Artesunate Induces Apoptosis of Bladder Cancer Cells by miR-16 Regulation of COX-2 Expression
Abstract
:1. Introduction
2. Results and Discussion
2.1. Results
2.1.1. Artesunate (ART) Inhibited Tumor Growth in the Bladder Cancer Rat
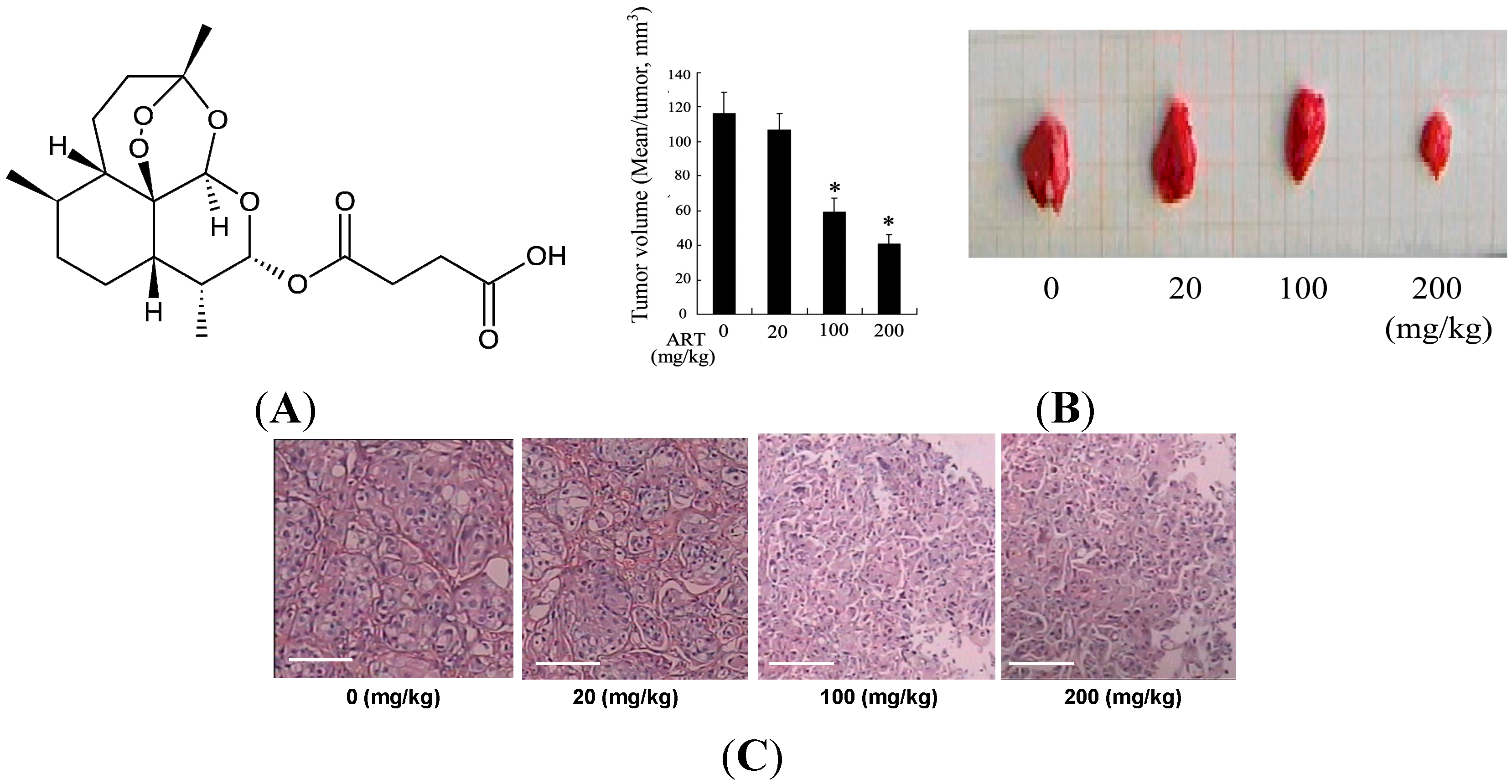
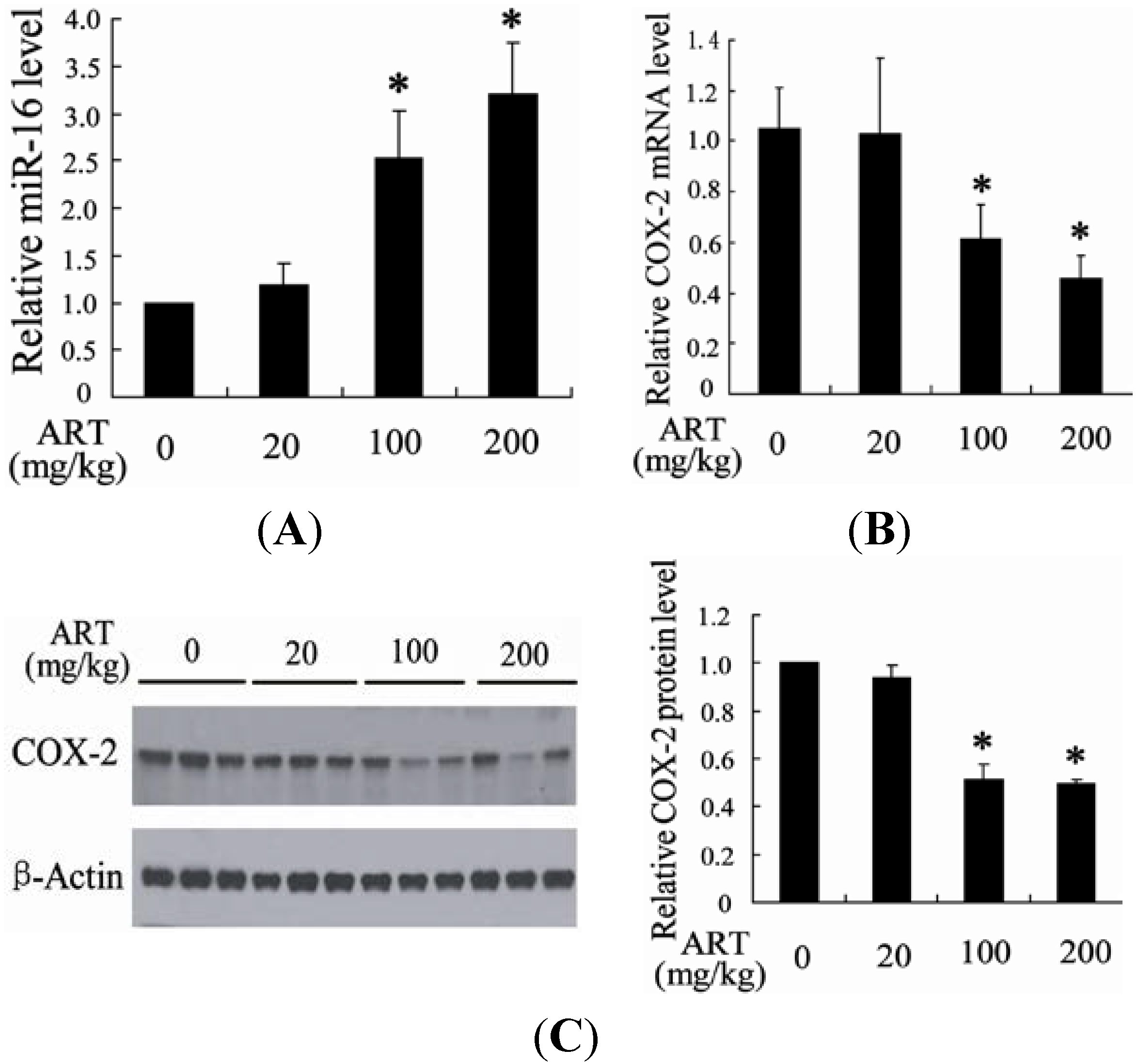
2.1.2. ART Significantly Increased miR-16 and Decreased Cyclooxygenase-2 (COX-2) Expression in Tumors
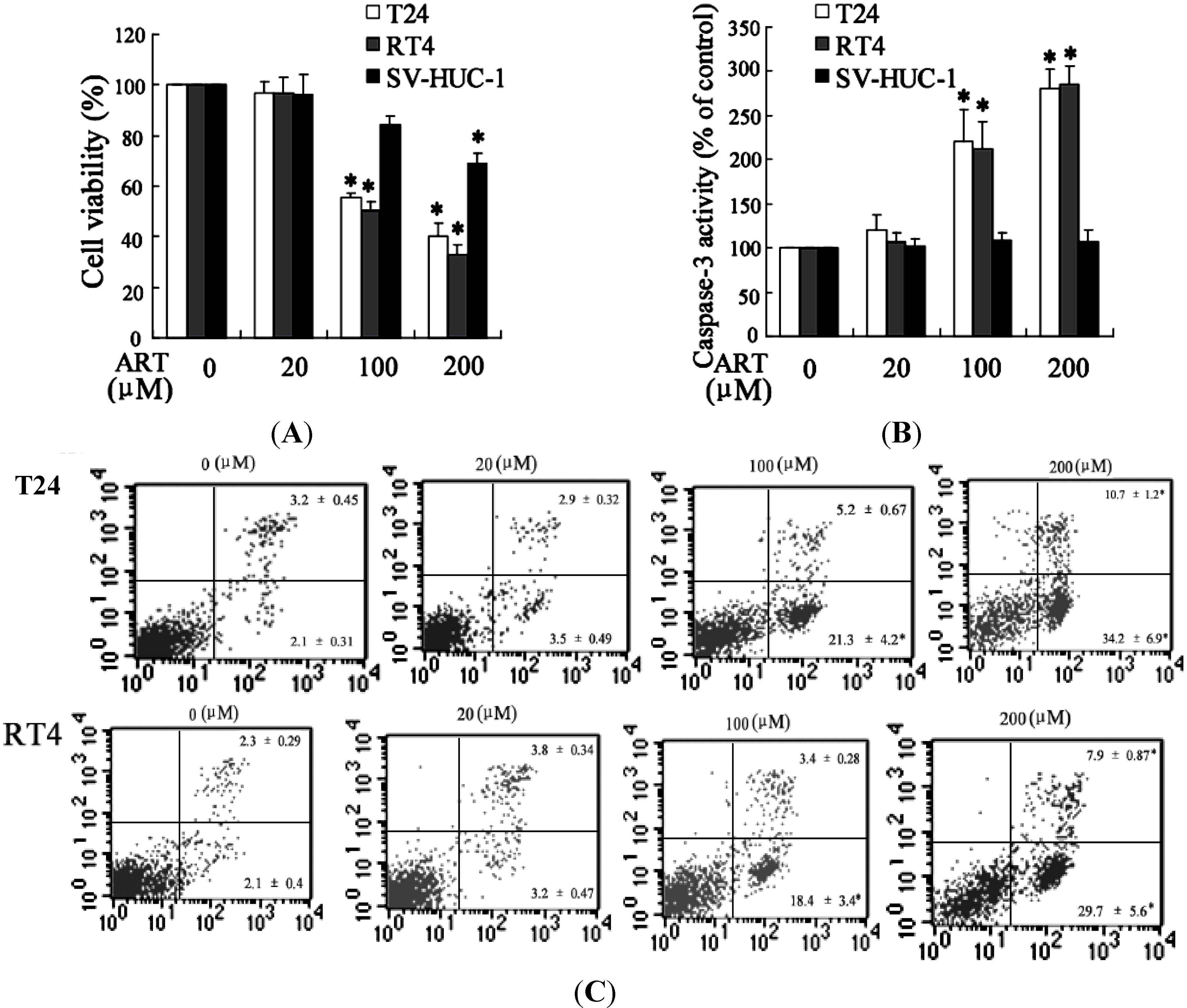
2.1.3. ART Induced Cytotoxicity and Apoptosis in Bladder Cancer Cells
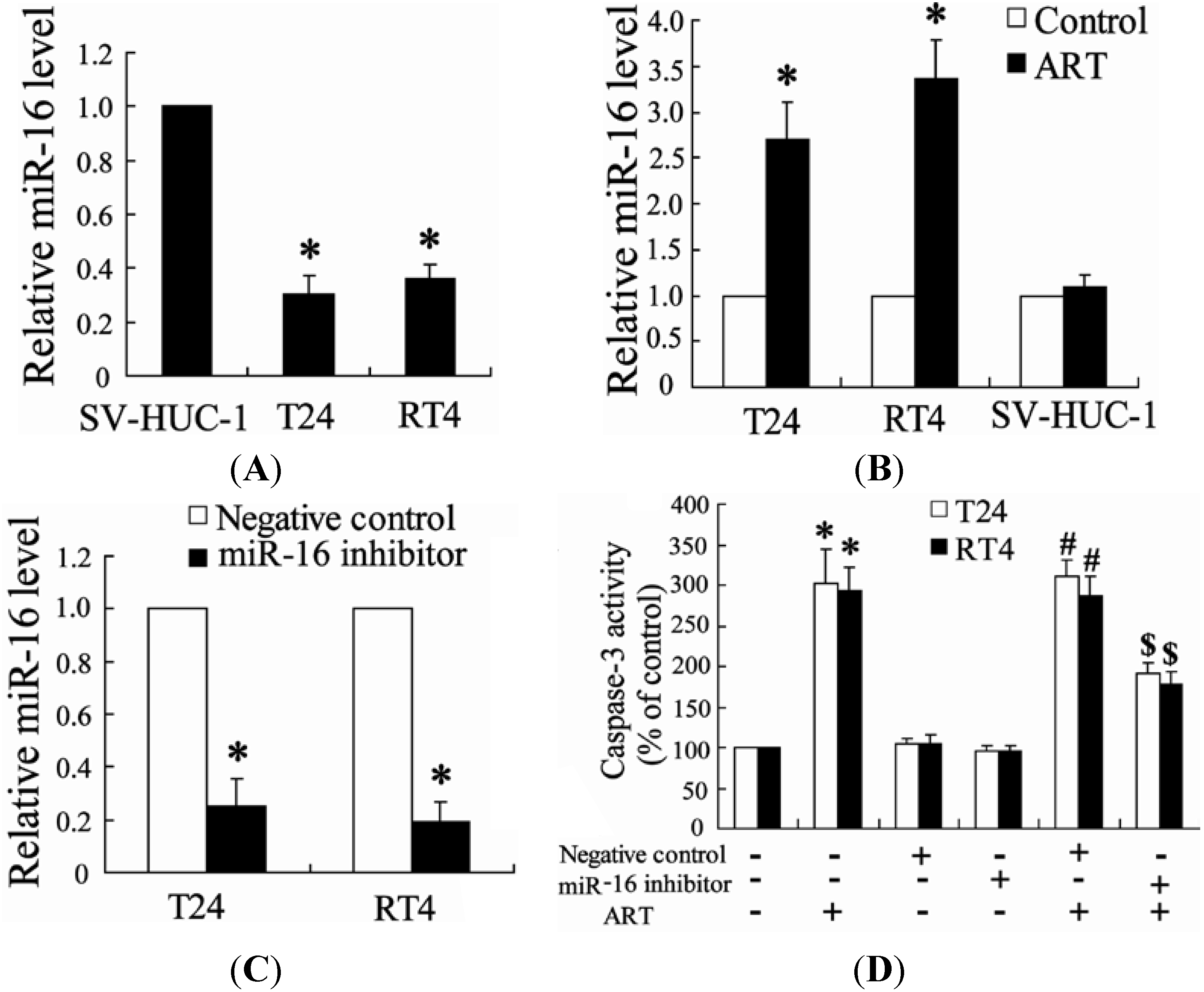
2.1.4. Down-Regulation of miR-16 Can Reverse the Effect of ART on Apoptosis of Bladder Cancer Cells
2.1.5. ART Decreases COX-2 Expression and Prostaglandin E2 (PGE2) Production in Bladder Cancer Cells
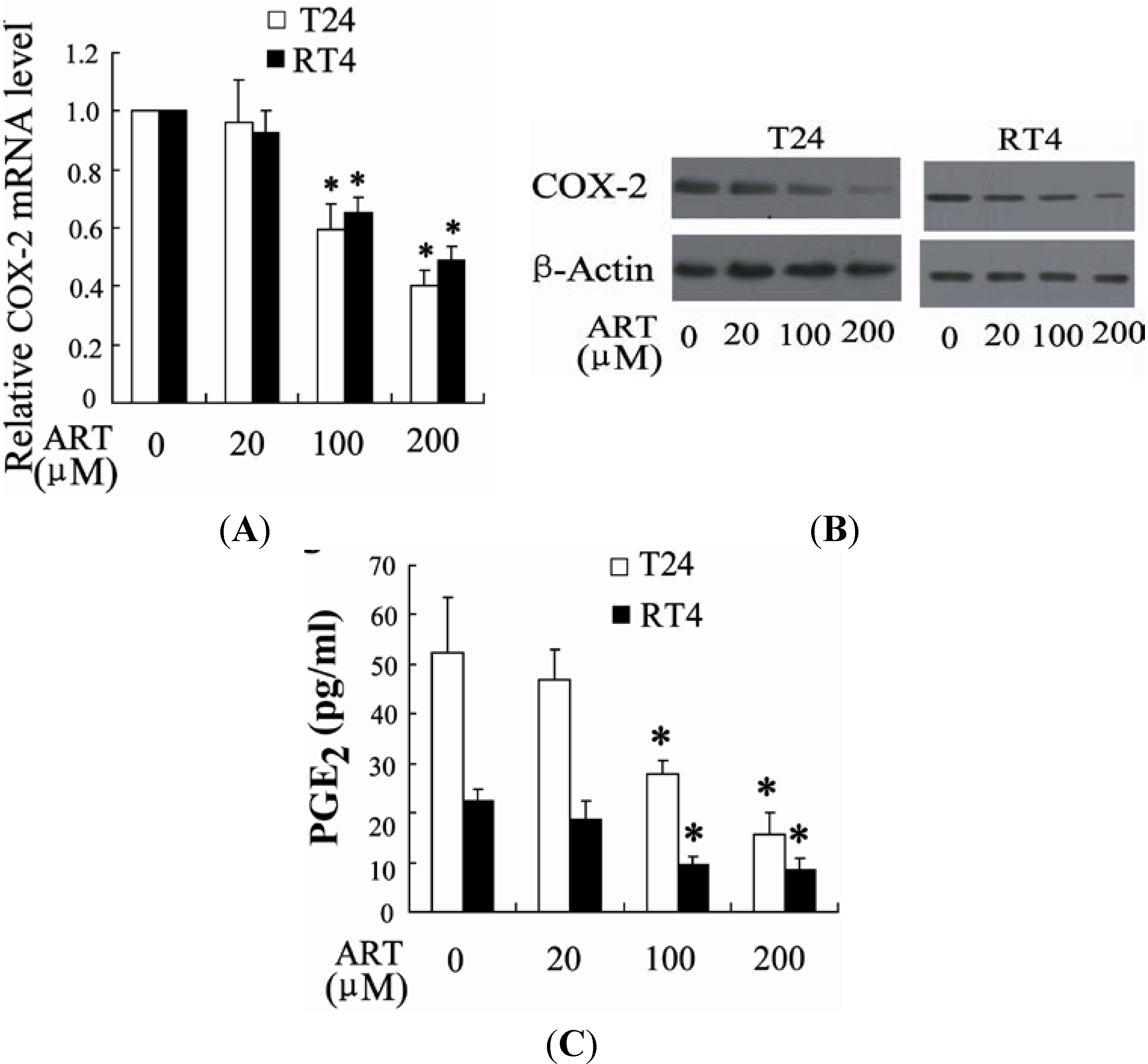
2.1.6. miR-16 Is Involved in ART Regulation of COX-2 Expression in Bladder Cancer Cells
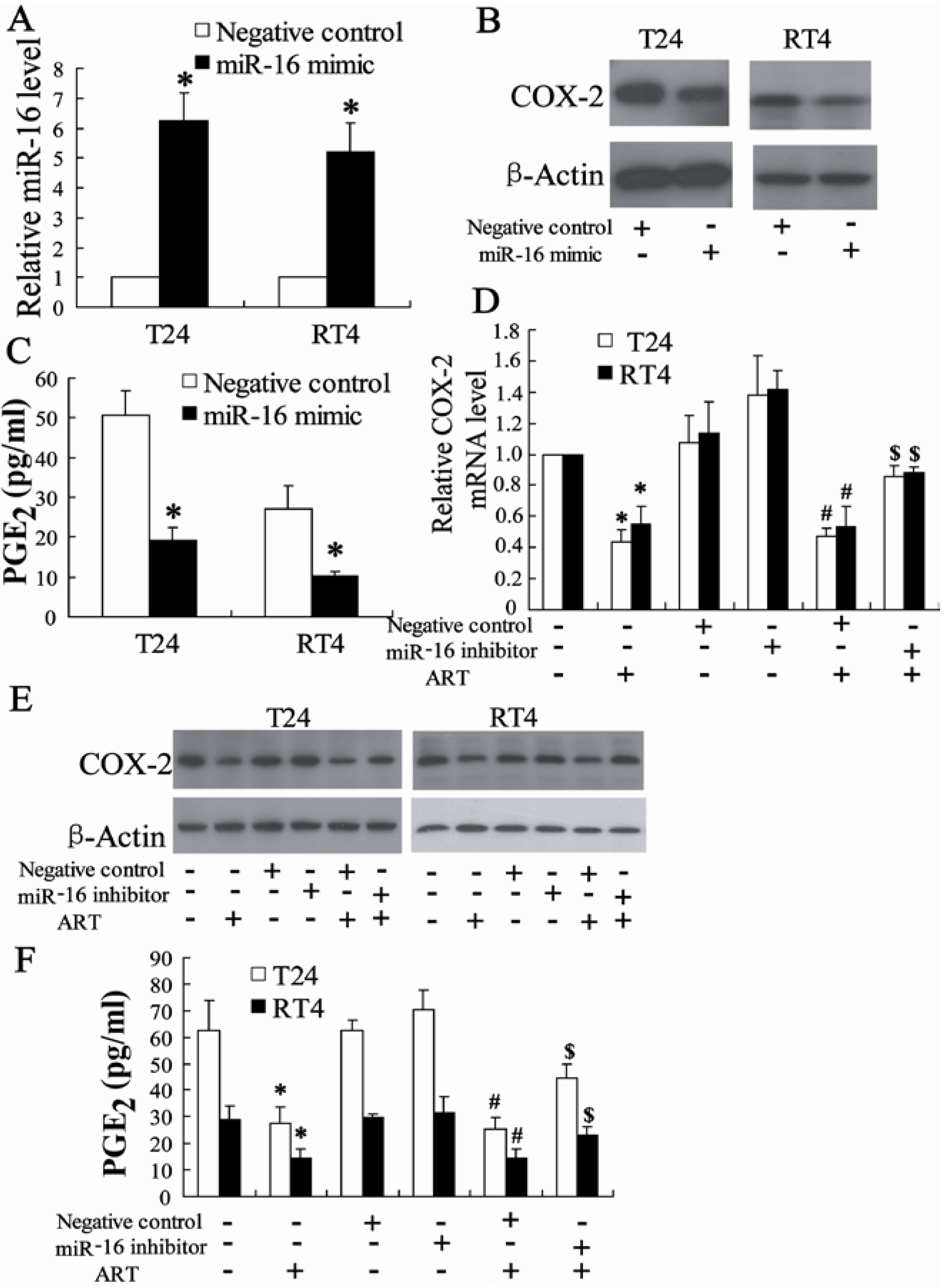
2.1.7. Exogenous PGE2 Can Reverse the Effect of ART on Inhibition of Bladder Cancer Cell Growth
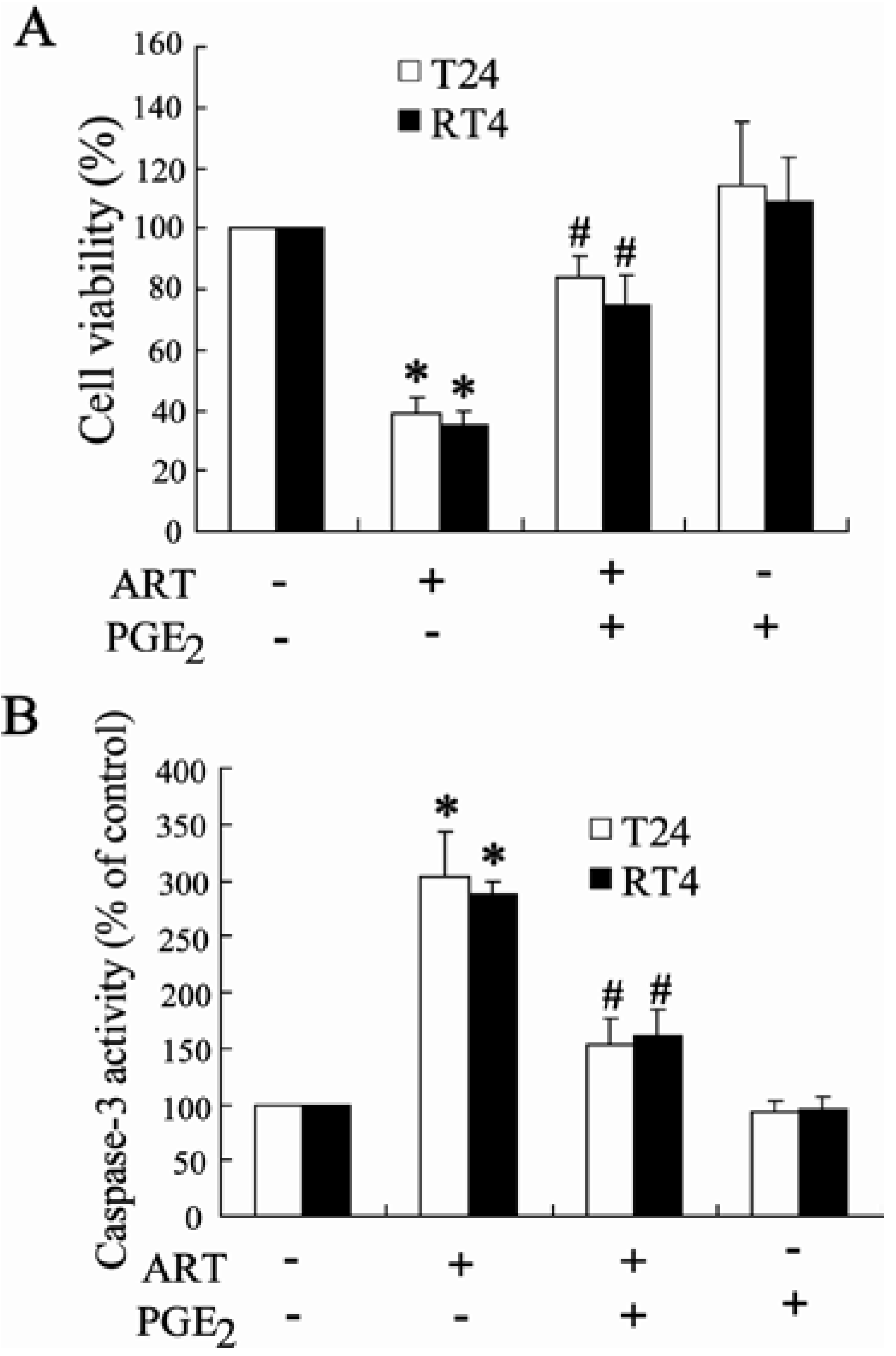
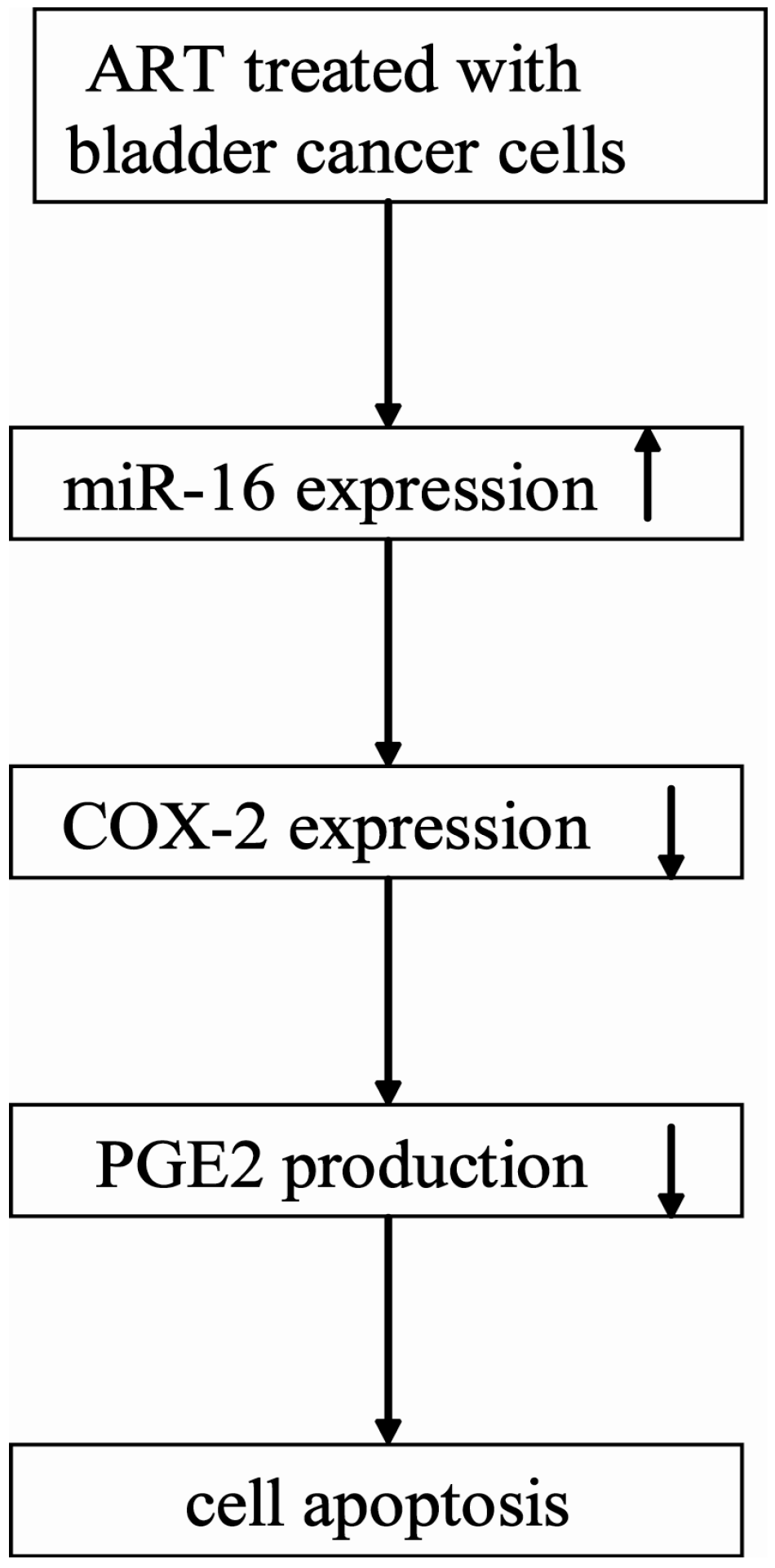
2.2. Discussion
3. Experimental Section
3.1. Chemicals and Reagents
3.2. Cell Culture
3.3. Animals
3.4. Dimethylthiazoly-2,5-diphenyltetrazolium Bromide (MTT) Assay
3.5. Flow Cytometry Analysis
3.6. Caspase-3 Activity Assays
3.7. ELISA
3.8. Real-Time PCR
3.9. Western Blot Analysis
3.10. Transfection Procedures
3.11. Statistical Analysis
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Kirkali, Z.; Chan, T.; Manoharan, M.; Algaba, F.; Busch, C.; Cheng, L.; Kiemeney, L.; Kriegmair, M.; Montironi, R.; Murphy, W.M.; et al. Bladder cancer: Epidemiology, staging and grading, and diagnosis. Urology 2005, 66, 4–34. [Google Scholar]
- Bellmunt, J.; Guix, M. New agents for bladder cancer. Ann. Oncol. 2010, 21, 56–58. [Google Scholar]
- Busby, J.E.; Kamat, A.M. Chemoprevention for bladder cancer. Cancer 2006, 176, 1914–1920. [Google Scholar]
- Bartel, D.P. MicroRNAs: Genomics, biogenesis, mechanism, and function. Cell 2004, 116, 281–297. [Google Scholar] [CrossRef]
- Winter, J.; Jung, S.; Keller, S.; Gregory, R.I.; Diederich, S. Many roads to maturity: MicroRNA biogenesis pathways and their regulation. Nat. Cell. Biol. 2009, 11, 228–234. [Google Scholar]
- Boggs, R.M.; Wright, Z.M.; Stickney, M.J.; Porter, W.W.; Murphy, K.E. MicroRNA expression in canine mammary cancer. Mamm. Genome 2008, 19, 561–569. [Google Scholar]
- Agra-Andrieu, N.; Motiño, O.; Mayoral, R.; Llorente-Izquierdo, C.; Fernández-Alvarez, A.; Boscá, L.; Casado, M.; Martín-Sanz, P. Cyclooxygenase-2 is a target of microRNA-16 in human hepatoma cells. PLoS One 2012, 7, e50935. [Google Scholar]
- Jiang, Q.Q.; Liu, B.; Yuan, T. MicroRNA-16 inhibits bladder cancer proliferation by targeting cyclin D1. Asian Pac. J. Cancer Prev. 2013, 14, 4127–4130. [Google Scholar]
- Kawai, N.; Tsujii, M.; Tsuji, S. Cyclooxygenases and colon cancer. Prostaglandins Other Lipid Mediat. 2002, 68–69, 187–196. [Google Scholar] [CrossRef]
- Shirahama, T.; Arima, J.; Akiba, S.; Sakakura, C. Relation between cyclooxygenase-2 expression and tumor invasiveness and patient survival in transitional cell carcinoma of the urinary bladder. Cancer 2001, 92, 188–193. [Google Scholar]
- Shirahama, T.; Sakakura, C. Over-expression of cyclooxygenase-2 in squamous cell carcinoma of the urinary bladder. Clin. Cancer Res. 2001, 7, 558–561. [Google Scholar]
- Smakman, N.; Schaap, N.; Snijckers, C.M.; Rinkes, I.H.B.; Kranenburg, O. NS-398, a selective cyclooxygenase-2 inhibitor, reduces experimental bladder carcinomaoutg owth by inhibiting tumor cell proliferation. Urology 2005, 66, 434–440. [Google Scholar] [CrossRef]
- Gee, J.; Sabichi, A.L.; Grossman, H.B. Chemoprevention of superficial bladder cancer. Crit. Rev. Oncol. Hematol. 2002, 43, 277–286. [Google Scholar]
- Okie, S. Raising the safety bar—The FDA’s coxib meeting. N. Engl. J. Med. 2005, 352, 1283–1285. [Google Scholar]
- Herfs, M.; Herman, L.; Hubert, P.; Minner, F.; Arafa, M.; Roncarati, P.; Henrotin, Y.; Boniver, J.; Delvenne, P. High expression of PGE2 enzymatic pathways in cervical (pre) neoplastic lesions and functional consequences for antigen-presenting cells. Cancer Immunol. Immunother. 2009, 58, 603–614. [Google Scholar] [CrossRef]
- Awad, M.I.; Alkadru, A.M.; Behrens, R.H.; Baraka, O.Z.; Eltayeb, I.B. Descriptive study on the efficacy and safety of artesunate suppository in combination with other antimalarials in the treatment of severe malaria in Sudan. Am. J. Trop. Med. Hyg. 2003, 68, 153–158. [Google Scholar]
- Woerdenbag, H.J.; Moskal, T.A.; Pras, N.; Malingré, T.M.; El-Feraly, F.S.; Kampinga, H.H.; Antonius, W.T.K. Cytotoxicity of artemisinin-related endoperoxides to ehrlich ascites tumor cells. J. Nat. Prod. 1993, 56, 849–856. [Google Scholar] [CrossRef]
- Dell’Eva, R.; Pfeffer, U.; Vené, R.; Anfosso, L.; Forlani, A.; Albini, A.; Efferth, T. Inhibition of angiogenesis in vivo and growth of Kaposi’s sarcoma xenograft tumors by the anti-malarial artesunate. Biochem. Pharmacol. 2004, 68, 2359–2366. [Google Scholar] [CrossRef]
- Jin, M.; Shen, X.; Zhao, C.; Qin, X.; Liu, H.; Huang, L.; Huang, L.; Qiu, Z.; Liu, Y. In vivo study of effects of artesunate nanoliposomes on human hepatocellular carcinomaxenografts in nude mice. Drug Deliv. 2013, 20, 127–133. [Google Scholar] [CrossRef]
- Ma, H.; Yao, Q.; Zhang, A.M.; Lin, S.; Wang, X.X.; Wu, L.; Sun, J.G.; Chen, Z.T. The effects of artesunate on the expression of EGFR and ABCG2 in A549 human lung cancer cells and a xenograft model. Molecules 2011, 16, 10556–10569. [Google Scholar]
- Xu, Q.; Li, Z.X.; Peng, H.Q.; Sun, Z.W.; Cheng, R.L.; Ye, Z.M.; Li, W.X. Artesunate inhibits growth and induces apoptosis in human osteosarcoma HOS cell line in vitro and in vivo. J. Zhejiang Univ. Sci. B 2011, 12, 247–255. [Google Scholar] [CrossRef]
- Earnshaw, W.C.; Martins, L.M.; Kaufmann, S.H. Mammalian caspases: Structure, activation, substrates, and functions during apoptosis. Annu. Rev. Biochem. 1999, 68, 383–424. [Google Scholar] [CrossRef]
- Pruthi, R.S.; Derksen, E.; Gaston, K. Cyclooxygenase-2 as a potential target in the prevention and treatment of genitourinary tumors: A review. J. Urol. 2003, 169, 2352–2359. [Google Scholar]
- Efferth, T.; Kaina, B. Toxicity of the antimalarial artemisinin and its dervatives. Crit. Rev. Toxicol. 2010, 40, 405–421. [Google Scholar]
- Zhang, L.X.; Liu, Z.N.; Ye, J.; Sha, M.; Qian, H.; Bu, X.H.; Luan, Z.Y.; Xu, X.L.; Huang, A.H.; Yuan, D.L.; et al. Artesunate exerts an anti-immunosuppressive effect on cervical cancer by inhibiting PGE2 production and Foxp3 expression. Cell. Biol. Int. 2014, 38, 639–646. [Google Scholar]
- Aqeilan, R.I.; Calin, G.A.; Croce, C.M. miR-15a and miR-16-1 in cancer: Discovery, function and future perspectives. Cell. Death Differ. 2010, 17, 215–220. [Google Scholar]
- Gatt, M.E.; Zhao, J.J.; Ebert, M.S.; Zhang, Y.; Chu, Z.; Mani, M.; Gazit, R.; Carrasco1, D.E.; Dutta-Simmons, J.; Adamia, S.; et al. MicroRNAs 15a/16-1 function as tumor suppressor genes in multiple myeloma. Blood 2010, 117, 7188. [Google Scholar]
- Young, L.E.; Moore, A.E.; Sokol, L.; Meisner-Kober, N.; Dixon, D.A. The mRNA stability factor HuR inhibits microRNA-16 targeting of COX-2. Mol. Cancer Res. 2012, 10, 167–180. [Google Scholar]
- Qin, J.; Yuan, J.; Li, L.; Liu, H.; Qin, R.; Qin, W.; Chen, B.Q.; Wang, H.; Wu, K.C. In vitro and in vivo inhibitory effect evaluation of cyclooxygenase-2 inhibitors, antisense cyclooxygenase-2 cDNA, and their combination on the growth of human bladder cancercells. Biomed. Pharmacother. 2009, 63, 241–248. [Google Scholar] [CrossRef]
- Parada, B.; Reis, F.; Cerejo, R.; Garrido, P.; Sereno, J.; Xavier-Cunha, M.; Neto, P.; Mota, A.; Figueiredo, A.; Teixeira, F.; et al. ω-3 fatty acids inhibit tumor growth in a rat model of bladder cancer. Biomed. Res. Int. 2013, 2013, 368178. [Google Scholar]
- Vermes, I.; Haanen, C.; Reutelingsperger, C. Flow cytometry of apoptotic cell death. J. Immunol. Methods 2000, 243, 167–190. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zuo, W.; Wang, Z.-Z.; Xue, J. Artesunate Induces Apoptosis of Bladder Cancer Cells by miR-16 Regulation of COX-2 Expression. Int. J. Mol. Sci. 2014, 15, 14298-14312. https://doi.org/10.3390/ijms150814298
Zuo W, Wang Z-Z, Xue J. Artesunate Induces Apoptosis of Bladder Cancer Cells by miR-16 Regulation of COX-2 Expression. International Journal of Molecular Sciences. 2014; 15(8):14298-14312. https://doi.org/10.3390/ijms150814298
Chicago/Turabian StyleZuo, Wei, Zhen-Zhong Wang, and Jun Xue. 2014. "Artesunate Induces Apoptosis of Bladder Cancer Cells by miR-16 Regulation of COX-2 Expression" International Journal of Molecular Sciences 15, no. 8: 14298-14312. https://doi.org/10.3390/ijms150814298
APA StyleZuo, W., Wang, Z.-Z., & Xue, J. (2014). Artesunate Induces Apoptosis of Bladder Cancer Cells by miR-16 Regulation of COX-2 Expression. International Journal of Molecular Sciences, 15(8), 14298-14312. https://doi.org/10.3390/ijms150814298



