Structural Diversity of the Microbial Surfactin Derivatives from Selective Esterification Approach
Abstract
:1. Introduction
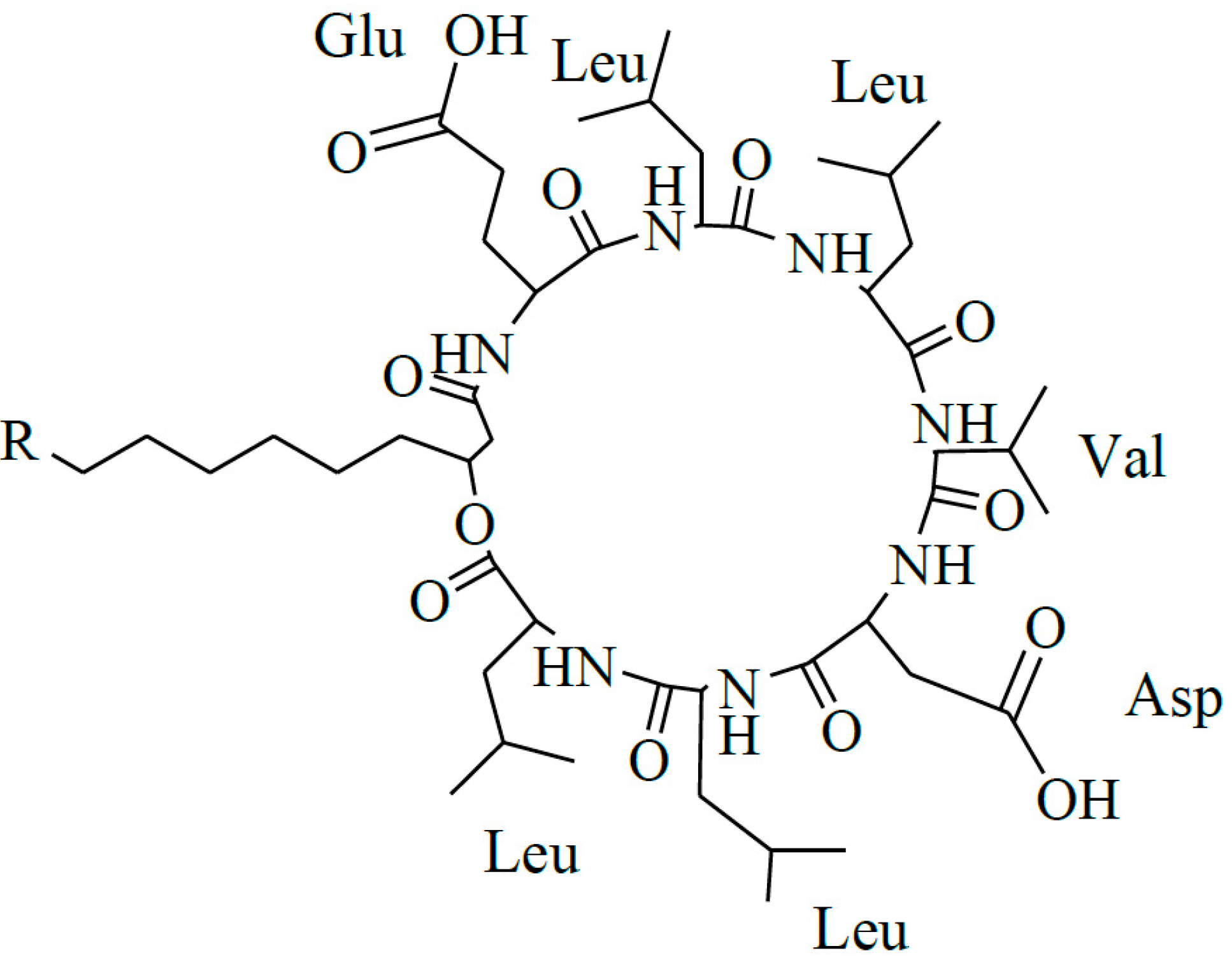
2. Results and Discussion
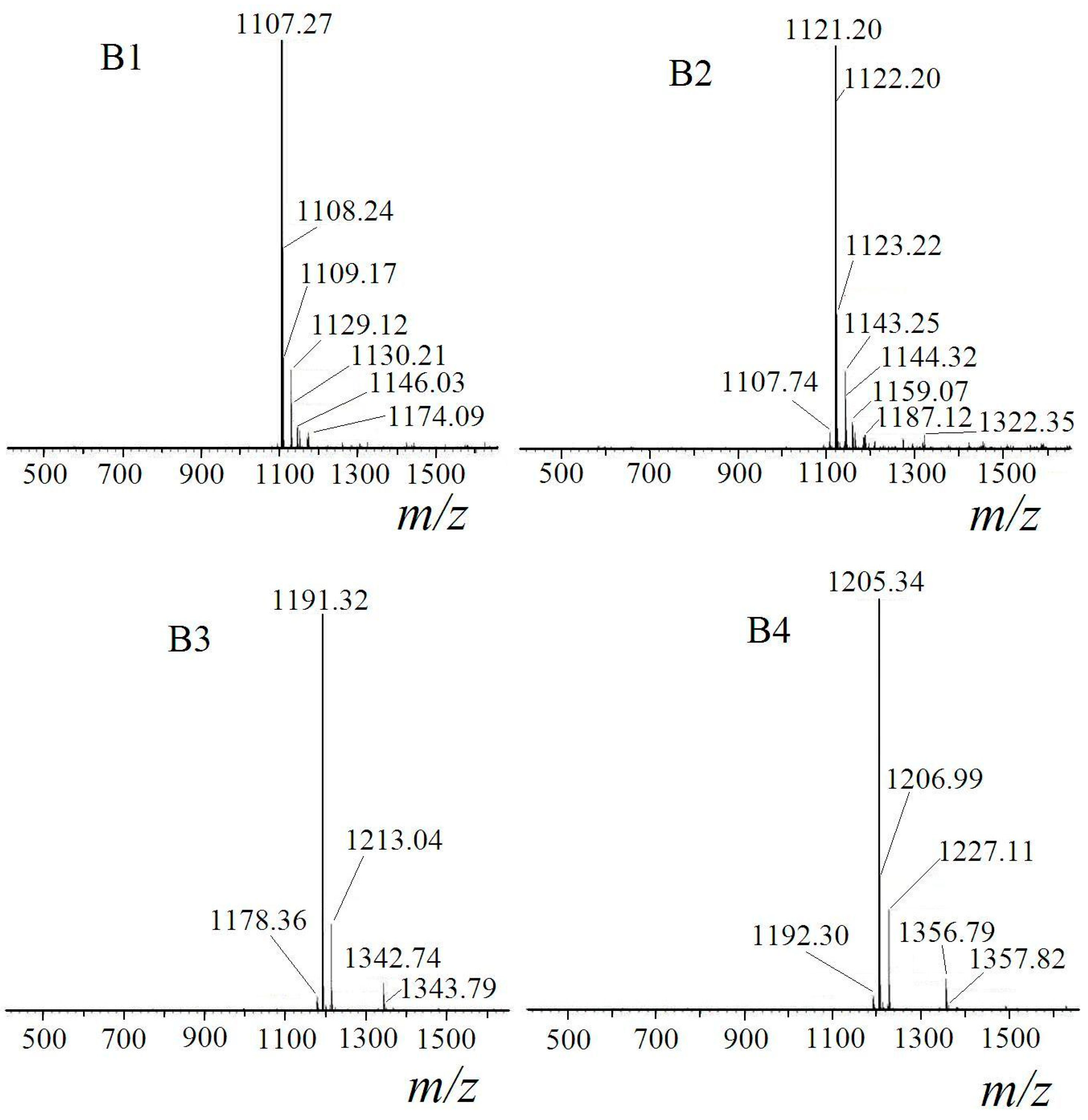
2.1. Identification of Surfactin-(Glu-γ, Asp-β)-Hexyl Ester
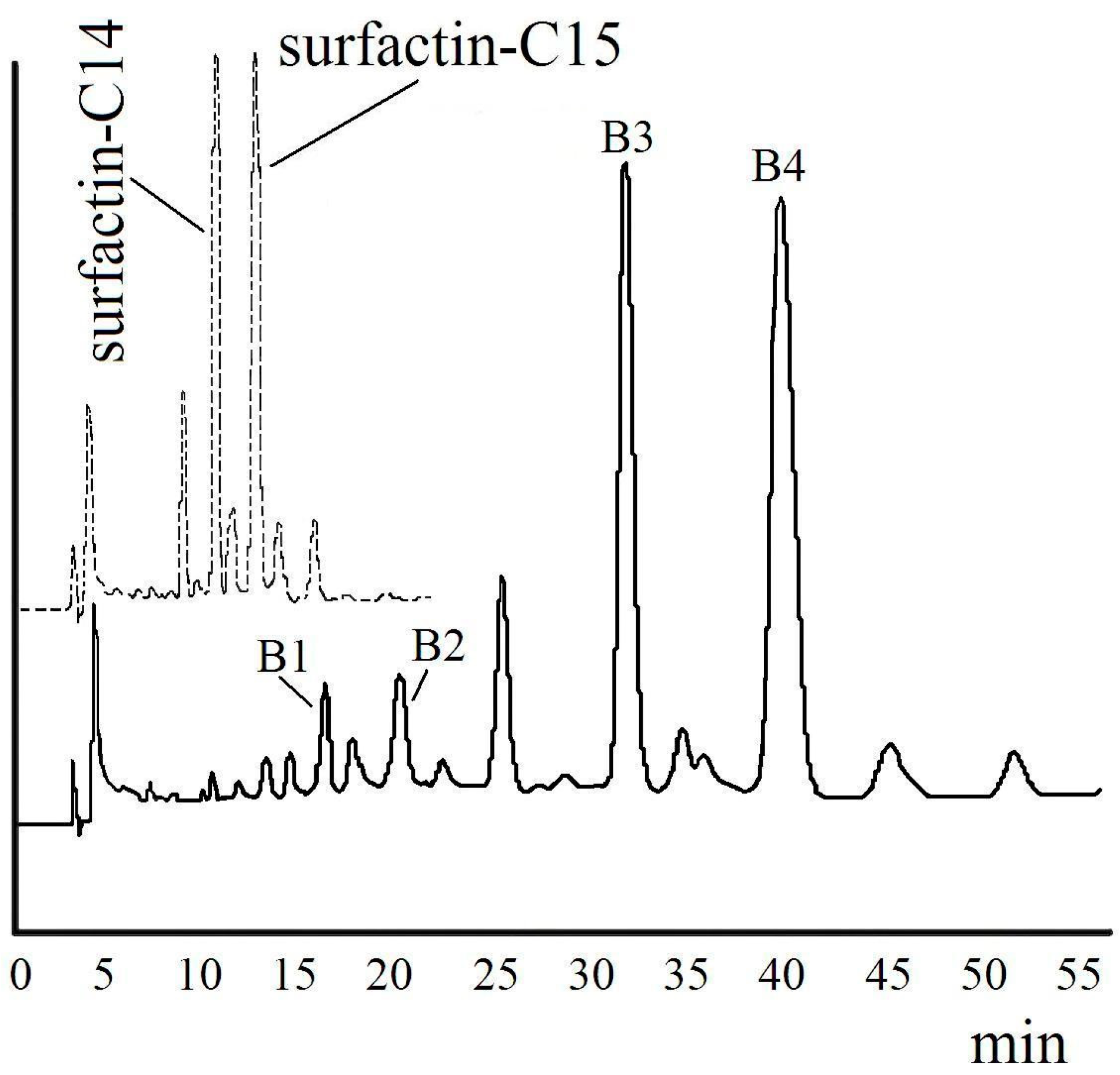
| Fraction | m/z | Ionization Mode | Molecular Weight | Compound |
|---|---|---|---|---|
| B1 | 1107 | [M + H]+ | 1106 | mono-hexyl-surfactin C14 ester |
| 1129 | [M + Na]+ | |||
| B2 | 1121 | [M + H]+ | 1120 | mono-hexyl-surfactin C15 ester |
| 1143 | [M + Na]+ | |||
| B3 | 1191 | [M + H]+ | 1190 | di-hexyl-surfactin C14 ester |
| 1213 | [M + Na]+ | |||
| B4 | 1205 | [M + H]+ | 1204 | di-hexyl-surfactin C15 ester |
| 1227 | [M + Na]+ |
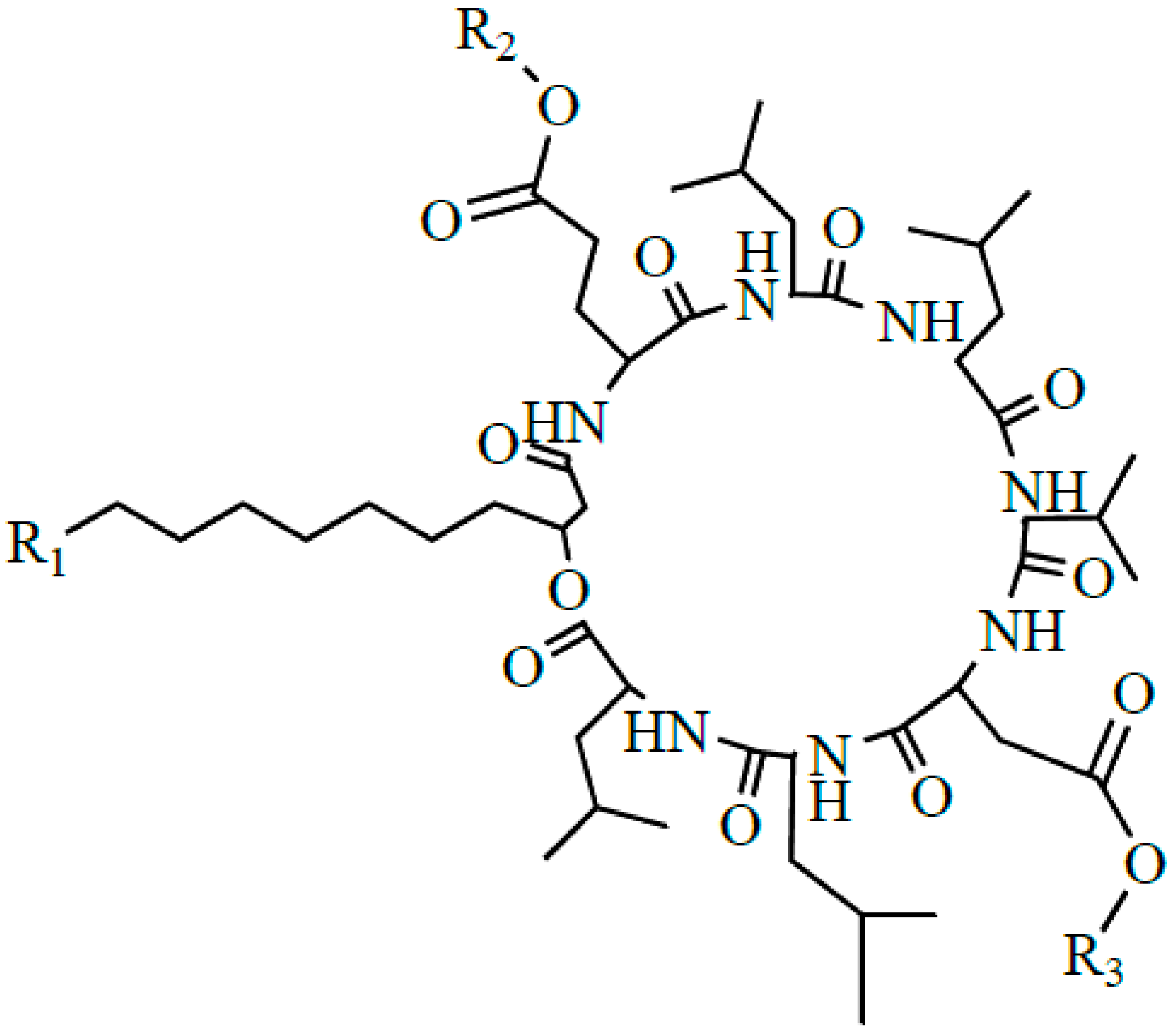
2.2. Identification of (Glu-γ, Asp-β)-2-Methoxy-Ethyl-Surfactin Ester
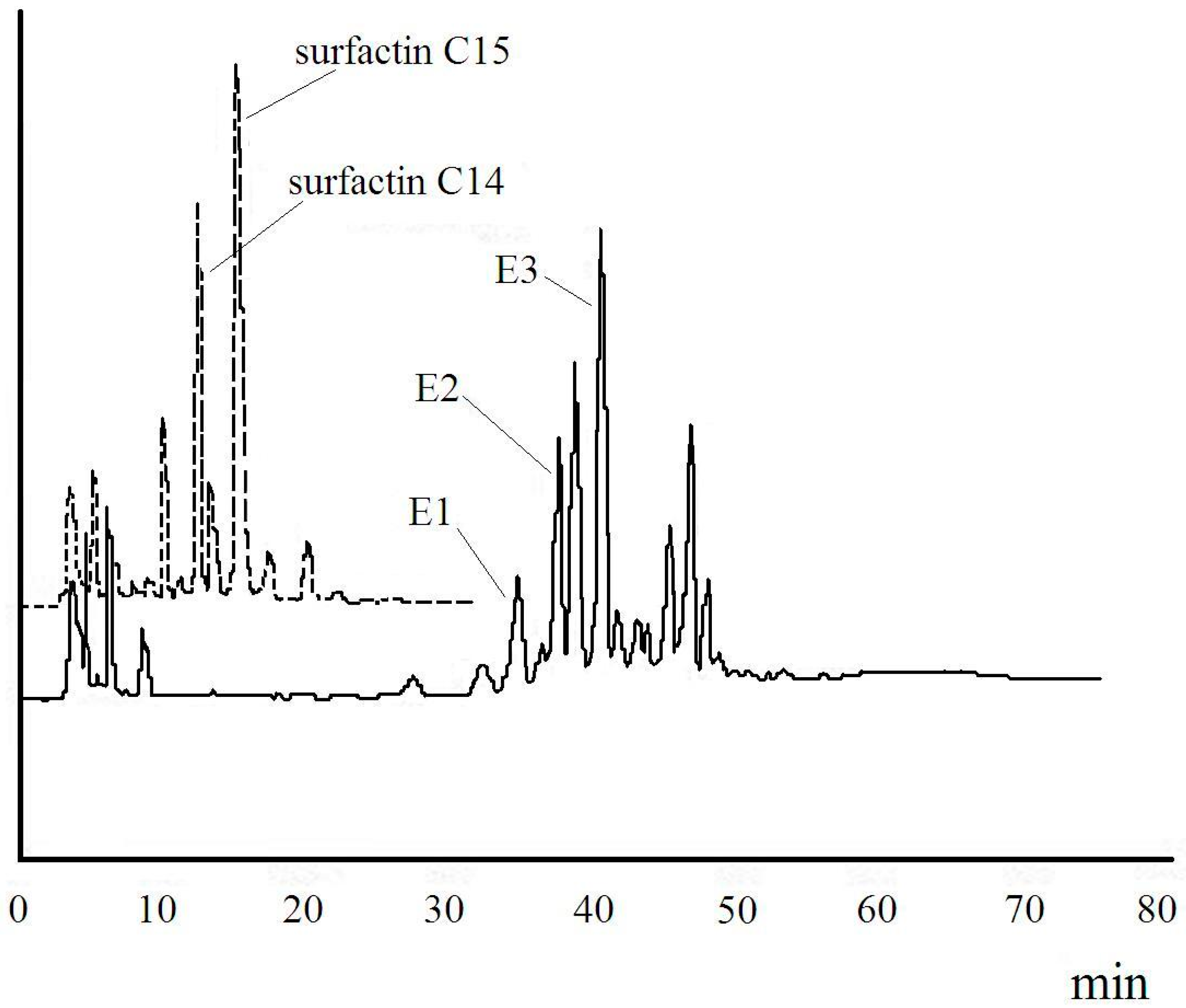
| Fraction | m/z | Ionization Mode | Molecular Weight | Compound |
|---|---|---|---|---|
| E1 | 1102 | [M + Na]+ | 1079 | mono-2-methoxy-ethyl-surfactin C14 ester |
| E2 | 1160 | [M + Na]+ | 1137 | di-2-methoxy-ethyl-surfactin C14 ester |
| E3 | 1174 | [M + Na]+ | 1151 | di-2-methoxy-ethyl-surfactin C15 ester |


2.3. Identification of Surfactin-(Glu-γ, Asp-β)-6-Hydoxyl-Hexyl Ester
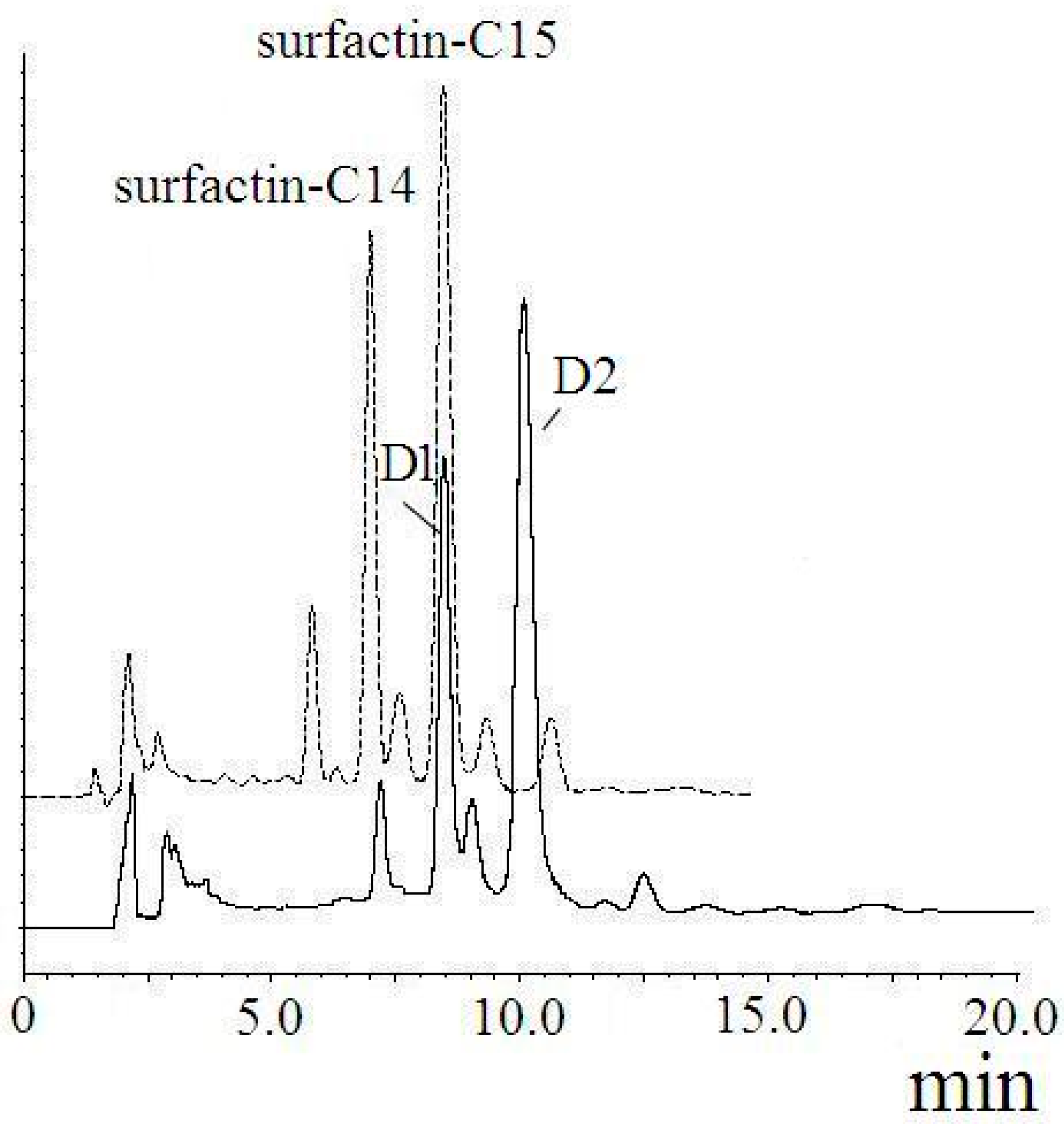
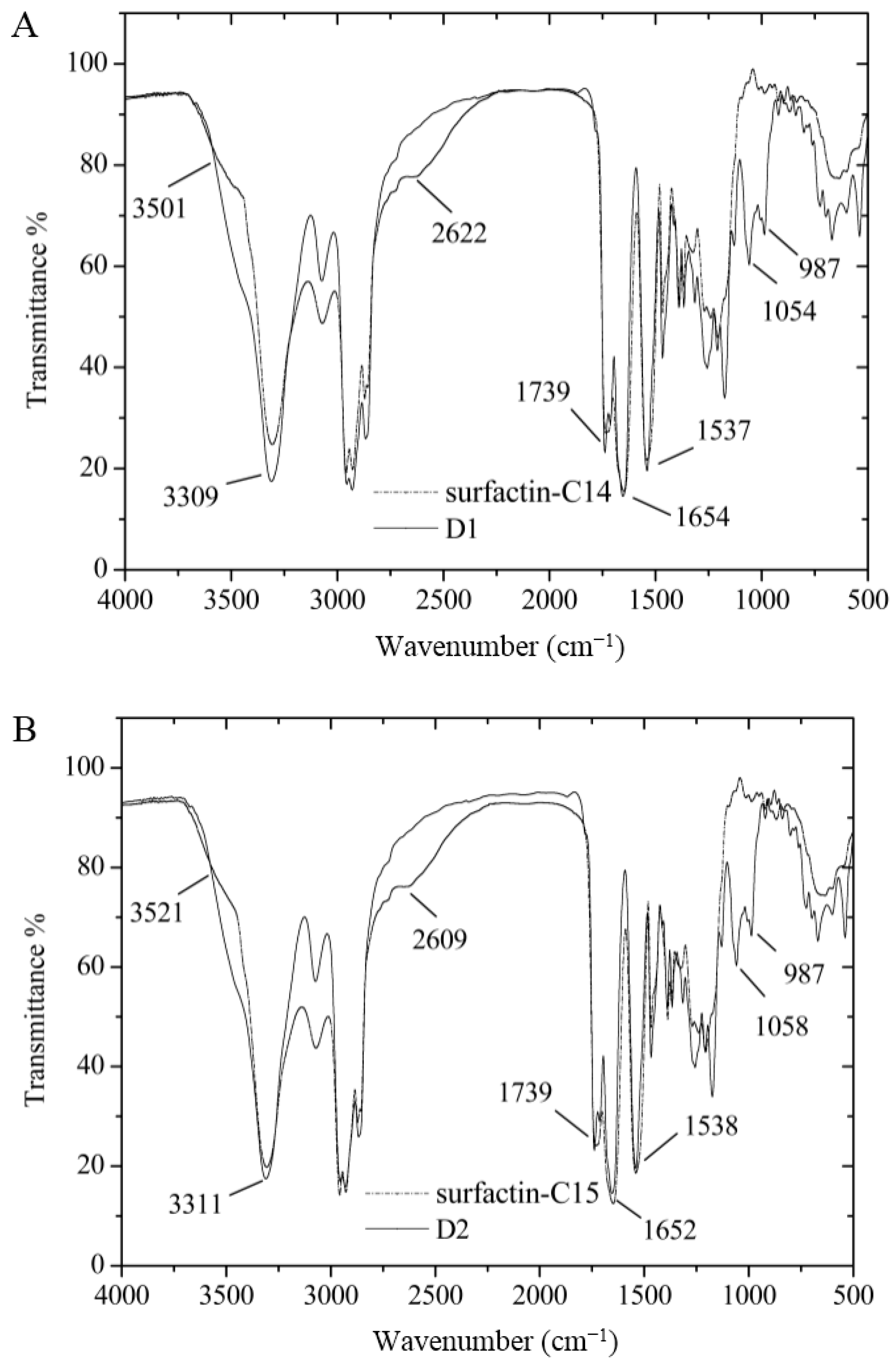
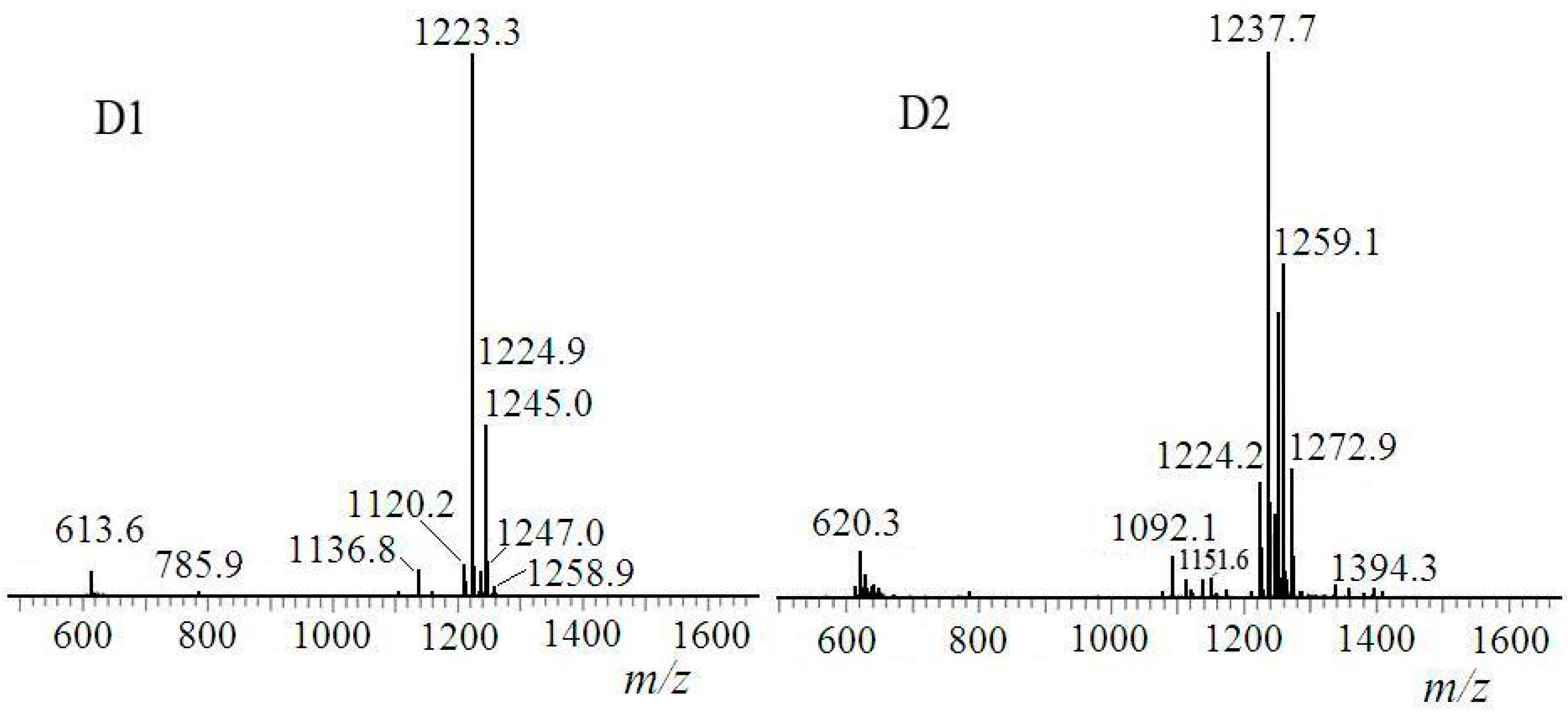
| Fraction | m/z | Ionization Mode | Molecular Weight | Compound |
|---|---|---|---|---|
| D1 | 1223 | [M + H]+ | 1222 | di-6-hydoxyl-hexyl-surfactin C14 ester |
| 1245 | [M + Na]+ | |||
| D2 | 1237 | [M + H]+ | 1236 | di-6-hydoxyl-hexyl-surfactin C15 ester |
| 1259 | [M + Na]+ |
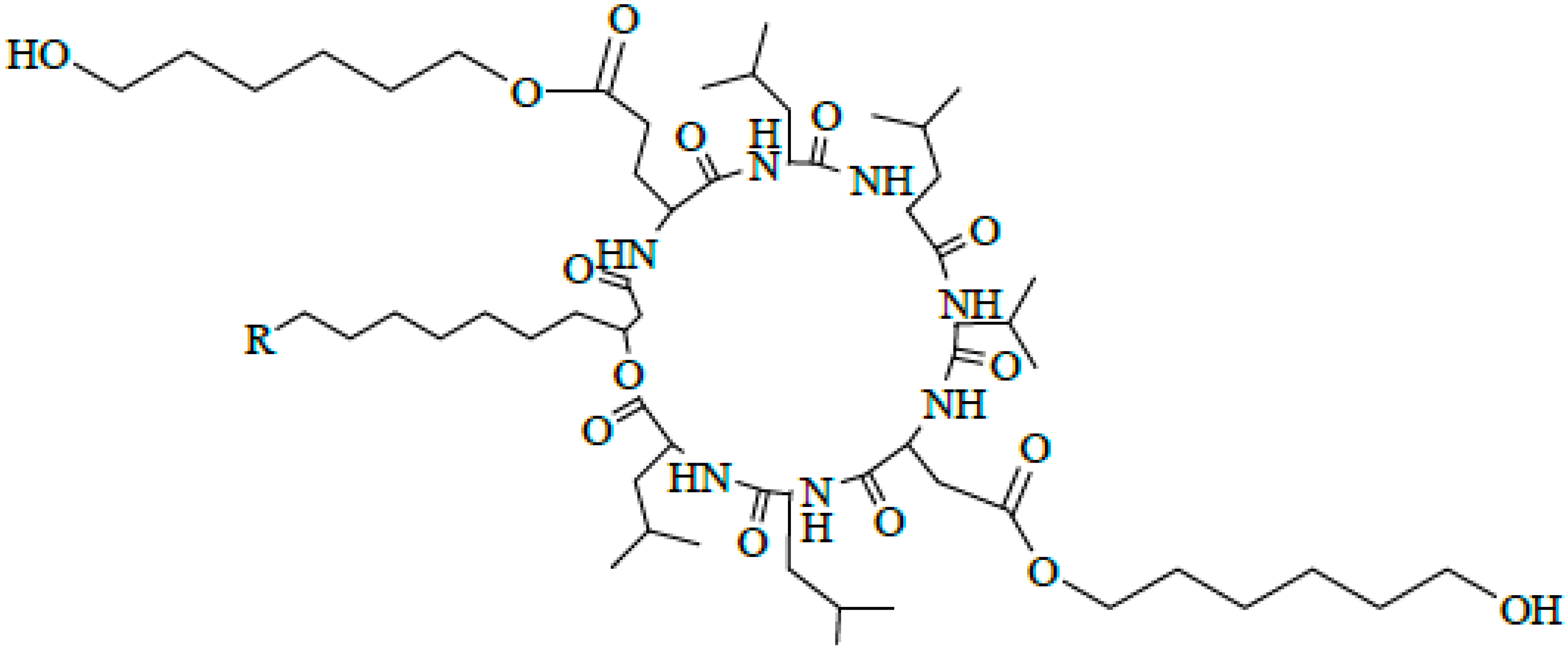
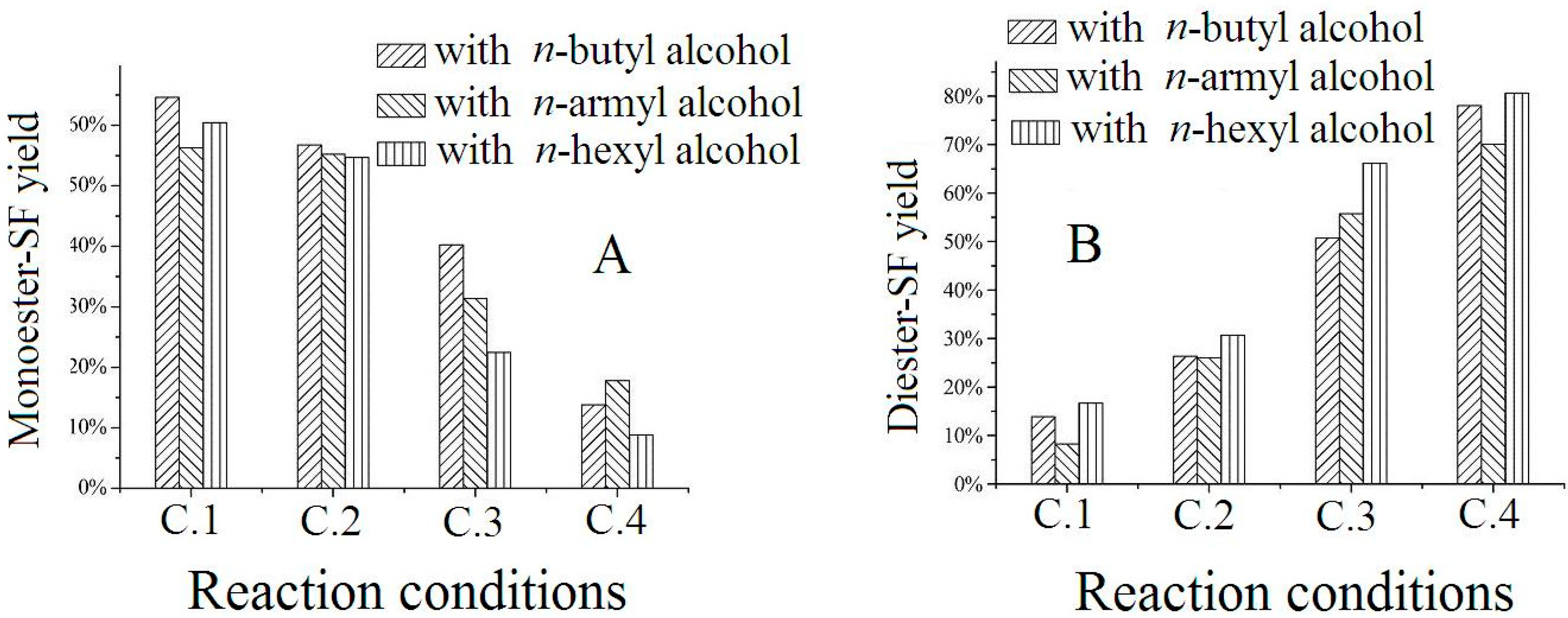
2.4. Discussion
3. Experimental Section
3.1. Reagents
3.2. Preparation of (Glu-γ, Asp-β)-Hexyl-Surfactin Ester
3.3. Preparation of (Glu-γ, Asp-β)-2-Methoxy-Ethyl-Surfactin Ester
3.4. Preparation of (Glu-γ, Asp-β)-6-Hydoxyl-Hexyl-Surfactin Ester
3.5. Analysis and Purification
3.6. Molecular Weight Determination
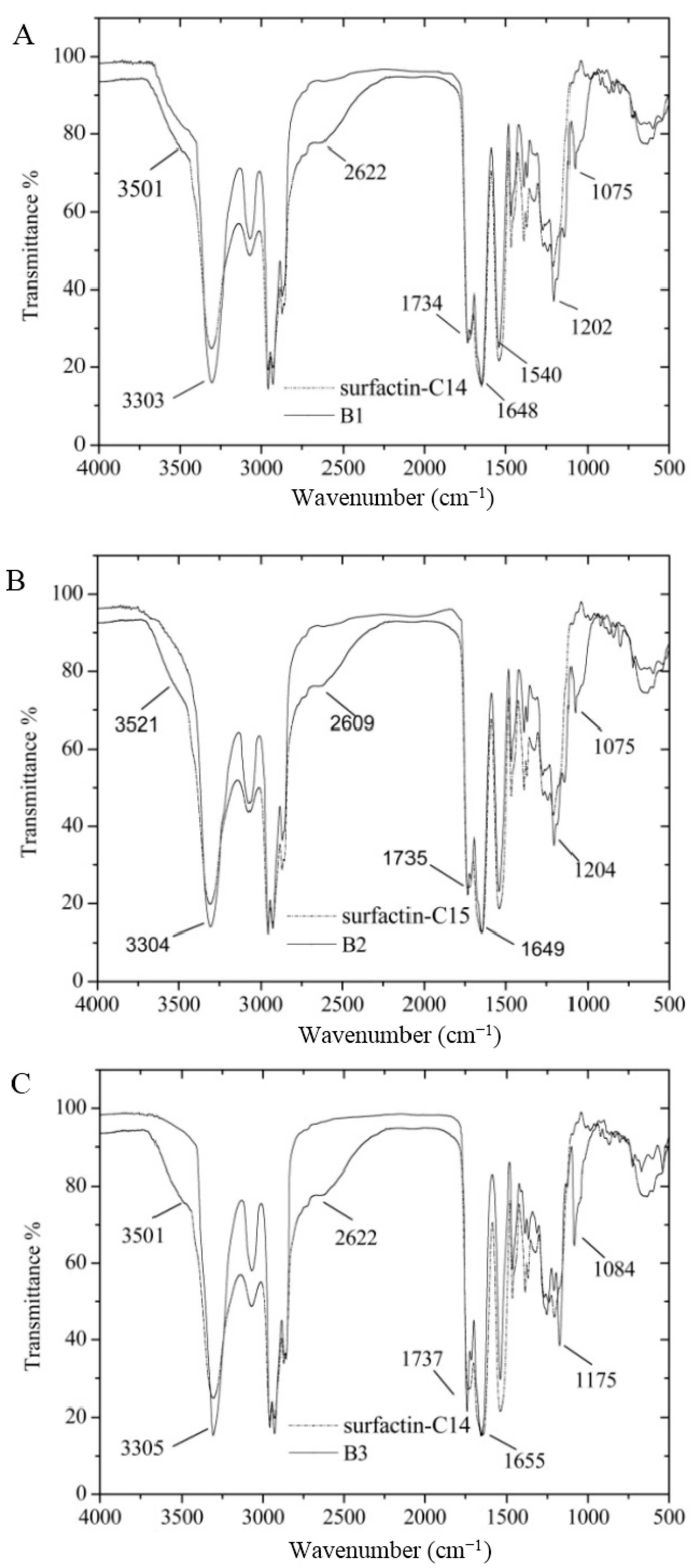
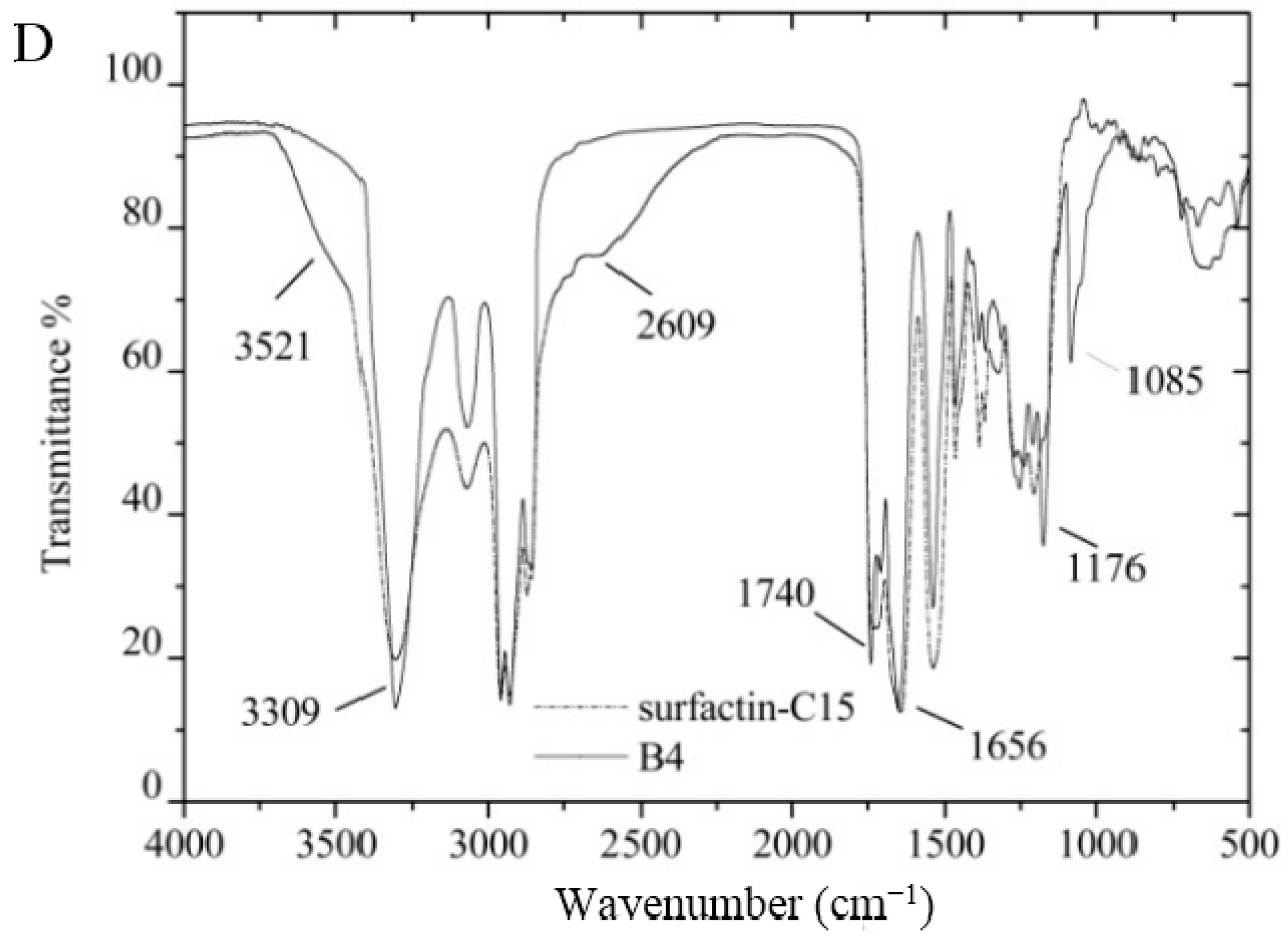
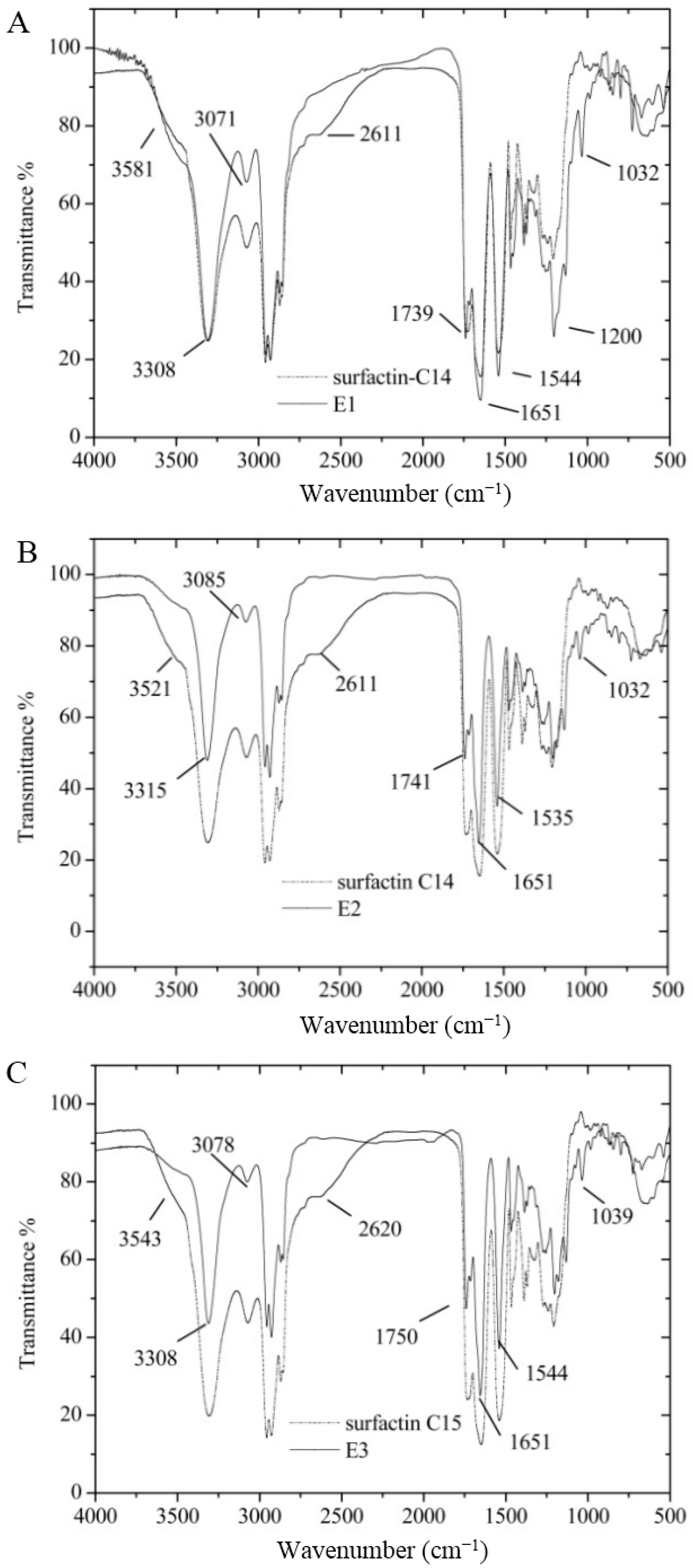
3.7. Certification of Characteristic Functional Groups
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Arima, K.; Kakinuma, A.; Tamura, G. Surfactin, a crystalline peptidelipid surfactant produced by Bacillus subtilis: Isolation, characterization and its inhibition of fibrin clot formation. Biochem. Biophys. Res. Commun. 1968, 31, 488–494. [Google Scholar] [CrossRef] [PubMed]
- Kakimuma, A.; Hori, M.; Isono, M.; Tamura, G.; Arima, K. Determination of fatty acid in surfactin and elucidation of the total structure of surfactin. Agric. Biol. Chem. 1969, 33, 971–972. [Google Scholar] [CrossRef]
- Kakimuma, A.; Ouchida, A.; Shima, T.; Sugino, H.; Isono, M.; Tamura, G.; Arima, K. Confirmation of the structure of surfactin by mass spectrometry. Agric. Biol. Chem. 1969, 33, 1669–1671. [Google Scholar] [CrossRef]
- Kakinuma, A.; Masatake, H.; Hiromu, S.; Isamu, Y.; Masao, I.; Gakuzo, T.; Kei, A. Determination of the location of lactone ring in surfactin. Agric. Biol. Chem. 1969, 33, 1523–1524. [Google Scholar] [CrossRef]
- Baumgart, F.; Kluge, B.; Ullrich, C.; Vater, J.; Ziessow, D. Identification of amino acid substitutions in the lipopeptide surfactin using 2D NMR spectroscopy. Biochem. Biophy. Res. Commun. 1991, 177, 998–1005. [Google Scholar] [CrossRef]
- Peypoux, F.; Bonmatin, J.M.; Labbe, H.; Grangemard, I.; Das, B.C.; Ptak, M.; Wallach, J.; Michel, G. [Ala4] surfactin, a novel isoform from Bacillus subtilis studied by mass and NMR spectroscopies. Eur. J. Biochem. 1994, 224, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Haddad, N.I.A.; Yang, S.; Mu, B. Structural characterization of eight cyclic lipopeptides produced by Bacillus subtilis HSO 121. Protein Pept. Lett. 2007, 14, 766–773. [Google Scholar] [CrossRef] [PubMed]
- Kanatomo, S.; Nagai, S.; Ohki, K.; Yasuda, Y. Study on surfactin, a cyclic depsipeptide. I: Isolation and structure of eight surfactin analogs produced by Bacillus natto KMD-2311. Yakugaku Zasshi 1995, 115, 756–764. [Google Scholar] [PubMed]
- Liu, X.; Yang, S.; Mu, B. Isolation and characterization of a C-12-lipopeptide produced by Bacillus subtilis HSO 121. J. Pept. Sci. 2008, 14, 864–875. [Google Scholar] [CrossRef] [PubMed]
- Gould, A.R.; May, B.K.; Elliott, W.H. Studies on the protoplast-bursting factor from Bacillus amyloliquefaciens. FEBS Lett. 1971, 14, 320–322. [Google Scholar] [CrossRef] [PubMed]
- Eeman, M.; Berquand, A.; Dufrene, Y.F.; Paquot, M.; Dufour, S.; Deleu, M. Penetration of surfactin into phospholipid monolayers: Nanoscale interfacial organization. Langmuir 2006, 22, 11337–11345. [Google Scholar] [CrossRef] [PubMed]
- Dufour, S.; Deleu, M.; Nott, K.; Wathelet, B.; Thonart, P.; Paquot, M. Hemolytic activity of new linear surfactin analogs in relation to their physico-chemical properties. Biochim. Biophys. Acta 2005, 1726, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Hosono, K.; Suzuki, H. Acylpeptides, the inhibitors of cyclic adenosine 3',5'-monophosphate phosphodiesterase. III: Inhibition of cyclic AMP phosphodiesterase. J. Antibiot. 1983, 36, 679–683. [Google Scholar] [CrossRef] [PubMed]
- Thimon, L.; Peypoux, F.; Das, B.C.; Wallach, J.; Michel, G. Selective esterification of surfactin: Preparation and properties of surfactin methyl esters. Biotechnol. Appl. Biochem. 1994, 20, 415–423. [Google Scholar]
- Ferré, G.; Besson, F.; Buchet, R. Conformational studies of the cyclic l,d-lipopeptide surfactin by fourier transform infrared spectroscopy. Spectrochim. Acta A Mol. Biomol. Spectrosc. 1997, 53, 623–635. [Google Scholar] [CrossRef]
- Morikawa, M.; Hirata, Y.; Imanaka, T. A study on the structure-function relationship of lipopeptide biosurfactants. Biochim. Biophys. Acta 2000, 1488, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Vass, E.; Besson, F.; Majer, Z.; Volpon, L.; Hollosi, M. Ca2+-induced changes of surfactin conformation: A FTIR and circular dichroism study. Biochem. Biophy. Res. Commun. 2001, 282, 361–367. [Google Scholar] [CrossRef]
- Liu, X.; Yang, S.; Mu, B. Production and characterization of a C-15-surfactin-O-methyl ester by a lipopeptide producing strain Bacillus subtilis HSO 121. Process Biochem. 2009, 44, 1144–1151. [Google Scholar] [CrossRef]
- Li, Y.; Yang, S.; Mu, B. Structural characterization of lipopeptide methyl esters produced by Bacillus licheniformis HSN 221. Chem. Biodivers. 2010, 7, 2065–2075. [Google Scholar] [CrossRef] [PubMed]
- Zhuravleva, O.I.; Afiyatullov, S.S.; Ermakova, S.P.; Nedashkovskaya, O.I.; Dmitrenok, P.S.; Denisenko, V.A.; Kuznetsova, T.A. New C-14-surfactin methyl ester from the marine bacterium Bacillus pumilus KMM 456. Russ. Chem. Bull. 2010, 59, 2137–2142. [Google Scholar] [CrossRef]
- Kracht, M.; Rokos, H.; Ozel, M.; Kowall, M.; Pauli, G.; Vater, J. Antiviral and hemolytic activities of surfactin isoforms and their methyl ester derivatives. J. Antibiot. 1999, 52, 613–619. [Google Scholar] [CrossRef] [PubMed]
- Nagai, S.; Okimura, K.; Kaizawa, N.; Ohki, K.; Kanatomo, S. Study on surfactin, a cyclic depsipeptide. II. Synthesis of surfactin B2 produced by Bacillus natto KMD 2311. Chem. Pharm. Bull. (Tokyo) 1996, 44, 5–10. [Google Scholar] [CrossRef]
- Pagadoy, M.; Peypoux, F.; Wallach, J. Solid-phase synthesis of surfactin, a powerful biosurfactant produced by Bacillus subtilis, and of four analogues. Int. J. Pept. Res. Ther. 2005, 11, 195–202. [Google Scholar] [CrossRef]
- Francius, G.; Dufour, S.; Deleu, M.; Papot, M.; Mingeot-Leclercq, M.P.; Dufrene, Y.F. Nanoscale membrane activity of surfactins: Influence of geometry, charge and hydrophobicity. Biochim. Biophys. Acta 2008, 1778, 2058–2068. [Google Scholar] [CrossRef]
- Razafindralambo, H.; Dufour, S.; Paquot, M.; Deleu, M. Thermodynamic studies of the binding interactions of surfactin analogues to lipid vesicles. J. Therm. Anal. Calorim. 2009, 95, 817–821. [Google Scholar] [CrossRef]
- Zhao, Y.; Yang, S.; Mu, B. Kinetic modeling of esterification reaction of surfactin-C15 in methanol solution. Appl. Biochem. Biotechnol. 2013, 169, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Neises, B.; Steglich, W. Simple method for the esterification of carboxylic acids. Angew. Chem. Int. Ed. 1978, 17, 522–524. [Google Scholar] [CrossRef]
- Sheehan, J.; Cruickshank, P.; Boshart, G. Notes—A convenient synthesis of water-soluble carbodiimides. J. Org. Chem. 1961, 26, 2525–2528. [Google Scholar] [CrossRef]
- Ostroumova, O.S.; Malev, V.V.; Ilin, M.G.; Schagina, L.V. Surfactin activity depends on the membrane dipole potential. Langmuir 2010, 26, 15092–15097. [Google Scholar] [CrossRef] [PubMed]
- Borders, D.B.; Francis, N.D.; Fantini, A.A. Extractive Purification of Lipopeptide Antibiotics. U.S. Patent 02/055537/A1, 18 July 2002. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shao, C.; Liu, L.; Gang, H.; Yang, S.; Mu, B. Structural Diversity of the Microbial Surfactin Derivatives from Selective Esterification Approach. Int. J. Mol. Sci. 2015, 16, 1855-1872. https://doi.org/10.3390/ijms16011855
Shao C, Liu L, Gang H, Yang S, Mu B. Structural Diversity of the Microbial Surfactin Derivatives from Selective Esterification Approach. International Journal of Molecular Sciences. 2015; 16(1):1855-1872. https://doi.org/10.3390/ijms16011855
Chicago/Turabian StyleShao, Chuanshi, Lin Liu, Hongze Gang, Shizhong Yang, and Bozhong Mu. 2015. "Structural Diversity of the Microbial Surfactin Derivatives from Selective Esterification Approach" International Journal of Molecular Sciences 16, no. 1: 1855-1872. https://doi.org/10.3390/ijms16011855
APA StyleShao, C., Liu, L., Gang, H., Yang, S., & Mu, B. (2015). Structural Diversity of the Microbial Surfactin Derivatives from Selective Esterification Approach. International Journal of Molecular Sciences, 16(1), 1855-1872. https://doi.org/10.3390/ijms16011855







