Biomedical Application of Low Molecular Weight Heparin/Protamine Nano/Micro Particles as Cell- and Growth Factor-Carriers and Coating Matrix
Abstract
:1. Introduction
2. Biomedical Applications of Low Molecular Weight Heparin/Protamine Nano/Micro Particles (LMWH/P N/MPs)
2.1. Preparation and Function of Growth Factors (GFs) and LMWH/P N/MPs
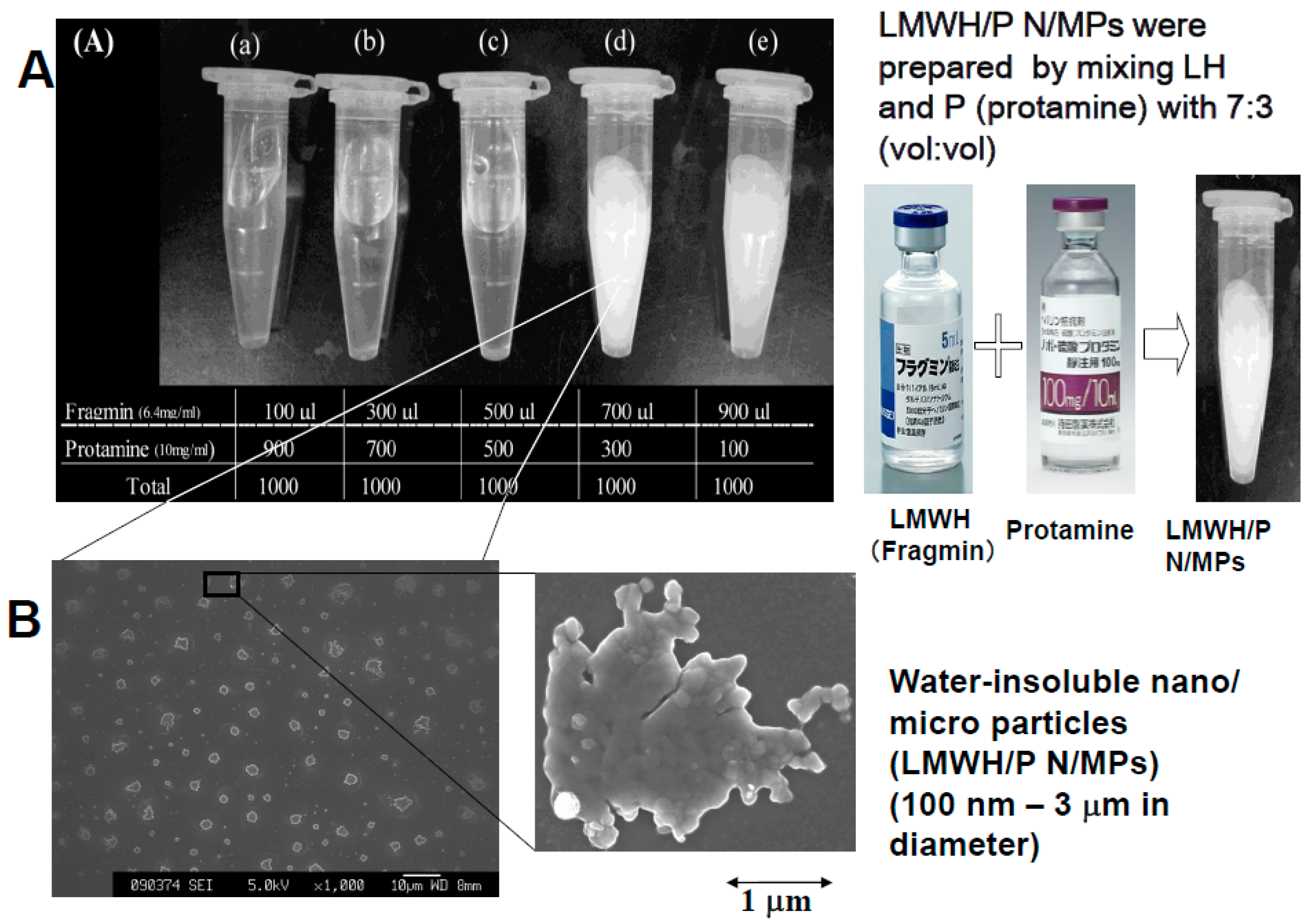
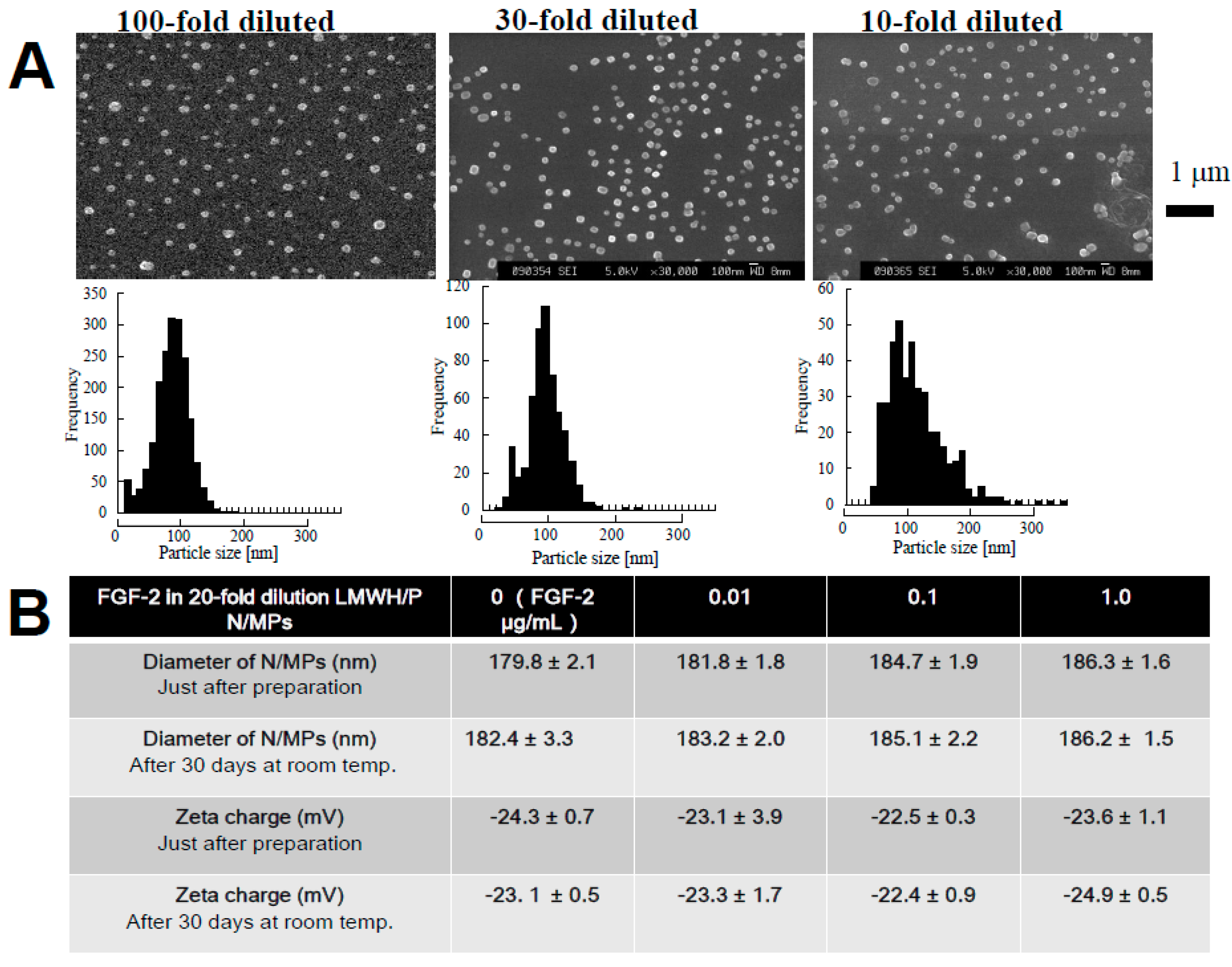
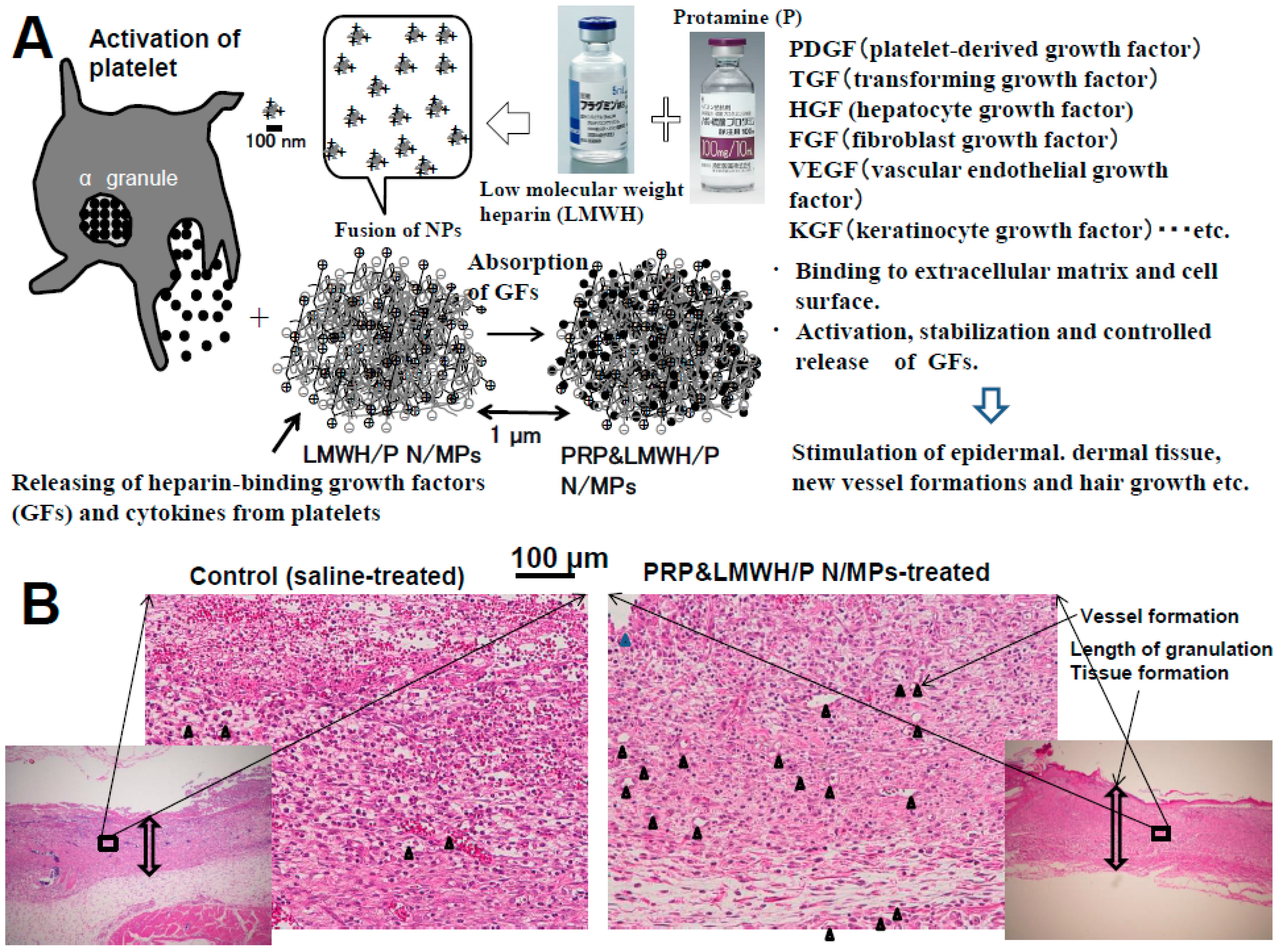
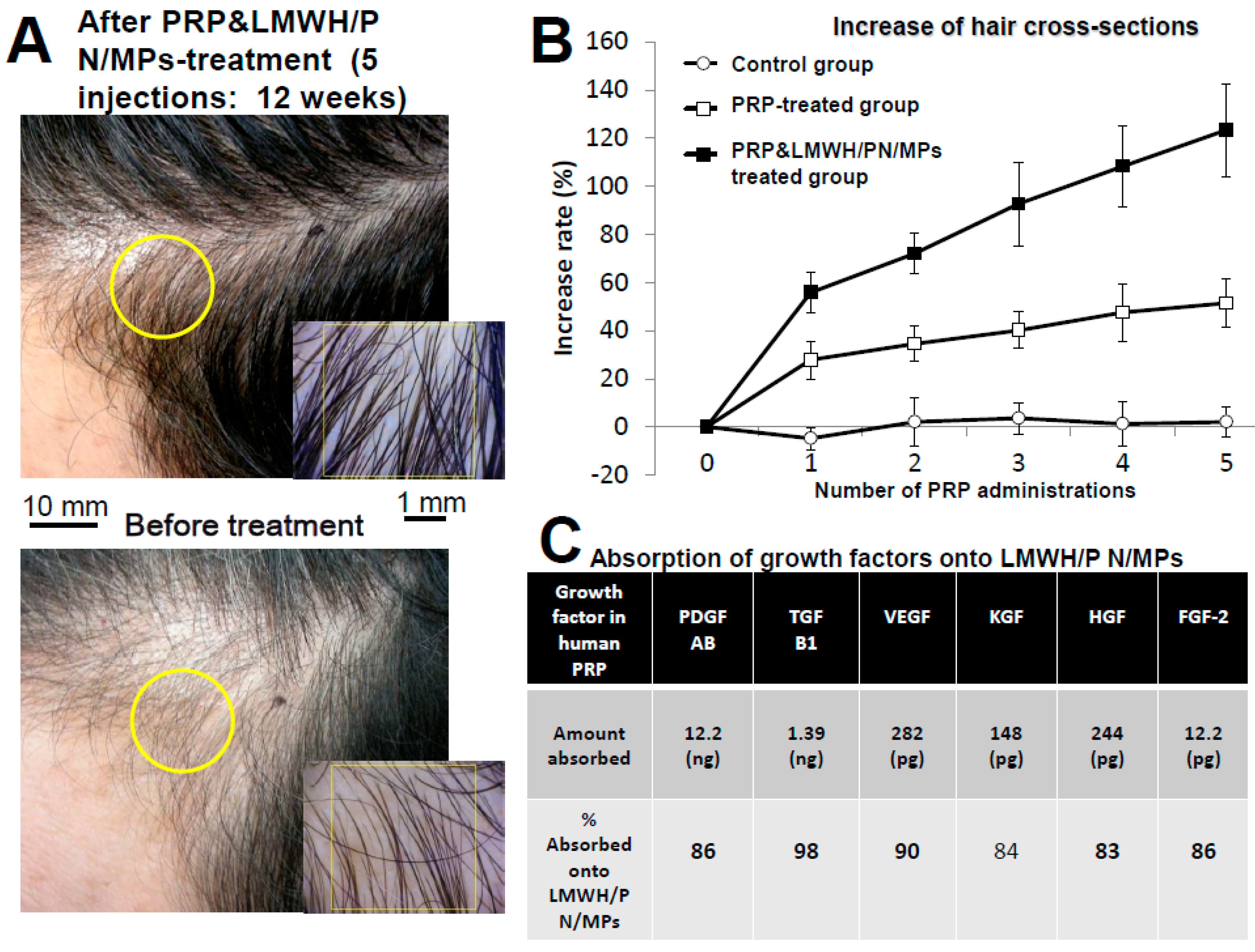
2.2. Preparation of GFs in Platelet-Rich-Plasma (PRP) and LMWH/P N/MPs and Their Application
2.3. LMWH/P N/MPs as ADSCs- and BMSCs-Carriers

2.4. Cell Cultures with Low Serum Using LMWH/P N/MPs-Coated Plates
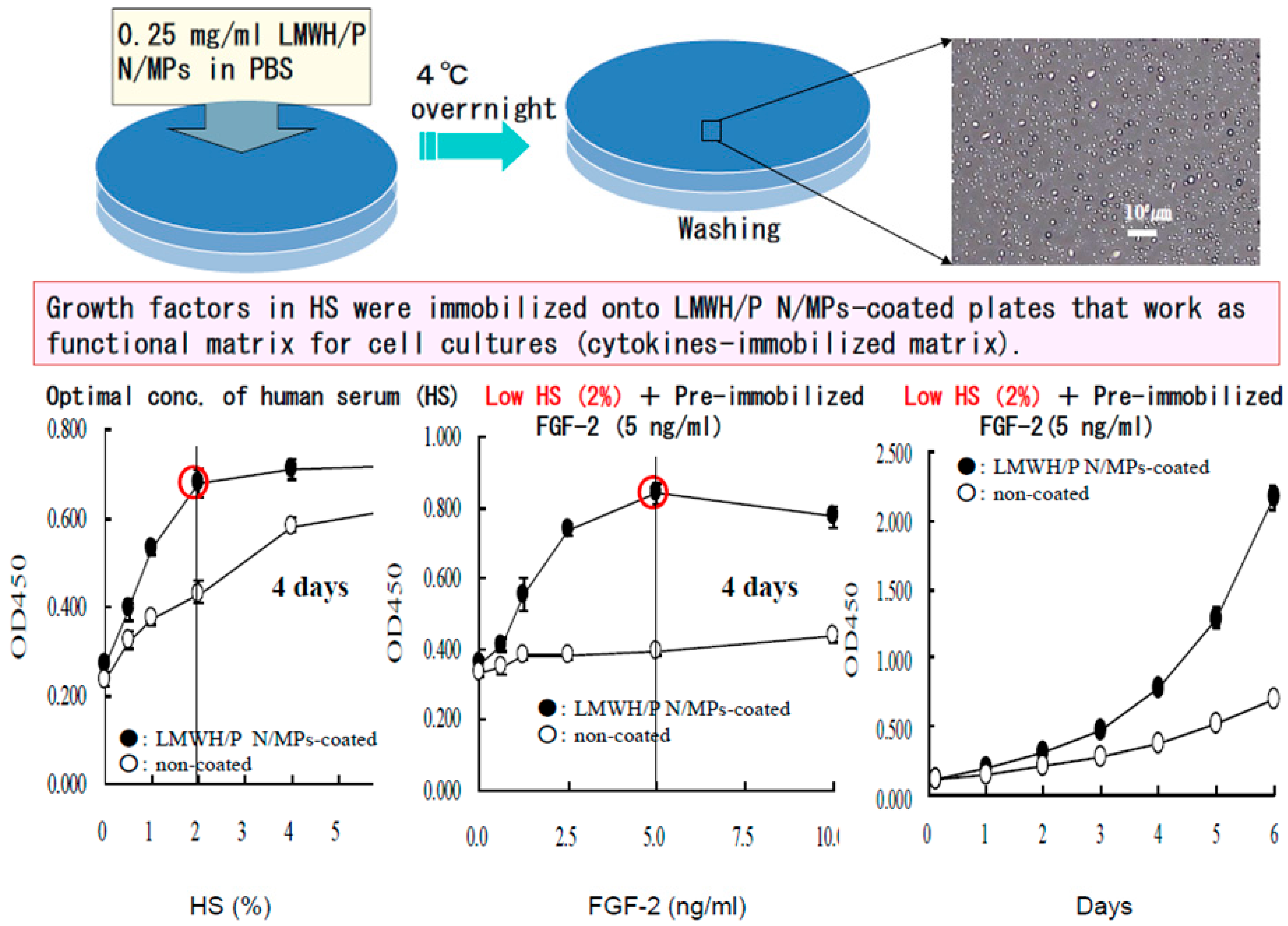
2.5. Proliferation of CD34+ Hematopoietic Progenitor Cells (CD34+ HCs) on LMWH/P N/MPs-Coated Plates
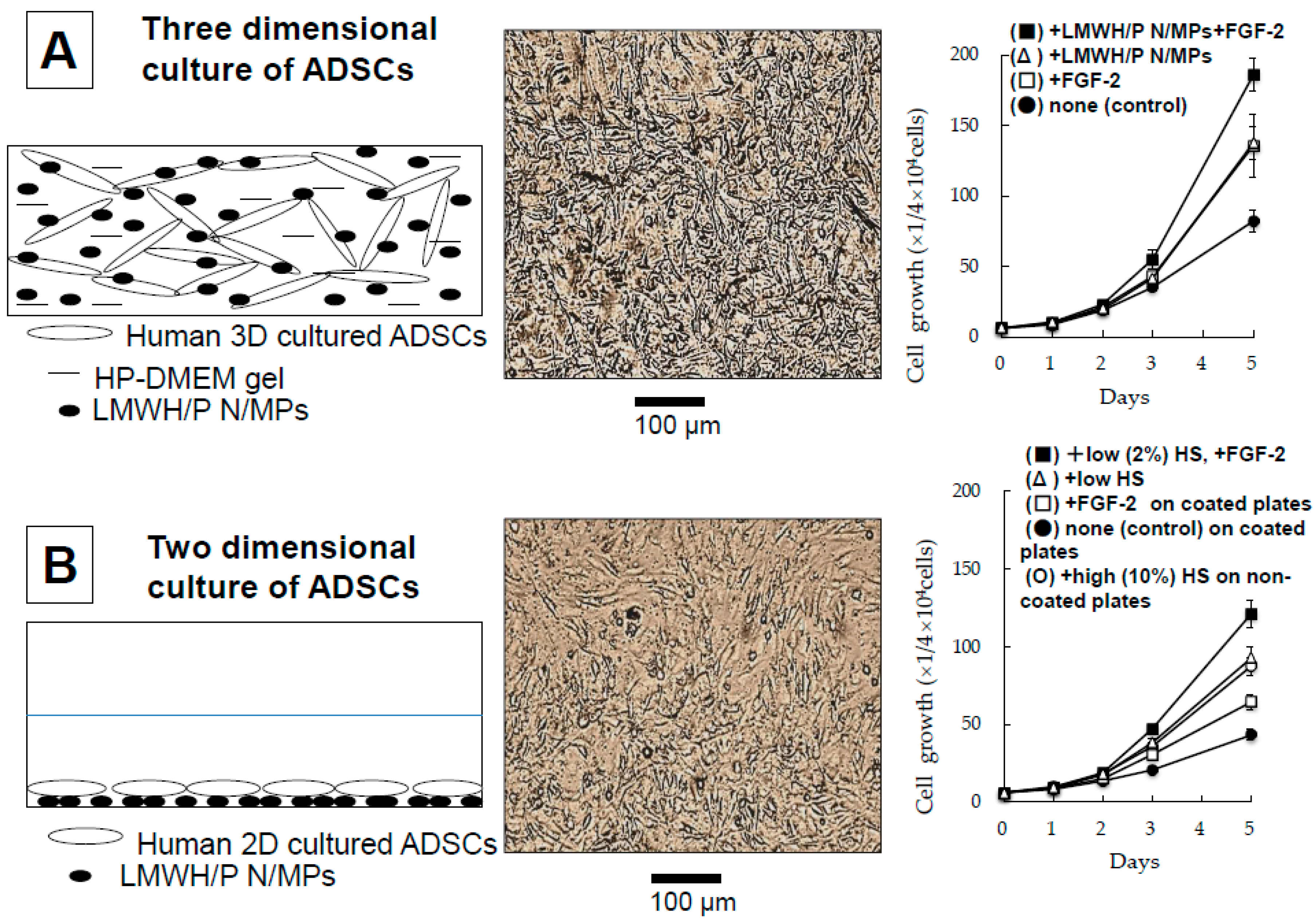
2.6. Three-Dimensional Expansion of ADSCs and BMSCs Using Plasma-Medium Gel with LMWH/P N/MPs
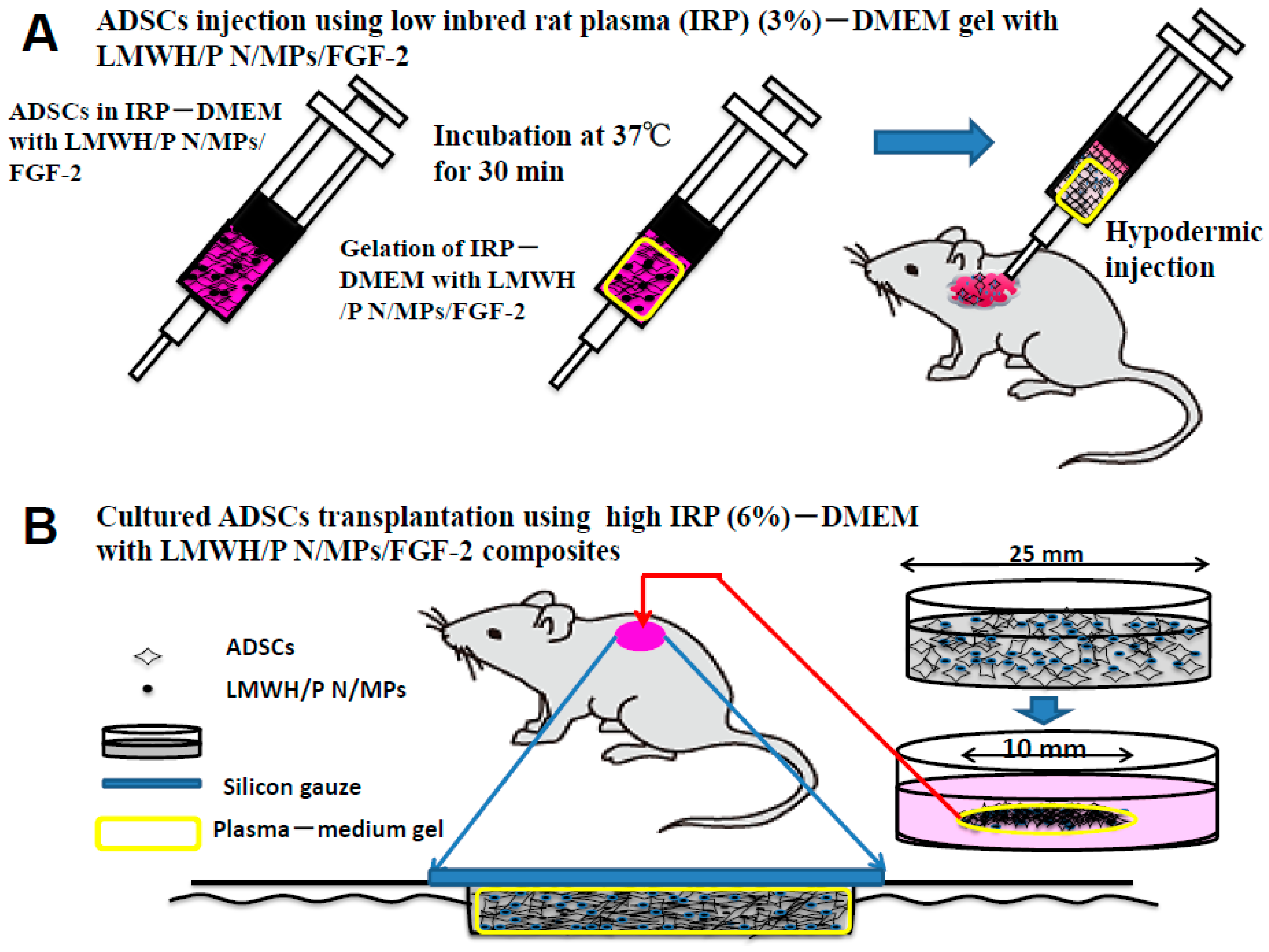
2.7. Transplantation of Inbred Rat Adipose-Derived Stromal Cells (IR-ADSCs) Using Plasma Gel with LMWH/P N/MPs/FGF-2
3. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Nemeno, J.G.E.; Lee, S.; Yang, W.; Lee, K.M.; Lee, J.I.K. Applications and implications of heparin and protamine in tissue engineering and regenerative medicine. Biomed. Res. Int. 2014. [Google Scholar] [CrossRef]
- Kishimoto, S.; Ishihara, M.; Takikawa, M.; Mori, Y.; Hattori, H.; Fujita, M.; Nakamura, S. Novel experimental and clinical therapeutic uses of low-molecular-weight heparin/protamine microparticles. Pharmaceutics 2012, 4, 42–57. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, S.; Kanatani, Y.; Kishimoto, S.; Nambu, M.; Ohno, C.; Hattori, H.; Takase, B.; Tanaka, Y.; Yura, H.; Kiyosawa, T.; et al. Controlled release of FGF-2 using fragmin/protamine microparticles and effect on neovascularization. J. Biomed. Mater. Res. A 2009, 91, 814–823. [Google Scholar] [CrossRef] [PubMed]
- Mori, Y.; Nakamura, S.; Kishimoto, S.; Kawakami, M.; Suzuki, S.; Matsui, T.; Ishihara, M. Preparation and characterization of low-molecular-weight heparin/protamine nanoparticles (LMW-H/P NPs) as FGF-2 carrier. Int. J. Nanomed. 2010, 5, 147–155. [Google Scholar] [CrossRef]
- Kishimoto, S.; Ishihara, M.; Nakamura, S.; Takikawa, M.; Fujita, M.; Sumi, Y.; Kiyosawa, T.; Sato, T.; Kanatani, Y. Fragmin/protamine microparticles to absorb and protect HGF and to function as local HGF carrier in vivo. Acta Biomater. 2013, 9, 4763–4770. [Google Scholar] [CrossRef] [PubMed]
- Takikawa, M.; Nakamura, S.I.; Nakamura, S.; Nambu, M.; Ishihara, M.; Fujita, M.; Kishimoto, S.; Doumoto, T.; Yanagibayashi, S.; Azuma, R.; et al. Enhancement of vascularization and granulation tissue formation by growth factors in human platelet-rich plasma-containing fragmin/protamine microparticles. J. Biomed. Mater. Res. B 2011, 97, 373–380. [Google Scholar] [CrossRef]
- Kishimoto, S.; Ishihara, M.; Mori, Y.; Takikawa, M.; Hattori, H.; Nakamura, S.; Sato, T. Effective expansion of human adipose-derived stromal cells and bone marrow-derived mesenchymal stem cells cultured on a fragmin/protamine nanoparticles-coated substratum with human platelet-rich plasma. J. Tissue Eng. Regen. Med. 2012, 7, 955–964. [Google Scholar] [CrossRef] [PubMed]
- Kishimoto, S.; Nakamura, S.; Nakamura, S.I.; Hattori, H.; Oomuma, F.; Kanatani, Y.; Tanaka, Y.; Harada, Y.; Tagawa, M.; Maehara, T.; et al. Cytokine-immobilized microparticle-coated plates for culturing hematopoietic progenitor cells. J. Control. Release 2009, 133, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, S.; Kishimoto, S.; Nakamura, S.I.; Nambu, M.; Fujita, M.; Tanaka, Y.; Mori, Y.; Tagawa, M.; Maehara, T.; Ishihara, M. Fragmin/protamine microparticles as cell carriers to enhance viability of adipose-derived stromal cells and their subsequent effect on in vivo neovascularization. J. Biomed. Mater. Res. 2010, 92, 1614–1622. [Google Scholar]
- Kishimoto, S.; Nakamura, S.; Nakamura, S.I.; Kanatani, Y.; Hattori, H.; Tanaka, Y.; Harada, Y.; Tagawa, M.; Mori, Y.; Maehara, T.; et al. Fragmin/protamine microparticle-coated matrix immobilized cytokines to stimulate various cell proliferations with low serum media. Artif. Org. 2009, 33, 431–438. [Google Scholar] [CrossRef]
- Kishimoto, S.; Ishihara, M.; Takikawa, M.; Takikawa, M.; Sumi, Y.; Nakamura, S.; Fujita, M.; Sato, T.; Kiyosawa, T. Three-dimensional culture using human plasma-medium gel with fragmin/protamine microparticles for proliferation of various human cells. Cytotechnology 2014, 66, 791–802. [Google Scholar] [CrossRef] [PubMed]
- Kishimoto, S.; Ishihara, M.; Mori, Y.; Takikawa, M.; Sumi, Y.; Nakamura, S.; Sato, T.; Kiyosawa, T. Three-dimensional expansion using plasma-medium gel with fragmin/protamine nanoparticles and FGF-2 to stimulate adipose-derived stromal cells and bone marrow-derived mesenchymal stem cells. BioRes. Open Access 2012, 1, 314–323. [Google Scholar] [CrossRef] [PubMed]
- Berth, G.; Voigh, A.; Dautzenberg, H.; Donath, E.; Mohwald, H. Polyelectrolyte complex and layer-by layer capsules from chitosan/chitosan sulfate. Biomacromolecules 2002, 3, 579–590. [Google Scholar] [CrossRef] [PubMed]
- Houska, M.; Brynda, E.; Bahata, K. The effect of polyelectrolyte chain length on layer-by layer protein/polyelectrolyte assembly—An experimental study. J. Colloid Interface Sci. 2004, 273, 140–147. [Google Scholar] [CrossRef] [PubMed]
- Sotiropoulou, M.; Bokias, G.; Staikos, G. Water-soluble complexes through coulombic interactions between bovine serum albumin and anionic polyelectrolytes grafted with hydrophilic nonionic side chains. Biomecromolecules 2005, 6, 1835–1838. [Google Scholar] [CrossRef]
- Hagiwara, K.; Kishimoto, S.; Ishihara, M.; Koyama, Y.; Mazda, O.; Sato, T. In vivo gene transfer using pDNA/chitosan/chondroitin sulfate ternary complexes: Influence of chondroitin sulfate on the stability of freeze-dried complexes and transfer gene expression in vivo. J. Gene Med. 2013, 15, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Salmivirta, M.; Lidhold, K.; Lindahl, U. Heparan sulfate: A piece of information. FASEB J. 1996, 10, 1270–1279. [Google Scholar] [PubMed]
- Lindahl, U.; Kjellen, L. Pathophsiology of heparin sulphate: Many diseases, few drug. J. Intern. Med. 2013, 273, 5555–5571. [Google Scholar] [CrossRef]
- Ishihara, M.; Ono, K. Structure and function of heparin and heparan sulfate: Heparinoid library and modification of FGF-activities. Trends Glycosci. Glycotechnol. 1998, 10, 223–233. [Google Scholar] [CrossRef]
- Hirsh, J.; Warkentin, T.E.; Shaughnessy, S.G.; Anand, S.S.; Halperin, J.L.; Raschke, R.; Granger, C. Heparin and low-molecular heparin, mechanisms of action, pharmacokinetics, dosing, monitoring, efficacy, and safety. Chest 2001, 119, 64–94. [Google Scholar] [CrossRef]
- Wolzt, M.; Wetermann, A.; Nieszpaur-Los, M.; Schneider, B.; Fassolt, A.; Lechner, K.; Eichler, H.; Kyrle, P.A. Studies on the neutralizing effects of protamine on unfractionated and low molecular weight heparin (Fragmin®) at the site of activation of the coagulation system in man. Thromb. Haemost. 1995, 73, 439–443. [Google Scholar] [PubMed]
- Pan, M.; Lezo, J.S.; Medina, A.; Romero, M.; Hernandez, E.; Segura, J.; Melian, F.; Wanguemert, F.; Landin, M.; Benitez, F.; et al. In-laboratory removal of femoral sheath following protamine administration in patients having intracoronary stent implantation. Am. J. Cardiol. 1997, 80, 1336–1338. [Google Scholar] [CrossRef] [PubMed]
- Marx, R.E. Platelet-rich plasma (PRP): What is PRP and what is not PRP? Implant. Dent. 2001, 10, 225–228. [Google Scholar] [CrossRef] [PubMed]
- Bhanot, S.; Alex, J.C. Current applications of platelet gels in facial plastic surgery. Facial Plast. Surg. 2002, 18, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, S.I.; Ishihara, M.; Takikawa, M.; Murakami, K.; Kishimoto, S.; Nakamura, S.; Yanagibayashi, S.; Mori, Y.; Fujita, M.; Kubo, S.; et al. Increased survival of free fat grafts and vascularization in rats with local delivery of fragmin/protamine microparticles containing FGF-2 (F/P MP-F). J. Biomed. Mater. Res. B 2011, 96, 234–241. [Google Scholar] [CrossRef]
- Takikawa, M.; Sumi, Y.; Ishihara, M.; Kishimoto, S.; Nakamura, S.; Yanagibayashi, S.; Hattori, H.; Azuma, R.; Yamamoto, N.; Kiyosawa, T. PRP&F/P MPs improved survival of dorsal paired pedicle skin flaps in rats. J. Surg. Res. 2011, 170, e189–e196. [Google Scholar] [CrossRef] [PubMed]
- Takikawa, M.; Nakamura, S.I.; Nakamura, S.; Nambu, M.; Ishihara, M.; Murakami, K.; Kishimoto, S.; Sasaki, K.; Yanagishita, S.; Azuma, R.; et al. Enhanced effect of platelet-rich plasma containing a new carrier on hair growth. Dermatol. Surg. 2011, 37, 1721–1729. [Google Scholar] [CrossRef] [PubMed]
- Pittenger, M.F.; Mackay, A.M.; Beck, S.C.; Jaiswal, R.K.; Douglas, R.; Mosca, J.D.; Moorman, M.A.; Simonetti, D.W.; Craig, S.; Marshak, D.R. Multilineage potential of adult human mesenchymal stem cells. Science 1999, 284, 143–147. [Google Scholar] [CrossRef] [PubMed]
- Prockop, D.J. Marrow stromal cells as stem cells for nonhematopoietic tissues. Science 1997, 276, 71–74. [Google Scholar] [CrossRef] [PubMed]
- Zuk, P.A.; Zhu, M.; Mizuno, H.; Huang, J.; Futrell, J.W.; Katz, A.J.; Benhaim, P.; Lorenz, H.P.; Hedrick, M.H. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 2001, 7, 211–228. [Google Scholar] [CrossRef] [PubMed]
- Rickard, D.J.; Kassem, M.; Hefferan, T.E.; Sarkar, G.; Spelsberg, T.C.; Riggs, B.L. Isolation and characterization of osteoblast precurcor cells from bone marrow. J. Bone Miner. Res. 1996, 11, 312–324. [Google Scholar] [CrossRef] [PubMed]
- Hattori, H.; Sato, M.; Masuoka, K.; Ishihara, M.; Kikuchi, T.; Matui, T.; Takase, B.; Ishizuka, T.; Kikuchi, M.; Fujikawa, K.; et al. Osteogenic potential of human adipose tissue-derived stromal cells as alternative stem cell source. Cells Tissues Organs 2004, 178, 2–12. [Google Scholar] [CrossRef] [PubMed]
- Johnstone, B.; Hering, T.M.; Caplan, A.I.; Goldberg, V.M.; Yoo, J.U. In vitro chondrogenesis of bone marrow-derived mesenchymal progenitor cells. Exp. Cell Res. 1998, 238, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Masuoka, K.; Asazuma, T.; Hattori, H.; Yoshihara, Y.; Sato, M.; Matsumura, K.; Matsui, T.; Takase, B.; Nemoto, K.; Ishihara, M. Tissue engineering of articular cartilage with autologous cultured adipose tissue-derived stromal cells using atelocollagen honeycomb-shaped scaffold with membrane sealing in rabbits. J. Biomed. Mater. Res. B 2006, 79, 25–34. [Google Scholar] [CrossRef]
- Beresford, J.N.; Bennett, J.H.; Devlin, C.; Leboy, P.S.; Owen, M.E. Evidence for an inverse relationship between the differentiation of adipocytic and osteogenic cells in rat marrow stromal cell cultures. J. Cell Sci. 1992, 102, 341–351. [Google Scholar] [PubMed]
- Wakitani, S.; Saito, T.; Caplan, A.I. Myogenic cells derived from rat bone marrow mesenchymal stem cells exposed to 5-azacytidine. Muscle Nerve 1995, 18, 1417–1426. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, G.; Cusella-DeAngelis, G.; Coletta, M.; Palucci, E.; Stomaiuolo, A.; Cossu, G.; Mavilio, F. Muscle regeneration by bone marrow-derived myogenic progenitors. Science 1998, 279, 1528–1530. [Google Scholar] [CrossRef] [PubMed]
- Woodbury, D.; Reynoldsk, K.; Black, I.B. Adult bone marrow stromal stem cells express germline, ectodermal, endodermal, and mesodermal genes prior to neurogenesis. J. Neurosci. Res. 2002, 96, 908–917. [Google Scholar] [CrossRef]
- Ashjian, P.H.; Elbarbary, A.S.; Edmonds, B.; DeUgarte, D.; Zhu, M.; Zuk, P.A.; Lorenz, H.P.; Benhaim, P.; Hedrick, M.H. In vitro differentiation of human processed lipoaspirate cells into early neural progenitors. Plast. Reconstr. Surg. 2003, 111, 1922–1931. [Google Scholar] [CrossRef] [PubMed]
- Nambu, M.; Ishihara, M.; Nakamura, S.; Mizuno, H.; Yanagibayashi, S.; Kanatani, Y.; Hattori, H.; Kiyosawa, T. Enhanced healing of mitomycin C-treated wounds in rats using inbred adipose tissue-derived stromal cells within an atelocollagen matrix. Wound Rep. Regen. 2007, 15, 505–510. [Google Scholar] [CrossRef]
- Wang, H.J.; Pieper, J.; Schotel, R.; van Blitterswijk, C.A.; Lamme, E.N. Stimulation of skin repair is dependent on fibroblast source and presence of extracellular matrix. Tissue Eng. 2004, 10, 1054–1064. [Google Scholar] [CrossRef] [PubMed]
- Sart, S.; Ma, T.; Li, Y. Preconditioning stem cells for in vivo delivery. BioRes. Open Access 2014, 3, 137–149. [Google Scholar] [CrossRef] [PubMed]
- De-Almeida, P.E.; Ransohoff, J.D.; Nahid, A.; Wu, J.C. Immunogenicity of pluripotent stem cells and their derivatives. Circ. Res. 2013, 112, 549–561. [Google Scholar] [CrossRef] [PubMed]
- Nambu, M.; Ishihara, M.; Kishimoto, S.; Yanagibayashi, S.; Yamamoto, N.; Azuma, R.; Kanatani, Y.; Kiyosawa, T.; Mizuno, H. Stimulatory effect of autologous adipose tissue-derived stromal cells in an atelocollagen matrix on wound healing in diabetic db/db mice. J. Tissue Eng. 2011, 2, 158105. [Google Scholar]
- Kumano, I.; Kishimoto, S.; Nakamura, S.; Hattori, H.; Tanaka, Y.; Nakata, M.; Sato, T.; Fujita, M.; Maehara, T.; Ishihara, M. Fragmin/protamine microparticles (F/P MPs) as cell carriers enhance the formation and growth of tumors in vivo. Cell Mol. BioEng. 2011, 4, 476–483. [Google Scholar] [CrossRef]
- Volpe, J.P.; Milasm, L. Influence of tumor transplantation methods on tumor growth rate and metastatic potential of solitary tumors derived metastasis. Clin. Exp. Metasitasis 1990, 8, 381–389. [Google Scholar] [CrossRef]
- Spees, J.L.; Gregorym, C.A.; Singhm, H.; Tucker, H.A.; Peister, A.; Lynch, P.J.; Hsu, S.C.; Smith, J.; Prockop, D.J. Internalized antigens must be removed to prepare hypoimmunogenic mesenchymal stem cells for cell and gene therapy. Mol. Ther. 2004, 9, 747–756. [Google Scholar] [CrossRef] [PubMed]
- Martin, M.J.; Muotri, A.; Gage, F.; Varki, A. Human embryonic stem cells express an immunogenic nonhuman sialic acid. Nat. Med. 2005, 11, 228–232. [Google Scholar] [CrossRef] [PubMed]
- Kishimoto, S.; Hattori, H.; Nakamura, S.; Amano, Y.; Kanatani, Y.; Tanaka, Y.; Mori, Y.; Harada, Y.; Tagawa, M.; Ishihara, M. Expansion and characterization of human bone marrow-derived mesenchymal stem cells cultured on fragmin/protamine microparticle-coated matrix with fibroblast growth factor-2 in low serum medium. Tissue Eng. C 2009, 15, 523–527. [Google Scholar] [CrossRef]
- Kishimoto, S.; Ishihara, M.; Kanatani, Y.; Nambu, M.; Takikawa, M.; Sumi, Y.; Nakamura, S.; Hattori, H.; Sato, T. Selective expansion of CD34+ cells from mouse bone marrow cultured on LH/P MP-coated plates with adequate cytokines. J. Tissue Eng. 2011, 2. [Google Scholar] [CrossRef]
- Gupta, P.; Oegema, T.R.; Brazilm, J.J.; Dudekm, A.Z.; Slungaard, A.; Verfaillie, C.M. Structurally specific heparan sulfates support primitive human hematopoiesis by formation of a multimolecular stem cell niche. Blood 1998, 92, 4641–4651. [Google Scholar] [PubMed]
- Alvarez-Silva, M.; Borojevic, R. GM-CSF and IL-3 activities in schistosomal liver granulomas are controlled by stroma-associated heparan sulfate proteoglycans. J. Leukoc. Biol. 1996, 59, 435–441. [Google Scholar] [PubMed]
- Roberts, R.; Gallagher, J.; Spooncer, E.; Allen, T.D.; Bloomfield, F.; Dexter, T.M. Heparan sulfate bound growth factors: A mechanism for stromal cell mediated haemopoiesis. Nature 1988, 332, 376–378. [Google Scholar] [CrossRef] [PubMed]
- Gordon, M.Y.; Riley, G.P.; Watt, S.M.; Greaves, M.F. Compartmentalization of a haematopoietic growth factor (GM-CSF) by glycosaminoglycans in the bone marrow microenvironment. Nature 1987, 326, 403–405. [Google Scholar] [CrossRef] [PubMed]
- Drayer, A.L.; Olthof, S.G.; Vellengam, E. Mammalian target of rapamycin is required for thrombopoietin-induced proliferation of megakaryocyte progenitors. Stem Cells 2006, 24, 105–114. [Google Scholar] [CrossRef] [PubMed]
- Schepers, H.; Wierenga, A.T.; Gasligam, D.V.; Eggen, B.J.; Vellenga, E.; Schuringa, J.J. Reintroduction of C/EBPα in leukemic CD34+ stem/progenitor cells impairs self-renewal and partially restores myelopoiesis. Blood 2007, 110, 1317–1325. [Google Scholar] [CrossRef] [PubMed]
- Rokstad, A.M.; Donati, I.; Borgogna, M.; Oberholzer, J.; Strand, B.L.; Espevikm, T.; Skjak-Braek, G. Cell-compatible covalently reinforced beads obtained from a chemoenzymatically engineered alginate. Biomaterials 2006, 27, 4726–4737. [Google Scholar] [CrossRef] [PubMed]
- Sumi, Y.; Ishihara, M.; Kishimoto, S.; Takikawa, M.; Doumoto, T.; Azuma, R.; Nakamura, S.; Hattori, H.; Fujita, M.; Kiyosawa, T. Transplantation of inbred adipose-derived stromal cells in rats with plasma gel containing fragmin/protamine microparticles and FGF-2. J. Biomed. Mater. Res. B 2013, 101, 78–791. [Google Scholar]
- Sumi, Y.; Ishihara, M.; Kishimoto, S.; Takikawa, M.; Hattori, H.; Takikawa, M.; Doumoto, T.; Azuma, R.; Nakamura, S.; Fujita, M.; et al. Effective wound healing in streptozotocin-induced diabetic rats by adipose-derived stromal cell-transplantation in plasma-gel containing fragmin/protamine microparticles. Ann. Plast. Surg. 2014, 72, 113–120. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ishihara, M.; Kishimoto, S.; Takikawa, M.; Hattori, H.; Nakamura, S.; Shimizu, M. Biomedical Application of Low Molecular Weight Heparin/Protamine Nano/Micro Particles as Cell- and Growth Factor-Carriers and Coating Matrix. Int. J. Mol. Sci. 2015, 16, 11785-11803. https://doi.org/10.3390/ijms160511785
Ishihara M, Kishimoto S, Takikawa M, Hattori H, Nakamura S, Shimizu M. Biomedical Application of Low Molecular Weight Heparin/Protamine Nano/Micro Particles as Cell- and Growth Factor-Carriers and Coating Matrix. International Journal of Molecular Sciences. 2015; 16(5):11785-11803. https://doi.org/10.3390/ijms160511785
Chicago/Turabian StyleIshihara, Masayuki, Satoko Kishimoto, Makoto Takikawa, Hidemi Hattori, Shingo Nakamura, and Masafumi Shimizu. 2015. "Biomedical Application of Low Molecular Weight Heparin/Protamine Nano/Micro Particles as Cell- and Growth Factor-Carriers and Coating Matrix" International Journal of Molecular Sciences 16, no. 5: 11785-11803. https://doi.org/10.3390/ijms160511785
APA StyleIshihara, M., Kishimoto, S., Takikawa, M., Hattori, H., Nakamura, S., & Shimizu, M. (2015). Biomedical Application of Low Molecular Weight Heparin/Protamine Nano/Micro Particles as Cell- and Growth Factor-Carriers and Coating Matrix. International Journal of Molecular Sciences, 16(5), 11785-11803. https://doi.org/10.3390/ijms160511785





