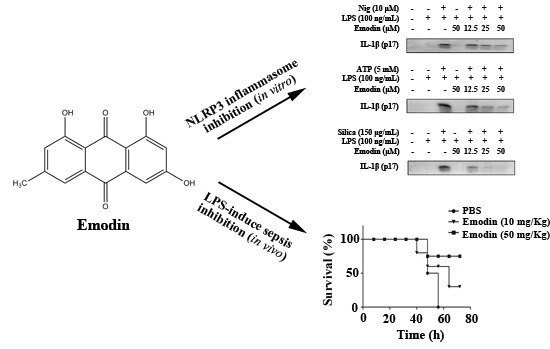
Anti-Inflammatory Effect of Emodin via Attenuation of NLRP3 Inflammasome Activation
Abstract
:1. Introduction
2. Results and Discussion
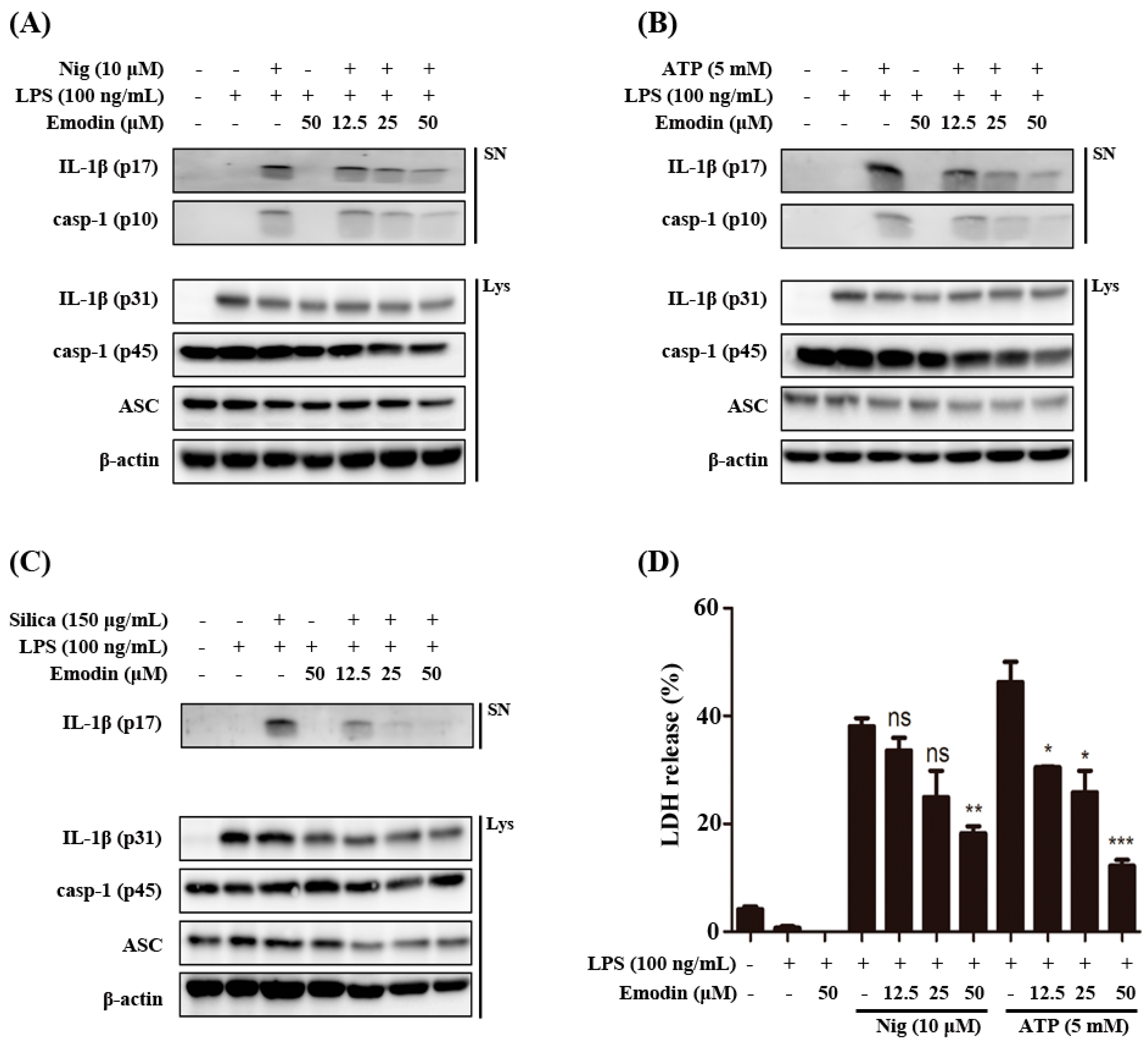
2.1. Effects of Emodin on Inflammasome-Induced Interleukin (IL)-1β Secretion in Vitro
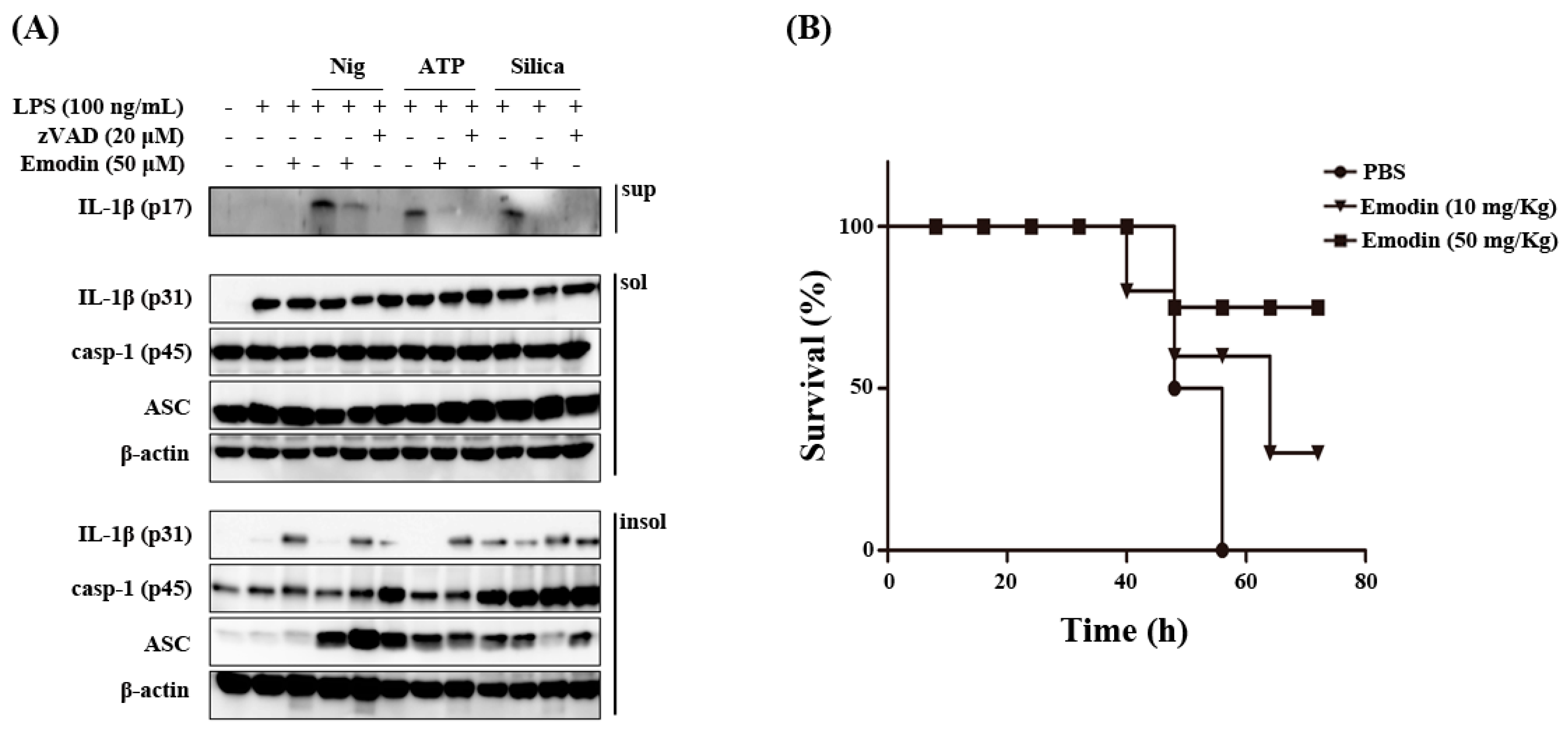
2.2. Effects of Emodin on Inflammasome Component and Sepsis in Vivo Model
3. Experimental Section
3.1. Reagents and Antibodies
3.2. Preparation and Culture of Bone Marrow-Derived Macrophages (BMDMs)
3.3. Animals
3.4. Inflammasome Activation
3.5. Immunoblot Analysis
3.6. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Schroder, K.; Tschopp, J. The inflammasomes. Cell 2010, 140, 821–832. [Google Scholar] [CrossRef] [PubMed]
- Martinon, F.; Burns, K.; Tschopp, J. The inflammasome: A molecular platform triggering activation of inflammatory caspases and processing of proIL-β. Mol. Cell 2002, 10, 417–426. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.; Yu, J.; Jiang, T.; Zhu, X.; Tan, L. The NLRP3 inflammasome in Alzheimer’s disease. Mol. Neurobiol. 2013, 48, 875–882. [Google Scholar] [CrossRef]
- Esser, N.; L’homme, L.; de Roover, A.; Kohnen, L.; Scheen, A.J.; Moutschen, M.; Piette, J.; Legrand-Poels, S.; Paquot, N. Obesity phenotype is related to NLRP3 inflammasome activity and immunological profile of visceral adipose tissue. Diabetologia 2013, 56, 2487–2497. [Google Scholar] [CrossRef] [PubMed]
- Alcocer-Gómez, E.; de Miguel, M.; Casas-Barquero, N.; Núñez-Vasco, J.; Sánchez-Alcazar, J.A.; Fernández-Rodríguez, A.; Cordero, M.D. NLRP3 Inflammasome is activated in mononuclear blood cells from patients with major depressive disorder. Brain Behav. Immun. 2014, 36, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Vandanmagsar, B.; Youm, Y.; Ravussin, A.; Galgani, J.E.; Stadler, K.; Mynatt, R.L.; Ravussin, E.; Stephens, J.M.; Dixit, V.D. The NLRP3 inflammasome instigates obesity-induced inflammation and insulin resistance. Nat. Med. 2011, 17, 179–188. [Google Scholar] [CrossRef] [PubMed]
- Cordero, M.D.; Alcocer-Gómez, E.; Culic, O.; Carrión, A.M.; de Miguel, M.; Díaz-Parrado, E.; Pérez-Villegas, E.M.; Bullón, P.; Battino, M.; Sánchez-Alcazar, J.A. NLRP3 inflammasome is activated in fibromyalgia: The effect of coenzyme Q10. Antioxid. Redox Signal. 2014, 20, 1169–1180. [Google Scholar] [CrossRef] [PubMed]
- Kuo, Y.; Tsai, W.; Meng, H.; Chen, W.; Yang, L.; Lin, C. Immune reponses in human mesangial cells regulated by emodin from Polygonum Hypoleucum Ohwi. Life Sci. 2001, 68, 1271–1286. [Google Scholar] [CrossRef] [PubMed]
- Basu, S.; Ghosh, A.; Hazra, B. Evaluation of the antibacterial activity of Ventilago madraspatana Gaertn., Rubia cordifolia Linn. and Lantana camara Linn.: Isolation of emodin and physcion as active antibacterial agents. Phytother. Res. 2005, 19, 888–894. [Google Scholar]
- Sato, M.; Maulik, G.; Bagchi, D.; Das, D.K. Myocardial protection by protykin, a novel extract of trans-resveratrol and emodin. Free Radic. Res. 2000, 32, 135–144. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.; Chang, H.; Shyue, S.; Hsu, S. Emodin Induces Apoptosis in human lung adenocarcinoma cells through a reactive oxygen species-dependent mitochondrial signaling pathway. Biochem. Pharmacol. 2005, 70, 229–241. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Yang, J.H.; Li, X.; Hwangbo, K.; Hwang, S.; Taketomi, Y.; Murakami, M.; Chang, Y.; Kim, C.; Son, J. Emodin, a naturally occurring anthraquinone derivative, suppresses LgE-mediated anaphylactic reaction and mast cell activation. Biochem. Pharmacol. 2011, 82, 1700–1708. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Kang, T.; Shim, D.; Sun, X.; Han, J.; Ji, Y.; Kim, T.; Koppula, S.; Lee, K. Emodin attenuates A23187-induced mast cell degranulation and tumor necrosis factor-α secretion through protein kinase C and IκB kinase 2 signaling. Eur. J. Pharmacol. 2014, 723, 501–506. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Tu, X.; Lin, G.; Xia, H.; Huang, H.; Wan, J.; Cheng, Z.; Liu, M.; Chen, G.; Zhang, H. Emodin-mediated protection from acute myocardial infarction via inhibition of inflammation and apoptosis in local ischemic myocardium. Life Sci. 2007, 81, 1332–1338. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Zhao, L.; Mei, H.; Zhang, S.; Huang, Z.; Duan, Y.; Ye, P. Exploration of emodin to treat α-naphthylisothiocyanate-induced cholestatic hepatitis via anti-inflammatory pathway. Eur. J. Pharmacol. 2008, 590, 377–386. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Dhawan, S.; Aggarwal, B.B. Emodin (3-methyl-1,6,8-trihydroxyanthraquinone) inhibits TNF-induced NF-κB activation, IκB degradation, and expression of cell surface adhesion proteins in human vascular endothelial cells. Oncogene 1998, 17, 913–918. [Google Scholar] [CrossRef] [PubMed]
- Fernandes-Alnemri, T.; Wu, J.; Yu, J.; Datta, P.; Miller, B.; Jankowski, W.; Rosenberg, S.; Zhang, J.; Alnemri, E. The pyroptosome: A supramolecular assembly of ASC dimers mediating inflammatory cell death via caspase-1 activation. Cell Death Differ. 2007, 14, 1590–1604. [Google Scholar] [CrossRef] [PubMed]
- Bryant, C.; Fitzgerald, K.A. Molecular mechanisms involved in inflammasome activation. Trends Cell Biol. 2009, 19, 455–464. [Google Scholar] [CrossRef] [PubMed]
- Zhou, R.; Yazdi, A.S.; Menu, P.; Tschopp, J. A role for mitochondria in NLRP3 inflammasome activation. Nature 2011, 469, 221–225. [Google Scholar] [CrossRef]
- Hara, H.; Tsuchiya, K.; Kawamura, I.; Fang, R.; Hernandez-Cuellar, E.; Shen, Y.; Mizuguchi, J.; Schweighoffer, E.; Tybulewicz, V.; Mitsuyama, M. Phosphorylation of the adaptor ASC acts as a molecular switch that controls the formation of speck-like aggregates and inflammasome activity. Nat. Immunol. 2013, 14, 1247–1255. [Google Scholar] [CrossRef]
- Yu, J.; Wu, J.; Zhang, Z.; Datta, P.; Ibrahimi, I.; Taniguchi, S.; Sagara, J.; Fernandes-Alnemri, T.; Alnemri, E. Cryopyrin and pyrin activate caspase-1, but not NF-κB, via ASC oligomerization. Cell Death Differ. 2006, 13, 236–249. [Google Scholar] [CrossRef]
- Mariathasan, S.; Weiss, D.S.; Newton, K.; McBride, J.; O'Rourke, K.; Roose-Girma, M.; Lee, W.P.; Weinrauch, Y.; Monack, D.M.; Dixit, V.M. Cryopyrin activates the inflammasome in response to toxins and ATP. Nature 2006, 440, 228–232. [Google Scholar] [CrossRef]
- Shim, D.; Han, J.; Sun, X.; Jang, C.; Koppula, S.; Kim, T.; Kang, T.; Lee, K. Lysimachia clethroides Duby extract attenuates inflammatory response in Raw 264.7 macrophages stimulated with lipopolysaccharide and in acute lung injury mouse model. J. Ethnopharmacol. 2013, 150, 1007–1015. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, J.-W.; Shim, D.-W.; Shin, W.-Y.; Heo, K.-H.; Kwak, S.-B.; Sim, E.-J.; Jeong, J.-H.; Kang, T.-B.; Lee, K.-H. Anti-Inflammatory Effect of Emodin via Attenuation of NLRP3 Inflammasome Activation. Int. J. Mol. Sci. 2015, 16, 8102-8109. https://doi.org/10.3390/ijms16048102
Han J-W, Shim D-W, Shin W-Y, Heo K-H, Kwak S-B, Sim E-J, Jeong J-H, Kang T-B, Lee K-H. Anti-Inflammatory Effect of Emodin via Attenuation of NLRP3 Inflammasome Activation. International Journal of Molecular Sciences. 2015; 16(4):8102-8109. https://doi.org/10.3390/ijms16048102
Chicago/Turabian StyleHan, Ji-Won, Do-Wan Shim, Woo-Young Shin, Kang-Hyuk Heo, Su-Bin Kwak, Eun-Jeong Sim, Jae-Hyun Jeong, Tae-Bong Kang, and Kwang-Ho Lee. 2015. "Anti-Inflammatory Effect of Emodin via Attenuation of NLRP3 Inflammasome Activation" International Journal of Molecular Sciences 16, no. 4: 8102-8109. https://doi.org/10.3390/ijms16048102
APA StyleHan, J.-W., Shim, D.-W., Shin, W.-Y., Heo, K.-H., Kwak, S.-B., Sim, E.-J., Jeong, J.-H., Kang, T.-B., & Lee, K.-H. (2015). Anti-Inflammatory Effect of Emodin via Attenuation of NLRP3 Inflammasome Activation. International Journal of Molecular Sciences, 16(4), 8102-8109. https://doi.org/10.3390/ijms16048102