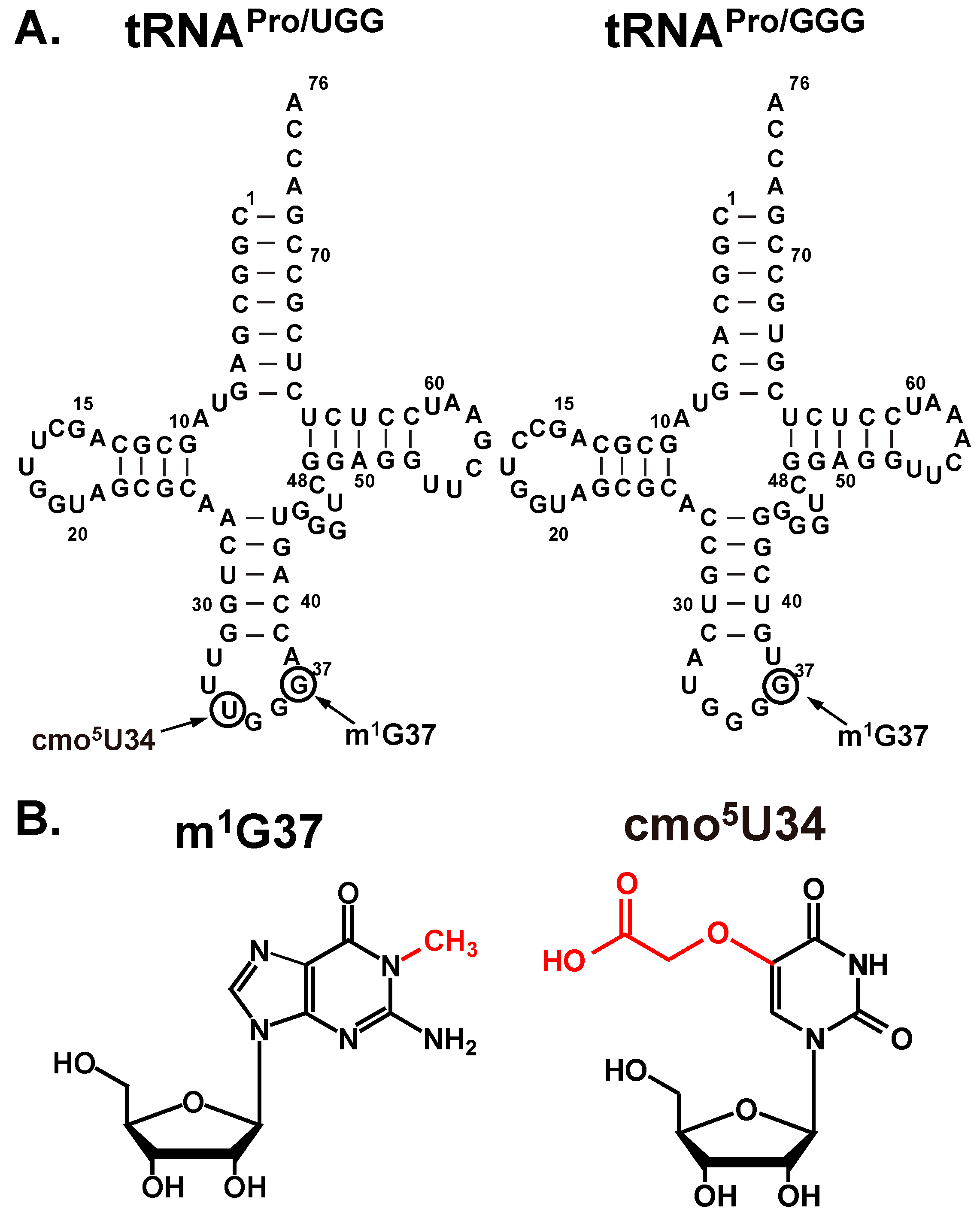
2.1. The Native-State UGG tRNAPro Is Unable to Fully Convert a Dipeptide to a Tripeptide
We have used the mRNA sequence 5ʹ-AUG-CCC-CGU-U to study non-programmed +1FS events (
Figure 2A) [
11]. In this sequence, the slippery CCC-C motif is next to the AUG start codon, which is the most shift-prone position in the entire reading frame [
11]. For a translating
E. coli ribosome carrying the fMP dipeptide on tRNAPro (fM = formyl methionine) in the post-complex, the correct reading of the slippery motif followed by one more round of peptide bond formation will generate the tripeptide fMPR, whereas the +1-frame reading will generate the tripeptide fMPV. These alternative products are readily resolved by electrophoretic thin layer chromatography (eTLC), allowing quantitative analysis of the fractional conversion from the dipeptide fMP to the tripeptide fMPR or fMPV at the site of the CCC-C motif. Using this mRNA sequence, we developed an assay to detect tRNA
Pro shifting into the +1-frame in a post-complex [
11]. Specifically, we assembled the post-complex by taking the
E. coli 70S initiation complex (70SIC) through the first round of peptide bond formation to place fMP-tRNA
Pro in the P-site next to an empty A-site (
Figure 2B). By allowing fMP-tRNA
Pro to shift into the +1-frame over time, we quenched the shift by simultaneously adding ternary complexes of tRNA
Arg and tRNA
Val to take the ribosome through the next peptide bond formation. It was with this assay that we observed the synthesis of fMPV tripeptide, indicating that tRNA
Pro can shift into the +1-frame [
11]. Control experiments confirmed that the synthesis of fMPV indeed reflected the +1-frame shift of tRNA
Pro, rather than the miscoding by tRNA
Val [
11]. However, the shift from a stalled post-complex occurs at a slow rate relative to the rate of peptide bond formation [
11], suggesting that it is relevant only when the A-site is empty or contains a rare codon.
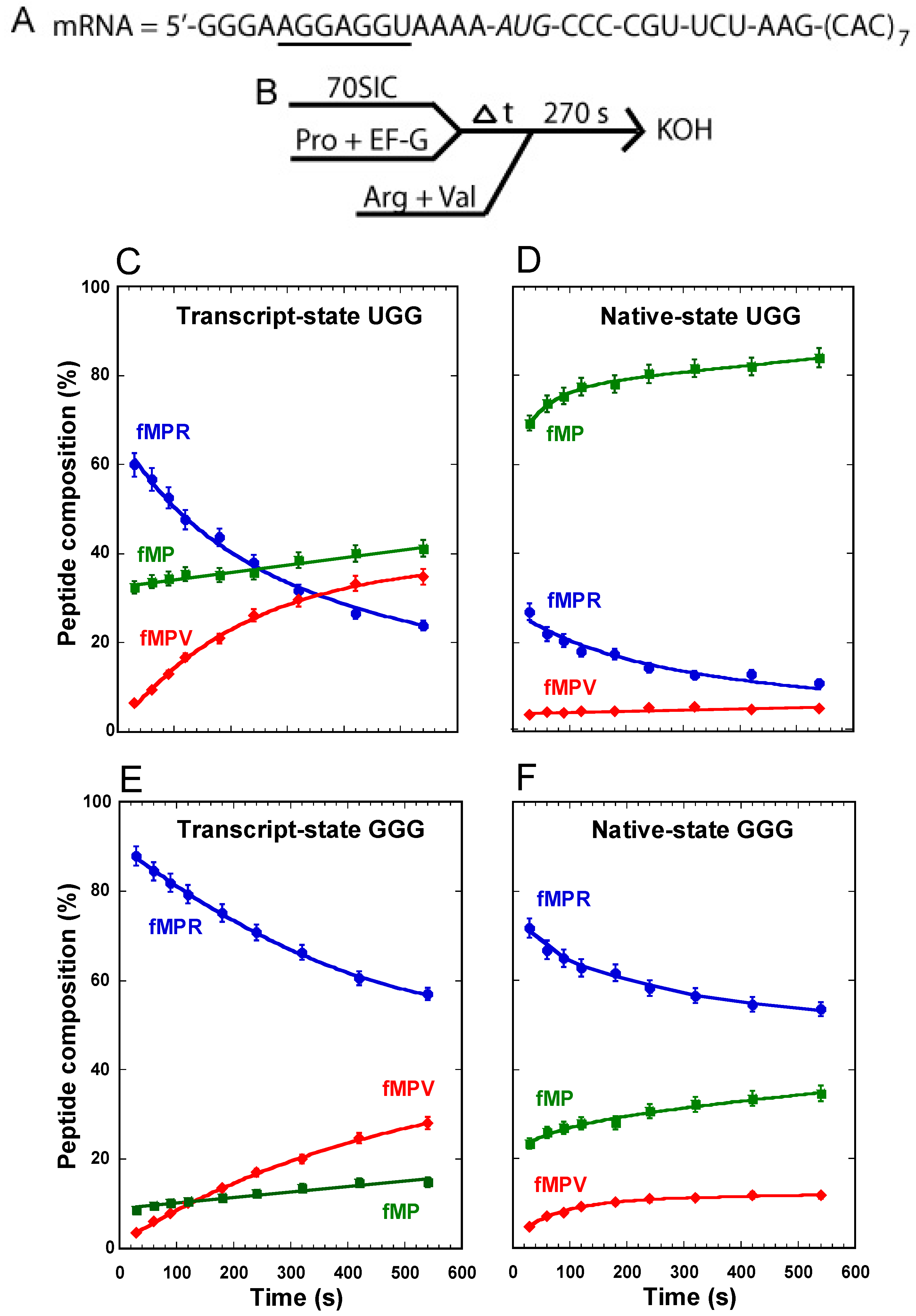
Our recent study with this assay primarily focused on the synthesis of fMPV [
11] without much attention to the unreacted fMP substrate or the 0-frame product fMPR. To gain more insight into all three components and their quantitative changes over time in the course of the reaction, we applied this assay to study the UGG isoacceptor of tRNA
Pro and analyzed the fractional composition of each peptide among all three. With the transcript-state of the tRNA, made by
in vitro transcription and lacking all natural post-transcriptional modifications, we observed a progressive accumulation of the fMPV product from background to 40%, a rapid synthesis of the fMPR product to 60% followed by a decrease to 20%, and a substantial consumption of the substrate fMP down to 40%, which remained relatively stable over the course of the 10-min reaction (
Figure 2C). The stable level of the remaining fMP dipeptide, together with the steady increase of the fMPV tripeptide and the concomitant decrease of the fMPR tripeptide, indicates that the substrate was readily consumed, as the tRNA gradually shifted from the 0-frame into the +1-frame for the next round of peptide bond formation.
With the native-state of the UGG tRNA, isolated from cells and containing all natural post-transcriptional modifications, we observed a suppression of the synthesis of the fMPV product to only ~2%, a suppression of the synthesis of the fMPR product to 10%, but surprisingly little consumption of the substrate fMP, which remained at 80% (
Figure 2D). Relative to the transcript-state, the suppression of the synthesis of fMPV in the native-state is attributable to the presence of m
1G37, but not cmo
5U34. In fact, a variant of the native-state, containing cmo
5U34 but lacking m
1G37, was as shift-prone as the transcript-state, showing high activity of synthesis of the fMPV product [
11]. This indicates that it is m
1G37 that is the major determinant for suppressing +1FS errors of this tRNA. However, the high level of unconsumed fMP in the native-state was unexpected.
For comparison, the transcript-state of the GGG isoacceptor behaved similarly as the UGG counterpart, enabling a steady increase of the synthesis of the fMPV product, a steady decrease of the fMPR product, and a substantial consumption of the substrate fMP down to 20% (
Figure 2E). In the native-state, we also observed a suppression of the synthesis of fMPV, attributable to the presence of m
1G37 (
Figure 2F) [
11]. While the suppression of the synthesis of fMPV was not as effective as in the UGG isoacceptor, this is because the reaction lacked the protein elongation factor EF-P, which is required to completely suppress +1FS errors of the GGG isoacceptor [
11]. However, a major difference was observed for the substrate fMP; while it was consumed to below 40% when attached to the native-state GGG isoacceptor, it was hardly consumed when attached to the native-state UGG isoacceptor (
Figure 2D
vs. F).
Figure 2.
Analysis of forward +1 frameshifts of tRNAPro from a stalled ribosome complex. (A) The template mRNA sequence, showing the 5ʹ-Shine Dalgarno sequence (underlined), the AUG start codon (italic), and the slippery CCC-C sequence immediately following the start codon; (B) The reaction scheme for the conversion from fMP to tripeptides, starting with mixing a 70SIC with the ternary complex of tRNAPro in the presence of EF-G to form a post-complex with fMP-tRNAPro in the P-site. After stalling for various time periods, the complex was reacted with ternary complexes of tRNAArg and tRNAVal for 270 s before being quenched by KOH. The changes in the peptide composition over time for the substrate fMP, the 0-frame product fMPR, and the +1-frame fMPV were monitored for (C) the transcript-state of the UGG isoacceptor of tRNAPro; (D) the native-state of the UGG isoacceptor; (E) the transcript-state of the GGG isoacceptor; and (F) the native-state of the GGG isoacceptor. Each data point is the average of 3 independent measurements.
Figure 2.
Analysis of forward +1 frameshifts of tRNAPro from a stalled ribosome complex. (A) The template mRNA sequence, showing the 5ʹ-Shine Dalgarno sequence (underlined), the AUG start codon (italic), and the slippery CCC-C sequence immediately following the start codon; (B) The reaction scheme for the conversion from fMP to tripeptides, starting with mixing a 70SIC with the ternary complex of tRNAPro in the presence of EF-G to form a post-complex with fMP-tRNAPro in the P-site. After stalling for various time periods, the complex was reacted with ternary complexes of tRNAArg and tRNAVal for 270 s before being quenched by KOH. The changes in the peptide composition over time for the substrate fMP, the 0-frame product fMPR, and the +1-frame fMPV were monitored for (C) the transcript-state of the UGG isoacceptor of tRNAPro; (D) the native-state of the UGG isoacceptor; (E) the transcript-state of the GGG isoacceptor; and (F) the native-state of the GGG isoacceptor. Each data point is the average of 3 independent measurements.
![Ijms 16 14866 g002]()
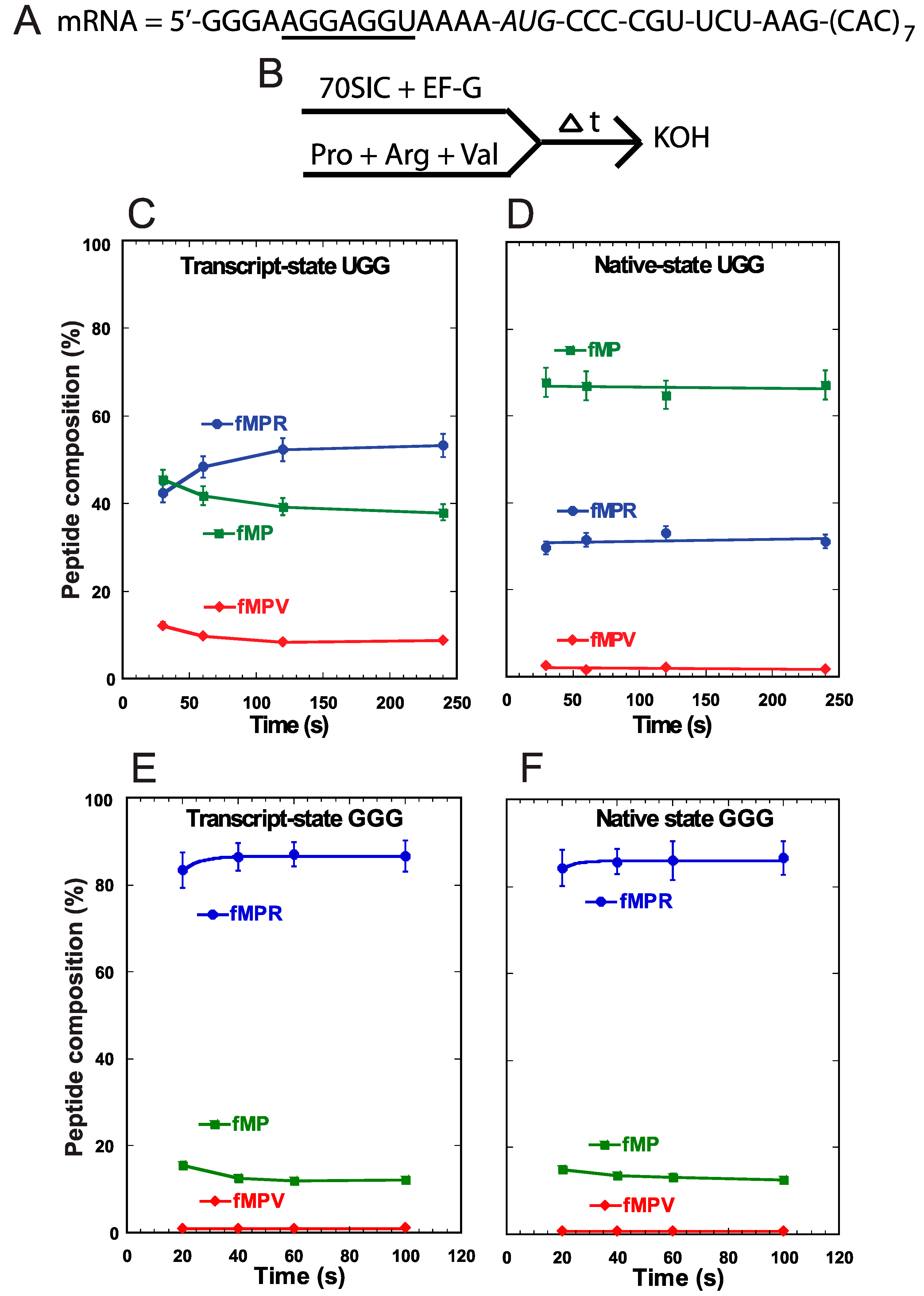
Figure 3.
Analysis of forward +1 frameshifts of tRNAPro during translocation into the P-site. (A) The template mRNA sequence, showing the 5ʹ-Shine Dalgarno sequence (underlined), the AUG start codon (italic), and the slippery CCC-C sequence immediately following the start codon; (B) The reaction scheme for the conversion from fMP to tripeptides, starting with rapid mixing of a 70SIC with ternary complexes of tRNAPro, tRNAArg, and tRNAVal, in the presence of EF-G. The reaction was quenched over time by KOH. The changes in the peptide composition over time for the substrate fMP, the 0-frame product fMPR, and the +1-frame fMPV were monitored for (C) the transcript-state of the UGG isoacceptor of tRNAPro; (D) the native-state of the UGG isoacceptor; (E) the transcript-state of the GGG isoacceptor; and (F) the native-state of the GGG isoacceptor. Each data point is the average of 3 independent measurements.
Figure 3.
Analysis of forward +1 frameshifts of tRNAPro during translocation into the P-site. (A) The template mRNA sequence, showing the 5ʹ-Shine Dalgarno sequence (underlined), the AUG start codon (italic), and the slippery CCC-C sequence immediately following the start codon; (B) The reaction scheme for the conversion from fMP to tripeptides, starting with rapid mixing of a 70SIC with ternary complexes of tRNAPro, tRNAArg, and tRNAVal, in the presence of EF-G. The reaction was quenched over time by KOH. The changes in the peptide composition over time for the substrate fMP, the 0-frame product fMPR, and the +1-frame fMPV were monitored for (C) the transcript-state of the UGG isoacceptor of tRNAPro; (D) the native-state of the UGG isoacceptor; (E) the transcript-state of the GGG isoacceptor; and (F) the native-state of the GGG isoacceptor. Each data point is the average of 3 independent measurements.
We performed a different assay to monitor +1FS errors during translocation of tRNA
Pro into the P-site. In this assay, we assembled the 70SIC on the same slippery mRNA sequence (
Figure 3A) and took the ribosome complex through two successive rounds of peptide bond formation in the presence of EF-G and ternary complexes of tRNA
Pro, tRNA
Arg, and tRNA
Val (
Figure 3B). Because the A-site was occupied throughout the assay, and because there is no evidence of tRNA shifting in the A-site [
11], the synthesis of fMPV indicated that tRNA
Pro had shifted to the +1-frame during its translocation into the P-site. We showed recently that the rate of +1-shifting during translocation was fast [
11], comparable to the rate of normal peptide bond formation, suggesting its potential to challenge the reading-frame maintenance in actively growing cells. Using this concerted assay, we detected low levels of accumulation of the +1-frame product fMPV with the UGG isoacceptor in both the transcript-state and the native-state (
Figure 3C,D). In fact, because the level of fMPV did not change appreciably between the two states, this supports the notion that +1FS errors during tRNA translocation are not suppressed by m
1G37 or other post-transcriptional modifications [
11]. Importantly, while both states also synthesized the 0-frame product fMPR to similar levels, they differed in the consumption of the fMP substrate. While the transcript-state showed a time-dependent consumption of fMP down to 40%, the native-state maintained a high level of fMP (>60%) with little decrease over time (
Figure 3C,D). The inability to convert fMP to either product was unique to the native-state of the UGG isoacceptor and was not observed with the GGG isoacceptor. For the latter, the fMP substrate was readily converted to the 0-frame product fMPR at high levels and to the +1-frame product fMPV at low levels, independent of the transcript- or native-state (
Figure 3E,F). These results confirmed that +1FS errors are typically low during tRNA translocation into the P-site and that such errors are not suppressible by post-transcriptional modifications. Importantly, unlike the UGG isoacceptor, the GGG isoacceptor in both the transcript- and native-state consumes the fMP substrate to below 20%.
2.2. Base Pairing Stability of cmo5U34 in the Native-State of the UGG Isoacceptor
The unusually high level of unreacted fMP was observed only with the native-state of the UGG isoacceptor, which set this tRNA apart from the other three studied above. One possibility was that this tRNA preferentially dropped off from the ribosome relative to the other three. This was unlikely, given that it is the major form of tRNA
Pro and that it is in the fully modified state, which is more robust in the binding and activity with the ribosome than the transcript-state [
11]. We thus considered the possibility that, due to the possession of cmo
5U34, the native-state UGG isoacceptor is highly shift-prone on a slippery mRNA sequence and can shift into alternative frames other than the +1-frame. This possibility is based on the unusual pairing stability of cmo
5U34. While the cmo
5U34-containing UGG isoacceptor can recognize all four 5ʹ-CCN codons for Pro, stability of the wobble base pair in the codon-anticodon duplex varies greatly among the four. Crystal structural analysis of ribosome-tRNA complexes suggested that the pairing of cmo
5U34 with A or G is the most stable, followed by the pairing with U, and by the pairing with C [
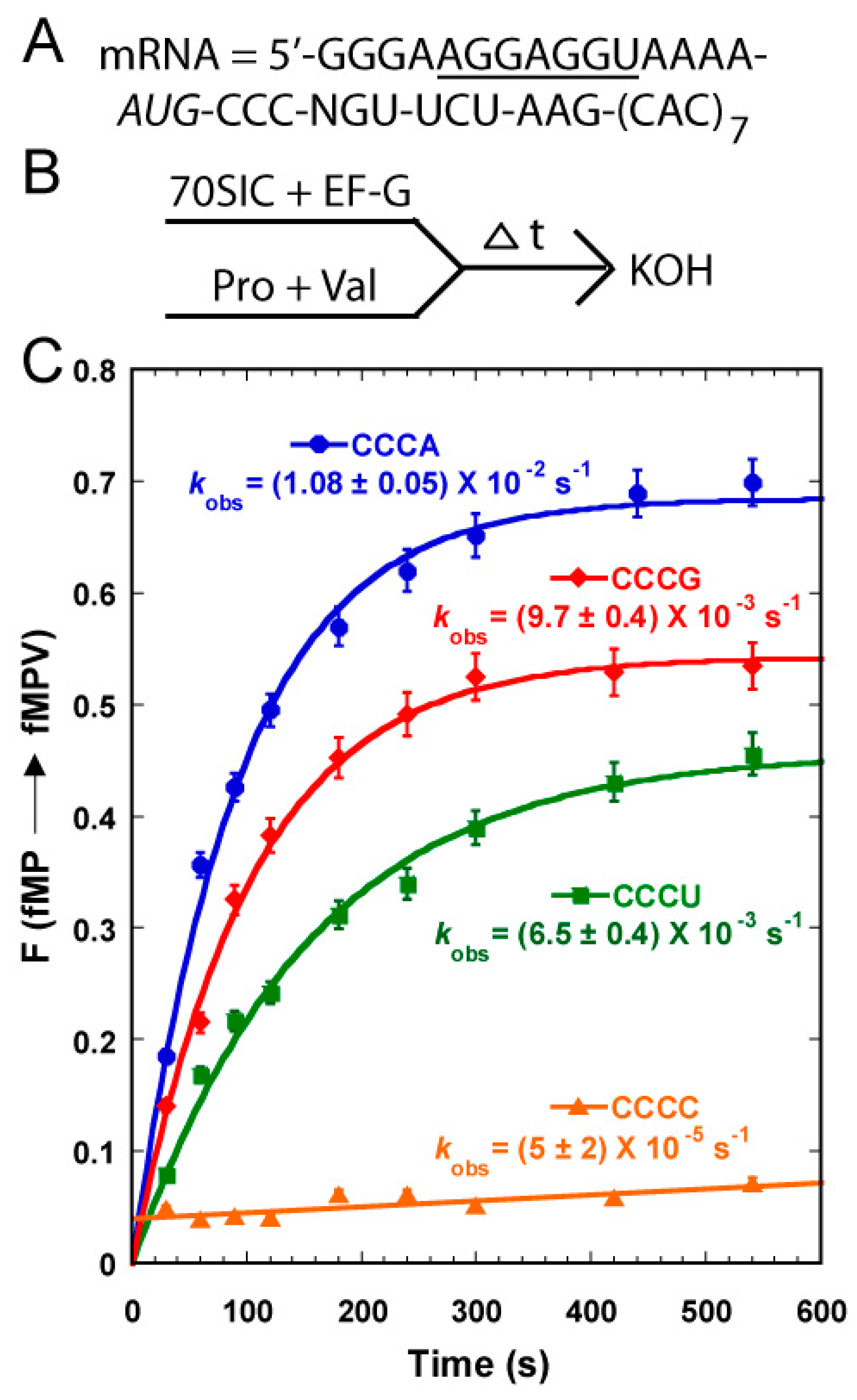
11]. To test this hypothesis, we took 70SIC through two rounds of peptide bond formation with ternary complexes of the native-state UGG isoacceptor and the +1-frame tRNA
Val (
Figure 4A,B). In this design, the post-complex of fMP-tRNA
Pro after the first peptide bond formation must shift into the +1-frame to react with tRNA
Val to synthesize the fMPV product. The speed and level of fMPV synthesis would be driven by the stability of the pairing between its cmo
5U34 and the N nucleotide on the mRNA. A time-dependent analysis showed that the fractional conversion was the fastest and reached the highest level with the CCC-A mRNA sequence, followed by the CCC-G mRNA, and by the CCC-U mRNA, with the conversion being the poorest with the CCC-C mRNA (
Figure 4C). This relative shifting propensity, in both the kinetics and the overall level, was in complete agreement with the structural prediction, supporting the notion that base pairing stability of cmo
5U34-N at the wobble position is the major determinant to drive the shift. In contrast, the transcript-state of the same tRNA shifted with similar efficiency among the four mRNA sequences without a preference [
11], suggesting that the pairing between the unmodified U34 and the N base lacked a thermodynamic selectivity. This comparison highlights the ability of the modified cmo
5U34 relative to the unmodified U34 to differentiate among base pairing partners.
Figure 4.
The propensity of shifting into the +1-frame of the native-state UGG isoacceptor of tRNAPro on mRNA CCC-N sequences. (A) The template mRNA sequences, showing the 5ʹ-Shine Dalgarno sequence (underlined), the AUG start codon (italic), and the slippery CCC-N sequence immediately following the start codon, where N = A, C, G, and U; (B) The reaction scheme for monitoring +1FS by rapid mixing of a 70SIC with ternary complexes of tRNAPro and tRNAVal in the presence of EF-G; and (C) Kinetics of the fractional conversion (F) from fMP to fMPV was monitored over time. The kobs for each mRNA sequence was indicated and each data point is the average of 3 independent measurements.
Figure 4.
The propensity of shifting into the +1-frame of the native-state UGG isoacceptor of tRNAPro on mRNA CCC-N sequences. (A) The template mRNA sequences, showing the 5ʹ-Shine Dalgarno sequence (underlined), the AUG start codon (italic), and the slippery CCC-N sequence immediately following the start codon, where N = A, C, G, and U; (B) The reaction scheme for monitoring +1FS by rapid mixing of a 70SIC with ternary complexes of tRNAPro and tRNAVal in the presence of EF-G; and (C) Kinetics of the fractional conversion (F) from fMP to fMPV was monitored over time. The kobs for each mRNA sequence was indicated and each data point is the average of 3 independent measurements.
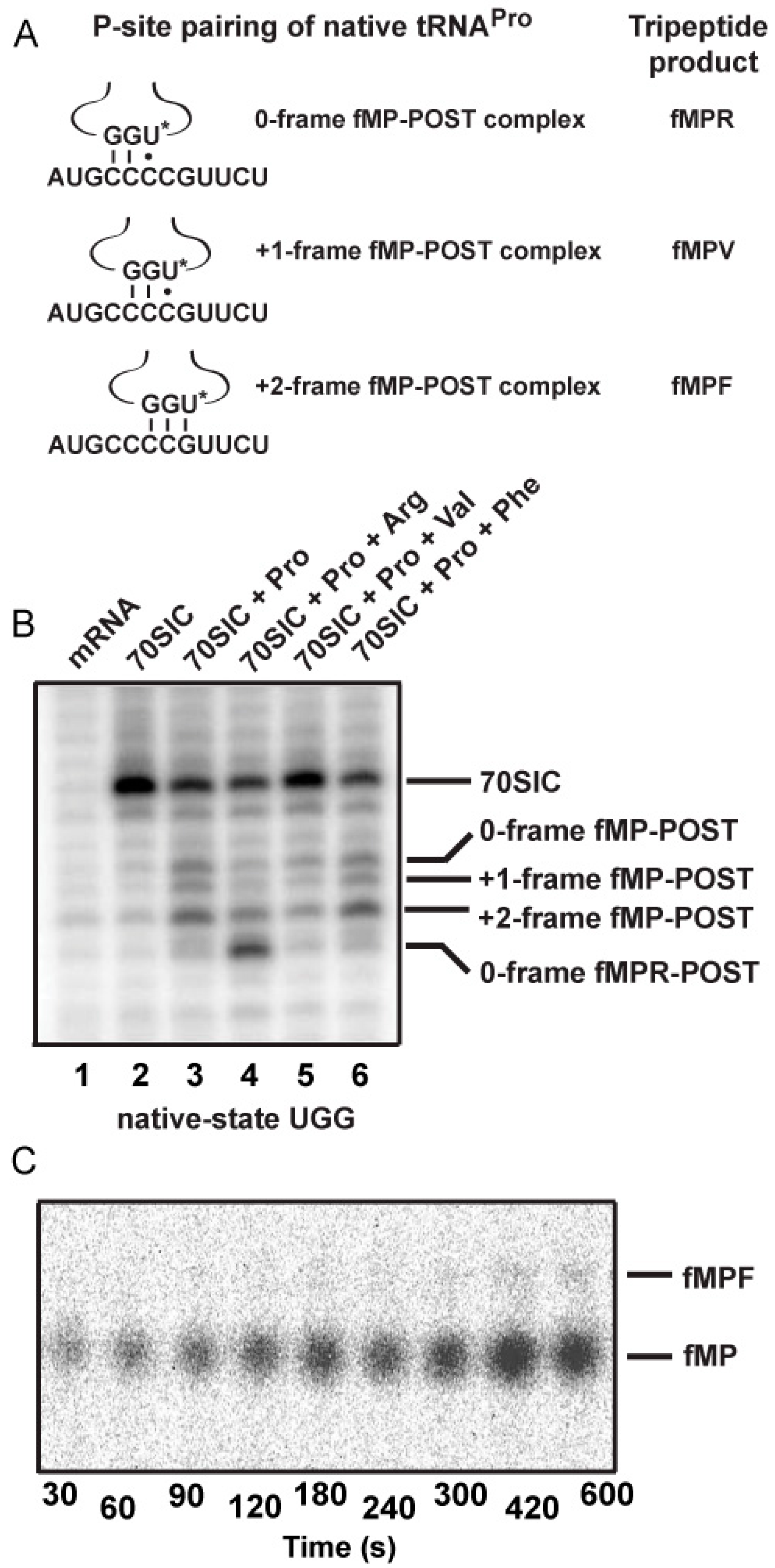
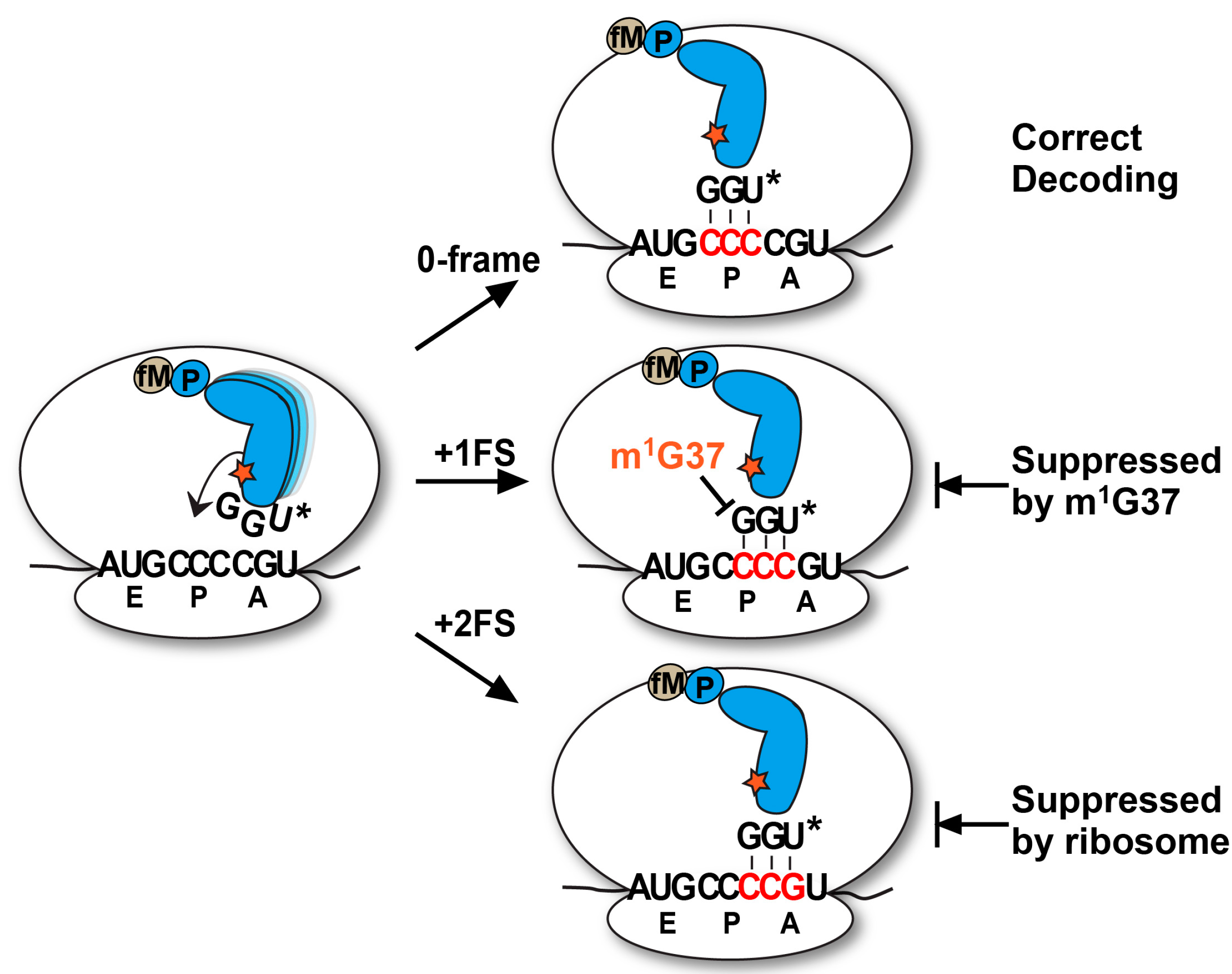
2.3. Evidence of the Native-State UGG tRNAPro Shifting into the +2-Frame
With the native-state UGG isoacceptor, we tested if the high levels of unreacted fMP substrate (
Figure 2C) was due to its shifting beyond the +1-frame to a position unable to participate in peptide bond formation. Based on the established order of relative stability, the reading of the cmo
5UGG anticodon in the 0-frame or the +1-frame of a post-complex would be in the least stable context, involving the cmo
5U-C base pair, whereas the reading in the +2-frame would be in a more stable context, involving the cmo
5U-G base pair (
Figure 5A). We hypothesized that the difference in stability would provide a strong thermodynamic drive in favor of the tRNA to shift, not only by one, but more preferably by two nucleotides into the +2-frame. In contrast, we hypothesized that the transcript-state containing the unmodified U34, which would pair with G poorly, lacked the thermodynamic drive to shift into the +2-frame. The poor pairing of an unmodified U34 with G is notable and has been reported previously [
11].
Toeprint analysis supported our hypothesis, showing the shift into the +2-frame of the native-state UGG isoacceptor (
Figure 5B). Using the same slippery mRNA sequence in the context of a longer mRNA, we formed post-complexes at various positions on the mRNA by mixing 70SIC with the ternary complex of the native-state UGG isoacceptor alone or in the presence of ternary complexes of the next 0-frame, +1-frame, or +2-frame tRNA. We then determined the position of the P-site in each complex by primer extension of an oligonucleotide annealed to the 3ʹ-end. In this design, the mRNA alone posed no block to primer extension (lane 1), while the 70SIC with fM-tRNA
fMet posed a block at the P-site (lane 2). The post-complex with each addition of an amino acid would move the P-site down by one codon distance toward the 3ʹ-end, thereby shortening the length of the primer extension accordingly. In the post-complex formed with 70SIC and the ternary complex of the native-state UGG tRNA
Pro, the synthesized fMP dipeptide was found at all three frames but most prominently in the +2-frame (43% of all possible frames, lane 3), indicating that the tRNA had shifted from the 0-frame, transiently passed the +1-frame, and stably ended up at the +2-frame. In the post-complex formed with ternary complexes of tRNA
Pro and tRNA
Arg (the next 0-frame tRNA), the synthesized tripeptide fMPR was primarily found 6-nucleotide down from the 70SIC (58% of all possible frames, lane 4), indicating the movement of the tRNA-ribosome complex by two in-frame codons. The in-frame movement of the fMPR-post complex served as a positive control for tripeptide formation, although a notable fraction of the post-complex was also found 5-nucleotide down (lane 4), indicating a shift into the +2-frame before settling at the 6-nucleotide down position. However, in the post-complex formed with ternary complexes of tRNA
Pro and tRNA
Val, primer extension stopped with the fMP dipeptide at the 0- or the +2-frame, but no stop with the fMPV tripeptide at the +1-frame (lane 5). This result is consistent with the notion that, once the fMP dipeptide was formed, a fraction of the native-state UGG tRNA stayed in the 0-frame and another fraction rapidly moved into the +2-frame, while the +1-frame fMPV tripeptide was barely detectable (
Figure 5C), due to the low yield of production (~5%) as shown in kinetic analysis (
Figure 4, mRNA = CCC-C).
Figure 5.
Dipeptidyl and tripeptidyl post-complexes formed on a slippery mRNA sequence. (
A) Post-complexes were formed on a long mRNA that contained the initial AUG-CCC-CGU-UCU coding sequence as used in
Figure 2 and
Figure 3. The fMP-post complex is shown with the native-state UGG isoacceptor in the 0-, +1-, and +2-frame. The codon-anticodon pairing interaction and the tripeptide product of each after one more round of peptide bond formation is illustrated. The U
* denotes cmo
5U34; (
B) Toeprints of ribosome post-complexes formed by interaction of 70SIC with ternary complexes of the indicated tRNAs in the presence of EF-G. Unbound mRNA was included as a control and the 70SIC with a P-site bound fMet-tRNA
fMet was used as a reference for the reading frame; and (
C) eTLC analysis of +2-frame fMPF formation. 70SIC with an mRNA with the initial coding sequence of AUG-CCC-CGU-UCU was mixed with ternary complexes of native tRNA
Pro/UGG and tRNA
Phe/GAA in the presence of EF-G and incubated at 20 °C. Aliquots were quenched in 0.5 M KOH prior to analysis by eTLC.
Figure 5.
Dipeptidyl and tripeptidyl post-complexes formed on a slippery mRNA sequence. (
A) Post-complexes were formed on a long mRNA that contained the initial AUG-CCC-CGU-UCU coding sequence as used in
Figure 2 and
Figure 3. The fMP-post complex is shown with the native-state UGG isoacceptor in the 0-, +1-, and +2-frame. The codon-anticodon pairing interaction and the tripeptide product of each after one more round of peptide bond formation is illustrated. The U
* denotes cmo
5U34; (
B) Toeprints of ribosome post-complexes formed by interaction of 70SIC with ternary complexes of the indicated tRNAs in the presence of EF-G. Unbound mRNA was included as a control and the 70SIC with a P-site bound fMet-tRNA
fMet was used as a reference for the reading frame; and (
C) eTLC analysis of +2-frame fMPF formation. 70SIC with an mRNA with the initial coding sequence of AUG-CCC-CGU-UCU was mixed with ternary complexes of native tRNA
Pro/UGG and tRNA
Phe/GAA in the presence of EF-G and incubated at 20 °C. Aliquots were quenched in 0.5 M KOH prior to analysis by eTLC.
![Ijms 16 14866 g005]()
Analysis of the post-complex formed with ternary complexes of tRNA
Pro and tRNA
Phe revealed an intriguing result. While tRNA
Phe was cognate to the UUC codon in the +2-frame (
Figure 5A), primer extension was blocked with the fMP dipeptide primarily at the +2-frame (46% of all frames, lane 6) without evidence for the formation of a tripeptidyl-tRNA. This result suggests that the +2-frame ribosome complex failed to support peptide synthesis even with the cognate tRNA
Phe. Indeed, eTLC analysis of reactions containing 70SIC and ternary complexes of the UGG isoacceptor and tRNA
Phe yielded a low yield of tripeptide (less than 1% of the substrate,
Figure 5C), supporting the notion that the +2-frame post-complex was a poor substrate for peptide bond formation. Thus, because of the inability of the +2-frame post-complex to catalyze peptide bond formation, the data explain why the fMP substrate was not consumed with the native-state UGG tRNA
Pro. In cellular conditions, such a +2-frame post-complex, stalled at the P-site and unable to perform peptidyl transfer, is likely resolved by the drop-off of the P-site tRNA or by the several rescue mechanisms available to bacterial ribosomes [
22,
23,
24].