Applications and Mechanisms of Ionic Liquids in Whole-Cell Biotransformation
Abstract
:1. Introduction
2. Characteristics of Ionic Liquids for Whole-Cell Catalysis
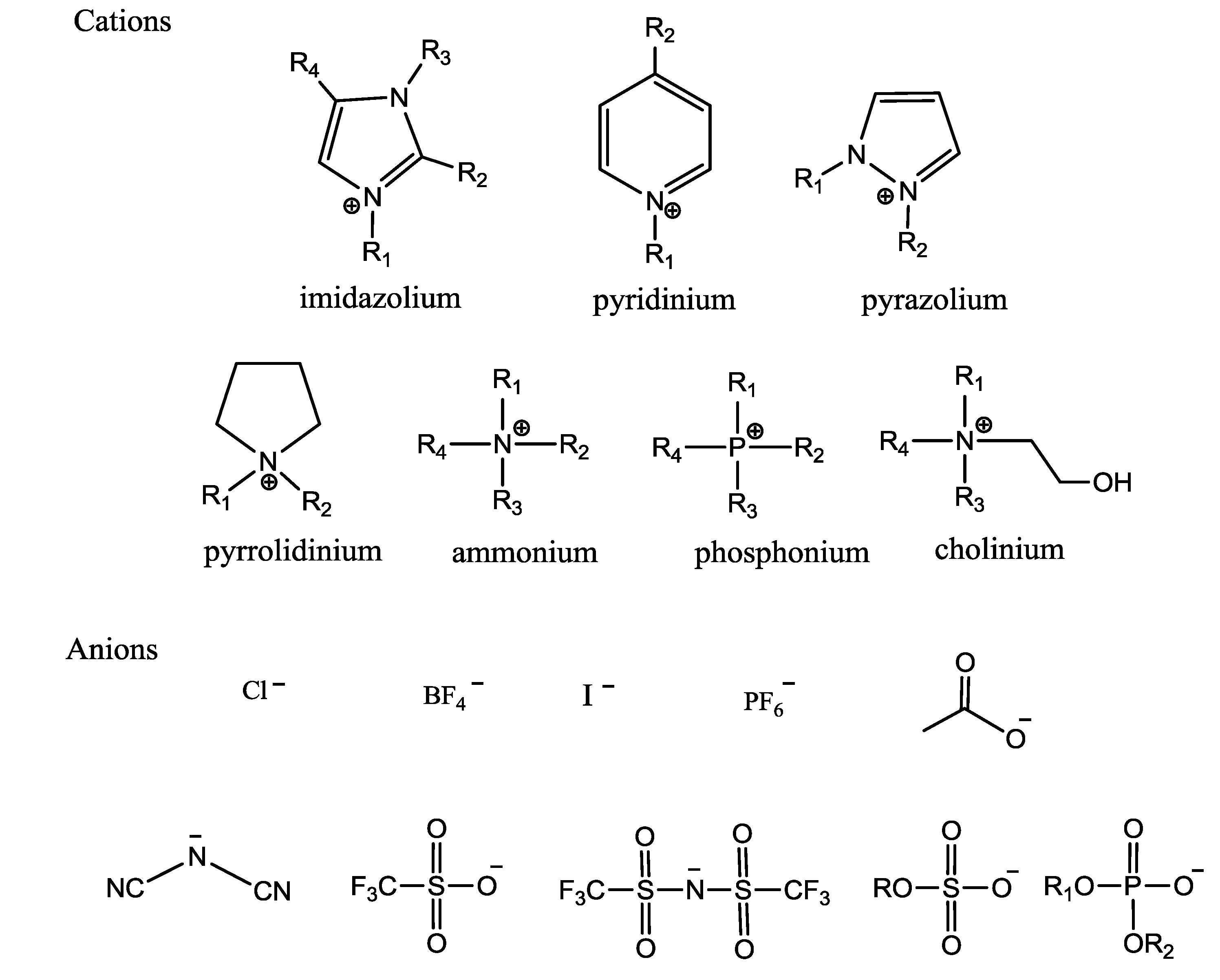
2.1. The Melting Point of ILs
2.2. The Viscosity of ILs
2.3. The Density of ILs
2.4. The Polarity of ILs
3. Whole-Cell Biocatalytic Transformations in Ionic Liquids
| Microorganism | ILs | Biocatalysis Reaction | Improvement | Reference | |||||
|---|---|---|---|---|---|---|---|---|---|
| Penicillium purpurogenum Li-3 | (BMIM)(BF4) | Biosynthesis of glycyrrhetic acid 3-O-mono-β-d-glucuronide (GAMG) | A yield of 2.62 g·L−1 after 62 h in IL co-solvent medium compared to 2.34 g·L−1 after 72 h in buffer medium. | [17] | |||||
| Rhodotorula glutinis ATCC201718 | (DEME)(Tf2N) (BMIM)(PF6) (HMIM)(PF6) (OMIN)(PF6) etc. | Hydrolysis reaction of racemic 1,2-epoxyhexane | The high enantiomeric ratio of (R)-diol (E > 100) without significant decrease in the reactivity was accomplished by adding 1-heptanol in minute amounts to dodecane. | [66] | |||||
| Saccharomyces cerevisiae Candida albicans Rhodotorula glutinis Geotrichum candidum Micrococcus luteus | (BMIM)(BF6) | Reductions of (Z)-C6H5CH=CXC (=O)CH3 (X = Cl, Br) | Better diastereoselectivity and enantiose-lectivity than in pure water. | [35] | |||||
| Penicillium purpurogenum Li-3 (w-PGUS) Escherichia coli BL21 Pichia pastoris GS115 | (BMIM)(BF6) | Hydrolysis of glycyrrhizin (GL) to glycyrrhetic acid 3-O-mono-β-d-glucuronide (GAMG) | The 60 g·L−1 (1.23 U/g) cell concentration, a GAMG yield of 87.63% was achieved after 60 h. | [67] | |||||
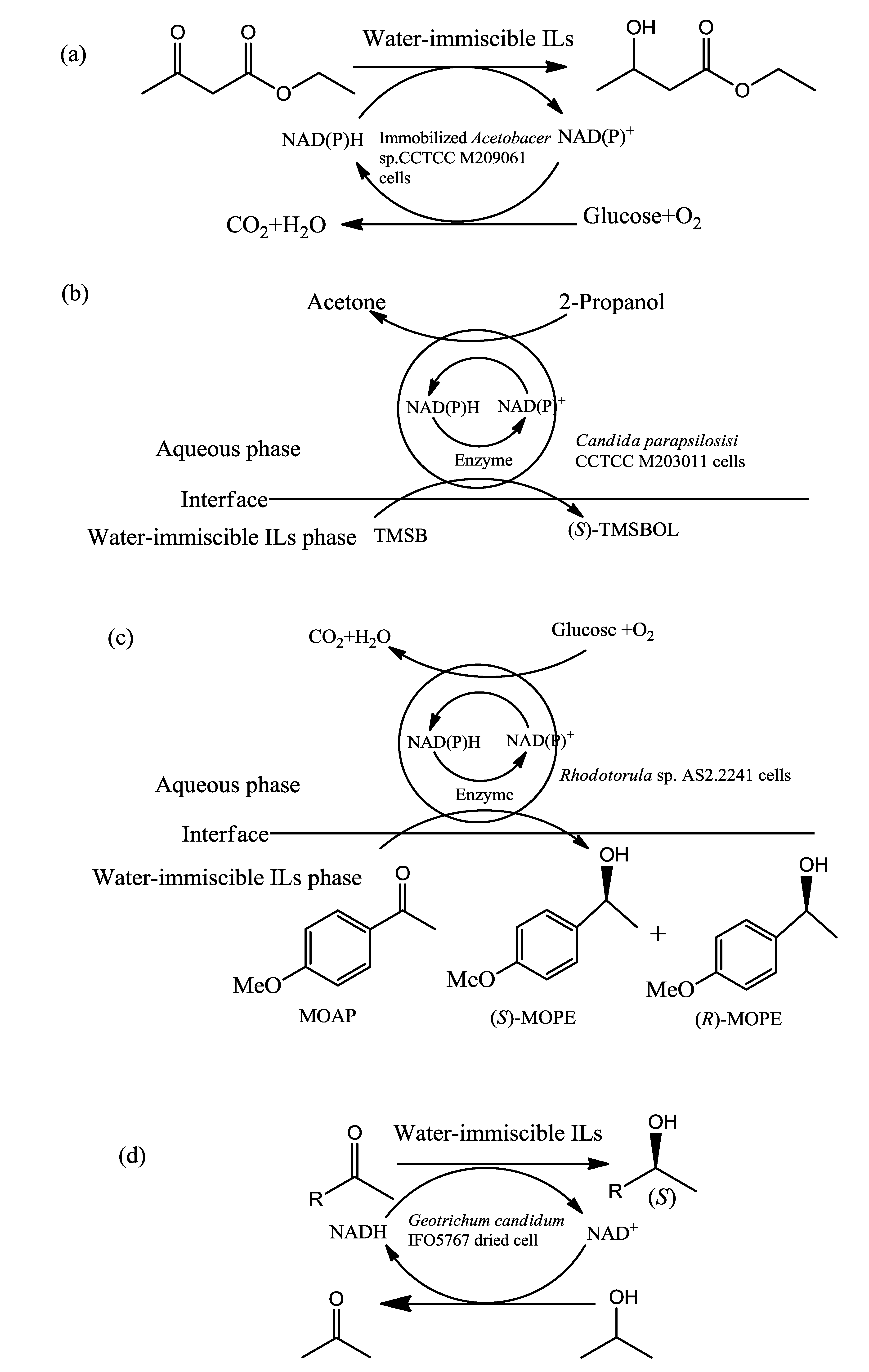
| Acetobacter sp. CCTCC M209061 | (C2OHMIM)(NO3) | Reduction of 4-(trimethylsilyl)-3-butyn-2-one reduction to (R)-4-(trimethylsilyl)-3-butyn-2-ol | The initial reaction rate, the maximum yield and the product e.e. were 14.0 mmol·min−1·(g·cell)−1, 91%, and 499%, respectively. | [68] | |||||
| Aspergillus ochraceus | (C3MIM)(PF6) | Reduction of 11α hydroxylation of 16α,17-epoxyprogesterone (HEP) | The substrate conversion reached 90% with a substrate concentration of 20 g·L−1 under the selected conditions. | [69] | |||||
| Escherichia coli | ((EO2E)MPL)(NTF) (HMPL)(NTF) (HPYR)(NTF) ((NEMM)EO2E)(NTF) | Reduction of 2-octanone to (R)-2-octanol | Various ionic liquids can be used for this reaction and the ionic liquid volume fractions are up to 40%. | [70] | |||||
| Escherichia coli | (HMPL)(NTF) | The asymmetric reduction of 2-octanone to (R)-2-octanol | The average conversion was 98.5 (±0.7)%, and enantiomeric excesses were constant at values ≥99.5% (R). A total of 999 (±6) g (R)-2-octanol·L−1 IL was produced. | [42] | |||||
| Rhizopus nigricans | (BMIM)(PF6) (BMIM)(NTf2) | 11α-Hydroxylation of 16α,17-epoxyprogesterone | The conversion was greatly increased to above 90% at 18 g·L−1 feeding concentration. | [71] | |||||
3.1. Whole-Cell Biotranformation in Ionic Liquids by Bacterium
3.2. Whole-Cell Biotranformation in Ionic Liquids by Yeasts
3.3. Whole-Cell Biotranformation in Ionic Liquids by Fungi
4. Interaction Mechanisms of Ionic Liquids with Cells in Whole-Cell Biotransformation
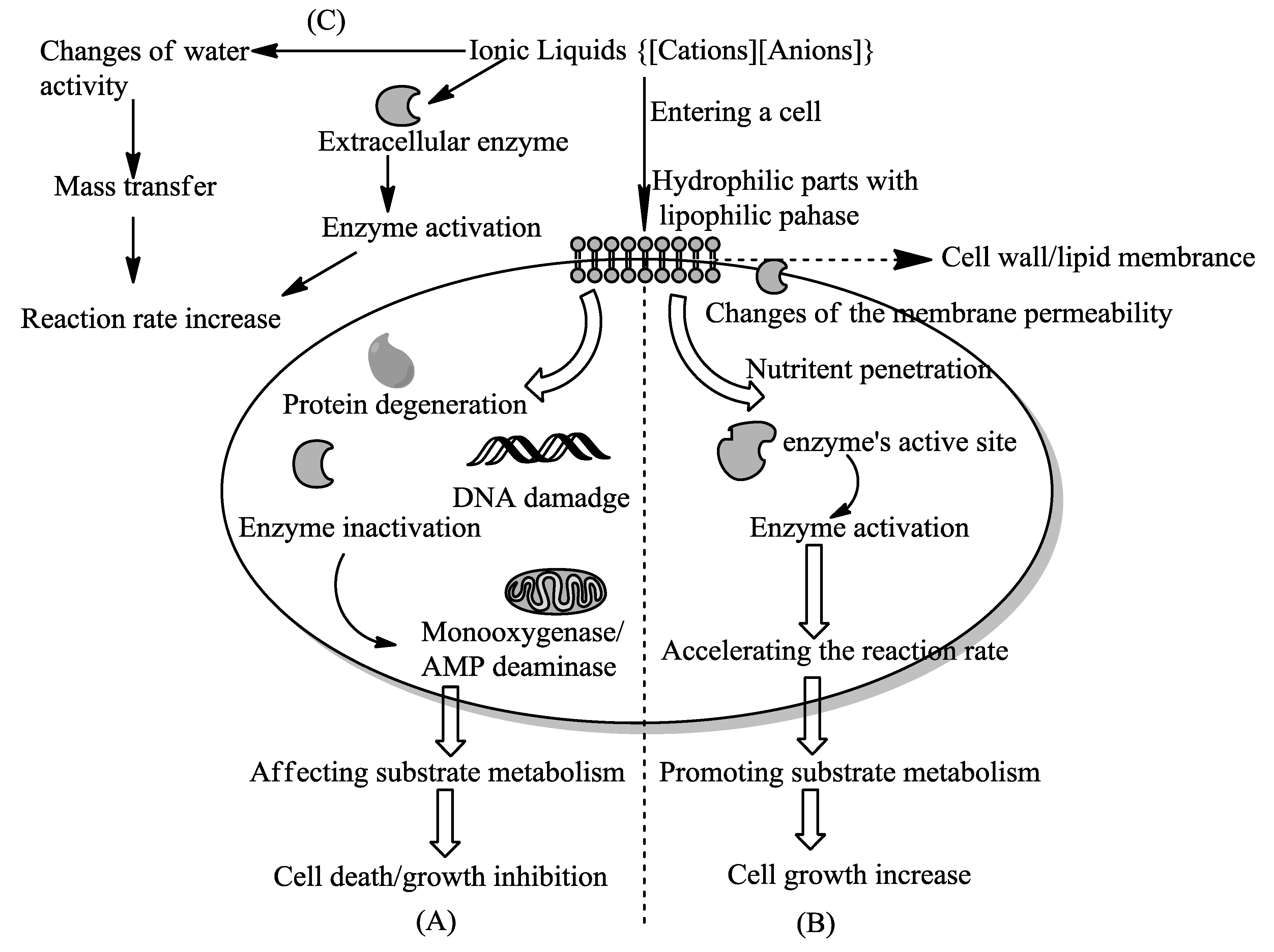
4.1. To Regulate The Cell Growth
4.2. To Enhance the Reaction Rate
5. Future Perspectives and Conclusions
5.1. Microstructure Changes of Cells in ILs
5.2. Safety Evaluations of the ILs in Whole-Cell Biotransformations
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hajfarajollah, H.; Mokhtarani, B.; Sharifi, A.; Mirzaei, M.; Afaghi, A. Toxicity of various kinds of ionic liquids towards the cell growth and end product formation of the probiotic strain, Propionibacterium freudenreichii. RSC Adv. 2014, 4, 13153–13160. [Google Scholar] [CrossRef]
- Dominguez de Maria, P.; Maugeri, Z. Ionic liquids in biotransformations: From proof-of-concept to emerging deep-eutectic-solvents. Curr. Opin. Chem. Biol. 2011, 15, 220–225. [Google Scholar] [CrossRef]
- Earle, M.J.; Seddon, K.R. Ionic liquids. Green solvents for the future. Pure Appl. Chem. 2000, 72, 1391–1398. [Google Scholar]
- Das, R.N.; Roy, K. Development of classification and regression models for Vibrio fischeri toxicity of ionic liquids: Green solvents for the future. Toxicol. Res. 2012, 1, 186–195. [Google Scholar] [CrossRef]
- Talavera-Prieto, N.M.C.; Ferreira, A.G.M.; Simoes, P.N.; Carvalho, P.J.; Mattedi, S.; Coutinho, J.A.P. Thermophysical characterization of N-methyl-2-hydroxyethylammonium carboxilate ionic liquids. J. Chem. Thermodyn. 2014, 68, 221–234. [Google Scholar] [CrossRef]
- Quijano, G.; Couvert, A.; Amrane, A. Ionic liquids: Applications and future trends in bioreactor technology. Bioresour. Technol. 2010, 101, 8923–8930. [Google Scholar] [CrossRef]
- Machado, M.F.; Queiros, R.P.; Santos, M.D.; Fidalgo, L.G.; Delgadillo, I.; Saraiva, J.A. Effect of ionic liquids alkyl chain length on horseradish peroxidase thermal inactivation kinetics and activity recovery after inactivation. World J. Microbiol. Biotechnol. 2014, 30, 487–494. [Google Scholar] [CrossRef]
- Van Rantwijk, F.; Madeira Lau, R.; Sheldon, R.A. Biocatalytic transformations in ionic liquids. Trends Biotechnol. 2003, 21, 131–138. [Google Scholar] [CrossRef]
- Ho, T.D.; Zhang, C.; Hantao, L.W.; Anderson, J.L. Ionic Liquids in analytical chemistry: Fundamentals, Advances, and Perspectives. Anal. Chem. 2014, 86, 262–285. [Google Scholar] [CrossRef]
- Sun, P.; Armstrong, D.W. Ionic liquids in analytical chemistry. Anal. Chim. Acta 2010, 661, 1–16. [Google Scholar] [CrossRef]
- Peng, W.-H.; Lee, Y.-Y.; Wu, C.; Wu, K.C.-W. Acid–base bi-functionalized, large-pored mesoporous silica nanoparticles for cooperative catalysis of one-pot cellulose-to-HMF conversion. J. Mater. Chem. 2012, 22, 23181–23185. [Google Scholar] [CrossRef]
- Qi, X.; Watanabe, M.; Aida, T.M.; Smith, R.L. Catalytic conversion of cellulose into 5-hydroxymethylfurfural in high yields via a two-step process. Cellulose 2011, 18, 1327–1333. [Google Scholar] [CrossRef]
- Hsu, W.-H.; Lee, Y.-Y.; Peng, W.-H.; Wu, K.C.W. Cellulosic conversion in ionic liquids (ILs): Effects of H2O/cellulose molar ratios, temperatures, times, and different ILs on the production of monosaccharides and 5-hydroxymethylfurfural (HMF). Catal. Today 2011, 174, 65–69. [Google Scholar] [CrossRef]
- Su, Y.; Brown, H.M.; Huang, X.; Zhou, X.-D.; Amonette, J.E.; Zhang, Z.C. Single-step conversion of cellulose to 5-hydroxymethylfurfural (HMF), a versatile platform chemical. Appl. Catal. A Gen. 2009, 361, 117–122. [Google Scholar] [CrossRef]
- Bräutigam, S.; Dennewald, D.; Schürmann, M.; Lutje-Spelberg, J.; Pitner, W.-R.; Weuster-Botz, D. Whole-cell biocatalysis: Evaluation of new hydrophobic ionic liquids for efficient asymmetric reduction of prochiral ketones. Enzym. Microb. Technol. 2009, 45, 310–316. [Google Scholar] [CrossRef]
- Kohlmann, C.; Robertz, N.; Leuchs, S.; Dogan, Z.; Lütz, S.; Bitzer, K.; Na’amnieh, S.; Greiner, L. Ionic liquid facilitates biocatalytic conversion of hardly water soluble ketones. J. Mol. Catal. B Enzym. 2011, 68, 147–153. [Google Scholar] [CrossRef]
- Kaleem, I.; Shen, H.; Lv, B.; Wei, B.; Rasool, A.; Li, C. Efficient biosynthesis of glycyrrhetic acid 3-O-mono-β-d-glucuronide (GAMG) in water-miscible ionic liquid by immobilized whole cells of Penicillium purpurogenum Li-3 in alginate gel. Chem. Eng. Sci. 2014, 106, 136–143. [Google Scholar] [CrossRef]
- Zhao, H. Methods for stabilizing and activating enzymes in ionic liquids-a review. J. Chem. Technol. Biotechnol. 2010, 85, 891–907. [Google Scholar] [CrossRef]
- Zhang, W.G.; Wei, D.Z.; Yang, X.P.; Song, Q.X. Penicillin acylase catalysis in the presence of ionic liquids. Bioprocess. Biosyst. Eng. 2006, 29, 379–383. [Google Scholar] [CrossRef]
- Wang, X.H.; Dai, J.T.; Wang, J.P.; Wu, H. Enhancement extraction of quercetin from suaeda glauca bge. Using ionic liquids as solvent. Asian J. Chem. 2014, 26, 1111–1115. [Google Scholar]
- Hussain, W.; Pollard, D.J.; Truppo, M.; Lye, G.J. Enzymatic ketone reductions with co-factor recycling: Improved reactions with ionic liquid co-solvents. J. Mol. Catal. B Enzym. 2008, 55, 19–29. [Google Scholar] [CrossRef]
- Kragl, U.; Eckstein, M.; Kaftzik, N. Enzyme catalysis in ionic liquids. Curr. Opin. Biotechnol. 2002, 13, 565–571. [Google Scholar] [CrossRef]
- Sun, S.; Qin, F.; Bi, Y.; Chen, J.; Yang, G.; Liu, W. Enhanced transesterification of ethyl ferulate with glycerol for preparing glyceryl diferulate using a lipase in ionic liquids as reaction medium. Biotechnol. Lett. 2013, 35, 1449–1454. [Google Scholar] [CrossRef]
- Wu, C.; Zhang, Z.; Chen, C.; He, F.; Zhuo, R. Synthesis of poly (epsilon-caprolactone) by an immobilized lipase coated with ionic liquids in a solvent-free condition. Biotechnol. Lett. 2013, 35, 1623–1630. [Google Scholar] [CrossRef]
- Bose, S.; Barnes, C.A.; Petrich, J.W. Enhanced stability and activity of cellulase in an ionic liquid and the effect of pretreatment on cellulose hydrolysis. Biotechnol. Bioeng. 2012, 109, 434–443. [Google Scholar] [CrossRef]
- Xiong, Y.; Zhang, Z.H.; Wang, X.; Liu, B.; Lin, J.T. Hydrolysis of cellulose in ionic liquids catalyzed by a magnetically-recoverable solid acid catalyst. Chem. Eng. J. 2014, 235, 349–355. [Google Scholar] [CrossRef]
- Rehmann, L.; Ivanova, E.; Gunaratne, H.Q.N.; Seddon, K.R.; Stephens, G. Enhanced laccase stability through mediator partitioning into hydrophobic ionic liquids. Green Chem. 2014, 16, 1462–1469. [Google Scholar] [CrossRef]
- Karjalainen, E.; Izquierdo, D.F.; Marti-Centelles, V.; Luis, S.V.; Tenhu, H.; Garcia-Verdugo, E. An enzymatic biomimetic system: Enhancement of catalytic efficiency with new polymeric chiral ionic liquids synthesised by controlled radical polymerisation. Polym. Chem. 2014, 5, 1437–1446. [Google Scholar] [CrossRef]
- Goldfeder, M.; Fishman, A. Modulating enzyme activity using ionic liquids or surfactants. Appl. Microbiol. Biot. 2014, 98, 545–554. [Google Scholar] [CrossRef]
- Soukup-Hein, R.J.; Warnke, M.M.; Armstrong, D.W. Ionic liquids in analytical chemistry. Annu. Rev. Anal. Chem. 2009, 2, 145–168. [Google Scholar] [CrossRef]
- Williams, H.D.; Sahbaz, Y.; Ford, L.; Nguyen, T.H.; Scammells, P.J.; Porter, C.J.H. Ionic liquids provide unique opportunities for oral drug delivery: Structure optimization and in vivo evidence of utility. Chem. Commun. 2014, 50, 1688–1690. [Google Scholar] [CrossRef]
- Khachatryan, K.S.; Smirnova, S.V.; Torocheshnikova, I.I.; Shvedene, N.V.; Formanovsky, A.A.; Pletnev, I.V. Solvent extraction and extraction-voltammetric determination of phenols using room temperature ionic liquid. Anal. Bioanal. Chem. 2005, 381, 464–470. [Google Scholar] [CrossRef]
- Smirnova, S.V.; Torocheshnikova, I.I.; Formanovsky, A.A.; Pletnev, I.V. Solvent extraction of amino acids into a room temperature ionic liquid with dicyclohexano-18-crown-6. Anal. Bioanal. Chem. 2004, 378, 1369–1375. [Google Scholar] [CrossRef]
- Wang, J.-H.; Cheng, D.-H.; Chen, X.-W.; Du, Z.; Fang, Z.-L. Direct extraction of double-stranded DNA into ionic liquid 1-butyl-3-methylimidazolium hexafluorophosphate and its quantification. Anal. Chem. 2007, 79, 620–625. [Google Scholar] [CrossRef]
- Zampieri, D.S.; de Paula, B.R.S.; Zampieri, L.A.; Vale, J.A.; Rodrigues, J.A.R.; Moran, P.J.S. Enhancements of enantio and diastereoselectivities in reduction of (Z)-3-halo-4-phenyl-3-buten-2-one mediated by microorganisms in ionic liquid/water biphasic system. J. Mol. Catal. B Enzym. 2013, 85–86, 61–64. [Google Scholar] [CrossRef]
- Quijano, G.; Couvert, A.; Amrane, A.; Darracq, G.; Couriol, C.; le Cloirec, P.; Paquin, L.; Carrié, D. Toxicity and biodegradability of ionic liquids: New perspectives towards whole-cell biotechnological applications. Chem. Eng. J. 2011, 174, 27–32. [Google Scholar] [CrossRef]
- Thi, P.T.P.; Cho, C.W.; Yun, Y.S. Environmental fate and toxicity of ionic liquids: A review. Water Res. 2010, 44, 352–372. [Google Scholar] [CrossRef]
- Zhang, C.; Malhotra, S.V.; Francis, A.J. Toxicity of ionic liquids to Clostridium sp. and effects on uranium biosorption. J. Hazard. Mater. 2014, 264, 246–253. [Google Scholar] [CrossRef]
- Matsumoto, M.; Mochiduki, K.; Kondo, K. Toxicity of ionic liquids and organic solvents to lactic acid-producing bacteria. J. Biosci. Bioeng. 2004, 98, 344–347. [Google Scholar] [CrossRef]
- Holbrey, J.D.; Seddon, K.R. Ionic liquids. Clean Prod. Process. 1999, 1, 223–236. [Google Scholar]
- Jain, N.; Kumar, A.; Chauhan, S.; Chauhan, S.M.S. Chemical and biochemical transformations in ionic liquids. Tetrahedron 2005, 61, 1015–1060. [Google Scholar] [CrossRef]
- Dennewald, D.; Pitner, W.-R.; Weuster-Botz, D. Recycling of the ionic liquid phase in process integrated biphasic whole-cell biocatalysis. Process. Biochem. 2011, 46, 1132–1137. [Google Scholar] [CrossRef]
- Welton, T. Ionic liquids in catalysis. Coordin. Chem. Rev. 2004, 248, 2459–2477. [Google Scholar] [CrossRef]
- Tsunashima, K.; Sakai, Y.; Matsumiya, M. Physical and electrochemical properties of phosphonium ionic liquids derived from trimethylphosphine. Electrochem. Commun. 2014, 39, 30–33. [Google Scholar] [CrossRef]
- Olivier-Bourbigou, H.; Magna, L.; Morvan, D. Ionic liquids and catalysis: Recent progress from knowledge to applications. Appl. Catal. A Gen. 2010, 373, 1–56. [Google Scholar] [CrossRef]
- Park, S.; Kazlauskas, R.J. Biocatalysis in ionic liquids—Advantages beyond green technology. Curr. Opin. Biotechnol. 2003, 14, 432–437. [Google Scholar] [CrossRef]
- Yang, Z.; Pan, W. Ionic liquids: Green solvents for nonaqueous biocatalysis. Enzym. Microb. Technol. 2005, 37, 19–28. [Google Scholar] [CrossRef]
- Kaar, J.L.; Jesionowski, A.M.; Berberich, J.A.; Moulton, R.; Russell, A.J. Impact of ionic liquid physical properties on lipase activity and stability. J. Am. Chem. Soc. 2003, 125, 4125–4131. [Google Scholar] [CrossRef]
- Reichardt, C. Solvatochromic dyes as solvent polarity indicators. Chem. Rev. 1994, 94, 2319–2358. [Google Scholar] [CrossRef]
- Reichardt, C.; Che, D.Q.; Heckenkemper, G.; Schafer, G. Syntheses and UV/Vis-spectroscopic properties of hydrophilic 2-, 3-, and 4-pyridyl-substituted solvatochromic and halochromic pyridinium N-phenolate betaine dyes as new empirical solvent polarity indicators. Eur. J. Org. Chem. 2001, 2001, 2343–2361. [Google Scholar] [CrossRef]
- Seddon, K.R.; Carmichael, A.J. Polarity study of some 1-alkyl-3-methylimidazolium ambient-temperature ionic liquids with the solvatochromic dye, Nile Red. J. Phys. Org. Chem. 2000, 13, 591–595. [Google Scholar] [CrossRef]
- Ropel, L.; Belvèze, L.S.; Aki, S.N.V.K.; Stadtherr, M.A.; Brennecke, J.F. Octanol-water partition coefficients of imidazolium-based ionic liquids. Green Chem. 2005, 7, 83–90. [Google Scholar] [CrossRef]
- Moniruzzaman, M.; Nakashima, K.; Kamiya, N.; Goto, M. Recent advances of enzymatic reactions in ionic liquids. Biochem. Eng. J. 2010, 48, 295–314. [Google Scholar] [CrossRef]
- Baker, S.N.; Baker, G.A.; Bright, F.V. Temperature-dependent microscopic solvent properties of “dry” and “wet” 1-butyl-3-methylimidazolium hexafluorophosphate: Correlation with ET (30) and Kamlet–Taft polarity scales. Green Chem. 2002, 4, 165–169. [Google Scholar] [CrossRef]
- Carothers, J.M.; Goler, J.A.; Keasling, J.D. Chemical synthesis using synthetic biology. Curr. Opin. Biotechnol. 2009, 20, 498–503. [Google Scholar] [CrossRef]
- Rozzell, J.D. Commercial scale biocatalysis: Myths and realities. Bioorg. Med. Chem. 1999, 7, 2253–2261. [Google Scholar] [CrossRef]
- Clomburg, J.M.; Gonzalez, R. Biofuel production in Escherichia coli: The role of metabolic engineering and synthetic biology. Appl. Microbiol. Biotechnol. 2010, 86, 419–434. [Google Scholar] [CrossRef]
- Wood, N.; Ferguson, J.L.; Gunaratne, H.Q.N.; Seddon, K.R.; Goodacre, R.; Stephens, G.M. Screening ionic liquids for use in biotransformations with whole microbial cells. Green Chem. 2011, 13, 1843–1851. [Google Scholar] [CrossRef]
- Athalye, S.; Sharma-Shivappa, R.; Peretti, S.; Kolar, P.; Davis, J.P. Producing biodiesel from cottonseed oil using Rhizopus. oryzae ATCC #34612 whole cell biocatalysts: Culture media and cultivation period optimization. Energy Sustain Dev. 2013, 17, 331–336. [Google Scholar] [CrossRef]
- Nakashima, K.; Arai, S.; Tanino, T.; Ogino, C.; Kondo, A.; Fukuda, H. Production of biodiesel fuel in ionic liquids catalyzed by whole-cell biocatalysts. J. Biosci. Bioeng. 2009, 108, S43–S43. [Google Scholar]
- Allen, C.C.R.; Boudet, C.J.; Hardacre, C.; Migaud, M.E. Enhancement of whole cell dioxygenase biotransformations of haloarenes by toxic ionic liquids. RSC Adv. 2014, 4, 19916–19924. [Google Scholar] [CrossRef]
- Yan, H.; Long, Y.; Song, K.; Tung, C.H.; Zheng, L.Q. Photo-induced transformation from wormlike to spherical micelles based on pyrrolidinium ionic liquids. Soft Matter 2014, 10, 115–121. [Google Scholar] [CrossRef]
- Baum, S.; van Rantwijk, F.; Stolz, A. Application of a recombinant Escherichia coli whole-cell catalyst expressing hydroxynitrile lyase and nitrilase activities in ionic liquids for the production of (S)-Mandelic Acid and (S)-Mandeloamide. Adv. Synth. Catal. 2012, 354, 113–122. [Google Scholar] [CrossRef]
- Lou, W.Y.; Zong, M.H.; Smith, T.J. Use of ionic liquids to improve whole-cell biocatalytic asymmetric reduction of acetyltrimethylsilane for efficient synthesis of enantiopure (S)-1-trimethylsilylethanol. Green Chem. 2006, 8, 147–155. [Google Scholar] [CrossRef]
- Sendovski, M.; Nir, N.; Fishman, A. Bioproduction of 2-Phenylethanol in a biphasic ionic liquid aqueous system. J. Agric. Food Chem. 2010, 58, 2260–2265. [Google Scholar] [CrossRef]
- Matsumoto, M.; Sugimoto, T.; Ishiguro, Y.; Yamaguchi, H.; Kondo, K. Effect of organic solvents and ionic liquids on resolution of 2-epoxyhexane by whole cells of Rhodotorula glutinisin a two-liquid phase system. J. Chem. Technol. Biot. 2014, 89, 522–527. [Google Scholar] [CrossRef]
- Chen, J.-Y.; Kaleem, I.; He, D.-M.; Liu, G.-Y.; Li, C. Efficient production of glycyrrhetic acid 3-O-mono-β-d-glucuronide by whole-cell biocatalysis in an ionic liquid/buffer biphasic system. Process. Biochem. 2012, 47, 908–913. [Google Scholar] [CrossRef]
- Xiao, Z.-J.; Du, P.-X.; Lou, W.-Y.; Wu, H.; Zong, M.-H. Using water-miscible ionic liquids to improve the biocatalytic anti-Prelog asymmetric reduction of prochiral ketones with whole cells of Acetobacter. sp. CCTCC M209061. Chem. Eng. Sci. 2012, 84, 695–705. [Google Scholar] [CrossRef]
- Mao, S.; Hua, B.; Wang, N.; Hu, X.; Ge, Z.; Li, Y.; Liu, S.; Lu, F. 11α hydroxylation of 16α,17-epoxyprogesterone in biphasic ionic liquid/water system by Aspergillus ochraceus. J. Chem. Technol. Biotechnol. 2013, 88, 287–292. [Google Scholar] [CrossRef]
- Dennewald, D.; Hortsch, R.; Weuster-Botz, D. Evaluation of parallel milliliter-scale stirred-tank bioreactors for the study of biphasic whole-cell biocatalysis with ionic liquids. J. Biotechnol. 2012, 157, 253–257. [Google Scholar] [CrossRef]
- Wu, D.X.; Guan, Y.X.; Wang, H.Q.; Yao, S.J. 11α-Hydroxylation of 16α,17-epoxyprogesterone by Rhizopus nigricans in a biphasic ionic liquid aqueous system. Bioresour. Technol. 2011, 102, 9368–9373. [Google Scholar] [CrossRef]
- Cull, S.G.; Holbrey, J.D.; Vargas-Mora, V.; Seddon, K.R.; Lye, G.J. Room-temperature ionic liquids as replacements for organic solvents in multiphase bioprocess operations. Biotechnol. Bioengin. 2000, 69, 227–233. [Google Scholar] [CrossRef]
- Pfruender, H.; Amidjojo, M.; Kragl, U.; Weuster-Botz, D. Efficient whole-cell biotransformation in a biphasic ionic liquid/water system. Angew. Chem. 2004, 43, 4529–4531. [Google Scholar] [CrossRef]
- Wang, X.-T.; Yue, D.-M.; Zong, M.-H.; Lou, W.-Y. Use of ionic liquid to significantly improve asymmetric reduction of ethyl acetoacetate catalyzed by Acetobacter. sp. CCTCC M209061 Cells. Ind. Eng. Chem. Res. 2013, 52, 12550–12558. [Google Scholar] [CrossRef]
- Pfruender, H.; Jones, R.; Weuster-Botz, D. Water immiscible ionic liquids as solvents for whole cell biocatalysis. J. Biotechnol. 2006, 124, 182–190. [Google Scholar] [CrossRef]
- Dipeolu, O.; Green, E.; Stephens, G. Effects of water-miscible ionic liquids on cell growth and nitro reduction using Clostridium sporogenes. Green Chem. 2009, 11, 397–401. [Google Scholar] [CrossRef]
- Weuster-Botz, D. Process intensification of whole-cell biocatalysis with ionic liquids. Chem. Rec. 2007, 7, 334–340. [Google Scholar] [CrossRef]
- Gangu, S.A.; Weatherley, L.R.; Scurto, A.M. Whole-Cell Biocatalysis with Ionic Liquids. Curr. Org. Chem. 2009, 13, 1242–1258. [Google Scholar] [CrossRef]
- Cornmell, R.J.; Winder, C.L.; Schuler, S.; Goodacre, R.; Stephens, G. Using a biphasic ionic liquid/water reaction system to improve oxygenase-catalysed biotransformation with whole cells. Green Chem. 2008, 10, 685–691. [Google Scholar] [CrossRef]
- Silva, V.D.; Carletto, J.S.; Carasek, E.; Stambuk, B.U.; Nascimento, M.D.G. Asymmetric reduction of (4S)-(+)-carvone catalyzed by baker's yeast: A green method for monitoring the conversion based on liquid–liquid–liquid microextraction with polypropylene hollow fiber membranes. Process. Biochem. 2013, 48, 1159–1165. [Google Scholar] [CrossRef]
- Howarth, J.; James, P.; Dai, J. Immobilized baker’s yeast reduction of ketones in an ionic liquid, [BMIM]PF6 and water mix. Tetrahedron Lett. 2001, 7517–7519. [Google Scholar] [CrossRef]
- Lou, W.Y.; Chen, L.; Zhang, B.B.; Smith, T.J.; Zong, M.H. Using a water-immiscible ionic liquid to improve asymmetric reduction of 4-(trimethylsilyl)-3-butyn-2-one catalyzed by immobilized Candida parapsilosis CCTCC M203011 cells. BMC Biotechnol. 2009. [Google Scholar] [CrossRef]
- Wang, W.; Zong, M.-H.; Lou, W.-Y. Use of an ionic liquid to improve asymmetric reduction of 4′-methoxyacetophenone catalyzed by immobilized Rhodotorula sp. AS2.2241 cells. J. Mol. Catal. B Enzym. 2009, 56, 70–76. [Google Scholar] [CrossRef]
- Adachi, D.; Hama, S.; Nakashima, K.; Bogaki, T.; Ogino, C.; Kondo, A. Production of biodiesel from plant oil hydrolysates using an Aspergillus oryzae whole-cell biocatalyst highly expressing Candida antarctica lipase B. Bioresour. Technol. 2013, 135, 410–416. [Google Scholar] [CrossRef]
- Arai, S.; Nakashima, K.; Tanino, T.; Ogino, C.; Kondo, A.; Fukuda, H. Production of biodiesel fuel from soybean oil catalyzed by fungus whole-cell biocatalysts in ionic liquids. Enzym. Microb. Technol. 2010, 46, 51–55. [Google Scholar] [CrossRef]
- Matsuda, T.; Yamagishi, Y.; Koguchi, S.; Iwai, N.; Kitazume, T. An effective method to use ionic liquids as reaction media for asymmetric reduction by Geotrichum candidum. Tetrahedron Lett. 2006, 47, 4619–4622. [Google Scholar] [CrossRef]
- Tanaka, T.; Iwai, N.; Matsuda, T.; Kitazume, T. Utility of ionic liquid for Geotrichum candidum-catalyzed synthesis of optically active alcohols. J. Mol. Catal. B Enzym. 2009, 57, 317–320. [Google Scholar] [CrossRef]
- Ming-Liang, F.; Jing, L.; Ya-Chen, D.; Yu, F.; Ruo-Si, F.; Qi-He, C.; Xiao-Jie, L. Effect of ionic liquid-containing system on betulinic acid production from betulin biotransformation by cultured Armillaria luteo-virens Sacc cells. Eur. Food Res. Technol. 2011, 233, 507–515. [Google Scholar] [CrossRef]
- Ventura, S.P.M.; Gurbisz, M.; Ghavre, M.; Ferreira, F.M.M.; Gonçalves, F.; Beadham, I.; Quilty, B.; Coutinho, J.A.P.; Gathergood, N. Imidazolium and pyridinium ionic liquids from mandelic acid derivatives: Synthesis and bacteria and algae toxicity evaluation. ACS Sustain. Chem. Eng. 2013, 1, 393–402. [Google Scholar] [CrossRef]
- Ganske, F.; Bornscheuer, U.T. Growth of Escherichia coli, Pichia pastoris and Bacillus cereus in the presence of the ionic liquids [BMIM][BF4] and [BMIM][PF6] and Organic Solvents. Biotechnol. Lett. 2006, 28, 465–469. [Google Scholar] [CrossRef]
- Pernak, J.; Sobaszkiewicz, K.; Mirska, I. Anti-microbial activities of ionic liquids. Green Chem. 2003, 5, 52–56. [Google Scholar] [CrossRef]
- Sikkema, Jan; De Bont, Jan A.M.; Poolman, B. Mechanisms of membrane toxicity of hydrocarbons. Microbiol. Rev. 1995, 6, 201–222. [Google Scholar]
- Moniruzzaman, M.; Kamiya, N.; Goto, M. Biocatalysis in water-in-ionic liquid microemulsions: A case study with horse- radish peroxidase. Langmuir 2009, 25, 977–982. [Google Scholar] [CrossRef]
- Petkovic, M.; Seddon, K.R.; Rebelo, L.P.; Silva Pereira, C. Ionic liquids: A pathway to environmental acceptability. Chem. Soc. Rev. 2011, 40, 1383–1403. [Google Scholar] [CrossRef]
- Tee, K.L.; Roccatano, D.; Stolte, S.; Arning, J.; Jastorff, B.; Schwaneberg, U. Ionic liquid effects on the activity of monooxygenase P450 BM-3. Green Chem. 2008, 10, 117–123. [Google Scholar] [CrossRef]
- Chefson, A.; Auclair, K. CYP3A4 activity in the presence of organic cosolvents, ionic liquids, or water-immiscible organic solvents. Chembiochem 2007, 8, 1189–1197. [Google Scholar] [CrossRef]
- Skladanowski, A.C.; Stepnowski, P.; Kleszczynski, K.; Dmochowska, B. AMP deaminase in vitro inhibition by xenobiotics A potential molecular method for risk assessment of synthetic nitro- and polycyclic musks, imidazolium ionic liquids and N-glucopyranosyl ammonium salts. Environ. Toxicol. Pharmacol. 2005, 19, 291–296. [Google Scholar] [CrossRef]
- Couling, D.J.; Bernot, R.J.; Docherty, K.M.; Dixon, J.K.; Maginn, E.J. Assessing the factors responsible for ionic liquid toxicity to aquatic organisms via quantitative structure–property relationship modeling. Green Chem. 2006, 8, 82–90. [Google Scholar] [CrossRef]
- Torrecilla, J.S.; Palomar, J.; Lemus, J.; Rodríguez, F. A quantum-chemical-based guide to analyze/quantify the cytotoxicity of ionic liquids. Green Chem. 2010, 12, 123–124. [Google Scholar] [CrossRef]
- Ranke, J.; Stolte, S.; Sto¨rmann, R.; Arning, J.; Jastorff, B. Design of sustainable chemical productss the example of ionic liquids. Chem. Rev. 2007, 107, 2183–2206. [Google Scholar] [CrossRef]
- Lovejoy, K.S.; Davis, L.E.; McClellan, L.M.; Lillo, A.M.; Welsh, J.D.; Schmidt, E.N.; Sanders, C.K.; Lou, A.J.; Fox, D.T.; Koppisch, A.T.; et al. Evaluation of ionic liquids on phototrophic microbes and their use in biofuel extraction and isolation. J. Appl. Phycol. 2012, 25, 973–981. [Google Scholar]
- Luis, P.; Ortiz, I.; Aldaco, R.; Irabien, A. A novel group contribution method in the development of a QSAR for predicting the toxicity (Vibrio fischeri EC50) of ionic liquids. Ecotoxicol. Environ. Saf. 2007, 67, 423–429. [Google Scholar] [CrossRef]
- Tourne-Peteilh, C.; Coasne, B.; In, M.; Brevet, D.; Devoisselle, J.M.; Vioux, A.; Viau, L. Surfactant behavior of ionic liquids involving a drug: From molecular interactions to self-assembly. Langmuir 2014, 30, 1229–1238. [Google Scholar] [CrossRef]
- Steudte, S.; Bemowsky, S.; Mahrova, M.; Bottin-Weber, U.; Tojo-Suarez, E.; Stepnowski, P.; Stolte, S. Toxicity and biodegradability of dicationic ionic liquids. RSC Adv. 2014, 4, 5198–5205. [Google Scholar] [CrossRef]
- Ruegg, T.L.; Kim, E.M.; Simmons, B.A.; Keasling, J.D.; Singer, S.W.; Soon Lee, T.; Thelen, M.P. An auto-inducible mechanism for ionic liquid resistance in microbial biofuel production. Nat. Commun. 2014. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Fan, L.-L.; Li, H.-J.; Chen, Q.-H. Applications and Mechanisms of Ionic Liquids in Whole-Cell Biotransformation. Int. J. Mol. Sci. 2014, 15, 12196-12216. https://doi.org/10.3390/ijms150712196
Fan L-L, Li H-J, Chen Q-H. Applications and Mechanisms of Ionic Liquids in Whole-Cell Biotransformation. International Journal of Molecular Sciences. 2014; 15(7):12196-12216. https://doi.org/10.3390/ijms150712196
Chicago/Turabian StyleFan, Lin-Lin, Hong-Ji Li, and Qi-He Chen. 2014. "Applications and Mechanisms of Ionic Liquids in Whole-Cell Biotransformation" International Journal of Molecular Sciences 15, no. 7: 12196-12216. https://doi.org/10.3390/ijms150712196
APA StyleFan, L.-L., Li, H.-J., & Chen, Q.-H. (2014). Applications and Mechanisms of Ionic Liquids in Whole-Cell Biotransformation. International Journal of Molecular Sciences, 15(7), 12196-12216. https://doi.org/10.3390/ijms150712196





