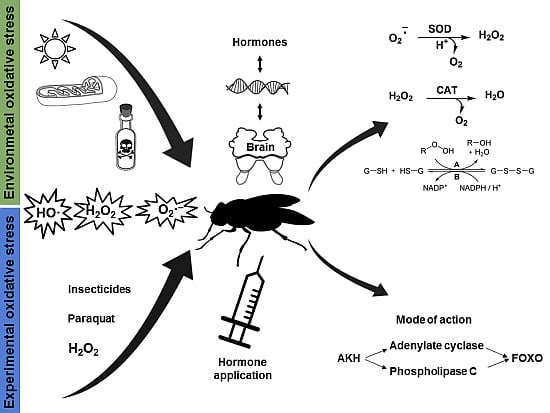
Hormonal Regulation of Response to Oxidative Stress in Insects—An Update
Abstract
:1. Introduction
2. Reactive Oxygen Species and the Phenomenon of Oxidative Stress
2.1. Endogenous Sources of Reactive Oxygen Species (ROS)
2.2. Exogenous Sources of ROS
3. Oxidative Stress and Antioxidant Responses in Insects
4. Insect Hormones: A Brief Overview
5. Adipokinetic Hormones—Main Stress Hormones in Insects
5.1. Role of Adipokinetic Hormones (AKH) in the Insect Anti-Oxidative Stress Response
5.2. Role of AKH in Oxidative Stress Elicited by Insecticides
| Insect Species | Treatment | Mortality (%) | Increase of Mortality (%) | Mortality Ratio |
|---|---|---|---|---|
| P. apterus | P 50 ng inj. | 26.6 ± 8.9 (7) | - | – |
| P 50 ng inj. + AKH 10 pmol inj. | 31.5 ± 13.6 (12) | 4.9 | 1.18 | |
| P 50 ng inj. + AKH 80 pmol inj. | 61.0 ± 8.5 * (6) | 34.4 | 2.29 | |
| P 50 ng inj. + AKH 80 pmol top. | 41.3 ± 12.1 (8) | 14.7 | 1.55 | |
| P 100 ng inj. | 52.0 ± 14.6 (13) | – | – | |
| P 100 ng inj. + AKH 10 pmol inj. | 61.2 ± 2.4 (6) | 9.2 | 1.18 | |
| P 100 ng inj. + AKH 80 pmol inj. | 76.1 ± 4.9 * (8) | 24.1 | 1.46 | |
| P 100 ng inj. + AKH 80 pmol top. | 82.9 ± 14.4 * (7) | 30.9 | 1.59 | |
| P 400 ng top. | 45.4 ± 9.0 (4) | – | – | |
| P 400 ng top. + AKH 80 pmol top. | 73.1 ± 7.2 * (5) | – | 1.61 | |
| E 200 ng inj. | 30.0 ± 4.0 (4) | – | – | |
| E 200 ng inj. + AKH 80 pmol inj. | 91.5 ± 2.5 * (4) | 61.5 | 3.05 | |
| E 250 ng inj. | 57.5 ± 2.9 (4) | – | – | |
| E 250 ng inj.+AKH 80 pmol inj. | 98.7 ± 2.5 * (4) | 41.2 | 1.72 | |
| M 300 ng inj. | 25.0 ± 7.1 (4) | – | – | |
| M 300 ng inj. + AKH 80 pmol inj. | 36.2 ± 8.5 (4) | 11.2 | 1.45 | |
| M 450 ng inj. | 46.2 ± 10.3 (4) | – | – | |
| M 450 ng inj. + AKH 80 pmol inj. | 73.7 ± 7.5 * (4) | 27.5 | 1.59 | |
| E 450 ng top. | 17.5 ± 2.8 (4) | – | – | |
| E 450 ng top. + AKH 80 pmol top. | 31.2 ± 4.7 * (4) | 13.7 | 1.78 | |
| E 1100 ng top. | 51.2 ± 4.7 (4) | – | – | |
| E 1100 ng top. + AKH 80 pmol top. | 88.7 ± 4.7 * (4) | 37.5 | 1.73 | |
| M 500 ng top. | 15.0 ± 4.0 (4) | – | – | |
| M 500 ng top. + AKH 80 pmol top. | 21.2 ± 4.7 (4) | 6.2 | 1.41 | |
| M 900 ng top. | 48.7 ± 4.7 (4) | – | – | |
| M 900 ng top. + AKH 80 pmol top. | 68.7 ± 8.5 * (4) | 20.0 | 1.41 | |
| T. castaneum | PM 0.53 µg/mL res. film | 48.8 ± 2.3 (5) | – | – |
| PM 0.53 µg/mL res. film + AKH 35 pmol/μL dip. | 71.2 ± 1.5 * (5) | 23.3 | 1.46 | |
| PM 0.53 µg/mL res. film-AKH-def. beetles | 26.0 ± 2.5 * (5) | −22.8 | 0.53 | |
| D 13.92 µg/mL res. film | 56.0 ± 2.8 (5) | – | – | |
| D 13.92 µg/mL res. film + AKH 35 pmol/μL dip. | 76.8 ± 2.3 * (5) | 20.8 | 1.37 | |
| D 13.92 µg/mL res. film-AKH-def. beetles | 32.0 ± 2.5 * (5) | −24.0 | 0.57 |
5.3. A Plausible Mechanism of AKH Action in Anti-Oxidative Stress Response

5.4. Drosophila Melanogaster—An Excellent Model for Anti-Oxidative Stress Response Investigation
6. Other Hormones Controlling the Anti-Oxidative Stress Response in Insects
6.1. Glucagon
6.2. Corazonin
6.3. Prothoracicotropic Hormones
6.4. Ecdysteroids
6.5. Juvenile Hormones
7. Conclusions
Abbreviations
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Fridovich, I. Mitochondria: Are they the seat of senescence? Aging Cell 2004, 3, 13–16. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B.; Gutteridge, J.M.C. Free radicals in biology and medicine, 3rd ed.; Oxford University Press: New York, NY, USA, 1999. [Google Scholar]
- Dupuy, C.; Virion, A.; Ohayon, R. Mechanism of hydrogen peroxide formation catalyzed by NADPH oxidase in thyroid plasma membrane. J. Biol. Chem. 1991, 266, 3739–3743. [Google Scholar] [PubMed]
- Granger, D.N. Role of xanthine oxidase and granulocytes in ischemia-reperfusion injury. Am. J. Physiol. Heart Circ. 1988, 255, H1269–H1275. [Google Scholar] [CrossRef]
- Fenton, H.J.H. Oxidation of tartaric acid in presence of iron. J. Chem. Soc. Trans. 1894, 65, 899–910. [Google Scholar] [CrossRef]
- Haber, F.; Weiss, J.J. The catalytic decomposition of hydrogen peroxide by iron salts. Proc. R. Soc. London Ser. A Math. Phys. Sci. 1934, 147, 332–351. [Google Scholar] [CrossRef]
- Liochev, S.I.; Fridovich, I. The Haber-Weiss cycle—70 years later: An alternative view. Redox Rep. 2002, 7, 55–57. [Google Scholar] [CrossRef] [PubMed]
- Biaglow, J.E.; Mitchell, J.B.; Held, K. The importance of peroxide and superoxide in the X-ray response. Int. J. Radiat. Oncol. Biol. Phys. 1992, 22, 665–669. [Google Scholar] [CrossRef]
- Chiu, S.M.; Xue, L.Y.; Friedman, L.R.; Oleinick, N.L. Copper ion-mediated sensitization of nuclear matrix attachment sites to ionizing radiation. Biochemistry 1993, 32, 6214–6219. [Google Scholar] [CrossRef] [PubMed]
- Hiltermann, J.T.; Lapperre, T.S.; van Bree, L.; Steerenberg, P.A.; Brahim, J.J.; Sont, J.K.; Sterk, P.J.; Hiemstra, P.S.; Stolk, J. Ozone-induced inflammation assessed in sputum and bronchial lavage fluid from asthmatics. Free Radic. Biol. Med. 1999, 27, 1448–1454. [Google Scholar] [CrossRef]
- Bagchi, D.; Bagchi, M.; Hassoun, E.A.; Stohs, S.J. In vitro and in vivo generation of reactive oxygen species, DNA damage and lactate dehydrogenase leakage by selected pesticides. Toxicology 1995, 104, 129–140. [Google Scholar] [CrossRef]
- Melchiorri, D.; Ortiz, G.G.; Reiter, R.J.; Sewerynek, E.; Daniels, W.M.; Pablos, M.I.; Nisticò, G. Melatonin reduces paraquat-induced genotoxicity in mice. Toxicol. Lett. 1998, 95, 103–108. [Google Scholar] [CrossRef]
- Ledirac, N.; Antherieu, S.; d’Uby, A.D.; Caron, J.-C.; Rahmani, R. Effects of organochlorine insecticides on MAP kinase pathways in human HaCaT keratinocytes: key role of reactive oxygen species. Toxicol. Sci. 2005, 86, 444–452. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S. Oxidative stress from environmental pollutants. Arch. Insect Biochem. Physiol. 1995, 29, 135–157. [Google Scholar] [CrossRef] [PubMed]
- Bi, J.L.; Felton, G.W. Foliar oxidative stress and insect herbivory: Primary compounds, secondary metabolites, and reactive oxygen species as components of induced resistance. J. Chem. Ecol. 1995, 21, 1511–1530. [Google Scholar] [CrossRef] [PubMed]
- Souza, A.V.G.; Petretski, J.H.; Demasi, M.; Bechara, E.J.H.; Oliveira, P.L. Urate protects a blood-sucking insect against hemin-induced oxidative stress. Free Radic. Biol. Med. 1997, 22, 209–214. [Google Scholar] [CrossRef]
- Lalouette, L.; Williams, C.M.; Hervant, F.; Sinclair, B.J.; Renault, D. Metabolic rate and oxidative stress in insects exposed to low temperature thermal fluctuations. Comp. Biochem. Physiol. 2011, 158, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Meng, J.-Y.; Zhang, C.-Y.; Zhu, F.; Wang, X.-P.; Lei, C.-L. Ultraviolet light-induced oxidative stress: Effects on antioxidant response of Helicoverpa armigera adults. J. Insect Physiol. 2009, 55, 588–592. [Google Scholar] [CrossRef] [PubMed]
- Felton, G.W.; Summers, C.B. Antioxidant systems in insects. Arch. Insect Biochem. Physiol. 1995, 29, 187–197. [Google Scholar] [CrossRef] [PubMed]
- Perić-Mataruga, V.; Nenadović, V.; Ivanović, J. Neurohormones in insect stress: A review. Arch. Biol. Sci. 2006, 58, 1–12. [Google Scholar] [CrossRef]
- Krishnan, N.; Kodrík, D. Endocrine control of oxidative stress in insects. In Oxidative Stress in Vertebrates and Invertebrates: Molecular Aspects of Cell Signaling; Farooqui, T., Farooqui, A.A., Eds.; Wiley-Blackwell: New Jersey, NJ, USA, 2012; pp. 261–270. [Google Scholar]
- Gilbert, L.I.; Yatrou, K.; Gill, S.S. Comprehensive Molecular Insect Science; Elsevier: Oxford, UK, 2005; Volume 3, p. 842. [Google Scholar]
- Nijhout, H.F. Insect Hormones; Princeton University Press: Princeton, NJ, USA, 1998. [Google Scholar]
- Gäde, G.; Hoffmann, K.H.; Spring, J.H. Hormonal regulation in insects: Facts, gaps, and future directions. Physiol. Rev. 1997, 77, 963–1032. [Google Scholar] [PubMed]
- Gäde, G.; Auerswald, L. Mode of action of neuropeptides from the adipokinetic hormone family. Gen. Comp. Endocrinol. 2003, 132, 10–20. [Google Scholar] [CrossRef]
- Kodrík, D. Adipokinetic hormone functions that are not associated with insect flight. Physiol. Entomol. 2008, 33, 171–180. [Google Scholar] [CrossRef]
- Scarborough, R.M.; Jamieson, G.C.; Kalish, F.; Kramer, S.J.; McEnroe, G.A.; Miller, C.A.; Schooley, D.A. Isolation and primary structure of two peptides with cardioacceleratory and hyperglycemic activity from the corpora cardiaca of Periplaneta americana. Proc. Natl. Acad. Sci. USA 1984, 81, 5575–5579. [Google Scholar] [CrossRef] [PubMed]
- Kodrík, D.; Socha, R.; Šimek, P.; Zemek, R.; Goldsworthy, G.J. A new member of the AKH/RPCH family that stimulates locomotory activity in the firebug, Pyrrhocoris apterus (Heteroptera). Insect Biochem. Mol. Biol. 2000, 30, 489–498. [Google Scholar] [CrossRef]
- Goldsworthy, G.; Opoku-Ware, K.; Mullen, L. Adipokinetic hormone enhances laminarin and bacterial lipopolysaccharide-induced activation of the prophenoloxidase cascade in the African migratory locust, Locusta migratoria. J. Insect Physiol. 2002, 48, 601–608. [Google Scholar] [CrossRef]
- Lee, G.; Park, J.H. Hemolymph sugar homeostasis and starvation-induced hyperactivity affected by genetic manipulations of the adipokinetic hormone-encoding gene in Drosophila melanogaster. Genetics 2004, 167, 311–323. [Google Scholar] [CrossRef] [PubMed]
- Kodrík, D.; Krishnan, N.; Habuštová, O. Is the titer of adipokinetic peptides in Leptinotarsa decemlineata fed on genetically modified potatoes increased by oxidative stress? Peptides 2007, 28, 974–980. [Google Scholar] [CrossRef] [PubMed]
- Kodrík, D.; Vinokurov, K.; Tomčala, A.; Socha, R. The effect of adipokinetic hormone on midgut characteristics in Pyrrhocoris apterus L. (Heteroptera). J. Insect Physiol. 2012, 58, 194–204. [Google Scholar] [CrossRef] [PubMed]
- Vinokurov, K.; Bednářová, A.; Tomčala, A.; Stašková, T.; Krishnan, N.; Kodrík, D. Role of adipokinetic hormone in stimulation of salivary gland activities: The fire bug Pyrrhocoris apterus L. (Heteroptera) as a model species. J. Insect Physiol. 2014, 60, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Bil, M.; Broeckx, V.; Landuyt, B.; Huybrechts, R. Differential peptidomics highlights adipokinetic hormone as key player in regulating digestion in anautogenous flesh fly, Sarcophaga crassipalpis. Gen. Comp. Endocrinol. 2014, 208, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Večeřa, J.; Krishnan, N.; Alquicer, G.; Kodrík, D.; Socha, R. Adipokinetic hormone-induced enhancement of antioxidant capacity of Pyrrhocoris apterus hemolymph in response to oxidative stress. Comp. Biochem. Physiol. C 2007, 146, 336–342. [Google Scholar] [CrossRef] [PubMed]
- Bednářová, A.; Krishnan, N.; Cheng, I.-C.; Večeřa, J.; Lee, H.-J.; Kodrík, D. Adipokinetic hormone counteracts oxidative stress elicited in insects by hydrogen peroxide: in vivo and in vitro study. Physiol. Entomol. 2013, 38, 54–62. [Google Scholar] [CrossRef]
- Velki, M.; Kodrík, D.; Večeřa, J.; Hackenberger, B.K.; Socha, R. Oxidative stress elicited by insecticides: A role for the adipokinetic hormone. Gen. Comp. Endocrinol. 2011, 172, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Plavšin, I.; Stašková, T.; Šerý, M.; Smýkal, V.; Hackenberger, B.K.; Kodrík, D. Hormonal enhancement of insecticide efficacy in Tribolium castaneum: Oxidative stress and metabolic aspects. Comp. Biochem. Physiol. C 2015, 170, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Kodrík, D.; Socha, R.; Syrová, Z. Developmental and diel changes of adipokinetic hormone in CNS and haemolymph of the flightless wing-polymorphic bug, Pyrrhocoris apterus (L.). J. Insect Physiol. 2003, 49, 53–61. [Google Scholar] [CrossRef]
- Diederen, J.H.B.; Oudejans, R.C.H.M.; Harthoorn, L.F.; van der Horst, D.J. Cell biology of the adipokinetic hormone-producing neurosecretory cells in the locust corpus cardiacum. Microsc. Res. Tech. 2002, 56, 227–236. [Google Scholar] [CrossRef] [PubMed]
- Scott, M.; Lubin, B.; Zuo, L.; Zuo, L.; Kuypers, F. Erythrocyte defense against hydrogen peroxide: preeminent importance of catalase. J. Lab. Clin. Med. 1991, 118, 7–16. [Google Scholar] [PubMed]
- Mendis, E.; Rajapakse, N.; Kim, S.-K. Antioxidant properties of a radical-scavenging peptide purified from enzymatically prepared fish skin gelatin hydrolysate. J. Agric. Food Chem. 2005, 53, 581–587. [Google Scholar] [CrossRef] [PubMed]
- Burlakova, E.B.; Zhizhina, G.P.; Gurevich, S.M.; Fatkullina, L.D.; Kozachenko, A.I.; Nagler, L.G.; Zavarykina, T.M.; Kashcheev, V.V. Biomarkers of oxidative stress and smoking in cancer patients. J. Cancer Res. Ther. 2010, 6, 47–53. [Google Scholar] [PubMed]
- Večeřa, J.; Krishnan, N.; Mithöfer, A.; Vogel, H.; Kodrík, D. Adipokinetic hormone-induced antioxidant response in Spodoptera littoralis. Comp. Biochem. Physiol. C 2012, 155, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Whiteside, C.; Hassan, H.M. Induction and inactivation of catalase and superoxide dismutase of Escherichia coli by ozone. Arch. Biochem. Biophys. 1987, 257, 464–471. [Google Scholar] [CrossRef]
- Farr, S.B.; Kogoma, T. Oxidative stress responses in Escherichia coli and Salmonella typhimurium. Microbiol. Mol. Biol. Rev. 1991, 55, 561–585. [Google Scholar]
- Mannervik, B.; Danielson, U.H.; Ketterer, B. Glutathione transferases—Structure and catalytic activity. CRC Crit. Rev. Biochem. 1988, 23, 283–337. [Google Scholar] [CrossRef] [PubMed]
- Sawicki, R.; Singh, S.P.; Mondal, A.K.; Benes, H.; Zimniak, P. Cloning, expression and biochemical characterization of one ɛ-class (GST-3) and ten Δ-class (GST-1) glutathione S-transferases from Drosophila melanogaster, and identification of additional nine members of the Epsilon class. Biochem. J. 2003, 370, 661–669. [Google Scholar] [CrossRef] [PubMed]
- Ploemen, J.H.T.M.; van Ommen, B.; de Haan, A.; Schefferlie, J.G.; van Bladeren, P.J. In vitro and in vivo reversible and irreversible inhibition of rat glutathione S-transferase isoenzymes by caffeic acid and its 2-S-glutathionyl conjugate. Food Chem. Toxicol. 1993, 31, 475–482. [Google Scholar] [CrossRef]
- Kosower, N.S.; Kosower, E.M. The glutathione status of cells. Int. Rev. Cytol. 1978, 54, 109–160. [Google Scholar] [PubMed]
- Lomaestro, B.M.; Malone, M. Glutathione in health and disease: Pharmacotherapeutic issues. Ann. Pharmacother. 1995, 29, 1263–1273. [Google Scholar] [PubMed]
- Chevion, M.; Berenshtein, E.; Stadtman, E.R. Human studies related to protein oxidation: Protein carbonyl content as a marker of damage. Free Radic. Res. 2000, 33, S99–S108. [Google Scholar] [PubMed]
- Kodrík, D.; Bártů, I.; Socha, R. Adipokinetic hormone (Pyrap-AKH) enhances the effect of a pyrethroid insecticide against the firebug Pyrrhocoris apterus. Pest. Manag. Sci. 2010, 66, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Kodrík, D.; Stašková, T.; Jedličková, V.; Weyda, F.; Závodská, R.; Pflegerová, J. Molecular characterization, tissue distribution, and ultrastructural localization of adipokinetic hormones in the CNS of the firebug Pyrrhocoris apterus (Heteroptera, Insecta). Gen. Comp. Endocrinol. 2015, 210, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Janero, D.R. Malondialdehyde and thiobarbituric acid-reactivity as diagnostic indices of lipid peroxidation and peroxidative tissue injury. Free Radic. Biol. Med. 1990, 9, 515–540. [Google Scholar] [CrossRef]
- Slocinska, M.; Antos-Krzeminska, N.; Rosinski, G.; Jarmuszkiewicz, W. Identification and characterization of uncoupling protein 4 in fat body and muscle mitochondria from the cockroach Gromphadorhina cocquereliana. J. Bioenerg. Biomembr. 2011, 43, 717–727. [Google Scholar] [CrossRef] [PubMed]
- Slocinska, M.; Antos-Krzeminska, N.; Golebiowski, M.; Kuczer, M.; Stepnowski, P.; Rosinski, G.; Jarmuszkiewicz, W. UCP4 expression changes in larval and pupal fat bodies of the beetle Zophobas atratus under adipokinetic hormone treatment. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2013, 166, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Sluse, F.E.; Jarmuszkiewicz, W.; Navet, R.; Douette, P.; Mathy, G.; Sluse-Goffart, C.M. Mitochondrial UCPs: New insights into regulation and impact. Biochim. Biophys. Acta 2006, 1757, 480–485. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Blanco, A.; Fridell, Y.-W.C.; Helfand, S.L. Involvement of Drosophila uncoupling protein 5 in metabolism and aging. Genetics 2006, 172, 1699–1710. [Google Scholar] [CrossRef] [PubMed]
- Gáliková, M.; Diesner, M.; Klepsatel, P.; Hehlert, P.; Xu, Y.; Bickmeyer, I.; Predel, R.; Kühnlein, R.P. Energy homeostasis control in Drosophila adipokinetic hormone mutants. Genetics 2015, 201. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.J.P.; Orchard, I. Is insecticide-induced release of insect neurohormones a secondary effect of hyperactivity of the central nervous system? Pestic. Biochem. Physiol. 1982, 17, 232–242. [Google Scholar] [CrossRef]
- Candy, D.J. Adipokinetic hormones concentrations in the haemolymph of Schistocerca gregaria, measured by radioimmunoassay. Insect Biochem. Mol. Biol. 2002, 32, 1361–1367. [Google Scholar] [CrossRef]
- Kodrík, D.; Socha, R. The effect of insecticide on adipokinetic hormone titre in the insect body. Pest. Manag. Sci. 2005, 61, 1077–1082. [Google Scholar] [CrossRef] [PubMed]
- Che-Mendoza, A.; Penilla, R.; Rodríguez, D. Insecticide resistance and glutathione S-transferases in mosquitoes: A review. Afr. J. Biotechnol. 2009, 8, 1386–1397. [Google Scholar]
- Vontas, J.G.; Small, G.J.; Hemingway, J. Glutathione S-transferases as antioxidant defence agents confer pyrethroid resistance in Nilaparvata lugens. Biochem. J. 2001, 357, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Fragoso, D.B.; Guedes, R.N.C.; Rezende, S.T. Glutathione S-transferase detoxification as a potential pyrethroid resistance mechanism in the maize weevil, Sitophilus zeamais. Entomol. Exp. Appl. 2003, 109, 21–29. [Google Scholar] [CrossRef]
- Kostaropoulos, I.; Papadopoulos, A.I.; Metaxakis, A.; Boukouvala, E.; Papadopoulou-Mourkidou, E. Glutathione S-transferase in the defence against pyrethroids in insects. Insect Biochem. Mol. Biol. 2001, 31, 313–319. [Google Scholar] [CrossRef]
- Staubli, F.; Jorgensen, T.J.D.; Cazzamali, G.; Williamson, M.; Lenz, C.; Sondergaard, L.; Roepstorff, P.; Grimmelikhuijzen, C.J.P. Molecular identification of the insect adipokinetic hormone receptors. Proc. Natl. Acad. Sci. USA 2002, 99, 3446–3451. [Google Scholar] [CrossRef] [PubMed]
- Caers, J.; Verlinden, H.; Zels, S.; Vandersmissen, H.P.; Vuerinckx, K.; Schoofs, L. More than two decades of research on insect neuropeptide GPCRs: An overview. Front. Endocrinol. 2012, 3, 151. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.; Kim, Y.-J.; Adams, M.E. Identification of G protein-coupled receptors for Drosophila PRXamide peptides, CCAP, corazonin, and AKH supports a theory of ligand-receptor coevolution. Proc. Natl. Acad. Sci. USA 2002, 99, 11423–11428. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.; Huang, H.; Hua, R.; Li, G.; Yang, D.; Luo, J.; Zhang, C.; Shi, L.; Benovic, J.L.; Zhou, N. Molecular and functional characterization of adipokinetic hormone receptor and its peptide ligands in Bombyx mori. FEBS Lett. 2009, 583, 1463–1468. [Google Scholar] [CrossRef] [PubMed]
- Hansen, K.K.; Hauser, F.; Cazzamali, G.; Williamson, M.; Grimmelikhuijzen, C.J.P. Cloning and characterization of the adipokinetic hormone receptor from the cockroach Periplaneta americana. Biochem. Biophys. Res. Commun. 2006, 343, 638–643. [Google Scholar] [CrossRef] [PubMed]
- Kaufmann, C.; Brown, M.R. Adipokinetic hormones in the African malaria mosquito, Anopheles gambiae: Identification and expression of genes for two peptides and a putative receptor. Insect Biochem. Mol. Biol. 2006, 36, 466–481. [Google Scholar] [CrossRef] [PubMed]
- Wicher, D.; Agricola, H.-J.; Söhler, S.; Gundel, M.; Heinemann, S.H.; Wollweber, L.; Stengl, M.; Derst, C. Differential receptor activation by cockroach adipokinetic hormones produces differential effects on ion currents, neuronal activity, and locomotion. J. Neurophysiol. 2006, 95, 2314–2325. [Google Scholar] [CrossRef] [PubMed]
- Kaufmann, C.; Merzendorfer, H.; Gäde, G. The adipokinetic hormone system in Culicinae (Diptera: Culicidae): molecular identification and characterization of two adipokinetic hormone (AKH) precursors from Aedes aegypti and Culex pipiens and two putative AKH receptor variants from A. aegypti. Insect Biochem. Mol. Biol. 2009, 39, 770–781. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, R.; Isoe, J.; Moore, W.; Riehle, M.A.; Wells, M.A. The putative AKH receptor of the tobacco hornworm, Manduca sexta, and its expression. J. Insect Sci. 2011, 11, 40. [Google Scholar] [CrossRef] [PubMed]
- Spencer, I.M.; Candy, D.J. Hormonal control of diacyl glycerol mobilization from fat body of the desert locust, Schistocerca gregaria. Insect Biochem. 1976, 6, 289–296. [Google Scholar] [CrossRef]
- Vroemen, S.F.; van Marrewijk, W.J.A.; de Meijer, J.; van den Broek, A.T.; van der Horst, D.J. Differential induction of inositol phosphate metabolism by three adipokinetic hormones. Mol. Cell. Endocrinol. 1997, 130, 131–139. [Google Scholar] [CrossRef]
- Van der Horst, D.J.; van Marrewijk, W.J.A.; Diederen, J.H. Adipokinetic hormones of insect: Release, signal transduction, and responses. Int. Rev. Cytol. 2001, 211, 179–240. [Google Scholar] [PubMed]
- Bednářová, A.; Kodrík, D.; Krishnan, N. Adipokinetic hormone exerts its anti-oxidative effects using a conserved signal-transduction mechanism involving both PKC and cAMP by mobilizing extra- and intracellular Ca2+ stores. Comp. Biochem. Physiol. C 2013, 158, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Bednářová, A.; Kodrík, D.; Krishnan, N. Knockdown of adipokinetic hormone synthesis increases susceptibility to oxidative stress in Drosophila—A role for dFoxO? Comp. Biochem. Physiol. C 2015, 171, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Yang, Z.; Yue, Z.; Alvarez, J.D.; Sehgal, A. FOXO and insulin signaling regulate sensitivity of the circadian clock to oxidative stress. Proc. Natl. Acad. Sci. USA 2007, 104, 15899–15904. [Google Scholar]
- Süren-Castillo, S.; Abrisqueta, M.; Maestro, J.L. FoxO is required for the activation of hypertrehalosemic hormone expression in cockroaches. Biochim. Biophys. Acta 2014, 1840, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Hay, N. Interplay between FOXO, TOR, and Akt. Biochim. Biophys. Acta 2011, 1813, 1965–1970. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Budanov, A.V.; Park, E.J.; Birse, R.; Kim, T.E.; Perkins, G.A.; Ocorr, K.; Ellisman, M.H.; Bodmer, R.; Bier, E.; Karin, M. Sestrin as a feedback inhibitor of TOR that prevents age-related pathologies. Science 2010, 327, 1223–1228. [Google Scholar] [CrossRef] [PubMed]
- Peeters, H.; Debeer, P.; Bairoch, A.; Wilquet, V.; Huysmans, C.; Parthoens, E.; Fryns, J.P.; Gewillig, M.; Nakamura, Y.; Niikawa, N.; et al. PA26 is a candidate gene for heterotaxia in humans: Identification of a novel PA26-related gene family in human and mouse. Hum. Genet. 2003, 112, 573–580. [Google Scholar] [PubMed]
- Budanov, A.V; Sablina, A.A.; Feinstein, E.; Koonin, E.V; Chumakov, P.M. Regeneration of peroxiredoxins by p53-regulated sestrins, homologs of bacterial AhpD. Science 2004, 304, 596–600. [Google Scholar] [CrossRef] [PubMed]
- Budanov, A.V; Karin, M. p53 target genes sestrin1 and sestrin2 connect genotoxic stress and mTOR signaling. Cell 2008, 134, 451–460. [Google Scholar] [CrossRef] [PubMed]
- Eijkelenboom, A.; Burgering, B.M.T. FOXOs: Signalling integrators for homeostasis maintenance. Nat. Rev. Mol. Cell Biol. 2013, 14, 83–97. [Google Scholar] [CrossRef] [PubMed]
- Jünger, M.A.; Rintelen, F.; Stocker, H.; Wasserman, J.D.; Végh, M.; Radimerski, T.; Greenberg, M.E.; Hafen, E. The Drosophila forkhead transcription factor FOXO mediates the reduction in cell number associated with reduced insulin signaling. J. Biol. 2003, 2, 20. [Google Scholar] [CrossRef] [PubMed]
- Mattila, J.; Bremer, A.; Ahonen, L.; Kostiainen, R.; Puig, O. Drosophila FoxO regulates organism size and stress resistance through an adenylate cyclase. Mol. Cell. Biol. 2009, 29, 5357–5365. [Google Scholar] [CrossRef] [PubMed]
- Tong, J.J.; Schriner, S.E.; McCleary, D.; Day, B.J.; Wallace, D.C. Life extension through neurofibromin mitochondrial regulation and antioxidant therapy for neurofibromatosis-1 in Drosophila melanogaster. Nat. Genet. 2007, 39, 476–485. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Goode, J.; Best, J.; Meltzer, J.; Schilman, P.E.; Chen, J.; Garza, D.; Thomas, J.B.; Montminy, M. The insulin-regulated CREB coactivator TORC promotes stress resistance in Drosophila. Cell Metab. 2008, 7, 434–444. [Google Scholar] [CrossRef] [PubMed]
- Essers, M.A.G.; Weijzen, S.; de Vries-Smits, A.M.M.; Saarloos, I.; de Ruiter, N.D.; Bos, J.L.; Burgering, B.M.T. FOXO transcription factor activation by oxidative stress mediated by the small GTPase Ral and JNK. EMBO J. 2004, 23, 4802–4812. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.C.; Bohmann, D.; Jasper, H. JNK extends life span and limits growth by antagonizing cellular and organism-wide responses to insulin signaling. Cell 2005, 121, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Kawamori, D.; Kaneto, H.; Nakatani, Y.; Matsuoka, T.-A.; Matsuhisa, M.; Hori, M.; Yamasaki, Y. The forkhead transcription factor Foxo1 bridges the JNK pathway and the transcription factor PDX-1 through its intracellular translocation. J. Biol. Chem. 2006, 281, 1091–1098. [Google Scholar] [CrossRef] [PubMed]
- Kops, G.J.; de Ruiter, N.D.; de Vries-Smits, A.M.; Powell, D.R.; Bos, J.L.; Burgering, B.M. Direct control of the Forkhead transcription factor AFX by protein kinase B. Nature 1999, 398, 630–634. [Google Scholar] [PubMed]
- Biggs, W.H.; Meisenhelder, J.; Hunter, T.; Cavenee, W.K.; Arden, K.C. Protein kinase B/Akt-mediated phosphorylation promotes nuclear exclusion of the winged helix transcription factor FKHR1. Proc. Natl. Acad. Sci. USA 1999, 96, 7421–7426. [Google Scholar] [CrossRef] [PubMed]
- Glauser, D.A.; Schlegel, W. The emerging role of FOXO transcription factors in pancreatic β cells. J. Endocrinol. 2007, 193, 195–207. [Google Scholar] [CrossRef] [PubMed]
- Buch, S.; Melcher, C.; Bauer, M.; Katzenberger, J.; Pankratz, M.J. Opposing effects of dietary protein and sugar regulate a transcriptional target of Drosophila insulin-like peptide signaling. Cell Metab. 2008, 7, 321–332. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.W.; Haddad, G.G. Review: Hypoxic and oxidative stress resistance in Drosophila melanogaster. Placenta 2011, 32, S104–S108. [Google Scholar] [CrossRef] [PubMed]
- Adams, M.D.; Celniker, S.E.; Holt, R.A.; Evans, C.A.; Gocayne, J.D.; Amanatides, P.G.; Scherer, S.E.; Li, P.W.; Hoskins, R.A.; et al. The genome sequence of Drosophila melanogaster. Science 2000, 287, 2185–2195. [Google Scholar] [CrossRef] [PubMed]
- Noyes, B.E.; Katz, F.N.; Schaffer, M.H. Identification and expression of the Drosophila adipokinetic hormone gene. Mol. Cell. Endocrinol. 1995, 109, 133–141. [Google Scholar] [CrossRef]
- Schaffer, M.H.; Noyes, B.E.; Slaughter, C.A.; Thorne, G.C.; Gaskell, S.J. The fruitfly Drosophila melanogaster contains a novel charged adipokinetic-hormone-family peptide. Biochem. J. 1990, 269, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Grönke, S.; Müller, G.; Hirsch, J.; Fellert, S.; Andreou, A.; Haase, T.; Jäckle, H.; Kühnlein, R.P. Dual lipolytic control of body fat storage and mobilization in Drosophila. PLoS Biol. 2007, 5, e137. [Google Scholar] [CrossRef] [PubMed]
- Bharucha, K.N.; Tarr, P.; Zipursky, S.L. A glucagon-like endocrine pathway in Drosophila modulates both lipid and carbohydrate homeostasis. J. Exp. Biol. 2008, 211, 3103–3110. [Google Scholar] [CrossRef] [PubMed]
- Baumbach, J.; Xu, Y.; Hehlert, P.; Kühnlein, R.P. Gαq, Gγ1 and Plc21C control Drosophila body fat storage. J. Genet. Genom. 2014, 41, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.K.; Rulifson, E.J. Conserved mechanisms of glucose sensing and regulation by Drosophila corpora cardiaca cells. Nature 2004, 431, 316–320. [Google Scholar] [CrossRef] [PubMed]
- Isabel, G.; Martin, J.-R.; Chidami, S.; Veenstra, J.A.; Rosay, P. AKH-producing neuroendocrine cell ablation decreases trehalose and induces behavioral changes in Drosophila. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2005, 288, R531–R538. [Google Scholar] [PubMed]
- Waterson, M.J.; Chung, B.Y.; Harvanek, Z.M.; Ostojic, I.; Alcedo, J.; Pletcher, S.D. Water sensor ppk28 modulates Drosophila lifespan and physiology through AKH signaling. Proc. Natl. Acad. Sci. USA 2014, 111, 8137–8142. [Google Scholar] [CrossRef] [PubMed]
- Katewa, S.D.; Demontis, F.; Kolipinski, M.; Hubbard, A.; Gill, M.S.; Perrimon, N.; Melov, S.; Kapahi, P. Intramyocellular fatty-acid metabolism plays a critical role in mediating responses to dietary restriction in Drosophila melanogaster. Cell Metab. 2012, 16, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Braco, J.T.; Gillespie, E.L.; Alberto, G.E.; Brenman, J.E.; Johnson, E.C. Energy-dependent modulation of glucagon-like signaling in Drosophila via the AMP-activated protein kinase. Genetics 2012, 192, 457–466. [Google Scholar] [CrossRef] [PubMed]
- Sajwan, S.; Sidorov, R.; Stašková, T.; Žaloudíková, A.; Takasu, Y.; Kodrík, D.; Zurovec, M. Targeted mutagenesis and functional analysis of adipokinetic hormone-encoding gene in Drosophila. Insect Biochem. Mol. Biol. 2015, 61, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Kannan, K.; Fridell, Y.-W.C. Functional implications of Drosophila insulin-like peptides in metabolism, aging, and dietary restriction. Front. Physiol. 2013, 4, 288. [Google Scholar] [CrossRef] [PubMed]
- Nässel, D.R.; Liu, Y.; Luo, J. Insulin/IGF signaling and its regulation in Drosophila. Gen. Comp. Endocrinol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Maier, V.; Fuchs, J.; Pfeiffer, E.F.; Bounias, M. Glucagon as a non species-specific regulator of the glycaemia in honeybee (Apis mellifica). Diabete Matab. 1990, 16, 428–434. [Google Scholar]
- Alquicer, G.; Kodrík, D.; Krishnan, N.; Vecera, J.; Socha, R. Activation of insect anti-oxidative mechanisms by mammalian glucagon. Comp. Biochem. Physiol. B 2009, 152, 226–233. [Google Scholar] [CrossRef] [PubMed]
- Bednářová, A.; Kodrík, D.; Krishnan, N. Unique roles of glucagon and glucagon-like peptides: Parallels in understanding the functions of adipokinetic hormones in stress responses in insects. Comp. Biochem. Physiol. A 2013, 164, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Veenstra, J.A. Isolation and structure of corazonin, a cardioactive peptide from the American cockroach. FEBS Lett. 1989, 250, 231–234. [Google Scholar] [CrossRef]
- Boerjan, B.; Verleyen, P.; Huybrechts, J.; Schoofs, L.; de Loof, A. In search for a common denominator for the diverse functions of arthropod corazonin: A role in the physiology of stress? Gen. Comp. Endocrinol. 2010, 166, 222–233. [Google Scholar] [CrossRef] [PubMed]
- Veenstra, J.A. Does corazonin signal nutritional stress in insects? Insect Biochem. Mol. Biol. 2009, 39, 755–762. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Bretz, C.A.; Hawksworth, S.A.; Hirsh, J.; Johnson, E.C. Corazonin neurons function in sexually dimorphic circuitry that shape behavioral responses to stress in Drosophila. PLoS ONE 2010, 5, e9141. [Google Scholar] [CrossRef] [PubMed]
- Ishizaki, H.; Suzuki, A. The brain secretory peptides that control moulting and metamorphosis of the silkmoth, Bombyx mori. Int. J. Dev. Biol. 1994, 38, 301–310. [Google Scholar] [PubMed]
- Kawakami, A.; Kataoka, H.; Oka, T.; Mizoguchi, A.; Kimura-Kawakami, M.; Adachi, T.; Iwami, M.; Nagasawa, H.; Suzuki, A.; Ishizaki, H. Molecular cloning of the Bombyx mori prothoracicotropic hormone. Science 1990, 247, 1333–1335. [Google Scholar] [CrossRef] [PubMed]
- Agui, N.; Bollenbacher, W.E.; Granger, N.A.; Gilbert, L.I. Corpus allatum is release site for insect prothoracicotropic hormone. Nature 1980, 285, 669–670. [Google Scholar] [CrossRef]
- Perić-Mataruga, V.; Vlahović, M.; Mrdaković, M.; Todorović, D.; Matić, D.; Gavrilović, A.; Ilijin, L. Prothoracicotropic hormone-producing neurosecretory neurons and antioxidative defense in midgut of Lymantria dispar in trophic stress. TURKISH J. Biol. 2014, 38, 403–411. [Google Scholar] [CrossRef]
- Krishnan, N.; Večeřa, J.; Kodrík, D.; Sehnal, F. 20-Hydroxyecdysone prevents oxidative stress damage in adult Pyrrhocoris apterus. Arch. Insect Biochem. Physiol. 2007, 65, 114–124. [Google Scholar] [CrossRef] [PubMed]
- Roesijadi, G.; Rezvankhah, S.; Binninger, D.M.; Weissbach, H. Ecdysone induction of MsrA protects against oxidative stress in Drosophila. Biochem. Biophys. Res. Commun. 2007, 354, 511–516. [Google Scholar] [CrossRef] [PubMed]
- Brot, N.; Weissbach, H. Biochemistry of methionine sulfoxide residues in proteins. Biofactors 1991, 3, 91–96. [Google Scholar] [PubMed]
- Weissbach, H.; Resnick, L.; Brot, N. Methionine sulfoxide reductases: History and cellular role in protecting against oxidative damage. Biochim. Biophys. Acta Proteins Proteom. 2005, 1703, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Cherbas, L.; Cherbas, P. Transcription activation by the ecdysone receptor (EcR/USP): Identification of activation functions. Mol. Endocrinol. 2003, 17, 716–731. [Google Scholar] [CrossRef] [PubMed]
- Moskovitz, J.; Bar-Noy, S.; Williams, W.M.; Requena, J.; Berlett, B.S.; Stadtman, E.R. Methionine sulfoxide reductase (MsrA) is a regulator of antioxidant defense and lifespan in mammals. Proc. Natl. Acad. Sci. USA 2001, 98, 12920–12925. [Google Scholar] [CrossRef] [PubMed]
- Kantorow, M.; Hawse, J.R.; Cowell, T.L.; Benhamed, S.; Pizarro, G.O.; Reddy, V.N.; Hejtmancik, J.F. Methionine sulfoxide reductase A is important for lens cell viability and resistance to oxidative stress. Proc. Natl. Acad. Sci. USA 2004, 101, 9654–9659. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Luo, C.X.; Chu, W.H.; Shan, Y.A.; Qian, Z.M.; Zhu, G.; Yu, Y.B.; Feng, H. 20-Hydroxyecdysone protects against oxidative stress-induced neuronal injury by scavenging free radicals and modulating NF-κB and JNK pathways. PLoS ONE 2012, 7, e50764. [Google Scholar] [CrossRef] [PubMed]
- Lacort, M.; Leal, A.M.; Liza, M.; Martín, C.; Martínez, R.; Ruiz-Larrea, M.B. Protective effect of estrogens and catecholestrogens against peroxidative membrane damagein vitro. Lipids 1995, 30, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, R.; Bai, H.; Dolezal, A.G.; Amdam, G.; Tatar, M. Juvenile hormone regulation of Drosophila aging. BMC Biol. 2013, 11, 85. [Google Scholar] [CrossRef] [PubMed]
- Sezer, B.; Ozalp, P. Effect of juvenile hormone analogue, pyriproxyfen on antioxidant enzymes of greater wax moth, Galleria mellonella (Lepidoptera: Pyralidae: Galleriinae) Larvae. Pak. J. Zool. 2015, 47, 665–669. [Google Scholar]
- Fahmy, N. Impact of two insect growth regulators on the enhancement of oxidative stress and antioxidant efficiency of the cotton leaf worm, Spodoptera littoralis (Biosd.). Egypt. Acad. J. Biol. Sci. 2012, 5, 137–149. [Google Scholar]
- Zou, F.M.; Lou, D.S.; Zhu, Y.H.; Wang, S.P.; Jin, B.R.; Gui, Z.Z. Expression profiles of glutathione S-transferase genes in larval midgut of Bombyx mori exposed to insect hormones. Mol. Biol. Rep. 2011, 38, 639–647. [Google Scholar] [CrossRef] [PubMed]
- Seehuus, S.C.; Norberg, K.; Gimsa, U.; Krekling, T.; Amdam, G.V. Reproductive protein protects functionally sterile honey bee workers from oxidative stress. Proc. Natl. Acad. Sci. USA 2006, 103, 962–967. [Google Scholar] [CrossRef] [PubMed]
- Jamroz, R.C.; Gasdaska, J.R.; Bradfield, J.Y.; Law, J.H. Transferrin in a cockroach: Molecular cloning, characterization, and suppression by juvenile hormone. Proc. Natl. Acad. Sci. USA 1993, 90, 1320–1324. [Google Scholar] [CrossRef] [PubMed]
- Harizanova, N.; Georgieva, T.; Dunkov, B.C.; Yoshiga, T.; Law, J.H. Aedes aegypti transferrin. Gene structure, expression pattern, and regulation. Insect Mol. Biol. 2005, 14, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Chapman, R.F. The insects: structure and function, 4th ed.; Cambridge University Press: Cambridge, UK; New York, NY, USA; Melbourne, Australia, 1998. [Google Scholar]
- Andrus, P.K.; Fleck, T.J.; Gurney, M.E.; Hall, E.D. Protein oxidative damage in a transgenic mouse model of familial amyotrophic lateral sclerosis. J. Neurochem. 1998, 71, 2041–2048. [Google Scholar] [CrossRef] [PubMed]
- Meneghini, R. Iron homeostasis, oxidative stress, and DNA damage. Free Radic. Biol. Med. 1997, 23, 783–792. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kodrík, D.; Bednářová, A.; Zemanová, M.; Krishnan, N. Hormonal Regulation of Response to Oxidative Stress in Insects—An Update. Int. J. Mol. Sci. 2015, 16, 25788-25816. https://doi.org/10.3390/ijms161025788
Kodrík D, Bednářová A, Zemanová M, Krishnan N. Hormonal Regulation of Response to Oxidative Stress in Insects—An Update. International Journal of Molecular Sciences. 2015; 16(10):25788-25816. https://doi.org/10.3390/ijms161025788
Chicago/Turabian StyleKodrík, Dalibor, Andrea Bednářová, Milada Zemanová, and Natraj Krishnan. 2015. "Hormonal Regulation of Response to Oxidative Stress in Insects—An Update" International Journal of Molecular Sciences 16, no. 10: 25788-25816. https://doi.org/10.3390/ijms161025788
APA StyleKodrík, D., Bednářová, A., Zemanová, M., & Krishnan, N. (2015). Hormonal Regulation of Response to Oxidative Stress in Insects—An Update. International Journal of Molecular Sciences, 16(10), 25788-25816. https://doi.org/10.3390/ijms161025788