Liquid Phase Sintered Ceramic Bone Scaffolds by Combined Laser and Furnace
Abstract
:1. Introduction
2. Results and Discussions
2.1. Porous Scaffolds
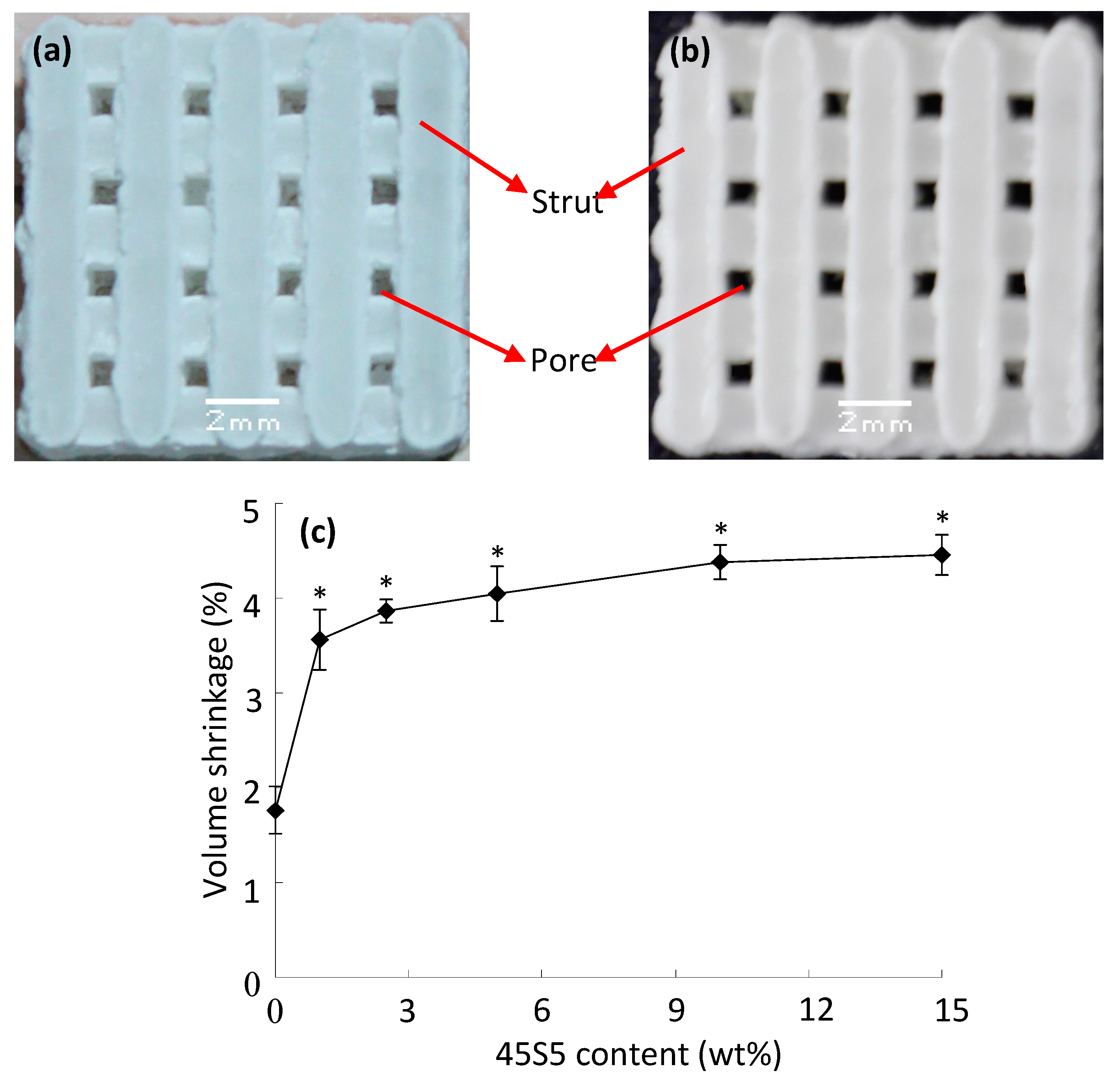
2.2. Mechanical Properties
 ) furnace sintering at 1100 °C. Statistical analysis showed that the mechanical properties of the scaffolds with 45S5 were significantly different from those without 45S5 (* p < 0.05, ** p < 0.01).
) furnace sintering at 1100 °C. Statistical analysis showed that the mechanical properties of the scaffolds with 45S5 were significantly different from those without 45S5 (* p < 0.05, ** p < 0.01).
 ) furnace sintering at 1100 °C. Statistical analysis showed that the mechanical properties of the scaffolds with 45S5 were significantly different from those without 45S5 (* p < 0.05, ** p < 0.01).
) furnace sintering at 1100 °C. Statistical analysis showed that the mechanical properties of the scaffolds with 45S5 were significantly different from those without 45S5 (* p < 0.05, ** p < 0.01).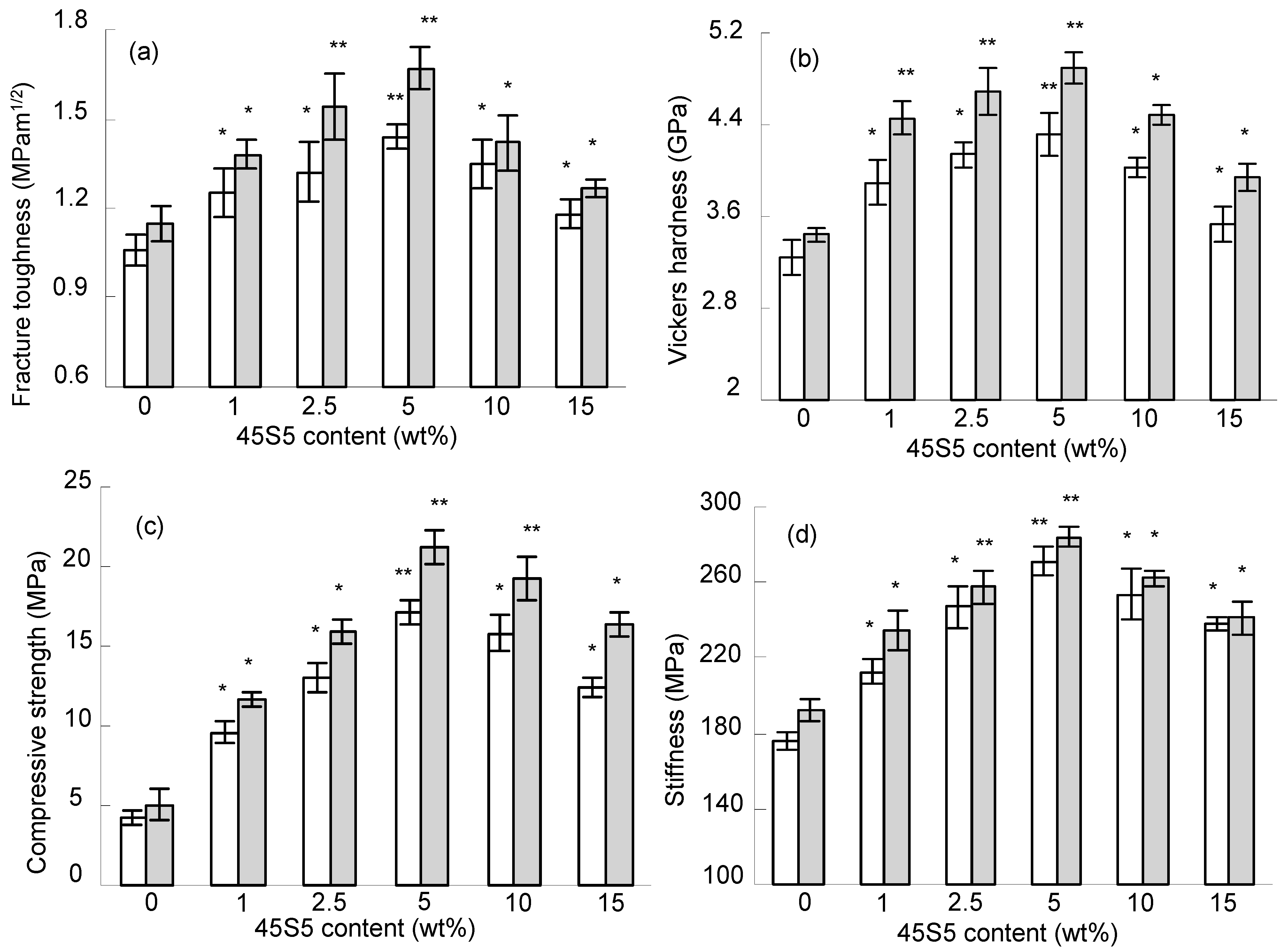
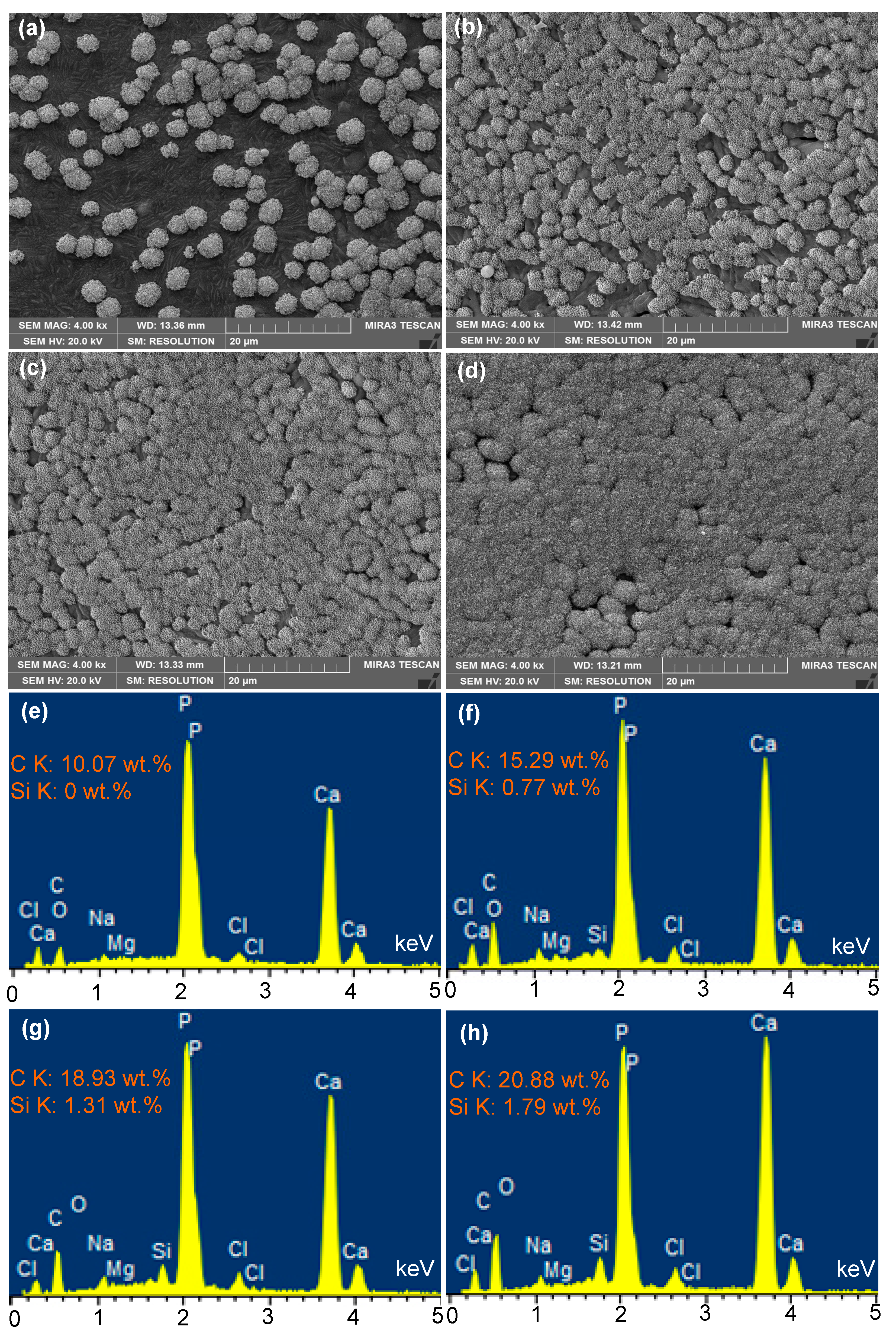
2.3. Microstructure
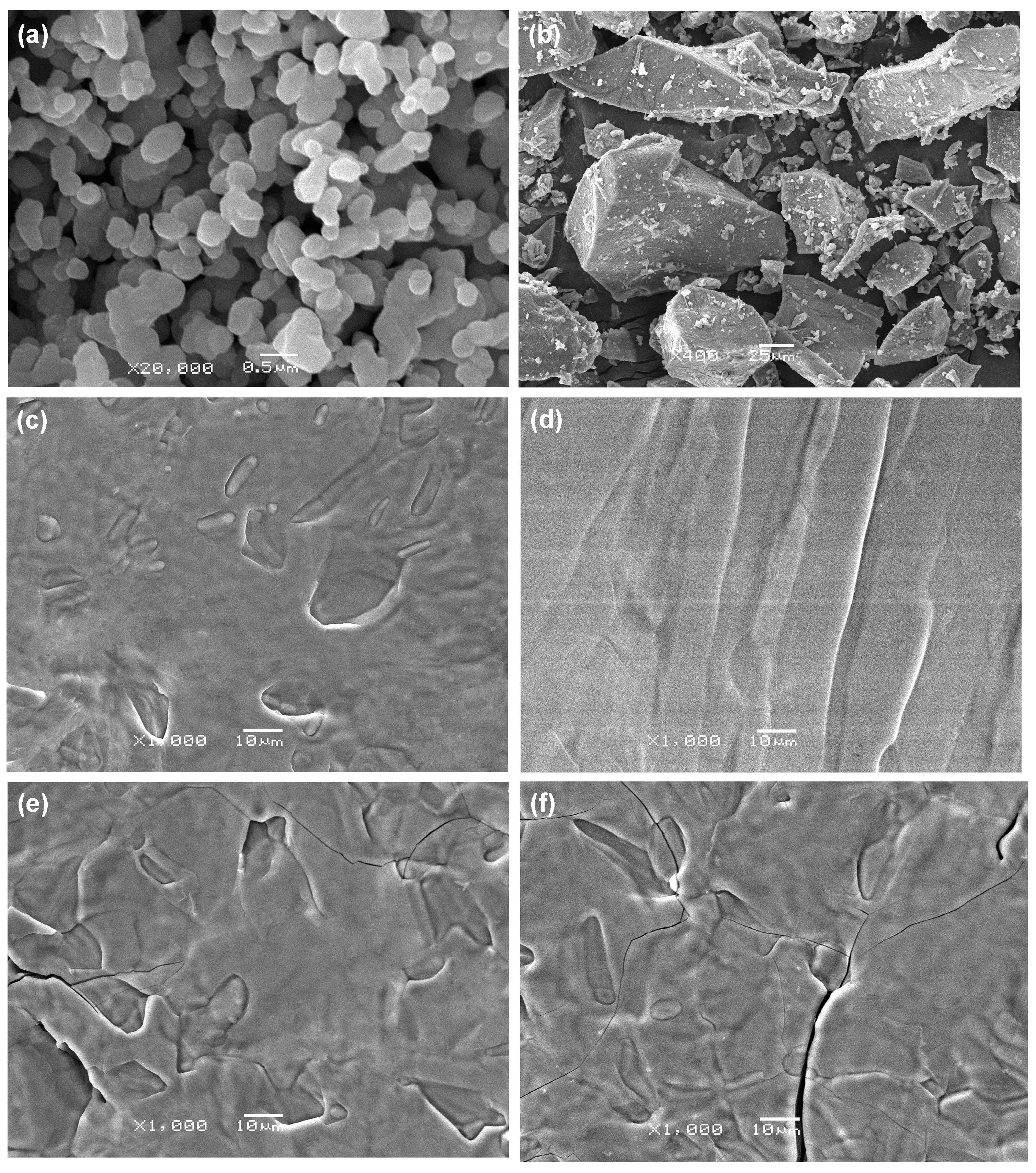

2.4. Phase Composition
 : CaSiO3 (JCPDF#43-1460).
: CaSiO3 (JCPDF#43-1460).
 : CaSiO3 (JCPDF#43-1460).
: CaSiO3 (JCPDF#43-1460).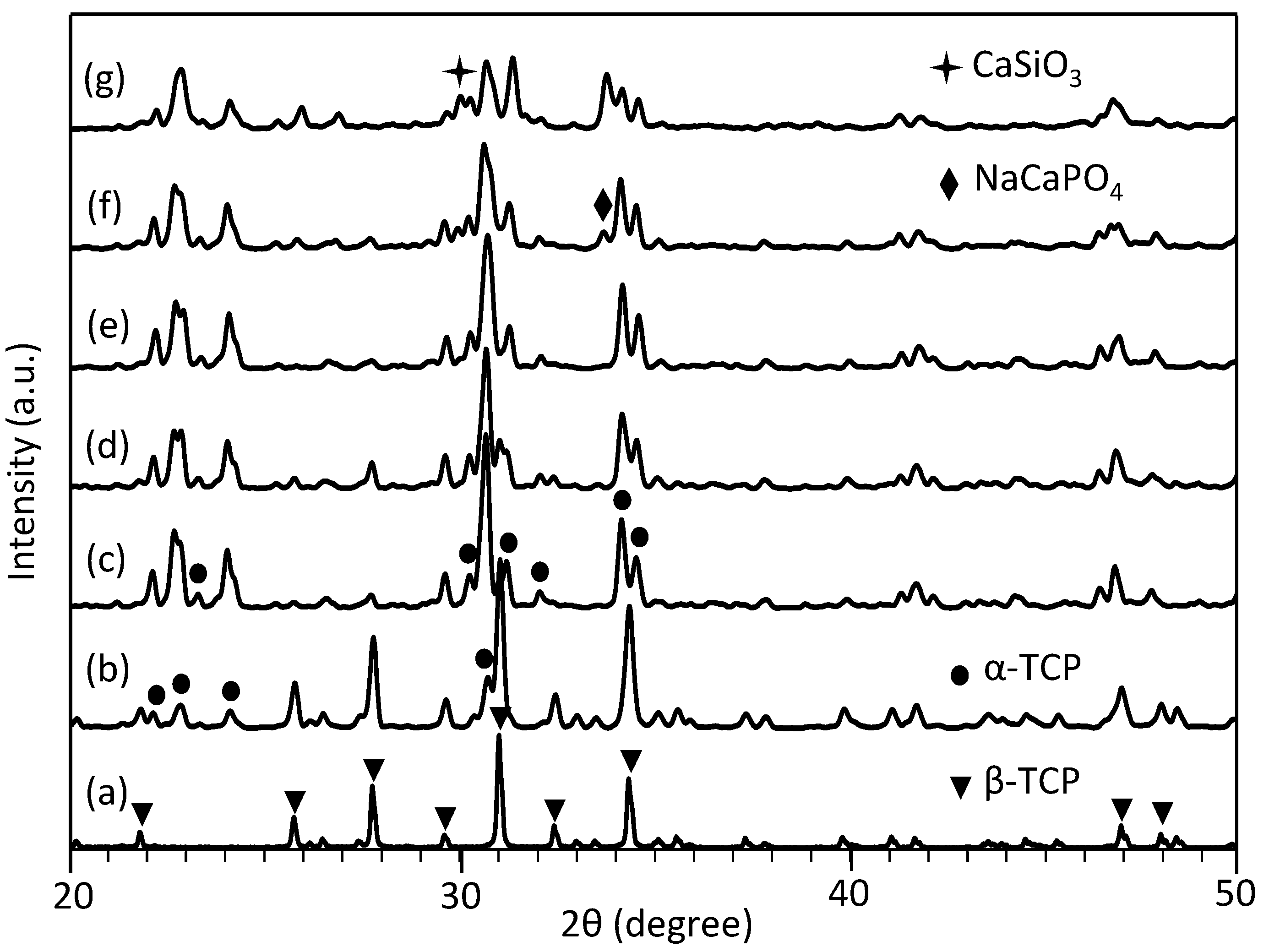
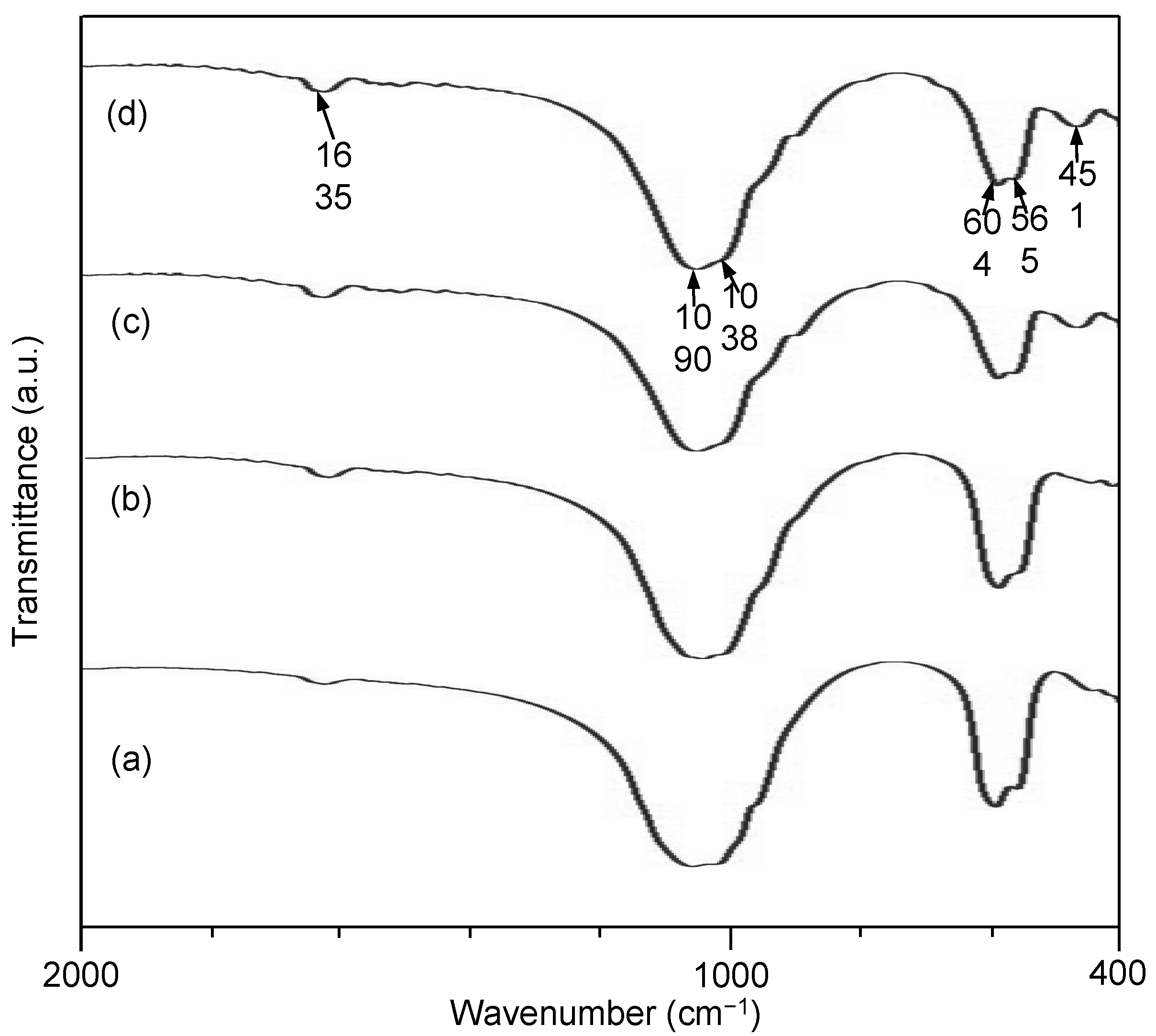
2.5. In Vitro Characterization

3. Experimental Section
3.1. Preparation of the Composite Powders
3.2. Fabrication and Characterization of β-Tricalcium phosphate (β-TCP) Scaffolds
3.3. Bioactivity in Simulated Body Fluid (SBF)
| Ion Species | SBF | HBP |
|---|---|---|
| Na+ | 142.0 | 142.0 |
| K+ | 5.0 | 5.0 |
| Mg2+ | 1.5 | 1.5 |
| Ca2+ | 2.5 | 2.5 |
| Cl− | 148.8 | 103.0 |
| HCO3− | 4.2 | 27.0 |
| HPO42− | 1.0 | 1.0 |
| SO42− | 0.5 | 0.5 |
3.4. Cell Attachment and Proliferation
3.5. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lu, Q.; Wang, X.; Lu, S.; Li, M.; Kaplan, D.L.; Zhu, H. Nanofibrous architecture of silk fibroin scaffolds prepared with a mild self-assembly process. Biomaterials 2011, 32, 1059–1067. [Google Scholar] [CrossRef]
- Salgado, C.L.; Sanchez, E.M.S.; Zavaglia, C.A.C.; Almeida, A.B.; Granja, P.L. Injectable biodegradable polycaprolactone-sebacic acid gels for bone tissue engineering. Tissue Eng. Part A 2011, 18, 137–146. [Google Scholar]
- Mijovic, B.; Trcin, M.T.; Agic, A.; Zdraveva, E.; Bujic, M.; Spoljaric, I.; Kosec, V. Study on cell adhesion detection onto biodegradable electrospun PCL scaffolds. JFBI 2012, 5, 33–40. [Google Scholar] [CrossRef]
- Li, J.; Liu, W.; Yin, A.; Wu, J.; al-Deyab, S.S.; el-Newehy, M.; Mo, X. Nano-yarns reinforced silk fibroin composites scaffold for bone tissue engineering. JFBI 2012, 5, 169–179. [Google Scholar] [CrossRef]
- Bian, S.; Lu, W.; Xu, C.; Fan, Y.; Zhang, X. In vitro cartilage tissue engineering using porous collagen/PLLA nanoparticle hybrid scaffold. J. Med. Biol. Eng. 2014, 34, 36–43. [Google Scholar] [CrossRef]
- Mijovic, B.; Agic, A. The effect of processing and rheological variables on the morphology of dermal electrospun scaffold. JFBI 2011, 3, 181–186. [Google Scholar] [CrossRef]
- Liu, D.; Zhuang, J.; Shuai, C.; Peng, S. Mechanical properties’ improvement of a tricalcium phosphate scaffold with poly-l-lactic acid in selective laser sintering. Biofabrication 2013, 5. [Google Scholar] [CrossRef]
- Liu, S.; Jin, F.; Lin, K.; Lu, J.; Sun, J.; Chang, J.; Dai, K.; Fan, C. The effect of calcium silicate on in vitro physiochemical properties and in vivo osteogenesis, degradability and bioactivity of porous β-tricalcium phosphate bioceramics. Biomed. Mater. 2013, 8. [Google Scholar] [CrossRef]
- Feng, P.; Wei, P.; Shuai, C.; Peng, S. Characterization of Mechanical and Biological Properties of 3-D Scaffolds Reinforced with Zinc Oxide for Bone Tissue Engineering. PLoS One 2014, 9, e87755. [Google Scholar] [CrossRef]
- Zhang, Y.; Kong, D.; Feng, X. Fabrication and properties of porous β-tricalcium phosphate ceramics prepared using a double slip-casting method using slips with different viscosities. Ceram. Int. 2012, 38, 2991–2996. [Google Scholar] [CrossRef]
- Gao, C.; Deng, Y.; Feng, P.; Mao, Z.; Li, P.; Yang, B.; Deng, J.; Cao, Y.; Shuai, C.; Peng, S. Current progress in bioactive ceramic scaffolds for bone repair and regeneration. Int. J. Mol. Sci. 2014, 15, 4714–4732. [Google Scholar] [CrossRef]
- Ma, J.; Ye, F.; Cao, Y.; Liu, C.F.; Zhang, H.J. Microstructure and mechanical properties of liquid phase sintered silicon carbide composites. J. Zhejiang Univ. Sci. A 2010, 11, 766–770. [Google Scholar] [CrossRef]
- Desimone, D.; Li, W.; Roether, J.A.; Schubert, D.W.; Crovace, M.C.; Rodrigues, A.C.M.; Zanotto, E.D.; Boccaccini, A.R. Biosilicate®-gelatine bone scaffolds by the foam replica technique: Development and characterization. Sci. Technol. Adv. Mater. 2013, 14. [Google Scholar] [CrossRef]
- Hotta, M.; Hojo, J. Inhibition of grain growth in liquid-phase sintered SiC ceramics by AlN additive and spark plasma sintering. J. Eur. Ceram. Soc. 2010, 30, 2117–2122. [Google Scholar] [CrossRef]
- Kalita, S.J.; Bose, S.; Hosick, H.L.; Bandyopadhyay, A. CaO-P2O5-Na2O-based sintering additives for hydroxyapatite (HAp) ceramics. Biomaterials 2004, 25, 2331–2339. [Google Scholar] [CrossRef]
- Hesaraki, S.; Safari, M.; Shokrgozar, M.A. Development of β-tricalcium phosphate/sol-gel derived bioactive glass composites: Physical, mechanical, and in vitro biological evaluations. J. Biomed. Mater. Res. B 2009, 91, 459–469. [Google Scholar] [CrossRef]
- Jung, S.B.; Day, D.E. Conversion kinetics of silicate, borosilicate, and borate bioactive glasses to hydroxyapatite. Phys. Chem. Glasses B 2009, 50, 85–88. [Google Scholar]
- Deng, M.; Wen, H.L.; Dong, X.L.; Li, F.; Xu, X.; Li, H.; Zhou, X.D. Effects of 45S5 bioglass on surface properties of dental enamel subjected to 35% hydrogen peroxide. Int. J. Oral Sci. 2013, 5, 103–110. [Google Scholar] [CrossRef]
- Rahaman, M.N.; Day, D.E.; Sonny, B.B.; Fu, Q.; Jung, S.B.; Bonewald, L.F.; Tomsia, A.P. Bioactive glass in tissue engineering. Acta Biomater. 2011, 7, 2355–2373. [Google Scholar] [CrossRef]
- Bahniuk, M.S.; Pirayesh, H.; Singh, H.D.; Nychka, J.A.; Unsworth, L.D. Bioactive glass 45S5 powders: Effect of synthesis route and resultant surface chemistry and crystallinity on protein adsorption from human plasma. Biointerphases 2012, 7, 1–15. [Google Scholar]
- Plewinski, M.; Schickle, K.; Lindner, M.; Kirsten, A.; Weber, M.; Fischer, H. The effect of crystallization of bioactive bioglass 45S5 on apatite formation and degradation. Dent. Mater. 2013, 29, 1256–1264. [Google Scholar] [CrossRef]
- Ryu, H.S.; Youn, H.J.; Hong, K.S.; Chang, B.S.; Lee, C.K.; Chung, S.S. An improvement in sintering property of β-tricalcium phosphate by addition of calcium pyrophosphate. Biomaterials 2002, 23, 909–914. [Google Scholar]
- Kwon, S.H.; Jun, Y.K.; Hong, S.H.; Kim, H.E. Synthesis and dissolution behavior of β-TCP and HA/β-TCP composite powders. J. Eur. Ceram. Soc. 2003, 23, 1039–1045. [Google Scholar] [CrossRef]
- Liu, W.; Ma, Y.; Cai, Q. Consolidation of MA W-Ni-Fe alloyed powder by microwave-assisted sintering. Mater. Sci. Appl. 2011, 2, 609–614. [Google Scholar]
- Miranzo, P.; González-Julián, J.; Osendi, M.I.; Belmonte, M. Enhanced particle rearrangement during liquid phase spark plasma sintering of silicon nitride-based ceramics. Ceram. Int. 2011, 37, 159–166. [Google Scholar] [CrossRef]
- Illingworth, T.C.; Golosnoy, I.O.; Gergely, V.; Clyne, T.W. Numerical modelling of transient liquid phase bonding and other diffusion controlled phase changes. J. Mater. Sci. 2005, 40, 2505–2511. [Google Scholar] [CrossRef]
- Amorim, F.L.; Lohrengel, A.; Schaefer, G.; Czelusniak, T. A study on the SLS manufacturing and experimenting of TiB2-CuNi EDM electrodes. Rapid. Prototyp. J. 2013, 19, 418–429. [Google Scholar] [CrossRef]
- Tilocca, A. Structural models of bioactive glasses from molecular dynamics simulations. Proc. R. Soc. AMath. Phys. 2009, 465, 1003–1027. [Google Scholar] [CrossRef]
- Jun, I.K.; Song, J.H.; Choi, W.Y.; Koh, Y.H.; Kim, H.E.; Kim, H.W. Porous hydroxyapatite scaffolds coated with bioactive apatite-wollastonite glass-ceramics. J. Am. Ceram. Soc. 2007, 90, 2703–2708. [Google Scholar] [CrossRef]
- Shahzad, K.; Deckers, J.; Boury, S.; Neirinck, B.; Kruth, J.; Vleugels, J. Preparation and indirect selective laser sintering of alumina/PA microspheres. Ceram. Int. 2012, 38, 1241–1247. [Google Scholar] [CrossRef]
- Dewidar, M.M.; Lim, J.; Dalgarno, K.W. A Comparison between direct and indirect laser sintering of metals. J. Mater. Sci. Technol. 2008, 24, 227–232. [Google Scholar]
- Murphy, C.M.; O’Brien, F.J. Understanding the effect of mean pore size on cell activity in collagen-glycosaminoglycan scaffolds. Cell. Adhes. Migr. 2010, 4, 377–381. [Google Scholar] [CrossRef]
- Fedorovich, N.E.; Alblas, J.; Hennink, W.E.; Öner, F.C.; Dhert, W.J. Organ printing: The future of bone regeneration? Trends Biotechnol. 2011, 29, 601–606. [Google Scholar] [CrossRef]
- Huang, S.; Huang, B.; Zhou, K.; Li, Z. Effects of coatings on the mechanical properties of carbon fiber reinforced HAP composites. Mater. Lett. 2004, 58, 3582–3585. [Google Scholar] [CrossRef]
- Guillon, O.; Cao, S.; Chang, J.; Wondraczek, L.; Boccaccini, A.R. Effect of uniaxial load on the sintering behaviour of 45S5 bioglass® powder compacts. J. Eur. Ceram. Soc. 2011, 31, 999–1007. [Google Scholar] [CrossRef]
- Abramović, B.; Šojić, D.; Anderluh, V. Visible-light-induced photocatalytic degradation of herbicide mecoprop in aqueous suspension of TiO2. Acta Chim. Slov. 2007, 54, 558–564. [Google Scholar]
- Soliman, M.H.; Gado, H.S.; Kouraim, M.N. Chemical studies on the removal of iron from crude phosphoric acid using an organosilicon compound. J. Chem. 2009, 6, S329–S341. [Google Scholar]
- Al-Muslet, N.A.; Ali, E.E. In vivo spectral analysis of bladder cancer using fourier transform infrared spectroscopy, a comparative study. Aust. J. Basic Appl. 2011, 5, 1734–1739. [Google Scholar]
- Neelgund, G.M.; Olurode, K.; Luo, Z.; Oki, A. A simple and rapid method to graft hydroxyapatite on carbon nanotubes. Mater. Sci. Eng. C 2011, 31, 1477–1481. [Google Scholar] [CrossRef]
- Qi, C.; Zhu, Y.; Zhao, X.; Zhao, J.; Chen, F.; Cheng, G.; Ruan, Y.J. High surface area carbonate apatite nanorod bundles: Surfactant-free sonochemical synthesis and drug loading and release properties. Mater. Res. Bull. 2013, 48, 1536–1540. [Google Scholar] [CrossRef]
- Yao, Q.; Nooeaid, P.; Roether, J.A.; Dong, Y.; Zhang, Q.; Boccaccini, A.R. Bioglass®-based scaffolds incorporating polycaprolactone and chitosan coatings for controlled vancomycin delivery. Ceram. Int. 2013, 39, 7517–7522. [Google Scholar] [CrossRef]
- Ahangaran, F.; Hassanzadeh, A.; Nouri, S. Surface modification of Fe3O4@SiO2 microsphere by silane coupling agent. Int. Nano Lett. 2013, 3, 1–5. [Google Scholar] [CrossRef]
- Vallet-Regí, M.; Salinas, A.J.; Ramírez-Castellanos, J.; González-Calbet, J.M. Nanostructure of bioactive sol-gel glasses and organic-inorganic hybrids. Chem. Mater. 2005, 17, 1874–1879. [Google Scholar] [CrossRef]
- Lefebvre, L.; Chevalier, J.; Gremillard, L.; Zenati, R.; Thollet, G.; Bernache-Assolant, D.; Govin, A. Structural transformations of bioactive glass 45S5 with thermal treatments. Acta Mater. 2007, 55, 3305–3313. [Google Scholar] [CrossRef]
- Qian, J.; Kang, Y.; Wei, Z.; Zhang, W. Fabrication and characterization of biomorphic 45S5 bioglass scaffold from sugarcane. Mater. Sci. Eng. C 2009, 29, 1361–1364. [Google Scholar] [CrossRef]
- Ohtsuki, C.; Kamitakahara, M.; Miyazaki, T. Bioactive ceramic-based materials with designed reactivity for bone tissue regeneration. J. R. Soc. Interface 2009, 6, S349–S360. [Google Scholar] [CrossRef]
- Alves, N.M.; Leonor, I.B.; Azevedo, H.S.; Reis, R.L.; Mano, J.F. Designing biomaterials based on biomineralization of bone. J. Mater. Chem. 2010, 20, 2911–2921. [Google Scholar] [CrossRef]
- Zhang, X.; Han, P.; Jaiprakash, A.; Wu, C.; Xiao, Y. Stimulatory effect of Ca3ZrSi2O9 bioceramics on cementogenic/osteogenic differentiation of periodontal ligament cells. J. Mater. Chem. B 2014, 2, 1415–1423. [Google Scholar] [CrossRef]
- Oryan, A.; Alidadi, S.; Moshiri, A.; Maffulli, N. Bone regenerative medicine: Classic options, novel strategies, and future directions. J. Orthop. Surg. Res. 2014, 9. [Google Scholar] [CrossRef]
- Shuai, C.; Feng, P.; Zhang, L.; Gao, C.; Hu, H.; Peng, S.; Min, A. Correlation between properties and microstructure of laser sintered porous β-tricalcium phosphate bone scaffolds. Sci. Technol. Adv. Mater. 2013, 14. [Google Scholar] [CrossRef]
- Bellucci, D.; Sola, A.; Gazzarri, M.; Chiellini, F.; Cannillo, V. A new hydroxyapatite-based biocomposite for bone replacement. Mater. Sci. Eng. C 2013, 33, 1091–1101. [Google Scholar]
- Evans, A.G.; Charles, E.A. Fracture toughness determination by indentation. J. Am. Ceram. Soc. 1976, 59, 371–372. [Google Scholar] [CrossRef]
- Kokubo, T.; Ito, S.; Huang, Z.T.; Hayashi, T.; Sakka, S.; Kitsugi, T.; Yamamuro, T. Ca, P-rich layer formed on high-strength bioactive glass-ceramic A-W. J. Biomed. Mater. Res. 1990, 24, 331–343. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Feng, P.; Deng, Y.; Duan, S.; Gao, C.; Shuai, C.; Peng, S. Liquid Phase Sintered Ceramic Bone Scaffolds by Combined Laser and Furnace. Int. J. Mol. Sci. 2014, 15, 14574-14590. https://doi.org/10.3390/ijms150814574
Feng P, Deng Y, Duan S, Gao C, Shuai C, Peng S. Liquid Phase Sintered Ceramic Bone Scaffolds by Combined Laser and Furnace. International Journal of Molecular Sciences. 2014; 15(8):14574-14590. https://doi.org/10.3390/ijms150814574
Chicago/Turabian StyleFeng, Pei, Youwen Deng, Songlin Duan, Chengde Gao, Cijun Shuai, and Shuping Peng. 2014. "Liquid Phase Sintered Ceramic Bone Scaffolds by Combined Laser and Furnace" International Journal of Molecular Sciences 15, no. 8: 14574-14590. https://doi.org/10.3390/ijms150814574
APA StyleFeng, P., Deng, Y., Duan, S., Gao, C., Shuai, C., & Peng, S. (2014). Liquid Phase Sintered Ceramic Bone Scaffolds by Combined Laser and Furnace. International Journal of Molecular Sciences, 15(8), 14574-14590. https://doi.org/10.3390/ijms150814574






