Melatonin Promotes Superovulation in Sika Deer (Cervus nippon)
Abstract
:1. Introduction
2. Results
2.1. The Concentration of MT, FSH, LH and PRL in Sika Deer after MT Implantation
2.1.1. Melatonin Concentrations
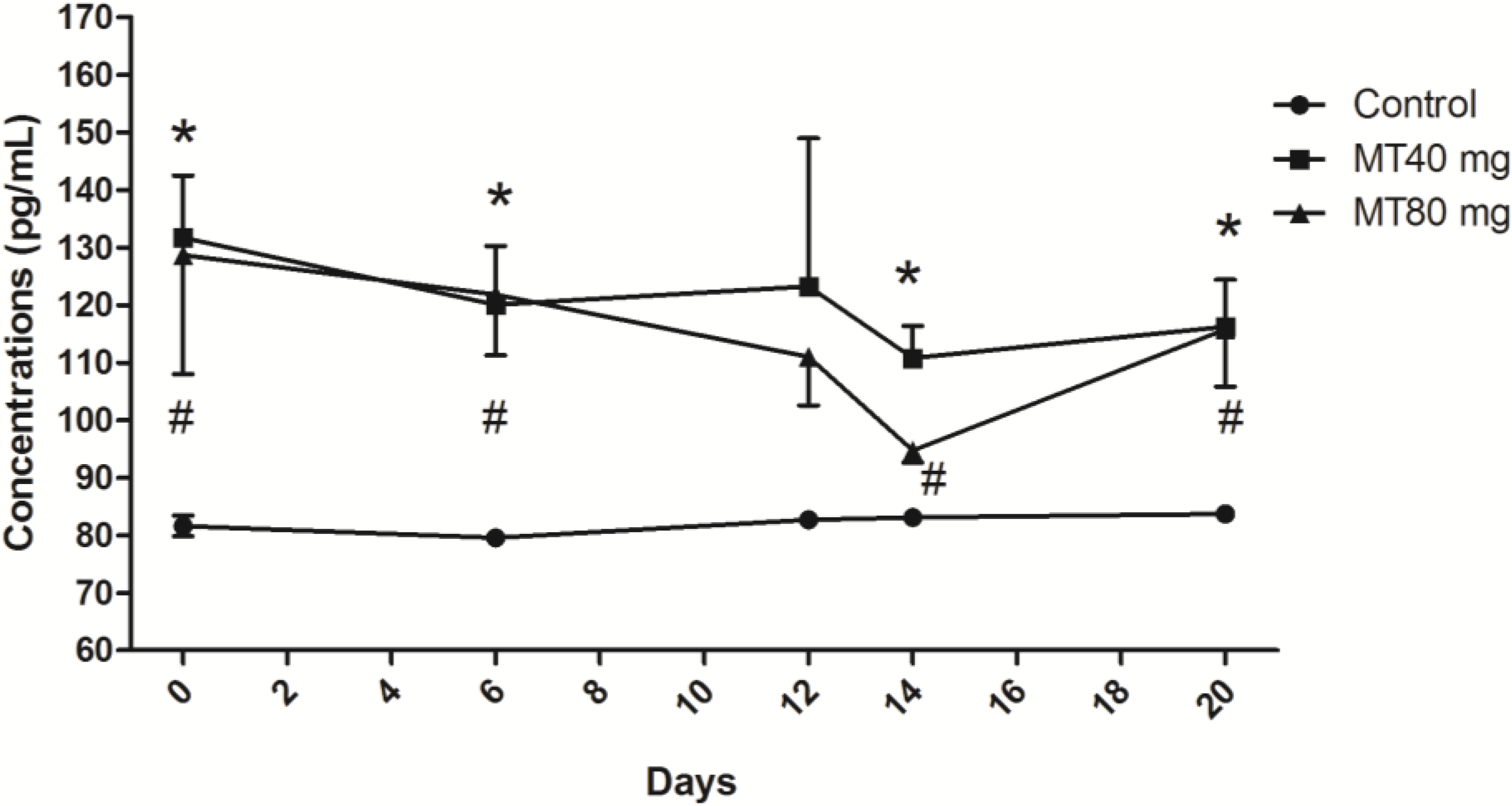
2.1.2. FSH Concentrations
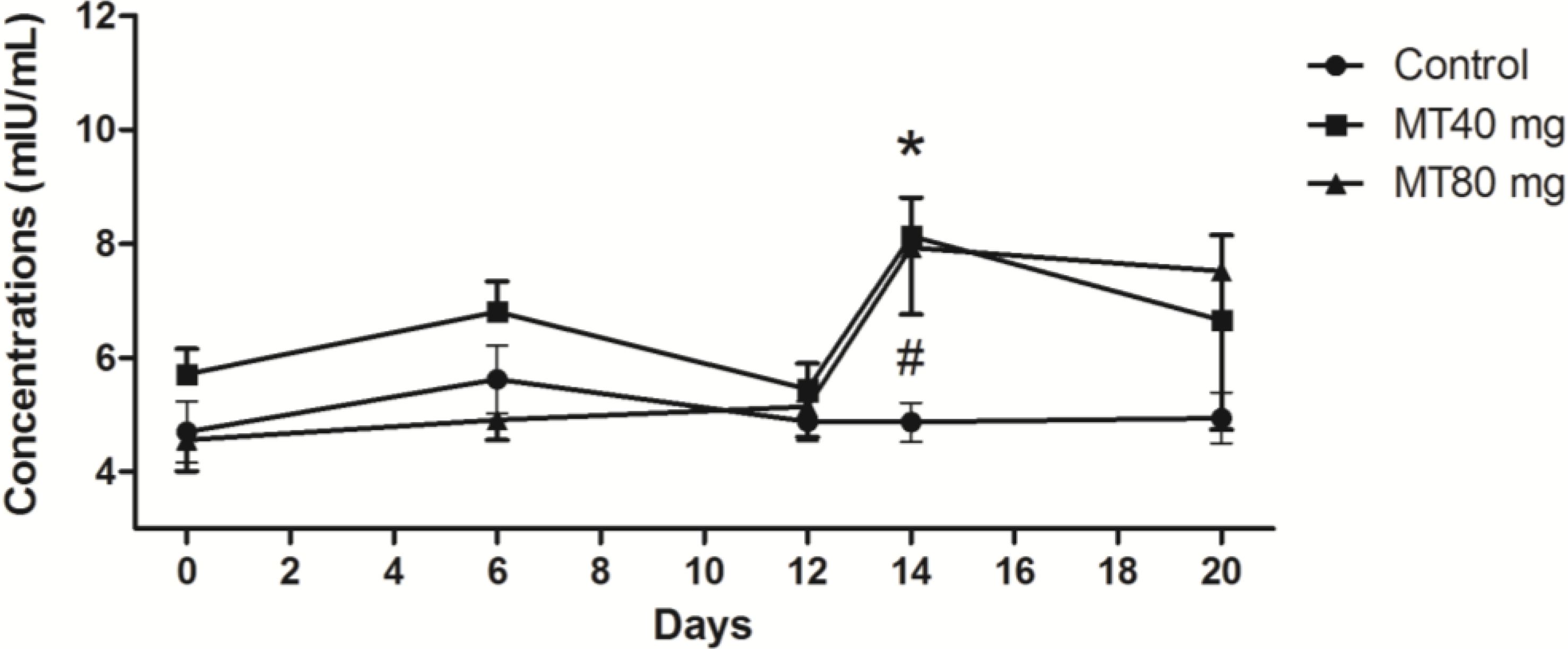
2.1.3. LH Concentrations

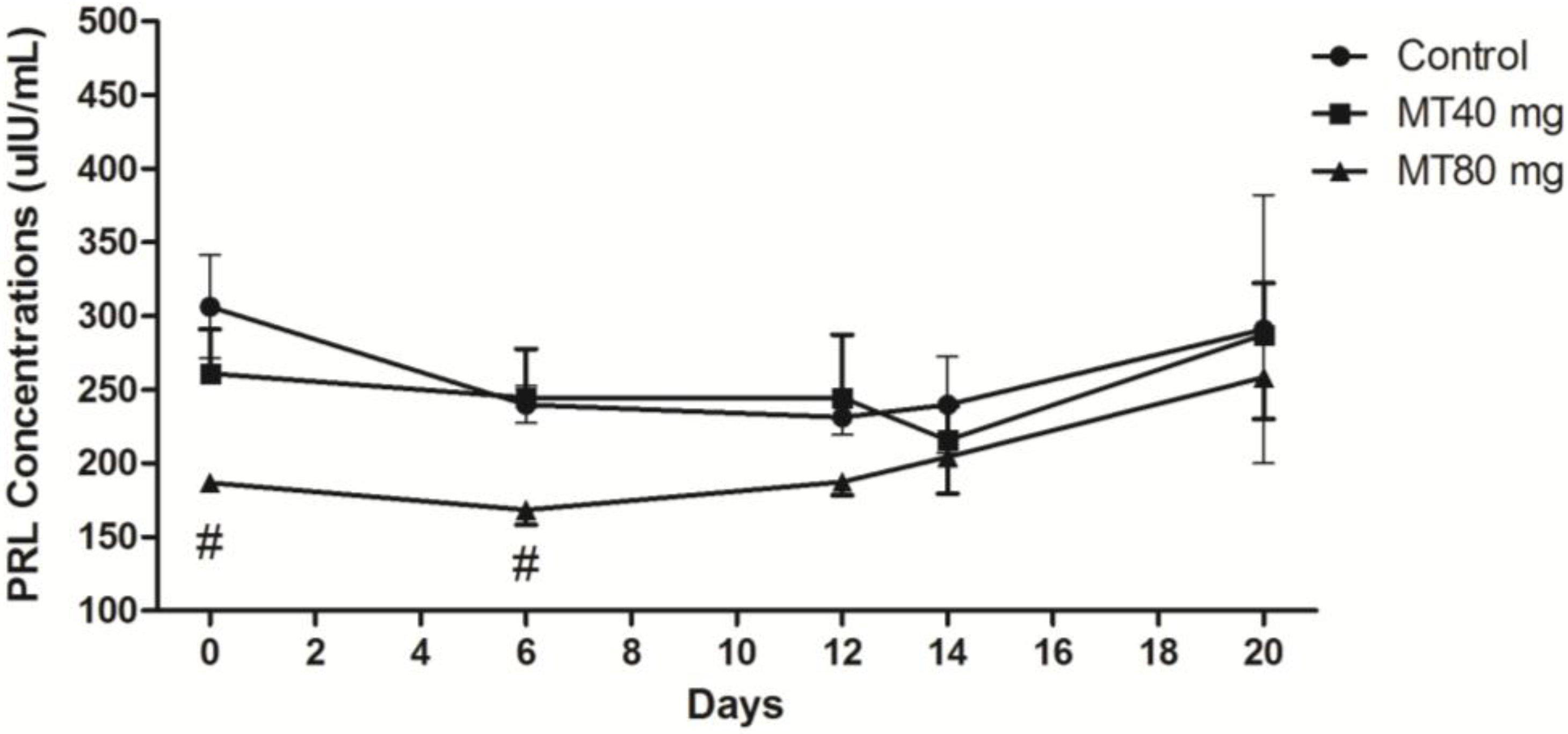
2.1.4. PRL Concentration
2.2. Correlation between the Levels of Melatonin and FSH, LH and PRL
| Blood Collection Stage | Hormone | Control Group | Group 1 (40 mg Implants) | Group 2 (80 mg Implants) | |
|---|---|---|---|---|---|
| CIDR insertion (Day 0) | MT | FSH | −0.629 | −0.494 | 0.912 |
| LH | −0.458 | 0.940 | 0.996 * | ||
| PRL | 0.854 | 0.464 | 0.659 | ||
| CIDR substitution (Day 6) | MT | FSH | −0.738 | 0.829 | 0.318 |
| LH | −0.863 | 0.529 | −0.074 | ||
| PRL | 0.794 | 0.567 | −0.169 | ||
| CIDR Removal (Day 12) | MT | FSH | −0.666 | 0.086 | −0.043 |
| LH | −0.105 | −0.056 | 0.991 * | ||
| PRL | 0.887 | −0.706 | −0.043 | ||
| Insemination (Day 14) | MT | FSH | −0.246 | −0.281 | 0.178 |
| LH | −0.179 | −0.832 | 0.432 | ||
| PRL | 0.293 | −0.855 | 0.311 | ||
| Embryo Recovery (Day 20) | MT | FSH | −0.103 | −0.960 * | 0.851 |
| LH | −0.095 | −0.268 | −0.384 | ||
| PRL | 0.076 | −0.644 | −0.313 | ||
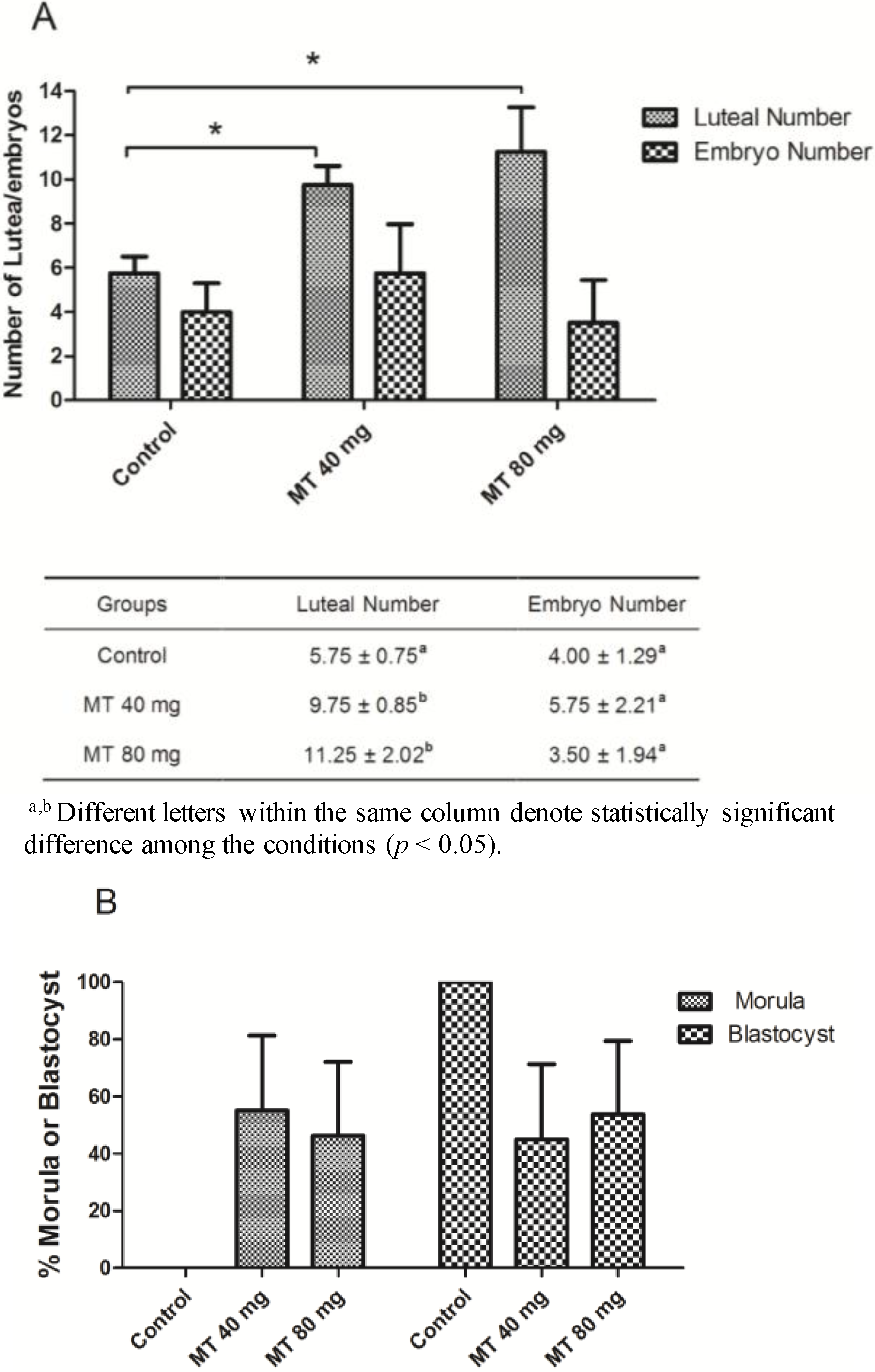
2.3. Effects of Melatonin Implantation on Superovulation in Female Sika Deer
3. Discussion
4. Experimental Section
4.1. Materials
4.2. Methods
4.2.1. Melatonin Implantation
4.2.2. Estrous Synchronization, Superovulation, Insemination and Embryo Recovery
4.2.3. Blood Collection
4.2.4. Preparation of Serum
4.2.5. Assay for Hormone Concentrations
4.3. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wang, L.; Zhou, G.B.; Shi, W.Q.; Shi, J.M.; Tian, X.Z.; Gao, C.; Zhang, L.; Zhu, S.E.; Zhang, T.T.; Zeng, S.M.; et al. First live offspring born in superovulated sika deer (Cervus nippon) after embryo vitrification. Theriogenology 2012, 78, 1627–1632. [Google Scholar] [CrossRef]
- Soler, J.P.; Mucci, N.; Kaiser, G.G.; Aller, J.; Hunter, J.W.; Dixon, T.E.; Alberio, R.H. Multiple ovulation and embryo transfer with fresh, frozen and vitrified red deer (Cervus elaphus) embryos in Argentina. Anim. Reprod. Sci. 2007, 102, 322–327. [Google Scholar] [CrossRef]
- Zhang, L.; Chai, M.; Tian, X.; Wang, F.; Fu, Y.; He, C.; Deng, S.; Lian, Z.; Feng, J.; Tan, D.X.; et al. Effects of melatonin on superovulation and transgenic embryo transplantation in small-tailed han sheep (Ovis aries). Neuro Endocrinol. Lett. 2013, 34, 294–301. [Google Scholar]
- Forcada, F.; Abecia, J.A.; Cebrian-Perez, J.A.; Muino-Blanco, T.; Valares, J.A.; Palacin, I.; Casao, A. The effect of melatonin implants during the seasonal anestrus on embryo production after superovulation in aged high-prolificacy Rasa Aragonesa ewes. Theriogenology 2006, 65, 356–365. [Google Scholar] [CrossRef]
- Barrett, P.; Bolborea, M. Molecular pathways involved in seasonal body weight and reproductive responses governed by melatonin. J. Pineal Res. 2012, 52, 376–388. [Google Scholar] [CrossRef]
- Gao, C.; Han, H.-B.; Tian, X.-Z.; Tan, D.-X.; Wang, L.; Zhou, G.-B.; Zhu, S.-E.; Liu, G.-S. Melatonin promotes embryonic development and reduces reactive oxygen species in vitrified mouse 2-cell embryos. J. Pineal Res. 2012, 52, 305–311. [Google Scholar] [CrossRef]
- Malpaux, B.; Migaud, M.; Tricoire, H.; Chemineau, P. Biology of mammalian photoperiodism and the critical role of the pineal gland and melatonin. J. Biol. Rhythms 2001, 16, 336–347. [Google Scholar] [CrossRef]
- Reiter, R.J.; Tan, D.-X.; Manchester, L.C.; Paredes, S.D.; Mayo, J.C.; Sainz, R.M. Melatonin and Reproduction Revisited. Biol. Reprod. 2009, 81, 445–456. [Google Scholar] [CrossRef]
- Wang, F.; Tian, X.; Zhang, L.; Tan, D.; Reiter, R.J.; Liu, G. Melatonin promotes the in vitro development of pronuclear embryos and increases the efficiency of blastocyst implantation in murine. J. Pineal Res. 2013, 55, 267–274. [Google Scholar] [CrossRef]
- Wang, F.; Tian, X.; Zhang, L.; Gao, C.; He, C.; Fu, Y.; Ji, P.; Li, Y.; Li, N.; Liu, G. Beneficial effects of melatonin on in vitro bovine embryonic development are mediated by melatonin receptor 1. J. Pineal Res. 2014, 56, 333–342. [Google Scholar] [CrossRef]
- Malpaux, B.; Thiéry, J.-C.; Chemineau, P. Melatonin and the seasonal control of reproduction. Reprod. Nutr. Dev. 1999, 39, 355–366. [Google Scholar] [CrossRef]
- Bubenik, G.; Smith, P.; Schams, D. The effect of orally administered melatonin on the seasonality of deer pelage exchange, antler development, LH, FSH, prolactin, testosterone, T3, T4, cortisol, and alkaline phosphatase. J. Pineal Res. 1986, 3, 331–349. [Google Scholar] [CrossRef]
- Chuffa, L.G.A.; Seiva, F.R.; Fávaro, W.J.; Teixeira, G.R.; Amorim, J.P.; Mendes, L.O.; Fioruci, B.A.; Pinheiro, P.F.F.; Fernandes, A.A.H.; Franci, J.A. Melatonin reduces LH, 17 beta-estradiol and induces differential regulation of sex steroid receptors in reproductive tissues during rat ovulation. Reprod. Biol. Endocrinol. 2011, 9, 108. [Google Scholar] [CrossRef]
- Wu, J.; Xu, B.; Wang, W. Effects of luteinizing hormone and follicle stimulating hormone on the developmental competence of porcine preantral follicle oocytes grown in vitro. J. Assist. Reprod. Genet. 2007, 24, 419–424. [Google Scholar] [CrossRef]
- Shi, J.M.; Tian, X.Z.; Zhou, G.B.; Wang, L.; Gao, C.; Zhu, S.E.; Zeng, S.M.; Tian, J.H.; Liu, G.S. Melatonin exists in porcine follicular fluid and improves in vitro maturation and parthenogenetic development of porcine oocytes. J. Pineal Res. 2009, 47, 318–323. [Google Scholar] [CrossRef]
- Rönnberg, L.; Kauppila, A.; Leppäluoto, J.; Martikainen, H.; Vakkuri, O. Circadian and seasonal variation in human preovulatory follicular fluid melatonin concentration. J. Clin. Endocrinol. Metab. 1990, 71, 493–496. [Google Scholar] [CrossRef]
- Nakamura, Y.; Tamura, H.; Takayama, H.; Kato, H. Increased endogenous level of melatonin in preovulatory human follicles does not directly influence progesterone production. Fertil. Steril. 2003, 80, 1012–1016. [Google Scholar] [CrossRef]
- Diaz, E.; Pazo, D.; Esquifino, A.; Diaz, B. Effects of ageing and exogenous melatonin on pituitary responsiveness to GnRH in rats. J. Reprod. Fertil. 2000, 119, 151–156. [Google Scholar]
- Delgadillo, J.; Carrillo, E.; Morán, J.; Duarte, G.; Chemineau, P.; Malpaux, B. Induction of sexual activity of male creole goats in subtropical northern Mexico using long days and melatonin. J. Anim. Sci. 2001, 79, 2245–2252. [Google Scholar]
- Anand, S.; Losee-Olson, S.; Turek, F.W.; Horton, T.H. Differential regulation of luteinizing hormone and follicle-stimulating hormone in male Siberian hamsters by exposure to females and photoperiod. Endocrinology 2002, 143, 2178–2188. [Google Scholar]
- Shiu, S.Y.; Ng, N.; Pang, S.F. A molecular perspective of the genetic relationships of G-protein coupled melatonin receptor subtypes. J. Pineal Res. 1996, 20, 198–204. [Google Scholar] [CrossRef]
- Reiter, R.J. Pineal melatonin: Cell biology of its synthesis and of its physiological interactions. Endocr. Rev. 1991, 12, 151–180. [Google Scholar] [CrossRef]
- Hafez, E.S.E. Studies on the breeding season and reproduction of the ewe Part III. The breeding season and artificial light Part IV. Studies on the reproduction of the ewe Part V. Mating behaviour and pregnancy diagnosis. J. Agric. Sci. 1952, 42, 232–265. [Google Scholar] [CrossRef]
- Adam, C.L.; Atkinson, T. Effect of feeding melatonin to red deer (Cervus elaphus) on the onset of the breeding season. J. Reprod. Fertil. 1984, 72, 463–466. [Google Scholar] [CrossRef]
- Falcon, J.; Besseau, L.; Fazzari, D.; Attia, J.; Gaildrat, P.; Beauchaud, M.; Boeuf, G. Melatonin modulates secretion of growth hormone and prolactin by trout pituitary glands and cells in culture. Endocrinology 2003, 144, 4648–4658. [Google Scholar] [CrossRef]
- Jiménez-Ortega, V.; Barquilla, P.C.; Pagano, E.S.; Fernández-Mateos, P.; Esquifino, A.I.; Cardinali, D.P. Melatonin supplementation decreases prolactin synthesis and release in rat adenohypophysis: Correlation with anterior pituitary redox state and circadian clock mechanisms. Chronobiol. Int. 2012, 29, 1–15. [Google Scholar]
- Webster, J.; Barrell, G. Advancement of reproductive activity, seasonal reduction in prolactin secretion and seasonal pelage changes in pubertal red deer hinds (Cervus elaphus) subjected to artificially shortened daily photoperiod or daily melatonin treatments. J. Reprod. Fertil. 1985, 73, 255–260. [Google Scholar] [CrossRef]
- Itoh, M.T.; Ishizuka, B.; Kuribayashi, Y.; Amemiya, A.; Sumi, Y. Melatonin, its precursors, and synthesizing enzyme activities in the human ovary. Mol. Hum. Reprod. 1999, 5, 402–408. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, L.; Zhuo, Z.-Y.; Shi, W.-Q.; Tan, D.-X.; Gao, C.; Tian, X.-Z.; Zhang, L.; Zhou, G.-B.; Zhu, S.-E.; Yun, P.; et al. Melatonin Promotes Superovulation in Sika Deer (Cervus nippon). Int. J. Mol. Sci. 2014, 15, 12107-12118. https://doi.org/10.3390/ijms150712107
Wang L, Zhuo Z-Y, Shi W-Q, Tan D-X, Gao C, Tian X-Z, Zhang L, Zhou G-B, Zhu S-E, Yun P, et al. Melatonin Promotes Superovulation in Sika Deer (Cervus nippon). International Journal of Molecular Sciences. 2014; 15(7):12107-12118. https://doi.org/10.3390/ijms150712107
Chicago/Turabian StyleWang, Liang, Zhi-Yong Zhuo, Wen-Qing Shi, Dun-Xian Tan, Chao Gao, Xiu-Zhi Tian, Lu Zhang, Guang-Bin Zhou, Shi-En Zhu, Peng Yun, and et al. 2014. "Melatonin Promotes Superovulation in Sika Deer (Cervus nippon)" International Journal of Molecular Sciences 15, no. 7: 12107-12118. https://doi.org/10.3390/ijms150712107
APA StyleWang, L., Zhuo, Z.-Y., Shi, W.-Q., Tan, D.-X., Gao, C., Tian, X.-Z., Zhang, L., Zhou, G.-B., Zhu, S.-E., Yun, P., & Liu, G.-S. (2014). Melatonin Promotes Superovulation in Sika Deer (Cervus nippon). International Journal of Molecular Sciences, 15(7), 12107-12118. https://doi.org/10.3390/ijms150712107






