The REST Gene Signature Predicts Drug Sensitivity in Neuroblastoma Cell Lines and Is Significantly Associated with Neuroblastoma Tumor Stage
Abstract
:1. Introduction
2. Results and Discussion
2.1. Results
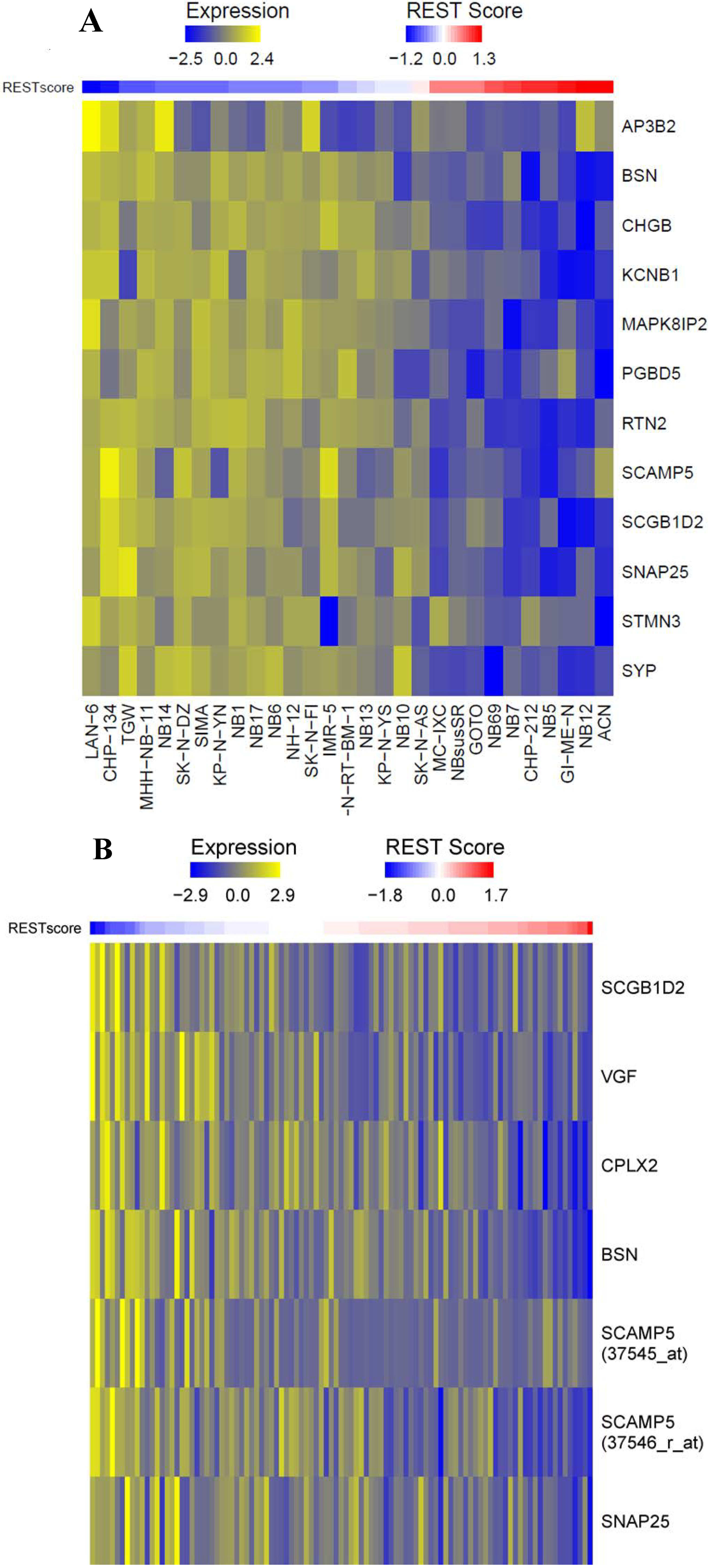
2.1.1. Repressor Element-1 Silencing Transcription (REST) Score in Neuroblastoma
| Symbol | Correlation | p Value |
|---|---|---|
| AP3B2 | −0.40135 | 0.030935 |
| BSN | −0.3463 | 0.065728 |
| CHGB | −0.39356 | 0.034663 |
| CPLX2 | 0.051692 | 0.790004 |
| HBA1 | 0.359446 | 0.055481 |
| HBA2 | 0.090065 | 0.642199 |
| KCNB1 | −0.40135 | 0.030936 |
| MAPK8IP2 | −0.19656 | 0.306799 |
| MMP24 | 0.240233 | 0.209381 |
| PGBD5 | −0.41019 | 0.027101 |
| RTN2 | −0.30055 | 0.113145 |
| SCAMP5 | −0.23286 | 0.224127 |
| SCGB1D2 | −0.42984 | 0.019953 |
| SNAP25 | −0.39209 | 0.03541 |
| STMN3 | −0.21973 | 0.252092 |
| SYP | −0.2474 | 0.195705 |
| VGF | 0.018048 | 0.925965 |
| Symbol | Probe ID | Correlation | p Value |
|---|---|---|---|
| AP3B2 | 38937_at | 0.121623 | 0.22567 |
| BSN | 33728_at | −0.19752 | 0.047712 |
| CHGB | 33426_at | 0.001979 | 0.984333 |
| CPLX2 | 33084_at | −0.59139 | 7.43 × 10−11 |
| KCNB1 | 40693_at | 0.112516 | 0.262606 |
| MAPK8IP2 | 37588_s_at | 0.227915 | 0.02189 |
| MMP24 | 32924_at | 0.364921 | 0.000175 |
| RTN2 | 34408_at | 0.120774 | 0.228953 |
| RUNDC3A | 36823_at | 0.12448 | 0.214878 |
| SCAMP5 | 37545_at | −0.03147 | 0.754758 |
| SCAMP5 | 37546_r_at | −0.55512 | 1.70 × 10−9 |
| SCGB1D2 | 32880_at | −0.42044 | 1.20 × 10−5 |
| SYP | 37182_at | 0.103264 | 0.304127 |
| SNAP25 | 38484_at | −0.06155 | 0.540931 |
| VGF | 32969_r_at | −0.10574 | 0.292595 |
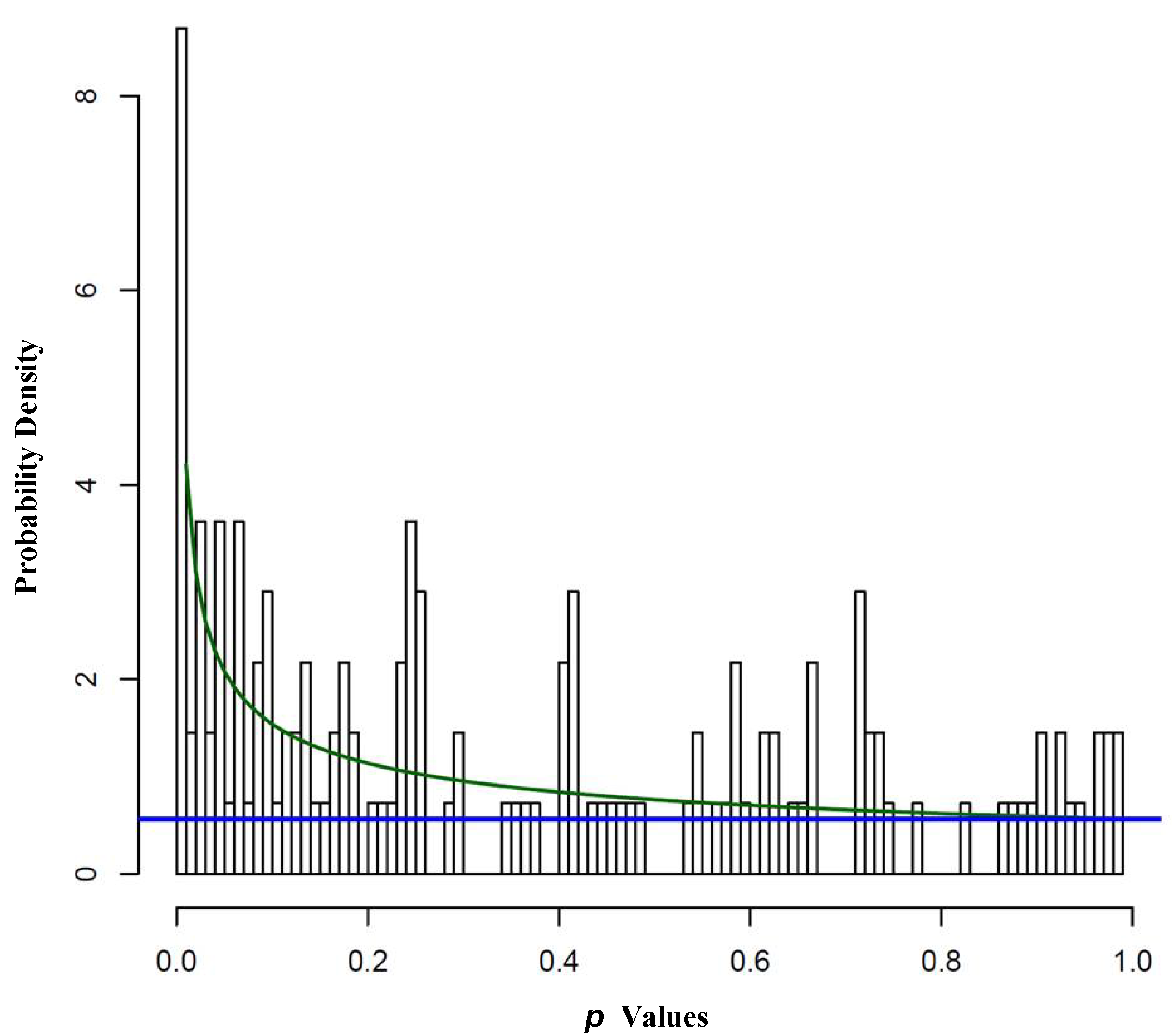
2.1.2. REST Signature and Drug Sensitivity in Neuroblastoma
| FDR | Number of Significant Associations | p Value Cutoff |
|---|---|---|
| 0.05 | 9 | 0.00389 |
| 0.10 | 14 | 0.01904 |
| 0.15 | 24 | 0.04820 |
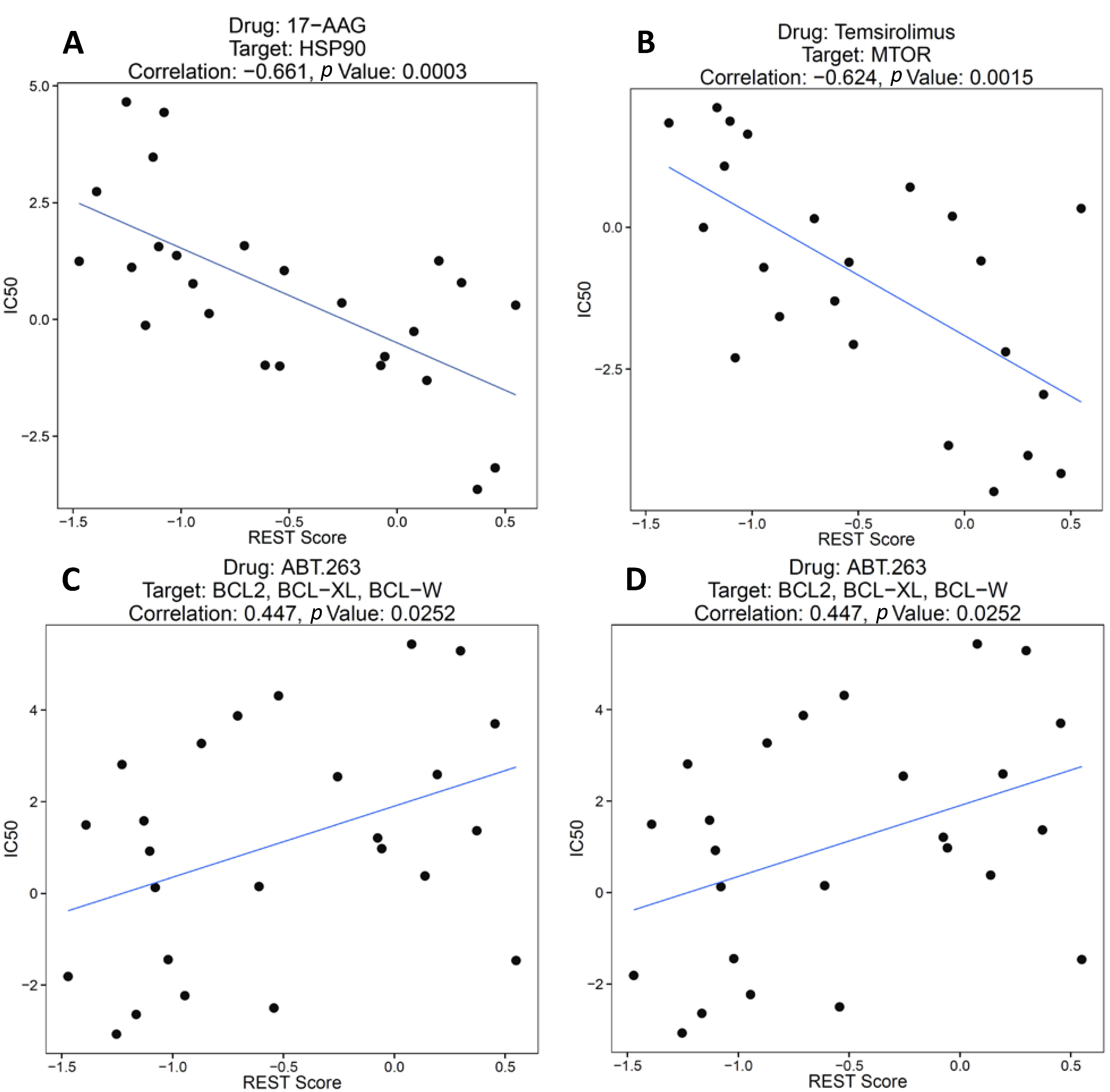
2.1.3. REST Signature and Tumor Stage in Neuroblastoma
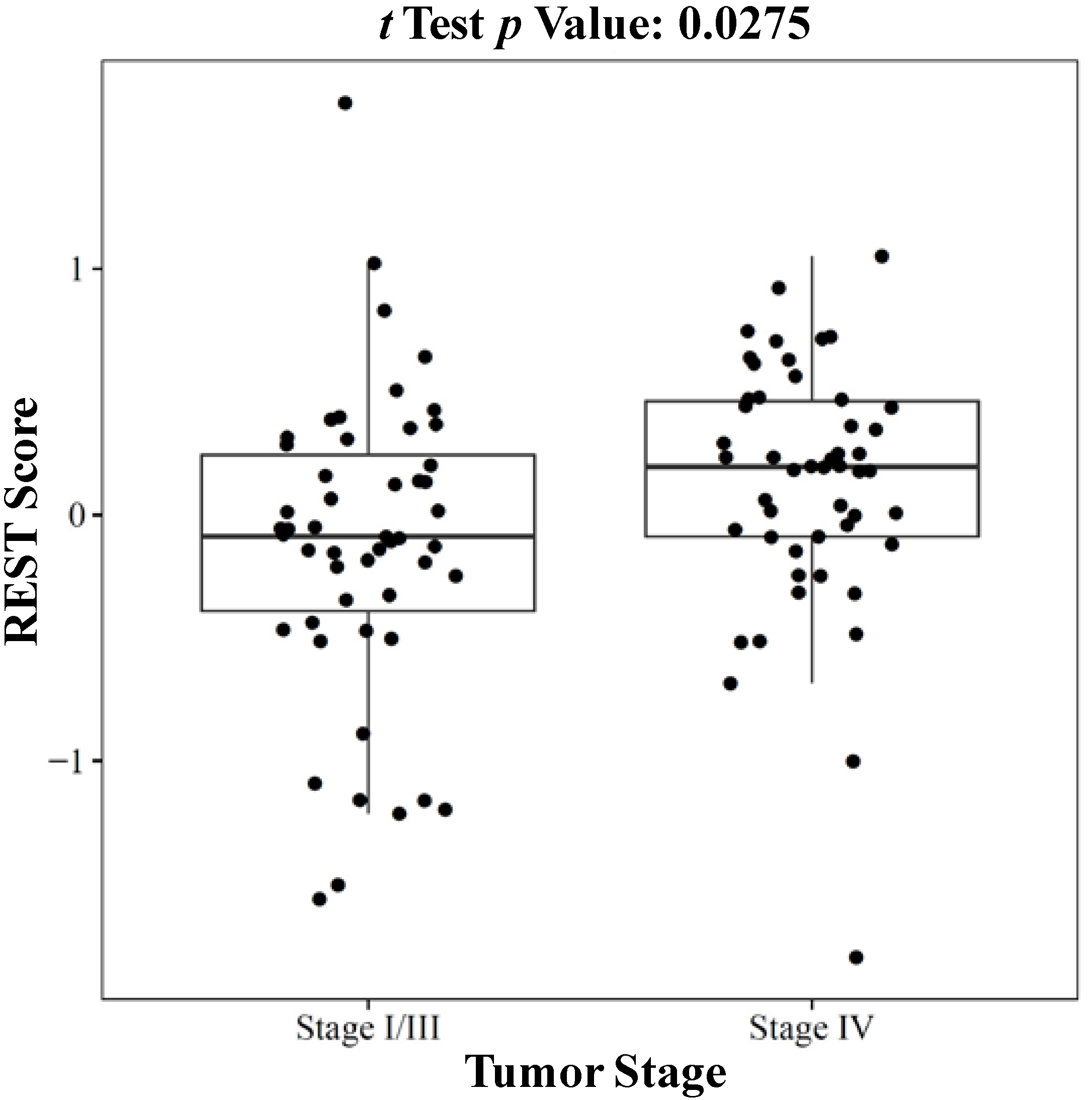
2.2. Discussion
3. Experimental Section
3.1. Data Collection
3.2. Data Analysis
4. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Maris, J.M. Recent advances in neuroblastoma. New Engl. J. Med. 2010, 362, 2202–2211. [Google Scholar] [CrossRef]
- Zirath, H.; Frenzel, A.; Oliynyk, G.; Segerström, L.; Westermark, U.K.; Larsson, K.; Persson, M.M.; Hultenby, K.; Lehtiö, J.; Einvik, C. MYC inhibition induces metabolic changes leading to accumulation of lipid droplets in tumor cells. Proc. Natl. Acad. Sci. USA 2013, 110, 10258–10263. [Google Scholar] [CrossRef]
- Stallings, R.L.; Foley, N.H.; Bryan, K.; Buckley, P.G.; Bray, I. Therapeutic targeting of miRNAs in neuroblastoma. Expert Opin. Ther.Targets 2010, 14, 951–962. [Google Scholar] [CrossRef]
- Ow, T.J.; Bell, D.; Kupferman, M.E.; Demonte, F.; Hanna, E.Y. Esthesioneuroblastoma. Neurosurg. Clin. N. Am. 2013, 24, 51–65. [Google Scholar] [CrossRef]
- Schipper, M.H.; van Duinen, S.G.; Taphoorn, M.J.; Kloet, A.; Walchenbach, R.; Wiggenraad, R.G.; Vecht, C.J. Cerebral ganglioneuroblastoma of adult onset: Two patients and a review of the literature. Clin. Neurol. Neurosurg. 2012, 114, 529–534. [Google Scholar] [CrossRef]
- Alisi, A.; Cho, W.C.; Locatelli, F.; Fruci, D. Multidrug resistance and cancer stem cells in neuroblastoma and hepatoblastoma. Int. J. Mol. Sci. 2013, 14, 24706–24725. [Google Scholar] [CrossRef]
- Krizkova, S.; Fabrik, I.; Huska, D.; Adam, V.; Babula, P.; Hrabeta, J.; Eckschlager, T.; Pochop, P.; Darsova, D.; Kukacka, J. An adsorptive transfer technique coupled with Brdicka reaction to reveal the importance of metallothionein in chemotherapy with platinum based cytostatics. Int. J. Mol. Sci. 2010, 11, 4826–4842. [Google Scholar] [CrossRef]
- Wang, Q.; Diskin, S.; Rappaport, E.; Attiyeh, E.; Mosse, Y.; Shue, D.; Seiser, E.; Jagannathan, J.; Shusterman, S.; Bansal, M. Integrative genomics identifies distinct molecular classes of neuroblastoma and shows that multiple genes are targeted by regional alterations in DNA copy number. Cancer Res. 2006, 66, 6050–6062. [Google Scholar] [CrossRef]
- Hsu, D.M.; Agarwal, S.; Benham, A.; Coarfa, C.; Trahan, D.N.; Chen, Z.; Stowers, P.N.; Courtney, A.N.; Lakoma, A.; Barbieri, E. G-CSF receptor positive neuroblastoma subpopulations are enriched in chemotherapy-resistant or relapsed tumors and are highly tumorigenic. Cancer Res. 2013, 73, 4134–4146. [Google Scholar] [CrossRef]
- Henderson, T.O.; Bhatia, S.; Pinto, N.; London, W.B.; McGrady, P.; Crotty, C.; Sun, C.L.; Cohn, S.L. Racial and ethnic disparities in risk and survival in children with neuroblastoma: A children’s oncology group study. J. Clin. Oncol. 2011, 29, 76–82. [Google Scholar] [CrossRef]
- Hirabayashi, Y.; Gotoh, Y. Epigenetic control of neural precursor cell fate during development. Nat. Rev. Neurosci. 2010, 11, 377–388. [Google Scholar] [CrossRef]
- Li, H.-T.; Jiang, F.-X.; Shi, P.; Zhang, T.; Liu, X.-Y.; Lin, X.-W.; Pang, X.-N. In vitro reprogramming of rat bone marrow-derived mesenchymal stem cells into insulin-producing cells by genetically manipulating negative and positive regulators. Biochem. Biophys. Res. Commun. 2012, 420, 793–798. [Google Scholar] [CrossRef]
- Sung, M.; Jeong, M.S.; Jang, S.B. Biological characterization and structure based prediction of insulin-like growth factor binding protein-5. Biochem. Biophys. Res. Commun. 2010, 403, 230–236. [Google Scholar] [CrossRef]
- Soldati, C.; Bithell, A.; Johnston, C.; Wong, K.Y.; Teng, S.W.; Beglopoulos, V.; Stanton, L.W.; Buckley, N.J. Repressor element 1 silencing transcription factor couples loss of pluripotency with neural induction and neural differentiation. Stem Cells 2012, 30, 425–434. [Google Scholar] [CrossRef]
- Majumder, S. REST in good times and bad: roles in tumor suppressor and oncogenic activities. Cell Cycle 2006, 5, 1929–1935. [Google Scholar]
- Negrini, S.; Prada, I.; D’Alessandro, R.; Meldolesi, J. REST: An oncogene or a tumor suppressor? Trends Cell Biol. 2013, 23, 289–295. [Google Scholar] [CrossRef]
- Wagoner, M.P.; Gunsalus, K.T.; Schoenike, B.; Richardson, A.L.; Friedl, A.; Roopra, A. The transcription factor REST is lost in aggressive breast cancer. PLoS Genet. 2010, 6, e1000979. [Google Scholar] [CrossRef]
- Taylor, P.; Fangusaro, J.; Rajaram, V.; Goldman, S.; Helenowski, I.B.; MacDonald, T.; Hasselblatt, M.; Riedemann, L.; Laureano, A.; Cooper, L. REST is a novel prognostic factor and therapeutic target for medulloblastoma. Mol. Cancer Ther. 2012, 11, 1713–1723. [Google Scholar]
- Singh, A.; Rokes, C.; Gireud, M.; Fletcher, S.; Baumgartner, J.; Fuller, G.; Stewart, J.; Zage, P.; Gopalakrishnan, V. Retinoic acid induces REST degradation and neuronal differentiation by modulating the expression of SCF (β-TRCP) in neuroblastoma cells. Cancer 2011, 117, 5189–5202. [Google Scholar] [CrossRef]
- Fuller, G.N.; Su, X.; Price, R.E.; Cohen, Z.R.; Lang, F.F.; Sawaya, R.; Majumder, S. Many human medulloblastoma tumors overexpress repressor element-1 silencing transcription (REST)/neuron-restrictive silencer factor, which can be functionally countered by REST-VP16. Mol. Cancer Ther. 2005, 4, 343–349. [Google Scholar]
- Lawinger, P.; Venugopal, R.; Guo, Z.S.; Immaneni, A.; Sengupta, D.; Lu, W.; Rastelli, L.; Marin Dias Carneiro, A.; Levin, V.; Fuller, G.N. The neuronal repressor REST/NRSF is an essential regulator in medulloblastoma cells. Nat. Med. 2000, 6, 826–831. [Google Scholar] [CrossRef]
- Wagoner, M.P.; Roopra, A. A REST derived gene signature stratifies glioblastomas into chemotherapy resistant and responsive disease. Biol. Med. Cent. Genomics 2012, 13, 686:1–686:12. [Google Scholar]
- Guo, C.; White, P.S.; Weiss, M.J.; Hogarty, M.D.; Thompson, P.M.; Stram, D.O.; Gerbing, R.; Matthay, K.K.; Seeger, R.C.; Brodeur, G.M. Allelic deletion at 11q23 is common in MYCN single copy neuroblastomas. Oncogene 1999, 18, 4948–4957. [Google Scholar] [CrossRef]
- Santo, E.E.; Ebus, M.E.; Koster, J.; Schulte, J.H.; Lakeman, A.; van Sluis, P.; Vermeulen, J.; Gisselsson, D.; Ora, I.; Lindner, S. Oncogenic activation of FOXR1 by 11q23 intrachromosomal deletion-fusions in neuroblastoma. Oncogene 2012, 31, 1571–1581. [Google Scholar] [CrossRef]
- Yagyu, S.; Iehara, T.; Gotoh, T.; Miyachi, M.; Katsumi, Y.; Kikuchi, K.; Tsuchiya, K.; Osone, S.; Kuroda, H.; Sugimoto, T. Preoperative analysis of 11q loss using circulating tumor-released DNA in serum: A novel diagnostic tool for therapy stratification of neuroblastoma. Cancer Lett. 2011, 309, 185–189. [Google Scholar] [CrossRef]
- Pounds, S.; Morris, S.W. Estimating the occurrence of false positives and false negatives in microarray studies by approximating and partitioning the empirical distribution of p-values. Bioinformatics 2003, 19, 1236–1242. [Google Scholar] [CrossRef]
- Cohn, S.L.; Tweddle, D.A. MYCN amplification remains prognostically strong 20 years after its “clinical debut”. Eur. J. Cancer 2004, 40, 2639–2642. [Google Scholar] [CrossRef]
- Brodeur, G.M.; Seeger, R.C.; Schwab, M.; Varmus, H.E.; Bishop, J.M. Amplification of N-myc in untreated human neuroblastomas correlates with advanced disease stage. Science 1984, 224, 1121–1124. [Google Scholar]
- Kang, J.H.; Rychahou, P.G.; Ishola, T.A.; Qiao, J.; Evers, B.M.; Chung, D.H. MYCN silencing induces differentiation and apoptosis in human neuroblastoma cells. Biochem. Biophys. Res. Commun. 2006, 351, 192–197. [Google Scholar] [CrossRef]
- Bell, E.; Chen, L.; Liu, T.; Marshall, G.M.; Lunec, J.; Tweddle, D.A. MYCN oncoprotein targets and their therapeutic potential. Cancer Lett. 2010, 293, 144–157. [Google Scholar] [CrossRef]
- Otto, S.J.; McCorkle, S.R.; Hover, J.; Conaco, C.; Han, J.J.; Impey, S.; Yochum, G.S.; Dunn, J.J.; Goodman, R.H.; Mandel, G. A new binding motif for the transcriptional repressor REST uncovers large gene networks devoted to neuronal functions. J. Neurosci. 2007, 27, 6729–6239. [Google Scholar] [CrossRef]
- Conti, L.; Crisafulli, L.; Caldera, V.; Tortoreto, M.; Brilli, E.; Conforti, P.; Zunino, F.; Magrassi, L.; Schiffer, D.; Cattaneo, E. REST controls self-renewal and tumorigenic competence of human glioblastoma cells. PLoS One 2012, 7, e38486. [Google Scholar] [CrossRef]
- Kamal, M.M.; Sathyan, P.; Singh, S.K.; Zinn, P.O.; Marisetty, A.L.; Liang, S.; Gumin, J.; El-Mesallamy, H.O.; Suki, D.; Colman, H. REST regulates oncogenic properties of glioblastoma stem cells. Stem Cells 2012, 30, 405–414. [Google Scholar] [CrossRef]
- Tivnan, A.; Zhao, J.; Johns, T.G.; Day, B.W.; Stringer, B.W.; Boyd, A.W.; Tiwari, S.; Giles, K.M.; Teo, C.; McDonald, K.L. The tumor suppressor microRNA, miR-124a, is regulated by epigenetic silencing and by the transcriptional factor, REST in glioblastoma. Tumor Biol. 2014, 35, 1459–1465. [Google Scholar] [CrossRef]
- Donev, R.M.; Gray, L.C.; Sivasankar, B.; Hughes, T.R.; van den Berg, C.W.; Morgan, B.P. Modulation of CD59 expression by restrictive silencer factor-derived peptides in cancer immunotherapy for neuroblastoma. Cancer Res. 2008, 68, 5979–5987. [Google Scholar] [CrossRef]
- Team, R.C. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Liang, J.; Tong, P.; Zhao, W.; Li, Y.; Zhang, L.; Xia, Y.; Yu, Y. The REST Gene Signature Predicts Drug Sensitivity in Neuroblastoma Cell Lines and Is Significantly Associated with Neuroblastoma Tumor Stage. Int. J. Mol. Sci. 2014, 15, 11220-11233. https://doi.org/10.3390/ijms150711220
Liang J, Tong P, Zhao W, Li Y, Zhang L, Xia Y, Yu Y. The REST Gene Signature Predicts Drug Sensitivity in Neuroblastoma Cell Lines and Is Significantly Associated with Neuroblastoma Tumor Stage. International Journal of Molecular Sciences. 2014; 15(7):11220-11233. https://doi.org/10.3390/ijms150711220
Chicago/Turabian StyleLiang, Jianfeng, Pan Tong, Wanni Zhao, Yaqiao Li, Li Zhang, Ying Xia, and Yanbing Yu. 2014. "The REST Gene Signature Predicts Drug Sensitivity in Neuroblastoma Cell Lines and Is Significantly Associated with Neuroblastoma Tumor Stage" International Journal of Molecular Sciences 15, no. 7: 11220-11233. https://doi.org/10.3390/ijms150711220
APA StyleLiang, J., Tong, P., Zhao, W., Li, Y., Zhang, L., Xia, Y., & Yu, Y. (2014). The REST Gene Signature Predicts Drug Sensitivity in Neuroblastoma Cell Lines and Is Significantly Associated with Neuroblastoma Tumor Stage. International Journal of Molecular Sciences, 15(7), 11220-11233. https://doi.org/10.3390/ijms150711220




