | Son [170] |
| DILI | Rivaroxaban | USA | 2015 | Baig [171] |
| DILI | Bupropion, doxycycline | USA | 2015 | Tang [172] |
| DILI HILI | Multiple synthetic drugs and dietary supplements | Germany Europe | 2016 | Teschke [17] |
| DILI HILI | Multiple synthetic drugs and herbs | Korea Asia | 2016 | Woo [173] |
9.1. International Registries and Regulatory Agencies
9.2. Published Reports
10. Perspectives
11. Conclusions
Author Contributions
Conflicts of Interest
References
- Andrade, R.J.; Lucena, M.I.; Fernández, M.C.; Pelaez, G.; Pachkoria, K.; García-Ruiz, E.; García-Muñoz, B.; Gonzalez-Grande, R.; Pizarro, A.; Durán, J.A.; et al. spanish group for the study of drug-induced liver disease. Drug-induced liver injury: An analysis of 461 incidences submitted to the Spanish registry over a 10-year period. Gastroenterology 2005, 129, 512–521. [Google Scholar] [CrossRef] [PubMed]
- Andrade, R.J.; Lucena, M.I.; Kaplowitz, N.; García-Muñoz, B.; Borraz, Y.; Pachkoria, K.; García-Cortés, M.; Fernández, M.C.; Pelaez, G.; Rodrigo, L.; et al. Outcome of acute idiosyncratic drug-induced liver injury: Long term follow-up in a hepatotoxicity registry. Hepatology 2006, 44, 1581–1588. [Google Scholar] [CrossRef] [PubMed]
- Chalasani, N.; Fontana, R.J.; Bonkovsky, H.L.; Watkins, P.B.; Davern, T.; Serrano, J.; Yang, H.; Rochon, J. Drug-induced liver injury network (DILIN). Causes, clinical features, and outcomes from a prospective study of drug-induced liver injury in the United States. Gastroenterology 2008, 135, 1924–1934. [Google Scholar] [CrossRef] [PubMed]
- Liss, G.; Lewis, J.H. Drug-induced liver injury: What was new in 2008? Expert Opin. Drug Metab. Toxicol. 2009, 5, 843–860. [Google Scholar] [CrossRef] [PubMed]
- Björnsson, E.; Jacobsen, E.I.; Kalaitzakis, E. Hepatotoxicity associated with statins: Reports of idiosyncratic liver injury post-marketing. J. Hepatol. 2012, 56, 374–380. [Google Scholar] [CrossRef] [PubMed]
- Chalasani, N.; Bonkovsky, H.L.; Fontana, R.; Lee, W.; Stolz, A.; Talwalkar, J.; Reddy, K.R.; Watkins, P.B.; Navarro, V.; Barnhart, H.; et al. Features and outcomes of 889 patients with drug-induced liver injury: The DILIN prospective study. Gastroenterology 2015, 148, 1340–1352. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Andrade, R. Editorial. Drug-induced liver injury: Expanding our knowledge by enlarging population analysis with prospective and scoring causality assessment. Gastroenterology 2015, 148, 1271–1273. [Google Scholar] [CrossRef] [PubMed]
- Danan, G.; Bénichou, C. Causality assessment of adverse reactions to drugs—I. A novel method based on the conclusions of international consensus meetings: Application to drug-induced liver injuries. J. Clin. Epidemiol. 1993, 46, 1323–1330. [Google Scholar] [CrossRef]
- Bénichou, C.; Danan, G.; Flahault, A. Causality assessment of adverse reactions to drugs—II. An original model for validation of drug causality assessment methods: Case reports with positive rechallenge. J. Clin. Epidemiol. 1993, 46, 1331–1336. [Google Scholar] [CrossRef]
- Seeff, K.B. Herbal hepatotoxicity. Clin. Liver Dis. 2007, 11, 577–596. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Schwarzenboeck, A.; Eickhoff, A.; Frenzel, C.; Wolff, A.; Schulze, J. Clinical and causality assessment in herbal hepatotoxicity. Expert Opin. Drug Saf. 2013, 12, 330–366. [Google Scholar] [CrossRef] [PubMed]
- Bunchorntavakul, C.; Reddy, K.R. Review article: Herbal and dietary supplement hepatotoxicity. Aliment. Pharmacol. Ther. 2013, 37, 3–17. [Google Scholar] [CrossRef] [PubMed]
- Navarro, V.J.; Barnhart, H.; Bonkovsky, H.L.; Davern, T.; Fontana, R.J.; Grant, L.; Reddy, K.R.; Seeff, L.B.; Serrano, J.; Sherker, A.H.; et al. Liver injury from herbals and dietary supplements in the U.S. drug-induced liver injury network. Hepatology 2014, 60, 1399–1408. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Eickhoff, A. Herbal hepatotoxicity in traditional and modern medicine: Actual key issues and new encouraging steps. Front. Pharmacol. 2015, 6, 1–40. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Eickhoff, A.; Schwarzenboeck, A.; Schmidt-Taenzer, W.; Genthner, A.; Frenzel, C.; Wolff, A.; Schulze, J. Clinical review: Herbal hepatotoxicity and the call for systematic data documentation of individual cases. J. Liver Clin. Res. 2015, 2, 1008. [Google Scholar]
- Teschke, R.; Schulze, J.; Eickhoff, A.; Wolff, A.; Frenzel, C. Mysterious Hawaii liver disease case—Naproxen overdose as cause rather than OxyELITE Pro? J. Liver Clin. Res. 2015, 2, 1013. [Google Scholar]
- Teschke, R.; Schwarzenboeck, A.; Frenzel, C.; Schulze, J.; Eickhoff, A.; Wolff, A. The mystery of the Hawaii liver disease cluster in summer 2013, a pragmatic and clinical approach to solve the problem. Ann. Hepatol. 2016, 15, 91–118. [Google Scholar] [CrossRef]
- Aithal, G.P.; Watkins, P.B.; Andrade, R.J.; Larrey, D.; Molokhia, M.; Takikawa, H.; Hunt, C.M.; Wilke, R.A.; Avigan, M.; Kaplowitz, N.; et al. Case definition and phenotype standardization in drug-induced liver injury. Clin. Pharmacol. Ther. 2011, 89, 806–815. [Google Scholar] [CrossRef] [PubMed]
- Urban, T.J.; Daly, A.K.; Aithal, G.P. Genetic basis of drug-induced liver injury: Present and future. Semin. Liver Dis. 2014, 34, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Andrade, R.J.; Robles, M.; Ulzurrun, E.; Lucena, M.I. Drug-induced liver injury: Insights from genetic studies. Pharmacogenomics 2009, 10, 1467–1487. [Google Scholar] [CrossRef] [PubMed]
- Sgro, C.; Clinard, F.; Ouazir, K.; Chanay, H.; Allard, C.; Guilleminet, C.; Lenoir, C.; Lemoine, A.; Hillon, P. Incidence of drug-induced hepatic injuries: A French population-based study. Hepatology 2002, 36, 451–455. [Google Scholar] [CrossRef] [PubMed]
- Lammert, C.; Einarsson, S.; Saha, C.; Niklasson, A.; Bjornsson, E.; Chalasani, N. Relationship between daily dose of oral medications and idiosyncratic drug-induced liver injury: Search for signals. Hepatology 2008, 47, 2003–2009. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Borlak, J.; Tong, W. High lipophilicity and high daily dose of oral medications are associated with significant risk for drug-induced liver injury. Hepatology 2013, 58, 388–396. [Google Scholar] [CrossRef] [PubMed]
- Khandelwal, N.; James, L.P.; Sanders, C.; Larson, A.M.; Lee, W.M. The acute liver failure study group. Unrecognized acetaminophen toxicity as a cause of indeterminate acute liver failure. Hepatology 2011, 53, 567–576. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Liang, X.S.; Li, C.Z. Sinusoidal obstruction syndrome associated with the ingestion of gynura root. Clin. Toxicol. 2010, 48, 962–964. [Google Scholar] [CrossRef] [PubMed]
- eschke, R.; Wolff, A.; Frenzel, C.; Schulze, J. Review article: herbal hepatotoxicity—An update on traditional Chinese medicine preparations. Aliment Pharmacol. Ther. 2014, 40, 32–50. [Google Scholar] [CrossRef] [PubMed]
- Larrey, D.; Faure, S. Herbal medicine hepatotoxicity: A new step with development of specific biomarkers. J. Hepatol. 2011, 54, 599–601. [Google Scholar] [CrossRef] [PubMed]
- Aithal, G.P.; Rawlins, M.D.; Day, C.P. Accuracy of hepatic adverse drug reaction reporting in one English health region. Br. Med. J. 1999, 319, 1541. [Google Scholar] [CrossRef]
- Dalton, H.R.; Fellows, H.J.; Stableforth, W.; Joseph, M.; Thurairajah, P.H.; Warshow, U.; Hazeldine, S.; Remnarace, R.; Ijaz, S.; Hussaini, S.H.; et al. The role of hepatitis E virus testing in drug-induced liver injury. Aliment. Pharmacol. Ther. 2007, 26, 1429–1435. [Google Scholar] [CrossRef] [PubMed]
- Davern, T.J.; Chalasani, N.; Fontana, R.J.; Hayashi, P.H.; Protiva, P.; Kleiner, D.E.; Engle, R.E.; Nguyen, H.; Emerson, S.U.; Purcell, R.H.; et al. Acute hepatitis E infection accounts for some cases of suspected drug-induced liver injury. Gastroenterology 2011, 141, 1665–1672. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Frenzel, C.; Wolff, A.; Eickhoff, A.; Schulze, J. Drug induced liver injury: Accuracy of diagnosis in published reports. Ann. Hepatol. 2014, 13, 248–255. [Google Scholar] [PubMed]
- Teschke, R.; Schulze, J.; Schwarzenboeck, A.; Eickhoff, A.; Frenzel, C. Herbal hepatotoxicity: Suspected cases assessed for alternative causes. Eur. J. Gastroenterol. Hepatol. 2013, 25, 1093–1098. [Google Scholar] [CrossRef] [PubMed]
- National Institutes of Health (NIH) and LiverTox: Causality. Available online: http://livertox.nlm.nih.gov/Causality.html (accessed on 7 November 2015).
- García-Cortés, M.; Stephens, C.; Lucena, M.I.; Fernández-Castañer, A.; Andrade, R.J. Causality assessment methods in drug induced liver injury: Strengths and weaknesses. J. Hepatol. 2011, 55, 683–691. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Wolff, A.; Frenzel, C.; Schwarzenboeck, A.; Schulze, J.; Eickhoff, A. Drug and herb induced liver injury: Council for international organizations of medical sciences scale for causality assessment. World J. Hepatol. 2014, 6, 17–32. [Google Scholar] [PubMed]
- Teschke, R.; Eickhoff, A.; Schulze, J. Drug and herb induced liver injury in clinical and translational hepatology: Causality assessment methods, quo vadis? J. Clin. Transl. Hepatol. 2013, 1, 59–74. [Google Scholar] [CrossRef] [PubMed]
- Bégaud, B.; Evreux, J.C.; Jouglard, J.; Lagier, G. Unexpected or toxic drug reaction assessment (imputation). Actualization of the method used in France. Therapie 1985, 40, 111–118. [Google Scholar] [PubMed]
- Danan, G.; Bénichou, C.; Begaud, B.; Biour, M.; Couzigou, P.; Evreux, J.C.; Lagier, G.; Berthelot, P.; Benhamou, J.P. Critères dimputation dune hépatite aiguë à un médicament. Résultats de réunions de consensus. Gastroenterol. Clin. Biol. 1987, 11, 581–585. [Google Scholar] [PubMed]
- Danan, G. Consensus meetings on: Causality assessment of drug-induced liver injury. J. Hepatol. 1988, 7, 132–136. [Google Scholar] [CrossRef]
- Bénichou, C. Criteria of drug-induced liver disorders. Report of an international consensus meeting. J. Hepatol. 1990, 11, 272–276. [Google Scholar] [PubMed]
- Maria, V.A.J.; Victorino, R.M.M. Development and validation of a clinical scale for the diagnosis of drug-induced hepatitis. Hepatology 1997, 26, 664–669. [Google Scholar] [CrossRef] [PubMed]
- Takikawa, H.; Takamori, Y.; Kumagi, T.; Onji, M.; Watanabe, M.; Shibuya, A.; Hisamochi, A.; Kumashiro, R.; Ito, T.; Mitsumoto, Y.; et al. Assessment of 287 Japanese cases of drug induced liver injury by the diagnostic scale of the International Consensus Meeting. Hepatol. Res. 2003, 27, 192–195. [Google Scholar] [CrossRef]
- Takikawa, H. Recent status of drug-induced liver injury and its problems in Japan. Jpn. Med. Assoc. J. 2010, 53, 243–247. [Google Scholar]
- Hutchinson, T.A.; Lane, D.A. Assessing methods for causality assessment of suspected adverse drug reactions. J. Clin. Epidemiol. 1989, 42, 5–16. [Google Scholar] [CrossRef]
- Hoofnagle, J.H.; Nelson, K.E.; Purcell, R.H. Review article: Hepatitis E. N. Engl. J. Med. 2012, 367, 1237–1244. [Google Scholar] [CrossRef] [PubMed]
- National Institutes of Health (NIH) and LiverTox: Agents Included in LiverTox by Drug Class. Available online: http://livertox.nlm.nih.gov/index.html (accessed on 7 November 2015).
- National Institutes of Health (NIH) and LiverTox: Drug Record. Herbals and Dietary Supplements. Available online: http://livertox.nlm.nih.gov/Herbals_and_Dietary_Supplements.htm (accessed on 7 November 2015).
- Teschke, R.; Genthner, A.; Wolff, A.; Frenzel, C.; Schulze, J.; Eickhoff, A. Herbal hepatotoxicity: Analysis of cases with initially reported positive reexposure tests. Dig. Liver Dis. 2014, 46, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Frenzel, C.; Schulze, J.; Schwarzenboeck, A.; Eickhoff, A. Herbalife hepatotoxicity: Evaluation of cases with positive reexposure tests. World J. Hepatol. 2013, 5, 353–363. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Frenzel, C. Drug induced liver injury: Do we still need a routine liver biopsy for diagnosis today? Ann. Hepatol. 2014, 13, 121–126. [Google Scholar]
- Roytman, M.M.; Pörzgen, P.; Lee, C.L.; Huddleston, L.; Kuo, T.T.; Bryant- Greenwood, P.; Wong, L.L.; Tsai, N. Letter to the editor: Outbreak of severe hepatitis linked to weight-loss supplement OxyElite Pro. Am. J. Gastroenterol. 2014, 109, 1296–1298. [Google Scholar] [CrossRef] [PubMed]
- Hennes, E.M.; Zeniya, M.; Czaja, A.J.; Parés, A.; Dalekos, G.N.; Krawitt, E.L.; Bittencourt, P.L.; Porta, G.; Boberg, K.M.; Hofer, H.; et al. Simplified criteria for the diagnosis of autoimmune hepatitis. Hepatology 2008, 48, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Frenzel, C.; Schulze, J.; Eickhoff, A. Herbal hepatotoxicity: Challenges and pitfalls of causality assessment methods. World J. Gastroenterol. 2013, 19, 2864–2882. [Google Scholar] [CrossRef] [PubMed]
- Fontana, R.J.; Watkins, P.B.; Bonkovsky, H.L.; Chalasani, N.; Davern, T.; Serrano, J.; Rochon, J. Drug-induced liver injury Network (DILIN) prospective study. Rationale, design and conduct. Drug Saf. 2009, 32, 55–68. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, V.K.; McHutchison, J.G.; Hoofnagle, J.H. Drug-Induced Liver Injury Network (DILIN). Important elements for the diagnosis of drug-induced liver injury. Clin. Gastroenterol. Hepatol. 2010, 8, 463–470. [Google Scholar] [CrossRef] [PubMed]
- Naranjo, C.A.; Busto, U.; Sellers, E.M.; Sandor, P.; Ruiz, I.; Roberts, E.A.; Janecek, E.; Domecq, C.; Greenblatt, D.J. A method for estimating the probability of adverse drug reactions. Clin. Pharmacol. Ther. 1981, 30, 239–245. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). The Use of the WHO-UMC System for Standardised Case Causality Assessment. WHO Collaborating Centre for International Drug Monitoring (Uppsala Monitoring Centre, UMC), Database 2000. Available online: http://who-umc.org/Graphics/24734.pdf (accessed on 7 November 2015).
- Kaplowitz, N. Causality assessment versus guilt-by-association in drug hepatotoxicity. Hepatology 2001, 33, 308–310. [Google Scholar] [CrossRef] [PubMed]
- Karch, F.E.; Lasagna, L. Adverse drug reaction. A critical review. JAMA 1975, 234, 1236–1241. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Schmidt-Taenzer, W.; Wolff, A. Spontaneous reports of assumed herbal hepatotoxicity by black cohosh: Is the liver unspecific Naranjo scale precise enough to ascertain causality? Pharmacoepidemiol. Drug Saf. 2011, 20, 567–582. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Schulze, J. Suspected herbal hepatotoxicity: Requirements for appropriate causality assessment by the US Pharmacopeia. Drug Saf. 2012, 35, 1091–1097. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Schwarzenboeck, A.; Schmidt-Taenzer, W.; Wolff, A.; Hennermann, K.H. Herb induced liver injury presumably caused by black cohosh: A survey of initially purported cases and herbal quality specifications. Ann. Hepatol. 2011, 11, 249–259. [Google Scholar]
- Teschke, R.; Schmidt-Taenzer, W.; Wolff, A. USP suspected herbal hepatotoxicity: Quality of causality assessment is more important than quantity of counted cases, not vice versa. Pharmacoepidemiol. Drug Saf. 2012, 21, 336–338. [Google Scholar] [CrossRef]
- Mahady, G.B.; Low Dog, T.; Barrett, M.L.; Chavez, M.L.; Gardiner, P.; Ko, R.; Marles, R.J.; Pellicore, L.S.; Giancaspro, G.I.; Sarma, D.N. United States pharmacopeia review of the black cohosh case reports of hepatotoxicity. Menopause 2008, 15, 628–638. [Google Scholar] [CrossRef] [PubMed]
- Sarma, D.N.; Barrett, M.L.; Chavez, M.L.; Gardiner, P.; Ko, R.; Mahady, G.B.; Marles, R.J.; Pellicore, L.S.; Giancaspro, G.I.; Low Dog, T. Safety of green tea extract: A systematic review by the US pharmacopeia. Drug Saf. 2008, 31, 469–484. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Wolff, A. Regulatory causality evaluation methods applied in kava hepatotoxicity: Are they appropriate? Regul. Toxicol. Pharmacol. 2011, 59, 1–7. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Assessments of the Risk of Hepatotoxicity with Kava Products; WHO Document Production Services: Geneva, Switzerland, 2007. [Google Scholar]
- Björnsson, E.; Olsson, R. Suspected drug-induced liver fatalities reported to the WHO database. Dig. Liver Dis. 2006, 38, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Eickhoff, A.; Wolff, A.; Frenzel, C.; Schulze, J. Herbal hepatotoxicity and WHO global introspection method. Ann. Hepatol. 2013, 12, 11–21. [Google Scholar] [PubMed]
- Teschke, R.; Frenzel, C.; Schulze, J.; Eickhoff, A. Spontaneous reports of primarily suspected herbal hepatotoxicity by Pelargonium sidoides: Was causality adequately ascertained? Regul. Toxicol. Pharmacol. 2012, 63, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Frenzel, C.; Wolff, A.; Herzog, J.; Glass, X.; Schulze, J.; Eickhoff, A. Initially purported hepatotoxicity by Pelargonium sidoides: The dilemma of pharmacovigilance and proposals for improvements. Ann. Hepatol. 2012, 11, 500–512. [Google Scholar] [PubMed]
- Rejection of Drug Risks, Step II. Available online: http://www.spc.int/cis/documents/02_0714_BfArM_Kava_Removal.pdf (accessed on 7 November 2015).
- Teschke, R.; Glass, X.; Schulze, J. Herbal hepatotoxicity by Greater Celandine (Chelidonium majus): Causality assessment of 22 spontaneous reports. Regul. Toxicol. Pharmacol. 2011, 61, 282–291. [Google Scholar] [CrossRef] [PubMed]
- Klontz, K.C.; DeBeck, H.J.; LeBlanc, P.; Mogen, K.M.; Wolpert, B.J.; Sabo, J.L.; Salter, M.; Seelman, S.L.; Lance, S.E.; Monahan, C.; et al. The role of adverse event reporting in the FDA response to multistate outbreak of liver disease associated with a dietary supplement. Public Health Rep. 2015, 130, 526–532. [Google Scholar] [PubMed]
- Teschke, R. Black cohosh and suspected hepatotoxicity—Inconsistencies, confounding variables, and prospective use of a diagnostic causality algorithm: A critical review. Menopause 2010, 17, 426–440. [Google Scholar] [CrossRef] [PubMed]
- García-Cortés, M.; Lucena, M.I.; Pachkoria, K.; Borraz, Y.; Hidalgo, R.; Andrade, R.J. Evaluation of Naranjo adverse drug reactions probability scale in causality assessment of drug-induced liver injury. Aliment. Pharmacol. Ther. 2008, 27, 780–789. [Google Scholar] [CrossRef] [PubMed]
- Tajiri, K.; Shimizu, Y. Practical guideline for diagnosis and early management of drug-induced liver injury. World J. Gastroenterol. 2008, 14, 6774–6785. [Google Scholar] [CrossRef] [PubMed]
- Fenkel, J.M.; Navarro, V.J. Herbal and dietary supplement-induced liver injury. Gastroenterol. Hepatol. 2011, 7, 695–696. [Google Scholar]
- Wai, C.T. Presentation of drug-induced liver injury in Singapore. Singap. Med. J. 2006, 47, 116–120. [Google Scholar]
- Björnsson, E.; Olsson, R. Serious adverse liver reactions associated with herbal weight loss supplements. J. Hepatol. 2007, 47, 295–297. [Google Scholar] [CrossRef] [PubMed]
- European Medicine Agency (EMA). Assessment of Case Reports Connected to Herbal Medicinal Products Containing Cimicifugae Racemosae Rhizoma (Black Cohosh, Root). Available online: http://www.ema.europa.eu/docs/en_GB/document_library/Herbal_-_HMPC_assessment_report/2010/02/WC500074167.pdf (accessed on 7 November 2015).
- García-Cortés, M.; Borraz, Y.; Lucena, M.I.; Peláez, G.; Salmerón, J.; Diago, M.; Martínez-Sierra, M.C.; Navarro, J.M.; Planas, R.; Soria, M.J.; et al. Liver injury induced by “natural remedies”: An analysis of cases submitted to the Spanish Liver Toxicity Registry. Rev. Esp. Enferm. Dig. 2008, 100, 688–695. [Google Scholar] [PubMed]
- Daly, A.K.; Donaldson, P.T.; Bhatnagar, P.; Shen, Y.; Pe’er, I.; Floratos, A.; Daly, M.J.; Goldstein, D.B.; John, S.; Nelson, M.R.; et al. HLA-B*5701 genotype is a major determinant of drug-induced liver injury due to flucloxacillin. Nat. Genet. 2009, 41, 816–819. [Google Scholar] [CrossRef] [PubMed]
- Miljkovic, M.M.; Dobric, S.; Dragojevic-Simic, V. Consistency between causality assessments obtained with two scales and their agreement with clinical judgments in hepatotoxicity. Pharmacoepidemiol. Drug Saf. 2011, 20, 272–285. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.A.; Min, H.J.; Yoo, S.S.; Kim, H.J.; Choi, S.N.; Ha, C.Y.; Kim, H.J.; Kim, T.H.; Jung, W.T.; Lee, O.J.; et al. Drug-induced liver injury: Twenty five cases of acute hepatitis following ingestion of Polygonum multiflorum Thun. Gut Liver 2011, 5, 493–499. [Google Scholar] [CrossRef] [PubMed]
- Chau, T.N.; Cheung, W.I.; Ngan, T.; Lin, J.; Lee, K.W.S.; Poon, W.T.; Leung, V.K.S.; Mak, T.; Tse, M.L. Causality assessment of herb-induced liver injury using multidisciplinary approach and the Roussel Uclaf Causality assessment Method (RUCAM). Clin. Toxicol. 2011, 49, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Lucena, M.I.; Molokhia, M.; Shen, Y.; Urban, T.J.; Aithal, G.P.; Andrade, R.J.; Day, C.P.; Ruiz–Cabello, F.; Donaldson, P.T.; Stephens, C.; et al. Susceptibility to amoxicillin-clavulanate-induced liver injury is influenced by multiple HLA class I and II alleles. Gastroenterology 2011, 141, 338–347. [Google Scholar] [CrossRef] [PubMed]
- Miljkovic, M.M.; Dobric, S.; Dragojevic-Simic, V. Accuracy and reproducibility of two scales in causality assessment of unexpected hepatotoxicity. J. Clin. Pharm. Ther. 2012, 37, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Bessone, F.; Hernandez, N.; Dávalos, M.; Paraná, R.; Schinoni, M.I.; Lizarzabal, M.; Kershenobich, D.; Loaeza, A.; Arrese, M.; Chirino, R.A.; et al. Building a Spanish-Latin American network on drug induced liver injury; much to get from a joint collaborative initiative. Ann. Hepatol. 2012, 11, 544–549. [Google Scholar] [PubMed]
- Cases of Liver Failure in Association with Flupirtine in the German Spontaneous Reporting System. Available online: http://www.akdae.de/Kommission/Organisation/Aufgaben/Publikationen/PDF/Stammschulte2012.pdf (accessed on 7 November 2015).
- Halegoua-De Marzio, D.; Vega, M.; Schifter Weber, J.; Aithal, G.P.; Andrade, R.J.; Bessone, F.; Bjornsson, E.; Bjornsson, H.K.; Larrey, D.G.; Lizarzabal, M.; et al. An international effort to assess hepatotoxicity associated with some herbalife® products. Hepatology 2013, 58, 383A–384A. [Google Scholar]
- Douros, A.; Bronder, E.; Andersohn, F.; Klimpel, A.; Thomae, M.; Orzechowski, H.D.; Kreutz, R.; Garbe, E. Flupirtine-induced liver injury—Seven cases from the Berlin case–control surveillance study and review of the German spontaneous adverse drug reaction reporting database. Eur. J. Clin. Pharmacol. 2014, 70, 453–459. [Google Scholar] [CrossRef] [PubMed]
- Robles-Diaz, M.; Gonzalez-Jimenez, A.; Medina-Caliz, I.; Stephens, C.; García-Cortes, M.; García-Muñoz, B.; Ortega-Alonso, A.; Blanco-Reina, E.; Gonzalez-Grande, R.; Jimenez-Perez, M.; et al. Distinct phenotype of hepatotoxicity associated with illicit use of anabolic androgenic steroids. Aliment. Pharmacol. Ther. 2015, 41, 116–125. [Google Scholar] [CrossRef] [PubMed]
- Douros, A.; Bronder, E.; Andersohn, F.; Klimpel, A.; Thomae, M.; Sarganas, G.; Kreutz, R.; Garbe, E. Drug-induced liver injury: Results from the hospital-based Berlin case–control surveillance study. Br. J. Clin. Pharmacol. 2015, 79, 988–999. [Google Scholar] [CrossRef] [PubMed]
- Johnstone, D.I.; Chang, A.; Viray, M.; Chatham-Stephens, K.; He, H.; Taylor, E.; Wong, L.L.; Schier, J.; Martin, C.; Fabricant, D.; et al. Hepatotoxicity associated with the dietary supplement OxyELITE Pro™—Hawaii 2013. Drug Test Anal. 2015. [Google Scholar] [CrossRef] [PubMed]
- Flamenbaum, M.; Abergel, A.; Marcato, N.; Zénut, M.; Kémény, J.L.; Cassan, P. Regressive fulminant hepatitis, acute pancreatitis and renal insufficiency after taking ketoprofen. Gastroenterol. Clin. Biol. 1998, 22, 975. [Google Scholar] [PubMed]
- Lucena, M.I.; Carvajal, A.; Andrade, R.J.; Velasco, A. Antidepressant-induced hepatotoxicity. Expert Opin. Drug Saf. 2003, 2, 249–262. [Google Scholar] [CrossRef] [PubMed]
- Stickel, F.; Baumüller, H.M.; Seitz, K.; Vasilakis, D.; Seitz, G.; Seitz, H.K.; Schuppan, D. Hepatitis induced by Kava (Piper methysticum rhizoma). J. Hepatol. 2003, 39, 62–67. [Google Scholar] [CrossRef]
- Masumoto, T.; Horiike, N.; Abe, M.; Kumaki, T.; Matsubara, H.; Fazle Akbar, S.M.; Michitaka, K.; Hyodo, I.; Onji, M. Diagnosis of drug-induced liver injury in Japanese patients by criteria of the Consensus Meetings in Europe. Hepatol. Res. 2003, 25, 1–7. [Google Scholar] [CrossRef]
- Andrade, R.J.; Lucena, M.I.; Alonso, A.; García-Cort́es, M.; García-Ruiz, E.; Benitez, R.; Fernández, M.C.; Pelaez, G.; Romero, M.; Corpas, R.; et al. HLA class II genotype influences the type of liver injury in drug-induced idiosyncratic liver disease. Hepatology 2004, 39, 1603–1612. [Google Scholar] [CrossRef] [PubMed]
- Arotcarena, R.; Bigué, J.P.; Etcharry, F.; Pariente, A. Pioglitazone-induced acute severe hepatitis. Gastroenterol. Clin. Biol. 2004, 28, 609–618. [Google Scholar]
- Lee, W.M.; Larrey, D.; Olsson, R.; Lewis, J.H.; Keisu, M.; Auclert, L.; Sheth, S. Hepatic findings in long-term clinical trials of ximelagatran. Drug Saf. 2005, 28, 351–370. [Google Scholar] [CrossRef] [PubMed]
- Jorge, O.A.; Jorge, A.D. Hepatotoxicity associated with the ingestion of Centella asiatica. Rev. Esp. Enferm. Dig. 2005, 97, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Gloro, R.; Hourmand-Ollivier, I.; Mosquet, B.; Mosquet, L.; Rousselot, P.; Salamé, E.; Piquet, M.A.; Dao, T. Fulminant hepatitis during self-medication with hydroalcoholic extract of green tea. Eur. J. Gastroenterol. Hepatol. 2005, 17, 1135–1137. [Google Scholar] [CrossRef] [PubMed]
- Fontana, R.J.; Shakil, O.; Greenson, J.K.; Boyd, I.; Lee, W.M. Acute liver failure due to amoxicillin and amoxicillin/clavulanate. Dig. Dis. Sci. 2005, 10, 1785–1790. [Google Scholar] [CrossRef] [PubMed]
- De Valle, M.B.; AV Klinteberg, V.; Alem, N.; Olsson, R.; Björnsson, E. Drug-induced liver injury in a Swedish University hospital out-patient hepatology clinic. Aliment. Pharmacol. Ther. 2006, 24, 1187–1195. [Google Scholar] [CrossRef] [PubMed]
- Yuen, M.F.; Tam, S.; Fung, J.; Wong, D.K.H.; Wong, B.C.Y.; Lai, C.L. Traditional Chinese Medicine causing hepatotoxicity in patients with chronic hepatitis B infection: A 1-year prospective study. Aliment. Pharmacol. Ther. 2006, 24, 1179–1186. [Google Scholar] [CrossRef] [PubMed]
- Jimenez-Saenz, M.; Martinez-Sanchez M del, C. Acute hepatitis associated with the use of green tea infusions. J. Hepatol. 2006, 44, 616–617. [Google Scholar] [CrossRef] [PubMed]
- Cárdenas, A.; Restrepo, J.C.; Sierra, F.; Correa, G. Acute hepatitis due to shen-min: A herbal product derived from Polygonum multiflorum. J. Clin. Gastroenterol. 2006, 40, 629–632. [Google Scholar] [CrossRef] [PubMed]
- Yan, B.; Leung, Y.; Urbanski, S.J.; Myers, R.P. Rofecoxib-induced hepatotoxicity: A forgotten complication of the coxibs. Can. J. Gastroenterol. 2006, 20, 351–355. [Google Scholar] [PubMed]
- Hussaini, S.H.; O’Brien, C.S.; Despott, E.J.; Dalton, H.R. Antibiotic therapy: A major cause of drug-induced jaundice in southwest England. Eur. J. Gastroenterol. Hepatol. 2007, 19, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Stojanovski, S.D.; Casavant, M.J.; Mousa, H.M.; Baker, P.; Nahata, M.C. Atomoxetine-induced hepatitis in a child. Clin. Toxicol. 2007, 45, 51–55. [Google Scholar] [CrossRef]
- Björnsson, E.; Kalaitzakis, E.; Klinteberg, V.A.V.; Alem, E.; Olsson, R. Long-term follow-up of patients with mild to moderate drug-induced liver injury. Alimdnt. Pharmacol. 2007, 26, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Rigato, I.; Cravatari, M.; Avellini, C.; Ponte, E.; Crocè, S.L.; Tiribelli, C. Drug-induced acute cholestatic liver damage in a patient with mutation of UGT1A1. Nat. Clin. Pract. Gastroenterol. Hepatol. 2007, 4, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Schwarzenboeck, A.; Hennermann, K.H. Kava hepatotoxicity: A clinical survey and critical analysis of 26 suspected cases. Eur. J. Gastroenterol. Hepatol. 2008, 20, 1182–1193. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.H.; Kim, J.I.; Jeong, K.H.; Ko, K.H.; Ko, P.G.; Hwang, S.W.; Kim, E.M.; Kim, S.H.; Lee, H.Y.; Lee, B.S. Clinical characteristics of 159 cases of acute toxic hepatitis. Korean J. Hepatol. 2008, 14, 483–492. [Google Scholar] [CrossRef] [PubMed]
- Sohn, C.H.; Cha, M.I.; Oh, B.J.; Yeo, W.H.; Lee, J.H.; Kim, W.; Lim, K.S. Liver transplantation for acute toxic hepatitis due to herbal medicines and preparations. J. Korean Soc. Clin. Toxicol. 2008, 6, 110–116. [Google Scholar]
- Choi, G.Y.; Yang, H.W.; Cho, S.H.; Kang, D.W.; Go, H.; Lee, W.C.; Lee, Y.J.; Jung, S.H.; Kim, A.N.; Cha, S.W. Drug-induced hepatitis caused by albendazole. J. Korean Med. Sci. 2008, 23, 903–905. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Bahre, R. Severe hepatotoxicity by Indian Ayurvedic herbal products: A structured causality assessment. Ann. Hepatol. 2009, 8, 258–266. [Google Scholar] [PubMed]
- Mazzanti, G.; Menniti-Ippolito, F.; Moro, P.A.; Cassetti, F.; Raschetti, R.; Santuccio, C.; Mastrangelo, S. Hepatotoxicity from green tea: A review of the literature and two unpublished cases. Eur. J. Clin. Pharmacol. 2009, 65, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Stickel, F.; Droz, S.; Patsenker, E.; Bögli-Stuber, K.; Aebi, B.; Leib, S.L. Severe hepatotoxicity following ingestion of Herbalife nutritionally supplements contaminated with Bacillus subtilis. J. Hepatol. 2009, 50, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.S.; Choi, H.S.; Yun, T.J.; Lee, K.G.; Seo, Y.S.; Yeon, J.E.; Byun, K.S.; Um, S.H.; Kim, C.D.; Ryu, H.S. A case of acute cholestatic hepatitis induced by Corydalis speciosa Max. Korean J. Hepatol. 2009, 15, 517–523. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Schwarzenboeck, A. Suspected hepatotoxicity by cimicifugae racemosae rhizoma (black cohosh, root): Critical analysis and structured causality assessment. Phytomedicine 2009, 16, 72–84. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Bahre, R.; Fuchs, J.; Wolff, A. Black cohosh hepatotoxicity: Quantitative causality evaluation in nine suspected cases. Menopause 2009, 16, 956–965. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Yim, H.J.; Ahn, J.H.; Kim, J.H.; Kim, J.N.; Yoon, I.; Kim, D.I.; Lee, H.S.; Lee, S.W.; Choi, J.H. Two cases of toxic hepatitis caused by arrowroot juice. Korean J. Hepatol. 2009, 15, 504–509. [Google Scholar] [CrossRef] [PubMed]
- Harugeri, A.; Parthasarathi, G.; Sharma, J.; DSouza, G.A.; Ramesh, M. Montelukast induced acute hepatocellular injury. J. Postgrad. Med. 2009, 55, 141–142. [Google Scholar] [PubMed]
- Licata, A.; Calvaruso, V.; Capello, M.; Craxi, A.; Almasio, P.L. Clinical course and outcomes of drug-induced liver injury: Nimesulide as the first implicated medication. Dig. Liver Dis. 2010, 42, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Essaid, A.; Timraz, A. Cholestatic acute hepatitis induced by tadalafil (Cialis®). Gastroenterol. Clin. Biol. 2010, 34, e1–e2. [Google Scholar] [CrossRef] [PubMed]
- Jóhannsson, M.; Ormarsdóttir, S.; Olafsson, S. Hepatotoxicity associated with the use of Herbalife. Laeknabladid 2010, 96, 167–172. [Google Scholar] [PubMed]
- Bae, S.H.; Kim, D.H.; Bae, Y.S.; Lee, K.J.; Kim, D.W.; Yoon, J.B.; Hong, J.H.; Kim, S.H. Toxic hepatitis associated with Polygoni multiflori. Korean J. Hepatol. 2010, 16, 182–186. [Google Scholar] [CrossRef] [PubMed]
- Treeprasertsuk, S.; Huntrakul, J.; Ridtitid, W.; Kullavanijaya, P.; Björnsson, E.S. The predictors of complications in patients with drug-induced liver injury caused by antimicrobial agents. Aliment. Pharmacol. Ther. 2010, 11, 1200–1207. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.N.; Kim, D.J.; Kim, Y.M.; Kim, B.H.; Sohn, K.M.; Choi, M.J.; Choi, Y.H. Aloe-induced toxic hepatitis. J. Korean Med. Sci. 2010, 25, 492–495. [Google Scholar] [CrossRef] [PubMed]
- Singla, A.; Hammad, H.T.; Hammoud, G.M. Uncommon cause of acute drug-induced liver injury following mammoplasty. Gastroenterol. Res. 2010, 3, 171–172. [Google Scholar] [CrossRef]
- Teschke, R. Kava hepatotoxicity: A clinical review. Ann. Hepatol. 2010, 9, 251–265. [Google Scholar] [PubMed]
- Lin, G.; Wang, J.Y.; Li, N.; Li, M.; Gao, H.; Ji, Y.; Zhang, F.; Wang, H.; Zhou, Y.; Ye, Y.; et al. Hepatic sinusoidal obstruction syndrome associated with consumption of Gynura segetum. J. Hepatol. 2011, 54, 666–673. [Google Scholar] [CrossRef] [PubMed]
- Gluck, N.; Fried, M.; Porat, R. Acute amiodarone liver toxicity likely due to ischemic hepatitis. Isr. Med. Assoc. J. 2011, 13, 748–752. [Google Scholar] [PubMed]
- Sabaté, M.; Ibáñez, L.; Pérez, E.; Vidal, X.; Buti, M.; Xiol, X.; Mas, A.; Guarner, C.; Forné, M.; Solà, R.; et al. Paracetamol in therapeutic dosages and acute liver injury: Causality assessment in a prospective case series. BMC Gastroenterol. 2011, 11, 80. [Google Scholar] [CrossRef] [PubMed]
- Timcheh-Hariri, A.; Balali-Mood, M.; Aryan, E.; Sadeghi, M.; Riahi-Zanjani, B. Toxic hepatitis in a group of 20 male body-builders taking dietary supplements. Food Chem. Toxicol. 2012, 50, 3826–3832. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Frenzel, C.; Glass, X.; Schulze, J.; Eickhoff, A. Greater Celandine hepatotoxicity: A clinical review. Ann. Hepatol. 2012, 11, 838–848. [Google Scholar] [PubMed]
- Moch, C.; Rocher, F.; Lainé, P.; Lacotte, J.; Biour, M.; Gouraud, A.; Bernard, N.; Descotes, J.; Vial, T. Etifoxine-induced acute hepatitis: A case series. Clin. Res. Gastroenterol. Hepatol. 2012, 36, e85–e88. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Ryu, S.L.; Shim, J.W.; Kim, D.S.; Shim, J.Y.; Park, M.S.; Jung, H.L. A pediatric case of toxic hepatitis induced by Hovenia dulcis. Pediatr. Gastroenterol. Hepatol. Nutr. 2012, 15, 111–116. [Google Scholar] [CrossRef]
- Gao, H.; Li, N.; Wang, J.Y.; Zhang, S.C.; Lin, G. Definitive diagnosis of hepatic sinusoidal obstruction syndrome induced by pyrrolizidine alkaloids. J. Dig. Dis. 2012, 13, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Sprague, D.; Bamha, K. Drug-induced liver injury due to varenicline. BMC Gastroenterol. 2012, 12, 65. [Google Scholar] [CrossRef] [PubMed]
- Suk, K.T.; Kim, D.J.; Kim, C.H.; Park, S.H.; Yoon, J.H.; Kim, Y.S.; Baik, G.H.; Kim, J.B.; Kweon, Y.O.; Kim, B.I.; et al. A prospective nationwide study of drug-induced liver injury in Korea. Am. J. Gastroenterol. 2012, 107, 1380–1387. [Google Scholar] [CrossRef] [PubMed]
- Hou, F.Q.; Zeng, Z.; Wang, G.Q. Hospital admissions for drug-induced liver injury: Clinical features, therapy, and outcomes. Cell Biochem. Biophys. 2012, 64, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Nabha, L.; Balba, G.P.; Tuanzon, C.; Kumar, P.N. Etravirine induced severe hpersensitivity reaction and fulminant hepatitis: A case report and review of the literature. J. AIDS Clin. Res. 2012. [Google Scholar] [CrossRef]
- Ripault, M.P.; Pinzani, V.; Fayolle, V.; Pageaux, G.P.; Larrey, D. Crizotinib-induced acute hepatitis: First case with relaps after reintroduction with reduced dose. Clin. Res. Gastroenterol. Hepatol. 2013, 37, e21–e23. [Google Scholar] [CrossRef] [PubMed]
- Carrier, P.; Godet, B.; Crepin, S.; Magy, L.; Debette-Gratien, M.; Pillegand, B.; Jacques, J.; Sautereau, D.; Vidal, E.; Labrousse, F.; et al. Acute liver toxicity due to methylprednisolone: Consider this diagnosis in the context of autoimmunity. Clin. Res. Gastroenterol. Hepatol. 2013, 37, 100–104. [Google Scholar] [CrossRef] [PubMed]
- Ríos, D.; Restrepo, J.C. Abendazole-induced liver injury: A case report. Colomb. Med. 2013, 44, 118–120. [Google Scholar] [PubMed]
- Goossens, N.; Spahr, L.; Rubbia-Brandt, L. Severe immune-mediated drug-induced liver injury linked to ibandronate: A case report. J. Hepatol. 2013, 59, 1139–1142. [Google Scholar] [CrossRef] [PubMed]
- Markova, S.M.; de Marco, T.; Bendjilali, N.; Kobashigawa, E.A.; Mefford, J.; Sodhi, J.; Le, H.; Zhang, C.; Halladay, J.; Rettie, A.E.; et al. Association of CYP2C9*2 with bosentan-induced liver injury. Clin. Pharmacol. Ther. 2013, 94, 678–686. [Google Scholar] [CrossRef] [PubMed]
- Abenavoli, L.; Milic, N.; Beaugrand, M. Severe hepatitis by cyproterone acetate: Role of corticosteroids. A case report. Ann. Hepatol. 2013, 1, 152–155. [Google Scholar]
- Björnsson, E.S.; Bergmann, O.M.; Björnsson, H.K.; Kvaran, R.B.; Olafsson, S. Incidence, presentation and outcomes in patients with drug-induced liver injury in the general population of Iceland. Gastroenterology 2013, 144, 1419–1425. [Google Scholar] [CrossRef] [PubMed]
- Marumoto, A.; Roytman, M.M.; Tsai, N.C.S. Trial and error: Investigational drug induced liver injury, a case series report. Hawaiì J. Med. Public Health 2013, 72, 30–33. [Google Scholar] [PubMed]
- Adnan, M.M.; Khan, M.; Hashmi, S.; Hamza, M.; AbdulMujeeb, S.; Amer, S. Black cohosh and liver toxicity: Is there a relationship? Case Rep. Gastrointest. Med. 2014. [Google Scholar] [CrossRef]
- Lin, J.; Moore, D.; Hockey, B.; Di Lernia, R.; Gorelik, A.; Liew, D.; Nicoll, A. Drug-induced hepatotoxicity: Incidence of abnormal liver function tests consistent with volatile anaesthetic hepatitis in traumatic patients. Liver Intern. 2014, 34, 576–586. [Google Scholar] [CrossRef] [PubMed]
- Cheetham, T.C.; Lee, J.; Hunt, C.M.; Niu, F.; Reisinger, S.; Murray, R.; Powell, G.; Papay, J. An automated causality assessment algorithm to detect drug-induced liver injury in electronic medical record data. Pharmacoepidemiol. Drug Saf. 2014, 23, 601–608. [Google Scholar] [CrossRef] [PubMed]
- Russmann, S.; Niedrig, D.F.; Budmiger, M.; Schmidt, C.; Stieger, B.; Hürlimann, S.; Kullak-Ublick, G.A. Rivaroxaban postmarketing risk of liver injury. J. Hepatol. 2014, 61, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Bohm, N.; Bohm, N.; Makowski, C.; Machado, M.; Davie, A.; Seabrook, N.; Wheless, L.; Bevill, B.; Clark, B.; Kyle, T.R., III. Case report and cohort analysis of drug-induced liver injury associated with daptomycin. Antimicrobial. Agents Chemother. 2014, 58, 4902–4903. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Saiful-Islam, M.; Wright, G.; Tanner, P.; Lucas, R. A case of anastrazole-related drug-induced autoimmune hepatitis. Clin. J. Gastroenterol. 2014, 7, 414–417. [Google Scholar] [CrossRef] [PubMed]
- Im, S.G.; Yoo, S.H.; Jeon, D.O.; Cho, H.J.; Choi, J.Y.; Paik, S.; Park, Y.M. Chelidonium majus-induced acute hepatitis. Ewha Med. J. 2014, 37, 60–63. [Google Scholar] [CrossRef]
- Lim, R.; Choundry, H.; Conner, K.; Karnsakul, W. A challenge for diagnosing acute liver injury with concomitant/sequential exposure to multiple drugs: Can causality assessment scales be utilized to identify the offending drug? Case Rep. Pediatr. 2014, 2014, 156389. [Google Scholar] [CrossRef] [PubMed]
- Hao, K.; Yu, Y.; He, C.; Wang, M.; Wang, S.; Li, X. RUCAM scale-based diagnosis, clinical features and prognosis of 140 cases of drug-induced liver injury. J. Zhonghua Gan Zang Bing 2014, 22, 938–994. [Google Scholar]
- Veluswamy, R.R.; Ward, S.C.; Yum, K.; Abramovitz, R.B.; Isola, L.M.; Jagannath, S.; Parekh, S. Adverse drug reaction: Pomalidomide-induced liver injury. Lancet 2014, 383, 2125–2126. [Google Scholar] [CrossRef]
- Lin, P.F. A Case of Amoxicillin Hepatocellular Injury. Available online: http://www.med.ucla.edu/modules/xfsection/article.php?articleid=804 (accessed on 7 November 2015).
- Dyson, J.K.; Hutchinson, J.; Harrison, L.; Rotimi, O.; Tiniakos, D.; Foster, G.R.; Aldersley, M.A.; McPherson, S. Liver toxicity associated with sofosbuvir, an NS5A inhibitor and ribavirin use. J. Hepatol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, B.; Yilmaz, B.; Aktaş, B.; Unlu, O.; Roach, E.C. Lesser celandine (pilewort) induced acute toxic liver injury: The first case report worldwide. World J. Hepatol. 2015, 7, 285–288. [Google Scholar] [CrossRef] [PubMed]
- Mazzanti, G.; Di Soto, A.; Vitalone, A. Hepatotoxicity of green tea: An update. Arch. Toxicol. 2015, 89, 1175–1191. [Google Scholar] [CrossRef] [PubMed]
- Tauqeer, A.; Pandey, R.; Shah, R.; Black, J. Resolution of ipilimumab induced severe hepatotoxicity with triple immunosuppressants therapy. BMJ Case Rep. 2015. [Google Scholar] [CrossRef]
- Son, C.G. Drug-induced liver injury by Western medication. J. Int. Korean Med. 2015, 36, 69–75. [Google Scholar]
- Baig, M.; Wool, K.J.; Halalnych, J.H.; Sarmad, R.A. Acute liver failure after initiation of rivaroxaban: A case report and review of the literature. N. Am. J. Med. Sci. 2015, 7, 407–410. [Google Scholar] [CrossRef] [PubMed]
- Tang, D.M.; Koh, C.; Twaddell, W.S.; von Rosenvinge, E.C.; Han, H. Acute hepatocellular drug-induced liver injury from bupropion and doxycycline. ACG Case Rep. J. 2015, 3, 66–68. [Google Scholar] [CrossRef] [PubMed]
- Woo, H.J.; Kim, H.Y.; Choi, E.S.; Cho, Y.; Kim, Y.; Lee, J.H.; Jang, E. Drug-induced liver injury: A 2-year retrospective study of 1169 hospitalized patients in a single medical center. Phytomedicine 2016, 22, 1201–1205. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Danan, G.; Teschke, R. RUCAM in Drug and Herb Induced Liver Injury: The Update. Int. J. Mol. Sci. 2016, 17, 14. https://doi.org/10.3390/ijms17010014
Danan G, Teschke R. RUCAM in Drug and Herb Induced Liver Injury: The Update. International Journal of Molecular Sciences. 2016; 17(1):14. https://doi.org/10.3390/ijms17010014
Chicago/Turabian StyleDanan, Gaby, and Rolf Teschke. 2016. "RUCAM in Drug and Herb Induced Liver Injury: The Update" International Journal of Molecular Sciences 17, no. 1: 14. https://doi.org/10.3390/ijms17010014
APA StyleDanan, G., & Teschke, R. (2016). RUCAM in Drug and Herb Induced Liver Injury: The Update. International Journal of Molecular Sciences, 17(1), 14. https://doi.org/10.3390/ijms17010014







