Microwave-Assisted Synthesis of Glutathione-Capped CdTe/CdSe Near-Infrared Quantum Dots for Cell Imaging
Abstract
:1. Introduction
2. Results and Discussion
2.1. Characterization of Glutathione (GSH)-Stabilized CdTe/CdSe Quantum Dots (QDs)
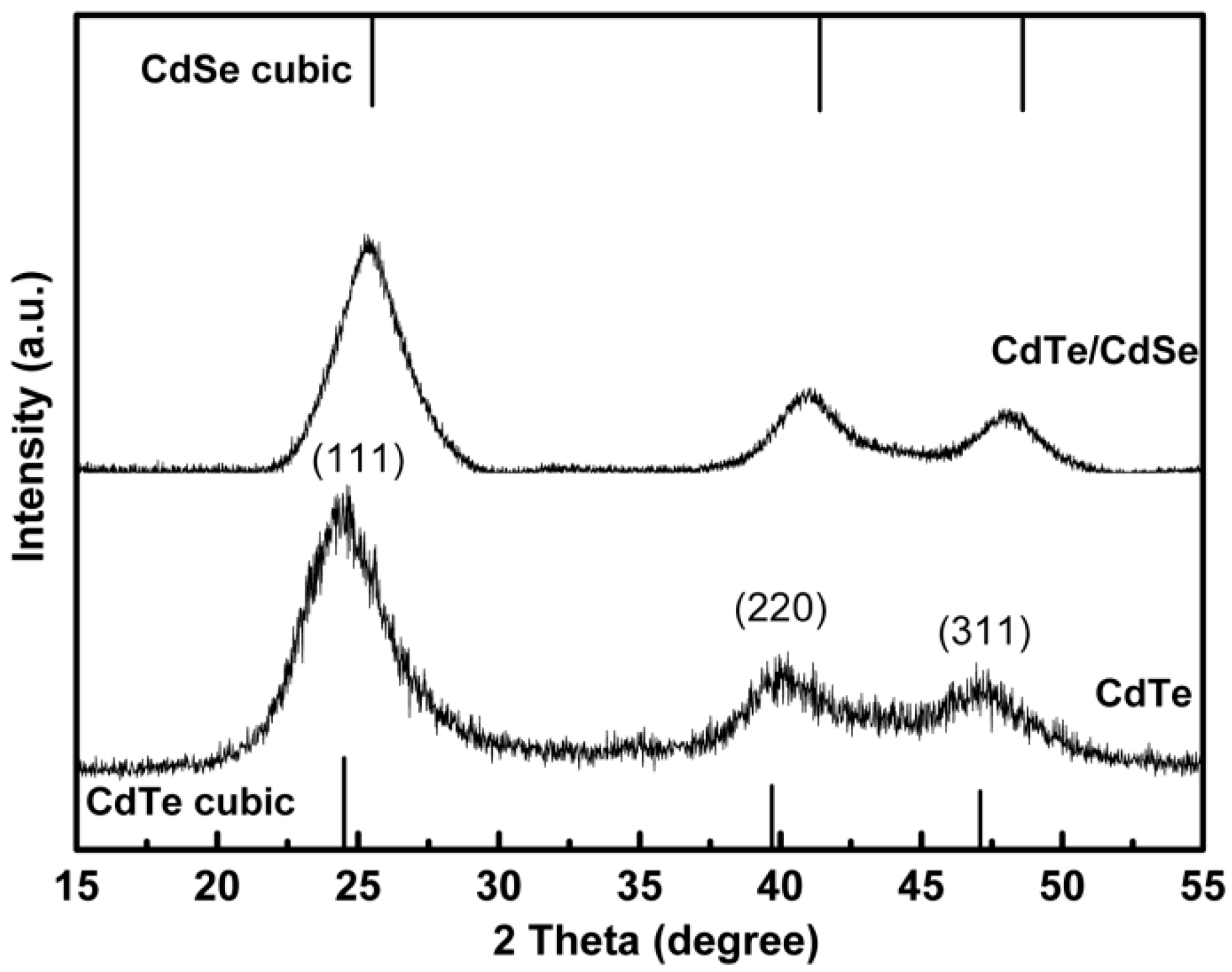
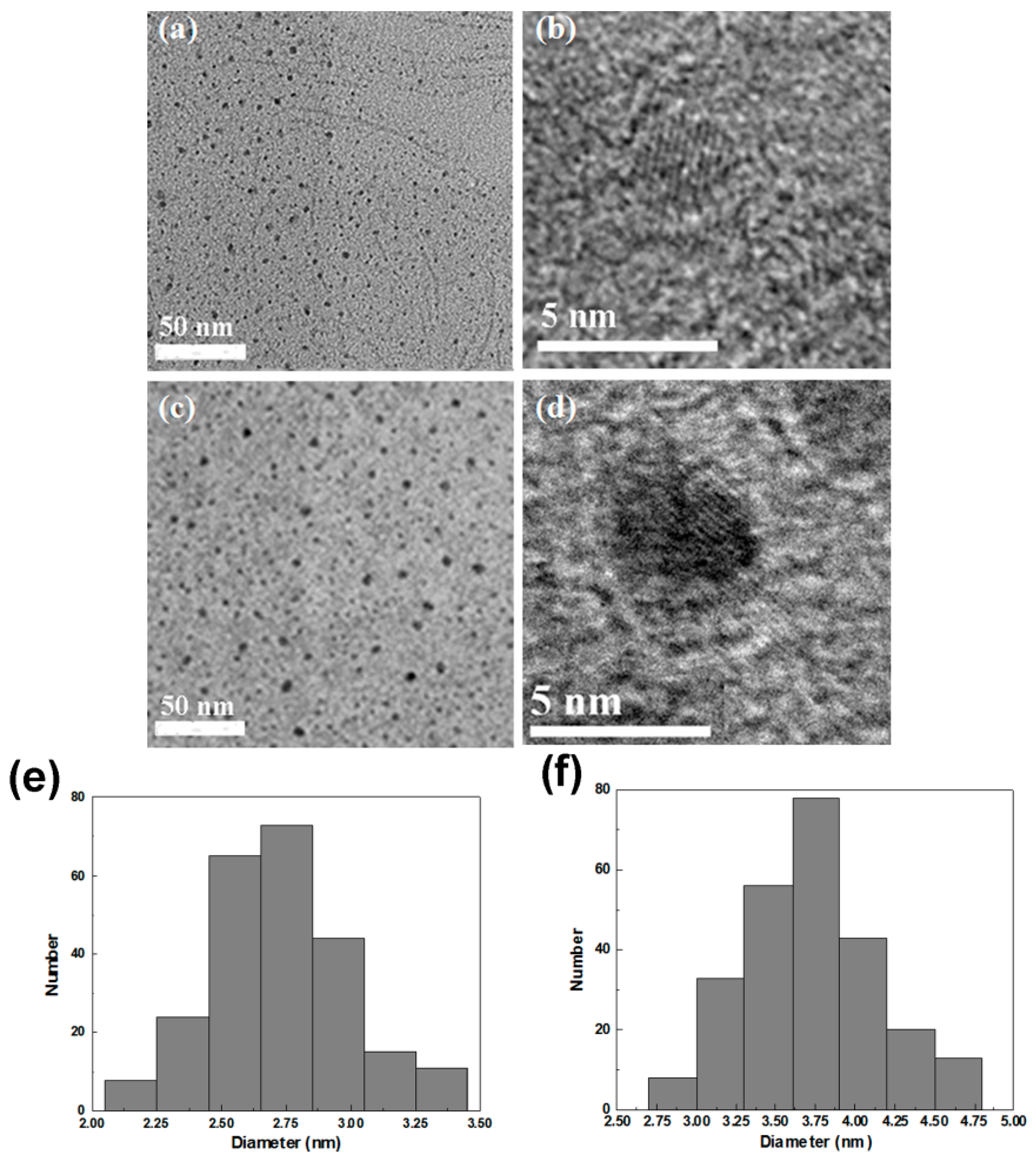
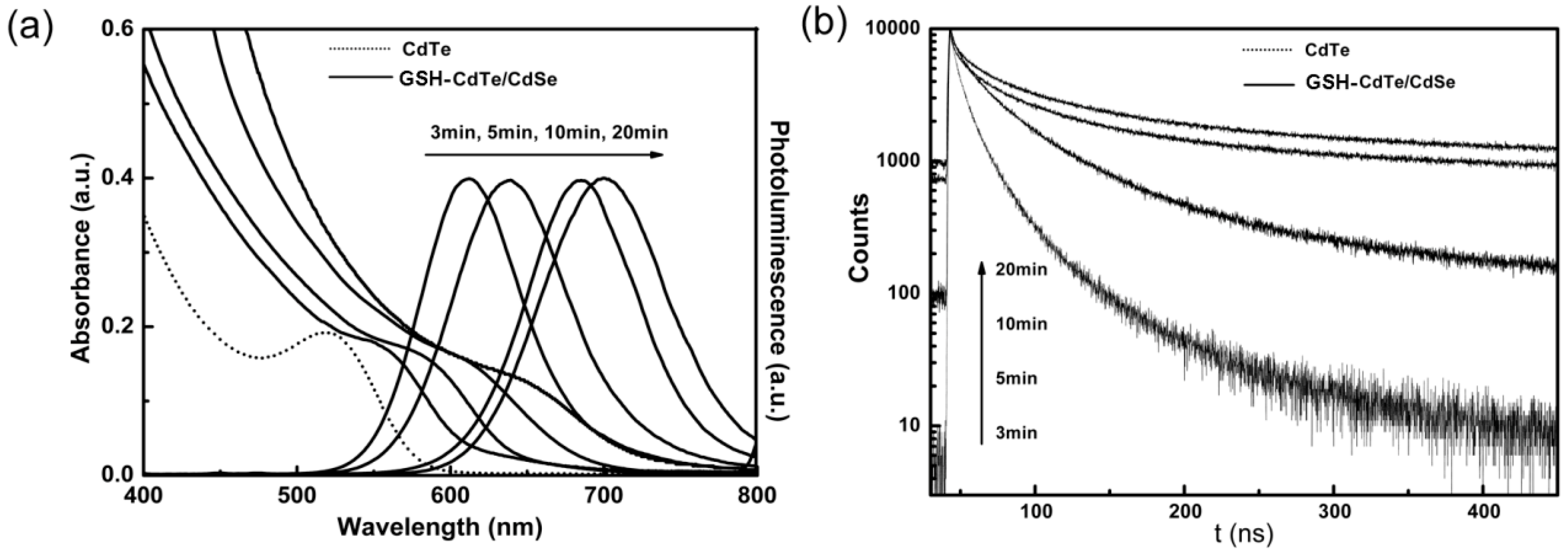
2.2. The Biocompatibility and Cell Imaging of GSH-CdTe/CdSe QDs
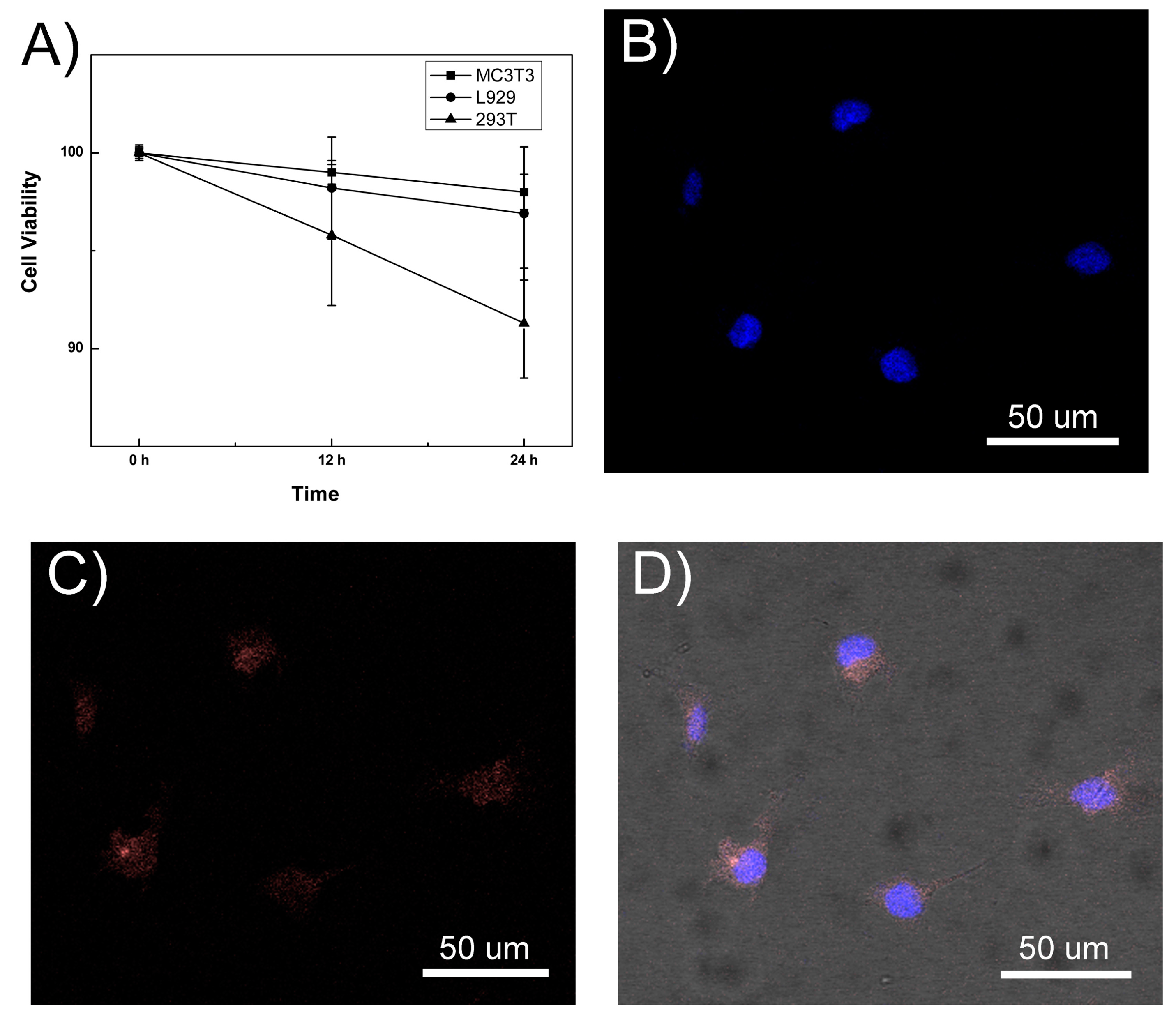
3. Experimental Section
3.1. Preparation of CdTe/CdSe Core/Shell Quantum Dots (QDs)
3.2. The Calculation of Photo Luminescence Quantum Yield (PLQY) and Photo Luminescence (PL) Lifetime
3.3. Cell Viability and Proliferation Assays
3.4. Preparation of RGD Conjugated GSH-CdTe/CdSe QDs and Cell Imaging
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Algar, W.R.; Susumu, K.; Delehanty, J.B.; Medintz, I.L. Semiconductor quantum dots in bioanalysis: Crossing the valley of death. Anal. Chem. 2011, 83, 8826–8837. [Google Scholar] [CrossRef] [PubMed]
- Aldeek, F.; Balan, L.; Medjahdi, G.; Roques-Carmes, T.; Malval, J.-P.; Mustin, C.; Ghanbaja, J.; Schneider, R. Enhanced optical properties of core/shell/shell CdTe/CdS/ZnO quantum dots prepared in aqueous solution. J. Phys. Chem. C 2009, 113, 19458–19467. [Google Scholar] [CrossRef]
- Zhong, X.; Feng, Y.; Knoll, W.; Han, M. Alloyed ZnxCd1−xS nanocrystals with highly narrow luminescence spectral width. J. Am. Chem. Soc. 2003, 125, 13559–13563. [Google Scholar] [CrossRef] [PubMed]
- Bailey, R.E.; Nie, S. Alloyed semiconductor quantum dots: Tuning the optical properties without changing the particle size. J. Am. Chem. Soc. 2003, 125, 7100–7106. [Google Scholar] [CrossRef] [PubMed]
- Hines, M.A.; Scholes, G.D. Colloidal PbS nanocrystals with size-tunable near-infrared emission: Observation of post-synthesis self-narrowing of the particle size distribution. Adv. Mater. 2003, 15, 1844–1849. [Google Scholar] [CrossRef]
- Kim, S.; Fisher, B.; Eisler, H.-J.; Bawendi, M. Type-II quantum dots: CdTe/CdSe (core/shell) and CdSe/ZnTe (core/shell) heterostructures. J. Am. Chem. Soc. 2003, 125, 11466–11467. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; He, Z.; Chan, W.; Choi, M.M. Synthesis and characterization of high-quality water-soluble near-infrared-emitting CdTe/CdS quantum dots capped by N-acetyl-l-cysteine via hydrothermal method. J. Phys. Chem. C 2009, 113, 1293–1300. [Google Scholar] [CrossRef]
- Chen, H.; Wang, Y.; Xu, J.; Ji, J.; Zhang, J.; Hu, Y.; Gu, Y. Non-invasive near-infrared fluorescence imaging of CdHgTe quantum dots in mouse model. J. Fluoresc. 2008, 18, 801–811. [Google Scholar] [CrossRef] [PubMed]
- Harrison, M.; Kershaw, S.; Burt, M.; Eychmüller, A.; Weller, H.; Rogach, A. Wet chemical synthesis and spectroscopic study of CdHgTe nanocrystals with strong near-infrared luminescence. Mater. Sci. Eng. B 2000, 69, 355–360. [Google Scholar] [CrossRef]
- Rogach, A.L.; Eychmüller, A.; Hickey, S.G.; Kershaw, S.V. Infrared-emitting colloidal nanocrystals: Synthesis, assembly, spectroscopy, and applications. Small 2007, 3, 536–557. [Google Scholar] [CrossRef] [PubMed]
- Mao, W.; Guo, J.; Yang, W.; Wang, C.; He, J.; Chen, J. Synthesis of high-quality near-infrared-emitting CdTeS alloyed quantum dots via the hydrothermal method. Nanotechnology 2007, 18, 485611. [Google Scholar] [CrossRef]
- Liang, G.-X.; Gu, M.-M.; Zhang, J.-R.; Zhu, J.-J. Preparation and bioapplication of high-quality, water-soluble, biocompatible, and near-infrared-emitting CdSeTe alloyed quantum dots. Nanotechnology 2009, 20, 415103. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Zhang, H.; Qiao, Y.; Su, X. One-pot synthesis of ternary CuInS2 quantum dots with near-infrared fluorescence in aqueous solution. RSC Adv. 2012, 2, 819–825. [Google Scholar] [CrossRef]
- Kim, S.; Lim, Y.T.; Soltesz, E.G.; de Grand, A.M.; Lee, J.; Nakayama, A.; Parker, J.A.; Mihaljevic, T.; Laurence, R.G.; Dor, D.M. Near-infrared fluorescent type II quantum dots for sentinel lymph node mapping. Nat. Biotechnol. 2003, 22, 93–97. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Qin, D.; Du, X.; Zhang, L.; Zhao, G.; Zhang, Q.; Wu, J. Aqueous synthesis and characterization of bovine hemoglobin-conjugated cadmium sulfide nanocrystals. J. Alloys Compd. 2014, 604, 181–187. [Google Scholar] [CrossRef]
- Kong, Y.; Chen, J.; Gao, F.; Brydson, R.; Johnson, B.; Heath, G.; Zhang, Y.; Wu, L.; Zhou, D. Near-infrared fluorescent ribonuclease-A-encapsulated gold nanoclusters: Preparation, characterization, cancer targeting and imaging. Nanoscale 2013, 5, 1009–1017. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Zhang, T.; Feng, L.; Zhang, X.; Zhang, M.; Cui, D. Synthesis of ribonuclease A-conjugated CdS quantum dots and its photocatalytic properties. Micro Nano Lett. 2012, 7, 1023–1025. [Google Scholar] [CrossRef]
- Samanta, A.; Deng, Z.; Liu, Y. Aqueous synthesis of glutathione-capped CdTe/CdS/ZnS and CdTe/CdSe/ZnS core/shell/shell nanocrystal heterostructures. Langmuir 2012, 28, 8205–8215. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Liu, S.; He, Y. Fluorescence quenching investigation on the interaction of glutathione-CdTe/CdS quantum dots with sanguinarine and its analytical application. Luminescence 2014, 29, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Gui, R.; An, X. Layer-by-layer aqueous synthesis, characterization and fluorescence properties of type-II CdTe/CdS core/shell quantum dots with near-infrared emission. RSC Adv. 2014, 3, 20959–20969. [Google Scholar] [CrossRef]
- Jing, L.; Kershaw, S.V.; Kipp, T.; Kalytchuk, S.; Ding, K.; Zeng, J.; Jiao, M.; Sun, X.; Mews, A.; Rogach, A.L.; et al. Insight into strain effects on band alignment shifts, carrier localization and recombination kinetics in CdTe/CdS core/shell quantum dots. J. Am. Chem. Soc. 2015, 137, 2073–2084. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Sun, X.; Wang, X.; Xu, H. Synthesis of highly luminescent and biocompatible CdTe/CdS/ZnS quantum dots using microwave irradiation: A comparative study of different ligands. Luminescence 2014, 29, 837–845. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Han, H. One-step synthesis of water-soluble ZnSe quantum dots via microwave irradiation. Mater. Lett. 2010, 64, 1099–1101. [Google Scholar] [CrossRef]
- Du, J.; Li, X.; Wang, S.; Wu, Y.; Hao, X.; Xu, C.; Zhao, X. Microwave-assisted synthesis of highly luminescent glutathione-capped Zn1−xCdxTe alloyed quantum dots with excellent biocompatibility. J. Mater. Chem. 2012, 22, 11390–11395. [Google Scholar] [CrossRef]
- Gaponik, N.; Talapin, D.V.; Rogach, A.L.; Hoppe, K.; Shevchenko, E.V.; Kornowski, A.; Eychmüller, A.; Weller, H. Thiol-capping of CdTe nanocrystals: An alternative to organometallic synthetic routes. J. Phys. Chem. B 2002, 106, 7177–7185. [Google Scholar] [CrossRef]
- Yu, K.; Zaman, B.; Romanova, S.; Wang, D.-S.; Ripmeester, J.A. Sequential synthesis of type II colloidal CdTe/CdSe core-shell nanocrystals. Small 2005, 1, 332–338. [Google Scholar] [CrossRef] [PubMed]
- Laheld, U.; Pedersen, F.; Hemmer, P. Excitons in type-II quantum dots: Finite offsets. Phys. Rev. B 1995, 52, 2697. [Google Scholar] [CrossRef]
- Schöps, O.; le Thomas, N.; Woggon, U.; Artemyev, M. Recombination dynamics of CdTe/CdS core-shell nanocrystals. J. Phys. Chem. B 2006, 110, 2074–2079. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.-Y.; Wang, S.-R.; Yang, C.-H. Synthesis and characterization of CdTe/CdS and CdTe/CdSe core/shell type-II quantum dots in a noncoordinating solvent. Nanotechnology 2007, 18, 345602. [Google Scholar] [CrossRef]
- James, D.R.; Liu, Y.-S.; de Mayo, P.; Ware, W.R. Distributions of fluorescence lifetimes: Consequences for the photophysics of molecules adsorbed on surfaces. Chem. Phys. Lett. 1985, 120, 460–465. [Google Scholar] [CrossRef]
- Ivanov, S.A.; Piryatinski, A.; Nanda, J.; Tretiak, S.; Zavadil, K.R.; Wallace, W.O.; Werder, D.; Klimov, V.I. Type-II core/shell CdS/ZnSe nanocrystals: Synthesis, electronic structures, and spectroscopic properties. J. Am. Chem. Soc. 2007, 129, 11708–11719. [Google Scholar] [CrossRef] [PubMed]
- Li, J.J.; Tsay, J.M.; Michalet, X.; Weiss, S. Wavefunction engineering: From quantum wells to near-infrared type-II colloidal quantum dots synthesized by layer-by-layer colloidal epitaxy. Chem. Phys. 2005, 318, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Qu, L.; Peng, X. Control of photoluminescence properties of CdSe nanocrystals in growth. J. Am. Chem. Soc. 2002, 124, 2049–2055. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, X.; Li, L.; Lai, Y.; Yan, J.; Tang, Y.; Wang, X. Microwave-Assisted Synthesis of Glutathione-Capped CdTe/CdSe Near-Infrared Quantum Dots for Cell Imaging. Int. J. Mol. Sci. 2015, 16, 11500-11508. https://doi.org/10.3390/ijms160511500
Chen X, Li L, Lai Y, Yan J, Tang Y, Wang X. Microwave-Assisted Synthesis of Glutathione-Capped CdTe/CdSe Near-Infrared Quantum Dots for Cell Imaging. International Journal of Molecular Sciences. 2015; 16(5):11500-11508. https://doi.org/10.3390/ijms160511500
Chicago/Turabian StyleChen, Xiaogang, Liang Li, Yongxian Lai, Jianna Yan, Yichen Tang, and Xiuli Wang. 2015. "Microwave-Assisted Synthesis of Glutathione-Capped CdTe/CdSe Near-Infrared Quantum Dots for Cell Imaging" International Journal of Molecular Sciences 16, no. 5: 11500-11508. https://doi.org/10.3390/ijms160511500
APA StyleChen, X., Li, L., Lai, Y., Yan, J., Tang, Y., & Wang, X. (2015). Microwave-Assisted Synthesis of Glutathione-Capped CdTe/CdSe Near-Infrared Quantum Dots for Cell Imaging. International Journal of Molecular Sciences, 16(5), 11500-11508. https://doi.org/10.3390/ijms160511500




