Notch Cooperates with Survivin to Maintain Stemness and to Stimulate Proliferation in Human Keratinocytes during Ageing
Abstract
:1. Introduction
2. Results and Discussion
2.1. Notch1 Decreases during Ageing and Differentiation in Human Keratinocytes
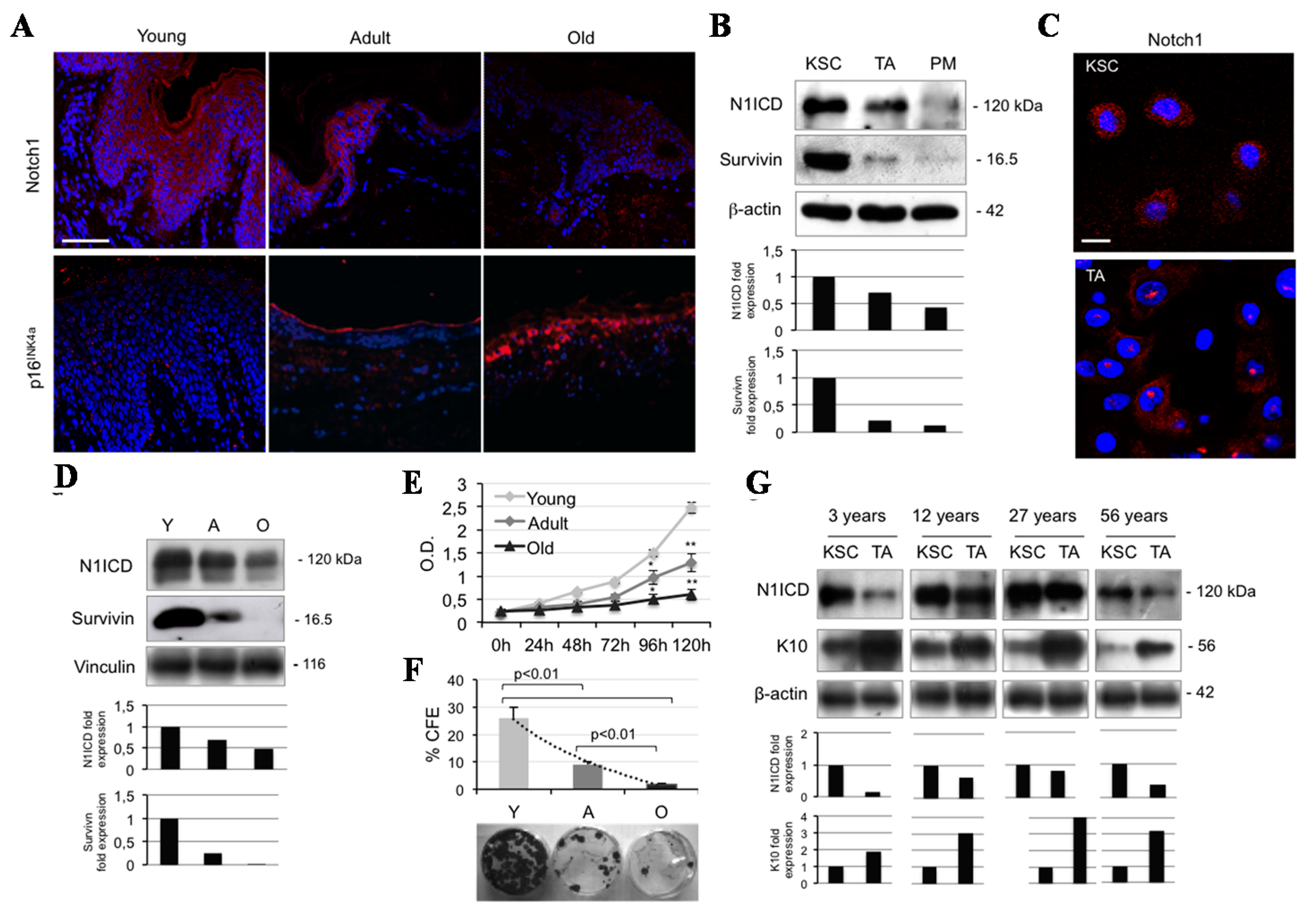
2.2. Calcium Reduces N1ICD in Human Keratinocytes during Ageing
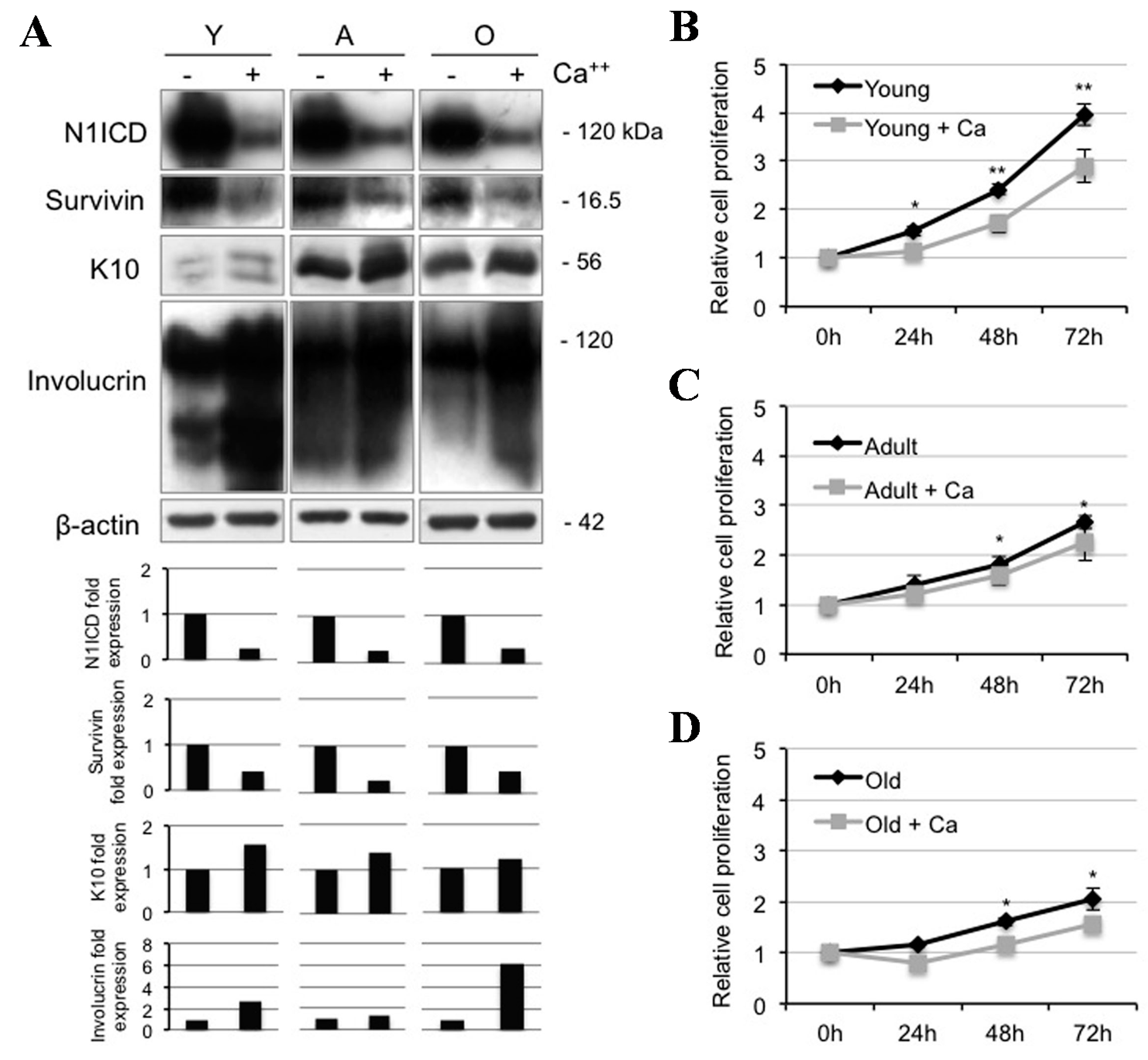
2.3. Notch1 Inhibition Favors Differentiation and Reduces Proliferation in Young Human Keratinocytes

2.4. Notch1 Inhibition Reduces Keratinocyte Proliferation in Adult and Old Keratinocytes
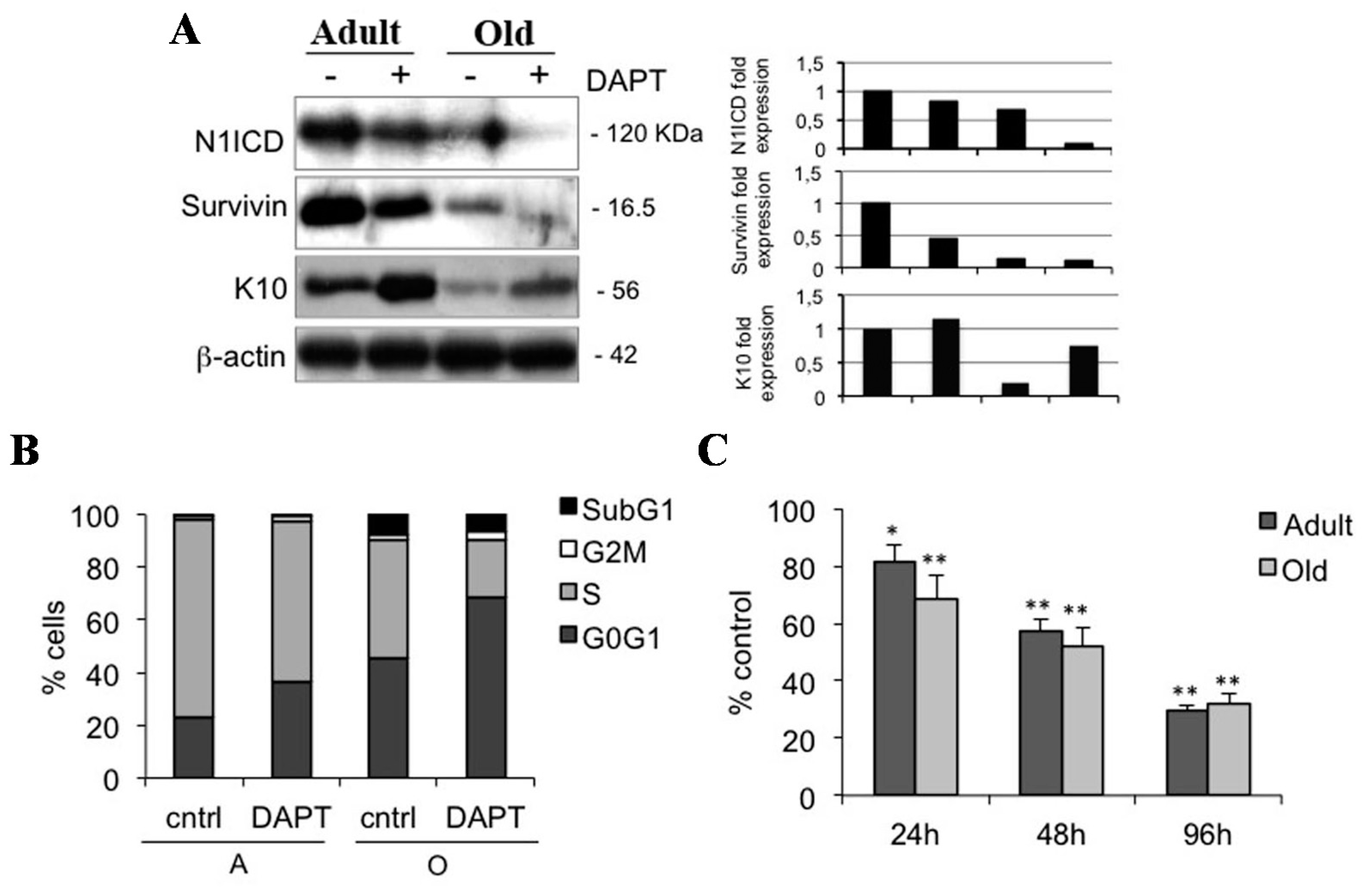
2.5. Notch1 Inhibition Down-Regulates Survivin and Inhibits Proliferation in Keratinocyte Subpopulations
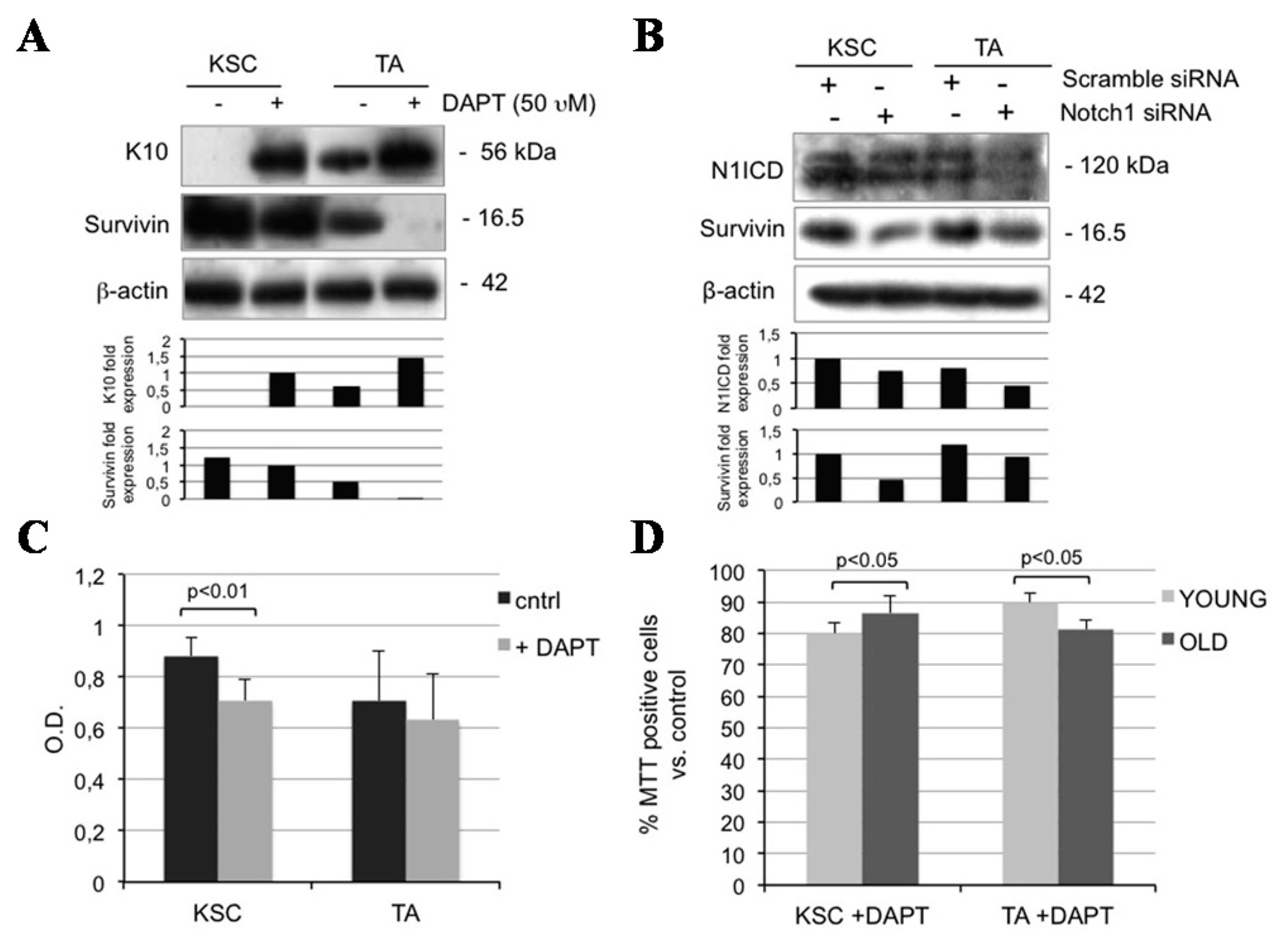
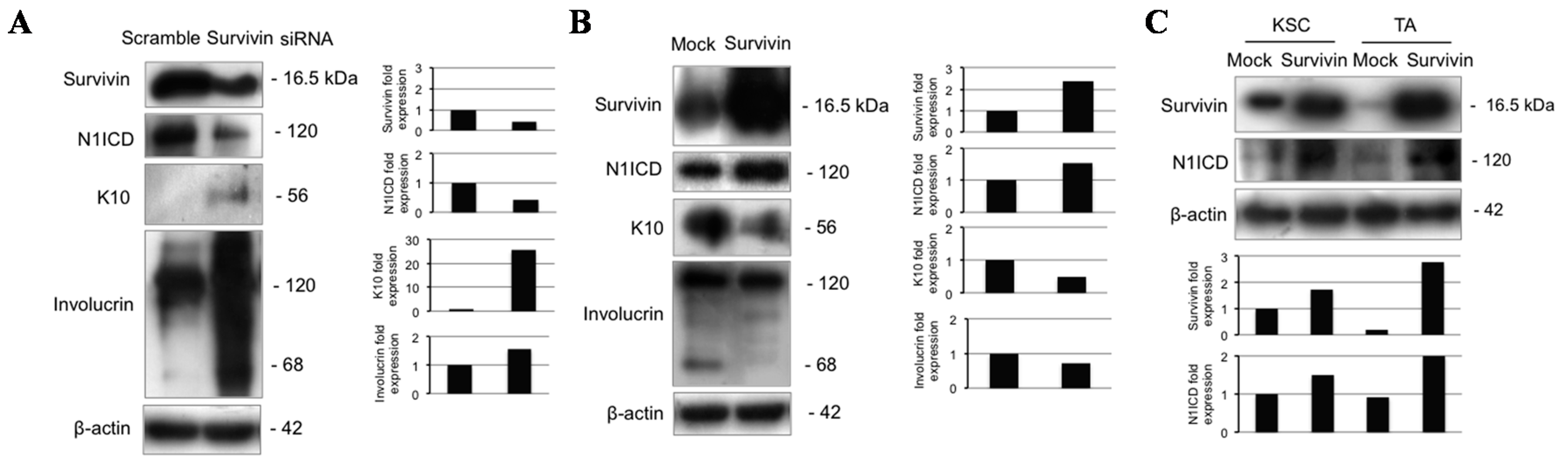
2.6. Survivin Up-Regulates Notch1 and Reduces Keratinocyte Differentiation
3. Experimental Section
3.1. Isolation of Primary Keratinocytes
3.2. MTT Assay
3.3. siRNA Transfection of Keratinocytes
3.4. Infection of Keratinocytes
3.5. Colony Forming Efficiency (CFE)
3.6. Flow Cytometry Analysis (FACS)
3.7. Western Blotting (WB)
3.8. Immunofluorescence (IF) in Situ
3.9. Immunofluorescence of Skin Biopsies
3.10. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Livshits, G.; Kobielak, A.; Fuchs, E. Governing epidermal homeostasis by coupling cell-cell adhesion to integrin and growth factor signaling, proliferation, and apoptosis. Proc. Natl. Acad. Sci. USA 2012, 109, 4886–4891. [Google Scholar] [CrossRef] [PubMed]
- Pincelli, C.; Marconi, A. Keratinocyte stem cells: Friends and foes. J. Cell Physiol. 2010, 225, 310–315. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dallaglio, K.; Marconi, A.; Truzzi, F.; Lotti, R.; Palazzo, E.; Petrachi, T.; Saltari, A.; Coppini, M.; Pincelli, C. E-FABP induces differentiation in normal human keratinocytes and modulates the differentiation process in psoriatic keratinocytes in vitro. Exp. Dermatol. 2013, 22, 255–261. [Google Scholar] [CrossRef] [PubMed]
- Radoja, N.; Gazel, A.; Banno, T.; Yano, S.; Blumenberg, M. Transcriptional profiling of epidermal differentiation. Physiol. Genom. 2006, 27, 65–78. [Google Scholar] [CrossRef] [PubMed]
- Truzzi, F.; Saltari, A.; Palazzo, E.; Lotti, R.; Petrachi, T.; Dallaglio, K.; Gemelli, C.; Grisendi, G.; Dominici, M.; Pincelli, C.; et al. CD271 mediates stem cells to early progeny transition in human epidermis. J. Investig. Dermatol. 2015, 135, 786–795. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Artavanis-Tsakonas, S.; Rand, M.D.; Lake, R.J. Notch signaling: Cell fate control and signal integration in development. Science 1999, 284, 770–776. [Google Scholar] [CrossRef] [PubMed]
- Mumm, J.S.; Kopan, R. Notch signaling: From the outside in. Dev. Biol. 2000, 228, 151–165. [Google Scholar] [CrossRef] [PubMed]
- Lai, E.C. Keeping a good pathway down: Transcriptional repression of Notch pathway target genes by CSL proteins. EMBO Rep. 2002, 3, 840–845. [Google Scholar] [CrossRef] [PubMed]
- Williams, S.E.; Beronja, S.; Pasolli, H.A.; Fuchs, E. Asymmetric cell divisions promote Notch-dependent epidermal differentiation. Nature 2011, 470, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Blanpain, C.; Lowry, W.E.; Pasolli, H.A.; Fuchs, E. Canonical notch signaling functions as a commitment switch in the epidermal lineage. Genes Dev. 2006, 20, 3022–3035. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.W.; Raskett, C.M.; Prudovsky, I.; Altieri, D.C. Molecular dependence of estrogen receptor-negative breast cancer on a notch-survivin signaling axis. Cancer Res. 2008, 68, 5273–5281. [Google Scholar] [CrossRef] [PubMed]
- Marconi, A.; Dallaglio, K.; Lotti, R.; Vaschieri, C.; Truzzi, F.; Fantini, F.; Pincelli, C. Survivin identifies keratinocyte stem cells and is downregulated by anti-β1 integrin during anoikis. Stem Cells 2007, 25, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Dallaglio, K.; Palazzo, E.; Marconi, A.; Dumas, M.; Truzzi, F.; Lotti, R.; Bontè, F.; Pincelli, C. Endogenous survivin modulates survival and proliferation in UVB-treated human keratinocytes. Exp. Dermatol. 2009, 18, 464–471. [Google Scholar] [CrossRef] [PubMed]
- Zobiri, O.; Deshayes, N.; Rathman-Josserand, M. Evolution of the clonogenic potential of human epidermal stem/progenitor cells with age. Stem Cells Cloning 2012, 5, 1–4. [Google Scholar] [PubMed]
- Burkhalter, M.D.; Rudolph, K.L.; Sperka, T. Genome instability of ageing stem cells-induction and defence mechanisms. Ageing Res. Rev. 2015, 23, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Giangreco, A.; Goldie, S.J.; Failla, V.; Saintigny, G.; Watt, F.M. Human skin aging is associated with reduced expression of the stem cell markers β1 integrin and MCSP. J. Investig. Dermatol. 2010, 130, 604–608. [Google Scholar] [CrossRef] [PubMed]
- Ota, T.; Takekoshi, S.; Takagi, T.; Kitatani, K.; Toriumi, K.; Kojima, T.; Kato, M.; Ikoma, N.; Mabuchi, T.; Ozawa, A. Notch signaling may be involved in the abnormal differentiation of epidermal keratinocytes in psoriasis. Acta Histochem. Cytochem. 2014, 47, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Nickoloff, B.J.; Qin, J.Z.; Chaturvedi, V.; Denning, M.F.; Bonish, B.; Miele, L. Jagged-1 mediated activation of notch signaling induces complete maturation of human keratinocytes through NF-κB and PPARγ. Cell Death Differ. 2002, 9, 842–855. [Google Scholar] [CrossRef] [PubMed]
- Thélu, J.; Rossio, P.; Favier, B. Notch signalling is linked to epidermal cell differentiation level in basal cell carcinoma, psoriasis and wound healing. BMC Dermatol. 2002, 2, 7. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, K.; Fujii, T.; Kawachi, H.; Miki, Y.; Omura, K.; Morita, K.; Kaymori, K.; Katsube, K.; Yamaguchi, A. Reduction of NOTCH1 expression pertains to maturation abnormalities of keratinocytes in squamous neoplasms. Lab Investig. 2012, 92, 688–702. [Google Scholar] [CrossRef] [PubMed]
- Nowell, C.; Radtke, F. Cutaneous Notch signaling in health and disease. Cold Spring Harb. Perspect. Med. 2013, 3, a017772. [Google Scholar] [CrossRef] [PubMed]
- Tiberio, R.; Marconi, A.; Fila, C.; Fumelli, C.; Pignatti, M.; Krajewski, S.; Giannetti, A.; Reed, J.C.; Pincelli, C. Keratinocytes enriched for stem cells are protected from anoikis via an integrin signaling pathway in a Bcl-2 dependent manner. FEBS Lett. 2002, 524, 139–144. [Google Scholar] [CrossRef]
- Tseng, C.Y.; Kao, S.H.; Wan, C.L.; Cho, Y.; Tung, S.Y.; Hsu, H.J. Notch signaling mediates the age-associated decrease in adhesion of germline stem cells to the niche. PLoS Genet. 2014, 10, e1004888. [Google Scholar] [CrossRef] [PubMed]
- Perdigoto, C.N.; Bardin, A.J. Sending the right signal: Notch and stem cells. Biochim. Biophys. Acta 2013, 1830, 2307–2322. [Google Scholar] [CrossRef] [PubMed]
- Dallaglio, K.; Petrachi, T.; Marconi, A.; Truzzi, F.; Lotti, R.; Saltari, A.; Morandi, P.; Puviani, M.; Maiorana, A.; Pincelli, C. Expression of nuclear survivin in normal skin and squamous cell carcinoma: A possible role in tumour invasion. Br. J. Cancer 2014, 110, 199–207. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nakamura, T.; Yoshitomi, Y.; Sakai, K.; Patel, V.; Fukumoto, S.; Yamada, Y. Epiprofin orchestrates epidermal keratinocyte proliferation and differentiation. J. Cell Sci. 2014, 127, 5261–5272. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Shu, B.; Yang, R.; Xu, Y.; Xing, B.; Liu, J.; Chen, L.; Qi, S.; Liu, X.; Wang, P.; et al. Wnt and Notch signaling pathway involved in wound healing by targeting c-Myc and Hes1 separately. Stem Cell Res. Ther. 2015, 6, 120. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.W.; Simin, K.; Liu, Q.; Plescia, J.; Guha, M.; Khan, A.; Hsieh, C.C.; Altieri, D.C. A functional Notch-survivin gene signature in basal breast cancer. Breast Cancer Res. 2008, 10, R97. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Li, D.; Liu, H.; Xu, H.; Zheng, H.; Qian, F.; Li, W.; Zhao, C.; Wang, Z.; Wang, X. Notch-1 signaling facilitates survivin expression in human non-small cell lung cancer cells. Cancer Biol. Ther. 2011, 11, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Baker, A.T.; Zlobin, A.; Osipo, C. Notch-EGFR/HER2 Bidirectional Crosstalk in Breast Cancer. Front. Oncol. 2014, 4, 360. [Google Scholar] [CrossRef] [PubMed]
- Pincelli, C.; Haake, A.R.; Benassi, L.; Grassilli, E.; Magnoni, C.; Ottani, D.; Polakowska, R.; Franceschi, C.; Giannetti, A. Autocrine nerve growth factor protects human keratinocytes from apoptosis through its high affinity receptor (TRK): A role for BCL-2. J. Investig. Dermatol. 1997, 109, 757–764. [Google Scholar] [CrossRef] [PubMed]
- Dallaglio, K.; Marconi, A.; Pincelli, C. Survivin: A dual player in healthy and diseased skin. J. Investig. Dermatol. 2012, 132, 18–27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mandinova, A.; Lefort, K.; Tommasi di Vignano, A.; Stonely, W.; Ostano, P.; Chiorino, G.; Iwaki, H.; Nakanishi, J.; Dotto, G.P. The FoxO3a gene is a key negative target of canonical Notch signalling in the keratinocyte UVB response. EMBO J. 2008, 27, 1243–1254. [Google Scholar] [CrossRef] [PubMed]
- Hsu, Y.C.; Li, L.; Fuchs, E. Emerging interactions between skin stem cells and their niches. Nat. Med. 2014, 20, 847–856. [Google Scholar] [CrossRef] [PubMed]
- Giangreco, A.; Qin, M.; Pintar, J.E.; Watt, F.M. Epidermal stem cells are retained in vivo throughout skin aging. Aging Cell 2008, 7, 250–259. [Google Scholar] [CrossRef] [PubMed]
- Piwko-Czuchra, A.; Koegel, H.; Meyer, H.; Bauer, M.; Werner, S.; Brakebusch, C.; Fässler, R. β1 integrin-mediated adhesion signalling is essential for epidermal progenitor cell expansion. PLoS ONE 2009, 4, e5488. [Google Scholar] [CrossRef] [PubMed]
- South, A.P.; Purdie, K.J.; Watt, S.A.; Haldenby, S.; den Breems, N.Y.; Dimon, M.; Arron, S.T.; Kluk, M.J.; Aster, J.C.; McHugh, A.; et al. NOTCH1 mutations occur early during cutaneous squamous cell carcinogenesis. J. Investig. Dermatol. 2014, 134, 2630–2638. [Google Scholar] [CrossRef] [PubMed]
- Dallaglio, K.; Petrachi, T.; Marconi, A.; Truzzi, F.; Lotti, R.; Saltari, A.; Morandi, P.; Puviani, M.; Maiorana, A.; Roop, D.R.; et al. Isolation and characterization of squamous cell carcinoma-derived stem-like cells: Role in tumor formation. Int. J. Mol. Sci. 2013, 14, 19540–19555. [Google Scholar] [CrossRef] [PubMed] [Green Version]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Palazzo, E.; Morandi, P.; Lotti, R.; Saltari, A.; Truzzi, F.; Schnebert, S.; Dumas, M.; Marconi, A.; Pincelli, C. Notch Cooperates with Survivin to Maintain Stemness and to Stimulate Proliferation in Human Keratinocytes during Ageing. Int. J. Mol. Sci. 2015, 16, 26291-26302. https://doi.org/10.3390/ijms161125948
Palazzo E, Morandi P, Lotti R, Saltari A, Truzzi F, Schnebert S, Dumas M, Marconi A, Pincelli C. Notch Cooperates with Survivin to Maintain Stemness and to Stimulate Proliferation in Human Keratinocytes during Ageing. International Journal of Molecular Sciences. 2015; 16(11):26291-26302. https://doi.org/10.3390/ijms161125948
Chicago/Turabian StylePalazzo, Elisabetta, Paolo Morandi, Roberta Lotti, Annalisa Saltari, Francesca Truzzi, Sylvianne Schnebert, Marc Dumas, Alessandra Marconi, and Carlo Pincelli. 2015. "Notch Cooperates with Survivin to Maintain Stemness and to Stimulate Proliferation in Human Keratinocytes during Ageing" International Journal of Molecular Sciences 16, no. 11: 26291-26302. https://doi.org/10.3390/ijms161125948
APA StylePalazzo, E., Morandi, P., Lotti, R., Saltari, A., Truzzi, F., Schnebert, S., Dumas, M., Marconi, A., & Pincelli, C. (2015). Notch Cooperates with Survivin to Maintain Stemness and to Stimulate Proliferation in Human Keratinocytes during Ageing. International Journal of Molecular Sciences, 16(11), 26291-26302. https://doi.org/10.3390/ijms161125948









