The Autonomous Glycosylation of Large DNA Viruses
Abstract
:1. Introduction
2. Nucleo-Cytoplasmic Large DNA Viruses (NCLDV)
3. Phycodnaviridae
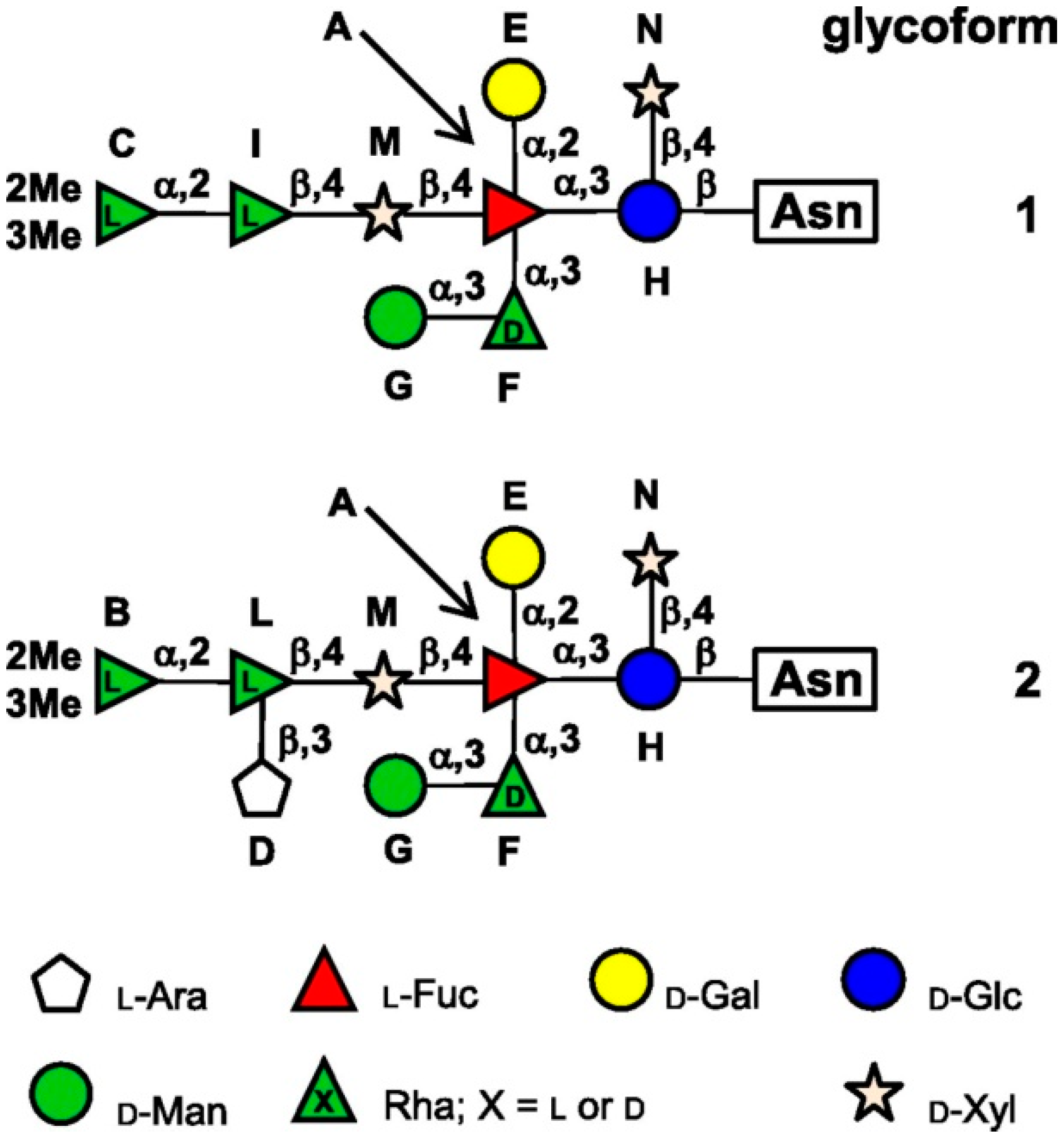
3.1. Chlorovirus Nucleotide-Sugar Biosynthetic Pathways.
3.1.1. GDP-l-Fucose Production
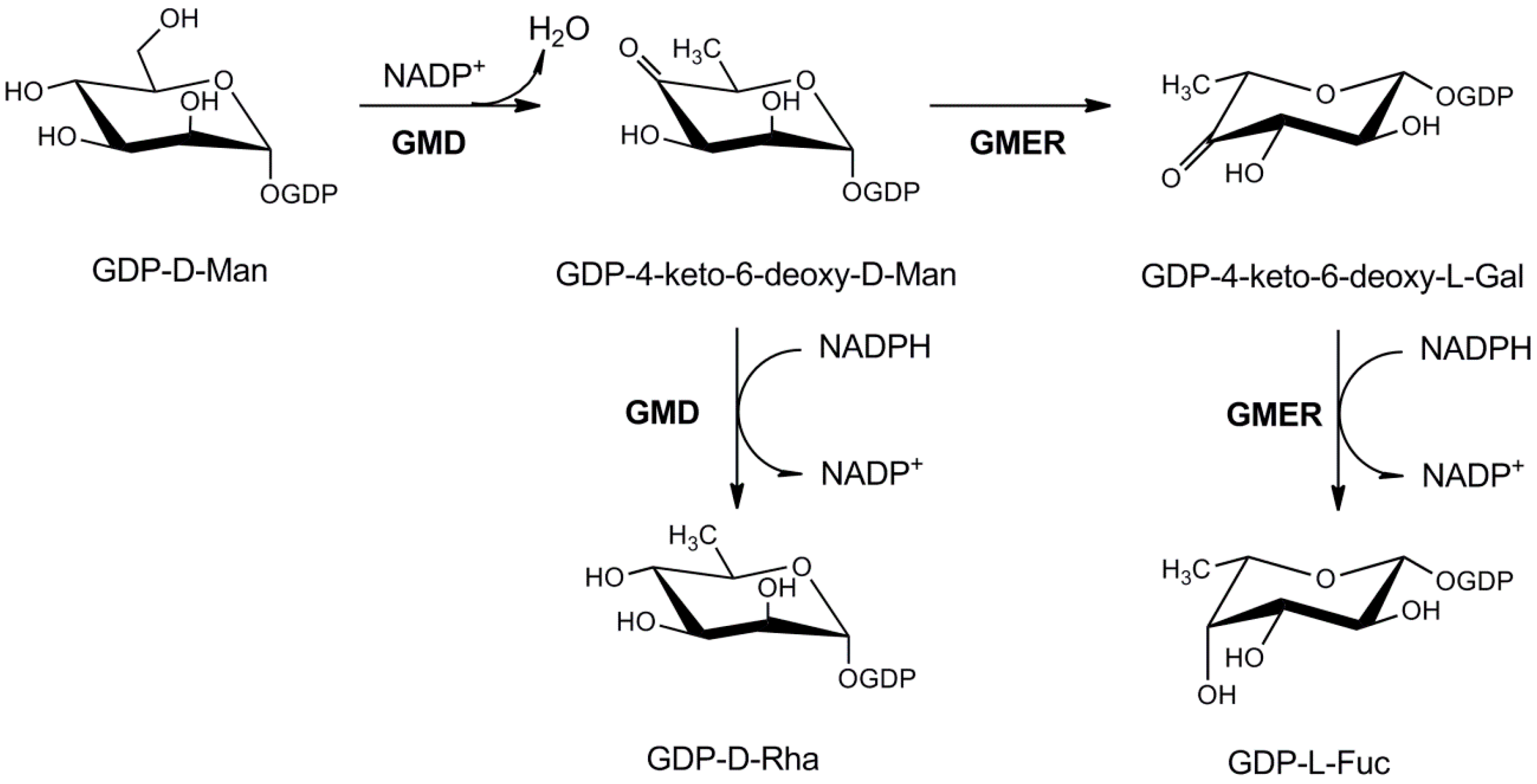
3.1.2. UDP-l-Rhamnose Pathway
3.1.3. Glucosamine-6P and UDP-d-Glucuronic Biosynthesis
3.2. Chlorovirus Glycosyltransferases
4. Mimiviridae
4.1. Nucleotide-Sugar Metabolism
4.1.1. UDP-d-GlcNAc Biosynthesis
4.1.2. UDP-l-Rhamnose Pathway
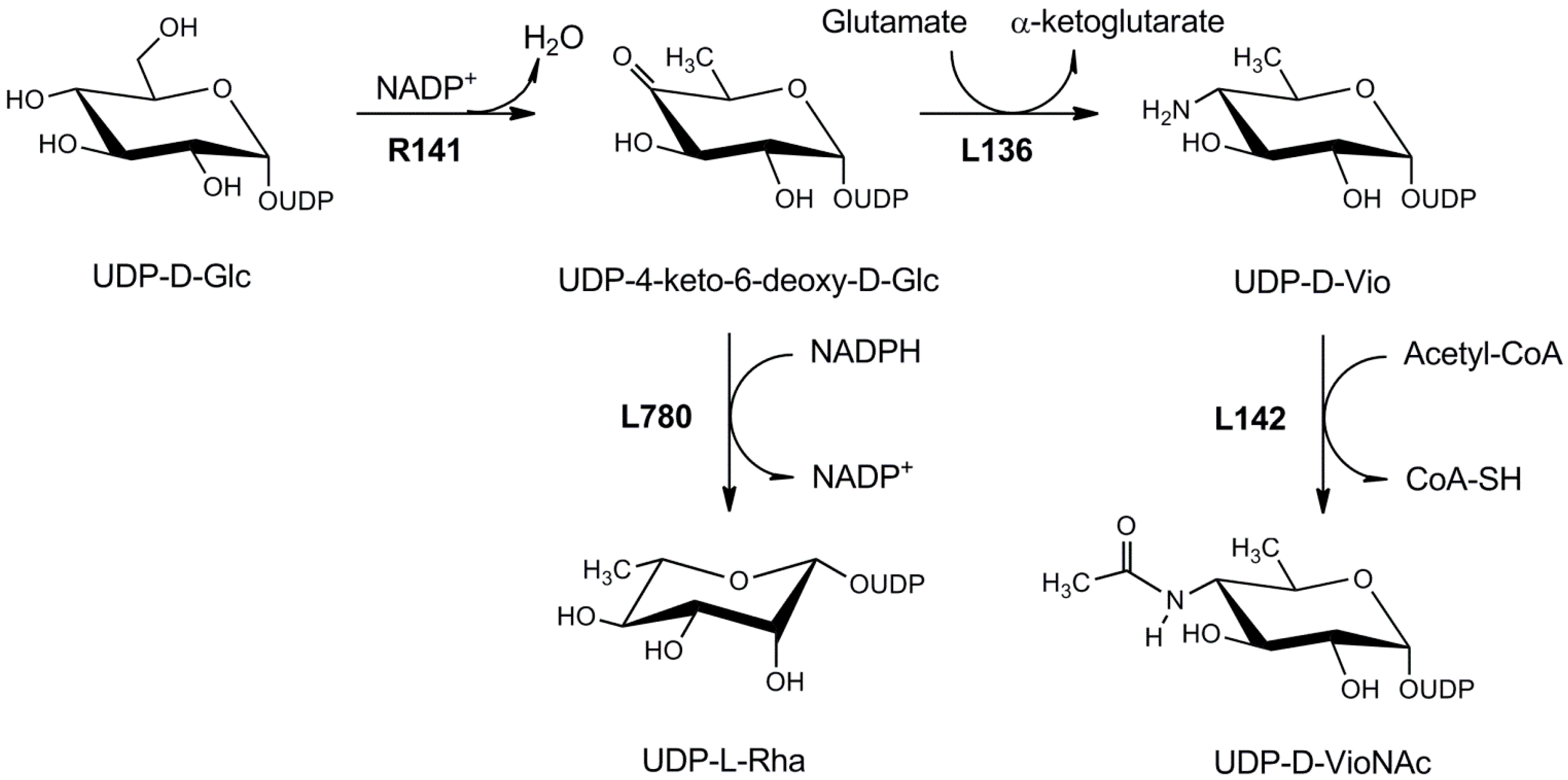
4.1.3. UDP-d-Viosamine Pathway
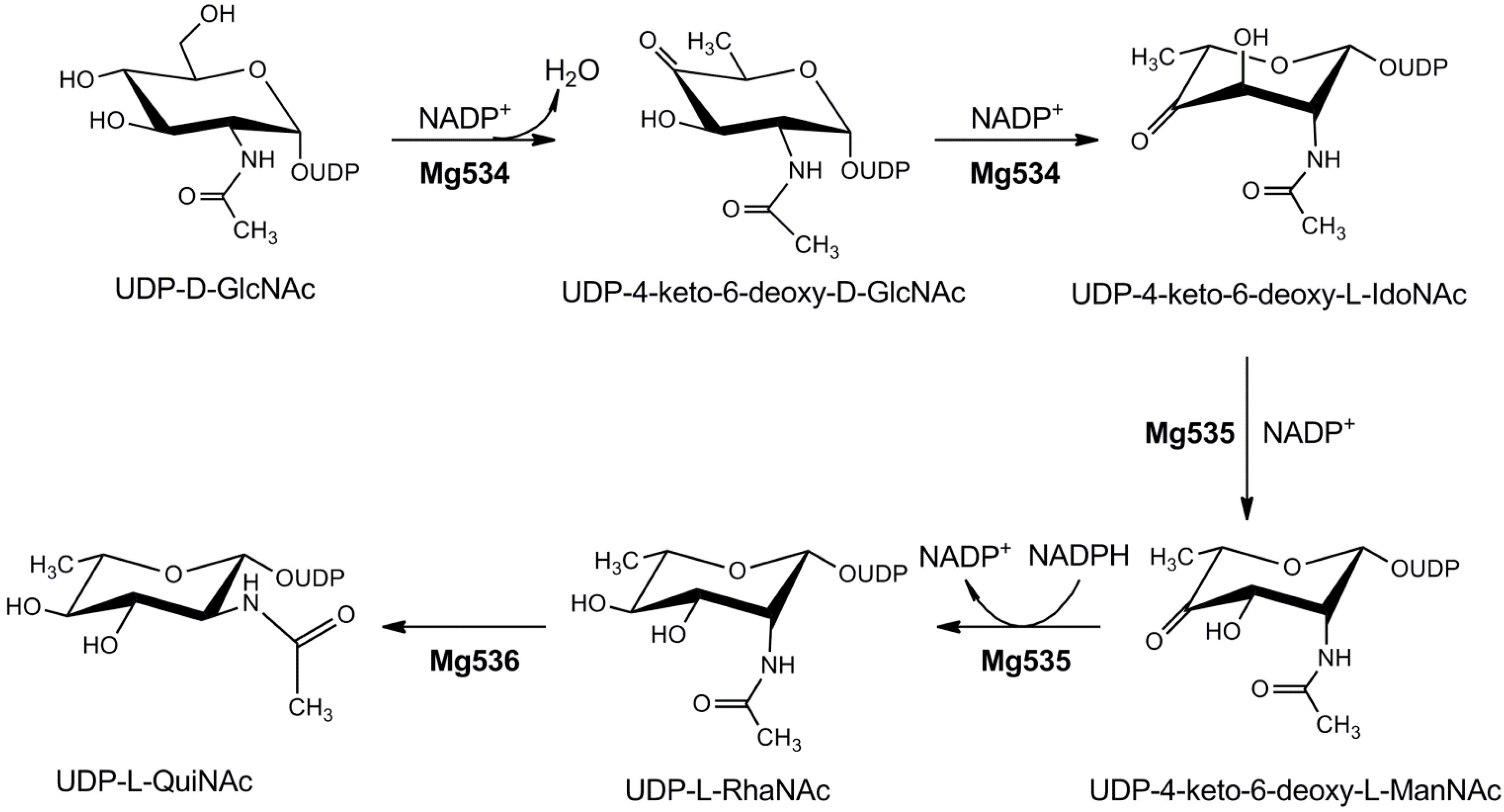
4.1.4. UDP-2-Acetamido-4,6-dideoxyhexoses
4.2. Mimiviridae Glycosyltransferases
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Vigerust, D.J.; Shepherd, V.L. Virus glycosylation: Role in virulence and immune interactions. Trends Microbiol. 2007, 15, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Tate, M.D.; Job, E.R.; Deng, Y.M.; Gunalan, V.; Maurer-Stroh, S.; Reading, P.C. Playing hide and seek: How glycosylation of the influenza virus hemagglutinin can modulate the immune response to infection. Viruses 2014, 6, 1294–1316. [Google Scholar] [CrossRef] [PubMed]
- Hiraiwa, N.; Yabuta, T.; Yoritomi, K.; Hiraiwa, M.; Tanaka, Y.; Suzuki, T.; Yoshida, M.; Kannagi, R. Transactivation of the fucosyltransferase VII gene by human T-cell leukemia virus type 1 Tax through a variant cAMP-responsive element. Blood 2003, 101, 3615–3621. [Google Scholar] [CrossRef] [PubMed]
- Markine-Goriaynoff, N.; Gillet, L.; van Etten, J.L.; Korres, H.; Verma, N.; Vanderplasschen, A. Glycosyltransferases encoded by viruses. J. Gen. Virol. 2004, 85, 2741–2754. [Google Scholar] [CrossRef] [PubMed]
- Van Etten, J.L.; Lane, L.C.; Dunigan, D.D. DNA viruses: The really big ones (giruses). Annu. Rev. Microbiol. 2010, 64, 83–99. [Google Scholar] [CrossRef] [PubMed]
- Colson, P.; de Lamballerie, X.; Yutin, N.; Asgari, S.; Bigot, Y.; Bideshi, D.K.; Cheng, X.W.; Federici, B.A.; van Etten, J.L.; Koonin, E.V.; et al. “Megavirales”, a proposed new order for eukaryotic nucleocytoplasmic large DNA viruses. Arch. Virol. 2013, 158, 2517–2521. [Google Scholar] [CrossRef] [PubMed]
- Claverie, J.M.; Abergel, C. Mimivirus: The emerging paradox of quasi-autonomous viruses. Trends Genet. 2010, 26, 431–437. [Google Scholar] [CrossRef] [PubMed]
- Abergel, C.; Legendre, M.; Claverie, J.M. The rapidly expanding universe of giant viruses: Mimivirus, Pandoravirus, Pithovirus and Mollivirus. FEMS Microbiol. Rev. 2015, 39, 779–796. [Google Scholar] [CrossRef] [PubMed]
- Van Etten, J.L.; Gurnon, J.R.; Yanai-Balser, G.M.; Dunigan, D.D.; Graves, M.V. Chlorella viruses encode most, if not all, of the machinery to glycosylate their glycoproteins independent of the endoplasmic reticulum and Golgi. Biochim. Biophys. Acta 2010, 1800, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Yutin, N.; Colson, P.; Raoult, D.; Koonin, E.V. Mimiviridae: Clusters of orthologous genes, reconstruction of gene repertoire evolution and proposed expansion of the giant virus family. Virol. J. 2013, 10, 106. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Filée, J. Route of NCLDV evolution: The genomic accordion. Curr. Opin. Virol. 2013, 3, 595–599. [Google Scholar] [CrossRef] [PubMed]
- Moreira, D.; Brochier-Armanet, C. Giant viruses, giant chimeras: The multiple evolutionary histories of Mimivirus genes. BMC Evol. Biol. 2008, 8, 12. [Google Scholar] [CrossRef] [PubMed]
- Filée, J.; Siguier, P.; Chandler, M. I am what I eat and I eat what I am: Acquisition of bacterial genes by giant viruses. Trends Genet. 2007, 23, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Yutin, N.; Wolf, Y.I.; Koonin, E.V. Origin of giant viruses from smaller DNA viruses not from a fourth domain of cellular life. Virology 2014, 466–467, 38–52. [Google Scholar]
- Milrot, E.; Mutsafi, Y.; Fridmann-Sirkis, Y.; Shimoni, E.; Rechav, K.; Gurnon, J.R.; van Etten, J.L.; Minsky, A. Virus-host interactions: Insights from the replication cycle of the large Paramecium bursaria chlorella virus. Cell Microbiol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Mutsafi, Y.; Zauberman, N.; Sabanay, I.; Minsky, A. Vaccinia-like cytoplasmic replication of the giant Mimivirus. Proc. Natl. Acad. Sci. USA 2010, 107, 5978–5982. [Google Scholar] [CrossRef] [PubMed]
- Kuznetsov, Y.G.; Klose, T.; Rossmann, M.; McPherson, A. Morphogenesis of mimivirus and its viral factories: An atomic force microscopy study of infected cells. J. Virol. 2013, 87, 11200–11213. [Google Scholar] [CrossRef] [PubMed]
- Suzan-Monti, M.; La Scola, B.; Barrassi, L.; Espinosa, L.; Raoult, D. Ultrastructural characterization of the giant volcano-like virus factory of Acanthamoeba polyphaga Mimivirus. PLoS ONE 2007, 2, e328. [Google Scholar] [CrossRef] [PubMed]
- De Castro, C.; Molinaro, A.; Piacente, F.; Gurnon, J.R.; Sturiale, L.; Palmigiano, A.; Lanzetta, R.; Parrili, M.; Garozzo, D.; Tonetti, M.G.; et al. Structure of N-linked oligosaccharides attached to chlorovirus PBCV-1 major capsid protein reveals unusual class of complex N-glycans. Proc. Natl. Acad. Sci. USA 2013, 110, 13956–13960. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Onimatsu, H.; van Etten, J.L. Chlorella viruses. Adv. Virus Res. 2006, 66, 293–336. [Google Scholar] [PubMed]
- Wilson, W.H.; van Etten, J.L.; Allen, M.J. The phycodnaviridae: The story of how tiny giants rule the world. Curr. Top. Microbiol. Immunol. 2009, 328, 1–42. [Google Scholar] [PubMed]
- Van Etten, J.L.; Dunigan, D.D. Chloroviruses: Not your everyday plant virus. Trends Plant Sci. 2012, 17, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Dunigan, D.D.; Fitzgerald, L.A.; van Etten, J.L. Phycodnaviruses: A peek at genetic diversity. Virus Res. 2006, 117, 119–132. [Google Scholar] [CrossRef] [PubMed]
- Dunigan, D.D.; Cerny, R.L.; Bauman, A.T.; Roach, J.C.; Lane, L.C.; Agarkova, I.V.; Wulser, K.; Yanai-Balser, G.M.; Gurnon, J.R.; Vitek, J.C.; et al. Paramecium bursaria chlorella virus 1 proteome reveals novel architectural and regulatory features of a giant virus. J. Virol. 2012, 86, 8821–8834. [Google Scholar] [CrossRef] [PubMed]
- Danovaro, R.; Corinaldesi, C.; Dell'Anno, A.; Fuhrman, J.A.; Middelburg, J.J.; Noble, R.T.; Suttle, C.A. Marine viruses and global climate change. FEMS Microbiol. Rev. 2011, 35, 993–1034. [Google Scholar] [CrossRef] [PubMed]
- Sharoni, S.; Trainic, M.; Schatz, D.; Lehahn, Y.; Flores, M.J.; Bidle, K.D.; Ben-Dor, S.; Rudich, Y.; Koren, I.; Vardi, A. Infection of phytoplankton by aerosolized marine viruses. Proc. Natl. Acad. Sci. USA 2015, 112, 6643–6647. [Google Scholar] [CrossRef] [PubMed]
- Van Etten, J.L.; Burbank, D.E.; Xia, Y.; Meints, R.H. Growth cycle of a virus, PBCV-1, that infects Chlorella-like algae. Virology 1983, 126, 117–125. [Google Scholar] [CrossRef]
- Jeanniard, A.; Dunigan, D.D.; Gurnon, J.R.; Agarkova, I.V.; Kang, M.; Vitek, J.; Duncan, G.; McClung, O.W.; Larsen, M.; Claverie, J.M.; et al. Towards defining the chloroviruses: A genomic journey through a genus of large DNA viruses. BMC Genom. 2013, 14, 158. [Google Scholar] [CrossRef] [PubMed]
- Graves, M.V.; Bernadt, C.T.; Cerny, R.; van Etten, J.L. Molecular and genetic evidence for a virus-encoded glycosyltransferase involved in protein glycosylation. Virology 2001, 285, 332–345. [Google Scholar] [CrossRef] [PubMed]
- Wang, I.N.; Li, Y.; Que, Q.; Bhattacharya, M.; Lane, L.C.; Chaney, W.G.; van Etten, J.L. Evidence for virus-encoded glycosylation specificity. Proc. Natl. Acad. Sci. USA. 1993, 90, 3840–3844. [Google Scholar] [CrossRef] [PubMed]
- Nandhagopal, N.; Simpson, A.A.; Gurnon, J.R.; Yan, X.; Baker, T.S.; Graves, M.V.; van Etten, J.L.; Rossmann, M.G. The structure and evolution of the major capsid protein of a large, lipid-containing DNA virus. Proc. Natl. Acad. Sci. USA 2002, 99, 14758–14763. [Google Scholar] [CrossRef] [PubMed]
- Allen, S.; Richardson, J.M.; Mehlert, A.; Ferguson, M.A. Structure of a complex phosphoglycan epitope from gp72 of trypanosoma cruzi. J. Biol. Chem. 2013, 288, 11093–11105. [Google Scholar] [CrossRef] [PubMed]
- Tonetti, M.; Zanardi, D.; Gurnon, J.R.; Fruscione, F.; Armirotti, A.; Damonte, G.; Sturla, L.; de Flora, A.; van Etten, J.L. Paramecium bursaria chlorella virus 1 encodes two enzymes involved in the biosynthesis of GDP-l-fucose and GDP-d-rhamnose. J. Biol. Chem. 2003, 278, 21559–21565. [Google Scholar] [CrossRef] [PubMed]
- Agarkova, I.; Hertel, B.; Zhang, X.; Lane, L.; Tchourbanov, A.; Dunigan, D.D.; Thiel, G.; Rossmann, M.G.; van Etten, J.L. Dynamic attachment of Chlorovirus PBCV-1 to chlorella variabilis. Virology 2014, 466–467, 95–102. [Google Scholar]
- De Angelis, P.L.; Jing, W.; Graves, M.V.; Burbank, D.E.; van Etten, J.L. Hyaluronan synthase of chlorella virus PBCV-1. Science 1997, 278, 1800–1803. [Google Scholar] [CrossRef]
- Graves, M.V.; Burbank, D.E.; Roth, R.; Heuser, J.; de Angelis, P.L.; van Etten, J.L. Hyaluronan synthesis in virus PBCV-1-infected chlorella-like green algae. Virology 1999, 257, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, T.; Tanaka, M.; Fujie, M.; Usami, S.; Sakai, K.; Yamada, T. Chitin Synthesis in Chlorovirus CVK2-Infected Chlorella Cells. Virology 2002, 302, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Fruscione, F.; Sturla, L.; Duncan, G.; van Etten, J.L.; Valbuzzi, P.; de Flora, A.; di Zanni, E.; Tonetti, M. Differential role of NADP+ and NADPH in the activity and structure of GDP-D-mannose 4,6-Dehydratase from two chlorella viruses. J. Biol. Chem. 2008, 283, 184–193. [Google Scholar] [CrossRef] [PubMed]
- De Castro, C.; Speciale, I.; Duncan, G.; Dunigan, D.D.; Agarkova, I.; Lanzetta, R.; Sturiale, L.; Palmigiano, A.; Garozzo, D.; Molinaro, A.; et al. N-linked glycans of chloroviruses sharing a core architecture without precedent. Angew. Chem. Int. Ed. Engl. 2015. [Google Scholar] [CrossRef]
- Rosano, C.; Zuccotti, S.; Sturla, L.; Fruscione, F.; Tonetti, M.; Bolognesi, M. Quaternary assembly and crystal structure of GDP-d-mannose 4,6 dehydratase from Paramecium bursaria Chlorella virus. Biochem. Biophys. Res. Commun. 2006, 339, 191–195. [Google Scholar] [CrossRef] [PubMed]
- Parakkottil Chothi, M.; Duncan, G.A.; Armirotti, A.; Abergel, C.; Gurnon, J.R.; van Etten, J.L.; Bernardi, C.; Damonte, G.; Tonetti, M. Identification of an L-rhamnose synthetic pathway in two nucleocytoplasmic large DNA viruses. J. Virol. 2010, 84, 8829–8838. [Google Scholar] [CrossRef] [PubMed]
- Landstein, D.; Graves, M.V.; Burbank, D.E.; de Angelis, P.; van Etten, J.L. Chlorella virus PBCV-1 encodes functional glutamine: Fructose-6-phosphate amidotransferase and UDP-glucose dehydrogenase enzymes. Virology 1998, 250, 388–396. [Google Scholar] [CrossRef] [PubMed]
- Mohammed Ali, A.M.; Kawasaki, T.; Yamada, T. Genetic rearrangements on the Chlorovirus genome that switch between hyaluronan synthesis and chitin synthesis. Virology 2005, 342, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Piacente, F.; Bernardi, C.; Marin, M.; Blanc, G.; Abergel, C.; Tonetti, M.G. Characterization of a UDP-N-acetylglucosamine biosynthetic pathway encoded by the giant DNA virus Mimivirus. Glycobiology 2014, 24, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Xiang, Y.; van Etten, J.L.; Rossmann, M.G. Structure and function of a chlorella virus-encoded glycosyltransferase. Structure 2007, 15, 1031–1039. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Y.; Baxa, U.; Zhang, Y.; Steven, A.C.; Lewis, G.L.; van Etten, J.L.; Rossmann, M.G. Crystal structure of a virus-encoded putative glycosyltransferase. J. Virol. 2010, 84, 12265–12273. [Google Scholar] [CrossRef] [PubMed]
- Raoult, D.; Audic, S.; Robert, C.; Abergel, C.; Renesto, P.; Ogata, H.; La Scola, B.; Suzan, M.; Claverie, J.M. The 1.2-megabase genome sequence of mimivirus. Science 2004, 306, 1344–1350. [Google Scholar] [CrossRef] [PubMed]
- Legendre, M.; Santini, S.; Rico, A.; Abergel, C.; Claverie, J.M. Breaking the 1000-gene barrier for mimivirus using ultra-deep genome and transcriptome sequencing. Virol. J. 2011, 8, 99. [Google Scholar] [CrossRef] [PubMed]
- Kuznetsov, Y.G.; Xiao, C.; Sun, S.; Raoult, D.; Rossmann, M.; McPherson, A. Atomic force microscopy investigation of the giant mimivirus. Virology 2010, 404, 127–137. [Google Scholar] [CrossRef] [PubMed]
- Xiao, C.; Kuznetsov, Y.G.; Sun, S.; Hafenstein, S.L.; Kostyuchenko, V.A.; Chipman, P.R.; Suzan-Monti, M.; Raoult, D.; McPherson, A.; Rossmann, M.G. Structural studies of the giant mimivirus. PLoS Biol. 2009, 7, e92. [Google Scholar] [CrossRef] [PubMed]
- Klose, T.; Rossmann, M.G. Structure of large dsDNA viruses. Biol. Chem. 2014, 395, 711–719. [Google Scholar] [CrossRef] [PubMed]
- Fischer, M.G.; Allen, M.J.; Wilson, W.H.; Suttle, C.A. Giant virus with a remarkable complement of genes infects marine zooplankton. Proc. Natl. Acad. Sci. USA 2010, 107, 19508–19513. [Google Scholar] [CrossRef] [PubMed]
- Arslan, D.; Legendre, M.; Seltzer, V.; Abergel, C.; Claverie, J.M. Distant Mimivirus relative with a larger genome highlights the fundamental features of Megaviridae. Proc. Natl. Acad. Sci. USA 2011, 108, 17486–17491. [Google Scholar] [CrossRef] [PubMed]
- Yoosuf, N.; Yutin, N.; Colson, P.; Shabalina, S.A.; Pagnier, I.; Robert, C.; Azza, S.; Klose, T.; Wong, J.; Rossmann, M.G.; et al. related giant viruses in distant locations and different habitats: Acanthamoeba polyphaga moumouvirus represents a third lineage of the mimiviridae that is close to the megavirus lineage. Genom. Biol. Evol. 2012, 4, 1324–1330. [Google Scholar] [CrossRef] [PubMed]
- Santini, S.; Jeudy, S.; Bartoli, J.; Poirot, O.; Lescot, M.; Abergel, C.; Barbe, V.; Wommack, K.E.; Noordeloos, A.A.M.; Brussaard, C.P.D.; et al. Genome of phaeocystis globosa virus PgV-16T highlights the common ancestry of the largest known DNA viruses infecting eukaryotes. Proc. Natl. Acad. Sci. USA 2013, 110, 10800–10805. [Google Scholar] [CrossRef] [PubMed]
- Andrade, K.R.; Boratto, P.P.; Rodrigues, F.P.; Silva, L.C.; Dornas, F.P.; Pilotto, M.R.; La Scola, B.; Almeida, G.M.; Kroon, E.G.; Abrahão, J.S. Oysters as hot spots for mimivirus isolation. Arch. Virol. 2015, 160, 477–482. [Google Scholar] [CrossRef] [PubMed]
- Saadi, H.; Pagnier, I.; Colson, P.; Cherif, J.K.; Beji, M.; Boughalmi, M.; Azza, S.; Armstrong, N.; Robert, C.; Fournous, G.; et al. First isolation of mimivirus in a patient with pneumonia. Clin. Infect. Dis. 2013, 57, e127–e134. [Google Scholar] [CrossRef] [PubMed]
- Piacente, F.; Marin, M.; Molinaro, A.; de Castro, C.; Seltzer, V.; Salis, A.; Damonte, G.; Bernardi, C.; Claverie, J.M.; Abergel, C.; et al. Giant DNA virus mimivirus encodes pathway for biosynthesis of unusual sugar 4-Amino-4,6-dideoxy-d-glucose (Viosamine). J. Biol. Chem. 2012, 287, 3009–3018. [Google Scholar] [CrossRef] [PubMed]
- Luther, K.B.; Hülsmeier, A.J.; Schegg, B.; Deuber, S.A.; Raoult, D.; Hennet, T. Mimivirus collagen is modified by bifunctional lysyl hydroxylase and glycosyltransferase enzyme. J. Biol. Chem. 2011, 286, 43701–43709. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hülsmeier, A.J.; Hennet, T. O-Linked glycosylation in acanthamoeba polyphaga mimivirus. Glycobiology 2014, 24, 703–714. [Google Scholar] [CrossRef] [PubMed]
- Piacente, F.; de Castro, C.; Jeudy, S.; Molinaro, A.; Salis, A.; Damonte, G.; Bernardi, C.; Abergel, C.; Tonetti, M.G. Giant virus megavirus chilensis encodes the biosynthetic pathway for uncommon acetamido sugars. J. Biol. Chem. 2014, 289, 24428–24439. [Google Scholar] [CrossRef] [PubMed]
- Milewski, S.; Gabriel, I.; Olchowy, J. Enzymes of UDP-GlcNAc biosynthesis in yeast. Yeast 2006, 23, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Dong, C.; Beis, K.; Giraud, M.F.; Blankenfeldt, W.; Allard, S.; Major, L.L.; Kerr, I.D.; Whitfield, C.; Naismith, J.H. A structural perspective on the enzymes that convert dTDP-d-glucose into dTDP-l-rhamnose. Biochem. Soc. Trans. 2003, 31, 532–536. [Google Scholar] [CrossRef] [PubMed]
- Oka, T.; Nemoto, T.; Jigami, Y. Functional analysis of arabidopsis thaliana RHM2/MUM4, a multidomain protein involved in UDP-d-glucose to UDP-L-rhamnose conversion. J. Biol. Chem. 2007, 282, 5389–5403. [Google Scholar] [CrossRef] [PubMed]
- Tonetti, M. Tissue specific transplantation antigen P35B (= GDP-4-keto-6-D-deoxymannose epimerase-reductase) (TSTA3). In Handbook of Glycosyltransferases and Related Genes, 2nd ed.; Taniguchi, N., Honke, K., Fukuda, M., Narimatsu, H., Yamaguchi, Y., Angata, T., Eds.; Springer Japan: Tokyo, Japan, 2014; pp. 1607–1621. [Google Scholar]
- Martinez, V.; Ingwers, M.; Smith, J.; Glushka, J.; Yang, T.; Bar-Peled, M. Biosynthesis of UDP-4-keto-6-deoxyglucose and UDP-rhamnose in pathogenic fungi magnaporthe grisea and botryotinia fuckeliana. J. Biol. Chem. 2012, 287, 879–892. [Google Scholar] [CrossRef] [PubMed]
- Boyer, M.; Azza, S.; Barrassi, L.; Klose, T.; Campocasso, A.; Pagnier, I.; Fournous, G.; Borg, A.; Robert, C.; Zhang, X.; et al. Mimivirus shows dramatic genome reduction after intraamoebal culture. Proc. Natl. Acad Sci. USA 2011, 108, 10296–10301. [Google Scholar] [CrossRef] [PubMed]
- Moreau, H.; Piganeau, G.; Desdevises, Y.; Cooke, R.; Derelle, E.; Grimsley, N. Marine prasinovirus genomes show low evolutionary divergence and acquisition of protein metabolism genes by horizontal gene transfer. J. Virol. 2010, 84, 12555–12563. [Google Scholar] [CrossRef] [PubMed]
- Philippe, N.; Legendre, M.; Doutre, G.; Couté, Y.; Poirot, O.; Lescot, M.; Arslan, D.; Seltzer, V.; Bertaux, L.; Bruley, C.; et al. Pandoraviruses: Amoeba viruses with genomes up to 2.5 Mb reaching that of parasitic eukaryotes. Science 2013, 341, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Legendre, M.; Bartoli, J.; Shmakova, L.; Jeudy, S.; Labadie, K.; Adrait, A.; Lescot, M.; Poirot, O.; Bertaux, L.; Bruley, C.; et al. Thirty-thousand-year-old distant relative of giant icosahedral DNA viruses with a pandoravirus morphology. Proc. Natl. Acad. Sci. USA 2014, 111, 4274–4279. [Google Scholar] [CrossRef] [PubMed]
- Legendre, M.; Lartigue, A.; Bertaux, L.; Jeudy, S.; Bartoli, J.; Lescot, M.; Alempic, J.M.; Ramus, C.; Bruley, C.; Labadie, K.; et al. In-depth study of Mollivirus sibericum, a new 30,000-y-old giant virus infecting Acanthamoeba. Proc. Natl. Acad. Sci. USA. 2015, 112, E5327–E5335. [Google Scholar] [CrossRef] [PubMed]
- Petro, T.M.; Agarkova, I.V.; Zhou, Y.; Yolken, R.H.; van Etten, J.L.; Dunigan, D.D. Response of mammalian macrophages to challenge with the Chlorovirus ATCV-1. J. Virol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Yolken, R.H.; Jones-Brando, L.; Dunigan, D.D.; Kannan, G.; Dickerson, F.; Severance, E.; Sabunciyan, S.; Talbot, C.C., Jr.; Prandovszky, E.; Gurnon, J.R.; et al. Chlorovirus ATCV-1 is part of the human oropharyngeal virome and is associated with changes in cognitive functions in humans and mice. Proc. Natl. Acad. Sci. USA 2014, 111, 16106–16111. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piacente, F.; Gaglianone, M.; Laugieri, M.E.; Tonetti, M.G. The Autonomous Glycosylation of Large DNA Viruses. Int. J. Mol. Sci. 2015, 16, 29315-29328. https://doi.org/10.3390/ijms161226169
Piacente F, Gaglianone M, Laugieri ME, Tonetti MG. The Autonomous Glycosylation of Large DNA Viruses. International Journal of Molecular Sciences. 2015; 16(12):29315-29328. https://doi.org/10.3390/ijms161226169
Chicago/Turabian StylePiacente, Francesco, Matteo Gaglianone, Maria Elena Laugieri, and Michela G. Tonetti. 2015. "The Autonomous Glycosylation of Large DNA Viruses" International Journal of Molecular Sciences 16, no. 12: 29315-29328. https://doi.org/10.3390/ijms161226169
APA StylePiacente, F., Gaglianone, M., Laugieri, M. E., & Tonetti, M. G. (2015). The Autonomous Glycosylation of Large DNA Viruses. International Journal of Molecular Sciences, 16(12), 29315-29328. https://doi.org/10.3390/ijms161226169





