The Historical Development of Immunoendocrine Concepts of Psychiatric Disorders and Their Therapy
Abstract
:1. Introduction
2. The Development of Endocrine Concepts of Psychiatric Disorders
2.1. Historical Preliminary Remarks
2.2. The Use of Castration to Modify Behavior in Animals and Human Being
2.3. Humorism and Mental Illness
2.4. Psychopathological Symptoms in Circumscribed Endocrine Disorders

2.5. The Discovery of the Hormones
2.6. The Emergence and Systematic Development of Endocrine Concepts of Illness in Psychiatry
2.7. Therapeutic Use of Various Hormones in Psychiatry
2.8. Technical Developments for Establishing Hormone Concentration in the Blood
3. The Development of Immunological Concepts of Psychiatric Disorders
3.1. Observations and Hypotheses in the Ancient World
3.2. Measuring Body Temperature and Blood Analysis
3.3. The Emergence of Immunology
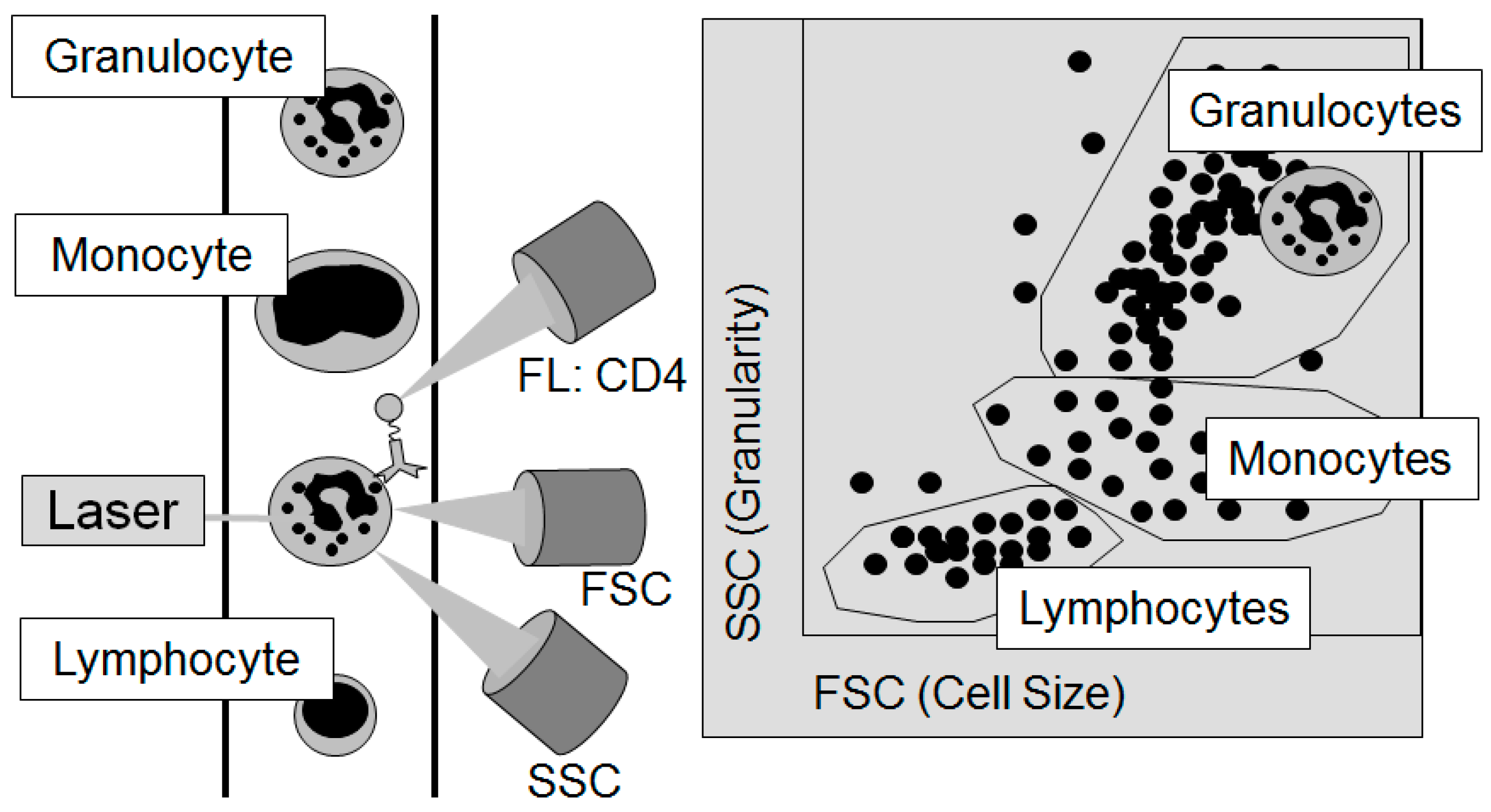
3.4. Technical Developments in Measurement of Cytokines and Immunologically Relevant Cells
4. Current Immunoendocrine Concepts of Psychiatric Disorders and Their Therapy
4.1. Current Hormone-Based Concepts of Depression
4.2. Current Hormone-Based Concepts of Other Psychiatric Disorders
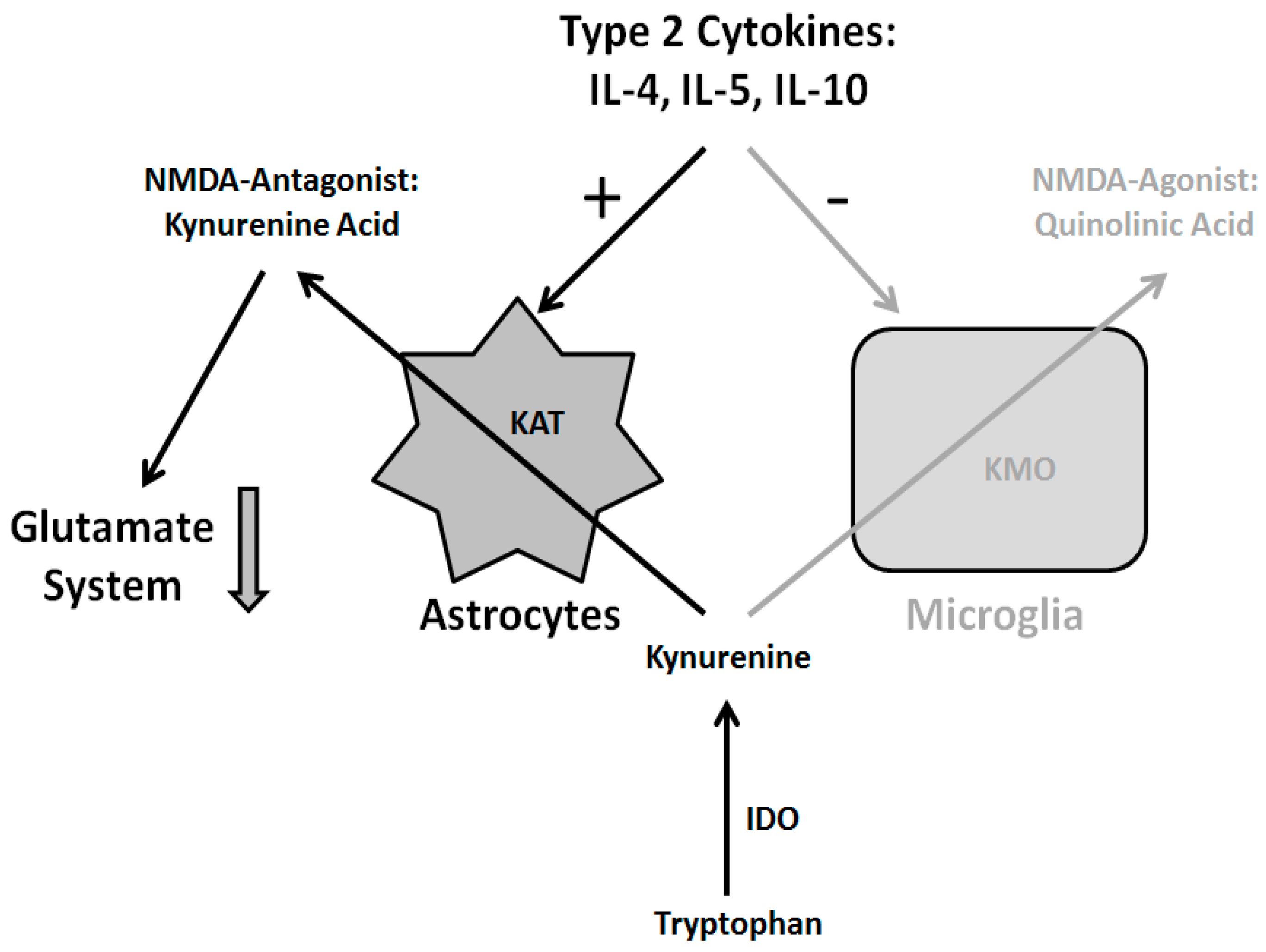
4.3. Current Immunological Concepts of Schizophrenia
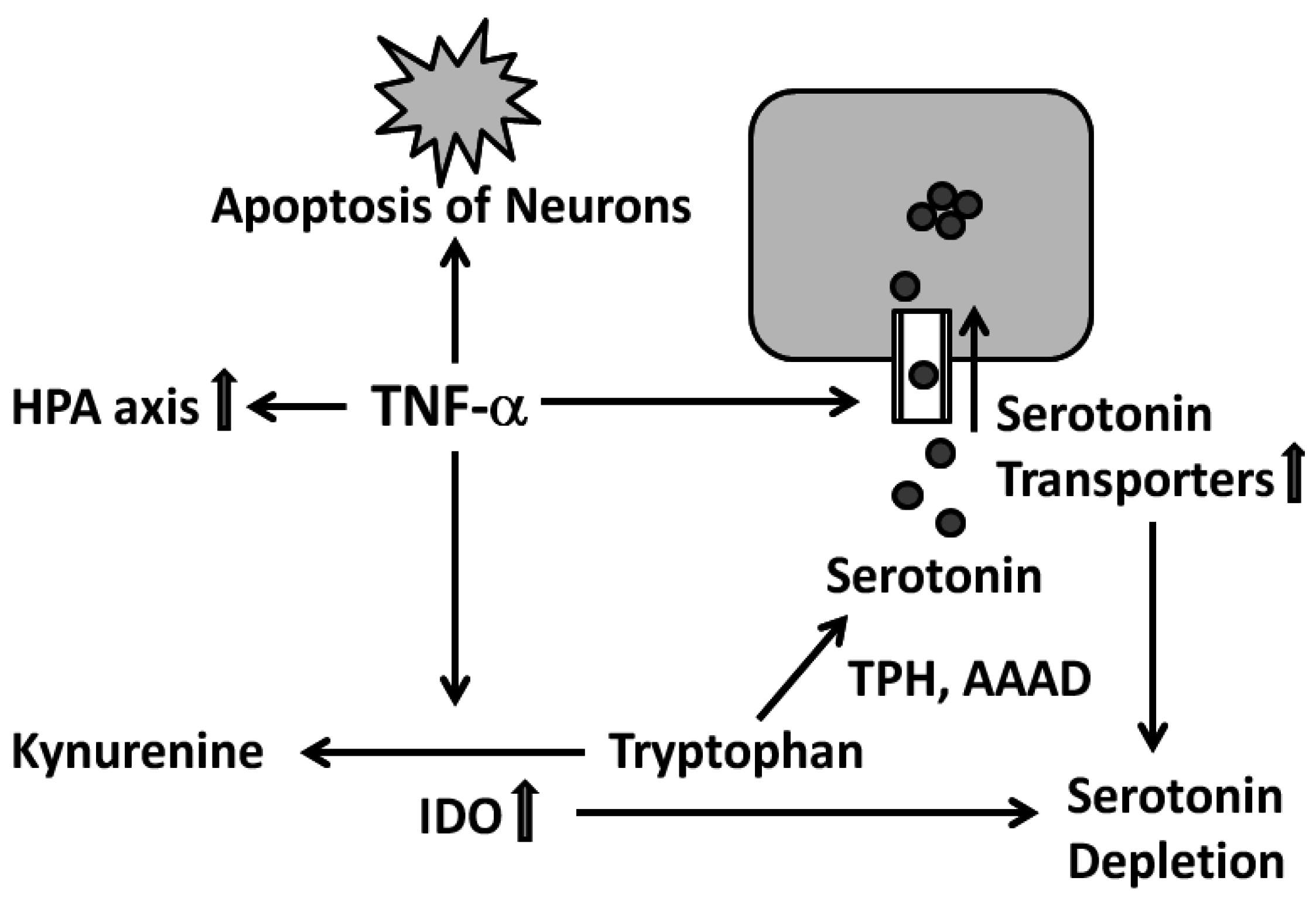
4.4. Current Immunological Concepts of Deprezssion
5. Perspectives of Immunoendocrine Concepts of Psychiatric Disorders and Their Therapy
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Müller, N.; Schwarz, M.J. Neuroimmune-endocrine crosstalk in schizophrenia and mood disorders. Expert Rev. Neurother. 2006, 6, 1017–1038. [Google Scholar] [CrossRef] [PubMed]
- Himmerich, H.; Steinberg, H. Hormonelle Krankheits- und Therapiekonzepte in der Psychiatrie. Fortschr. Neurol. Psychiatr. 2011, 79, 384–394. [Google Scholar] [CrossRef] [PubMed]
- Kleine, B.; Rossmanith, W.G. Hormone und Hormonsystem. Lehrbuch der Endokrinologie, 2nd ed.; Springer: Berlin, Germany, 2010. [Google Scholar]
- Nadler, M. Tierische Arbeitskraft im Neolithikum?—Belege von Ochsen im frühen Jungneolithikum von Marktbergel, Mittelfranken. In Schleife, Schlitten, Rad und Wagen. Zur Frage früher Transportmittel nördlich der Alpen; Köninger, J., Mainberger, M., Schlichtherle, H., Eds.; Janus: Freiburg, Germany, 2002; pp. 109–110. [Google Scholar]
- Walkowitz, J.E. Logistik im Neolithikum und Chalcolithikum. In Varia Neolithica IV; Beier, H.J., Ed.; Beier & Beran: Langenweißbach, Germany, 2006; pp. 123–151. [Google Scholar]
- King James Bible (KJV) quoted from The King James Bible Online—Authorized Version. Available online: http://www.kingjamesbibleonline.org/Matthew-Chapter-19 (accessed on 14 August 2015).
- Steinberg, H. Paul Julius Möbius (1853–1907). J. Neurol. 2005, 252, 624–625. [Google Scholar] [CrossRef] [PubMed]
- Steinberg, H. Als ob ich zu einer steinernen Wand spräche. In Der Nervenarzt Paul Julius Möbius-Eine Werkbiografie; Hans Huber Verlag: Bern, Switzerland, 2005. [Google Scholar]
- Steinberg, H. Zum 150. Geburtstag von Paul Julius Möbius (1853–1907). Anmerkungen zum Titelbild. Nervenarzt 2004, 75, 97–100. [Google Scholar] [CrossRef] [PubMed]
- Möbius, P.J. Über die Wirkungen der Kastration; Marhold: Halle, Germany, 1903. [Google Scholar]
- Splett, T.; Steinberg, H. Die Therapie der Hysterie im 19. Jahrhundert—Wie stand die deutsche Psychiatrie zur Kastration? Fortschr. Neurol. Psychiatr. 2003, 71, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Steinberg, H. Diesmal hat der Referent Kraepelins neue Eintheilung mit Stolz und Befriedigung begrüsst, denn es ist seine eigene. Die ätiologische Klassifikation der Nerven- und psychischen Krankheiten von Paul Julius Möbius. Fortschr. Neurol. Psychiatr. 2006, 74, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Huonker, T. Anstaltseinweisungen, Kindswegnahmen, Eheverbote, Sterilisationen, Kastrationen-Fürsorge, Zwangsmassnahmen, “Eugenik” und Psychiatrie in Zürich zwischen 1890 und 1970; Sozialdepartement: Zürich, Switzerland, 2002. [Google Scholar]
- Druhm, K.W. A welcome return to Draconia: California Penal Law 645, the castration of sex offenders and the constitution. Albany Law Rev. 1997, 61, 285–343. [Google Scholar]
- Scott, C.L.; Holmberg, T. Castration of sex offenders: Prisoners’ rights versus public safety. J. Am. Acad. Psychiatr. Law 2003, 31, 502–509. [Google Scholar]
- Lombroso, C. Der Verbrecher in Anthropologischer, Ärztlicher und Juristischer Beziehung; Richter: Hamburg, Germany, 1887. [Google Scholar]
- Gadebusch-Bondio, M. Die Rezeption der Kriminalanthropologischen Theorien von Cesare Lombroso in Deutschland von 1880–1914; Matthiesen: Husum, Germany, 1995. [Google Scholar]
- Jensch, N. Untersuchungen an Entmannten Sittlichkeitsverbrechern. Sammlung psychiatrischer und neurologischer Einzeldarstellungen; Thieme: Leipzig, Germany, 1944. [Google Scholar]
- Wolfert, R.; Steinberg, H. Der Homosexuelle als “störende Persönlichkeit” Nikolaus Jensch und seine psychiatrisch-genetischen Studien. Fortschr. Neurol. Psychiatr. 2009, 77, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Lemke, R. Neue Auffassungen zur Pathogenese, Klinik und strafrechtlichen Stellung der männlichen und weiblichen Homosexualität. Med. Klin. 1940, 36, 1355–1357. [Google Scholar]
- Moll, A. Behandlung der Homosexualität: Biochemisch oder Psychisch? Marcus &Weber: Bonn, Germany, 1921. [Google Scholar]
- Eckart, W.U. Das Krankheits- und Therapiekonzept der hippokratischen Medizin. In Geschichte der Medizin; Eckart, W.U., Ed.; Springer: Heidelberg, Germany, 2009; pp. 33–34. [Google Scholar]
- Lanczik, M.; Bergant, A.; Klier, C. Are severe psychiatric disorders in childbed of endogenous or organic nature? German contributions to the biological, nosological and psychopathological research in postpartum psychosis in the 18th and 19th century. Arch. Womens Ment. Health 2006, 9, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Schott, H. Medizingeschichte(n) Psychiatrie. Wahn (Schwangerschaftspsychose). Dtsch. Arztebl. 2004, 101, A-2397/B-2017/C-1945. [Google Scholar]
- Wagner-Jauregg, J. Myxödem und Kretinismus; Deuticke: Leipzig, Germany, 1912. [Google Scholar]
- Kocher, T. Über Kropfexstirpation und ihre Folgen. Arch. Klin. Chir. 1883, 29, 254–337. [Google Scholar]
- Strümpell, A. Lehrbuch der Speciellen Pathologie und Therapie der Inneren Krankheiten. Für Studierende und Ärzte. Vol II: Krankheiten des Nervensystems; Vogel: Leipzig, Germany, 1884. [Google Scholar]
- Von Basedow, K.A. Exophthalmus durch Hypertrophie des Zellgewebes in der Augenhöhle. Wschrft. Ges. Heilkd. 1840, 6, 197–204. [Google Scholar]
- Möbius, P.J. Ueber das Wesen der Basedow’schen Krankheit. Cbl. Nervenheilkd. Psychiatr. 1887, 10, 225–229. [Google Scholar]
- Möbius, P.J. Vom Verhältnisse der Poliomyelencephalitis zur Basedow’schen Krankheit von Dr. E. Jendrassik. Schmidts Jb. Ges. Med. 1886, 210, 237–238. [Google Scholar]
- Hertz, S.; Roberts, A. Application of radioactive iodine in therapy of Graves’ disease. J. Clin. Investig. 1942, 21, 624. [Google Scholar]
- Astwood, E.B. Treatment of hyperthyroidism with thiourea and thiouracil. JAMA 1943, 122, 78–81. [Google Scholar] [CrossRef]
- Addison, T. On the constitutional and local effects of disease of the suprarenal capsule. In A Collection of the Published Writings of the Late Thomas Addison MD; Daldy, T.M., Wilks, S., Eds.; New Sydenham Society: London, UK, 1868; pp. 211–239. [Google Scholar]
- Cushing, H. The Pituitary Body and Its Disorders; Lippincott: Philadelphia, PA, USA; London, UK, 1912; p. 217. [Google Scholar]
- Cushing, H. The basophil adenomas of the pituitary body and their clinical manifestations (pituitary basophilism). Bull. Johns Hopkins Hosp. 1932, 50, 137–195. [Google Scholar] [CrossRef]
- Cushing, H. Psychiatric disturbances associated with disorders of the ductless glands. Am. J. Insanity 1913, 69, 965–990. [Google Scholar]
- Aldrich, T.B. A preliminary report on the active principle of the suprarenal gland. Am. J. Physiol. 1901, 5, 457–461. [Google Scholar]
- Takamine, J. Adrenalin; the active principle of the suprarenal gland. Am. J. Pharm. 1901, 73, 523–531. [Google Scholar]
- Bayliss, W.M.; Starling, E.H. The mechanism of pancreatic secretion. J. Physiol. 1902, 28, 325–353. [Google Scholar] [CrossRef] [PubMed]
- Kendall, E.C.; Osterberg, A.E. The chemical identification of thyroxin. J. Biol. Chem. 1919, 40, 265–334. [Google Scholar]
- Banting, F.G. Early work on Insulin. Science 1937, 85, 594–596. [Google Scholar] [CrossRef] [PubMed]
- Piosik, R. Adolf Butenandt und sein Wirken an der Technischen Hochschule Danzig. Chemkon 2003, 10, 135–138. [Google Scholar] [CrossRef]
- Zhang, Y.; Proenca, R.; Maffei, M.; Barone, M.; Leopold, L.; Friedman, J.M. Positional cloning of the mouse obese gene and its human homologue. Nature 1994, 372, 425–432. [Google Scholar] [CrossRef] [PubMed]
- Kojima, M.; Hosoda, H.; Date, Y.; Nakazato, M.; Matsuo, H.; Kangawa, K. Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature 1999, 402, 656–660. [Google Scholar] [CrossRef] [PubMed]
- Bernhardt, D. Introduction and Overview. In Hormones and Human Behaviour; Donovan, B.T., Ed.; Cambridge University Press: New York, NY, USA; Cambridge, UK, 1985; pp. 1–16. [Google Scholar]
- Bleuler, M. Untersuchungen aus dem Grenzgebiet zwischen Psychopathologie und Endokrinologie. Arch. Psychiatr. Nervenkrankht. 1948, 180, 271–528. [Google Scholar]
- Bleuler, M. Endokrinologische Psychiatrie; Thieme: Stuttgart, Germany, 1954. [Google Scholar]
- Schüssler, P.; Yassouridis, A.; Uhr, M.; Kluge, M.; Bleninger, P.; Holsboer, F.; Steiger, A. Sleep and active renin levels—Interaction with age, gender, growth hormone and cortisol. Neuropsychobiology 2010, 61, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Bleuler, M. Endokrinologische Psychiatrie. In Psychiatrie der Gegenwart, 2nd ed.; Kisker, K.P., Meyer, J.E., Müller, M., Eds.; Springer: Berlin, Germany, 1979; pp. 257–342. [Google Scholar]
- Himmerich, H.; Veselinović, T.; Habel, U. Affektive Störungen bei Frauen. Die Stimmung im Spiegel der weiblichen Lebensphasen. Inf. Neurol. Psychiatr. 2009, 11, 42–26. [Google Scholar]
- Ripping, L.H. Die Geistesstörungen der Schwangeren, Wöchnerinnen und Säugenden; Enke: Stuttgart, Germany, 1877. [Google Scholar]
- Schmidt, M. Beiträge zur Kenntnis der Puerperalpsychosen. Arch. Psychiatr. Nervenkrankht. 1881, 11, 75–95. [Google Scholar] [CrossRef]
- Boyce, P.; Barriball, E. Puerperal psychosis. Arch. Womens Ment. Health 2010, 13, 45–47. [Google Scholar] [CrossRef] [PubMed]
- Mayer, L. Die Beziehungen der krankhaften Zustände in den Sexual-Organen des Weibes zu Geistesstörungen; Hirschwald: Berlin, Germany, 1869. [Google Scholar]
- Von Reitzenstein, F. Iwan Bloch †. Sexualreform 1922, 11, 113–126. [Google Scholar]
- Sakel, M. Neue Behandlungsmethode der Schizophrenie; Perles: Vienna, Austria, 1935. [Google Scholar]
- Walther, T. Die “Insulin-Koma-Behandlung”. Erfindung und Einführung des Ersten Modernen Psychiatrischen Schockverfahrens; Lehmann Antipsychiatrieverlag: Berlin, Germany, 2004. [Google Scholar]
- Dengler, K. Erfahrungen bei Behandlung von 16 männlichen Schizophrenen mit Choriongonadotropin/Depottestosteron. Arch. Psychiatr. Nervenkrankht. Z. Ges. Neurol. Psychiatr. 1961, 202, 481–503. [Google Scholar] [CrossRef]
- Bujanow, W. Hormones in the treatment of psychoses. Br. Med. J. 1972, 4, 298. [Google Scholar] [CrossRef] [PubMed]
- Linde, O.K. Historischer Abriss: Geschichte der Psychopharmaka. In Neuro-Psychopharmaka; Riederer, P., Laux, G., Pöldinger, W., Eds.; Springer: Vienna, Austria, 1992; Volume 1, pp. 41–66. [Google Scholar]
- Benkert, O.; Hippius, H. Kompendium der Psychiatrischen Pharmakotherapie, 7th ed.; Springer: Heidelberg, Germany, 2009. [Google Scholar]
- Yalow, R.S.; Berson, S.A. Assay of plasma insulin in human subjects by immunological methods. Nature 1959, 184, 1648–1649. [Google Scholar] [CrossRef] [PubMed]
- Berghoff, E. Zur Beeinflussung chronischer Krankheiten durch interkurrentes Fieber. Klin. Wochenschr. 1931, 10, 2138–2140. [Google Scholar] [CrossRef]
- Ilberg, J. Über Fiebertherapie bei Nervenkrankheiten im Altertum. Allg. Z. Psychiatr. 1929, 91, 353–354. [Google Scholar]
- Stahl, G.E. Theoria Medica Vera; Orphanotrophei: Halle, Germany, 1707. [Google Scholar]
- Steinberg, H. Kraepelin in Leipzig. Eine Begegnung von Psychiatrie und Psychologie; Psychiatrie-Verlag: Bonn, Germany, 2001. [Google Scholar]
- Kraepelin, E. Ueber den Einfluss acuter Krankheiten auf die Entstehung von Geisteskrankheiten. Arch. Psychiatr. Nervenkrankht. 1881, 11, 137–183. [Google Scholar] [CrossRef]
- Steinberg, H.; Himmerich, H. Kraepelin’s habilitation and his thesis: A pioneer work for modern systematic reviews, psychoimmunological research and categories of psychiatric diseases. World J. Biol. Psychiatry 2013, 14, 248–257. [Google Scholar] [CrossRef] [PubMed]
- Carswell, E.A.; Old, L.J.; Kassel, R.L.; Green, S.; Fiore, N.; Williamson, B. An endotoxin-induced serum factor that causes necrosis of tumors. Proc. Natl. Acad. Sci. USA 1975, 72, 3666–3670. [Google Scholar] [CrossRef] [PubMed]
- Bruce, L.C.; Peebles, A.M.S. Quantitative and qualitative leukocyte counts in the various forms of mental disease. J. Ment. Sci. 1904, 50, 409–412. [Google Scholar]
- Bruce, L.C.; Peebles, A.M.S. Clinical and experimental observations on catatonia. J. Ment. Sci. 1903, 49, 614–628. [Google Scholar]
- Bruce, L.C. On the experimental use of antiserums in acute insanity. J. Ment. Sci. 1904, 50, 259–262. [Google Scholar] [CrossRef]
- Dameshek, W. White blood cells in dementia praecox and dementia paralytica. Arch. Neurol. Psychiatry 1930, 24, 855. [Google Scholar]
- Whitrow, M. Wagner-Jauregg and fever therapy. Med. Hist. 1990, 34, 294–310. [Google Scholar] [CrossRef] [PubMed]
- Whitrow, M. Julius Wagner-Jauregg (1857–1940); Facultas: Vienna, Austria, 2001. [Google Scholar]
- Brown, E.M. Why Wagner-Jauregg won the Nobel-Prize for discovering malaria therapy for general paresis of the insane. Hist. Psychiatry 2000, 11, 371–382. [Google Scholar] [CrossRef]
- Wagner-Jauregg, J. Über die Einwirkung der Malaria auf die progressive Paralyse. Psychiatr. Neurol. Wochenschr. 1918, 20, 132–134. [Google Scholar]
- Himmerich, H.; Kirkby, K.C.; Steinberg, H. From Julius Wagner-Jauregg to biomarkers and immunological therapies. World J. Biol. Psychiatry 2010, 11, 154–155. [Google Scholar] [CrossRef] [PubMed]
- Wagner-Jauregg, J. Fieberbehandlung bei Psychosen. Wien. Klin. Wochenschr. 1926, 76, 79–82. [Google Scholar]
- Wagner-Jauregg, J. Infektions- und Fiebertherapie; Weidmann: Vienna, Austria, 1936. [Google Scholar]
- Wagner-Jauregg, J. Derzeitige Behandlung der progressiven Paralyse. Wien. Klin. Wochenschr. 1939, 52, 1075–1078. [Google Scholar]
- Noll, R. Kraepelin’s lost biological psychiatry? Autointoxication, organotherapy and surgery for dementia praecox. Hist. Psychiatry 2007, 18, 301–320. [Google Scholar] [CrossRef] [PubMed]
- Noll, R. Historical review: Autointoxication and focal infection theories of dementia praecox. World J. Biol. Psychiatry 2004, 5, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Lehmann-Facius, H. Über die Liquordiagnose der Schizophrenien. Klin. Wochenschr. 1937, 16, 1646–1648. [Google Scholar] [CrossRef]
- Lehmann-Facius, H. Serologisch-analytische Versuche mit Liquoren und Seren von Schizophrenen. Allg. Z. Psychiatr. 1939, 110, 232–243. [Google Scholar]
- Roeder, F. Über die serologische Diagnostik der Schizophrenie aus dem Liquor nach der Methode von Lehmann-Facius. Z. Ges. Neurol. Psychiatr. 1939, 165, 462–467. [Google Scholar]
- Göhde, W.; Dittrich, W. Impulse fluorometry of single cells in suspension. Z. Naturforsch. B 1969, 24, 360–361. [Google Scholar]
- Köhler, G.; Milstein, C. Continuous cultures of fused cells secreting antibody of predefined specificity. Nature 1975, 256, 495–497. [Google Scholar] [CrossRef] [PubMed]
- Gibbons, J.L.; McHugh, P.R. Plasma cortisol in depressive illness. J. Psychiatr. Res. 1962, 1, 162–171. [Google Scholar] [CrossRef]
- Gibbons, J.L. Cortisol depression rate in depressive illness. Arch. Gen. Psychiatry 1964, 10, 572–575. [Google Scholar] [CrossRef] [PubMed]
- Holsboer, F.; von Bardeleben, U.; Wiedemann, K.; Müller, O.A.; Stalla, G.K. Serial assessment of corticotropin-releasing hormone response after dexamethasone in depression. Implications for pathophysiology of DST nonsuppression. Biol. Psychiatry 1987, 22, 228–234. [Google Scholar] [CrossRef]
- Ising, M.; Künzel, H.E.; Binder, E.B.; Nickel, T.; Modell, S.; Holsboer, F. The combined dexamethasone/CRH test as a potential surrogate marker in depression. Prog. Neuropsychopharmacol. Biol. Psychiatry 2005, 29, 1085–1093. [Google Scholar] [CrossRef] [PubMed]
- Ströhle, A. Die Neuroendokrinologie von Stress und die Pathophysiologie und Therapie von Depression und Angst. Nervenarzt 2003, 74, 279–291. [Google Scholar] [CrossRef] [PubMed]
- Gilpin, N.W.; Richardson, H.N.; Koob, G.F. Effects of CRF1-receptor and opioid-receptor antagonists on dependence-induced increases in alcohol drinking by alcohol-preferring (P) rats. Alcohol. Clin. Exp. Res. 2008, 32, 1535–1542. [Google Scholar] [CrossRef] [PubMed]
- Jahn, H.; Döring, W.K.; Krampe, H.; Sieg, S.; Werner, C.; Poser, W.; Brunner, E.; Ehrenreich, H. Preserved vasopressin response to osmostimulation despite decreased basal vasopressin levels in longterm abstinent alcoholics. Alcohol. Clin. Exp. Res. 2004, 28, 1925–1930. [Google Scholar] [CrossRef] [PubMed]
- Hundt, W.; Zimmermann, U.; Pöttig, M.; Spring, K.; Holsboer, F. The combined dexamethasone-suppression/CRH-stimulation test in alcoholics during and after acute withdrawal. Alcohol. Clin. Exp. Res. 2001, 25, 687–691. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, U.; Spring, K.; Kunz-Ebrecht, S.R.; Uhr, M.; Wittchen, H.U.; Holsboer, F. Effect of ethanol on hypothalamic-pituitary-adrenal system response to psychosocial stress in sons of alcohol-dependent fathers. Neuropsychopharmacology 2004, 29, 1156–1165. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, U.; Spring, K.; Wittchen, H.U.; Himmerich, H.; Landgraf, R.; Uhr, M.; Holsboer, F. Arginine vasopressin and adrenocorticotropin secretion in response to psychosocial stress is attenuated by ethanol in sons of alcohol-dependent fathers. J. Psychiatr. Res. 2004, 38, 385–393. [Google Scholar] [CrossRef] [PubMed]
- Andrade, T.G.; Nakamuta, J.S.; Avanzi, V.; Graeff, F.G. Anxiolytic effect of estradiol in the median raphe nucleus mediated by 5-HT1A receptors. Behav. Brain Res. 2005, 163, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Estrada-Camarena, E.; Fernandez-Guasti, A.; Lopez-Rubalcava, C. Antidepressant-like effect of different estrogenic compounds in the forced swimming test. Neuropsychopharmacology 2003, 28, 830–838. [Google Scholar] [CrossRef] [PubMed]
- Zarrouf, F.A.; Artz, S.; Griffith, J.; Sirbu, C.; Kommor, M. Testosterone and depression: Systematic review and meta-analysis. J. Psychiatr. Pract. 2009, 15, 289–305. [Google Scholar] [CrossRef] [PubMed]
- Martel, M.M.; Klump, K.; Nigg, J.T.; Breedlove, S.M.; Sisk, C.L. Potential hormonal mechanisms of attention-deficit/hyperactivity disorder and major depressive disorder: A new perspective. Horm. Behav. 2009, 55, 465–479. [Google Scholar] [CrossRef] [PubMed]
- Kanayama, G.; Hudson, J.I.; Pope, H.G., Jr. Long-term psychiatric and medical consequences of anabolic-androgenic steroid abuse: A looming public health concern? Drug Alcohol Depend. 2008, 98, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Malone, D.A., Jr.; Dimeff, R.J.; Lombardo, J.A.; Sample, R.H. Psychiatric effects and psychoactive substance use in anabolic-androgenic steroid users. Clin. J. Sport Med. 1995, 5, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Maayan, R.; Shaltiel, G.; Poyurovsky, M.; Ramadan, E.; Morad, O.; Nechmad, A.; Weizman, A.; Agam, G. Chronic lithium treatment affects rat brain and serum dehydroepiandrosterone (DHEA) and DHEAsulphate (DHEA-S) levels. Int. J. Neuropsychopharmacol. 2004, 7, 71–75. [Google Scholar] [CrossRef] [PubMed]
- Ambar, G.; Chiavegatto, S. Anabolic-androgenic steroid treatment induces behavioral disinhibition and downregulation of serotonin receptor messenger RNA in the prefrontal cortex and amygdala of male mice. Genes Brain Behav. 2009, 8, 161–173. [Google Scholar] [CrossRef] [PubMed]
- Pichlo, H.G. Transsexualismus–leistungsrechtliche und gutachterliche Kriterien für geschlechtsangleichende somatische Maßnahmen aus Sicht des MDK-Nordrhein. In Transsexualität und Intersexualität; Groß, D., Neuschäfer-Rube, C., Steinbetzer, J., Eds.; Medizinisch Wissenschaftliche Verlagsgesellschaft: Berlin, Germany, 2008; pp. 119–130. [Google Scholar]
- Adli, M.; Bauer, M.; Rush, A.J. Algorithms and collaborative-care systems for depression: Are they effective and why? A Systematic Review. Biol. Psychiatry 2006, 59, 1029–1038. [Google Scholar] [CrossRef] [PubMed]
- Prange, A.J. Novel uses of thyroid hormones in patients with affective disorders. Thyroid 1996, 6, 537–543. [Google Scholar] [CrossRef] [PubMed]
- Aronson, R.; Offman, H.J.; Joffe, R.T.; Naylor, C.D. Triiodothyronine augmentation in the treatment of refractory depression. Arch. Gen. Psychiatry 1996, 53, 842–848. [Google Scholar] [CrossRef] [PubMed]
- Altshuler, L.L.; Bauer, M.; Frye, M.A.; Gitlin, M.J.; Mintz, J.; Szuba, M.P.; Leight, K.L.; Whybrow, P.C. Does thyroid supplementation accelerate tricyclic antidepressant response? A review and meta-analysis of the literature. Am. J. Psychiatry 2001, 158, 1617–1622. [Google Scholar] [CrossRef] [PubMed]
- Morton, G.J.; Cummings, D.E.; Baskin, D.G.; Barsh, G.S.; Schwartz, M.W. Central nervous system control of food intake and body weight. Nature 2006, 443, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Baumann, A.; Heitmann, S.; Bubendorff, V.; Himmerich, H. Laborwertveränderungen bei Anorexia nervosa. Praxis 2010, 99, 661–667. [Google Scholar] [CrossRef] [PubMed]
- Himmerich, H.; Schönknecht, P.; Heitmann, S.; Sheldrick, A.J. Laboratory parameters and appetite regulators in patients with anorexia nervosa. J. Psychiatr. Pract. 2010, 16, 82–92. [Google Scholar] [CrossRef] [PubMed]
- Nicolás, J.M.; Fernández-Solà, J.; Fatjó, F.; Casamitjana, R.; Bataller, R.; Sacanella, E.; Tobías, E.; Badía, E.; Estruch, R. Increased circulating leptin levels in chronic alcoholism. Alcohol. Clin. Exp. Res. 2001, 25, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Motivala, S.J.; Dang, J.; Obradovic, T.; Meadows, G.G.; Butch, A.W.; Irwin, M.R. Leptin and cellular and innate immunity in abstinent alcoholics and controls. Alcohol. Clin. Exp. Res. 2003, 27, 1819–1824. [Google Scholar] [CrossRef] [PubMed]
- Fulda, S.; Linseisen, J.; Wolfram, G.; Himmerich, S.; Gedrich, K.; Pollmächer, T.; Himmerich, H. Leptin plasma levels in the general population: Influence of age, gender, bodyweight and medical history. Protein Pept. Lett. 2010, 17, 1436–1440. [Google Scholar] [CrossRef] [PubMed]
- Kraus, T.; Reulbach, U.; Bayerlein, K.; Mugele, B.; Hillemacher, T.; Sperling, W.; Kornhuber, J.; Bleich, S. Leptin is associated with craving in females with alcoholism. Addict. Biol. 2004, 9, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Wurst, F.M.; Junghanns, K.; Thon, N. Alkoholabhängigkeit, craving und appetitregulierende hormone. Psychiatrie Psychother. 2009, 5, 106–109. [Google Scholar] [CrossRef]
- Kraus, T.; Schanze, A.; Gröschl, M.; Salomé, N.; Heilig, M.; Moechars, D.; Datta, R.; Perrissoud, D.; Dickson, S.L.; Engel, J.A. Ghrelin levels are increased in alcoholism. Alcohol. Clin. Exp. Res. 2005, 29, 2154–2157. [Google Scholar] [CrossRef] [PubMed]
- Jerlhag, E.; Egecioglu, E.; Landgren, S.; Salomé, N.; Heilig, M.; Moechars, D.; Datta, R.; Perrissoud, D.; Dickson, S.L.; Engel, J.A. Requirement of central ghrelin signaling for alcohol reward. Proc. Natl. Acad. Sci. USA 2009, 106, 11318–11323. [Google Scholar] [CrossRef] [PubMed]
- Himmerich, H. Neuroimmunologie. In Handbuch der Psychopharmakologie; Holsboer, F., Gründer, G., Benkert, O., Eds.; Springer: Heidelberg, Germany, 2008; pp. 369–374. [Google Scholar]
- Himmerich, H.; Sorge, S.; Kirkby, K.C.; Steinberg, H. Schizophrenic disorders. The development of immunological concepts and therapy in psychiatry. Nervenarzt 2012, 83, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Müller, N.; Schwarz, M.J. A psychoneuroimmunological perspective to Emil Kraepelin’s dichotomy: Schizophrenia and major depression as inflammatory CNS disorders. Eur. Arch. Psychiatry Clin. Neurosci. 2008, 258, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Müller, N.; Schwarz, M.J. Immunologische Aspekte bei schizophrenen Störungen. Nervenarzt 2007, 78, 253–263. [Google Scholar] [CrossRef] [PubMed]
- Stöber, G.; Ben-Shachar, D.; Cardon, M.; Falkai, P.; Fonteh, A.N.; Gawlik, M.; Glenthoj, B.Y.; Grunblatt, E.; Jablensky, A.; Kim, Y.K.; et al. Schizophrenia: From the brain to peripheral markers. A consensus paper of the WFSBP task force on biological markers. World J. Biol. Psychiatry 2009, 10, 127–155. [Google Scholar] [CrossRef] [PubMed]
- Müller, N.; Riedel, M.; Scheppach, C.; Brandstätter, B.; Sokullu, S.; Krampe, K.; Ulmschneider, M.; Engel, R.R.; Möller, H.J.; Schwarz, M.J. Beneficial antipsychotic effects of celecoxib add-on therapy compared to risperidone alone in schizophrenia. Am. J. Psychiatry 2002, 159, 1029–1034. [Google Scholar] [CrossRef] [PubMed]
- Wagner-Jauregg, J. Über die Einwirkung fieberhafter Erkrankungen auf Psychosen. Jb. Psychiatr. 1887, 7, 94–131. [Google Scholar]
- Hänel, F.; Legatis, G. Therapeutic use of megaphen in pulmonary tuberculosis. Tuberkulosearzt 1954, 8, 636–638. [Google Scholar] [PubMed]
- Pollmächer, T.; Hinze-Selch, D.; Mullington, J. Effects of clozapine on plasma cytokine and soluble cytokine receptor levels. J. Clin. Psychopharmacol. 1996, 16, 403–409. [Google Scholar] [CrossRef] [PubMed]
- Dalmau, J.; Gleichman, A.J.; Hughes, E.G.; Rossi, J.E.; Peng, X.; Lai, M.; Dessain, S.K.; Rosenfeld, M.R.; Balice-Gordon, R.; Lynch, D.R. Anti-NMDA-receptor encephalitis: Case series and analysis of the effects of antibodies. Lancet Neurol. 2008, 12, 1091–1098. [Google Scholar] [CrossRef]
- Margari, F.; Petruzzelli, M.G.; Mianulli, R.; Campa, M.G.; Pastore, A.; Tampoia, M. Circulating anti-brain autoantibodies in schizophrenia and mood disorders. Psychiatry Res. 2015. [Google Scholar] [CrossRef] [PubMed]
- Steiner, J.; Schiltz, K.; Bernstein, H.G.; Bogerts, B. Antineuronal antibodies against neurotransmitter receptors and synaptic proteins in schizophrenia: Current knowledge and clinical implications. CNS Drugs 2015, 29, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Steiner, J.; Walter, M.; Glanz, W.; Sarnyai, Z.; Bernstein, H.G.; Vielhaber, S.; Kästner, A.; Skalej, M.; Jordan, W.; Schiltz, K.; et al. Increased prevalence of diverse N-methyl-d-aspartate glutamate receptor antibodies in patients with an initial diagnosis of schizophrenia: Specific relevance of IgG NR1a antibodies for distinction from N-methyl-d-aspartate glutamate receptor encephalitis. JAMA Psychiatry 2013, 70, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.S.; Begg, M.D.; Gravenstein, S.; Schaefer, C.A.; Wyatt, R.J.; Bresnahan, M.; Babulas, V.P.; Susser, E.S. Serologic evidence of prenatal influenza in the etiology of schizophrenia. Arch. Gen. Psychiatry 2004, 61, 774–780. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.E.; Li, J.; Garbett, K.; Mirnics, K.; Patterson, P.H. Maternal immune activation alters fetal brain development through interleukin-6. J. Neurosci. 2007, 27, 10695–10702. [Google Scholar] [CrossRef] [PubMed]
- Lipina, T.V.; Zai, C.; Hlousek, D.; Roder, J.C.; Wong, A.H. Maternal immune activation during gestation interacts with Disc1 point mutation to exacerbate schizophrenia-related behaviors in mice. J. Neurosci. 2013, 33, 7654–7666. [Google Scholar] [CrossRef] [PubMed]
- Hsiao, E.Y.; McBride, S.W.; Hsien, S.; Sharon, G.; Hyde, E.R.; McCue, T.; Codelli, J.A.; Chow, J.; Reisman, S.E.; Petrosino, J.F.; et al. Microbiota modulate behavioral and physiological abnormalities associated with neurodevelopmental disorders. Cell 2013, 155, 1451–1463. [Google Scholar] [CrossRef] [PubMed]
- Bode, L.; Zimmermann, W.; Ferszt, R.; Steinbach, F.; Ludwig, H. Borna disease virus genome transcribed and expressed in psychiatric patients. Nat. Med. 1995, 1, 232–236. [Google Scholar] [CrossRef] [PubMed]
- House, P.K.; Vyas, A.; Sapolsky, R. Predator cat odors activate sexual arousal pathways in brains of Toxoplasma gondii infected rats. PLoS ONE 2011, 6, e23277. [Google Scholar] [CrossRef] [PubMed]
- Monroe, J.M.; Buckley, P.F.; Miller, B.J. Meta-analysis of anti-Toxoplasma gondii IgM antibodies in acute psychosis. Schizophr. Bull. 2015, 41, 989–998. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.K.; Myint, A.M.; Verkerk, R.; Scharpe, S.; Steinbusch, H.; Leonard, B. Cytokine changes and tryptophan metabolites in medication-naive and medication-free schizophrenic patients. Neuropsychobiology 2009, 59, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Dantzer, R.; O‘Connor, J.C.; Freund, G.G.; Johnson, R.W.; Kelley, K.W. From inflammation to sickness and depression: When the immune system subjugates the brain. Nat. Rev. Neurosci. 2008, 9, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Reichenberg, A.; Yirmiya, R.; Schuld, A.; Kraus, T.; Haack, M.; Morag, A.; Pollmächer, T. Cytokine-associated emotional and cognitive disturbances in humans. Arch. Gen. Psychiatry 2001, 58, 445–452. [Google Scholar] [CrossRef] [PubMed]
- Himmerich, H.; Fulda, S.; Linseisen, J.; Seiler, H.; Wolfram, G.; Himmerich, S.; Gedrich, K.; Kloiber, S.; Lucae, S.; Ising, M.; et al. Depression, comorbidities and the TNF-α system. Eur. Psychiatry 2008, 23, 421–429. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, F.M.; Lichtblau, N.; Minkwitz, J.; Chittka, T.; Thormann, J.; Kirkby, K.C.; Sander, C.; Mergl, R.; Faßhauer, M.; Stumvoll, M.; et al. Cytokine levels in depressed and non-depressed subjects, and masking effects of obesity. J. Psychiatr. Res. 2014, 55, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Lichtblau, N.; Schmidt, F.M.; Schumann, R.; Kirkby, K.C.; Himmerich, H. Cytokines as biomarkers in depressive disorder: Current standing and prospects. Int. Rev. Psychiatry 2013, 25, 592–603. [Google Scholar] [CrossRef] [PubMed]
- Tyring, S.; Gottlieb, A.; Papp, K.; Gordon, K.; Leonardi, C.; Wang, A.; Lalla, D.; Woolley, M.; Jahreis, A.; Zitnik, R.; et al. Etanercept and clinical outcomes, fatigue, and depression in psoriasis: Double-blind placebo-controlled randomised phase III trial. Lancet 2006, 367, 29–35. [Google Scholar] [CrossRef]
- Krügel, U.; Fischer, J.; Radicke, S.; Sack, U.; Himmerich, H. Antidepressant effects of TNF-α blockade in an animal model of depression. J. Psychiatr. Res. 2013, 47, 611–616. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, F.M.; Kirkby, K.C.; Himmerich, H. The TNF-α inhibitor etanercept as monotherapy in treatment-resistant depression—Report of two cases. Psychiatr. Danub. 2014, 26, 288–290. [Google Scholar] [PubMed]
- Müller, N.; Schwarz, M.J.; Dehning, S.; Douhe, A.; Cerovecki, A.; Goldstein-Müller, B.; Spellmann, I.; Hetzel, G.; Maino, K.; Kleindienst, N.; et al. The cyclooxygenase-2 inhibitor celecoxib has therapeutic effects in major depression: Results of a double-blind, randomized, placebo controlled, add-on pilot study to reboxetine. Mol. Psychiatry 2006, 11, 680–684. [Google Scholar] [CrossRef] [PubMed]
- Kenis, G.; Maes, M. Effects of antidepressants on the production of cytokines. Int. J. Neuropsychopharmacol. 2002, 5, 401–412. [Google Scholar] [CrossRef] [PubMed]
- Himmerich, H.; Fulda, S.; Sheldrick, A.J.; Plümäkers, B.; Rink, L. IFN-γ reduction by tricyclic antidepressants. Int. J. Psychiatry Med. 2010, 40, 413–424. [Google Scholar] [CrossRef] [PubMed]
- Petersein, C.; Sack, U.; Mergl, R.; Schönherr, J.; Schmidt, F.M.; Lichtblau, N.; Kirkby, K.C.; Bauer, K.; Himmerich, H. Impact of lithium alone and in combination with antidepressants on cytokine production in vitro. J. Neural Transm. 2015, 122, 109–122. [Google Scholar] [CrossRef] [PubMed]
- Munzer, A.; Sack, U.; Mergl, R.; Schönherr, J.; Petersein, C.; Bartsch, S.; Kirkby, K.C.; Bauer, K.; Himmerich, H. Impact of antidepressants on cytokine production of depressed patients in vitro. Toxins 2013, 5, 2227–2240. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Leung, B.P. CD4+CD25+ regulatory T cells in health and disease. Clin. Exp. Pharmacol. Physiol. 2006, 33, 519–524. [Google Scholar] [CrossRef] [PubMed]
- Himmerich, H.; Milenović, S.; Fulda, S.; Plümäkers, B.; Sheldrick, A.J.; Michel, T.M.; Kircher, T.; Rink, L. Regulatory T cells increased while IL-1β decreased during antidepressant therapy. J. Psychiatr. Res. 2010, 44, 1052–1057. [Google Scholar] [CrossRef] [PubMed]
- Millan, M.J. Dual- and triple-acting agents for treating core and comorbid symptoms of major depression: Novel concepts, new drugs. Neurotherapeutics 2009, 6, 53–77. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, P.J.; Rubinow, D.R. Sex hormones and mood in the perimenopause. Ann. N. Y. Acad. Sci. 2009, 1179, 70–85. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Steinberg, H.; Kirkby, K.C.; Himmerich, H. The Historical Development of Immunoendocrine Concepts of Psychiatric Disorders and Their Therapy. Int. J. Mol. Sci. 2015, 16, 28841-28869. https://doi.org/10.3390/ijms161226136
Steinberg H, Kirkby KC, Himmerich H. The Historical Development of Immunoendocrine Concepts of Psychiatric Disorders and Their Therapy. International Journal of Molecular Sciences. 2015; 16(12):28841-28869. https://doi.org/10.3390/ijms161226136
Chicago/Turabian StyleSteinberg, Holger, Kenneth C. Kirkby, and Hubertus Himmerich. 2015. "The Historical Development of Immunoendocrine Concepts of Psychiatric Disorders and Their Therapy" International Journal of Molecular Sciences 16, no. 12: 28841-28869. https://doi.org/10.3390/ijms161226136
APA StyleSteinberg, H., Kirkby, K. C., & Himmerich, H. (2015). The Historical Development of Immunoendocrine Concepts of Psychiatric Disorders and Their Therapy. International Journal of Molecular Sciences, 16(12), 28841-28869. https://doi.org/10.3390/ijms161226136





