Applications of Micro-Fourier Transform Infrared Spectroscopy (FTIR) in the Geological Sciences—A Review
Abstract
:1. Introduction
1.1. Conventional FTIR Techniques for Bulk Sample Characterization
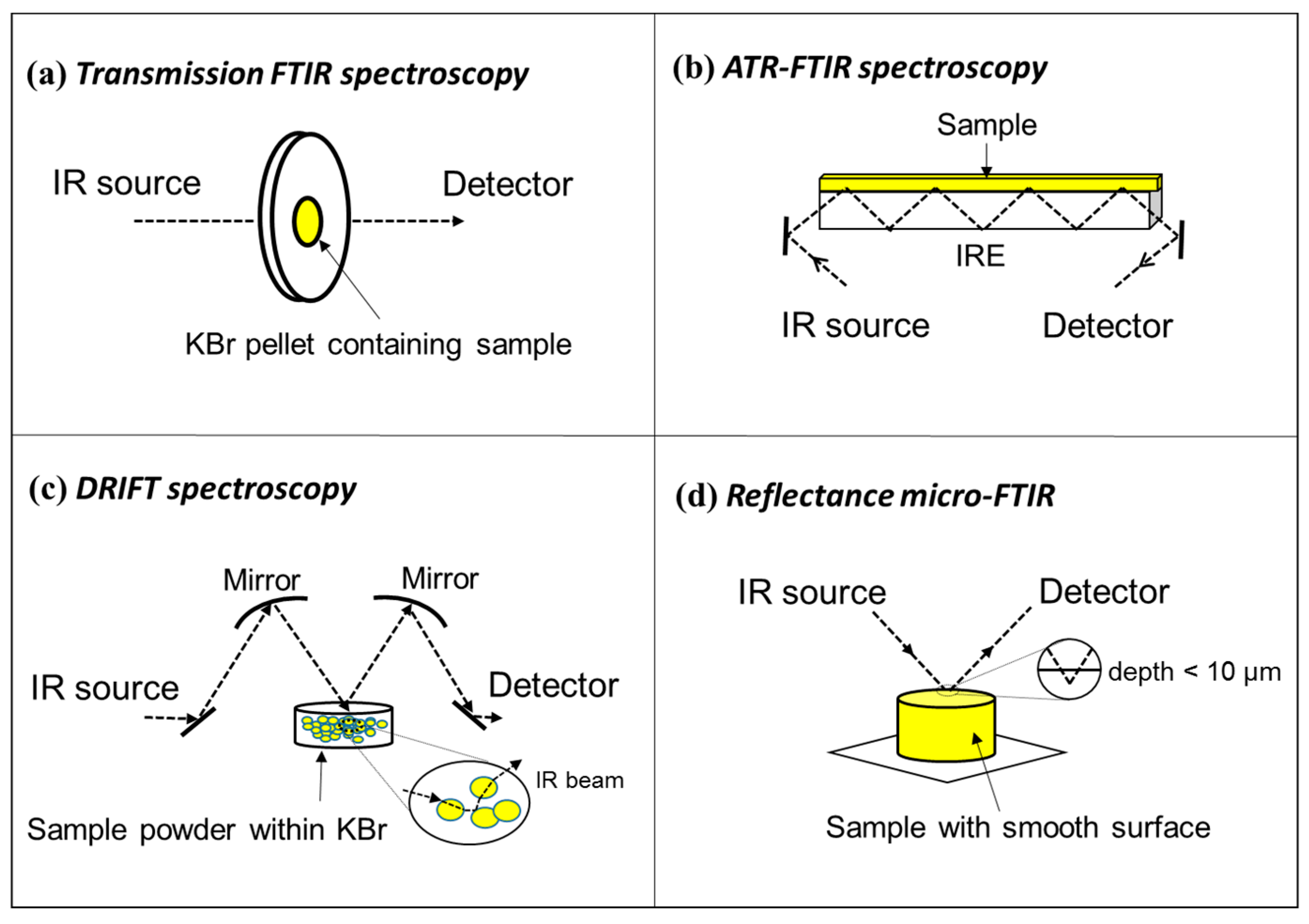
1.2. Micro-FTIR for in Situ High Resolution Characterization
2. Application of FTIR to the Chemical Characterization of Coal and Shale
2.1. Coal
| Organic Functional Groups | Inorganic Functional Groups | ||||
|---|---|---|---|---|---|
| Wavenumber (cm−1) | Assignment | Ref | Wavenumber (cm−1) | Assignment | Ref |
| 3300 | OH stretch intermolecular bonding | [50,52] | 3698, 3652,1095, 1034, 908, 689, 528, 338 | Kaolinite, Al2(Si2O5)(OH)4 | [52,54] |
| 3010 | Aromatic C–H | [50,51,52] | 1464, 705, 245 | Aragonite, CaCO3 | [54] |
| 2950 | Aliphatic CH3 | [50,51,52] | 1450, 882, 729 | Dolomite, CaMg(CO3)2 | [54] |
| 2920, 2850 | Aliphatic CH, CH2, and CH3 | [50,51,52] | 1431, 869, 307 | Calcite, CaCO3 | [54] |
| 1835 | C=O, anhydride | [50] | 3550, 3400,1615, 1155, 1132, 1106, 660 | Gypsum, CaSO4·2H2O | [54] |
| 1775–1765 | C=O, ester with electron withdrawing group attached to single bonded oxygen | [50] | 1115, 1148, 669 | Anhydrite, CaSO4 | [54] |
| 1735 | C=O ester | [50] | 1075, 790, 452 | Quartz, SiO2 | [50,54] |
| 1690–1720 | C=O, ketone, aldehyde, and -COOH | [50] | 1052 | Smectite, A0.3D2–3[T4O10]Z2·nH2O ¶ | [55] |
| 1650–1630 | C=O highly conjugated | [50] | 1015 | Oligoclase, (Na,Ca)[Al(Si,Al)Si2O8] | [55] |
| 1600 | Aromatic ring stretch | [50,51,52] | 1006 | Glauconite, (K,Na)(Mg,Fe)(Fe,Al)(Si,Al)4O10(OH)2 | [55] |
| ~1600 | High conjugated hydrogen bounded C=O | [50] | 1001 | Muscovite, KAl2(AlSi3O10)(OH)2 | [55] |
| 1560–1590 | Carboxyl group in salt from –COO− | [50] | 984 | Chlorite, A5–6T4Z18 § | [55] |
| 1490 | Aromatic ring stretch | [50] | 876, 727, 713 | Ankerite, Ca(Fe,Mg)(CO3)2 | [54] |
| 1450 | CH2 and CH3 bend, possibility of some aromatic ring modes | [50,51] | 407, 396 | Marcasite, FeS2 | [54] |
| 1375 | CH3 groups | [50,52] | 406, 340 | Pyrite, FeS2 | [54] |
| 1300–1100 | C-O stretch and O–H bend in phenoxy structures, ethers | [50] | |||
| 1100–1000 | Aliphatic ethers, alcohols | [50] | |||
| 900–700 | Aromatic C–H out-of-plane bending modes | [50] | |||
| 860 | Isolated aromatic H | [50] | |||
| 833 | 1,4-substituted aromatic groups | [50] | |||
| 815 | Isolated H and/or 2 neighboring H | [50] | |||
| 750 | 1,2-substituted, i.e., 4 neighboring H | [50] | |||
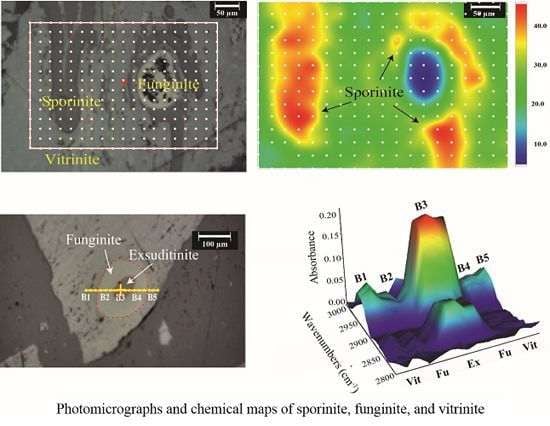
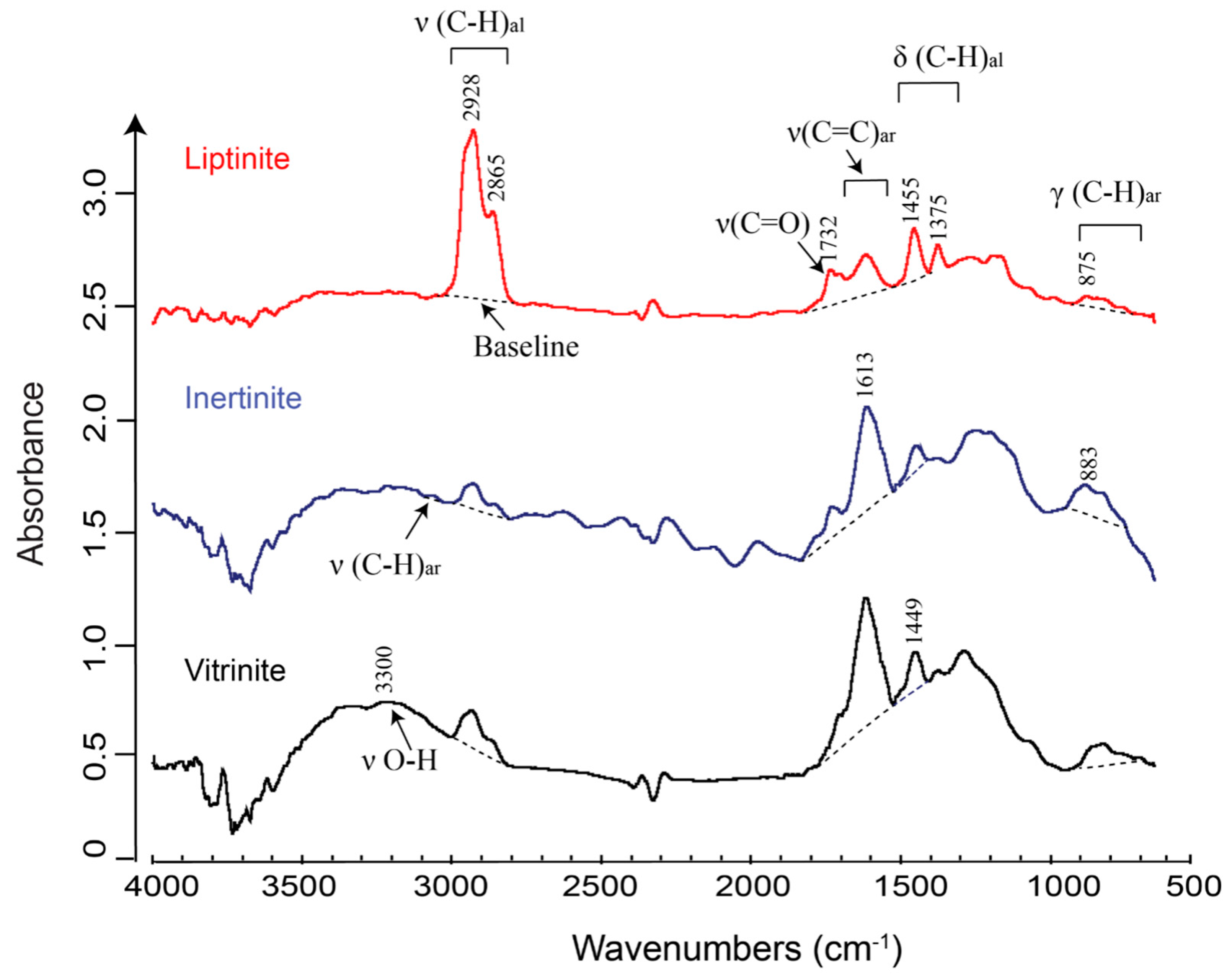
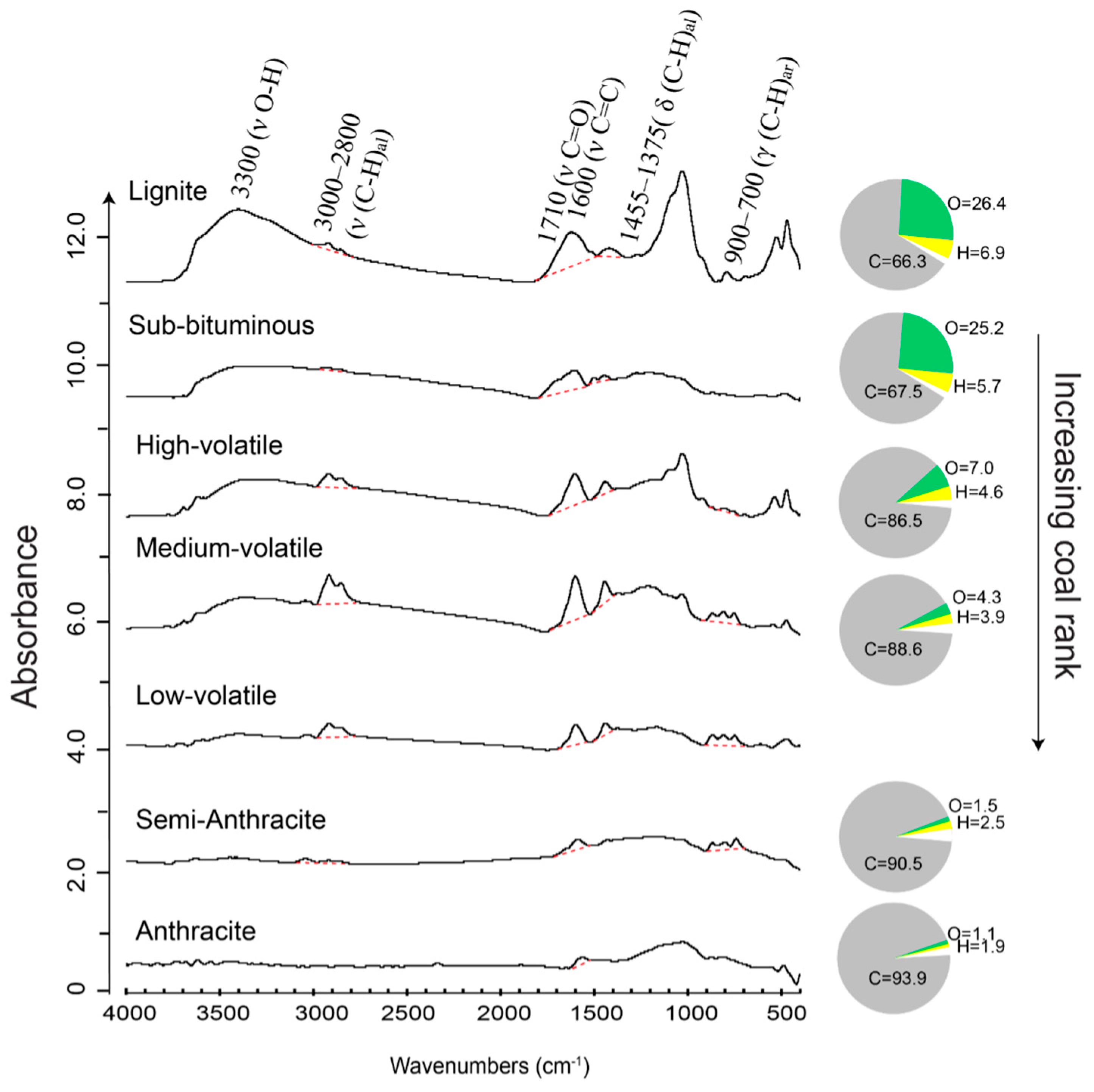
2.1.1. Geochemistry of Coal Macerals Investigated by Micro-FTIR
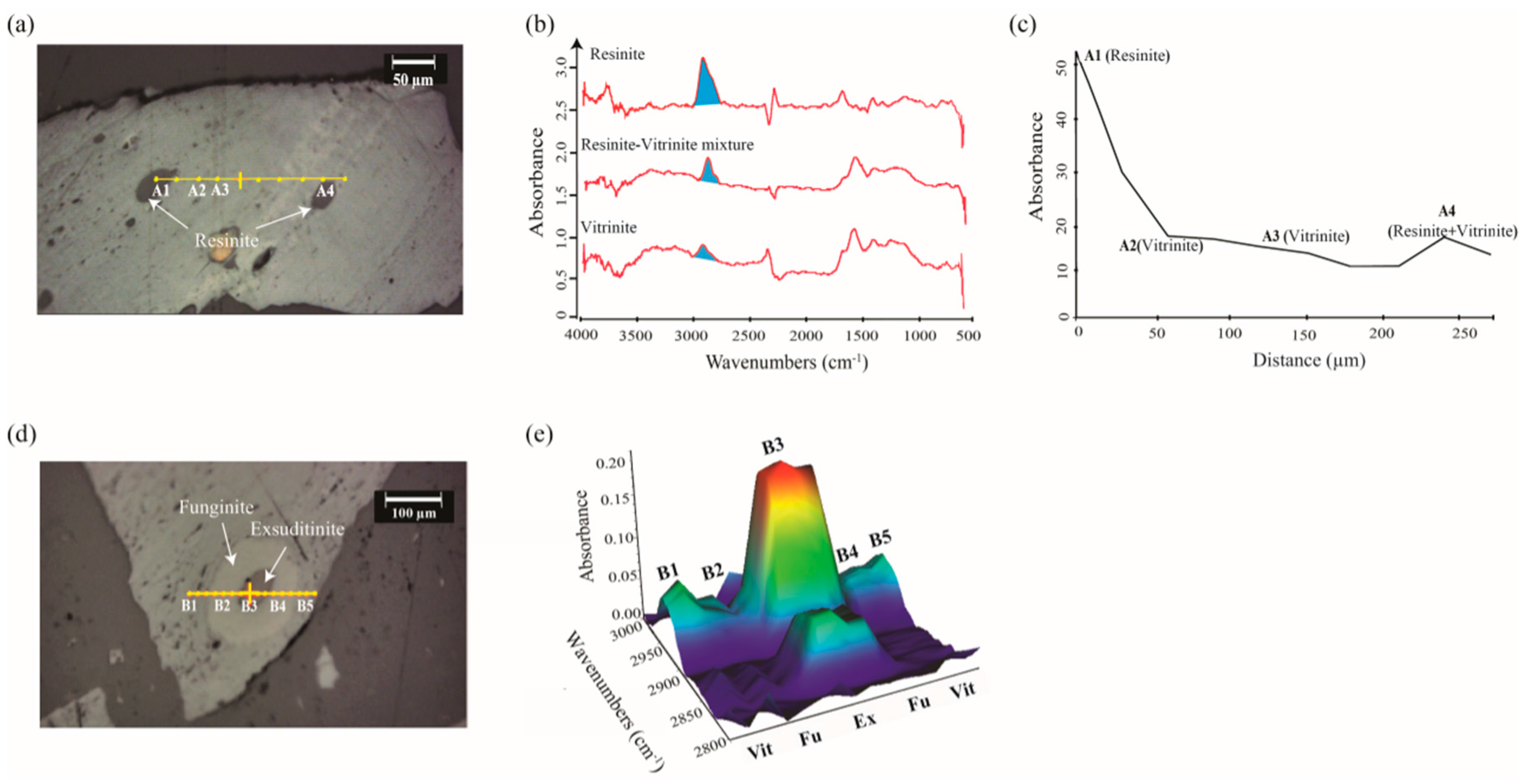
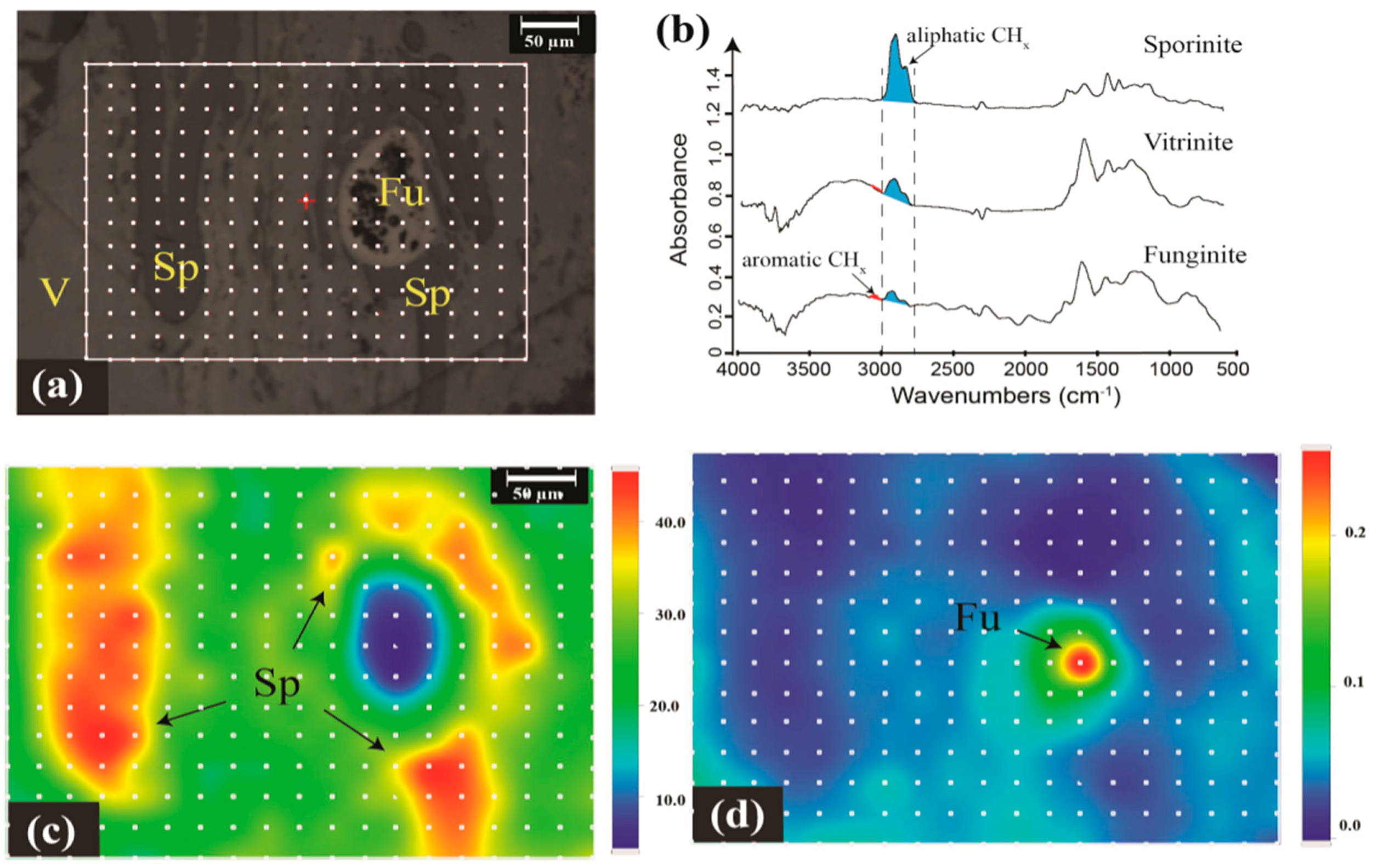
2.1.2. Maceral Coalification
2.2. Shale
2.2.1. Geochemistry of Organic Matter and Minerals in Shale
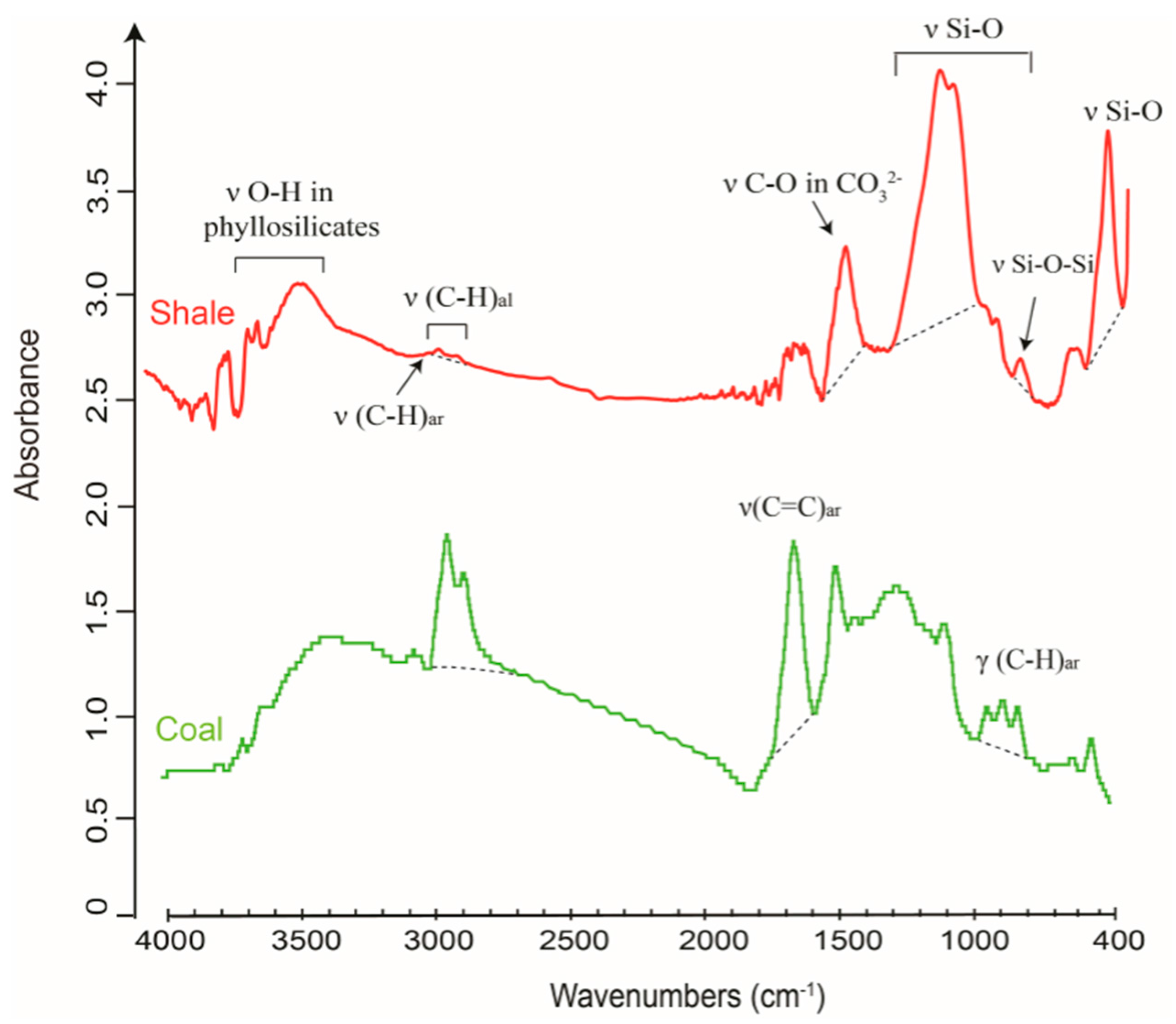
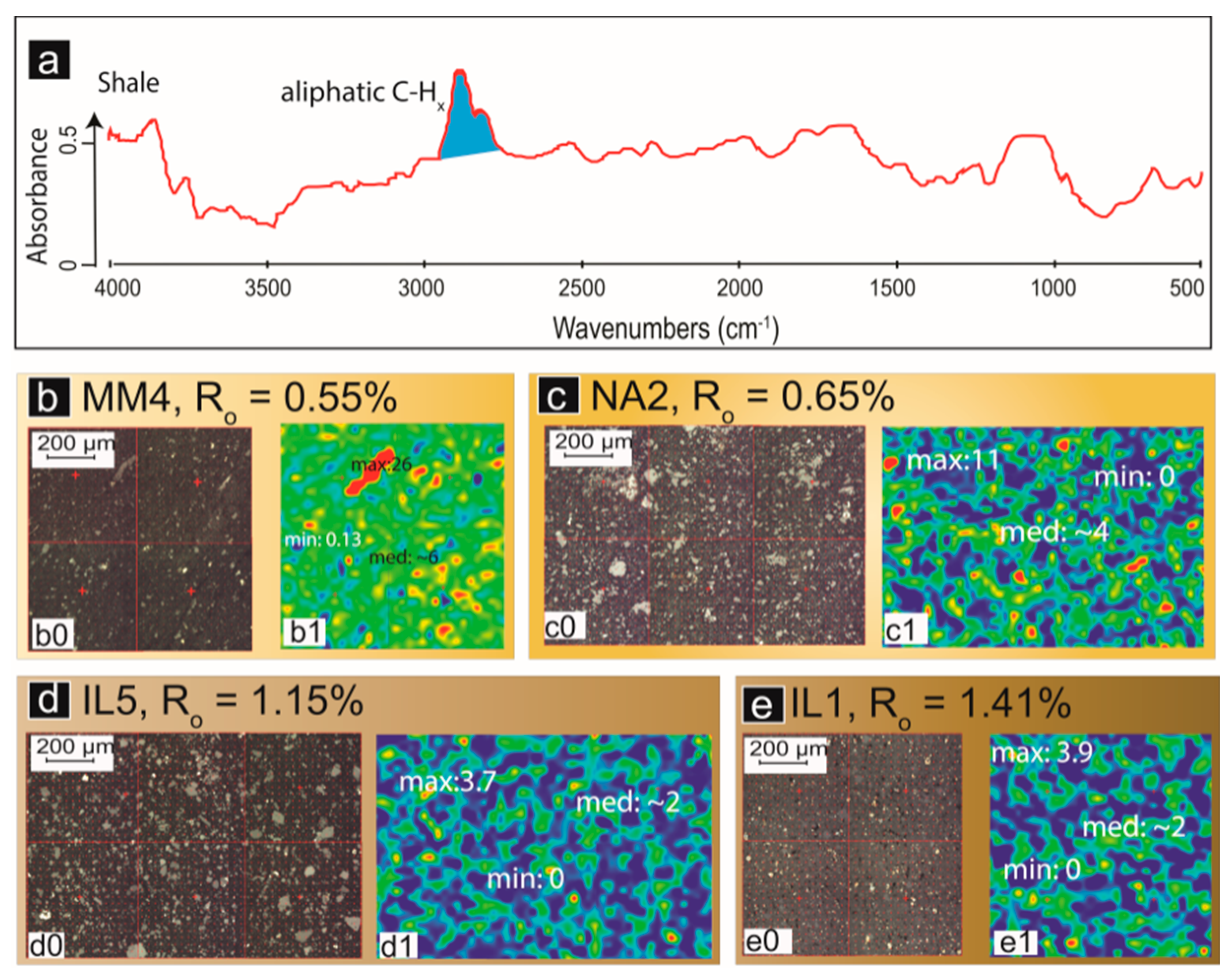
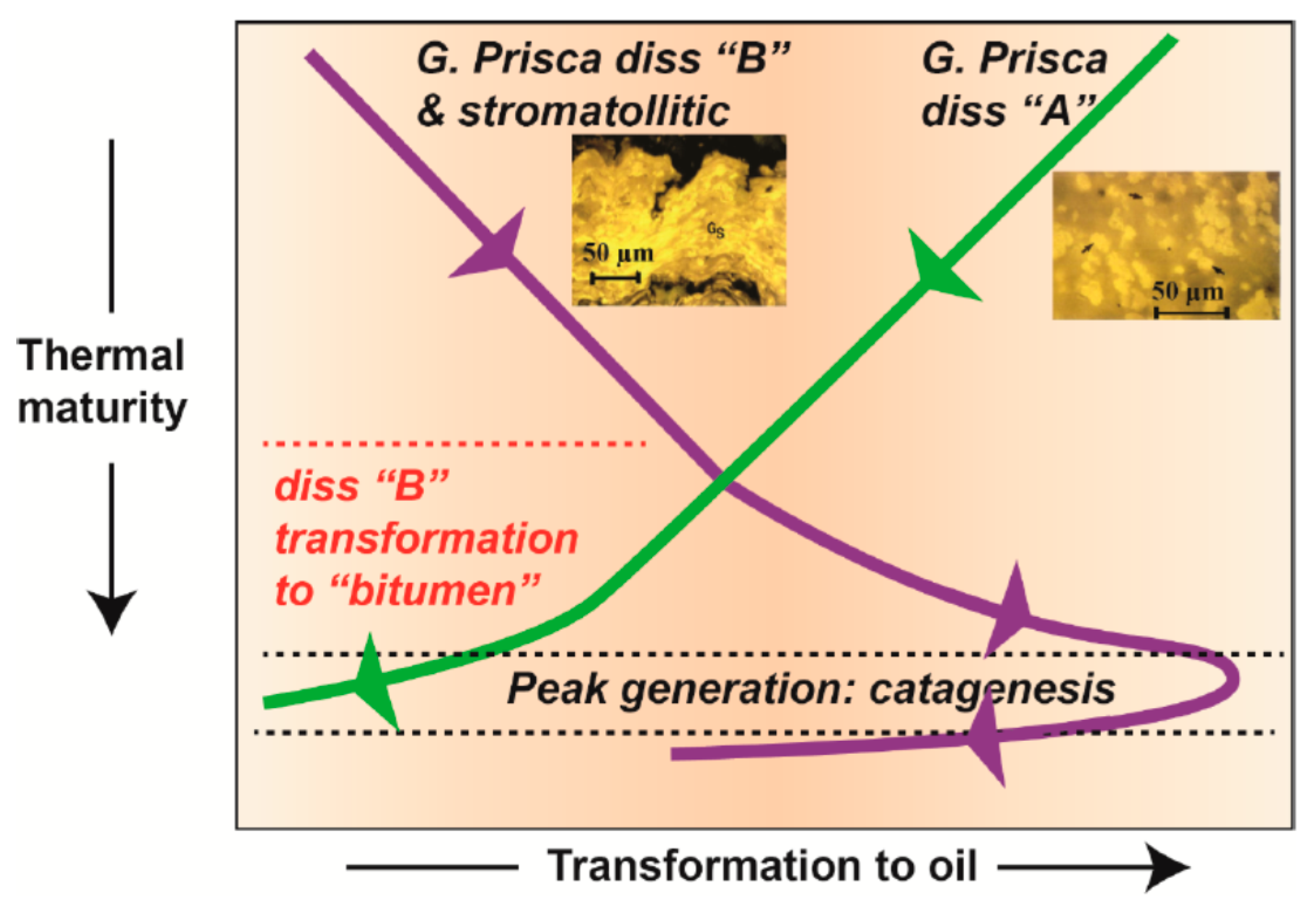
2.2.2. FTIR Spectral Data as a Proxy for Porosity and Permeability
2.3. Quantification of Organic Matter and Minerals in Coal and Shale
2.4. Limitations of Micro-FTIR in Coal and Shale Studies
3. Other Geological Application of Micro-FTIR
3.1. Application to the Analysis of Volatiles in Glasses and Minerals
3.2. Application to the Characterization of Fluid and Melt Inclusions
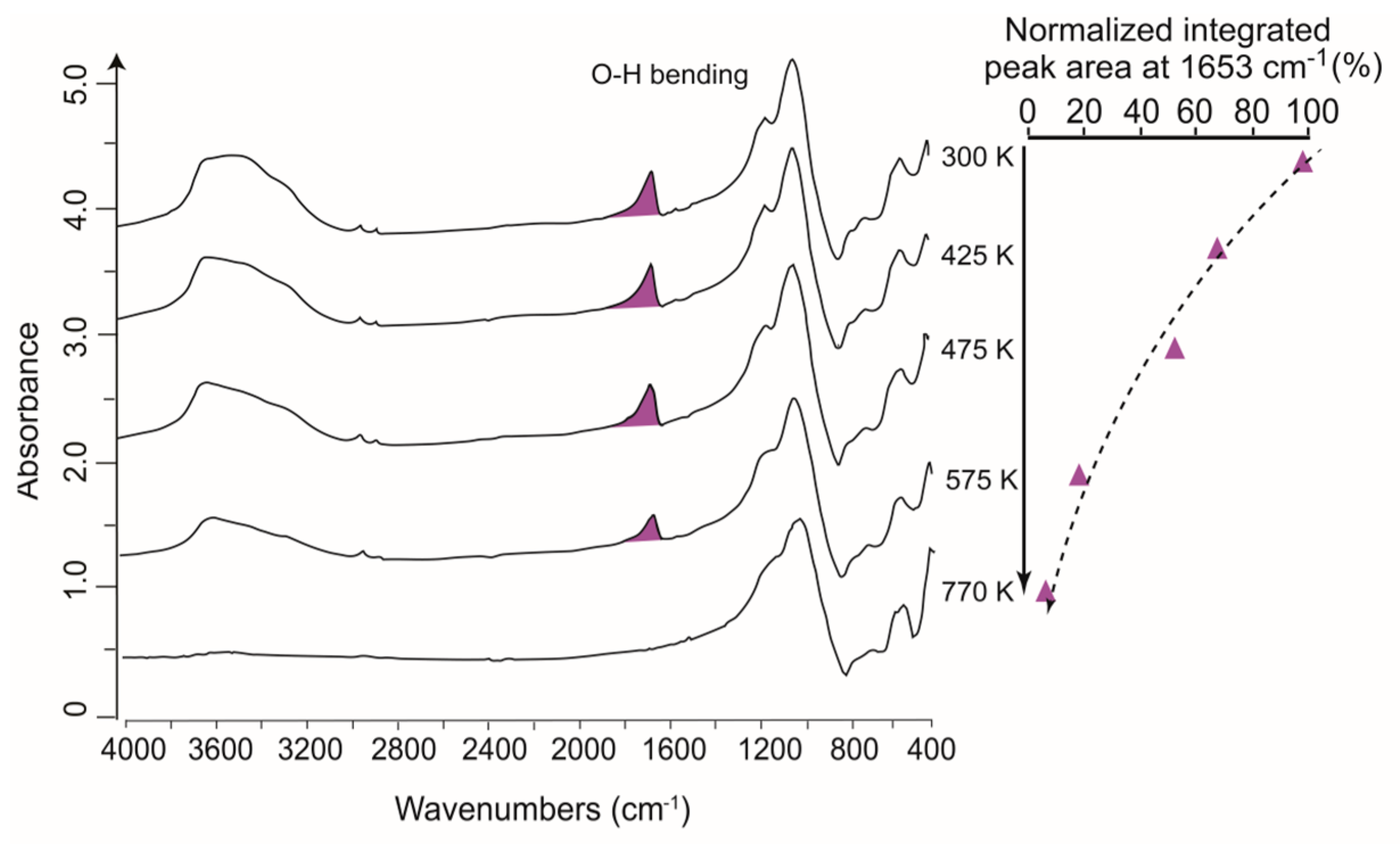
3.3. Application to High-Temperature Studies
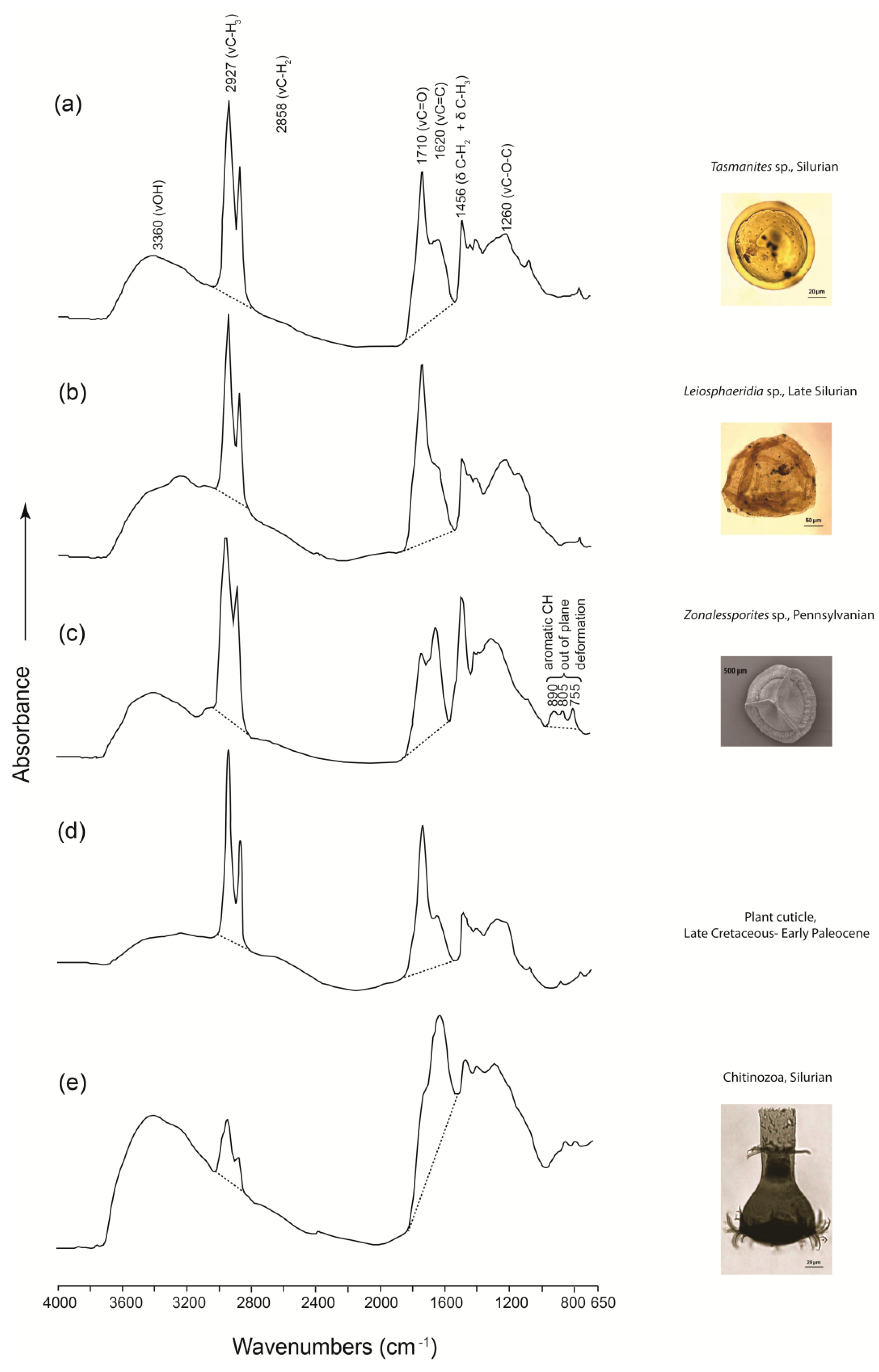
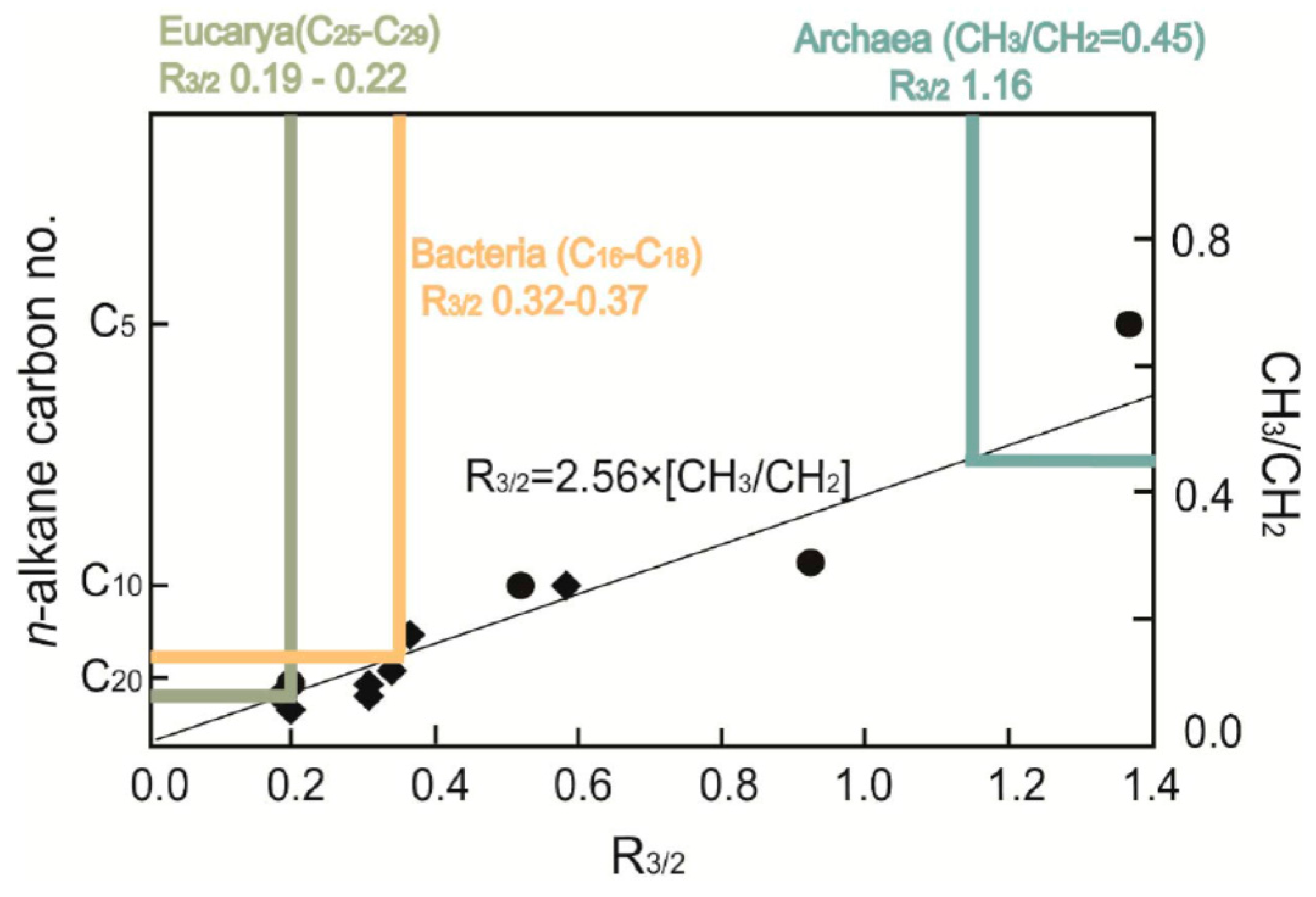
3.4. Application to Microfossil Characterization and Chemotaxonomy
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Iglesias, M.J.; Jiménez, A.; Laggoun-Défarge, F.; Suárez-Ruiz, I. FTIR study of pure vitrains and associated coals. Energy Fuels 1995, 9, 458–466. [Google Scholar] [CrossRef]
- Lis, G.P.; Mastalerz, M.; Schimmelmann, A.; Lewan, M.D.; Stankiewicz, B.A. FTIR absorption indices for thermal maturity in comparison with vitrinite reflectance R0 in type-II kerogens from Devonian black shales. Org. Geochem. 2005, 36, 1533–1552. [Google Scholar] [CrossRef]
- Behrens, H.; Romano, C.; Nowak, M.; Holtz, F.; Dingwell, D.B. Near-infrared spectroscopic determination of water species in glasses of the system MAlSi3O8 (M = Li, Na, K): An interlaboratory study. Chem. Geol. 1996, 128, 41–64. [Google Scholar] [CrossRef]
- Rossman, G.R. Analytical methods for measuring water in nominally anhydrous minerals. Rev. Mineral. Geochem. 2006, 62, 1–28. [Google Scholar] [CrossRef]
- Von Aulock, F.W.; Kennedy, B.M.; Schipper, C.I.; Castro, J.M.; Martin, D.E.; Watkins, J.M.; Wallace, P.J.; Puskar, L.; Bégué, F.; Nochols, A.R.L.; et al. Advances in Fourier transform infrared spectroscopy of natural glasses: From sample prepration to data analysis. Lithos 2014, 206–207, 52–64. [Google Scholar] [CrossRef]
- D’Angelo, J.A.; Zodrow, E.L. Chemometric study of functional groups in different layers of Trigonocarpus grandis ovules (Pennsylvanian seed fern, Canada). Org. Geochem. 2011, 42, 1039–1054. [Google Scholar] [CrossRef]
- Griffiths, P.R.; de Haseth, J.A. Fourier Transform Infrared Spectrometry, 2nd ed.; John Wiley & Sons, Inc.: New York, NY, USA, 2007. [Google Scholar]
- Parikh, S.J.; Chorover, J. FTIR spectroscopic study of biogenic Mn-oxide formation by Pseudomonas putida GB-1. Geomicrobiol. J. 2005, 22, 207–218. [Google Scholar] [CrossRef]
- Smith, B.C. Fundamentals of Fourier Infrared Spectroscopy, 2nd ed.; CRC Press, Taylor & Francis Group: Boca Raton, FL, USA, 2011. [Google Scholar]
- Zhang, Y.; Xu, Z.; Behrens, H. Hydrous species geospeedmeter in rhyolite: Improved calibration and application. Geochim. Cosmochim. Acta 2000, 64, 3347–3355. [Google Scholar] [CrossRef]
- Di Matteo, V.; Carroll, M.R.; Behrens, H.; Vetere, F.; Brooker, R.A. Water solubility in trachytic melts. Chem. Geol. 2004, 213, 187–196. [Google Scholar] [CrossRef]
- Ohlhorst, S.; Behrens, H.; Holtz, F. Compositional dependence of molar absorptivities of near-infrared OH- and H2O bands in rhyolitic to basaltic glasses. Chem. Geol. 2001, 174, 5–20. [Google Scholar] [CrossRef]
- Dixon, J.E.; Stopler, E.; Delaney, J.R. Infrared spectropic measurements of CO2 and H2O IN Juan de Fuca Ridge basaltic glasses. Earth Planet. Sci. Lett. 1988, 90, 87–104. [Google Scholar] [CrossRef]
- Stopler, E.M. The speciation of water in silicate melts. Geochim. Cosmochim. Acta 1982, 46, 2609–2620. [Google Scholar]
- Stopler, E.M. Water in silicate glasses: An infrared spectroscopic study. Contrib. Mineral. Petrol. 1982, 81, 1–17. [Google Scholar]
- Ibarra, J.V.; Muñoz, E.; Moliner, R. FTIR study of the evolution of coal structure during the coalification process. Org. Geochem. 1996, 24, 725–735. [Google Scholar] [CrossRef]
- Schrader, B. Infrared and Raman Spectroscopy: Methods and Applications; VCH Verlagsgesellschaft mbH: Weinheim, Germany, 1995. [Google Scholar]
- Li, Z.; Fredericks, P.M.; Rintoul, L.; Ward, C.R. Application of attenuated total reflectance micro- Fourier transform infrared (ATR-FTIR) spectroscopy ot the study of coal macerals: Examples from the Bowen Basin, Australia. Int. J. Coal Geol. 2007, 70, 87–94. [Google Scholar] [CrossRef]
- Li, Z.; Fredericks, P.M.; Ward, C.R.; Rintoul, L. Chemical functionalities of high and low sulfur Australian coals: A case study using micro attenuated total reflectance–Fourier transform infrared (ATR-FTIR) spectrometry. Org. Geochem. 2010, 41, 554–558. [Google Scholar] [CrossRef]
- Borer, P.; Hug, S.J.; Sulzberger, B.; Kraemer, S.M.; Kretzschmar, R. ATR-FTIR spectroscopic study of the adsorption of desferrioxamine B and aerobactin to the surface of lepidocrocite (γ-FeOOH). Geochim. Cosmochim. Acta 2009, 73, 4661–4672. [Google Scholar] [CrossRef]
- Goyne, K.W.; Chorover, J.; Kubicki, J.D.; Zimmerman, A.R.; Brantley, S.L. Sorption of the antibiotic ofloxacin to mesoporous and nonporous alumina and silica. J. Colloid Interface Sci. 2005, 283, 160–170. [Google Scholar] [CrossRef] [PubMed]
- Harrick, N.J. Internal Reflection Spectroscopy; Wiley-Interscience: New York, NY, USA, 1967. [Google Scholar]
- Griffiths, P.R.; de Haseth, J.A. Fourier Transform Infrared Spectrometry-Application to Chemical Systems; Wiley-Interscience: New York, NY, USA, 1986. [Google Scholar]
- Greene, E.F.; Tauch, S.; Webb, E.; Amarasiriwardena, D. Application of diffuse reflectance infrared Fourier transform spectroscopy (DRIFTS) for the identification of potential diagensis and crystallinity changes in teeth. Microchem. J. 2004, 76, 141–149. [Google Scholar] [CrossRef]
- Supaluknari, S.; Larkins, F.P. An FTIR study of Australian coals: Characterization of oxygen functional groups. Fuel Process. Technol. 1988, 19, 123–140. [Google Scholar] [CrossRef]
- Chen, Y.; Mastalerz, M.; Schimmelmann, A. Characterization of chemical functional groups in macerals across different coal ranks via micro-FTIR spectroscopy. Int. J. Coal Geol. 2012, 104, 22–33. [Google Scholar] [CrossRef]
- Landais, P.; Rochdi, A.; Largeau, C.; Derenne, S. Chemical characterization of torbanites by transmission micro-FTIR spectroscopy: Origin and extent of compositional heterogeneities. Geochim. Cosmochim. Acta 1993, 57, 2529–2539. [Google Scholar] [CrossRef]
- Kramers, H.A. La diffusion de la lumiere par les atomes. Atti Congr. Int. Fis. 1927, 2, 545–557. [Google Scholar]
- Kronig, R. On the theory of the dispersion of X-rays. J. Opt. Soc. Am. 1926, 12, 547–557. [Google Scholar] [CrossRef]
- International Committee for Coal Petrology (ICCP). International Handbook of Coal Petrography, 2nd ed.; Centre National de la Recherche Scientifique: Paris, France, 1963. [Google Scholar]
- Lin, R.; Ritz, G.P. Studying individual macerals using i.r. microspectroscopy, and implications on oil versus gas/condensate proneness and “low-rank” generation. Org. Geochem. 1993, 20, 695–706. [Google Scholar] [CrossRef]
- Mastalerz, M.; Bustin, R.M. Electron microprobe and micro-FTIR analyses applied to maceral chemistry. Int. J. Coal Geol. 1993, 24, 333–345. [Google Scholar] [CrossRef]
- Mastalerz, M.; Bustin, R.M. Variation in maceral chemistry within and between coals of varying rank: An electronic microprobe and micro-Fourier transform infra-red investigation. J. Microsc. 1993, 171, 153–166. [Google Scholar] [CrossRef]
- Mastalerz, M.; Bustin, R.M. Application of reflectance micro-Fourier transform infrared spectrometry in studying coal macerals: Comparison with other Fourier transform infrared techniques. Fuel 1995, 74, 536–542. [Google Scholar] [CrossRef]
- Mastalerz, M.; Bustin, R.M. Application of reflectance micro-Fourier Transform infrared analysis to the study of coal macerals: An example from the Late Jurassic to Early Cretaceous coals of the Mist Mountain Formation, British Columbia, Canada. Int. J. Coal Geol. 1996, 32, 55–67. [Google Scholar] [CrossRef]
- Garcia, D.; Black, J. Fourier transform infrared micro spectroscopy mapping. Applications to the vinyl siding industry. J. Vinyl Addit. Technol. 1997, 3, 200–204. [Google Scholar] [CrossRef]
- Guo, Y.; Bustin, R.M. Micro-FTIR spectroscopy of liptinite macerals in coal. Int. J. Coal Geol. 1998, 36, 259–275. [Google Scholar] [CrossRef]
- Chen, Y.; Caro, L.; Mastalerz, M.; Schimmelmann, A.; Blandón, A. Mapping the chemistry of resinite, funginite, and associated vitrinite in coal with micro-FTIR. J. Microsc. 2013, 249, 69–81. [Google Scholar] [CrossRef] [PubMed]
- Martin, M.C.; Dabat-Blondeau, C.; Unger, M.; Sedlmair, J.; Parkinson, D.Y.; Bechtel, H.A.; Illman, B.; Castro, J.M.; Keiluweit, M.; Buschke, D.; et al. 3D spectral imaging with synchrotron Fourier transform infrared spectro-microtomography. Nat. Methods 2013, 10, 861–864. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Mastalerz, M.; Schimmelmann, A. Heterogeneity of shale documented by micro-FTIR and image analysis. J. Microsc. 2014. [Google Scholar] [CrossRef] [PubMed]
- Mastalerz, M.; Hower, J.C.; Chen, Y. Microanalysis of barkinite from Chinese coals of high volatile bituminous rank. Int. J. Coal Geol. 2015, 141, 103–108. [Google Scholar] [CrossRef]
- King, P.L.; Dalby, K.D.; Lui, D.K.; Griffin, T. A new reflectance IR spectroscopy method for analyzing H2O in rhyolitic to basaltic glasses. Geochim. Cosmochim. Acta 2005, 69 (Suppl. S1), A744. [Google Scholar]
- Mongrain, J.; Larsen, J.F.; King, P.L. Rapid water exsolution, degasing, and bubble collapse observed experimentaly in K-phonolite melts. J. Volcanol. Geotherm. Res. 2008, 173, 178–184. [Google Scholar] [CrossRef]
- King, P.L.; Larsen, J.F. A micro-reflectance IR spectroscopy method for analyzing volatile species in basaltic, andesitic, phonolitic, and rhyolitic glasses. Am. Mineral. 2013, 98, 1162–1171. [Google Scholar] [CrossRef]
- Yasuda, A. A new technique using FT-IR micro-reflectance spectroscopy for measurement of water concentrations in melt inclusions. Earth Planets Space 2014, 66, 34–44. [Google Scholar] [CrossRef]
- Della Ventura, G.; Bellatreccia, F.; Marcelli, A.; Cestelli Guidi, M.; Piccinini, M.; Cavallo, A.; Piochi, M. Application of micro-FTIR imaging in the Earth Sciences. Anal. Bioanal. Chem. 2010, 297, 2039–2049. [Google Scholar] [CrossRef] [PubMed]
- Stach, E. The macerals of coal. In Stach's Textbook of Coal Petrology; Stach, E., Mackowsky, M., Teichmüller, M., Taylor, G.H., Chandra, D., Teichmüller, R., Eds.; Gebrüder Borntraeger: Berlin-Stuttgart, Germany, 1982; pp. 87–140. [Google Scholar]
- Tissot, B.; Welte, D.H. Petroleum Formation and Occurrence; Springer-Verlag: Berlin, Germany, 1978; p. 538. [Google Scholar]
- O’keefe, J.M.K.; Bechtel, A.; Christanis, K.; Dai, S.; DiMichele, W.A.; Eble, C.F.; Esterle, J.S.; Mastalerz, M.; Raymond, A.L.; Valentim, B.V.; et al. On the fundamental difference between coal rank and coal type. Int. J. Coal Geol. 2013, 118, 58–87. [Google Scholar] [CrossRef]
- Painter, P.C.; Starsinic, M.; Coleman, M.M. Determination of functional groups in coal by Fourier transform interferometry. In Fourier Transform Infrared Spectroscopy; Ferraro, J.R., Basile, L.J., Eds.; Academic Press: New York, NY, USA, 1985; pp. 169–240. [Google Scholar]
- Wang, S.; Griffiths, P.R. Resolution enhancement of diffuse reflectance I.R. spectra of coals by Fourier self-deconvolution. 1. C–H stretching and bending modes. Fuel 1985, 64, 229–236. [Google Scholar] [CrossRef]
- Solomon, P.R.; Carangelo, R.M. FTIR analysis of coal. 2. Aliphatic and aromatic hydrogen concentration. Fuel 1988, 67, 949–959. [Google Scholar] [CrossRef]
- Fredericks, P.M. Infrared Spectroscopy. In Spectroscopic Analysis of Coal Liquids; Kershaw, J.R., Ed.; Elsevier: New York, NY, USA, 1989; p. 395. [Google Scholar]
- Estep, P.A.; Kovach, J.J.; Karr, C., Jr. Quantitative infrared multicomponent analysis of minerals occurring in coal. Pet. Sci. Eng. 2008, 60, 1–17. [Google Scholar]
- Matteson, A.; Herron, M.M. Quantitative Mineral Analysis by Fourier Trannsform Infrared Spectroscopy. Paper No. 9308. In Pooceedings of the Society of Core Analysts Conference, Houston, TX, USA, 9–11 August 1993; pp. 1–15.
- Morga, R. Chemical structure of semifusinite and fusinite of steam and coking coal from the Upper Silesian Coal Basin (Poland) and its changes during heating as inferred from micro-FTIR analysis. Int. J. Coal Geol. 2010, 84, 1–15. [Google Scholar] [CrossRef]
- Kruge, M.A.; Landais, P.; Bensley, D.F.; Stankiewicz, B.A.; Elie, M.; Ruau, O. Speration and artificial maturation of macerals from type II kerogen. Energy Fuels 1997, 11, 503–514. [Google Scholar] [CrossRef]
- Pradier, B.; Landais, P.; Rochdi, A.; Davis, A. Chemical basis of fluorescence alteration of crude oils and kerogens—II. Fluorescence and infrared micro-spectrometric analysis of vitrinite and liptinite. Org. Geochem. 1992, 18, 241–248. [Google Scholar] [CrossRef]
- Hacura, A.; Wrzalik, R.; Matuszewska, A. Application of reflectance micro-infrared spectroscopy in coal structure studies. Anal. Bioanal. Chem. 2003, 375, 324–326. [Google Scholar] [PubMed]
- Sun, X. The investigation of chemical structure of coal macerals via transmitted-light FT-IR microspectroscopy. Spectrochim. Acta Part A 2005, 62, 557–564. [Google Scholar]
- Stankiewicz, B.A.; Kruge, M.A.; Mastalerz, M. A geochemical study of macerals from a Miocene lignite and an Eocene bituminous coal, Indonesia. Org. Geochem. 1996, 24, 531–545. [Google Scholar] [CrossRef]
- Killops, S.; Killops, V. Introduction to Organic Geochemistry, 2nd ed.; Blackwell Science Ltd.: Malden, MA, USA, 2005. [Google Scholar]
- Hutton, A.C.; Cook, A.C. Influence of alginite on the reflectance of vitrinite from Joadja, NSW, and some other coals and oil shales containing alginite. Fuel 1980, 59, 711–714. [Google Scholar] [CrossRef]
- Suárez-Ruiz, I.; Jiménez, A.; Iglesias, M.J.; Laggoun-Defarge, F.; Prado, J.G. Influence of resinite on huminite properties. Energy Fuels 1994, 8, 1417–1424. [Google Scholar] [CrossRef]
- Seewald, J.S. Organic-inorganic interactions in petroleum-producing sedimentary basins. Nature 2003, 426, 327–333. [Google Scholar] [CrossRef] [PubMed]
- Drobniak, A.; Mastalerz, M. Chemical evolution of Miocene wood: Example from the Belchatow brown coal deposit, central Poland. Int. J. Coal Geol. 2006, 66, 157–178. [Google Scholar] [CrossRef]
- Mursito, A.R.; Hirajima, T.; Sasaki, Keiko. Upgrading and dewatering of raw tropical peat by hydrothermal treatment. Fuel 2010, 89, 635–641. [Google Scholar] [CrossRef]
- Schimmelmann, A.; Mastalerz, M.; Gao, L.; Sauer, P.E.; Topalov, K. Dike intrusions into bituminous coal, Illinois Basin: H, C, N, O isotopic responses to rapid and brief heating. Geochim. Cosmochim. Acta 2009, 73, 6264–6281. [Google Scholar] [CrossRef]
- Ruau, O.; Landais, P.; Michels, R.; Langlois, E. Evaluation of organic matter reactivity during pyrolysis by micro-FTIR technqiues. Prepr. Am. Chem. Soc. 1997, 42, 164–167. [Google Scholar]
- Robl, T.L.; Davis, B.H. Comparison of the HF-HCl and HF-BF3 maceration techniques and the chemistry of resultant organic concentrates. Org. Geochem. 1993, 20, 249–255. [Google Scholar] [CrossRef]
- Schimmelmann, A.; Lewan, M.D.; Wintsch, R.P. D/H isotope ratios of kerogen, bitumen, oil, and water in hydrous pyrolysis of source rocks containing kerogen types I, II, IIS, and III. Geochim. Cosmochim. Acta 1999, 63, 3751–3766. [Google Scholar] [CrossRef]
- Alstadt, K.N.; Katti, D.R.; Katti, K.S. An in situ FTIR step-scan photoacoustic investigation of kerogen and mienrals in oil shale. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2011, 89, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Furmann, A.; Mastalerz, M.; Schimmelmann, A. Quantitative analysis of shale with KBr-FTIR and novel micro-FTIR. Fuel 2014b, 116, 538–549. [Google Scholar] [CrossRef]
- Jarvie, D.M. Total organic carbon (TOC) analysis. In Source and Migration Processes and Evalutaion Techniques: AAPG Treatise of Petroleum Geology, Handbook of Petroleum Geology; Merrill, R.K., Ed.; American Association pf Petroleum Geoologist: Tulsa, OH, USA, 1991; pp. 113–118. [Google Scholar]
- Jarvie, D.M.; Hill, R.J.; Ruble, T.E.; Pollastro, R.M. Unconventional shale-gas systems: The Mississippian Barnett Shale of north-central Texas as one model for thermogenic shale-gas assessment. AAPG Bull. 2007, 91, 475–499. [Google Scholar] [CrossRef]
- Loucks, R.G.; Reed, R.M.; Ruppel, S.C.; Jarvie, D.M. Morphology, genesis, and distriubtion of nanometer-scale pores in siliceous mudstons of the Mississippian Barnett Shale. J. Sediment. Res. 2009, 79, 848–861. [Google Scholar] [CrossRef]
- Stasiuk, L.D.; Kybett, B.D.; Bend, S.L. Reflected light microscopy and micro-FTIR of Upper Ordovician Gloeocapsomorpha prisca alginite in relation to paleoenvironment and petroleum generation, Saskatchewan, Canada. Org. Geochem. 1993, 20, 707–719. [Google Scholar] [CrossRef]
- Suarez-Rivera, R.; Deenadayalu, C.; Chertov, M.; Hartanto, R.N.; Gathogo, P.; Kunjir, R. Improving Horizontal Completions on Heterogeneous Tight Shales. In Proceedings of the Canadian Unconventional Resources Conference, Clagary, AB, Canda, 15–17 November 2011.
- Suarez-Rivera, R.; Chertov, M.; Dean, W.; Sidney, G.; John, K. Understanding Permeability Measruments in Tight Shales Promotes Enhanced Determination of Reservoir Quality. In Proceedings of the SPE Canadian Unconventional Resources Conderence, Clagary, AB, Canda, 30 October–1 November 2012.
- Ross, D.J.K.; Bustin, R.M. The importance of shale composition and pore structure upon gas storage potential of shale gas reservoirs. Mar. Petr. Geol. 2009, 26, 916–927. [Google Scholar] [CrossRef]
- Mastalerz, M.; He, L.; Melnichenko, Y.B.; Rupp, J.A. Porosity of coal and shale: Insights from gas adsorption and SANS/USANS techniques. Energy Fuels 2012, 26, 5109–5120. [Google Scholar] [CrossRef]
- Sorby, H.C. On the application of quantitative methods to the study of the structure and history of rocks. Q. J. Geol. Soc. Lond. 1908, 64, 171–232. [Google Scholar] [CrossRef]
- Painter, P.C.; Coleman, M.M.; Jenkins, R.G.; Whang, P.W.; Walker, P.L. Fourier Transform Infrared study of mineral matter in coal. A novel method for quantitative mineralogical analysis. Fuel 1978, 57, 337–344. [Google Scholar] [CrossRef]
- Cronauer, D.C.; Snyder, R.W.; Painter, P.C. Characterization of oil shale by FTIR spectroscopy. Abstr. Pap. Am. Chem. Soc. 1982, 27, 122–130. [Google Scholar]
- Brown, J.M.; Elliott, J.J. Quantitative analysis of minerals in oil shales by Fourier transform infrared spectroscopy. Am. Chem. Soc. Div. Petr. Chem. 1987, 32, 65–70. [Google Scholar]
- Charsky, A.; Herron, M.M. Quantitative Analysis of Kerogen Content and Mineralogy in Shale Cuttings by Diffuse Reflectance Infrared Fourier Transform Spectroscopy. In Proceedings of the International Symposium of the Society of Core Analysts, Aberdeen, UK, 27–30 August 2012.
- Solomon, P.R.; Miknis, F.P. Use of Fourier transform infrared spectroscopy for determining oil shale properties. Fuel 1980, 59, 893–896. [Google Scholar] [CrossRef]
- Bertaux, J.; Fröhich, F.; Ildefonse, P. Multicomponent analysis of FTIR spectra: Quantification of amorphous and crystallized mineral phases in synthetic and natural sediments. J. Sediment. Res. 1998, 68, 440–447. [Google Scholar] [CrossRef]
- Ballard, B.D. Quantitative mineralogy of reservoir rocks using Fourier transform infrared spectroscopy. In Proceedings of the Annual Technical Conference and Exhibition, Anaheim, CA, USA, 11–14 November 2007.
- Antoon, M.K.; Koenig, J.H.; Koenig, J.L. Least-squres curve-fitting of Fourier transform infrared spectra with appplications to polymer systems. Appl. Spectrosc. 1977, 31, 518–524. [Google Scholar] [CrossRef]
- Saarinen, P.; Kauppinen, J. Multicomponent analysis of FT-IR spectra. Appl. Spectrosc. 1991, 45, 953–963. [Google Scholar] [CrossRef]
- Herron, M.M.; Matteson, A.; Gustavson, G. Dual Range FT-IR Mineralogy and the Analysis of Sedimentary Formation. In Proceedings of the International Symposium of the Society of Core Analysts, Calgary, Canada, 7–10 September 1997; pp. 1–12.
- Breen, C.; Clegg, F.; Herron, M.M.; Hild, G.P.; Hillier, S.; Hughes, T.L.; Jones, T.G.J.; Matteson, A.; Yarwood, J. Bulk mineralogical characterization of oilfield reservoir rocks and sandstones using Diffuse Reflectance Infrared Fourier Transform Spectroscopy and Partial Least Squares analysis. Pet. Sci. Eng. 2008, 60, 1–17. [Google Scholar] [CrossRef]
- Washburn, K.E.; Birdwell, J.E. Multivariate analysis of ATR-FTIR spectra for assessment of oil shale organic geochemical properties. Org. Geochem. 2013, 63, 1–7. [Google Scholar] [CrossRef]
- Sparks, R.S.J. The dynamics of buble formation and growth in magmas: A review and analysis. J. Volcanol. Geotherm. Res. 1978, 3, 1–37. [Google Scholar] [CrossRef]
- Johnson, E.A. Water in nominally anhydrous crustal minerals: Speciation, concentration, and geological significance. Rev. Mineral. Geochem. 2006, 62, 117–154. [Google Scholar] [CrossRef]
- McIntosh, I.M.; Llewellin, E.W.; Humphreys, M.C.S.; Nichols, A.R.L.; Burgisser, A.; Schipper, C.I.; Larsen, J.F. Distribution of dissolved water in magmatic glass records growth and resorption of bubbles. Earth Planet. Sci. Lett. 2014, 401, 1–11. [Google Scholar] [CrossRef]
- Turek, A.; Riddle, C.; Cozens, B.; Tetley, N. Determination of chemical water in rock analysis by Karl Fischer titration. Chem. Geol. 1976, 17, 261–267. [Google Scholar] [CrossRef]
- von Aulock, F.W.; Nichols, A.R.L.; Kenndy, B.M.; Oze, C. Timescales of texture development in a cooling lava dome. Geochim. Cosmochim. Acta 2013, 114, 72–80. [Google Scholar] [CrossRef]
- Ryan, A.G.; Russell, J.K.; Nichols, A.R.L.; Hess, K.-U.; Porritt, L.A. Experiments and models on H2O retrograde solubility in volcanic systems. Am. Mineral. 2015, 100, 774–786. [Google Scholar] [CrossRef]
- Behrens, H.; Roux, J.; Neuville, D.R.; Siemann, M. Quantification of dissolved H2O in silicate glasses using confocal microRaman spectroscopy. Chem. Geol. 2006, 229, 96–112. [Google Scholar] [CrossRef]
- Di Muro, A.; Villemant, B.; Montagnac, G.; Scaillet, B.; Reynard, B. Quantification of water content and speciation in natural silicic glasses (phonolite, dacite, rhyolite) by confocal microRaman spectdrometry. Geochim. Cosmochim. Acta 2006, 70, 2868–2884. [Google Scholar] [CrossRef]
- Hervig, R.L.; Mazdab, F.K.; Moore, G.; McMillan, P.F. Analyzing hydrogen (H2O) in silicate glass by secondary ion mass spectrometry and reflectance Fourier transform infrared spectroscopy. In Melt Inclusions in Volcanic Systems: Methods, Applications and Problems; de Vivo, B., Bodnar, R.J., Eds.; Elsevier: New York, NY, USA, 2003; pp. 83–103. [Google Scholar]
- Humphreys, M.C.S.; Menand, T.; Blundy, J.D.; Klimm, K. Magma ascent rates in explosive eruptions: Constraints from H2O diffusion in melt inclusions. Earth Planet. Sci. Lett. 2008, 270, 25–40. [Google Scholar] [CrossRef]
- Berlo, K.; Tuffen, H.; Smith, V.; Castro, J.M.; Pyle, D.; Mather, T.; Geraki, K. Element variations in rhyolitic magma resulting from gas transport. Geochim. Cosmochim. Acta 2013, 121, 436–451. [Google Scholar] [CrossRef]
- Ihinger, P.D.; Hervig, R.L.; McMillan, P.F. Analytical methods for volatiles in glasses. Rev. Mineral. 1994, 30, 67–121. [Google Scholar]
- Nieuwoudt, M.K.; Simpson, M.P.; Tobin, M.; Puskar, L. Synchrotron FTIR microscopy of synthetic and natural CO2-H2O fluid inclusions. Vib. Spectrosc. 2014, 75, 136–148. [Google Scholar] [CrossRef]
- Lowenstern, J.B.; Pitcher, B.W. Analysis of H2O in silicate glass using attenuated total reflectance (ATR) micro-FTIR spectroscopy. Am. Mineral. 2013, 98, 1660–1668. [Google Scholar] [CrossRef]
- Dixon, J.E.; Pan, V. Determination of the molar absorptivity of dissolved carbonate in basaltic glass. Am. Mineral. 1995, 80, 1339–1342. [Google Scholar]
- Mormone, A.; Piochi, M.; Bellatreccia, F.; de Astis, G.; Moretti, R.; Della Ventura, G.; Cavallo, A.; Mangiacapra, A. A CO2-rich magma source beneath the Phlegraean Volcanic District (Southern Italy): Evidence from a melt inclusion study. Chem. Geol. 2011, 287, 66–80. [Google Scholar] [CrossRef]
- Lange, R.A.; Carmichael, I.S. Densities of Na2O-K2O-MgO-FeO-Fe2O3-Al2O3-TiO2-SiO2 liquids: New measurements and derived partial molar properties. Geochim. Cosmochim. Acta 1987, 51, 2931–2946. [Google Scholar] [CrossRef]
- Ochs, F.A., III; Lange, R.A. The density of hydrous magmatic liquids. Science 1999, 283, 1314–1317. [Google Scholar] [CrossRef] [PubMed]
- Tamic, N.; Behrens, H.; Holtz, F. The solubility of H2O and CO2 in rhyolitic melts in equilibrium with a mixed CO2-H2O fluid phase. Chem. Geol. 2001, 174, 333–347. [Google Scholar] [CrossRef]
- Wysoczanski, R.; Tani, K. Spectroscopic FTIR imaging of water species in silicic volcanic glasses and melt inclusions: An example from the Izu-Bonin arc. J. Volcanol. Geotherm. Res. 2006, 156, 302–314. [Google Scholar] [CrossRef]
- Nichols, A.R.L.; Wysoczanski, R.J. Using micro-FTIR spectroscopy to measure volatile contents in small and unexposed inclusions hosted in olivine crystals. Chem. Geol. 2007, 242, 371–384. [Google Scholar] [CrossRef]
- Castro, J.M.; Manga, M.; Martin, M.C. Vesiculation rates of obsidian domes inferred from H2O concentration profiles. Geophys. Res. Lett. 2005, 32, 1–5. [Google Scholar] [CrossRef]
- Castro, J.M.; Dingwell, D.B. Rapid ascent of rhyolitic magma at Chaitén volcano, Chile. Nature 2009, 461, 780–784. [Google Scholar] [CrossRef] [PubMed]
- Watkins, J.M.; Manga, M.; DePaolo, D.J. Bubble geobarometry: A record of pressure changes, degassing, and regassing at Mono Craters, California. Geology 2012, 40, 699–702. [Google Scholar] [CrossRef]
- Della Ventura, G.; Marcelli, A.; Bellatreccia, F. SR-FTIR microscopy and FTIR imaging in the Earth sciences. Rev. Mineral. Geochem. 2014, 78, 447–479. [Google Scholar] [CrossRef]
- Peslier, A.H. A review of water contents of nominally anhydrous natural minerals in the mantlels of Earth, Mars, and the Moon. J. Volcanol. Geotherm. Res. 2010, 197, 239–258. [Google Scholar] [CrossRef]
- Della Ventura, G.; Bellatreccia, F.; Parodi, G.C.; Cámara, F.; Piccinini, M. Single-crystal FTIR and X-ray study of vishnevite, ideally [Na2(SO4)][Na2(H2O)2](Si6Al6O24). Am. Mineral. 2007, 92, 713–721. [Google Scholar] [CrossRef]
- Bellatreccia, F.; Della Ventura, G.; Ottolini, L.; Libowitzky, E.; Beran, A. The quantitative analysis of OH in vesuviantie: A polirized FTIR and SIMS study. Phys. Chem. Miner. 2005, 32, 65–76. [Google Scholar] [CrossRef]
- Roedder, E. Fluid Inclusions; Mineralogical Society of America: Chantilly, VA, USA, 1984; p. 644. [Google Scholar]
- Danyushevsky, L.V.; McNeill, A.W.; Sobolev, A.V. Experimental and petrological studies of melt inclusions in phenocrysts from mantle-derived magmas: An overview of techniques, advantages and complications. Chem. Geol. 2002, 183, 5–24. [Google Scholar] [CrossRef]
- Thomas, R.; Kamenetsky, V.S.; Davidson, P. Laser Raman spectroscopic measurements of water in unexposed glass inclusions. Am. Mineral. 2006, 91, 467–470. [Google Scholar] [CrossRef]
- Guilhaumou, N.; Sautter, V.; Dumas, P. Synchrotron FTIR microanalysis of volatiles in melt inclusions and exsolved particles in ultramafic deep-seated garnets. Chem. Geol. 2005, 223, 82–92. [Google Scholar] [CrossRef]
- Mercier, M.; Di Muro, A.; Métrich, N.; Giordano, D.; Belhadj, O.; Mandeville, C.W. Spectroscopic analysis (FTIR, Raman) of water in mafic and intermediate glasses and glass inclusions. Geochim. Cosmochim. Acta 2010, 74, 5641–5656. [Google Scholar] [CrossRef]
- Pironon, J.; Barres, O. Semi-quantitative FT-IR microanalysis limits: Evidence from synthetic hydrocarbon fluid inclusions in sylvite. Geochim. Cosmochim. Acta 1990, 54, 509–518. [Google Scholar] [CrossRef]
- Pironon, J.; Barres, O. FT-IR microanalysis of hydrocarbon fluid inclusions. Chem. Geol. 1990, 84, 224–226. [Google Scholar] [CrossRef]
- Barrès, O.; Burneau, A.; Dubessy, J.; Pagel, M. Application of micro-FT-IR spectroscopy to individual hydrocarbon fluid inclusion analysis. Appl. Spectrosc. 1987, 41, 1000–1008. [Google Scholar] [CrossRef]
- Bourdet, J.; Burruss, R.C.; Chou, I-M.; Kempton, R.; Liu, K.; Hung, N.V. Evidence for a palaeo-oil column and alteration of residual oil in a gas-condensate field: Integrated oil inclusion and experimental results. Geochim. Cosmochim. Acta 2014, 142, 362–385. [Google Scholar] [CrossRef]
- Nowak, M.; Behrens, H. Water in rhyolitic magmas: Getting a grip on a slippery problem. Earth Planet. Sci. Lett. 2001, 184, 515–522. [Google Scholar] [CrossRef]
- Behrens, H.; Nowak, M. Quantification of H2O speciation in silicate glasses and melts by IR spectroscopy- in situ versus quench techniques. Phase Transit. 2003, 76, 45–61. [Google Scholar] [CrossRef]
- Prasad, R.S.R.; Prasad, K.S.; Murthy, S.R. Dehydration of natural stilbite: An in situ FTIR study. Am. Mineral. 2005, 90, 1636–1640. [Google Scholar] [CrossRef]
- Kolesov, B.A.; Geiger, C.A. Behavior of H2O molecules in the channels of natrolite and scolecite: A Raman and IR spectroscopic investigation of hydrous microporous silicates. Am. Mineral. 2006, 91, 1039–1048. [Google Scholar] [CrossRef]
- Prasad, R.S.R.; Prasad, K.S.; Chaitanya, V.K.; Babu, E.V.S.S.K.; Sreedhar, B.; Murthy, S.R. In situ FTIR study on the dehydration of natural geothite. J. Asian Earth Sci. 2006, 27, 503–511. [Google Scholar] [CrossRef]
- Prasad, R.S.R.; Prasad, K.S. Dehydration and rehydration of mesolite: An in situ FTIR study. Microporous Mesoporous Mater. 2007, 100, 287–294. [Google Scholar] [CrossRef]
- Tokiwai, K.; Nakashima, S. Integral molar absorptivities of OH in muscovite at 20 to 650 °C by in situ high-temperature IR microspectroscopy. Am. Mineral. 2010, 95, 1052–1059. [Google Scholar] [CrossRef]
- Dutta, S.; Hartkopf-Fröder, C.; Witte, K.; Brocke, R.; Mann, U. Molecular characterization of fossil palynomorphs by transmission micro-FTIR spectroscopy: Implications for hydrocarbon source evaluation. Int. J. Coal Geol. 2013, 115, 13–23. [Google Scholar] [CrossRef]
- Dutta, S.; Brocke, R.; Hartkopf-Fröder, C.; Littke, R.; Wilkes, H.; Mann, U. Highly aromatic character of biogeomacromolecules in Chitinozoa: A spectroscopic and pyrolytic study. Org. Geochem. 2007, 38, 1625–1642. [Google Scholar] [CrossRef]
- Dutta, S.; Hartkopf-Fröder, C.; Mann, U.; Wilkes, H.; Brocke, R.; Bertram, N. Macromolecular composition of Palaeozoic scolecodonts: Insights into the molecular taphonomy of zoomorphs. Lethaia 2009, 43, 334–343. [Google Scholar] [CrossRef]
- Igisu, M.; Ueno, Y.; Shimojima, M.; Nakashima, S.; Awramik, S.M.; Ohta, H.; Maruyama, S. Micro-FTIR spectroscopic signatures of Bacterial lipids in Proterozoic microfossils. Precambrian Res. 2009, 173, 19–26. [Google Scholar] [CrossRef]
- Han, J.; Calvin, M. Hydrocarbon distribution of algae and bacteria, and microbiological activity in sediments. Proc. Natl. Acad. Sci. USA 1983, 64, 436–443. [Google Scholar] [CrossRef]
- Ladygina, N.; Dedyukhina, E.G.; Vainshtein, M.B. A review on microbial synthesis of hydrocarbons. Process Biochem. 2006, 41, 1001–1014. [Google Scholar] [CrossRef]
- Igisu, M.; Komiya, T.; Kawashima, M.; Nakashima, S.; Ueno, Y.; Han, J.; Shu, D.; Li, Y.; Guo, J.; Maruyama, S.; Takai, K. FTIR microspectroscopy of Ediacaran phosphatized microfossils from the Doushantuo Formation, Weng’an, South China. Gondwana Res. 2014, 25, 1120–1138. [Google Scholar] [CrossRef]
- Zodrow, E.L.; Mastalerz, M.; Orem, W.H.; Šimůnek, Z.; Bashforth, A.R. Functional groups and elemental analyses of cuticular morphotypes of Cordaites principalies (Germar) Geinitz, Carboniferous Martitimes Basin, Canada. Int. J. Coal Geol. 2000, 45, 1–19. [Google Scholar] [CrossRef]
- Zodrow, E.L.; D'Angelo, J.A.; Mastalerz, M.; Keefe, D. Compression-cuticle relationship of seed ferns: Insights from liquid-solid states FTIR (Late Palaeozoic-Early Mesozoic, Canada-Spain-Argentina). Int. J. Coal Geol. 2009, 779, 61–73. [Google Scholar] [CrossRef]
- D’Angelo, J.A.; Zodrow, E.L.; Camargo, A. Chemometric study of functional groups in Pennsylvanian gymnosperm plant organs (Sydney Coalfield, Canada): Implications for chemotaxonomy and assessment of kerogen formation. Org. Geochem. 2010, 41, 1312–1325. [Google Scholar] [CrossRef]
- D’Angelo, J.A.; Zodrow, E.L.; Mastalerz, M. Compression map, funcitonal groups and fossilization: A chemometric approach (Pennsylvanian neuropteroid foliage, Canada). Int. J. Coal Geol. 2012, 90–91, 149–155. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, Y.; Zou, C.; Mastalerz, M.; Hu, S.; Gasaway, C.; Tao, X. Applications of Micro-Fourier Transform Infrared Spectroscopy (FTIR) in the Geological Sciences—A Review. Int. J. Mol. Sci. 2015, 16, 30223-30250. https://doi.org/10.3390/ijms161226227
Chen Y, Zou C, Mastalerz M, Hu S, Gasaway C, Tao X. Applications of Micro-Fourier Transform Infrared Spectroscopy (FTIR) in the Geological Sciences—A Review. International Journal of Molecular Sciences. 2015; 16(12):30223-30250. https://doi.org/10.3390/ijms161226227
Chicago/Turabian StyleChen, Yanyan, Caineng Zou, Maria Mastalerz, Suyun Hu, Carley Gasaway, and Xiaowan Tao. 2015. "Applications of Micro-Fourier Transform Infrared Spectroscopy (FTIR) in the Geological Sciences—A Review" International Journal of Molecular Sciences 16, no. 12: 30223-30250. https://doi.org/10.3390/ijms161226227
APA StyleChen, Y., Zou, C., Mastalerz, M., Hu, S., Gasaway, C., & Tao, X. (2015). Applications of Micro-Fourier Transform Infrared Spectroscopy (FTIR) in the Geological Sciences—A Review. International Journal of Molecular Sciences, 16(12), 30223-30250. https://doi.org/10.3390/ijms161226227